These Highlights Do Not Include All The Information Needed To Use Saxagliptin And Metformin Hydrochloride Extended-release Tablets
cbf8677c-cafe-48bc-8844-0cc6547886bd
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
• Post-marketing cases of metformin-associated lactic acidosis have resulted in death, hypothermia, hypotension, and resistant bradyarrhythmias. The onset of metformin-associated lactic acidosis is often subtle, accompanied only by nonspecific symptoms such as malaise, myalgias, respiratory distress, somnolence, and abdominal pain. Metformin-associated lactic acidosis was characterized by elevated blood lactate levels (> 5 mmol/Liter), anion gap acidosis (without evidence of ketonuria or ketonemia), an increased lactate/pyruvate ratio; and metformin plasma levels generally > 5 mcg/mL [ see Warnings and Precautions (5.1) ]. • Risk factors for metformin-associated lactic acidosis include renal impairment, concomitant use of certain drugs (e.g., carbonic anhydrase inhibitors such as topiramate), age 65 years old or greater, having a radiological study with contrast, surgery and other procedures, hypoxic states (e.g., acute congestive heart failure), excessive alcohol intake, and hepatic impairment. • Steps to reduce the risk of and manage metformin-associated lactic acidosis in these high risk groups are provided in the full prescribing information [ see Dosage and Administration (2.2) , Contraindications (4) , Warnings and Precautions (5.1) , Drug Interactions (7) , and Use in Specific Populations (8.6 , 8.7) ]. • If metformin-associated lactic acidosis is suspected, immediately discontinue saxagliptin and metformin hydrochloride extended-release tablets and institute general supportive measures in a hospital setting. Prompt hemodialysis is recommended [ see Warnings and Precautions (5.1) ].
Indications and Usage
Saxagliptin and metformin hydrochloride extended-release tablets are indicated as an adjunct to diet and exercise to improve glycemic control in adults with type 2 diabetes mellitus [ see Clinical Studies (14) ].
Dosage and Administration
• Administer once daily with the evening meal. ( 2.1 ) • Individualize the starting dosage based on the patient’s current regimen then adjust the dosage based on effectiveness and tolerability. ( 2.1 ) • Do not exceed a daily dosage of 5 mg saxagliptin/2,000 mg metformin HCl extended-release. ( 2.1 ) • Swallow whole. Never crush, cut, or chew. ( 2.1 ) • Limit the saxagliptin dosage to 2.5 mg daily for patients also taking strong cytochrome P450 3A4/5 inhibitors (e.g., ketoconazole). ( 2.3 , 7.1 ) • Assess renal function prior to initiation of saxagliptin and metformin hydrochloride extended-release tablets and periodically thereafter. ( 2.2 ) o Do not use in patients with eGFR below 30 mL/min/1.73 m 2 . o Initiation is not recommended in patients with eGFR between 30 - 45 mL/min/1.73 m 2 . o Assess risk/benefit of continuing if eGFR falls below 45 mL/min/1.73 m 2 . o Limit the saxagliptin component to 2.5 mg daily if eGFR is less than 45 mL/min/1.73 m 2 . o Discontinue if eGFR falls below 30 mL/min/1.73 m 2 . • Saxagliptin and metformin hydrochloride extended-release tablets may need to be discontinued at time of, or prior to, iodinated contrast imaging procedures. ( 2.4 )
Warnings and Precautions
• Pancreatitis: There have been postmarketing reports of acute pancreatitis. If pancreatitis is suspected, promptly discontinue saxagliptin and metformin hydrochloride extended-release tablets. ( 5.2 ) • Heart Failure: Consider the risks and benefits of saxagliptin and metformin hydrochloride extended-release tablets in patients who have known risk factors for heart failure. Monitor patients for signs and symptoms. ( 5.3 ) • Vitamin B 12 Deficiency: Metformin may lower vitamin B 12 levels. Measure hematological parameters annually. ( 5.4 ) • Hypoglycemia with Concomitant Use of Insulin or Insulin Secretagogues: Consider a lower dosage of insulin or insulin secretagogue when used in combination with saxagliptin and metformin hydrochloride extended-release tablets. ( 5.5 ) • Hypersensitivity-Related Events: There have been post-marketing reports of serious hypersensitivity reactions, such as anaphylaxis, angioedema, and exfoliative skin conditions in patients treated with saxagliptin. If hypersensitivity reactions occur, discontinue saxagliptin and metformin hydrochloride extended-release tablets, treat promptly, and monitor until signs and symptoms resolve. ( 5.6 ) • Arthralgia: Severe and disabling arthralgia has been reported in patients taking DPP4 inhibitors. Consider as a possible cause for severe joint pain and discontinue drug if appropriate. ( 5.7 ) • Bullous Pemphigoid: There have been postmarketing reports of bullous pemphigoid requiring hospitalization in patients taking DPP-4 inhibitors. Tell patients to report development of blisters or erosions. If bullous pemphigoid is suspected, discontinue saxagliptin and metformin hydrochloride extended-release tablets ( 5.8 ).
Contraindications
Saxagliptin and metformin hydrochloride extended-release tablets are contraindicated in patients with: • Severe renal impairment (eGFR below 30 mL/min/1.73 m 2 ). • Acute or chronic metabolic acidosis, including diabetic ketoacidosis. Diabetic ketoacidosis should be treated with insulin. • A history of a serious hypersensitivity reaction to saxagliptin, metformin HCl, or any of the ingredients in saxagliptin and metformin hydrochloride extended-release tablets. Reactions such as anaphylaxis, angioedema, or exfoliative skin conditions have been reported [ see Warnings and Precautions (5.6) and Adverse Reactions (6.2) ].
Adverse Reactions
In the add-on to insulin trial [ see Clinical Studies (14.1) ], the incidence of adverse events, including serious adverse events and discontinuations due to adverse events, was similar between saxagliptin and placebo, except for confirmed hypoglycemia [ see Adverse Reactions (6.1) ].
Drug Interactions
Table 3: Effect of Coadministered Drug on Systemic Exposures of Saxagliptin and its Active Metabolite, 5-hydroxy Saxagliptin ND = not determined; QD = once daily; q6h = every 6 hours; q12h = every 12 hours; BID = twice daily; LA = long acting. Coadministered Drug Dosage of Coadministered Drug Single dose unless otherwise noted. The 10 mg saxagliptin dose is not an approved dosage. Dosage of Saxagliptin Geometric Mean Ratio (ratio with/without coadministered drug) No Effect = 1.00 AUC AUC = AUC(INF) for drugs given as single dose and AUC = AUC(TAU) for drugs given in multiple doses. C max No dosing adjustments required for the following: Metformin 1,000 mg 100 mg saxagliptin 0.98 0.79 5-hydroxy saxagliptin 0.99 0.88 Glyburide 5 mg 10 mg saxagliptin 0.98 1.08 5-hydroxy saxagliptin ND ND Pioglitazone Results exclude one patient. 45 mg QD for 10 days 10 mg QD for 5 days saxagliptin 1.11 1.11 5-hydroxy saxagliptin ND ND Digoxin 0.25 mg q6h first day followed by q12h second day followed by QD for 5 days 10 mg QD for 7 days saxagliptin 1.05 0.99 5-hydroxy saxagliptin 1.06 1.02 Dapagliflozin 10 mg single dose 5 mg single dose saxagliptin ↓ 1% ↓ 7% 5-hydroxy saxagliptin ↑ 9% ↑ 6% Simvastatin 40 mg QD for 8 days 10 mg QD for 4 days saxagliptin 1.12 1.21 5-hydroxy saxagliptin 1.02 1.08 Diltiazem 360 mg LA QD for 9 days 10 mg saxagliptin 2.09 1.63 5-hydroxy saxagliptin 0.66 0.57 Rifampin The plasma dipeptidyl peptidase-4 (DPP4) activity inhibition over a 24-hour dose interval was not affected by rifampin. 600 mg QD for 6 days 5 mg saxagliptin 0.24 0.47 5-hydroxy saxagliptin 1.03 1.39 Omeprazole 40 mg QD for 5 days 10 mg saxagliptin 1.13 0.98 5-hydroxy saxagliptin ND ND Aluminum hydroxide + magnesium hydroxide + simethicone aluminum hydroxide: 2400 mg magnesium hydroxide: 2400 mg simethicone: 240 mg 10 mg saxagliptin 0.97 0.74 5-hydroxy saxagliptin ND ND Famotidine 40 mg 10 mg saxagliptin 1.03 1.14 5-hydroxy saxagliptin ND ND Limit saxagliptin and metformin hydrochloride extended-release tablet dose to 2.5 mg/1,000 mg once daily when coadministered with strong CYP3A4/5 inhibitors [ see Drug Interactions (7.1) and Dosage and Administration (2.2) ]: Ketoconazole 200 mg BID for 9 days 100 mg saxagliptin 2.45 1.62 5-hydroxy saxagliptin 0.12 0.05 Ketoconazole 200 mg BID for 7 days 20 mg saxagliptin 3.67 2.44 5-hydroxy saxagliptin ND ND Table 4: Effect of Saxagliptin on Systemic Exposures of Coadministered Drugs ND = not determined; QD = once daily; q6h = every 6 hours; q12h = every 12 hours; BID = twice daily; LA = long acting. Coadministered Drug Dosage of Coadministered Drug Single dose unless otherwise noted. The 10 mg saxagliptin dose is not an approved dosage. Dosage of Saxagliptin Geometric Mean Ratio (ratio with/without saxagliptin) No Effect = 1.00 AUC AUC = AUC(INF) for drugs given as single dose and AUC = AUC(TAU) for drugs given in multiple doses. C max No dosing adjustments required for the following: Metformin 1,000 mg 100 mg metformin 1.20 1.09 Glyburide 5 mg 10 mg glyburide 1.06 1.16 Pioglitazone Results include all patients. 45 mg QD for 10 days 10 mg QD for 5 days pioglitazone 1.08 1.14 hydroxy-pioglitazone ND ND Digoxin 0.25 mg q6h first day followed by q12h second day followed by QD for 5 days 10 mg QD for 7 days digoxin 1.06 1.09 Simvastatin 40 mg QD for 8 days 10 mg QD for 4 days simvastatin 1.04 0.88 simvastatin acid 1.16 1.00 Diltiazem 360 mg LA QD for 9 days 10 mg diltiazem 1.10 1.16 Ketoconazole 200 mg BID for 9 days 100 mg ketoconazole 0.87 0.84 Ethinyl estradiol and norgestimate ethinyl estradiol 0.035 mg and norgestimate 0.250 mg for 21 days 5 mg QD for 21 days ethinyl estradiol 1.07 0.98 norelgestromin 1.10 1.09 norgestrel 1.13 1.17 Table 5: Effect of Coadministered Drug on Plasma Metformin Systemic Exposure Coadministered Drug Dose of Coadministered Drug All metformin and coadministered drugs were given as single doses. Dose of Metformin Geometric Mean Ratio (ratio with/without coadministered drug) No Effect = 1.00 AUC AUC = AUC(INF). C max No dosing adjustments required for the following: Glyburide 5 mg 850 mg metformin 0.91 Ratio of arithmetic means. 0.93 Furosemide 40 mg 850 mg metformin 1.09 1.22 Nifedipine 10 mg 850 mg metformin 1.16 1.21 Propranolol 40 mg 850 mg metformin 0.90 0.94 Ibuprofen 400 mg 850 mg metformin 1.05 1.07 Drugs that are eliminated by renal tubular secretion may increase the accumulation of metformin [ see Drug Interactions (7.3) ]. Cimetidine 400 mg 850 mg metformin 1.40 1.61 Table 6: Effect of Metformin on Coadministered Drug Systemic Exposure Coadministered Drug Dose of Coadministered Drug All metformin and coadministered drugs were given as single doses. Dose of Metformin Geometric Mean Ratio (ratio with/without metformin) No Effect = 1.00 AUC AUC = AUC(INF) unless otherwise noted. C max No dosing adjustments required for the following: Glyburide 5 mg 850 mg glyburide 0.78 Ratio of arithmetic means, p-value of difference < 0.05. 0.63 Furosemide 40 mg 850 mg furosemide 0.87 0.69 Nifedipine 10 mg 850 mg nifedipine 1.10 AUC(0-24 hr) reported. 1.08 Propranolol 40 mg 850 mg propranolol 1.01 1.02 Ibuprofen 400 mg 850 mg ibuprofen 0.97 Ratio of arithmetic means. 1.01 Cimetidine 400 mg 850 mg cimetidine 0.95 1.01
Storage and Handling
How Supplied Saxagliptin and Metformin Hydrochloride Extended-Release Tablets, 5 mg/500 mg, are pink, film-coated, round, unscored tablets imprinted with SM3 over M in black ink on one side of the tablet and blank on the other side. They are available as follows: NDC 0378-8175-93 bottles of 30 tablets Saxagliptin and Metformin Hydrochloride Extended-Release Tablets, 5 mg/1000 mg, are pink, film-coated, capsule shaped, unscored tablets imprinted with SM6 over M in black ink on one side of the tablet and blank on the other side. They are available as follows: NDC 0378-8177-93 bottles of 30 tablets Saxagliptin and Metformin Hydrochloride Extended-Release Tablets, 2.5 mg/1000 mg, are light peach, film-coated, capsule shaped, unscored tablets imprinted with SM4 over M in black ink on one side of the tablet and blank on the other side. They are available as follows: NDC 0378-8176-91 bottles of 60 tablets Storage and Handling Store at 20° to 25°C (68° to 77°F). [See USP Controlled Room Temperature.] Dispense in a tight, light-resistant container as defined in the USP using a child-resistant closure. PHARMACIST: Dispense a Medication Guide with each prescription.
How Supplied
How Supplied Saxagliptin and Metformin Hydrochloride Extended-Release Tablets, 5 mg/500 mg, are pink, film-coated, round, unscored tablets imprinted with SM3 over M in black ink on one side of the tablet and blank on the other side. They are available as follows: NDC 0378-8175-93 bottles of 30 tablets Saxagliptin and Metformin Hydrochloride Extended-Release Tablets, 5 mg/1000 mg, are pink, film-coated, capsule shaped, unscored tablets imprinted with SM6 over M in black ink on one side of the tablet and blank on the other side. They are available as follows: NDC 0378-8177-93 bottles of 30 tablets Saxagliptin and Metformin Hydrochloride Extended-Release Tablets, 2.5 mg/1000 mg, are light peach, film-coated, capsule shaped, unscored tablets imprinted with SM4 over M in black ink on one side of the tablet and blank on the other side. They are available as follows: NDC 0378-8176-91 bottles of 60 tablets Storage and Handling Store at 20° to 25°C (68° to 77°F). [See USP Controlled Room Temperature.] Dispense in a tight, light-resistant container as defined in the USP using a child-resistant closure. PHARMACIST: Dispense a Medication Guide with each prescription.
Medication Information
Warnings and Precautions
• Pancreatitis: There have been postmarketing reports of acute pancreatitis. If pancreatitis is suspected, promptly discontinue saxagliptin and metformin hydrochloride extended-release tablets. ( 5.2 ) • Heart Failure: Consider the risks and benefits of saxagliptin and metformin hydrochloride extended-release tablets in patients who have known risk factors for heart failure. Monitor patients for signs and symptoms. ( 5.3 ) • Vitamin B 12 Deficiency: Metformin may lower vitamin B 12 levels. Measure hematological parameters annually. ( 5.4 ) • Hypoglycemia with Concomitant Use of Insulin or Insulin Secretagogues: Consider a lower dosage of insulin or insulin secretagogue when used in combination with saxagliptin and metformin hydrochloride extended-release tablets. ( 5.5 ) • Hypersensitivity-Related Events: There have been post-marketing reports of serious hypersensitivity reactions, such as anaphylaxis, angioedema, and exfoliative skin conditions in patients treated with saxagliptin. If hypersensitivity reactions occur, discontinue saxagliptin and metformin hydrochloride extended-release tablets, treat promptly, and monitor until signs and symptoms resolve. ( 5.6 ) • Arthralgia: Severe and disabling arthralgia has been reported in patients taking DPP4 inhibitors. Consider as a possible cause for severe joint pain and discontinue drug if appropriate. ( 5.7 ) • Bullous Pemphigoid: There have been postmarketing reports of bullous pemphigoid requiring hospitalization in patients taking DPP-4 inhibitors. Tell patients to report development of blisters or erosions. If bullous pemphigoid is suspected, discontinue saxagliptin and metformin hydrochloride extended-release tablets ( 5.8 ).
Indications and Usage
Saxagliptin and metformin hydrochloride extended-release tablets are indicated as an adjunct to diet and exercise to improve glycemic control in adults with type 2 diabetes mellitus [ see Clinical Studies (14) ].
Dosage and Administration
• Administer once daily with the evening meal. ( 2.1 ) • Individualize the starting dosage based on the patient’s current regimen then adjust the dosage based on effectiveness and tolerability. ( 2.1 ) • Do not exceed a daily dosage of 5 mg saxagliptin/2,000 mg metformin HCl extended-release. ( 2.1 ) • Swallow whole. Never crush, cut, or chew. ( 2.1 ) • Limit the saxagliptin dosage to 2.5 mg daily for patients also taking strong cytochrome P450 3A4/5 inhibitors (e.g., ketoconazole). ( 2.3 , 7.1 ) • Assess renal function prior to initiation of saxagliptin and metformin hydrochloride extended-release tablets and periodically thereafter. ( 2.2 ) o Do not use in patients with eGFR below 30 mL/min/1.73 m 2 . o Initiation is not recommended in patients with eGFR between 30 - 45 mL/min/1.73 m 2 . o Assess risk/benefit of continuing if eGFR falls below 45 mL/min/1.73 m 2 . o Limit the saxagliptin component to 2.5 mg daily if eGFR is less than 45 mL/min/1.73 m 2 . o Discontinue if eGFR falls below 30 mL/min/1.73 m 2 . • Saxagliptin and metformin hydrochloride extended-release tablets may need to be discontinued at time of, or prior to, iodinated contrast imaging procedures. ( 2.4 )
Contraindications
Saxagliptin and metformin hydrochloride extended-release tablets are contraindicated in patients with: • Severe renal impairment (eGFR below 30 mL/min/1.73 m 2 ). • Acute or chronic metabolic acidosis, including diabetic ketoacidosis. Diabetic ketoacidosis should be treated with insulin. • A history of a serious hypersensitivity reaction to saxagliptin, metformin HCl, or any of the ingredients in saxagliptin and metformin hydrochloride extended-release tablets. Reactions such as anaphylaxis, angioedema, or exfoliative skin conditions have been reported [ see Warnings and Precautions (5.6) and Adverse Reactions (6.2) ].
Adverse Reactions
In the add-on to insulin trial [ see Clinical Studies (14.1) ], the incidence of adverse events, including serious adverse events and discontinuations due to adverse events, was similar between saxagliptin and placebo, except for confirmed hypoglycemia [ see Adverse Reactions (6.1) ].
Drug Interactions
Table 3: Effect of Coadministered Drug on Systemic Exposures of Saxagliptin and its Active Metabolite, 5-hydroxy Saxagliptin ND = not determined; QD = once daily; q6h = every 6 hours; q12h = every 12 hours; BID = twice daily; LA = long acting. Coadministered Drug Dosage of Coadministered Drug Single dose unless otherwise noted. The 10 mg saxagliptin dose is not an approved dosage. Dosage of Saxagliptin Geometric Mean Ratio (ratio with/without coadministered drug) No Effect = 1.00 AUC AUC = AUC(INF) for drugs given as single dose and AUC = AUC(TAU) for drugs given in multiple doses. C max No dosing adjustments required for the following: Metformin 1,000 mg 100 mg saxagliptin 0.98 0.79 5-hydroxy saxagliptin 0.99 0.88 Glyburide 5 mg 10 mg saxagliptin 0.98 1.08 5-hydroxy saxagliptin ND ND Pioglitazone Results exclude one patient. 45 mg QD for 10 days 10 mg QD for 5 days saxagliptin 1.11 1.11 5-hydroxy saxagliptin ND ND Digoxin 0.25 mg q6h first day followed by q12h second day followed by QD for 5 days 10 mg QD for 7 days saxagliptin 1.05 0.99 5-hydroxy saxagliptin 1.06 1.02 Dapagliflozin 10 mg single dose 5 mg single dose saxagliptin ↓ 1% ↓ 7% 5-hydroxy saxagliptin ↑ 9% ↑ 6% Simvastatin 40 mg QD for 8 days 10 mg QD for 4 days saxagliptin 1.12 1.21 5-hydroxy saxagliptin 1.02 1.08 Diltiazem 360 mg LA QD for 9 days 10 mg saxagliptin 2.09 1.63 5-hydroxy saxagliptin 0.66 0.57 Rifampin The plasma dipeptidyl peptidase-4 (DPP4) activity inhibition over a 24-hour dose interval was not affected by rifampin. 600 mg QD for 6 days 5 mg saxagliptin 0.24 0.47 5-hydroxy saxagliptin 1.03 1.39 Omeprazole 40 mg QD for 5 days 10 mg saxagliptin 1.13 0.98 5-hydroxy saxagliptin ND ND Aluminum hydroxide + magnesium hydroxide + simethicone aluminum hydroxide: 2400 mg magnesium hydroxide: 2400 mg simethicone: 240 mg 10 mg saxagliptin 0.97 0.74 5-hydroxy saxagliptin ND ND Famotidine 40 mg 10 mg saxagliptin 1.03 1.14 5-hydroxy saxagliptin ND ND Limit saxagliptin and metformin hydrochloride extended-release tablet dose to 2.5 mg/1,000 mg once daily when coadministered with strong CYP3A4/5 inhibitors [ see Drug Interactions (7.1) and Dosage and Administration (2.2) ]: Ketoconazole 200 mg BID for 9 days 100 mg saxagliptin 2.45 1.62 5-hydroxy saxagliptin 0.12 0.05 Ketoconazole 200 mg BID for 7 days 20 mg saxagliptin 3.67 2.44 5-hydroxy saxagliptin ND ND Table 4: Effect of Saxagliptin on Systemic Exposures of Coadministered Drugs ND = not determined; QD = once daily; q6h = every 6 hours; q12h = every 12 hours; BID = twice daily; LA = long acting. Coadministered Drug Dosage of Coadministered Drug Single dose unless otherwise noted. The 10 mg saxagliptin dose is not an approved dosage. Dosage of Saxagliptin Geometric Mean Ratio (ratio with/without saxagliptin) No Effect = 1.00 AUC AUC = AUC(INF) for drugs given as single dose and AUC = AUC(TAU) for drugs given in multiple doses. C max No dosing adjustments required for the following: Metformin 1,000 mg 100 mg metformin 1.20 1.09 Glyburide 5 mg 10 mg glyburide 1.06 1.16 Pioglitazone Results include all patients. 45 mg QD for 10 days 10 mg QD for 5 days pioglitazone 1.08 1.14 hydroxy-pioglitazone ND ND Digoxin 0.25 mg q6h first day followed by q12h second day followed by QD for 5 days 10 mg QD for 7 days digoxin 1.06 1.09 Simvastatin 40 mg QD for 8 days 10 mg QD for 4 days simvastatin 1.04 0.88 simvastatin acid 1.16 1.00 Diltiazem 360 mg LA QD for 9 days 10 mg diltiazem 1.10 1.16 Ketoconazole 200 mg BID for 9 days 100 mg ketoconazole 0.87 0.84 Ethinyl estradiol and norgestimate ethinyl estradiol 0.035 mg and norgestimate 0.250 mg for 21 days 5 mg QD for 21 days ethinyl estradiol 1.07 0.98 norelgestromin 1.10 1.09 norgestrel 1.13 1.17 Table 5: Effect of Coadministered Drug on Plasma Metformin Systemic Exposure Coadministered Drug Dose of Coadministered Drug All metformin and coadministered drugs were given as single doses. Dose of Metformin Geometric Mean Ratio (ratio with/without coadministered drug) No Effect = 1.00 AUC AUC = AUC(INF). C max No dosing adjustments required for the following: Glyburide 5 mg 850 mg metformin 0.91 Ratio of arithmetic means. 0.93 Furosemide 40 mg 850 mg metformin 1.09 1.22 Nifedipine 10 mg 850 mg metformin 1.16 1.21 Propranolol 40 mg 850 mg metformin 0.90 0.94 Ibuprofen 400 mg 850 mg metformin 1.05 1.07 Drugs that are eliminated by renal tubular secretion may increase the accumulation of metformin [ see Drug Interactions (7.3) ]. Cimetidine 400 mg 850 mg metformin 1.40 1.61 Table 6: Effect of Metformin on Coadministered Drug Systemic Exposure Coadministered Drug Dose of Coadministered Drug All metformin and coadministered drugs were given as single doses. Dose of Metformin Geometric Mean Ratio (ratio with/without metformin) No Effect = 1.00 AUC AUC = AUC(INF) unless otherwise noted. C max No dosing adjustments required for the following: Glyburide 5 mg 850 mg glyburide 0.78 Ratio of arithmetic means, p-value of difference < 0.05. 0.63 Furosemide 40 mg 850 mg furosemide 0.87 0.69 Nifedipine 10 mg 850 mg nifedipine 1.10 AUC(0-24 hr) reported. 1.08 Propranolol 40 mg 850 mg propranolol 1.01 1.02 Ibuprofen 400 mg 850 mg ibuprofen 0.97 Ratio of arithmetic means. 1.01 Cimetidine 400 mg 850 mg cimetidine 0.95 1.01
Storage and Handling
How Supplied Saxagliptin and Metformin Hydrochloride Extended-Release Tablets, 5 mg/500 mg, are pink, film-coated, round, unscored tablets imprinted with SM3 over M in black ink on one side of the tablet and blank on the other side. They are available as follows: NDC 0378-8175-93 bottles of 30 tablets Saxagliptin and Metformin Hydrochloride Extended-Release Tablets, 5 mg/1000 mg, are pink, film-coated, capsule shaped, unscored tablets imprinted with SM6 over M in black ink on one side of the tablet and blank on the other side. They are available as follows: NDC 0378-8177-93 bottles of 30 tablets Saxagliptin and Metformin Hydrochloride Extended-Release Tablets, 2.5 mg/1000 mg, are light peach, film-coated, capsule shaped, unscored tablets imprinted with SM4 over M in black ink on one side of the tablet and blank on the other side. They are available as follows: NDC 0378-8176-91 bottles of 60 tablets Storage and Handling Store at 20° to 25°C (68° to 77°F). [See USP Controlled Room Temperature.] Dispense in a tight, light-resistant container as defined in the USP using a child-resistant closure. PHARMACIST: Dispense a Medication Guide with each prescription.
How Supplied
How Supplied Saxagliptin and Metformin Hydrochloride Extended-Release Tablets, 5 mg/500 mg, are pink, film-coated, round, unscored tablets imprinted with SM3 over M in black ink on one side of the tablet and blank on the other side. They are available as follows: NDC 0378-8175-93 bottles of 30 tablets Saxagliptin and Metformin Hydrochloride Extended-Release Tablets, 5 mg/1000 mg, are pink, film-coated, capsule shaped, unscored tablets imprinted with SM6 over M in black ink on one side of the tablet and blank on the other side. They are available as follows: NDC 0378-8177-93 bottles of 30 tablets Saxagliptin and Metformin Hydrochloride Extended-Release Tablets, 2.5 mg/1000 mg, are light peach, film-coated, capsule shaped, unscored tablets imprinted with SM4 over M in black ink on one side of the tablet and blank on the other side. They are available as follows: NDC 0378-8176-91 bottles of 60 tablets Storage and Handling Store at 20° to 25°C (68° to 77°F). [See USP Controlled Room Temperature.] Dispense in a tight, light-resistant container as defined in the USP using a child-resistant closure. PHARMACIST: Dispense a Medication Guide with each prescription.
Description
• Post-marketing cases of metformin-associated lactic acidosis have resulted in death, hypothermia, hypotension, and resistant bradyarrhythmias. The onset of metformin-associated lactic acidosis is often subtle, accompanied only by nonspecific symptoms such as malaise, myalgias, respiratory distress, somnolence, and abdominal pain. Metformin-associated lactic acidosis was characterized by elevated blood lactate levels (> 5 mmol/Liter), anion gap acidosis (without evidence of ketonuria or ketonemia), an increased lactate/pyruvate ratio; and metformin plasma levels generally > 5 mcg/mL [ see Warnings and Precautions (5.1) ]. • Risk factors for metformin-associated lactic acidosis include renal impairment, concomitant use of certain drugs (e.g., carbonic anhydrase inhibitors such as topiramate), age 65 years old or greater, having a radiological study with contrast, surgery and other procedures, hypoxic states (e.g., acute congestive heart failure), excessive alcohol intake, and hepatic impairment. • Steps to reduce the risk of and manage metformin-associated lactic acidosis in these high risk groups are provided in the full prescribing information [ see Dosage and Administration (2.2) , Contraindications (4) , Warnings and Precautions (5.1) , Drug Interactions (7) , and Use in Specific Populations (8.6 , 8.7) ]. • If metformin-associated lactic acidosis is suspected, immediately discontinue saxagliptin and metformin hydrochloride extended-release tablets and institute general supportive measures in a hospital setting. Prompt hemodialysis is recommended [ see Warnings and Precautions (5.1) ].
Excretion
Saxagliptin
Saxagliptin is eliminated by both renal and hepatic pathways. Following a single 50 mg dose of 14C-saxagliptin, 24%, 36%, and 75% of the dose was excreted in the urine as saxagliptin, its active metabolite, and total radioactivity, respectively. The average renal clearance of saxagliptin (~ 230 mL/min) was greater than the average estimated glomerular filtration rate (~ 120 mL/min), suggesting some active renal excretion. A total of 22% of the administered radioactivity was recovered in feces representing the fraction of the saxagliptin dose excreted in bile and/or unabsorbed drug from the gastrointestinal tract. Following a single oral dose of saxagliptin 5 mg to healthy subjects, the mean plasma terminal half-life (t1/2) for saxagliptin and its active metabolite was 2.5 and 3.1 hours, respectively.
Metformin HCl
Renal clearance is approximately 3.5 times greater than creatinine clearance, which indicates that tubular secretion is the major route of metformin elimination. Following oral administration, approximately 90% of the absorbed drug is eliminated via the renal route within the first 24 hours, with a plasma elimination half-life of approximately 6.2 hours. In blood, the elimination half-life is approximately 17.6 hours, suggesting that the erythrocyte mass may be a compartment of distribution.
Human Data
Published clinical lactation studies report that metformin is present in human milk which resulted in infant doses approximately 0.11% to 1% of the maternal weight-adjusted dosage and a milk/plasma ratio ranging between 0.13 and 1. However, the studies were not designed to definitely establish the risk of use of metformin during lactation because of small sample size and limited adverse event data collected in infants.
Metabolism
Saxagliptin
The metabolism of saxagliptin is primarily mediated by cytochrome P450 3A4/5 (CYP3A4/5). The major metabolite of saxagliptin is also a DPP4 inhibitor, which is one-half as potent as saxagliptin. Therefore, strong CYP3A4/5 inhibitors and inducers will alter the pharmacokinetics of saxagliptin and its active metabolite [see Drug Interactions (7.1) ].
Metformin HCl
Intravenous single-dose studies in healthy subjects demonstrate that metformin is excreted unchanged in the urine and does not undergo hepatic metabolism (no metabolites have been identified in humans) or biliary excretion.
Metabolism studies with extended-release metformin tablets have not been conducted.
7.4 Alcohol
Alcohol is known to potentiate the effect of metformin on lactate metabolism. Warn patients against excessive alcohol intake while receiving saxagliptin and metformin hydrochloride extended-release tablets.
Animal Data
Saxagliptin
In embryo-fetal development studies, saxagliptin was administered to pregnant rats and rabbits during the period of organogenesis, corresponding to the first trimester of human pregnancy. No adverse developmental effects were observed in either species at exposures 1503- and 152-times the 5 mg clinical dose in rats and rabbits, respectively, based on AUC. Saxagliptin crosses the placenta into the fetus following dosing in pregnant rats.
In a prenatal and postnatal development study, no adverse developmental effects were observed in maternal rats administered saxagliptin from gestation day 6 through lactation day 21 at exposures up to 470-times the 5 mg clinical dose, based on AUC.
Metformin HCl
Metformin HCl did not cause adverse developmental effect when administered to pregnant Sprague Dawley rats and rabbits up to 600 mg/kg/day during the period of organogenesis. This represents an exposure of about 2- and 6-times a 2,000 mg clinical dose based on body surface area (mg/m2) for rats and rabbits, respectively.
Saxagliptin and Metformin
Saxagliptin and metformin coadministered to pregnant rats and rabbits during the period of organogenesis did not result in adverse developmental effects considered clinically relevant in either species. Doses tested in rats provided exposure up to 100- and 10-times clinical exposure, and doses tested in rabbits provided exposure up to 249- and 1-times clinical exposure relative to the clinical dose of 5 mg saxagliptin and 2,000 mg metformin. Minor skeletal abnormalities associated with maternal toxicity were observed in rats. In rabbits, coadministration was poorly tolerated in a subset of mothers (12 of 30), resulting in death, moribundity, or abortion. However, among surviving mothers with evaluable litters, maternal toxicity was limited to marginal reductions in body weight over the course of gestation days 21 to 29, associated with fetal body weight decrements of 7%, and a low incidence of delayed ossification of the fetal hyoid bone.
Saxagliptin
When saxagliptin was used in combination with insulin or an insulin secretagogue, the incidence of confirmed hypoglycemia was increased over that of placebo used in combination with insulin or an insulin secretagogue [see Adverse Reactions (6.1) ]. Therefore, a lower dosage of insulin or an insulin secretagogue may be required to reduce the risk of hypoglycemia when used in combination with saxagliptin and metformin hydrochloride extended-release tablets.
Hypoglycemia
In the saxagliptin clinical trials, adverse reactions of hypoglycemia were based on all reports of hypoglycemia. A concurrent glucose measurement was not required or was normal in some patients. Therefore, it is not possible to conclusively determine that all these reports reflect true hypoglycemia.
The incidence of reported hypoglycemia for saxagliptin 2.5 mg and saxagliptin 5 mg versus placebo given as monotherapy was 4% and 5.6% versus 4.1%, respectively. In the add-on to metformin HCl immediate-release trial, the incidence of reported hypoglycemia was 7.8% with saxagliptin 2.5 mg, 5.8% with saxagliptin 5 mg, and 5% with placebo. When saxagliptin and metformin HCl immediate-release were coadministered in treatment-naive patients, the incidence of reported hypoglycemia was 3.4% in patients given saxagliptin 5 mg + metformin HCl immediate-release and 4% in patients given placebo + metformin HCl immediate-release.
In the active-controlled trial comparing add-on therapy with saxagliptin 5 mg to glipizide in patients inadequately controlled on metformin HCl alone, the incidence of reported hypoglycemia was 3% (19 events in 13 patients) with saxagliptin 5 mg versus 36.3% (750 events in 156 patients) with glipizide. Confirmed symptomatic hypoglycemia (accompanying fingerstick blood glucose ≤ 50 mg/dL) was reported in none of the saxagliptin-treated patients and in 35 glipizide-treated patients (8.1%) (p < 0.0001).
In the saxagliptin add-on to insulin trial, the overall incidence of reported hypoglycemia was 18.4% for saxagliptin 5 mg and 19.9% for placebo. However, the incidence of confirmed symptomatic hypoglycemia (accompanying fingerstick blood glucose ≤ 50 mg/dL) was higher with saxagliptin 5 mg (5.3%) versus placebo (3.3%). Among the patients using insulin in combination with metformin HCl, the incidence of confirmed symptomatic hypoglycemia was 4.8% with saxagliptin versus 1.9% with placebo.
In the saxagliptin add-on to metformin HCl plus sulfonylurea trial, the overall incidence of reported hypoglycemia was 10.1% for saxagliptin 5 mg and 6.3% for placebo. Confirmed hypoglycemia was reported in 1.6% of the saxagliptin-treated patients and in none of the placebo-treated patients [see Warnings and Precautions (5.5) ].
Risk Summary
Limited available data with saxagliptin and metformin hydrochloride extended-release tablets or saxagliptin in pregnant women are not sufficient to determine a drug-associated risk for major birth defects and miscarriage. Published trials with metformin use during pregnancy have not reported a clear association with metformin and major birth defect or miscarriage risk [see Data ].
No adverse developmental effects independent of maternal toxicity were observed when saxagliptin and metformin were administered separately or in combination to pregnant rats and rabbits during the period of organogenesis [see Data ].
The estimated background risk of major birth defects is 6 to 10% in women with pre-gestational diabetes with an HbA1c greater than 7 and has been reported to be as high as 20 to 25% in women with an HbA1c greater than 10. The estimated background risk of miscarriage for the indicated population is unknown. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2 to 4% and 15 to 20%, respectively.
10 Overdosage
Saxagliptin
In a controlled clinical trial, once-daily, orally administered saxagliptin in healthy subjects at doses up to 400 mg daily for 2 weeks (80-times the MRHD) had no dose-related clinical adverse reactions and no clinically meaningful effect on QTc interval or heart rate.
In the event of an overdose, initiate appropriate supportive treatment as dictated by the patient’s clinical status. Saxagliptin and its active metabolite are removed by hemodialysis (23% of dose over 4 hours). Contact the Poison Help line (1-800-222-1222) or a medical toxicologist for additional overdosage management recommendations.
Metformin HCl
Overdose of metformin HCl has occurred, including ingestion of amounts greater than 50 grams. Hypoglycemia was reported in approximately 10% of cases, but no causal association with metformin HCl has been established. Lactic acidosis has been reported in approximately 32% of metformin overdose cases [see Warnings and Precautions (5.1) ]. Metformin is dialyzable with a clearance of up to 170 mL/min under good hemodynamic conditions. Therefore, hemodialysis may be useful for removal of accumulated drug from patients in whom metformin overdosage is suspected.
Metformin Hcl
Hypoglycemia does not occur in patients receiving metformin alone under usual circumstances of use, but could occur when caloric intake is deficient, when strenuous exercise is not compensated by caloric supplementation, or during concomitant use with other glucose-lowering agents (such as sulfonylureas and insulin) or ethanol. Elderly, debilitated, or malnourished patients and those with adrenal or pituitary insufficiency or alcohol intoxication are particularly susceptible to hypoglycemic effects. Hypoglycemia may be difficult to recognize in the elderly and in people who are taking beta-adrenergic blocking drugs.
Inform patients using these concomitant medications of the risk of hypoglycemia and educate them on the signs and symptoms of hypoglycemia.
11 Description
Saxagliptin and metformin hydrochloride extended-release tablets contain two oral antihyperglycemic medications used in the management of type 2 diabetes mellitus: saxagliptin and metformin HCl.
Saxagliptin
Saxagliptin is an orally active inhibitor of the dipeptidyl-peptidase-4 (DPP4) enzyme.
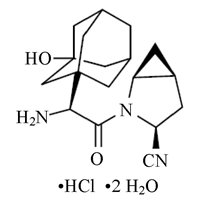
Saxagliptin hydrochloride dihydrate is described chemically as (1S,3S,5S)-2-[(2S)-Amino-(3-hydroxytricyclo[3.3.1.13,7]dec-1-yl)acetyl]-2-azabicyclo[3.1.0]hexane-3-carbonitrile hydrochloride dihydrate or (1S,3S,5S)-2-[(2S)-2-Amino-2-(3-hydroxyadamantan-1-yl)acetyl]-2-azabicyclo[3.1.0]hexane-3-carbonitrile hydrochloride dihydrate. The empirical formula is C18H25N3O2•HCl•2 H2O and the molecular weight is 387.92. The structural formula is:
Saxagliptin hydrochloride dihydrate is a white to off white, non-hygroscopic, crystalline powder. It is sparingly soluble in water at 24°C ± 3°C, slightly soluble in ethyl acetate, and soluble in methanol, ethanol, isopropyl alcohol, acetonitrile, acetone, and polyethylene glycol 400 (PEG 400).
Metformin HCl
Metformin hydrochloride, USP (1,1-Dimethylbiguanide monohydrochloride) is a white, crystalline powder with a molecular formula of C4H11N5•HCl and a molecular weight of 165.62. Metformin HCl is freely soluble in water, slightly soluble in alcohol, and is practically insoluble in acetone, ether, and chloroform. The pKa of metformin is 12.4. The pH of a 1% aqueous solution of metformin HCl is 6.36. The structural formula is:
Saxagliptin and Metformin Hydrochloride Extended-Release Tablets
Saxagliptin and metformin hydrochloride extended-release tablets are available for oral administration as tablets containing either 6.149 mg saxagliptin hydrochloride dihydrate equivalent to 5 mg saxagliptin and 500 mg metformin hydrochloride (saxagliptin and metformin hydrochloride extended-release tablets 5 mg/500 mg), or 6.149 mg saxagliptin hydrochloride dihydrate equivalent to 5 mg saxagliptin and 1000 mg metformin hydrochloride (saxagliptin and metformin hydrochloride extended-release tablets 5 mg/1000 mg), or 3.075 mg saxagliptin hydrochloride dihydrate equivalent to 2.5 mg saxagliptin and 1000 mg metformin hydrochloride (saxagliptin and metformin hydrochloride extended-release tablets 2.5 mg/1000 mg). Each film-coated tablet of saxagliptin and metformin hydrochloride extended-release contains the following inactive ingredients: carbomer homopolymer type A, colloidal silicon dioxide, hypromellose, magnesium stearate, polyethylene glycol, polyvinyl alcohol, povidone, red iron oxide, stearic acid, talc and titanium dioxide. The 2.5 mg/1000 mg tablets also contain yellow iron oxide. In addition, the black imprinting ink contains ammonium hydroxide, black iron oxide, propylene glycol and shellac glaze.
The biologically inert components of the tablet may occasionally remain intact during gastrointestinal transit and will be eliminated in the feces as a soft, hydrated mass.
5.4 Vitamin B12
In controlled clinical trials of metformin of 29-week duration, a decrease to subnormal levels of previously normal serum vitamin B12 levels, without clinical manifestations, was observed in approximately 7% of patients. Such decrease, possibly due to interference with B12 absorption from the B12-intrinsic factor complex, may be associated with anemia but appears to be rapidly reversible with discontinuation of metformin or vitamin B12 supplementation. Certain individuals (those with inadequate vitamin B12 or calcium intake or absorption) appear to be predisposed to developing subnormal vitamin B12 levels. Measure hematologic parameters on an annual basis and vitamin B12 at 2- to 3-year intervals in patients on saxagliptin and metformin hydrochloride extended-release tablets and manage any abnormalities [see Adverse Reactions (6.1) ].
Body Mass Index
Saxagliptin
No dosage adjustment is recommended based on body mass index (BMI) which was not identified as a significant covariate on the apparent clearance of saxagliptin or its active metabolite in the population pharmacokinetic analysis.
5.2 Pancreatitis
There have been post-marketing reports of acute pancreatitis in patients taking saxagliptin. In a cardiovascular outcomes trial enrolling participants with established atherosclerotic cardiovascular disease (ASCVD) or multiple risk factors for ASCVD (SAVOR trial), cases of definite acute pancreatitis were confirmed in 17 of 8240 (0.2%) patients receiving saxagliptin compared to 9 of 8173 (0.1%) receiving placebo. Pre-existing risk factors for pancreatitis were identified in 88% (15/17) of those patients receiving saxagliptin and in 100% (9/9) of those patients receiving placebo.
After initiation of saxagliptin and metformin hydrochloride extended-release tablets, observe patients for signs and symptoms of pancreatitis. If pancreatitis is suspected, promptly discontinue saxagliptin and metformin hydrochloride extended-release tablets and initiate appropriate management. It is unknown whether patients with a history of pancreatitis are at increased risk for the development of pancreatitis while using saxagliptin and metformin hydrochloride extended-release tablets.
Laboratory Tests
Absolute Lymphocyte Counts
Saxagliptin
There was a dose-related mean decrease in absolute lymphocyte count observed with saxagliptin. From a baseline mean absolute lymphocyte count of approximately 2200 cells/microL, mean decreases of approximately 100 and 120 cells/microL with saxagliptin 5 mg and 10 mg, respectively, relative to placebo were observed at 24 weeks in a pooled analysis of five placebo-controlled clinical trials. Similar effects were observed when saxagliptin 5 mg and metformin HCl were coadministered in treatment-naive patients compared to placebo and metformin HCl. There was no difference observed for saxagliptin 2.5 mg relative to placebo. The proportion of patients who were reported to have a lymphocyte count ≤ 750 cells/microL was 0.5%, 1.5%, 1.4%, and 0.4% in the saxagliptin 2.5 mg, 5 mg, 10 mg, and placebo groups, respectively. In most patients, recurrence was not observed with repeated exposure to saxagliptin although some patients had recurrent decreases upon rechallenge that led to discontinuation of saxagliptin. The decreases in lymphocyte count were not associated with clinically relevant adverse reactions. The 10 mg saxagliptin dosage is not an approved dosage.
In the SAVOR trial mean decreases of approximately 84 cells/microL with saxagliptin relative to placebo was observed. The proportion of patients who experienced a decrease in lymphocyte counts to a count of ≤ 750 cells/microL was 1.6% (136/8280) and 1.0% (78/8212) on saxagliptin and placebo, respectively.
The clinical significance of this decrease in lymphocyte count relative to placebo is not known. When clinically indicated, such as in settings of unusual or prolonged infection, lymphocyte count should be measured. The effect of saxagliptin on lymphocyte counts in patients with lymphocyte abnormalities (e.g., human immunodeficiency virus) is unknown.
Vitamin B12 Concentrations
Metformin HCl
In metformin clinical trials of 29-week duration, a decrease to subnormal levels of previously normal serum vitamin B12 levels was observed in approximately 7% of patients.
Medication Guide
|
Saxagliptin and Metformin Hydrochloride Extended-Release Tablets, for oral use
(sax″ a glip′ tin met for′ min hye″ droe klor′ ide) |
|||
|
What is the most important information I should know about saxagliptin and metformin hydrochloride extended-release tablets?
Serious side effects can happen in people taking saxagliptin and metformin hydrochloride extended-release tablets, including:
Stop taking saxagliptin and metformin hydrochloride extended-release tablets and call your healthcare provider right away or go to the nearest hospital emergency room if you have any of the following symptoms of lactic acidosis:
Most people who have had lactic acidosis with metformin have other things that, combined with the metformin, led to the lactic acidosis. Tell your doctor if you have any of the following, because you have a higher chance for getting lactic acidosis with saxagliptin and metformin hydrochloride extended-release tablets if you:
The best way to keep from having a problem with lactic acidosis from metformin is to tell your doctor if you have any of the problems in the list above. Your doctor may decide to stop your saxagliptin and metformin hydrochloride extended-release tablets for a while if you have any of these things.
Saxagliptin and metformin hydrochloride extended-release tablets can have other serious side effects. See “What are the possible side effects of saxagliptin and metformin hydrochloride extended-release tablets?”.
|
|||
|
|
||
|
|||
|
|
||
|
|||
|
What are saxagliptin and metformin hydrochloride extended-release tablets?
It is not known if saxagliptin and metformin hydrochloride extended-release tablets are safe and effective in children. |
|||
|
Who should not take saxagliptin and metformin hydrochloride extended-release tablets?
Do not take saxagliptin and metformin hydrochloride extended-release tablets if you:
|
|||
|
|
||
|
|||
|
Before taking saxagliptin and metformin hydrochloride extended-release tablets, tell your healthcare provider about all of your medical conditions, including if you:
Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements.
Know the medicines you take. Keep a list of them to show your healthcare provider and pharmacist when you get a new medicine. Saxagliptin and metformin hydrochloride extended-release tablets may affect the way other medicines work, and other medicines may affect how saxagliptin and metformin hydrochloride extended-release tablets work.
Tell your healthcare provider if you will be starting or stopping certain other types of medicines, such as antibiotics, or medicines that treat fungus or HIV/AIDS, because your dose of saxagliptin and metformin hydrochloride extended-release tablets might need to be changed. |
|||
|
How should I take saxagliptin and metformin hydrochloride extended-release tablets?
|
|||
|
What are the possible side effects of saxagliptin and metformin hydrochloride extended-release tablets?
Saxagliptin and metformin hydrochloride extended-release tablets can cause serious side effects, including:
|
|||
|
|
|
|
|
|||
|
|
||
|
|||
Common side effects of saxagliptin and metformin hydrochloride extended-release tablets include: |
|||
|
|
|
|
|
Swelling or fluid retention in your hands, feet, or ankles (peripheral edema) may become worse in people who also take a thiazolidinedione to treat diabetes. If you do not know whether you are already on this type of medication, ask your healthcare provider.
These are not all of the possible side effects of saxagliptin and metformin hydrochloride extended-release tablets.
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
|
|||
|
How should I store saxagliptin and metformin hydrochloride extended-release tablets?
Store saxagliptin and metformin hydrochloride extended-release tablets between 20° to 25°C (68° to 77°F).
Keep saxagliptin and metformin hydrochloride extended-release tablets and all medicines out of the reach of children.
|
|||
|
General information about the use of saxagliptin and metformin hydrochloride extended-release tablets
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use saxagliptin and metformin hydrochloride extended-release tablets for a condition for which they were not prescribed. Do not give saxagliptin and metformin hydrochloride extended-release tablets to other people, even if they have the same symptoms you have. They may harm them.
You can ask your pharmacist or healthcare provider for information about saxagliptin and metformin hydrochloride extended-release tablets that is written for health professionals.
|
|||
|
What are the ingredients of saxagliptin and metformin hydrochloride extended-release tablets?
Active ingredients: saxagliptin and metformin HCl.
Inactive ingredients in each tablet: carbomer homopolymer type A, colloidal silicon dioxide, hypromellose, magnesium stearate, polyethylene glycol, polyvinyl alcohol, povidone, red iron oxide, stearic acid, talc and titanium dioxide. The 2.5 mg/1000 mg tablets also contain yellow iron oxide. In addition, the black imprinting ink contains ammonium hydroxide, black iron oxide, propylene glycol and shellac glaze.
|
|||
|
Manufactured for: Mylan Pharmaceuticals Inc., Morgantown, WV 26505 U.S.A. Manufactured by: Mylan Laboratories Limited, Hyderabad — 500 096, India
For more information, call Mylan at 1-877-446-3679 (1-877-4-INFO-RX).
The brands listed are trademarks of their respective owners.
|
This Medication Guide has been approved by the U.S. Food and Drug Administration.
Manufactured for:
Mylan Pharmaceuticals Inc.
Morgantown, WV 26505 U.S.A.
Manufactured by:
Mylan Laboratories Limited
Hyderabad — 500 096, India
75108500
Revised: 2/2025
MXA:SAXMET:R2m/MXA:MG:SAXMET:R2m
Renal Impairment
In the SAVOR trial, adverse reactions related to renal impairment, including laboratory changes (i.e., doubling of serum creatinine compared with baseline and serum creatinine > 6 mg/dL), were reported in 5.8% (483/8280) of saxagliptin-treated patients and 5.1% (422/8212) of placebo-treated patients. The most frequently reported adverse reactions included renal impairment (2.1% vs. 1.9%), acute renal failure (1.4% vs. 1.2%), and renal failure (0.8% vs. 0.9%), in the saxagliptin versus placebo groups, respectively. From baseline to the end of treatment, there was a mean decrease in eGFR of 2.5 mL/min/1.73 m2 for saxagliptin-treated patients and a mean decrease of 2.4 mL/min/1.73 m2 for placebo-treated patients. More patients randomized to saxagliptin (421/5227, 8.1%) compared to patients randomized to placebo (344/5073, 6.8%) had downward shifts in eGFR from > 50 mL/min/1.73 m2 (i.e., normal or mild renal impairment) to ≤ 50 mL/min/1.73 m2 (i.e., moderate or severe renal impairment). The proportions of patients with renal adverse reactions increased with worsening baseline renal function and increased age, regardless of treatment assignment.
5.3 Heart Failure
In a cardiovascular outcomes trial enrolling participants with established ASCVD or multiple risk factors for ASCVD (SAVOR trial), more patients randomized to saxagliptin (289/8280, 3.5%) were hospitalized for heart failure compared to patients randomized to placebo (228/8212, 2.8%). In a time-to-first-event analysis the risk of hospitalization for heart failure was higher in the saxagliptin group (estimated Hazard Ratio: 1.27; 95% CI: 1.07, 1.51). Patients with a prior history of heart failure and patients with renal impairment had a higher risk for hospitalization for heart failure, irrespective of treatment assignment.
Consider the risks and benefits of saxagliptin and metformin hydrochloride extended-release tablets prior to initiating treatment in patients at a higher risk for heart failure. Observe patients for signs and symptoms of heart failure during therapy. Advise patients of the characteristic symptoms of heart failure, and to immediately report such symptoms. If heart failure develops, evaluate and manage according to current standards of care and consider discontinuation of saxagliptin and metformin hydrochloride extended-release tablets.
8.4 Pediatric Use
The safety and effectiveness of saxagliptin and metformin hydrochloride extended-release tablets as an adjunct to diet and exercise to improve glycemic control in patients with type 2 diabetes mellitus have not been established in pediatric patients.
Effectiveness of saxagliptin was not demonstrated in a 26-week, placebo-controlled, double-blind randomized clinical trial with a 26-week safety extension (NCT03199053) in 164 pediatric patients aged 10 to 17 years with inadequately controlled type 2 diabetes mellitus.
Geriatric Patients
Saxagliptin
No dosage adjustment is recommended based on age alone. Elderly subjects (65-80 years) had 23% and 59% higher geometric mean Cmax and geometric mean AUC values, respectively, for saxagliptin than young subjects (18-40 years). Differences in active metabolite pharmacokinetics between elderly and young subjects generally reflected the differences observed in saxagliptin pharmacokinetics. The difference between the pharmacokinetics of saxagliptin and the active metabolite in young and elderly subjects is likely due to multiple factors including declining renal function and metabolic capacity with increasing age. Age was not identified as a significant covariate on the apparent clearance of saxagliptin and its active metabolite in the population pharmacokinetic analysis.
Metformin HCl
Limited data from controlled pharmacokinetic studies of metformin in healthy elderly subjects suggest that total plasma clearance of metformin is decreased, the half-life is prolonged, and Cmax is increased, compared to healthy young subjects. From these data, it appears that the change in metformin pharmacokinetics with aging is primarily accounted for by a change in renal function.
4 Contraindications
Saxagliptin and metformin hydrochloride extended-release tablets are contraindicated in patients with:
-
•Severe renal impairment (eGFR below 30 mL/min/1.73 m2).
-
•Acute or chronic metabolic acidosis, including diabetic ketoacidosis. Diabetic ketoacidosis should be treated with insulin.
-
•A history of a serious hypersensitivity reaction to saxagliptin, metformin HCl, or any of the ingredients in saxagliptin and metformin hydrochloride extended-release tablets. Reactions such as anaphylaxis, angioedema, or exfoliative skin conditions have been reported [see Warnings and Precautions (5.6) and Adverse Reactions (6.2) ].
5.1 Lactic Acidosis
There have been postmarketing cases of metformin-associated lactic acidosis, including fatal cases. These cases had a subtle onset and were accompanied by nonspecific symptoms such as malaise, myalgias, abdominal pain, respiratory distress, or increased somnolence; however, hypothermia, hypotension and resistant bradyarrhythmias have occurred with severe acidosis.
Metformin-associated lactic acidosis was characterized by elevated blood lactate concentrations (> 5 mmol/Liter), anion gap acidosis (without evidence of ketonuria or ketonemia), and an increased lactate: pyruvate ratio; metformin plasma levels generally > 5 mcg/mL. Metformin decreases liver uptake of lactate increasing lactate blood levels which may increase the risk of lactic acidosis, especially in patients at risk.
If metformin-associated lactic acidosis is suspected, general supportive measures should be instituted promptly in a hospital setting, along with immediate discontinuation of saxagliptin and metformin hydrochloride extended-release tablets.
In saxagliptin and metformin hydrochloride extended-release tablet-treated patients with a diagnosis or strong suspicion of lactic acidosis, prompt hemodialysis is recommended to correct the acidosis and remove accumulated metformin (metformin HCl is dialyzable, with a clearance of up to 170 mL/minute under good hemodynamic conditions). Hemodialysis has often resulted in reversal of symptoms and recovery.
Educate patients and their families about the symptoms of lactic acidosis and if these symptoms occur instruct them to discontinue saxagliptin and metformin hydrochloride extended-release tablets and report these symptoms to their health care provider.
For each of the known and possible risk factors for metformin-associated lactic acidosis, recommendations to reduce the risk of and manage metformin-associated lactic acidosis are provided below:
Renal Impairment: The post-marketing metformin-associated lactic acidosis cases primarily occurred in patients with significant renal impairment. The risk of metformin accumulation and metformin-associated lactic acidosis increases with the severity of renal impairment because metformin is substantially excreted by the kidney. Clinical recommendations based upon the patient’s renal function include [see Clinical Pharmacology (12.3) ]:
-
•Before initiating saxagliptin and metformin hydrochloride extended-release tablets, obtain an estimated glomerular filtration rate (eGFR).
-
•Saxagliptin and metformin hydrochloride extended-release tablets are contraindicated in patients with an eGFR less than 30 mL/minute/1.73 m2 [see Contraindications (4) ].
-
•Initiation of saxagliptin and metformin hydrochloride extended-release tablets is not recommended in patients with eGFR between 30 and 45 mL/minute/1.73 m2.
-
•Obtain an eGFR at least annually in all patients taking saxagliptin and metformin hydrochloride extended-release tablets. In patients at increased risk for the development of renal impairment (e.g., the elderly), renal function should be assessed more frequently.
-
•In patients taking saxagliptin and metformin hydrochloride extended-release tablets whose eGFR later falls below 45 mL/minute/1.73 m2, assess the benefit and risk of continuing therapy.
Drug Interactions: The concomitant use of saxagliptin and metformin hydrochloride extended-release tablets with specific drugs may increase the risk of metformin-associated lactic acidosis: those that impair renal function, result in significant hemodynamic change, interfere with acid-base balance or increase metformin accumulation [see Drug Interactions (7) ]. Therefore, consider more frequent monitoring of patients.
Age 65 or Greater: The risk of metformin-associated lactic acidosis increases with the patient’s age because elderly patients have a greater likelihood of having hepatic, renal, or cardiac impairment than younger patients. Assess renal function more frequently in elderly patients [see Use in Specific Populations (8.5) ].
Radiological Studies with Contrast: Administration of intravascular iodinated contrast agents in metformin-treated patients has led to an acute decrease in renal function and the occurrence of lactic acidosis. Stop saxagliptin and metformin hydrochloride extended-release tablets at the time of, or prior to, an iodinated contrast imaging procedure in patients with an eGFR between 30 and 60 mL/min/1.73 m2; in patients with a history of hepatic impairment, alcoholism, or heart failure; or in patients who will be administered intra-arterial iodinated contrast. Re-evaluate eGFR 48 hours after the imaging procedure, and restart saxagliptin and metformin hydrochloride extended-release tablets if renal function is stable.
Surgery and Other Procedures: Withholding of food and fluids during surgical or other procedures may increase the risk for volume depletion, hypotension and renal impairment. Saxagliptin and metformin hydrochloride extended-release tablets should be temporarily discontinued while patients have restricted food and fluid intake.
Hypoxic States: Several of the post-marketing cases of metformin-associated lactic acidosis occurred in the setting of acute congestive heart failure (particularly when accompanied by hypoperfusion and hypoxemia). Cardiovascular collapse (shock), acute myocardial infarction, sepsis, and other conditions associated with hypoxemia have been associated with lactic acidosis and may also cause prerenal azotemia. When such events occur, discontinue saxagliptin and metformin hydrochloride extended-release tablets.
Excessive Alcohol Intake: Alcohol potentiates the effect of metformin on lactate metabolism and this may increase the risk of metformin-associated lactic acidosis. Warn patients against excessive alcohol intake while receiving saxagliptin and metformin hydrochloride extended-release tablets.
Hepatic Impairment: Patients with hepatic impairment have developed with cases of metformin-associated lactic acidosis. This may be due to impaired lactate clearance resulting in higher lactate blood levels. Therefore, avoid use of saxagliptin and metformin hydrochloride extended-release tablets in patients with clinical or laboratory evidence of hepatic disease.
6 Adverse Reactions
The following serious adverse reactions are described below or elsewhere in the prescribing information:
-
•Lactic Acidosis [see Boxed Warning and Warnings and Precautions (5.1) ]
-
•Pancreatitis [see Warnings and Precautions (5.2) ]
-
•Heart Failure [see Warnings and Precautions (5.3) ]
-
•Vitamin B12 Concentrations [see Warnings and Precautions (5.4) ]
-
•Hypoglycemia with Concomitant Use of Insulin or Insulin Secretagogues [see Warnings and Precautions (5.5) ]
-
•Hypersensitivity Reactions [see Warnings and Precautions (5.6) ]
-
•Severe and disabling arthralgia [see Warnings and Precautions (5.7) ]
-
•Bullous pemphigoid [see Warnings and Precautions (5.8) ]
7 Drug Interactions
-
•Strong CYP3A4/5 inhibitors (e.g., ketoconazole): Coadministration with saxagliptin and metformin hydrochloride extended-release tablets significantly increases saxagliptin concentrations. Limit saxagliptin and metformin hydrochloride extended-release tablets dosage to 2.5 mg/1,000 mg once daily when coadministered with a strong CYP3A4/5 inhibitor. (2.3, 7.1)
-
•Carbonic anhydrase inhibitors: May increase risk of lactic acidosis. Consider more frequent monitoring. (7.2)
-
•Drugs that reduce metformin clearance: May increase risk of lactic acidosis. Consider benefits and risks of concomitant use. (7.3)
-
•See full prescribing information for additional drug interactions. (7)
1.1 Limitations of Use
Saxagliptin and metformin hydrochloride extended-release tablets are not recommended for the treatment of type 1 diabetes mellitus or diabetic ketoacidosis.
5.8 Bullous Pemphigoid
Postmarketing cases of bullous pemphigoid requiring hospitalization have been reported with DPP-4 inhibitor use. In reported cases, patients typically recovered with topical or systemic immunosuppressive treatment and discontinuation of the DPP-4 inhibitor. Tell patients to report development of blisters or erosions while receiving saxagliptin and metformin hydrochloride extended-release tablets. If bullous pemphigoid is suspected, saxagliptin and metformin hydrochloride extended-release tablets should be discontinued and referral to a dermatologist should be considered for diagnosis and appropriate treatment.
8.7 Hepatic Impairment
Use of metformin in patients with hepatic impairment has been associated with some cases of lactic acidosis. Saxagliptin and metformin hydrochloride extended-release tablets are not recommended in patients with hepatic impairment [see Warnings and Precautions (5.1) ].
1 Indications and Usage
Saxagliptin and metformin hydrochloride extended-release tablets are indicated as an adjunct to diet and exercise to improve glycemic control in adults with type 2 diabetes mellitus [see Clinical Studies (14) ].
Racial Or Ethnic Groups
Saxagliptin
No dosage adjustment is recommended based on race. The population pharmacokinetic analysis compared the pharmacokinetics of saxagliptin and its active metabolite in 309 White subjects with 105 subjects of other races (consisting of six racial groups). No significant difference in the pharmacokinetics of saxagliptin and its active metabolite were detected between these two populations.
Metformin HCl
No studies of metformin pharmacokinetic parameters according to race have been performed. In controlled clinical studies of metformin in patients with type 2 diabetes mellitus, the antihyperglycemic effect was comparable in Whites (n = 249), Black or African American (n = 51), and Hispanic or Latino ethnicity (n = 24).
Drug Interaction Studies
Specific pharmacokinetic drug interaction studies with saxagliptin and metformin hydrochloride extended-release tablets have not been performed, although such studies have been conducted with the individual saxagliptin and metformin components.
Male and Female Patients
Saxagliptin
No dosage adjustment is recommended based on gender. There were no differences observed in saxagliptin pharmacokinetics between males and females. Compared to males, females had approximately 25% higher exposure values for the active metabolite than males, but this difference is unlikely to be of clinical relevance. Gender was not identified as a significant covariate on the apparent clearance of saxagliptin and its active metabolite in the population pharmacokinetic analysis.
Metformin HCl
Metformin pharmacokinetic parameters did not differ significantly between healthy subjects and patients with type 2 diabetes mellitus when analyzed according to gender (males = 19, females = 16). Similarly, in controlled clinical studies in patients with type 2 diabetes mellitus, the antihyperglycemic effect of metformin was comparable in males and females.
Warning: Lactic Acidosis
-
•Post-marketing cases of metformin-associated lactic acidosis have resulted in death, hypothermia, hypotension, and resistant bradyarrhythmias. The onset of metformin-associated lactic acidosis is often subtle, accompanied only by nonspecific symptoms such as malaise, myalgias, respiratory distress, somnolence, and abdominal pain. Metformin-associated lactic acidosis was characterized by elevated blood lactate levels (> 5 mmol/Liter), anion gap acidosis (without evidence of ketonuria or ketonemia), an increased lactate/pyruvate ratio; and metformin plasma levels generally > 5 mcg/mL [see Warnings and Precautions (5.1) ].
-
•Risk factors for metformin-associated lactic acidosis include renal impairment, concomitant use of certain drugs (e.g., carbonic anhydrase inhibitors such as topiramate), age 65 years old or greater, having a radiological study with contrast, surgery and other procedures, hypoxic states (e.g., acute congestive heart failure), excessive alcohol intake, and hepatic impairment.
-
•Steps to reduce the risk of and manage metformin-associated lactic acidosis in these high risk groups are provided in the full prescribing information [see Dosage and Administration (2.2), Contraindications (4), Warnings and Precautions (5.1), Drug Interactions (7), and Use in Specific Populations (8.6, 8.7) ].
-
•If metformin-associated lactic acidosis is suspected, immediately discontinue saxagliptin and metformin hydrochloride extended-release tablets and institute general supportive measures in a hospital setting. Prompt hemodialysis is recommended [see Warnings and Precautions (5.1) ].
5 Warnings and Precautions
-
•Pancreatitis: There have been postmarketing reports of acute pancreatitis. If pancreatitis is suspected, promptly discontinue saxagliptin and metformin hydrochloride extended-release tablets. (5.2)
-
•Heart Failure: Consider the risks and benefits of saxagliptin and metformin hydrochloride extended-release tablets in patients who have known risk factors for heart failure. Monitor patients for signs and symptoms. (5.3)
-
•Vitamin B12 Deficiency: Metformin may lower vitamin B12 levels. Measure hematological parameters annually. (5.4)
-
•Hypoglycemia with Concomitant Use of Insulin or Insulin Secretagogues: Consider a lower dosage of insulin or insulin secretagogue when used in combination with saxagliptin and metformin hydrochloride extended-release tablets. (5.5)
-
•Hypersensitivity-Related Events: There have been post-marketing reports of serious hypersensitivity reactions, such as anaphylaxis, angioedema, and exfoliative skin conditions in patients treated with saxagliptin. If hypersensitivity reactions occur, discontinue saxagliptin and metformin hydrochloride extended-release tablets, treat promptly, and monitor until signs and symptoms resolve. (5.6)
-
•Arthralgia: Severe and disabling arthralgia has been reported in patients taking DPP4 inhibitors. Consider as a possible cause for severe joint pain and discontinue drug if appropriate. (5.7)
-
•Bullous Pemphigoid: There have been postmarketing reports of bullous pemphigoid requiring hospitalization in patients taking DPP-4 inhibitors. Tell patients to report development of blisters or erosions. If bullous pemphigoid is suspected, discontinue saxagliptin and metformin hydrochloride extended-release tablets (5.8).
2 Dosage and Administration
-
•Administer once daily with the evening meal. (2.1)
-
•Individualize the starting dosage based on the patient’s current regimen then adjust the dosage based on effectiveness and tolerability. (2.1)
-
•Do not exceed a daily dosage of 5 mg saxagliptin/2,000 mg metformin HCl extended-release. (2.1)
-
•Swallow whole. Never crush, cut, or chew. (2.1)
-
•Limit the saxagliptin dosage to 2.5 mg daily for patients also taking strong cytochrome P450 3A4/5 inhibitors (e.g., ketoconazole). (2.3, 7.1)
-
•Assess renal function prior to initiation of saxagliptin and metformin hydrochloride extended-release tablets and periodically thereafter. (2.2)
-
oDo not use in patients with eGFR below 30 mL/min/1.73 m2.
-
oInitiation is not recommended in patients with eGFR between 30 - 45 mL/min/1.73 m2.
-
oAssess risk/benefit of continuing if eGFR falls below 45 mL/min/1.73 m2.
-
oLimit the saxagliptin component to 2.5 mg daily if eGFR is less than 45 mL/min/1.73 m2.
-
oDiscontinue if eGFR falls below 30 mL/min/1.73 m2.
-
-
•Saxagliptin and metformin hydrochloride extended-release tablets may need to be discontinued at time of, or prior to, iodinated contrast imaging procedures. (2.4)
3 Dosage Forms and Strengths
Extended-Release Tablets:
-
•Saxagliptin and Metformin Hydrochloride Extended-Release Tablets, 5 mg/500 mg, are pink, film-coated, round, unscored tablets imprinted with SM3 over M in black ink on one side of the tablet and blank on the other side.
-
•Saxagliptin and Metformin Hydrochloride Extended-Release Tablets, 5 mg/1000 mg, are pink, film-coated, capsule shaped, unscored tablets imprinted with SM6 over M in black ink on one side of the tablet and blank on the other side.
-
•Saxagliptin and Metformin Hydrochloride Extended-Release Tablets, 2.5 mg/1000 mg, are light peach, film-coated, capsule shaped, unscored tablets imprinted with SM4 over M in black ink on one side of the tablet and blank on the other side.
6.2 Postmarketing Experience
Additional adverse reactions have been identified during post-approval use of saxagliptin and metformin hydrochloride extended-release tablets, saxagliptin, or metformin HCl. Because these reactions are reported voluntarily from a population of uncertain size, it is generally not possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Saxagliptin
-
•Gastrointestinal Disorders: Pancreatitis
-
•Immune System Disorders: Hypersensitivity reactions including anaphylaxis, angioedema, and exfoliative skin conditions
-
•Musculoskeletal and Connective Tissue Disorders: Rhabdomyolysis, severe and disabling arthralgia
-
•Skin and Subcutaneous Tissue Disorders: Bullous pemphigoid
Metformin HCl
-
•Hepatobiliary Disorders: Cholestatic, hepatocellular, and mixed hepatocellular liver injury
14.1 Glycemic Efficacy Trials
The effectiveness of saxagliptin and metformin hydrochloride extended-release tablets has been established in clinical trials of coadministration of oral saxagliptin and metformin HCl immediate-release tablets in adults with type 2 diabetes mellitus inadequately controlled on metformin HCl alone and in treatment-naive patients inadequately controlled on diet and exercise alone. In these two trials, treatment with saxagliptin dosed in the morning plus metformin HCl immediate-release tablets at all doses produced statistically significant improvements in A1C, fasting plasma glucose (FPG), and 2-hour postprandial glucose (PPG) following a standard oral glucose tolerance test (OGTT), compared to control. Reductions in A1C were seen across subgroups including gender, age, race, and baseline BMI.
In these two trials, decrease in body weight in the treatment groups given saxagliptin in combination with metformin HCl immediate-release was similar to that in the groups given metformin HCl immediate-release alone. Saxagliptin plus metformin HCl immediate-release was not associated with significant changes from baseline in fasting serum lipids compared to metformin HCl alone.
The coadministration of saxagliptin and metformin HCl immediate-release tablets has also been evaluated in an active-controlled trial comparing add-on therapy with saxagliptin to glipizide in 858 patients inadequately controlled on metformin HCl alone, in a placebo-controlled trial where a subgroup of 314 patients inadequately controlled on insulin plus metformin HCl received add-on therapy with saxagliptin or placebo, a trial comparing saxagliptin to placebo in 257 patients inadequately controlled on metformin HCl plus a sulfonylurea, and a trial comparing saxagliptin to placebo in 315 patients inadequately controlled on dapagliflozin and metformin HCl.
In a 24-week, double-blind, randomized trial, patients treated with metformin HCl immediate-release 500 mg twice daily for at least 8 weeks were randomized to continued treatment with metformin HCl immediate-release 500 mg twice daily or to metformin HCl extended-release either 1,000 mg once daily or 1,500 mg once daily. The mean change in A1C from baseline to Week 24 was 0.1% (95% confidence interval 0%, 0.3%) for the metformin HCl immediate-release treatment arm, 0.3% (95% confidence interval 0.1%, 0.4%) for the 1,000 mg metformin HCl extended-release treatment arm, and 0.1% (95% confidence interval 0%, 0.3%) for the 1,500 mg metformin HCl extended-release treatment arm. Results of this trial suggest that patients receiving metformin HCl immediate-release treatment may be safely switched to metformin HCl extended-release once daily at the same total daily dose, up to 2,000 mg once daily. Following a switch from metformin HCl immediate-release to metformin HCl extended-release, glycemic control should be closely monitored and dosage adjustments made accordingly.
8 Use in Specific Populations
5.6 Hypersensitivity Reactions
There have been post-marketing reports of serious hypersensitivity reactions in patients treated with saxagliptin. These reactions include anaphylaxis, angioedema, and exfoliative skin conditions. Onset of these reactions occurred within the first 3 months after initiation of treatment with saxagliptin, with some reports occurring after the first dose. If a serious hypersensitivity reaction is suspected, discontinue saxagliptin and metformin hydrochloride extended-release tablets, assess for other potential causes for the event, and institute alternative treatment for diabetes [see Adverse Reactions (6.2) ].
Use caution in a patient with a history of angioedema to another dipeptidyl peptidase-4 (DPP4) inhibitor because it is unknown whether such patients will be predisposed to angioedema with saxagliptin and metformin hydrochloride extended-release tablets.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
Patients With Renal Impairment
Saxagliptin
A single-dose, open-label trial was conducted to evaluate the pharmacokinetics of saxagliptin (10 mg dose) in subjects with varying degrees of chronic renal impairment compared to subjects with normal renal function. The 10 mg dosage is not an approved dosage. The degree of renal impairment did not affect Cmax of saxagliptin or its metabolite. In subjects with moderate renal impairment with eGFR 30 to less than 45 mL/min/1.73 m2, severe renal impairment (eGFR 15 to less than 30 mL/min/1.73 m2) and ESRD patient on hemodialysis, the AUC values of saxagliptin or its active metabolite were > 2 fold higher than AUC values in subjects with normal renal function.
Metformin HCl
In patients with decreased renal function, the plasma and blood half-life of metformin is prolonged and the renal clearance is decreased [see Contraindications (4) and Warnings and Precautions (5.1) ].
14.2 Cardiovascular Safety Trial
The cardiovascular risk of saxagliptin was evaluated in SAVOR, a multicenter, multinational, randomized, double-blind trial comparing saxagliptin (N = 8280) to placebo (N = 8212), both administered in combination with standard of care, in adult patients with type 2 diabetes mellitus at high risk for atherosclerotic cardiovascular disease. Of the randomized trial patients, 97.5% completed the trial, and the median duration of follow-up was approximately 2 years. The trial was event-driven, and patients were followed until a sufficient number of events were accrued.
Patients were at least 40 years of age, had A1C ≥ 6.5%, and multiple risk factors (21% of randomized patients) for cardiovascular disease (age ≥ 55 years for men and ≥ 60 years for women plus at least one additional risk factor of dyslipidemia, hypertension, or current cigarette smoking) or established (79% of the randomized patients) cardiovascular disease defined as a history of ischemic heart disease, peripheral vascular disease, or ischemic stroke. The majority of patients were male (67%) and White (75%) with a mean age of 65 years. Approximately 16% of the population had moderate (estimated glomerular filtration rate [eGFR] ≥ 30 to ≤ 50 mL/min) to severe (eGFR < 30 mL/min) renal impairment, and 13% had a prior history of heart failure. Patients had a median duration of type 2 diabetes mellitus of approximately 10 years, and a mean baseline A1C level of 8.0%. Approximately 5% of patients were treated with diet and exercise only at baseline. Overall, the use of diabetes medications was balanced across treatment groups (metformin 69%, insulin 41%, sulfonylureas 40%, and TZDs 6%). The use of cardiovascular disease medications was also balanced (angiotensin-converting enzyme [ACE] inhibitors or angiotensin receptor blockers [ARBs] 79%, statins 78%, aspirin 75%, beta-blockers 62%, and non-aspirin antiplatelet medications 24%).
The primary analysis in SAVOR was time to first occurrence of a Major Adverse Cardiac Event (MACE). A major adverse cardiac event in SAVOR was defined as a cardiovascular death, or a nonfatal myocardial infarction (MI) or a nonfatal ischemic stroke. The trial was designed as a non-inferiority trial with a pre-specified risk margin of 1.3 for the hazard ratio of MACE, and was also powered for a superiority comparison if non-inferiority was demonstrated.
The results of SAVOR, including the contribution of each component to the primary composite endpoint are shown in Table 13. The incidence rate of MACE was similar in both treatment arms: 3.8 MACE per 100 patient-years on placebo vs. 3.8 MACE per 100 patient-years on saxagliptin. The estimated hazard ratio of MACE associated with saxagliptin relative to placebo was 1.00 with a 95.1% confidence interval of (0.89, 1.12). The upper bound of this confidence interval, 1.12, excluded a risk margin larger than 1.3.
|
Saxagliptin |
Placebo |
Hazard Ratio |
|||
|
Number of |
Rate per |
Number of |
Rate per 100 |
(95.1% CI) |
|
|
Composite of first event of CV death, non-fatal MI or non-fatal ischemic stroke (MACE) |
N = 8280 |
Total PY = 16308.8 |
N = 8212 |
Total PY = 16156.0 |
|
|
613 (7.4) |
3.8 |
609 (7.4) |
3.8 |
1.00 (0.89, 1.12) |
|
|
CV death |
245 (3.0) |
1.5 |
234 (2.8) |
1.4 |
|
|
Non-fatal MI |
233 (2.8) |
1.4 |
260 (3.2) |
1.6 |
|
|
Non-fatal |
135 (1.6) |
0.8 |
115 (1.4) |
0.7 |
The Kaplan-Meier-based cumulative event probability is presented in Figure 2 for time to first occurrence of the primary MACE composite endpoint by treatment arm. The curves for both saxagliptin and placebo arms are close together throughout the duration of the trial. The estimated cumulative event probability is approximately linear for both arms, indicating that the incidence of MACE for both arms was constant over the trial duration.
Vital status was obtained for 99% of patients in the trial. There were 798 deaths in the SAVOR trial. Numerically more patients (5.1%) died in the saxagliptin group than in the placebo group (4.6%). The risk of deaths from all cause (Table 14) was not statistically different between the treatment groups (HR: 1.11; 95.1% CI: 0.96, 1.27).
|
Saxagliptin |
Placebo |
Hazard Ratio |
|||
|
Number of |
Rate per 100 |
Number of |
Rate per 100 |
(95.1% CI) |
|
|
N = 8280 |
PY = 16645.3 |
N = 8212 |
PY = 16531.5 |
||
|
All-cause |
420 (5.1) |
2.5 |
378 (4.6) |
2.3 |
1.11 (0.96, 1.27) |
|
CV death |
269 (3.2) |
1.6 |
260 (3.2) |
1.6 |
|
|
Non-CV death |
151 (1.8) |
0.9 |
118 (1.4) |
0.7 |
Patients With Hepatic Impairment
No pharmacokinetic studies of metformin have been conducted in patients with hepatic impairment.
17 Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Medication Guide).
Lactic Acidosis
The risks of lactic acidosis due to the metformin component, its symptoms and conditions that predispose to its development, as noted in Warnings and Precautions (5.1), should be explained to patients. Patients should be advised to discontinue saxagliptin and metformin hydrochloride extended-release tablets immediately and to promptly notify their health care provider if unexplained hyperventilation, myalgia, malaise, unusual somnolence, dizziness, slow or irregular heartbeat, sensation of feeling cold (especially in the extremities), or other nonspecific symptoms occur. Gastrointestinal symptoms are common during initiation of metformin treatment and may occur during initiation of saxagliptin and metformin hydrochloride extended-release tablet therapy; however, patients should consult their physician if they develop unexplained symptoms. Although gastrointestinal symptoms that occur after stabilization are unlikely to be drug related, such an occurrence of symptoms should be evaluated to determine if it may be due to lactic acidosis or other serious disease.
Patients should be counseled against excessive alcohol intake while receiving saxagliptin and metformin hydrochloride extended-release tablets.
Patients should be informed about the importance of regular testing of renal function and hematological parameters when receiving treatment with saxagliptin and metformin hydrochloride extended-release tablets.
Instruct patients to inform their doctor that they are taking saxagliptin and metformin hydrochloride extended-release tablets prior to any surgical or radiological procedure, as temporary discontinuation of saxagliptin and metformin hydrochloride extended-release tablets may be required until renal function has been confirmed to be normal [see Warnings and Precautions (5.1) ].
Pancreatitis
Inform patients that acute pancreatitis has been reported during post-marketing use of saxagliptin. Educate patients that persistent severe abdominal pain, sometimes radiating to the back, which may or may not be accompanied by vomiting, is the hallmark symptom of acute pancreatitis. Instruct patients to promptly discontinue saxagliptin and metformin hydrochloride extended-release tablets and contact their health care provider if persistent severe abdominal pain occurs [see Warnings and Precautions (5.2) ].
Heart Failure
Inform patients of the signs and symptoms of heart failure. Before initiating saxagliptin and metformin hydrochloride extended-release tablets, ask patients if they have a history of heart failure or other risk factors for heart failure including moderate to severe renal impairment. Instruct patients to contact their health care provider as soon as possible if they experience symptoms of heart failure, including increasing shortness of breath, rapid increase in weight or swelling of the feet [see Warnings and Precautions (5.3) ].
Hypoglycemia with Concomitant Use with Insulin or Insulin Secretagogues
Inform patients that hypoglycemia can occur, particularly when insulin or an insulin secretagogue is used in combination with saxagliptin and metformin hydrochloride extended-release tablets. Educate patients about the risks, symptoms and appropriate management of hypoglycemia [see Warnings and Precautions (5.5) ].
Hypersensitivity Reactions
Patients should be informed that serious allergic (hypersensitivity) reactions, such as angioedema, anaphylaxis, and exfoliative skin conditions, have been reported during post-marketing use of saxagliptin. If symptoms of these allergic reactions (such as rash, skin flaking or peeling, urticaria, swelling of the skin, or swelling of the face, lips, tongue, and throat that may cause difficulty in breathing or swallowing) occur, patients must stop taking saxagliptin and metformin hydrochloride extended-release tablets and seek medical advice promptly [see Warnings and Precautions (5.6) ].
Severe and Disabling Arthralgia
Inform patients that severe and disabling joint pain may occur with this class of drugs. The time to onset of symptoms can range from one day to years. Instruct patients to seek medical advice if severe joint pain occurs [see Warnings and Precautions (5.7) ].
Bullous Pemphigoid
Inform patients that bullous pemphigoid may occur with this class of drugs. Instruct patients to seek medical advice if blisters or erosions occur [see Warnings and Precautions (5.8) ].
Administration Instructions
Patients should be informed that saxagliptin and metformin hydrochloride extended-release tablets must be swallowed whole and not crushed or chewed, and that the inactive ingredients may occasionally be eliminated in the feces as a soft mass that may resemble the original tablet.
Missed Dose
If a dose is missed, advise patients to take saxagliptin and metformin hydrochloride extended-release tablets as soon as they remember unless it is time for their next dose. Instruct patients not to take two doses of saxagliptin and metformin hydrochloride extended-release tablets at the same time.
7.2 Carbonic Anhydrase Inhibitors
Topiramate or other carbonic anhydrase inhibitors (e.g., zonisamide, acetazolamide or dichlorphenamide) frequently causes a decrease in serum bicarbonate and induce non-anion gap, hyperchloremic metabolic acidosis. Concomitant use of these drugs with saxagliptin and metformin hydrochloride extended-release tablets may increase the risk for lactic acidosis.
5.7 Severe and Disabling Arthralgia
There have been post-marketing reports of severe and disabling arthralgia in patients taking DPP4 inhibitors. The time to onset of symptoms following initiation of drug therapy varied from one day to years. Patients experienced relief of symptoms upon discontinuation of the medication. A subset of patients experienced a recurrence of symptoms when restarting the same drug or a different DPP4 inhibitor. Consider DPP4 inhibitors as a possible cause for severe joint pain and discontinue drug if appropriate.
16 How Supplied/storage and Handling
How Supplied
Saxagliptin and Metformin Hydrochloride Extended-Release Tablets, 5 mg/500 mg, are pink, film-coated, round, unscored tablets imprinted with SM3 over M in black ink on one side of the tablet and blank on the other side. They are available as follows:
NDC 0378-8175-93
bottles of 30 tablets
Saxagliptin and Metformin Hydrochloride Extended-Release Tablets, 5 mg/1000 mg, are pink, film-coated, capsule shaped, unscored tablets imprinted with SM6 over M in black ink on one side of the tablet and blank on the other side. They are available as follows:
NDC 0378-8177-93
bottles of 30 tablets
Saxagliptin and Metformin Hydrochloride Extended-Release Tablets, 2.5 mg/1000 mg, are light peach, film-coated, capsule shaped, unscored tablets imprinted with SM4 over M in black ink on one side of the tablet and blank on the other side. They are available as follows:
NDC 0378-8176-91
bottles of 60 tablets
Storage and Handling
Store at 20° to 25°C (68° to 77°F). [See USP Controlled Room Temperature.]
Dispense in a tight, light-resistant container as defined in the USP using a child-resistant closure.
PHARMACIST: Dispense a Medication Guide with each prescription.
7.5 Insulin Or Insulin Secretagogues
Insulin and insulin secretagogues are known to cause hypoglycemia. Concomitant use of saxagliptin and metformin hydrochloride extended-release tablets with insulin or an insulin secretagogue may require lower dosages of insulin or the insulin secretagogue to reduce the risk of hypoglycemia [see Warnings and Precautions (5.5) ].
7.6 Drugs Affecting Glycemic Control
Certain drugs tend to produce hyperglycemia and may lead to loss of glycemic control. These medications include the thiazides and other diuretics, corticosteroids, phenothiazines, thyroid products, estrogens, oral contraceptives, phenytoin, nicotinic acid, sympathomimetics, calcium channel blocking drugs, and isoniazid. When such drugs are administered to a patient receiving saxagliptin and metformin hydrochloride extended-release tablets, observe the patient closely for loss of blood glucose control. When such drugs are withdrawn from a patient receiving saxagliptin and metformin hydrochloride extended-release tablets, observe the patient closely for hypoglycemia.
Saxagliptin Morning and Evening Dosing
A 24-week monotherapy trial was conducted to assess a range of dosing regimens for saxagliptin. Treatment-naive patients with inadequately controlled diabetes (A1C ≥ 7% to ≤ 10%) underwent a 2-week, single-blind diet, exercise, and placebo lead-in period. A total of 365 patients were randomized to 2.5 mg every morning, 5 mg every morning, 2.5 mg with possible titration to 5 mg every morning, or 5 mg every evening of saxagliptin, or placebo. Patients who failed to meet specific glycemic goals during the trial were treated with metformin HCl rescue therapy added on to placebo or saxagliptin; the number of patients randomized per treatment group ranged from 71 to 74.
Treatment with either saxagliptin 5 mg every morning or 5 mg every evening provided significant improvements in A1C versus placebo (mean placebo-corrected reductions of -0.4% and -0.3%, respectively).
Coadministration of Saxagliptin with Metformin HCl Immediate-Release in Treatment-Naive Patients
A total of 1306 treatment-naive patients with type 2 diabetes mellitus participated in this 24-week, randomized, double-blind, active-controlled trial to evaluate the efficacy and safety of saxagliptin coadministered with metformin HCl immediate-release in patients with inadequate glycemic control (A1C ≥ 8% to ≤ 12%) on diet and exercise alone. Patients were required to be treatment-naive to be enrolled in this trial.
Patients who met eligibility criteria were enrolled in a single-blind, 1-week, dietary and exercise placebo lead-in period. Patients were randomized to one of four treatment arms: saxagliptin 5 mg + metformin HCl immediate-release 500 mg, saxagliptin 10 mg + metformin HCl immediate-release 500 mg, saxagliptin 10 mg + placebo, or metformin HCl immediate-release 500 mg + placebo (the maximum recommended approved saxagliptin dose is 5 mg daily; the 10 mg daily dose of saxagliptin does not provide greater efficacy than the 5 mg daily dose and the 10 mg saxagliptin dosage is not an approved dosage). Saxagliptin was dosed once daily. In the 3 treatment groups using metformin HCl immediate-release, the metformin HCl dose was up-titrated weekly in 500 mg per day increments, as tolerated, to a maximum of 2,000 mg per day based on FPG. Patients who failed to meet specific glycemic goals during this trial were treated with pioglitazone rescue as add-on therapy.
Coadministration of saxagliptin 5 mg plus metformin HCl immediate-release provided significant improvements in A1C, FPG, and PPG compared with placebo plus metformin HCl immediate-release (Table 7).
|
Efficacy Parameter |
Saxagliptin 5 mg + Metformin HCl N = 320 |
Placebo + Metformin HCl N = 328 |
|
Hemoglobin A1C (%) |
N = 306 |
N = 313 |
|
Baseline (mean) |
9.4 |
9.4 |
|
Change from baseline (adjusted mean Least squares mean adjusted for baseline value. )
|
-2.5 |
-2.0 |
|
Difference from placebo + metformin HCl (adjusted mean ) |
-0.5 p-value < 0.0001 compared to placebo + metformin HCl.
|
|
|
95% Confidence Interval |
(-0.7, -0.4) |
|
|
Percent of patients achieving A1C < 7% |
60%§ (185/307) |
41% (129/314) |
|
Fasting Plasma Glucose (mg/dL) |
N = 315 |
N = 320 |
|
Baseline (mean) |
199 |
199 |
|
Change from baseline (adjusted mean ) |
-60 |
-47 |
|
Difference from placebo + metformin HCl (adjusted mean ) |
-13 p-value < 0.05 compared to placebo + metformin HCl.
|
|
|
95% Confidence Interval |
(-19, -6) |
|
|
2-hour Postprandial Glucose (mg/dL) |
N = 146 |
N = 141 |
|
Baseline (mean) |
340 |
355 |
|
Change from baseline (adjusted mean ) |
-138 |
-97 |
|
Difference from placebo + metformin HCl (adjusted mean ) |
-41 |
|
|
95% Confidence Interval |
(-57, -25) |
Addition of Saxagliptin to Metformin HCl Immediate-Release
A total of 743 patients with type 2 diabetes mellitus participated in this 24-week, randomized, double-blind, placebo-controlled trial to evaluate the efficacy and safety of saxagliptin in combination with metformin HCl immediate-release in patients with inadequate glycemic control (A1C ≥ 7% and ≤ 10%) on metformin HCl alone. To qualify for enrollment, patients were required to be on a stable dose of metformin HCl (1,500-2,550 mg daily) for at least 8 weeks.
Patients who met eligibility criteria were enrolled in a single-blind, 2-week, dietary and exercise placebo lead-in period during which patients received metformin HCl immediate-release at their pre-trial dose, up to 2500 mg daily, for the duration of the trial. Following the lead-in period, eligible patients were randomized to 2.5 mg, 5 mg, or 10 mg of saxagliptin or placebo in addition to their current dose of open-label metformin HCl immediate-release (the maximum recommended approved saxagliptin dose is 5 mg daily; the 10 mg daily dose of saxagliptin does not provide greater efficacy than the 5 mg daily dose and the 10 mg dosage is not an approved dosage). Patients who failed to meet specific glycemic goals during the trial were treated with pioglitazone rescue therapy, added on to existing trial medications. Dose titrations of saxagliptin and metformin HCl immediate-release were not permitted.
Saxagliptin 2.5 mg and 5 mg add-on to metformin HCl immediate-release provided significant improvements in A1C, FPG, and PPG compared with placebo add-on to metformin HCl immediate-release (Table 8). Mean changes from baseline for A1C over time and at endpoint are shown in Figure 1. The proportion of patients who discontinued for lack of glycemic control or who were rescued for meeting prespecified glycemic criteria was 15% in the saxagliptin 2.5 mg add-on to metformin HCl immediate-release group, 13% in the saxagliptin 5 mg add-on to metformin HCl immediate-release group, and 27% in the placebo add-on to metformin HCl immediate-release group.
|
Efficacy Parameter |
Saxagliptin 2.5 mg + Metformin HCl N = 192 |
Saxagliptin 5 mg + Metformin HCl N = 191 |
Placebo + Metformin HCl N = 179 |
|
Hemoglobin A1C (%) |
N = 186 |
N = 186 |
N = 175 |
|
Baseline (mean) |
8.1 |
8.1 |
8.1 |
|
Change from baseline (adjusted mean Least squares mean adjusted for baseline value. )
|
-0.6 |
-0.7 |
+0.1 |
|
Difference from placebo (adjusted mean ) |
-0.7 p-value < 0.0001 compared to placebo + metformin HCl.
|
-0.8 |
|
|
95% Confidence Interval |
(-0.9, -0.5) |
(-1.0, -0.6) |
|
|
Percent of patients achieving A1C < 7% |
37% p-value < 0.05 compared to placebo + metformin HCl. (69/186)
|
44% (81/186) |
17% (29/175) |
|
Fasting Plasma Glucose (mg/dL) |
N = 188 |
N = 187 |
N = 176 |
|
Baseline (mean) |
174 |
179 |
175 |
|
Change from baseline (adjusted mean ) |
-14 |
-22 |
+1 |
|
Difference from placebo (adjusted mean ) |
-16 |
-23 |
|
|
95% Confidence Interval |
(-23, -9) |
(-30, -16) |
|
|
2-hour Postprandial Glucose (mg/dL) |
N = 155 |
N = 155 |
N = 135 |
|
Baseline (mean) |
294 |
296 |
295 |
|
Change from baseline (adjusted mean ) |
-62 |
-58 |
-18 |
|
Difference from placebo (adjusted mean ) |
-44 |
-40 |
|
|
95% Confidence Interval |
(-60, -27) |
(-56, -24) |
Saxagliptin Add-On Combination Therapy with Metformin HCl Immediate-Release versus Glipizide Add-On Combination Therapy with Metformin HCl Immediate-Release
In this 52-week, active-controlled trial, a total of 858 patients with type 2 diabetes mellitus and inadequate glycemic control (A1C > 6.5% and ≤ 10%) on metformin HCl immediate-release alone were randomized to double-blind add-on therapy with saxagliptin or glipizide. Patients were required to be on a stable dose of metformin HCl immediate-release (at least 1,500 mg daily) for at least 8 weeks prior to enrollment.
Patients who met eligibility criteria were enrolled in a single-blind, 2-week, dietary and exercise placebo lead-in period during which patients received metformin HCl immediate-release (1,500-3,000 mg based on their pre-trial dose). Following the lead-in period, eligible patients were randomized to 5 mg of saxagliptin or 5 mg of glipizide in addition to their current dose of open-label metformin HCl immediate-release. Patients in the glipizide plus metformin HCl immediate-release group underwent blinded titration of the glipizide dose during the first 18 weeks of the trial up to a maximum glipizide dose of 20 mg per day. Titration was based on a goal FPG ≤ 110 mg/dL or the highest tolerable glipizide dose. Fifty percent (50%) of the glipizide-treated patients were titrated to the 20-mg daily dose; 21% of the glipizide-treated patients had a final daily glipizide dose of 5 mg or less. The mean final daily dose of glipizide was 15 mg.
After 52 weeks of treatment, saxagliptin and glipizide resulted in similar mean reductions from baseline in A1C when added to metformin HCl immediate-release therapy (Table 9). This conclusion may be limited to patients with baseline A1C comparable to those in the trial (91% of patients had baseline A1C < 9%).
From a baseline mean body weight of 89 kg, there was a statistically significant mean reduction of 1.1 kg in patients treated with saxagliptin compared to a mean weight gain of 1.1 kg in patients treated with glipizide (p < 0.0001).
|
Efficacy Parameter |
Saxagliptin 5 mg + Metformin HCl N = 428 |
Titrated Glipizide + Metformin HCl N = 430 |
|
Hemoglobin A1C (%) |
N = 423 |
N = 423 |
|
Baseline (mean) |
7.7 |
7.6 |
|
Change from baseline (adjusted mean) |
-0.6 |
-0.7 |
|
Difference from glipizide + metformin HCl (adjusted mean Least squares mean adjusted for baseline value. )
|
0.1 |
|
|
95% Confidence Interval |
(-0.02, 0.2) Saxagliptin + metformin HCl is considered non-inferior to glipizide + metformin HCl because the upper limit of this confidence interval is less than the prespecified non-inferiority margin of 0.35%.
|
|
|
Fasting Plasma Glucose (mg/dL) |
N = 420 |
N = 420 |
|
Baseline (mean) |
162 |
161 |
|
Change from baseline (adjusted mean ) |
-9 |
-16 |
|
Difference from glipizide + metformin HCl (adjusted mean ) |
6 |
|
|
95% Confidence Interval |
(2, 11) Significance not tested.
|
Saxagliptin Add-On Combination Therapy with Insulin (with or without Metformin HCl Immediate-Release)
A total of 455 patients with type 2 diabetes mellitus participated in this 24-week, randomized, double-blind, placebo-controlled trial to evaluate the efficacy and safety of saxagliptin in combination with insulin in patients with inadequate glycemic control (A1C ≥ 7.5% and ≤ 11%) on insulin alone (N = 141) or on insulin in combination with a stable dose of metformin HCl immediate-release (N = 314). Patients were required to be on a stable dose of insulin (≥ 30 units to ≤ 150 units daily) with ≤ 20% variation in total daily dose for ≥ 8 weeks prior to screening. Patients entered the trial on intermediate- or long-acting (basal) insulin or premixed insulin. Patients using short-acting insulins were excluded unless the short-acting insulin was administered as part of a premixed insulin.
Patients who met eligibility criteria were enrolled in a single-blind, four-week, dietary and exercise placebo lead-in period during which patients received insulin (and metformin HCl immediate-release if applicable) at their pretrial dose(s). Following the lead-in period, eligible patients were randomized to add-on therapy with either saxagliptin 5 mg or placebo. Doses of the antidiabetic therapies were to remain stable but patients were rescued and allowed to adjust the insulin regimen if specific glycemic goals were not met or if the investigator learned that the patient had self-increased the insulin dose by > 20%. Data after rescue were excluded from the primary efficacy analyses.
Add-on therapy with saxagliptin 5 mg provided significant improvements from baseline to Week 24 in A1C and PPG compared with add-on placebo (Table 10). Similar mean reductions in A1C versus placebo were observed for patients using saxagliptin 5 mg add-on to insulin alone and saxagliptin 5 mg add-on to insulin in combination with metformin HCl immediate-release (-0.4% and -0.4%, respectively). The percentage of patients who discontinued for lack of glycemic control or who were rescued was 23% in the saxagliptin group and 32% in the placebo group.
The mean daily insulin dose at baseline was 53 units in patients treated with saxagliptin 5 mg and 55 units in patients treated with placebo. The mean change from baseline in daily dose of insulin was 2 units for the saxagliptin 5 mg group and 5 units for the placebo group.
|
Efficacy Parameter |
Saxagliptin 5 mg + Insulin (+/- Metformin HCl) N = 304 |
Placebo + Insulin (+/- Metformin HCl) N = 151 |
|
Hemoglobin A1C (%) |
N = 300 |
N = 149 |
|
Baseline (mean) |
8.7 |
8.7 |
|
Change from baseline (adjusted mean Least squares mean adjusted for baseline value and metformin HCl use at baseline. )
|
-0.7 |
-0.3 |
|
Difference from placebo (adjusted mean ) |
-0.4 p-value < 0.0001 compared to placebo + insulin.
|
|
|
95% Confidence Interval |
(-0.6, -0.2) |
|
|
2-hour Postprandial Glucose (mg/dL) |
N = 262 |
N = 129 |
|
Baseline (mean) |
251 |
255 |
|
Change from baseline (adjusted mean ) |
-27 |
-4 |
|
Difference from placebo (adjusted mean ) |
-23 p-value < 0.05 compared to placebo + insulin.
|
|
|
95% Confidence Interval |
(-37, -9) |
The change in fasting plasma glucose from baseline to Week 24 was also tested, but was not statistically significant. The percent of patients achieving an A1C < 7% was 17% (52/300) with saxagliptin in combination with insulin compared to 7% (10/149) with placebo. Significance was not tested.
Saxagliptin Add-On Combination Therapy with Metformin HCl plus Sulfonylurea
A total of 257 patients with type 2 diabetes mellitus participated in this 24-week, randomized, double-blind, placebo-controlled trial to evaluate the efficacy and safety of saxagliptin in combination with metformin HCl plus a sulfonylurea in patients with inadequate glycemic control (A1C ≥ 7% and ≤ 10%). Patients were to be on a stable combined dose of metformin HCl extended-release or immediate-release (at maximum tolerated dose, with minimum dose for enrollment being 1,500 mg) and a sulfonylurea (at maximum tolerated dose, with minimum dose for enrollment being ≥ 50% of the maximum recommended dose) for ≥ 8 weeks prior to enrollment.
Patients who met eligibility criteria were entered in a 2-week enrollment period to allow assessment of inclusion/exclusion criteria. Following the 2-week enrollment period, eligible patients were randomized to either double-blind saxagliptin (5 mg once daily) or double-blind matching placebo for 24 weeks. During the 24-week double-blind treatment period, patients were to receive metformin HCl and a sulfonylurea at the same constant dose ascertained during enrollment. Sulfonylurea dose could be down titrated once in the case of a major hypoglycemic event or recurring minor hypoglycemic events. In the absence of hypoglycemia, titration (up or down) of trial medication during the treatment period was prohibited.
Saxagliptin in combination with metformin HCl plus a sulfonylurea provided significant improvements in A1C and PPG compared with placebo in combination with metformin HCl plus a sulfonylurea (Table 11). The percentage of patients who discontinued for lack of glycemic control was 6% in the saxagliptin group and 5% in the placebo group.
|
Efficacy Parameter |
Saxagliptin 5 mg + Metformin HCl plus Sulfonylurea N = 129 |
Placebo + Metformin HCl plus Sulfonylurea N = 128 |
|
Hemoglobin A1C (%) |
N = 127 |
N = 127 |
|
Baseline (mean) |
8.4 |
8.2 |
|
Change from baseline (adjusted mean Least squares mean adjusted for baseline value. )
|
-0.7 |
-0.1 |
|
Difference from placebo (adjusted mean ) |
-0.7 p-value < 0.0001 compared to placebo + metformin HCl plus sulfonylurea.
|
|
|
95% Confidence Interval |
(-0.9, -0.5) |
|
|
2-hour Postprandial Glucose (mg/dL) |
N = 115 |
N = 113 |
|
Baseline (mean) |
268 |
262 |
|
Change from baseline (adjusted mean ) |
-12 |
5 |
|
Difference from placebo (adjusted mean ) |
-17 p-value < 0.05 compared to placebo + metformin HCl plus sulfonylurea.
|
|
|
95% Confidence Interval |
(-32, -2) |
The change in fasting plasma glucose from baseline to Week 24 was also tested, but was not statistically significant. The percent of patients achieving an A1C < 7% was 31% (39/127) with saxagliptin in combination with metformin HCl plus a sulfonylurea compared to 9% (12/127) with placebo. Significance was not tested.
Saxagliptin Add-on Combination Therapy with Metformin HCl plus an SGLT2 Inhibitor
A total of 315 patients with type 2 diabetes mellitus participated in this 24-week randomized, double-blind, placebo-controlled trial to evaluate the efficacy and safety of saxagliptin added to dapagliflozin (an SGLT2 inhibitor) and metformin HCl in patients with a baseline of HbA1c ≥ 7% to ≤ 10.5%. The mean age of these patients was 54.6 years, 1.6% were 75 years or older and 52.7% were female. The population was 87.9% White, 6.3% Black or African American, 4.1% Asian, and 1.6% other races. At baseline the population had diabetes for an average of 7.7 years and a mean HbA1c of 7.9%. The mean eGFR at baseline was 93.4 mL/min/1.73 m2. Patients were required to be on a stable dose of metformin HCl (≥ 1,500 mg per day) for at least 8 weeks prior to enrollment. Eligible patients who completed the screening period entered the lead-in treatment period, which included 16 weeks of open-label metformin HCl and 10 mg dapagliflozin treatment. Following the lead-in period, eligible patients were randomized to saxagliptin 5 mg (N = 153) or placebo (N = 162).
The group treated with add-on saxagliptin had statistically significant greater reductions in HbA1c from baseline versus the group treated with placebo (see Table 12).
|
Saxagliptin 5 mg (N = 153) Number of randomized and treated patients.
|
Placebo (N = 162) |
|
|
In combination with Dapagliflozin and Metformin |
||
|
Hemoglobin A1C (%) Analysis of Covariance including all post-baseline data regardless of rescue or treatment discontinuation. Model estimates calculated using multiple imputation to model washout of the treatment effect using placebo data for all patients having missing Week 24 data.
|
||
|
Baseline (mean) |
8.0 |
7.9 |
|
Change from baseline (adjusted mean Least squares mean adjusted for baseline value. )
95% Confidence Interval |
-0.5 (-0.6, -0.4) |
-0.2 (-0.3, -0.1) |
|
Difference from placebo (adjusted mean) 95% Confidence Interval |
-0.4 p-value < 0.0001.
(-0.5, -0.2) |
The known proportion of patients achieving HbA1c < 7% at Week 24 was 35.3% in the saxagliptin-treated group compared to 23.1% in the placebo-treated group.
In Vivo Assessment of Drug Interactions
| ND = not determined; QD = once daily; q6h = every 6 hours; q12h = every 12 hours; BID = twice daily; LA = long acting. | |||||
|
Coadministered Drug |
Dosage of Coadministered Drug Single dose unless otherwise noted. The 10 mg saxagliptin dose is not an approved dosage.
|
Dosage of Saxagliptin |
Geometric Mean Ratio (ratio with/without coadministered drug) No Effect = 1.00 |
||
|
AUC AUC = AUC(INF) for drugs given as single dose and AUC = AUC(TAU) for drugs given in multiple doses.
|
Cmax |
||||
|
No dosing adjustments required for the following: |
|||||
|
Metformin |
1,000 mg |
100 mg |
saxagliptin |
0.98 |
0.79 |
|
5-hydroxy saxagliptin |
0.99 |
0.88 |
|||
|
Glyburide |
5 mg |
10 mg |
saxagliptin |
0.98 |
1.08 |
|
5-hydroxy saxagliptin |
ND |
ND |
|||
|
Pioglitazone Results exclude one patient.
|
45 mg QD for 10 days |
10 mg QD for 5 days |
saxagliptin |
1.11 |
1.11 |
|
5-hydroxy saxagliptin |
ND |
ND |
|||
|
Digoxin |
0.25 mg q6h first day followed by q12h second day followed by QD for 5 days |
10 mg QD for 7 days |
saxagliptin |
1.05 |
0.99 |
|
5-hydroxy saxagliptin |
1.06 |
1.02 |
|||
|
Dapagliflozin |
10 mg single dose |
5 mg single dose |
saxagliptin |
↓ 1% |
↓ 7% |
|
5-hydroxy saxagliptin |
↑ 9% |
↑ 6% |
|||
|
Simvastatin |
40 mg QD for 8 days |
10 mg QD for 4 days |
saxagliptin |
1.12 |
1.21 |
|
5-hydroxy saxagliptin |
1.02 |
1.08 |
|||
|
Diltiazem |
360 mg LA QD for 9 days |
10 mg |
saxagliptin |
2.09 |
1.63 |
|
5-hydroxy saxagliptin |
0.66 |
0.57 |
|||
|
Rifampin The plasma dipeptidyl peptidase-4 (DPP4) activity inhibition over a 24-hour dose interval was not affected by rifampin.
|
600 mg QD for 6 days |
5 mg |
saxagliptin |
0.24 |
0.47 |
|
5-hydroxy saxagliptin |
1.03 |
1.39 |
|||
|
Omeprazole |
40 mg QD for 5 days |
10 mg |
saxagliptin |
1.13 |
0.98 |
|
5-hydroxy saxagliptin |
ND |
ND |
|||
|
Aluminum hydroxide + magnesium hydroxide + simethicone |
aluminum hydroxide: 2400 mg magnesium hydroxide: 2400 mg simethicone: 240 mg |
10 mg |
saxagliptin |
0.97 |
0.74 |
|
5-hydroxy saxagliptin |
ND |
ND |
|||
|
Famotidine |
40 mg |
10 mg |
saxagliptin |
1.03 |
1.14 |
|
5-hydroxy saxagliptin |
ND |
ND |
|||
|
Limit saxagliptin and metformin hydrochloride extended-release tablet dose to 2.5 mg/1,000 mg once daily when coadministered with strong CYP3A4/5 inhibitors [see Drug Interactions (7.1) and Dosage and Administration (2.2) ]: |
|||||
|
Ketoconazole |
200 mg BID for 9 days |
100 mg |
saxagliptin |
2.45 |
1.62 |
|
5-hydroxy saxagliptin |
0.12 |
0.05 |
|||
|
Ketoconazole |
200 mg BID for 7 days |
20 mg |
saxagliptin |
3.67 |
2.44 |
|
5-hydroxy saxagliptin |
ND |
ND |
| ND = not determined; QD = once daily; q6h = every 6 hours; q12h = every 12 hours; BID = twice daily; LA = long acting. | |||||
|
Coadministered Drug |
Dosage of Coadministered Drug Single dose unless otherwise noted. The 10 mg saxagliptin dose is not an approved dosage.
|
Dosage of Saxagliptin |
Geometric Mean Ratio (ratio with/without saxagliptin) No Effect = 1.00 |
||
|
AUC AUC = AUC(INF) for drugs given as single dose and AUC = AUC(TAU) for drugs given in multiple doses.
|
Cmax |
||||
|
No dosing adjustments required for the following: |
|||||
|
Metformin |
1,000 mg |
100 mg |
metformin |
1.20 |
1.09 |
|
Glyburide |
5 mg |
10 mg |
glyburide |
1.06 |
1.16 |
|
Pioglitazone Results include all patients.
|
45 mg QD for 10 days |
10 mg QD for 5 days |
pioglitazone |
1.08 |
1.14 |
|
hydroxy-pioglitazone |
ND |
ND |
|||
|
Digoxin |
0.25 mg q6h first day followed by q12h second day followed by QD for 5 days |
10 mg QD for 7 days |
digoxin |
1.06 |
1.09 |
|
Simvastatin |
40 mg QD for 8 days |
10 mg QD for 4 days |
simvastatin |
1.04 |
0.88 |
|
simvastatin acid |
1.16 |
1.00 |
|||
|
Diltiazem |
360 mg LA QD for 9 days |
10 mg |
diltiazem |
1.10 |
1.16 |
|
Ketoconazole |
200 mg BID for 9 days |
100 mg |
ketoconazole |
0.87 |
0.84 |
|
Ethinyl estradiol and norgestimate |
ethinyl estradiol 0.035 mg and norgestimate 0.250 mg for 21 days |
5 mg QD for 21 days |
ethinyl estradiol |
1.07 |
0.98 |
|
norelgestromin |
1.10 |
1.09 |
|||
|
norgestrel |
1.13 |
1.17 |
|
Coadministered Drug |
Dose of Coadministered Drug All metformin and coadministered drugs were given as single doses.
|
Dose of Metformin |
Geometric Mean Ratio (ratio with/without coadministered drug) No Effect = 1.00 |
||
|
AUC AUC = AUC(INF).
|
Cmax |
||||
|
No dosing adjustments required for the following: |
|||||
|
Glyburide |
5 mg |
850 mg |
metformin |
0.91 Ratio of arithmetic means.
|
0.93 |
|
Furosemide |
40 mg |
850 mg |
metformin |
1.09 |
1.22 |
|
Nifedipine |
10 mg |
850 mg |
metformin |
1.16 |
1.21 |
|
Propranolol |
40 mg |
850 mg |
metformin |
0.90 |
0.94 |
|
Ibuprofen |
400 mg |
850 mg |
metformin |
1.05 |
1.07 |
|
Drugs that are eliminated by renal tubular secretion may increase the accumulation of metformin [see Drug Interactions (7.3) ]. |
|||||
|
Cimetidine |
400 mg |
850 mg |
metformin |
1.40 |
1.61 |
|
Coadministered |
Dose of All metformin and coadministered drugs were given as single doses.
|
Dose of |
Geometric Mean Ratio (ratio with/without metformin) No Effect = 1.00 |
||
|
AUC AUC = AUC(INF) unless otherwise noted.
|
Cmax |
||||
|
No dosing adjustments required for the following: |
|||||
|
Glyburide |
5 mg |
850 mg |
glyburide |
0.78 Ratio of arithmetic means, p-value of difference < 0.05.
|
0.63 |
|
Furosemide |
40 mg |
850 mg |
furosemide |
0.87 |
0.69 |
|
Nifedipine |
10 mg |
850 mg |
nifedipine |
1.10 AUC(0-24 hr) reported.
|
1.08 |
|
Propranolol |
40 mg |
850 mg |
propranolol |
1.01 |
1.02 |
|
Ibuprofen |
400 mg |
850 mg |
ibuprofen |
0.97 Ratio of arithmetic means.
|
1.01 |
|
Cimetidine |
400 mg |
850 mg |
cimetidine |
0.95 |
1.01 |
Principal Display Panel – 5 Mg/1000mg
NDC 0378-8177-93
Saxagliptin and
Metformin HCl
Extended-Release
Tablets
5 mg/1000 mg
PHARMACIST: Dispense the accompanying
Medication Guide to each patient.
Do not crush, cut, or chew tablets.
Tablets must be swallowed whole.
Rx only 30 Tablets
Each film-coated extended-release tablet
contains 6.149 mg saxagliptin hydrochloride
dihydrate equivalent to 5 mg saxagliptin and
1000 mg metformin hydrochloride, USP.
Usual Dosage: See accompanying
prescribing information.
Keep this and all medication out of
the reach of children.
Store at 20° to 25°C (68° to 77°F).
[See USP Controlled Room
Temperature.]
Manufactured for:
Mylan Pharmaceuticals Inc.
Morgantown, WV 26505 U.S.A.
Made in India
Mylan.com
RMXA8177H1
Dispense in a tight, light-resistant
container as defined in the USP
using a child-resistant closure.
Keep container tightly closed.
Code No.: MH/DRUGS/AD/089
Principal Display Panel – 5 Mg/500 Mg
NDC 0378-8175-93
Saxagliptin and
Metformin HCl
Extended-Release
Tablets
5 mg/500 mg
PHARMACIST: Dispense the accompanying
Medication Guide to each patient.
Do not crush, cut, or chew tablets.
Tablets must be swallowed whole.
Rx only 30 Tablets
Each film-coated extended-release tablet
contains 6.149 mg saxagliptin hydrochloride
dihydrate equivalent to 5 mg saxagliptin and
500 mg metformin hydrochloride, USP.
Usual Dosage: See accompanying
prescribing information.
Keep this and all medication out of
the reach of children.
Store at 20° to 25°C (68° to 77°F). [See
USP Controlled Room Temperature.]
Manufactured for:
Mylan Pharmaceuticals Inc.
Morgantown, WV 26505 U.S.A.
Made in India
Mylan.com
RMXA8175H1
Dispense in a tight, light-resistant
container as defined in the USP
using a child-resistant closure.
Keep container tightly closed.
Code No.: MH/DRUGS/AD/089
In Vitro Assessment of Drug Interactions
In in vitro studies, saxagliptin and its active metabolite did not inhibit CYP1A2, 2A6, 2B6, 2C9, 2C19, 2D6, 2E1, or 3A4, or induce CYP1A2, 2B6, 2C9, or 3A4. Therefore, saxagliptin is not expected to alter the metabolic clearance of coadministered drugs that are metabolized by these enzymes. Saxagliptin is a P-glycoprotein (P-gp) substrate, but is not a significant inhibitor or inducer of P-gp.
2.1 Recommended Dosage and Administration
Individualize the starting dosage of saxagliptin and metformin hydrochloride extended-release tablets based on the patient’s current regimen and the available strengths of saxagliptin and metformin hydrochloride extended-release tablets [see Dosage Forms and Strengths (3) ].
Administer saxagliptin and metformin hydrochloride extended-release tablets once daily with the evening meal, with gradual dose titration to reduce the gastrointestinal side effects associated with metformin HCl [see Adverse Reactions (6.1) ].
The recommended starting dosage of saxagliptin and metformin hydrochloride extended-release tablets in patients who need 5 mg of saxagliptin and who are not currently treated with metformin HCl is one saxagliptin and metformin hydrochloride extended-release tablet containing 5 mg saxagliptin and 500 mg metformin HCl extended-release once daily with gradual dose escalation to reduce the gastrointestinal side effects due to metformin HCl.
In patients treated with metformin HCl, the recommended starting dosage of saxagliptin and metformin hydrochloride extended-release tablets should provide metformin HCl at the dose already being taken, or the nearest therapeutically appropriate dose. Following a switch from metformin HCl immediate-release to saxagliptin and metformin hydrochloride extended-release tablets, closely monitor glycemic control and adjust the dosage accordingly.
Patients who need 2.5 mg saxagliptin in combination with metformin HCl extended-release may be treated with saxagliptin and metformin hydrochloride extended-release tablets 2.5 mg/1,000 mg. Patients who need 2.5 mg saxagliptin who are either metformin HCl naive or who require a dose of metformin HCl higher than 1,000 mg should use the individual components.
Gradually titrate the dosage of saxagliptin and metformin hydrochloride extended-release tablets, as needed, after assessing therapeutic response and tolerability, up to a maximum recommended dosage of saxagliptin and metformin hydrochloride extended-release tablets (5 mg for saxagliptin and 2,000 mg for metformin HCl extended-release orally once daily).
Inform patients that saxagliptin and metformin hydrochloride extended-release tablets must be swallowed whole and never crushed, cut, or chewed. Occasionally, the inactive ingredients of saxagliptin and metformin hydrochloride extended-release tablets will be eliminated in the feces as a soft, hydrated mass that may resemble the original tablet.
If a dose is missed, advise patients not to take an extra dose. Resume treatment with the next dose.
7.1 Strong Inhibitors of Cyp3a4/5 Enzymes
Ketoconazole significantly increased saxagliptin exposure. Similar significant increases in plasma concentrations of saxagliptin are anticipated with other strong CYP3A4/5 inhibitors (e.g., atazanavir, clarithromycin, indinavir, itraconazole, nefazodone, nelfinavir, ritonavir, saquinavir, and telithromycin). The dose of saxagliptin and metformin hydrochloride extended-release tablets should be limited to 2.5 mg of saxagliptin when coadministered with a strong CYP3A4/5 inhibitor [see Dosage and Administration (2.3) and Clinical Pharmacology (12.3) ].
7.3 Drugs That Reduce Metformin Clearance
Concomitant use of drugs that interfere with common renal tubular transport systems involved in the renal elimination of metformin (e.g., organic cationic transporter-2 [OCT2]/multidrug and toxin extrusion [MATE] inhibitors such as ranolazine, vandetanib, dolutegravir, and cimetidine) could increase systemic exposure to metformin and may increase the risk for lactic acidosis [see Clinical Pharmacology (12.3) ]. Consider the benefits and risks of concomitant use.
Principal Display Panel – 2.5 Mg/1000 Mg
NDC 0378-8176-91
Saxagliptin and
Metformin HCl
Extended-Release
Tablets
2.5 mg/1000 mg
PHARMACIST: Dispense the accompanying
Medication Guide to each patient.
Do not crush, cut, or chew tablets.
Tablets must be swallowed whole.
Rx only 60 Tablets
Each film-coated extended-release tablet
contains 3.075 mg saxagliptin hydrochloride
dihydrate equivalent to 2.5 mg saxagliptin and
1000 mg metformin hydrochloride, USP.
Usual Dosage: See accompanying
prescribing information.
Keep this and all medication out of
the reach of children.
Store at 20° to 25°C (68° to 77°F).
[See USP Controlled Room
Temperature.]
Manufactured for:
Mylan Pharmaceuticals Inc.
Morgantown, WV 26505 U.S.A.
Made in India
Mylan.com
RMXA8176D1
Dispense in a tight, light-resistant
container as defined in the USP
using a child-resistant closure.
Keep container tightly closed.
Code No.: MH/DRUGS/AD/089
Adverse Reactions With Concomitant Use With Insulin
In the add-on to insulin trial [see Clinical Studies (14.1) ], the incidence of adverse events, including serious adverse events and discontinuations due to adverse events, was similar between saxagliptin and placebo, except for confirmed hypoglycemia [see Adverse Reactions (6.1) ].
Disease Associated Maternal And/or Embryo/fetal Risk
Poorly controlled diabetes in pregnancy increases the maternal risk for diabetic ketoacidosis, preeclampsia, spontaneous abortions, preterm delivery, still birth and delivery complications. Poorly controlled diabetes increases the fetal risk for major birth defects, stillbirth, and macrosomia related morbidity.
2.4 Discontinuation for Iodinated Contrast Imaging Procedures
Discontinue saxagliptin and metformin hydrochloride extended-release tablets at the time of, or prior to, an iodinated contrast imaging procedure in patients with an eGFR less than 60 mL/min/1.73 m2; a history of liver disease, alcoholism or heart failure; or in any patient who will be administered intra-arterial iodinated contrast. Re-evaluate eGFR 48 hours after the imaging procedure; restart saxagliptin and metformin hydrochloride extended-release tablets if renal function is stable [see Warnings and Precautions (5.1) ].
Saxagliptin and Metformin Hydrochloride Extended Release Tablets
Elderly patients are more likely to have decreased renal function. Assess renal function more frequently in the elderly [see Warnings and Precautions (5.1) and Clinical Pharmacology (12.3) ].
2.2 Recommendations for Dosage and Administration in Renal Impairment
Assess renal function prior to initiation of saxagliptin and metformin hydrochloride extended-release tablets and then as clinically indicated [see Use in Specific Populations (8.6) ].
The recommended dosage of saxagliptin and metformin hydrochloride extended-release tablets in patients with an estimated glomerular filtration rate (eGFR) greater than or equal to 45 mL/minute/1.73 m2 is the same as the recommended dosage in patients with normal renal function [see Dosage and Administration (2.1) ].
In patients taking saxagliptin and metformin hydrochloride extended-release tablets whose eGFR later falls below 45 mL/minute/1.73 m2, assess the benefit risk of continuing therapy and limit dose of the saxagliptin component to 2.5 mg once daily.
Initiation of saxagliptin and metformin hydrochloride extended-release tablets in patients with an eGFR between 30 – 45 mL/minute/1.73 m2 is not recommended.
Saxagliptin and metformin hydrochloride extended-release tablets are contraindicated in patients with an eGFR below 30 mL/minute/1.73 m2.
Discontinue saxagliptin and metformin hydrochloride extended-release tablets if the patient’s eGFR later falls below 30 mL/minute/1.73 m2 [see Contraindications (4) and Warnings and Precautions (5.1) ].
2.3 Dosage Modifications With Concomitant Use of Strong Cyp3a4/5 Inhibitors
The maximum recommended dosage of saxagliptin and metformin hydrochloride extended-release tablets is 2.5 mg of saxagliptin and 1,000 mg of metformin HCl given orally once daily when used concomitantly with strong cytochrome P450 3A4/5 (CYP3A4/5) inhibitors (e.g., ketoconazole, atazanavir, clarithromycin, indinavir, itraconazole, nefazodone, nelfinavir, ritonavir, saquinavir, and telithromycin) [see Dosage and Administration (2.1) , Drug Interactions (7.1), and Clinical Pharmacology (12.3) ].
Adverse Reactions Associated With Saxagliptin Coadministered With Metformin Hcl Immediate Release in Treatment Naive Patients With Type 2 Diabetes Mellitus
Table 2 shows the adverse reactions reported (regardless of investigator assessment of causality) in ≥ 5% of patients participating in an additional 24-week, active-controlled trial of coadministered saxagliptin and metformin HCl in treatment-naive patients.
|
Number (%) of Patients |
||
|
Saxagliptin 5 mg + Metformin HCl Metformin HCl immediate-release was initiated at a starting dose of 500 mg daily and titrated up to a maximum of 2,000 mg daily.
N = 320 |
Placebo + Metformin HCl N = 328 |
|
|
Headache |
24 (7.5) |
17 (5.2) |
|
Nasopharyngitis |
22 (6.9) |
13 (4.0) |
In patients treated with the combination of saxagliptin and metformin HCl immediate-release, either as saxagliptin add-on to metformin HCl immediate-release therapy or as coadministration in treatment-naive patients, diarrhea was the only gastrointestinal-related event that occurred with an incidence ≥ 5% in any treatment group in both trials. In the saxagliptin add-on to metformin HCl immediate-release trial, the incidence of diarrhea was 9.9%, 5.8%, and 11.2% in the saxagliptin 2.5 mg, 5 mg, and placebo groups, respectively. When saxagliptin and metformin HCl immediate-release were coadministered in treatment-naive patients, the incidence of diarrhea was 6.9% in the saxagliptin 5 mg + metformin HCl immediate-release group and 7.3% in the placebo + metformin HCl immediate-release group.
Structured Label Content
Excretion
Saxagliptin
Saxagliptin is eliminated by both renal and hepatic pathways. Following a single 50 mg dose of 14C-saxagliptin, 24%, 36%, and 75% of the dose was excreted in the urine as saxagliptin, its active metabolite, and total radioactivity, respectively. The average renal clearance of saxagliptin (~ 230 mL/min) was greater than the average estimated glomerular filtration rate (~ 120 mL/min), suggesting some active renal excretion. A total of 22% of the administered radioactivity was recovered in feces representing the fraction of the saxagliptin dose excreted in bile and/or unabsorbed drug from the gastrointestinal tract. Following a single oral dose of saxagliptin 5 mg to healthy subjects, the mean plasma terminal half-life (t1/2) for saxagliptin and its active metabolite was 2.5 and 3.1 hours, respectively.
Metformin HCl
Renal clearance is approximately 3.5 times greater than creatinine clearance, which indicates that tubular secretion is the major route of metformin elimination. Following oral administration, approximately 90% of the absorbed drug is eliminated via the renal route within the first 24 hours, with a plasma elimination half-life of approximately 6.2 hours. In blood, the elimination half-life is approximately 17.6 hours, suggesting that the erythrocyte mass may be a compartment of distribution.
Human Data
Published clinical lactation studies report that metformin is present in human milk which resulted in infant doses approximately 0.11% to 1% of the maternal weight-adjusted dosage and a milk/plasma ratio ranging between 0.13 and 1. However, the studies were not designed to definitely establish the risk of use of metformin during lactation because of small sample size and limited adverse event data collected in infants.
Metabolism
Saxagliptin
The metabolism of saxagliptin is primarily mediated by cytochrome P450 3A4/5 (CYP3A4/5). The major metabolite of saxagliptin is also a DPP4 inhibitor, which is one-half as potent as saxagliptin. Therefore, strong CYP3A4/5 inhibitors and inducers will alter the pharmacokinetics of saxagliptin and its active metabolite [see Drug Interactions (7.1) ].
Metformin HCl
Intravenous single-dose studies in healthy subjects demonstrate that metformin is excreted unchanged in the urine and does not undergo hepatic metabolism (no metabolites have been identified in humans) or biliary excretion.
Metabolism studies with extended-release metformin tablets have not been conducted.
7.4 Alcohol
Alcohol is known to potentiate the effect of metformin on lactate metabolism. Warn patients against excessive alcohol intake while receiving saxagliptin and metformin hydrochloride extended-release tablets.
Animal Data
Saxagliptin
In embryo-fetal development studies, saxagliptin was administered to pregnant rats and rabbits during the period of organogenesis, corresponding to the first trimester of human pregnancy. No adverse developmental effects were observed in either species at exposures 1503- and 152-times the 5 mg clinical dose in rats and rabbits, respectively, based on AUC. Saxagliptin crosses the placenta into the fetus following dosing in pregnant rats.
In a prenatal and postnatal development study, no adverse developmental effects were observed in maternal rats administered saxagliptin from gestation day 6 through lactation day 21 at exposures up to 470-times the 5 mg clinical dose, based on AUC.
Metformin HCl
Metformin HCl did not cause adverse developmental effect when administered to pregnant Sprague Dawley rats and rabbits up to 600 mg/kg/day during the period of organogenesis. This represents an exposure of about 2- and 6-times a 2,000 mg clinical dose based on body surface area (mg/m2) for rats and rabbits, respectively.
Saxagliptin and Metformin
Saxagliptin and metformin coadministered to pregnant rats and rabbits during the period of organogenesis did not result in adverse developmental effects considered clinically relevant in either species. Doses tested in rats provided exposure up to 100- and 10-times clinical exposure, and doses tested in rabbits provided exposure up to 249- and 1-times clinical exposure relative to the clinical dose of 5 mg saxagliptin and 2,000 mg metformin. Minor skeletal abnormalities associated with maternal toxicity were observed in rats. In rabbits, coadministration was poorly tolerated in a subset of mothers (12 of 30), resulting in death, moribundity, or abortion. However, among surviving mothers with evaluable litters, maternal toxicity was limited to marginal reductions in body weight over the course of gestation days 21 to 29, associated with fetal body weight decrements of 7%, and a low incidence of delayed ossification of the fetal hyoid bone.
Saxagliptin
When saxagliptin was used in combination with insulin or an insulin secretagogue, the incidence of confirmed hypoglycemia was increased over that of placebo used in combination with insulin or an insulin secretagogue [see Adverse Reactions (6.1) ]. Therefore, a lower dosage of insulin or an insulin secretagogue may be required to reduce the risk of hypoglycemia when used in combination with saxagliptin and metformin hydrochloride extended-release tablets.
Hypoglycemia
In the saxagliptin clinical trials, adverse reactions of hypoglycemia were based on all reports of hypoglycemia. A concurrent glucose measurement was not required or was normal in some patients. Therefore, it is not possible to conclusively determine that all these reports reflect true hypoglycemia.
The incidence of reported hypoglycemia for saxagliptin 2.5 mg and saxagliptin 5 mg versus placebo given as monotherapy was 4% and 5.6% versus 4.1%, respectively. In the add-on to metformin HCl immediate-release trial, the incidence of reported hypoglycemia was 7.8% with saxagliptin 2.5 mg, 5.8% with saxagliptin 5 mg, and 5% with placebo. When saxagliptin and metformin HCl immediate-release were coadministered in treatment-naive patients, the incidence of reported hypoglycemia was 3.4% in patients given saxagliptin 5 mg + metformin HCl immediate-release and 4% in patients given placebo + metformin HCl immediate-release.
In the active-controlled trial comparing add-on therapy with saxagliptin 5 mg to glipizide in patients inadequately controlled on metformin HCl alone, the incidence of reported hypoglycemia was 3% (19 events in 13 patients) with saxagliptin 5 mg versus 36.3% (750 events in 156 patients) with glipizide. Confirmed symptomatic hypoglycemia (accompanying fingerstick blood glucose ≤ 50 mg/dL) was reported in none of the saxagliptin-treated patients and in 35 glipizide-treated patients (8.1%) (p < 0.0001).
In the saxagliptin add-on to insulin trial, the overall incidence of reported hypoglycemia was 18.4% for saxagliptin 5 mg and 19.9% for placebo. However, the incidence of confirmed symptomatic hypoglycemia (accompanying fingerstick blood glucose ≤ 50 mg/dL) was higher with saxagliptin 5 mg (5.3%) versus placebo (3.3%). Among the patients using insulin in combination with metformin HCl, the incidence of confirmed symptomatic hypoglycemia was 4.8% with saxagliptin versus 1.9% with placebo.
In the saxagliptin add-on to metformin HCl plus sulfonylurea trial, the overall incidence of reported hypoglycemia was 10.1% for saxagliptin 5 mg and 6.3% for placebo. Confirmed hypoglycemia was reported in 1.6% of the saxagliptin-treated patients and in none of the placebo-treated patients [see Warnings and Precautions (5.5) ].
Risk Summary
Limited available data with saxagliptin and metformin hydrochloride extended-release tablets or saxagliptin in pregnant women are not sufficient to determine a drug-associated risk for major birth defects and miscarriage. Published trials with metformin use during pregnancy have not reported a clear association with metformin and major birth defect or miscarriage risk [see Data ].
No adverse developmental effects independent of maternal toxicity were observed when saxagliptin and metformin were administered separately or in combination to pregnant rats and rabbits during the period of organogenesis [see Data ].
The estimated background risk of major birth defects is 6 to 10% in women with pre-gestational diabetes with an HbA1c greater than 7 and has been reported to be as high as 20 to 25% in women with an HbA1c greater than 10. The estimated background risk of miscarriage for the indicated population is unknown. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2 to 4% and 15 to 20%, respectively.
10 Overdosage (10 OVERDOSAGE)
Saxagliptin
In a controlled clinical trial, once-daily, orally administered saxagliptin in healthy subjects at doses up to 400 mg daily for 2 weeks (80-times the MRHD) had no dose-related clinical adverse reactions and no clinically meaningful effect on QTc interval or heart rate.
In the event of an overdose, initiate appropriate supportive treatment as dictated by the patient’s clinical status. Saxagliptin and its active metabolite are removed by hemodialysis (23% of dose over 4 hours). Contact the Poison Help line (1-800-222-1222) or a medical toxicologist for additional overdosage management recommendations.
Metformin HCl
Overdose of metformin HCl has occurred, including ingestion of amounts greater than 50 grams. Hypoglycemia was reported in approximately 10% of cases, but no causal association with metformin HCl has been established. Lactic acidosis has been reported in approximately 32% of metformin overdose cases [see Warnings and Precautions (5.1) ]. Metformin is dialyzable with a clearance of up to 170 mL/min under good hemodynamic conditions. Therefore, hemodialysis may be useful for removal of accumulated drug from patients in whom metformin overdosage is suspected.
Metformin Hcl (Metformin HCl)
Hypoglycemia does not occur in patients receiving metformin alone under usual circumstances of use, but could occur when caloric intake is deficient, when strenuous exercise is not compensated by caloric supplementation, or during concomitant use with other glucose-lowering agents (such as sulfonylureas and insulin) or ethanol. Elderly, debilitated, or malnourished patients and those with adrenal or pituitary insufficiency or alcohol intoxication are particularly susceptible to hypoglycemic effects. Hypoglycemia may be difficult to recognize in the elderly and in people who are taking beta-adrenergic blocking drugs.
Inform patients using these concomitant medications of the risk of hypoglycemia and educate them on the signs and symptoms of hypoglycemia.
11 Description (11 DESCRIPTION)
Saxagliptin and metformin hydrochloride extended-release tablets contain two oral antihyperglycemic medications used in the management of type 2 diabetes mellitus: saxagliptin and metformin HCl.
Saxagliptin
Saxagliptin is an orally active inhibitor of the dipeptidyl-peptidase-4 (DPP4) enzyme.
Saxagliptin hydrochloride dihydrate is described chemically as (1S,3S,5S)-2-[(2S)-Amino-(3-hydroxytricyclo[3.3.1.13,7]dec-1-yl)acetyl]-2-azabicyclo[3.1.0]hexane-3-carbonitrile hydrochloride dihydrate or (1S,3S,5S)-2-[(2S)-2-Amino-2-(3-hydroxyadamantan-1-yl)acetyl]-2-azabicyclo[3.1.0]hexane-3-carbonitrile hydrochloride dihydrate. The empirical formula is C18H25N3O2•HCl•2 H2O and the molecular weight is 387.92. The structural formula is:
Saxagliptin hydrochloride dihydrate is a white to off white, non-hygroscopic, crystalline powder. It is sparingly soluble in water at 24°C ± 3°C, slightly soluble in ethyl acetate, and soluble in methanol, ethanol, isopropyl alcohol, acetonitrile, acetone, and polyethylene glycol 400 (PEG 400).
Metformin HCl
Metformin hydrochloride, USP (1,1-Dimethylbiguanide monohydrochloride) is a white, crystalline powder with a molecular formula of C4H11N5•HCl and a molecular weight of 165.62. Metformin HCl is freely soluble in water, slightly soluble in alcohol, and is practically insoluble in acetone, ether, and chloroform. The pKa of metformin is 12.4. The pH of a 1% aqueous solution of metformin HCl is 6.36. The structural formula is:
Saxagliptin and Metformin Hydrochloride Extended-Release Tablets
Saxagliptin and metformin hydrochloride extended-release tablets are available for oral administration as tablets containing either 6.149 mg saxagliptin hydrochloride dihydrate equivalent to 5 mg saxagliptin and 500 mg metformin hydrochloride (saxagliptin and metformin hydrochloride extended-release tablets 5 mg/500 mg), or 6.149 mg saxagliptin hydrochloride dihydrate equivalent to 5 mg saxagliptin and 1000 mg metformin hydrochloride (saxagliptin and metformin hydrochloride extended-release tablets 5 mg/1000 mg), or 3.075 mg saxagliptin hydrochloride dihydrate equivalent to 2.5 mg saxagliptin and 1000 mg metformin hydrochloride (saxagliptin and metformin hydrochloride extended-release tablets 2.5 mg/1000 mg). Each film-coated tablet of saxagliptin and metformin hydrochloride extended-release contains the following inactive ingredients: carbomer homopolymer type A, colloidal silicon dioxide, hypromellose, magnesium stearate, polyethylene glycol, polyvinyl alcohol, povidone, red iron oxide, stearic acid, talc and titanium dioxide. The 2.5 mg/1000 mg tablets also contain yellow iron oxide. In addition, the black imprinting ink contains ammonium hydroxide, black iron oxide, propylene glycol and shellac glaze.
The biologically inert components of the tablet may occasionally remain intact during gastrointestinal transit and will be eliminated in the feces as a soft, hydrated mass.
5.4 Vitamin B12
In controlled clinical trials of metformin of 29-week duration, a decrease to subnormal levels of previously normal serum vitamin B12 levels, without clinical manifestations, was observed in approximately 7% of patients. Such decrease, possibly due to interference with B12 absorption from the B12-intrinsic factor complex, may be associated with anemia but appears to be rapidly reversible with discontinuation of metformin or vitamin B12 supplementation. Certain individuals (those with inadequate vitamin B12 or calcium intake or absorption) appear to be predisposed to developing subnormal vitamin B12 levels. Measure hematologic parameters on an annual basis and vitamin B12 at 2- to 3-year intervals in patients on saxagliptin and metformin hydrochloride extended-release tablets and manage any abnormalities [see Adverse Reactions (6.1) ].
Body Mass Index
Saxagliptin
No dosage adjustment is recommended based on body mass index (BMI) which was not identified as a significant covariate on the apparent clearance of saxagliptin or its active metabolite in the population pharmacokinetic analysis.
5.2 Pancreatitis
There have been post-marketing reports of acute pancreatitis in patients taking saxagliptin. In a cardiovascular outcomes trial enrolling participants with established atherosclerotic cardiovascular disease (ASCVD) or multiple risk factors for ASCVD (SAVOR trial), cases of definite acute pancreatitis were confirmed in 17 of 8240 (0.2%) patients receiving saxagliptin compared to 9 of 8173 (0.1%) receiving placebo. Pre-existing risk factors for pancreatitis were identified in 88% (15/17) of those patients receiving saxagliptin and in 100% (9/9) of those patients receiving placebo.
After initiation of saxagliptin and metformin hydrochloride extended-release tablets, observe patients for signs and symptoms of pancreatitis. If pancreatitis is suspected, promptly discontinue saxagliptin and metformin hydrochloride extended-release tablets and initiate appropriate management. It is unknown whether patients with a history of pancreatitis are at increased risk for the development of pancreatitis while using saxagliptin and metformin hydrochloride extended-release tablets.
Laboratory Tests
Absolute Lymphocyte Counts
Saxagliptin
There was a dose-related mean decrease in absolute lymphocyte count observed with saxagliptin. From a baseline mean absolute lymphocyte count of approximately 2200 cells/microL, mean decreases of approximately 100 and 120 cells/microL with saxagliptin 5 mg and 10 mg, respectively, relative to placebo were observed at 24 weeks in a pooled analysis of five placebo-controlled clinical trials. Similar effects were observed when saxagliptin 5 mg and metformin HCl were coadministered in treatment-naive patients compared to placebo and metformin HCl. There was no difference observed for saxagliptin 2.5 mg relative to placebo. The proportion of patients who were reported to have a lymphocyte count ≤ 750 cells/microL was 0.5%, 1.5%, 1.4%, and 0.4% in the saxagliptin 2.5 mg, 5 mg, 10 mg, and placebo groups, respectively. In most patients, recurrence was not observed with repeated exposure to saxagliptin although some patients had recurrent decreases upon rechallenge that led to discontinuation of saxagliptin. The decreases in lymphocyte count were not associated with clinically relevant adverse reactions. The 10 mg saxagliptin dosage is not an approved dosage.
In the SAVOR trial mean decreases of approximately 84 cells/microL with saxagliptin relative to placebo was observed. The proportion of patients who experienced a decrease in lymphocyte counts to a count of ≤ 750 cells/microL was 1.6% (136/8280) and 1.0% (78/8212) on saxagliptin and placebo, respectively.
The clinical significance of this decrease in lymphocyte count relative to placebo is not known. When clinically indicated, such as in settings of unusual or prolonged infection, lymphocyte count should be measured. The effect of saxagliptin on lymphocyte counts in patients with lymphocyte abnormalities (e.g., human immunodeficiency virus) is unknown.
Vitamin B12 Concentrations
Metformin HCl
In metformin clinical trials of 29-week duration, a decrease to subnormal levels of previously normal serum vitamin B12 levels was observed in approximately 7% of patients.
Medication Guide
|
Saxagliptin and Metformin Hydrochloride Extended-Release Tablets, for oral use
(sax″ a glip′ tin met for′ min hye″ droe klor′ ide) |
|||
|
What is the most important information I should know about saxagliptin and metformin hydrochloride extended-release tablets?
Serious side effects can happen in people taking saxagliptin and metformin hydrochloride extended-release tablets, including:
Stop taking saxagliptin and metformin hydrochloride extended-release tablets and call your healthcare provider right away or go to the nearest hospital emergency room if you have any of the following symptoms of lactic acidosis:
Most people who have had lactic acidosis with metformin have other things that, combined with the metformin, led to the lactic acidosis. Tell your doctor if you have any of the following, because you have a higher chance for getting lactic acidosis with saxagliptin and metformin hydrochloride extended-release tablets if you:
The best way to keep from having a problem with lactic acidosis from metformin is to tell your doctor if you have any of the problems in the list above. Your doctor may decide to stop your saxagliptin and metformin hydrochloride extended-release tablets for a while if you have any of these things.
Saxagliptin and metformin hydrochloride extended-release tablets can have other serious side effects. See “What are the possible side effects of saxagliptin and metformin hydrochloride extended-release tablets?”.
|
|||
|
|
||
|
|||
|
|
||
|
|||
|
What are saxagliptin and metformin hydrochloride extended-release tablets?
It is not known if saxagliptin and metformin hydrochloride extended-release tablets are safe and effective in children. |
|||
|
Who should not take saxagliptin and metformin hydrochloride extended-release tablets?
Do not take saxagliptin and metformin hydrochloride extended-release tablets if you:
|
|||
|
|
||
|
|||
|
Before taking saxagliptin and metformin hydrochloride extended-release tablets, tell your healthcare provider about all of your medical conditions, including if you:
Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements.
Know the medicines you take. Keep a list of them to show your healthcare provider and pharmacist when you get a new medicine. Saxagliptin and metformin hydrochloride extended-release tablets may affect the way other medicines work, and other medicines may affect how saxagliptin and metformin hydrochloride extended-release tablets work.
Tell your healthcare provider if you will be starting or stopping certain other types of medicines, such as antibiotics, or medicines that treat fungus or HIV/AIDS, because your dose of saxagliptin and metformin hydrochloride extended-release tablets might need to be changed. |
|||
|
How should I take saxagliptin and metformin hydrochloride extended-release tablets?
|
|||
|
What are the possible side effects of saxagliptin and metformin hydrochloride extended-release tablets?
Saxagliptin and metformin hydrochloride extended-release tablets can cause serious side effects, including:
|
|||
|
|
|
|
|
|||
|
|
||
|
|||
Common side effects of saxagliptin and metformin hydrochloride extended-release tablets include: |
|||
|
|
|
|
|
Swelling or fluid retention in your hands, feet, or ankles (peripheral edema) may become worse in people who also take a thiazolidinedione to treat diabetes. If you do not know whether you are already on this type of medication, ask your healthcare provider.
These are not all of the possible side effects of saxagliptin and metformin hydrochloride extended-release tablets.
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
|
|||
|
How should I store saxagliptin and metformin hydrochloride extended-release tablets?
Store saxagliptin and metformin hydrochloride extended-release tablets between 20° to 25°C (68° to 77°F).
Keep saxagliptin and metformin hydrochloride extended-release tablets and all medicines out of the reach of children.
|
|||
|
General information about the use of saxagliptin and metformin hydrochloride extended-release tablets
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use saxagliptin and metformin hydrochloride extended-release tablets for a condition for which they were not prescribed. Do not give saxagliptin and metformin hydrochloride extended-release tablets to other people, even if they have the same symptoms you have. They may harm them.
You can ask your pharmacist or healthcare provider for information about saxagliptin and metformin hydrochloride extended-release tablets that is written for health professionals.
|
|||
|
What are the ingredients of saxagliptin and metformin hydrochloride extended-release tablets?
Active ingredients: saxagliptin and metformin HCl.
Inactive ingredients in each tablet: carbomer homopolymer type A, colloidal silicon dioxide, hypromellose, magnesium stearate, polyethylene glycol, polyvinyl alcohol, povidone, red iron oxide, stearic acid, talc and titanium dioxide. The 2.5 mg/1000 mg tablets also contain yellow iron oxide. In addition, the black imprinting ink contains ammonium hydroxide, black iron oxide, propylene glycol and shellac glaze.
|
|||
|
Manufactured for: Mylan Pharmaceuticals Inc., Morgantown, WV 26505 U.S.A. Manufactured by: Mylan Laboratories Limited, Hyderabad — 500 096, India
For more information, call Mylan at 1-877-446-3679 (1-877-4-INFO-RX).
The brands listed are trademarks of their respective owners.
|
This Medication Guide has been approved by the U.S. Food and Drug Administration.
Manufactured for:
Mylan Pharmaceuticals Inc.
Morgantown, WV 26505 U.S.A.
Manufactured by:
Mylan Laboratories Limited
Hyderabad — 500 096, India
75108500
Revised: 2/2025
MXA:SAXMET:R2m/MXA:MG:SAXMET:R2m
Renal Impairment
In the SAVOR trial, adverse reactions related to renal impairment, including laboratory changes (i.e., doubling of serum creatinine compared with baseline and serum creatinine > 6 mg/dL), were reported in 5.8% (483/8280) of saxagliptin-treated patients and 5.1% (422/8212) of placebo-treated patients. The most frequently reported adverse reactions included renal impairment (2.1% vs. 1.9%), acute renal failure (1.4% vs. 1.2%), and renal failure (0.8% vs. 0.9%), in the saxagliptin versus placebo groups, respectively. From baseline to the end of treatment, there was a mean decrease in eGFR of 2.5 mL/min/1.73 m2 for saxagliptin-treated patients and a mean decrease of 2.4 mL/min/1.73 m2 for placebo-treated patients. More patients randomized to saxagliptin (421/5227, 8.1%) compared to patients randomized to placebo (344/5073, 6.8%) had downward shifts in eGFR from > 50 mL/min/1.73 m2 (i.e., normal or mild renal impairment) to ≤ 50 mL/min/1.73 m2 (i.e., moderate or severe renal impairment). The proportions of patients with renal adverse reactions increased with worsening baseline renal function and increased age, regardless of treatment assignment.
5.3 Heart Failure
In a cardiovascular outcomes trial enrolling participants with established ASCVD or multiple risk factors for ASCVD (SAVOR trial), more patients randomized to saxagliptin (289/8280, 3.5%) were hospitalized for heart failure compared to patients randomized to placebo (228/8212, 2.8%). In a time-to-first-event analysis the risk of hospitalization for heart failure was higher in the saxagliptin group (estimated Hazard Ratio: 1.27; 95% CI: 1.07, 1.51). Patients with a prior history of heart failure and patients with renal impairment had a higher risk for hospitalization for heart failure, irrespective of treatment assignment.
Consider the risks and benefits of saxagliptin and metformin hydrochloride extended-release tablets prior to initiating treatment in patients at a higher risk for heart failure. Observe patients for signs and symptoms of heart failure during therapy. Advise patients of the characteristic symptoms of heart failure, and to immediately report such symptoms. If heart failure develops, evaluate and manage according to current standards of care and consider discontinuation of saxagliptin and metformin hydrochloride extended-release tablets.
8.4 Pediatric Use
The safety and effectiveness of saxagliptin and metformin hydrochloride extended-release tablets as an adjunct to diet and exercise to improve glycemic control in patients with type 2 diabetes mellitus have not been established in pediatric patients.
Effectiveness of saxagliptin was not demonstrated in a 26-week, placebo-controlled, double-blind randomized clinical trial with a 26-week safety extension (NCT03199053) in 164 pediatric patients aged 10 to 17 years with inadequately controlled type 2 diabetes mellitus.
Geriatric Patients
Saxagliptin
No dosage adjustment is recommended based on age alone. Elderly subjects (65-80 years) had 23% and 59% higher geometric mean Cmax and geometric mean AUC values, respectively, for saxagliptin than young subjects (18-40 years). Differences in active metabolite pharmacokinetics between elderly and young subjects generally reflected the differences observed in saxagliptin pharmacokinetics. The difference between the pharmacokinetics of saxagliptin and the active metabolite in young and elderly subjects is likely due to multiple factors including declining renal function and metabolic capacity with increasing age. Age was not identified as a significant covariate on the apparent clearance of saxagliptin and its active metabolite in the population pharmacokinetic analysis.
Metformin HCl
Limited data from controlled pharmacokinetic studies of metformin in healthy elderly subjects suggest that total plasma clearance of metformin is decreased, the half-life is prolonged, and Cmax is increased, compared to healthy young subjects. From these data, it appears that the change in metformin pharmacokinetics with aging is primarily accounted for by a change in renal function.
4 Contraindications (4 CONTRAINDICATIONS)
Saxagliptin and metformin hydrochloride extended-release tablets are contraindicated in patients with:
-
•Severe renal impairment (eGFR below 30 mL/min/1.73 m2).
-
•Acute or chronic metabolic acidosis, including diabetic ketoacidosis. Diabetic ketoacidosis should be treated with insulin.
-
•A history of a serious hypersensitivity reaction to saxagliptin, metformin HCl, or any of the ingredients in saxagliptin and metformin hydrochloride extended-release tablets. Reactions such as anaphylaxis, angioedema, or exfoliative skin conditions have been reported [see Warnings and Precautions (5.6) and Adverse Reactions (6.2) ].
5.1 Lactic Acidosis
There have been postmarketing cases of metformin-associated lactic acidosis, including fatal cases. These cases had a subtle onset and were accompanied by nonspecific symptoms such as malaise, myalgias, abdominal pain, respiratory distress, or increased somnolence; however, hypothermia, hypotension and resistant bradyarrhythmias have occurred with severe acidosis.
Metformin-associated lactic acidosis was characterized by elevated blood lactate concentrations (> 5 mmol/Liter), anion gap acidosis (without evidence of ketonuria or ketonemia), and an increased lactate: pyruvate ratio; metformin plasma levels generally > 5 mcg/mL. Metformin decreases liver uptake of lactate increasing lactate blood levels which may increase the risk of lactic acidosis, especially in patients at risk.
If metformin-associated lactic acidosis is suspected, general supportive measures should be instituted promptly in a hospital setting, along with immediate discontinuation of saxagliptin and metformin hydrochloride extended-release tablets.
In saxagliptin and metformin hydrochloride extended-release tablet-treated patients with a diagnosis or strong suspicion of lactic acidosis, prompt hemodialysis is recommended to correct the acidosis and remove accumulated metformin (metformin HCl is dialyzable, with a clearance of up to 170 mL/minute under good hemodynamic conditions). Hemodialysis has often resulted in reversal of symptoms and recovery.
Educate patients and their families about the symptoms of lactic acidosis and if these symptoms occur instruct them to discontinue saxagliptin and metformin hydrochloride extended-release tablets and report these symptoms to their health care provider.
For each of the known and possible risk factors for metformin-associated lactic acidosis, recommendations to reduce the risk of and manage metformin-associated lactic acidosis are provided below:
Renal Impairment: The post-marketing metformin-associated lactic acidosis cases primarily occurred in patients with significant renal impairment. The risk of metformin accumulation and metformin-associated lactic acidosis increases with the severity of renal impairment because metformin is substantially excreted by the kidney. Clinical recommendations based upon the patient’s renal function include [see Clinical Pharmacology (12.3) ]:
-
•Before initiating saxagliptin and metformin hydrochloride extended-release tablets, obtain an estimated glomerular filtration rate (eGFR).
-
•Saxagliptin and metformin hydrochloride extended-release tablets are contraindicated in patients with an eGFR less than 30 mL/minute/1.73 m2 [see Contraindications (4) ].
-
•Initiation of saxagliptin and metformin hydrochloride extended-release tablets is not recommended in patients with eGFR between 30 and 45 mL/minute/1.73 m2.
-
•Obtain an eGFR at least annually in all patients taking saxagliptin and metformin hydrochloride extended-release tablets. In patients at increased risk for the development of renal impairment (e.g., the elderly), renal function should be assessed more frequently.
-
•In patients taking saxagliptin and metformin hydrochloride extended-release tablets whose eGFR later falls below 45 mL/minute/1.73 m2, assess the benefit and risk of continuing therapy.
Drug Interactions: The concomitant use of saxagliptin and metformin hydrochloride extended-release tablets with specific drugs may increase the risk of metformin-associated lactic acidosis: those that impair renal function, result in significant hemodynamic change, interfere with acid-base balance or increase metformin accumulation [see Drug Interactions (7) ]. Therefore, consider more frequent monitoring of patients.
Age 65 or Greater: The risk of metformin-associated lactic acidosis increases with the patient’s age because elderly patients have a greater likelihood of having hepatic, renal, or cardiac impairment than younger patients. Assess renal function more frequently in elderly patients [see Use in Specific Populations (8.5) ].
Radiological Studies with Contrast: Administration of intravascular iodinated contrast agents in metformin-treated patients has led to an acute decrease in renal function and the occurrence of lactic acidosis. Stop saxagliptin and metformin hydrochloride extended-release tablets at the time of, or prior to, an iodinated contrast imaging procedure in patients with an eGFR between 30 and 60 mL/min/1.73 m2; in patients with a history of hepatic impairment, alcoholism, or heart failure; or in patients who will be administered intra-arterial iodinated contrast. Re-evaluate eGFR 48 hours after the imaging procedure, and restart saxagliptin and metformin hydrochloride extended-release tablets if renal function is stable.
Surgery and Other Procedures: Withholding of food and fluids during surgical or other procedures may increase the risk for volume depletion, hypotension and renal impairment. Saxagliptin and metformin hydrochloride extended-release tablets should be temporarily discontinued while patients have restricted food and fluid intake.
Hypoxic States: Several of the post-marketing cases of metformin-associated lactic acidosis occurred in the setting of acute congestive heart failure (particularly when accompanied by hypoperfusion and hypoxemia). Cardiovascular collapse (shock), acute myocardial infarction, sepsis, and other conditions associated with hypoxemia have been associated with lactic acidosis and may also cause prerenal azotemia. When such events occur, discontinue saxagliptin and metformin hydrochloride extended-release tablets.
Excessive Alcohol Intake: Alcohol potentiates the effect of metformin on lactate metabolism and this may increase the risk of metformin-associated lactic acidosis. Warn patients against excessive alcohol intake while receiving saxagliptin and metformin hydrochloride extended-release tablets.
Hepatic Impairment: Patients with hepatic impairment have developed with cases of metformin-associated lactic acidosis. This may be due to impaired lactate clearance resulting in higher lactate blood levels. Therefore, avoid use of saxagliptin and metformin hydrochloride extended-release tablets in patients with clinical or laboratory evidence of hepatic disease.
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following serious adverse reactions are described below or elsewhere in the prescribing information:
-
•Lactic Acidosis [see Boxed Warning and Warnings and Precautions (5.1) ]
-
•Pancreatitis [see Warnings and Precautions (5.2) ]
-
•Heart Failure [see Warnings and Precautions (5.3) ]
-
•Vitamin B12 Concentrations [see Warnings and Precautions (5.4) ]
-
•Hypoglycemia with Concomitant Use of Insulin or Insulin Secretagogues [see Warnings and Precautions (5.5) ]
-
•Hypersensitivity Reactions [see Warnings and Precautions (5.6) ]
-
•Severe and disabling arthralgia [see Warnings and Precautions (5.7) ]
-
•Bullous pemphigoid [see Warnings and Precautions (5.8) ]
7 Drug Interactions (7 DRUG INTERACTIONS)
-
•Strong CYP3A4/5 inhibitors (e.g., ketoconazole): Coadministration with saxagliptin and metformin hydrochloride extended-release tablets significantly increases saxagliptin concentrations. Limit saxagliptin and metformin hydrochloride extended-release tablets dosage to 2.5 mg/1,000 mg once daily when coadministered with a strong CYP3A4/5 inhibitor. (2.3, 7.1)
-
•Carbonic anhydrase inhibitors: May increase risk of lactic acidosis. Consider more frequent monitoring. (7.2)
-
•Drugs that reduce metformin clearance: May increase risk of lactic acidosis. Consider benefits and risks of concomitant use. (7.3)
-
•See full prescribing information for additional drug interactions. (7)
1.1 Limitations of Use
Saxagliptin and metformin hydrochloride extended-release tablets are not recommended for the treatment of type 1 diabetes mellitus or diabetic ketoacidosis.
5.8 Bullous Pemphigoid
Postmarketing cases of bullous pemphigoid requiring hospitalization have been reported with DPP-4 inhibitor use. In reported cases, patients typically recovered with topical or systemic immunosuppressive treatment and discontinuation of the DPP-4 inhibitor. Tell patients to report development of blisters or erosions while receiving saxagliptin and metformin hydrochloride extended-release tablets. If bullous pemphigoid is suspected, saxagliptin and metformin hydrochloride extended-release tablets should be discontinued and referral to a dermatologist should be considered for diagnosis and appropriate treatment.
8.7 Hepatic Impairment
Use of metformin in patients with hepatic impairment has been associated with some cases of lactic acidosis. Saxagliptin and metformin hydrochloride extended-release tablets are not recommended in patients with hepatic impairment [see Warnings and Precautions (5.1) ].
1 Indications and Usage (1 INDICATIONS AND USAGE)
Saxagliptin and metformin hydrochloride extended-release tablets are indicated as an adjunct to diet and exercise to improve glycemic control in adults with type 2 diabetes mellitus [see Clinical Studies (14) ].
Racial Or Ethnic Groups (Racial or Ethnic Groups)
Saxagliptin
No dosage adjustment is recommended based on race. The population pharmacokinetic analysis compared the pharmacokinetics of saxagliptin and its active metabolite in 309 White subjects with 105 subjects of other races (consisting of six racial groups). No significant difference in the pharmacokinetics of saxagliptin and its active metabolite were detected between these two populations.
Metformin HCl
No studies of metformin pharmacokinetic parameters according to race have been performed. In controlled clinical studies of metformin in patients with type 2 diabetes mellitus, the antihyperglycemic effect was comparable in Whites (n = 249), Black or African American (n = 51), and Hispanic or Latino ethnicity (n = 24).
Drug Interaction Studies
Specific pharmacokinetic drug interaction studies with saxagliptin and metformin hydrochloride extended-release tablets have not been performed, although such studies have been conducted with the individual saxagliptin and metformin components.
Male and Female Patients
Saxagliptin
No dosage adjustment is recommended based on gender. There were no differences observed in saxagliptin pharmacokinetics between males and females. Compared to males, females had approximately 25% higher exposure values for the active metabolite than males, but this difference is unlikely to be of clinical relevance. Gender was not identified as a significant covariate on the apparent clearance of saxagliptin and its active metabolite in the population pharmacokinetic analysis.
Metformin HCl
Metformin pharmacokinetic parameters did not differ significantly between healthy subjects and patients with type 2 diabetes mellitus when analyzed according to gender (males = 19, females = 16). Similarly, in controlled clinical studies in patients with type 2 diabetes mellitus, the antihyperglycemic effect of metformin was comparable in males and females.
Warning: Lactic Acidosis (WARNING: LACTIC ACIDOSIS)
-
•Post-marketing cases of metformin-associated lactic acidosis have resulted in death, hypothermia, hypotension, and resistant bradyarrhythmias. The onset of metformin-associated lactic acidosis is often subtle, accompanied only by nonspecific symptoms such as malaise, myalgias, respiratory distress, somnolence, and abdominal pain. Metformin-associated lactic acidosis was characterized by elevated blood lactate levels (> 5 mmol/Liter), anion gap acidosis (without evidence of ketonuria or ketonemia), an increased lactate/pyruvate ratio; and metformin plasma levels generally > 5 mcg/mL [see Warnings and Precautions (5.1) ].
-
•Risk factors for metformin-associated lactic acidosis include renal impairment, concomitant use of certain drugs (e.g., carbonic anhydrase inhibitors such as topiramate), age 65 years old or greater, having a radiological study with contrast, surgery and other procedures, hypoxic states (e.g., acute congestive heart failure), excessive alcohol intake, and hepatic impairment.
-
•Steps to reduce the risk of and manage metformin-associated lactic acidosis in these high risk groups are provided in the full prescribing information [see Dosage and Administration (2.2), Contraindications (4), Warnings and Precautions (5.1), Drug Interactions (7), and Use in Specific Populations (8.6, 8.7) ].
-
•If metformin-associated lactic acidosis is suspected, immediately discontinue saxagliptin and metformin hydrochloride extended-release tablets and institute general supportive measures in a hospital setting. Prompt hemodialysis is recommended [see Warnings and Precautions (5.1) ].
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
-
•Pancreatitis: There have been postmarketing reports of acute pancreatitis. If pancreatitis is suspected, promptly discontinue saxagliptin and metformin hydrochloride extended-release tablets. (5.2)
-
•Heart Failure: Consider the risks and benefits of saxagliptin and metformin hydrochloride extended-release tablets in patients who have known risk factors for heart failure. Monitor patients for signs and symptoms. (5.3)
-
•Vitamin B12 Deficiency: Metformin may lower vitamin B12 levels. Measure hematological parameters annually. (5.4)
-
•Hypoglycemia with Concomitant Use of Insulin or Insulin Secretagogues: Consider a lower dosage of insulin or insulin secretagogue when used in combination with saxagliptin and metformin hydrochloride extended-release tablets. (5.5)
-
•Hypersensitivity-Related Events: There have been post-marketing reports of serious hypersensitivity reactions, such as anaphylaxis, angioedema, and exfoliative skin conditions in patients treated with saxagliptin. If hypersensitivity reactions occur, discontinue saxagliptin and metformin hydrochloride extended-release tablets, treat promptly, and monitor until signs and symptoms resolve. (5.6)
-
•Arthralgia: Severe and disabling arthralgia has been reported in patients taking DPP4 inhibitors. Consider as a possible cause for severe joint pain and discontinue drug if appropriate. (5.7)
-
•Bullous Pemphigoid: There have been postmarketing reports of bullous pemphigoid requiring hospitalization in patients taking DPP-4 inhibitors. Tell patients to report development of blisters or erosions. If bullous pemphigoid is suspected, discontinue saxagliptin and metformin hydrochloride extended-release tablets (5.8).
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
-
•Administer once daily with the evening meal. (2.1)
-
•Individualize the starting dosage based on the patient’s current regimen then adjust the dosage based on effectiveness and tolerability. (2.1)
-
•Do not exceed a daily dosage of 5 mg saxagliptin/2,000 mg metformin HCl extended-release. (2.1)
-
•Swallow whole. Never crush, cut, or chew. (2.1)
-
•Limit the saxagliptin dosage to 2.5 mg daily for patients also taking strong cytochrome P450 3A4/5 inhibitors (e.g., ketoconazole). (2.3, 7.1)
-
•Assess renal function prior to initiation of saxagliptin and metformin hydrochloride extended-release tablets and periodically thereafter. (2.2)
-
oDo not use in patients with eGFR below 30 mL/min/1.73 m2.
-
oInitiation is not recommended in patients with eGFR between 30 - 45 mL/min/1.73 m2.
-
oAssess risk/benefit of continuing if eGFR falls below 45 mL/min/1.73 m2.
-
oLimit the saxagliptin component to 2.5 mg daily if eGFR is less than 45 mL/min/1.73 m2.
-
oDiscontinue if eGFR falls below 30 mL/min/1.73 m2.
-
-
•Saxagliptin and metformin hydrochloride extended-release tablets may need to be discontinued at time of, or prior to, iodinated contrast imaging procedures. (2.4)
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Extended-Release Tablets:
-
•Saxagliptin and Metformin Hydrochloride Extended-Release Tablets, 5 mg/500 mg, are pink, film-coated, round, unscored tablets imprinted with SM3 over M in black ink on one side of the tablet and blank on the other side.
-
•Saxagliptin and Metformin Hydrochloride Extended-Release Tablets, 5 mg/1000 mg, are pink, film-coated, capsule shaped, unscored tablets imprinted with SM6 over M in black ink on one side of the tablet and blank on the other side.
-
•Saxagliptin and Metformin Hydrochloride Extended-Release Tablets, 2.5 mg/1000 mg, are light peach, film-coated, capsule shaped, unscored tablets imprinted with SM4 over M in black ink on one side of the tablet and blank on the other side.
6.2 Postmarketing Experience
Additional adverse reactions have been identified during post-approval use of saxagliptin and metformin hydrochloride extended-release tablets, saxagliptin, or metformin HCl. Because these reactions are reported voluntarily from a population of uncertain size, it is generally not possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Saxagliptin
-
•Gastrointestinal Disorders: Pancreatitis
-
•Immune System Disorders: Hypersensitivity reactions including anaphylaxis, angioedema, and exfoliative skin conditions
-
•Musculoskeletal and Connective Tissue Disorders: Rhabdomyolysis, severe and disabling arthralgia
-
•Skin and Subcutaneous Tissue Disorders: Bullous pemphigoid
Metformin HCl
-
•Hepatobiliary Disorders: Cholestatic, hepatocellular, and mixed hepatocellular liver injury
14.1 Glycemic Efficacy Trials
The effectiveness of saxagliptin and metformin hydrochloride extended-release tablets has been established in clinical trials of coadministration of oral saxagliptin and metformin HCl immediate-release tablets in adults with type 2 diabetes mellitus inadequately controlled on metformin HCl alone and in treatment-naive patients inadequately controlled on diet and exercise alone. In these two trials, treatment with saxagliptin dosed in the morning plus metformin HCl immediate-release tablets at all doses produced statistically significant improvements in A1C, fasting plasma glucose (FPG), and 2-hour postprandial glucose (PPG) following a standard oral glucose tolerance test (OGTT), compared to control. Reductions in A1C were seen across subgroups including gender, age, race, and baseline BMI.
In these two trials, decrease in body weight in the treatment groups given saxagliptin in combination with metformin HCl immediate-release was similar to that in the groups given metformin HCl immediate-release alone. Saxagliptin plus metformin HCl immediate-release was not associated with significant changes from baseline in fasting serum lipids compared to metformin HCl alone.
The coadministration of saxagliptin and metformin HCl immediate-release tablets has also been evaluated in an active-controlled trial comparing add-on therapy with saxagliptin to glipizide in 858 patients inadequately controlled on metformin HCl alone, in a placebo-controlled trial where a subgroup of 314 patients inadequately controlled on insulin plus metformin HCl received add-on therapy with saxagliptin or placebo, a trial comparing saxagliptin to placebo in 257 patients inadequately controlled on metformin HCl plus a sulfonylurea, and a trial comparing saxagliptin to placebo in 315 patients inadequately controlled on dapagliflozin and metformin HCl.
In a 24-week, double-blind, randomized trial, patients treated with metformin HCl immediate-release 500 mg twice daily for at least 8 weeks were randomized to continued treatment with metformin HCl immediate-release 500 mg twice daily or to metformin HCl extended-release either 1,000 mg once daily or 1,500 mg once daily. The mean change in A1C from baseline to Week 24 was 0.1% (95% confidence interval 0%, 0.3%) for the metformin HCl immediate-release treatment arm, 0.3% (95% confidence interval 0.1%, 0.4%) for the 1,000 mg metformin HCl extended-release treatment arm, and 0.1% (95% confidence interval 0%, 0.3%) for the 1,500 mg metformin HCl extended-release treatment arm. Results of this trial suggest that patients receiving metformin HCl immediate-release treatment may be safely switched to metformin HCl extended-release once daily at the same total daily dose, up to 2,000 mg once daily. Following a switch from metformin HCl immediate-release to metformin HCl extended-release, glycemic control should be closely monitored and dosage adjustments made accordingly.
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
5.6 Hypersensitivity Reactions
There have been post-marketing reports of serious hypersensitivity reactions in patients treated with saxagliptin. These reactions include anaphylaxis, angioedema, and exfoliative skin conditions. Onset of these reactions occurred within the first 3 months after initiation of treatment with saxagliptin, with some reports occurring after the first dose. If a serious hypersensitivity reaction is suspected, discontinue saxagliptin and metformin hydrochloride extended-release tablets, assess for other potential causes for the event, and institute alternative treatment for diabetes [see Adverse Reactions (6.2) ].
Use caution in a patient with a history of angioedema to another dipeptidyl peptidase-4 (DPP4) inhibitor because it is unknown whether such patients will be predisposed to angioedema with saxagliptin and metformin hydrochloride extended-release tablets.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
Patients With Renal Impairment (Patients with Renal Impairment)
Saxagliptin
A single-dose, open-label trial was conducted to evaluate the pharmacokinetics of saxagliptin (10 mg dose) in subjects with varying degrees of chronic renal impairment compared to subjects with normal renal function. The 10 mg dosage is not an approved dosage. The degree of renal impairment did not affect Cmax of saxagliptin or its metabolite. In subjects with moderate renal impairment with eGFR 30 to less than 45 mL/min/1.73 m2, severe renal impairment (eGFR 15 to less than 30 mL/min/1.73 m2) and ESRD patient on hemodialysis, the AUC values of saxagliptin or its active metabolite were > 2 fold higher than AUC values in subjects with normal renal function.
Metformin HCl
In patients with decreased renal function, the plasma and blood half-life of metformin is prolonged and the renal clearance is decreased [see Contraindications (4) and Warnings and Precautions (5.1) ].
14.2 Cardiovascular Safety Trial
The cardiovascular risk of saxagliptin was evaluated in SAVOR, a multicenter, multinational, randomized, double-blind trial comparing saxagliptin (N = 8280) to placebo (N = 8212), both administered in combination with standard of care, in adult patients with type 2 diabetes mellitus at high risk for atherosclerotic cardiovascular disease. Of the randomized trial patients, 97.5% completed the trial, and the median duration of follow-up was approximately 2 years. The trial was event-driven, and patients were followed until a sufficient number of events were accrued.
Patients were at least 40 years of age, had A1C ≥ 6.5%, and multiple risk factors (21% of randomized patients) for cardiovascular disease (age ≥ 55 years for men and ≥ 60 years for women plus at least one additional risk factor of dyslipidemia, hypertension, or current cigarette smoking) or established (79% of the randomized patients) cardiovascular disease defined as a history of ischemic heart disease, peripheral vascular disease, or ischemic stroke. The majority of patients were male (67%) and White (75%) with a mean age of 65 years. Approximately 16% of the population had moderate (estimated glomerular filtration rate [eGFR] ≥ 30 to ≤ 50 mL/min) to severe (eGFR < 30 mL/min) renal impairment, and 13% had a prior history of heart failure. Patients had a median duration of type 2 diabetes mellitus of approximately 10 years, and a mean baseline A1C level of 8.0%. Approximately 5% of patients were treated with diet and exercise only at baseline. Overall, the use of diabetes medications was balanced across treatment groups (metformin 69%, insulin 41%, sulfonylureas 40%, and TZDs 6%). The use of cardiovascular disease medications was also balanced (angiotensin-converting enzyme [ACE] inhibitors or angiotensin receptor blockers [ARBs] 79%, statins 78%, aspirin 75%, beta-blockers 62%, and non-aspirin antiplatelet medications 24%).
The primary analysis in SAVOR was time to first occurrence of a Major Adverse Cardiac Event (MACE). A major adverse cardiac event in SAVOR was defined as a cardiovascular death, or a nonfatal myocardial infarction (MI) or a nonfatal ischemic stroke. The trial was designed as a non-inferiority trial with a pre-specified risk margin of 1.3 for the hazard ratio of MACE, and was also powered for a superiority comparison if non-inferiority was demonstrated.
The results of SAVOR, including the contribution of each component to the primary composite endpoint are shown in Table 13. The incidence rate of MACE was similar in both treatment arms: 3.8 MACE per 100 patient-years on placebo vs. 3.8 MACE per 100 patient-years on saxagliptin. The estimated hazard ratio of MACE associated with saxagliptin relative to placebo was 1.00 with a 95.1% confidence interval of (0.89, 1.12). The upper bound of this confidence interval, 1.12, excluded a risk margin larger than 1.3.
|
Saxagliptin |
Placebo |
Hazard Ratio |
|||
|
Number of |
Rate per |
Number of |
Rate per 100 |
(95.1% CI) |
|
|
Composite of first event of CV death, non-fatal MI or non-fatal ischemic stroke (MACE) |
N = 8280 |
Total PY = 16308.8 |
N = 8212 |
Total PY = 16156.0 |
|
|
613 (7.4) |
3.8 |
609 (7.4) |
3.8 |
1.00 (0.89, 1.12) |
|
|
CV death |
245 (3.0) |
1.5 |
234 (2.8) |
1.4 |
|
|
Non-fatal MI |
233 (2.8) |
1.4 |
260 (3.2) |
1.6 |
|
|
Non-fatal |
135 (1.6) |
0.8 |
115 (1.4) |
0.7 |
The Kaplan-Meier-based cumulative event probability is presented in Figure 2 for time to first occurrence of the primary MACE composite endpoint by treatment arm. The curves for both saxagliptin and placebo arms are close together throughout the duration of the trial. The estimated cumulative event probability is approximately linear for both arms, indicating that the incidence of MACE for both arms was constant over the trial duration.
Vital status was obtained for 99% of patients in the trial. There were 798 deaths in the SAVOR trial. Numerically more patients (5.1%) died in the saxagliptin group than in the placebo group (4.6%). The risk of deaths from all cause (Table 14) was not statistically different between the treatment groups (HR: 1.11; 95.1% CI: 0.96, 1.27).
|
Saxagliptin |
Placebo |
Hazard Ratio |
|||
|
Number of |
Rate per 100 |
Number of |
Rate per 100 |
(95.1% CI) |
|
|
N = 8280 |
PY = 16645.3 |
N = 8212 |
PY = 16531.5 |
||
|
All-cause |
420 (5.1) |
2.5 |
378 (4.6) |
2.3 |
1.11 (0.96, 1.27) |
|
CV death |
269 (3.2) |
1.6 |
260 (3.2) |
1.6 |
|
|
Non-CV death |
151 (1.8) |
0.9 |
118 (1.4) |
0.7 |
Patients With Hepatic Impairment (Patients with Hepatic Impairment)
No pharmacokinetic studies of metformin have been conducted in patients with hepatic impairment.
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient to read the FDA-approved patient labeling (Medication Guide).
Lactic Acidosis
The risks of lactic acidosis due to the metformin component, its symptoms and conditions that predispose to its development, as noted in Warnings and Precautions (5.1), should be explained to patients. Patients should be advised to discontinue saxagliptin and metformin hydrochloride extended-release tablets immediately and to promptly notify their health care provider if unexplained hyperventilation, myalgia, malaise, unusual somnolence, dizziness, slow or irregular heartbeat, sensation of feeling cold (especially in the extremities), or other nonspecific symptoms occur. Gastrointestinal symptoms are common during initiation of metformin treatment and may occur during initiation of saxagliptin and metformin hydrochloride extended-release tablet therapy; however, patients should consult their physician if they develop unexplained symptoms. Although gastrointestinal symptoms that occur after stabilization are unlikely to be drug related, such an occurrence of symptoms should be evaluated to determine if it may be due to lactic acidosis or other serious disease.
Patients should be counseled against excessive alcohol intake while receiving saxagliptin and metformin hydrochloride extended-release tablets.
Patients should be informed about the importance of regular testing of renal function and hematological parameters when receiving treatment with saxagliptin and metformin hydrochloride extended-release tablets.
Instruct patients to inform their doctor that they are taking saxagliptin and metformin hydrochloride extended-release tablets prior to any surgical or radiological procedure, as temporary discontinuation of saxagliptin and metformin hydrochloride extended-release tablets may be required until renal function has been confirmed to be normal [see Warnings and Precautions (5.1) ].
Pancreatitis
Inform patients that acute pancreatitis has been reported during post-marketing use of saxagliptin. Educate patients that persistent severe abdominal pain, sometimes radiating to the back, which may or may not be accompanied by vomiting, is the hallmark symptom of acute pancreatitis. Instruct patients to promptly discontinue saxagliptin and metformin hydrochloride extended-release tablets and contact their health care provider if persistent severe abdominal pain occurs [see Warnings and Precautions (5.2) ].
Heart Failure
Inform patients of the signs and symptoms of heart failure. Before initiating saxagliptin and metformin hydrochloride extended-release tablets, ask patients if they have a history of heart failure or other risk factors for heart failure including moderate to severe renal impairment. Instruct patients to contact their health care provider as soon as possible if they experience symptoms of heart failure, including increasing shortness of breath, rapid increase in weight or swelling of the feet [see Warnings and Precautions (5.3) ].
Hypoglycemia with Concomitant Use with Insulin or Insulin Secretagogues
Inform patients that hypoglycemia can occur, particularly when insulin or an insulin secretagogue is used in combination with saxagliptin and metformin hydrochloride extended-release tablets. Educate patients about the risks, symptoms and appropriate management of hypoglycemia [see Warnings and Precautions (5.5) ].
Hypersensitivity Reactions
Patients should be informed that serious allergic (hypersensitivity) reactions, such as angioedema, anaphylaxis, and exfoliative skin conditions, have been reported during post-marketing use of saxagliptin. If symptoms of these allergic reactions (such as rash, skin flaking or peeling, urticaria, swelling of the skin, or swelling of the face, lips, tongue, and throat that may cause difficulty in breathing or swallowing) occur, patients must stop taking saxagliptin and metformin hydrochloride extended-release tablets and seek medical advice promptly [see Warnings and Precautions (5.6) ].
Severe and Disabling Arthralgia
Inform patients that severe and disabling joint pain may occur with this class of drugs. The time to onset of symptoms can range from one day to years. Instruct patients to seek medical advice if severe joint pain occurs [see Warnings and Precautions (5.7) ].
Bullous Pemphigoid
Inform patients that bullous pemphigoid may occur with this class of drugs. Instruct patients to seek medical advice if blisters or erosions occur [see Warnings and Precautions (5.8) ].
Administration Instructions
Patients should be informed that saxagliptin and metformin hydrochloride extended-release tablets must be swallowed whole and not crushed or chewed, and that the inactive ingredients may occasionally be eliminated in the feces as a soft mass that may resemble the original tablet.
Missed Dose
If a dose is missed, advise patients to take saxagliptin and metformin hydrochloride extended-release tablets as soon as they remember unless it is time for their next dose. Instruct patients not to take two doses of saxagliptin and metformin hydrochloride extended-release tablets at the same time.
7.2 Carbonic Anhydrase Inhibitors
Topiramate or other carbonic anhydrase inhibitors (e.g., zonisamide, acetazolamide or dichlorphenamide) frequently causes a decrease in serum bicarbonate and induce non-anion gap, hyperchloremic metabolic acidosis. Concomitant use of these drugs with saxagliptin and metformin hydrochloride extended-release tablets may increase the risk for lactic acidosis.
5.7 Severe and Disabling Arthralgia
There have been post-marketing reports of severe and disabling arthralgia in patients taking DPP4 inhibitors. The time to onset of symptoms following initiation of drug therapy varied from one day to years. Patients experienced relief of symptoms upon discontinuation of the medication. A subset of patients experienced a recurrence of symptoms when restarting the same drug or a different DPP4 inhibitor. Consider DPP4 inhibitors as a possible cause for severe joint pain and discontinue drug if appropriate.
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
How Supplied
Saxagliptin and Metformin Hydrochloride Extended-Release Tablets, 5 mg/500 mg, are pink, film-coated, round, unscored tablets imprinted with SM3 over M in black ink on one side of the tablet and blank on the other side. They are available as follows:
NDC 0378-8175-93
bottles of 30 tablets
Saxagliptin and Metformin Hydrochloride Extended-Release Tablets, 5 mg/1000 mg, are pink, film-coated, capsule shaped, unscored tablets imprinted with SM6 over M in black ink on one side of the tablet and blank on the other side. They are available as follows:
NDC 0378-8177-93
bottles of 30 tablets
Saxagliptin and Metformin Hydrochloride Extended-Release Tablets, 2.5 mg/1000 mg, are light peach, film-coated, capsule shaped, unscored tablets imprinted with SM4 over M in black ink on one side of the tablet and blank on the other side. They are available as follows:
NDC 0378-8176-91
bottles of 60 tablets
Storage and Handling
Store at 20° to 25°C (68° to 77°F). [See USP Controlled Room Temperature.]
Dispense in a tight, light-resistant container as defined in the USP using a child-resistant closure.
PHARMACIST: Dispense a Medication Guide with each prescription.
7.5 Insulin Or Insulin Secretagogues (7.5 Insulin or Insulin Secretagogues)
Insulin and insulin secretagogues are known to cause hypoglycemia. Concomitant use of saxagliptin and metformin hydrochloride extended-release tablets with insulin or an insulin secretagogue may require lower dosages of insulin or the insulin secretagogue to reduce the risk of hypoglycemia [see Warnings and Precautions (5.5) ].
7.6 Drugs Affecting Glycemic Control
Certain drugs tend to produce hyperglycemia and may lead to loss of glycemic control. These medications include the thiazides and other diuretics, corticosteroids, phenothiazines, thyroid products, estrogens, oral contraceptives, phenytoin, nicotinic acid, sympathomimetics, calcium channel blocking drugs, and isoniazid. When such drugs are administered to a patient receiving saxagliptin and metformin hydrochloride extended-release tablets, observe the patient closely for loss of blood glucose control. When such drugs are withdrawn from a patient receiving saxagliptin and metformin hydrochloride extended-release tablets, observe the patient closely for hypoglycemia.
Saxagliptin Morning and Evening Dosing
A 24-week monotherapy trial was conducted to assess a range of dosing regimens for saxagliptin. Treatment-naive patients with inadequately controlled diabetes (A1C ≥ 7% to ≤ 10%) underwent a 2-week, single-blind diet, exercise, and placebo lead-in period. A total of 365 patients were randomized to 2.5 mg every morning, 5 mg every morning, 2.5 mg with possible titration to 5 mg every morning, or 5 mg every evening of saxagliptin, or placebo. Patients who failed to meet specific glycemic goals during the trial were treated with metformin HCl rescue therapy added on to placebo or saxagliptin; the number of patients randomized per treatment group ranged from 71 to 74.
Treatment with either saxagliptin 5 mg every morning or 5 mg every evening provided significant improvements in A1C versus placebo (mean placebo-corrected reductions of -0.4% and -0.3%, respectively).
Coadministration of Saxagliptin with Metformin HCl Immediate-Release in Treatment-Naive Patients
A total of 1306 treatment-naive patients with type 2 diabetes mellitus participated in this 24-week, randomized, double-blind, active-controlled trial to evaluate the efficacy and safety of saxagliptin coadministered with metformin HCl immediate-release in patients with inadequate glycemic control (A1C ≥ 8% to ≤ 12%) on diet and exercise alone. Patients were required to be treatment-naive to be enrolled in this trial.
Patients who met eligibility criteria were enrolled in a single-blind, 1-week, dietary and exercise placebo lead-in period. Patients were randomized to one of four treatment arms: saxagliptin 5 mg + metformin HCl immediate-release 500 mg, saxagliptin 10 mg + metformin HCl immediate-release 500 mg, saxagliptin 10 mg + placebo, or metformin HCl immediate-release 500 mg + placebo (the maximum recommended approved saxagliptin dose is 5 mg daily; the 10 mg daily dose of saxagliptin does not provide greater efficacy than the 5 mg daily dose and the 10 mg saxagliptin dosage is not an approved dosage). Saxagliptin was dosed once daily. In the 3 treatment groups using metformin HCl immediate-release, the metformin HCl dose was up-titrated weekly in 500 mg per day increments, as tolerated, to a maximum of 2,000 mg per day based on FPG. Patients who failed to meet specific glycemic goals during this trial were treated with pioglitazone rescue as add-on therapy.
Coadministration of saxagliptin 5 mg plus metformin HCl immediate-release provided significant improvements in A1C, FPG, and PPG compared with placebo plus metformin HCl immediate-release (Table 7).
|
Efficacy Parameter |
Saxagliptin 5 mg + Metformin HCl N = 320 |
Placebo + Metformin HCl N = 328 |
|
Hemoglobin A1C (%) |
N = 306 |
N = 313 |
|
Baseline (mean) |
9.4 |
9.4 |
|
Change from baseline (adjusted mean Least squares mean adjusted for baseline value. )
|
-2.5 |
-2.0 |
|
Difference from placebo + metformin HCl (adjusted mean ) |
-0.5 p-value < 0.0001 compared to placebo + metformin HCl.
|
|
|
95% Confidence Interval |
(-0.7, -0.4) |
|
|
Percent of patients achieving A1C < 7% |
60%§ (185/307) |
41% (129/314) |
|
Fasting Plasma Glucose (mg/dL) |
N = 315 |
N = 320 |
|
Baseline (mean) |
199 |
199 |
|
Change from baseline (adjusted mean ) |
-60 |
-47 |
|
Difference from placebo + metformin HCl (adjusted mean ) |
-13 p-value < 0.05 compared to placebo + metformin HCl.
|
|
|
95% Confidence Interval |
(-19, -6) |
|
|
2-hour Postprandial Glucose (mg/dL) |
N = 146 |
N = 141 |
|
Baseline (mean) |
340 |
355 |
|
Change from baseline (adjusted mean ) |
-138 |
-97 |
|
Difference from placebo + metformin HCl (adjusted mean ) |
-41 |
|
|
95% Confidence Interval |
(-57, -25) |
Addition of Saxagliptin to Metformin HCl Immediate-Release
A total of 743 patients with type 2 diabetes mellitus participated in this 24-week, randomized, double-blind, placebo-controlled trial to evaluate the efficacy and safety of saxagliptin in combination with metformin HCl immediate-release in patients with inadequate glycemic control (A1C ≥ 7% and ≤ 10%) on metformin HCl alone. To qualify for enrollment, patients were required to be on a stable dose of metformin HCl (1,500-2,550 mg daily) for at least 8 weeks.
Patients who met eligibility criteria were enrolled in a single-blind, 2-week, dietary and exercise placebo lead-in period during which patients received metformin HCl immediate-release at their pre-trial dose, up to 2500 mg daily, for the duration of the trial. Following the lead-in period, eligible patients were randomized to 2.5 mg, 5 mg, or 10 mg of saxagliptin or placebo in addition to their current dose of open-label metformin HCl immediate-release (the maximum recommended approved saxagliptin dose is 5 mg daily; the 10 mg daily dose of saxagliptin does not provide greater efficacy than the 5 mg daily dose and the 10 mg dosage is not an approved dosage). Patients who failed to meet specific glycemic goals during the trial were treated with pioglitazone rescue therapy, added on to existing trial medications. Dose titrations of saxagliptin and metformin HCl immediate-release were not permitted.
Saxagliptin 2.5 mg and 5 mg add-on to metformin HCl immediate-release provided significant improvements in A1C, FPG, and PPG compared with placebo add-on to metformin HCl immediate-release (Table 8). Mean changes from baseline for A1C over time and at endpoint are shown in Figure 1. The proportion of patients who discontinued for lack of glycemic control or who were rescued for meeting prespecified glycemic criteria was 15% in the saxagliptin 2.5 mg add-on to metformin HCl immediate-release group, 13% in the saxagliptin 5 mg add-on to metformin HCl immediate-release group, and 27% in the placebo add-on to metformin HCl immediate-release group.
|
Efficacy Parameter |
Saxagliptin 2.5 mg + Metformin HCl N = 192 |
Saxagliptin 5 mg + Metformin HCl N = 191 |
Placebo + Metformin HCl N = 179 |
|
Hemoglobin A1C (%) |
N = 186 |
N = 186 |
N = 175 |
|
Baseline (mean) |
8.1 |
8.1 |
8.1 |
|
Change from baseline (adjusted mean Least squares mean adjusted for baseline value. )
|
-0.6 |
-0.7 |
+0.1 |
|
Difference from placebo (adjusted mean ) |
-0.7 p-value < 0.0001 compared to placebo + metformin HCl.
|
-0.8 |
|
|
95% Confidence Interval |
(-0.9, -0.5) |
(-1.0, -0.6) |
|
|
Percent of patients achieving A1C < 7% |
37% p-value < 0.05 compared to placebo + metformin HCl. (69/186)
|
44% (81/186) |
17% (29/175) |
|
Fasting Plasma Glucose (mg/dL) |
N = 188 |
N = 187 |
N = 176 |
|
Baseline (mean) |
174 |
179 |
175 |
|
Change from baseline (adjusted mean ) |
-14 |
-22 |
+1 |
|
Difference from placebo (adjusted mean ) |
-16 |
-23 |
|
|
95% Confidence Interval |
(-23, -9) |
(-30, -16) |
|
|
2-hour Postprandial Glucose (mg/dL) |
N = 155 |
N = 155 |
N = 135 |
|
Baseline (mean) |
294 |
296 |
295 |
|
Change from baseline (adjusted mean ) |
-62 |
-58 |
-18 |
|
Difference from placebo (adjusted mean ) |
-44 |
-40 |
|
|
95% Confidence Interval |
(-60, -27) |
(-56, -24) |
Saxagliptin Add-On Combination Therapy with Metformin HCl Immediate-Release versus Glipizide Add-On Combination Therapy with Metformin HCl Immediate-Release
In this 52-week, active-controlled trial, a total of 858 patients with type 2 diabetes mellitus and inadequate glycemic control (A1C > 6.5% and ≤ 10%) on metformin HCl immediate-release alone were randomized to double-blind add-on therapy with saxagliptin or glipizide. Patients were required to be on a stable dose of metformin HCl immediate-release (at least 1,500 mg daily) for at least 8 weeks prior to enrollment.
Patients who met eligibility criteria were enrolled in a single-blind, 2-week, dietary and exercise placebo lead-in period during which patients received metformin HCl immediate-release (1,500-3,000 mg based on their pre-trial dose). Following the lead-in period, eligible patients were randomized to 5 mg of saxagliptin or 5 mg of glipizide in addition to their current dose of open-label metformin HCl immediate-release. Patients in the glipizide plus metformin HCl immediate-release group underwent blinded titration of the glipizide dose during the first 18 weeks of the trial up to a maximum glipizide dose of 20 mg per day. Titration was based on a goal FPG ≤ 110 mg/dL or the highest tolerable glipizide dose. Fifty percent (50%) of the glipizide-treated patients were titrated to the 20-mg daily dose; 21% of the glipizide-treated patients had a final daily glipizide dose of 5 mg or less. The mean final daily dose of glipizide was 15 mg.
After 52 weeks of treatment, saxagliptin and glipizide resulted in similar mean reductions from baseline in A1C when added to metformin HCl immediate-release therapy (Table 9). This conclusion may be limited to patients with baseline A1C comparable to those in the trial (91% of patients had baseline A1C < 9%).
From a baseline mean body weight of 89 kg, there was a statistically significant mean reduction of 1.1 kg in patients treated with saxagliptin compared to a mean weight gain of 1.1 kg in patients treated with glipizide (p < 0.0001).
|
Efficacy Parameter |
Saxagliptin 5 mg + Metformin HCl N = 428 |
Titrated Glipizide + Metformin HCl N = 430 |
|
Hemoglobin A1C (%) |
N = 423 |
N = 423 |
|
Baseline (mean) |
7.7 |
7.6 |
|
Change from baseline (adjusted mean) |
-0.6 |
-0.7 |
|
Difference from glipizide + metformin HCl (adjusted mean Least squares mean adjusted for baseline value. )
|
0.1 |
|
|
95% Confidence Interval |
(-0.02, 0.2) Saxagliptin + metformin HCl is considered non-inferior to glipizide + metformin HCl because the upper limit of this confidence interval is less than the prespecified non-inferiority margin of 0.35%.
|
|
|
Fasting Plasma Glucose (mg/dL) |
N = 420 |
N = 420 |
|
Baseline (mean) |
162 |
161 |
|
Change from baseline (adjusted mean ) |
-9 |
-16 |
|
Difference from glipizide + metformin HCl (adjusted mean ) |
6 |
|
|
95% Confidence Interval |
(2, 11) Significance not tested.
|
Saxagliptin Add-On Combination Therapy with Insulin (with or without Metformin HCl Immediate-Release)
A total of 455 patients with type 2 diabetes mellitus participated in this 24-week, randomized, double-blind, placebo-controlled trial to evaluate the efficacy and safety of saxagliptin in combination with insulin in patients with inadequate glycemic control (A1C ≥ 7.5% and ≤ 11%) on insulin alone (N = 141) or on insulin in combination with a stable dose of metformin HCl immediate-release (N = 314). Patients were required to be on a stable dose of insulin (≥ 30 units to ≤ 150 units daily) with ≤ 20% variation in total daily dose for ≥ 8 weeks prior to screening. Patients entered the trial on intermediate- or long-acting (basal) insulin or premixed insulin. Patients using short-acting insulins were excluded unless the short-acting insulin was administered as part of a premixed insulin.
Patients who met eligibility criteria were enrolled in a single-blind, four-week, dietary and exercise placebo lead-in period during which patients received insulin (and metformin HCl immediate-release if applicable) at their pretrial dose(s). Following the lead-in period, eligible patients were randomized to add-on therapy with either saxagliptin 5 mg or placebo. Doses of the antidiabetic therapies were to remain stable but patients were rescued and allowed to adjust the insulin regimen if specific glycemic goals were not met or if the investigator learned that the patient had self-increased the insulin dose by > 20%. Data after rescue were excluded from the primary efficacy analyses.
Add-on therapy with saxagliptin 5 mg provided significant improvements from baseline to Week 24 in A1C and PPG compared with add-on placebo (Table 10). Similar mean reductions in A1C versus placebo were observed for patients using saxagliptin 5 mg add-on to insulin alone and saxagliptin 5 mg add-on to insulin in combination with metformin HCl immediate-release (-0.4% and -0.4%, respectively). The percentage of patients who discontinued for lack of glycemic control or who were rescued was 23% in the saxagliptin group and 32% in the placebo group.
The mean daily insulin dose at baseline was 53 units in patients treated with saxagliptin 5 mg and 55 units in patients treated with placebo. The mean change from baseline in daily dose of insulin was 2 units for the saxagliptin 5 mg group and 5 units for the placebo group.
|
Efficacy Parameter |
Saxagliptin 5 mg + Insulin (+/- Metformin HCl) N = 304 |
Placebo + Insulin (+/- Metformin HCl) N = 151 |
|
Hemoglobin A1C (%) |
N = 300 |
N = 149 |
|
Baseline (mean) |
8.7 |
8.7 |
|
Change from baseline (adjusted mean Least squares mean adjusted for baseline value and metformin HCl use at baseline. )
|
-0.7 |
-0.3 |
|
Difference from placebo (adjusted mean ) |
-0.4 p-value < 0.0001 compared to placebo + insulin.
|
|
|
95% Confidence Interval |
(-0.6, -0.2) |
|
|
2-hour Postprandial Glucose (mg/dL) |
N = 262 |
N = 129 |
|
Baseline (mean) |
251 |
255 |
|
Change from baseline (adjusted mean ) |
-27 |
-4 |
|
Difference from placebo (adjusted mean ) |
-23 p-value < 0.05 compared to placebo + insulin.
|
|
|
95% Confidence Interval |
(-37, -9) |
The change in fasting plasma glucose from baseline to Week 24 was also tested, but was not statistically significant. The percent of patients achieving an A1C < 7% was 17% (52/300) with saxagliptin in combination with insulin compared to 7% (10/149) with placebo. Significance was not tested.
Saxagliptin Add-On Combination Therapy with Metformin HCl plus Sulfonylurea
A total of 257 patients with type 2 diabetes mellitus participated in this 24-week, randomized, double-blind, placebo-controlled trial to evaluate the efficacy and safety of saxagliptin in combination with metformin HCl plus a sulfonylurea in patients with inadequate glycemic control (A1C ≥ 7% and ≤ 10%). Patients were to be on a stable combined dose of metformin HCl extended-release or immediate-release (at maximum tolerated dose, with minimum dose for enrollment being 1,500 mg) and a sulfonylurea (at maximum tolerated dose, with minimum dose for enrollment being ≥ 50% of the maximum recommended dose) for ≥ 8 weeks prior to enrollment.
Patients who met eligibility criteria were entered in a 2-week enrollment period to allow assessment of inclusion/exclusion criteria. Following the 2-week enrollment period, eligible patients were randomized to either double-blind saxagliptin (5 mg once daily) or double-blind matching placebo for 24 weeks. During the 24-week double-blind treatment period, patients were to receive metformin HCl and a sulfonylurea at the same constant dose ascertained during enrollment. Sulfonylurea dose could be down titrated once in the case of a major hypoglycemic event or recurring minor hypoglycemic events. In the absence of hypoglycemia, titration (up or down) of trial medication during the treatment period was prohibited.
Saxagliptin in combination with metformin HCl plus a sulfonylurea provided significant improvements in A1C and PPG compared with placebo in combination with metformin HCl plus a sulfonylurea (Table 11). The percentage of patients who discontinued for lack of glycemic control was 6% in the saxagliptin group and 5% in the placebo group.
|
Efficacy Parameter |
Saxagliptin 5 mg + Metformin HCl plus Sulfonylurea N = 129 |
Placebo + Metformin HCl plus Sulfonylurea N = 128 |
|
Hemoglobin A1C (%) |
N = 127 |
N = 127 |
|
Baseline (mean) |
8.4 |
8.2 |
|
Change from baseline (adjusted mean Least squares mean adjusted for baseline value. )
|
-0.7 |
-0.1 |
|
Difference from placebo (adjusted mean ) |
-0.7 p-value < 0.0001 compared to placebo + metformin HCl plus sulfonylurea.
|
|
|
95% Confidence Interval |
(-0.9, -0.5) |
|
|
2-hour Postprandial Glucose (mg/dL) |
N = 115 |
N = 113 |
|
Baseline (mean) |
268 |
262 |
|
Change from baseline (adjusted mean ) |
-12 |
5 |
|
Difference from placebo (adjusted mean ) |
-17 p-value < 0.05 compared to placebo + metformin HCl plus sulfonylurea.
|
|
|
95% Confidence Interval |
(-32, -2) |
The change in fasting plasma glucose from baseline to Week 24 was also tested, but was not statistically significant. The percent of patients achieving an A1C < 7% was 31% (39/127) with saxagliptin in combination with metformin HCl plus a sulfonylurea compared to 9% (12/127) with placebo. Significance was not tested.
Saxagliptin Add-on Combination Therapy with Metformin HCl plus an SGLT2 Inhibitor
A total of 315 patients with type 2 diabetes mellitus participated in this 24-week randomized, double-blind, placebo-controlled trial to evaluate the efficacy and safety of saxagliptin added to dapagliflozin (an SGLT2 inhibitor) and metformin HCl in patients with a baseline of HbA1c ≥ 7% to ≤ 10.5%. The mean age of these patients was 54.6 years, 1.6% were 75 years or older and 52.7% were female. The population was 87.9% White, 6.3% Black or African American, 4.1% Asian, and 1.6% other races. At baseline the population had diabetes for an average of 7.7 years and a mean HbA1c of 7.9%. The mean eGFR at baseline was 93.4 mL/min/1.73 m2. Patients were required to be on a stable dose of metformin HCl (≥ 1,500 mg per day) for at least 8 weeks prior to enrollment. Eligible patients who completed the screening period entered the lead-in treatment period, which included 16 weeks of open-label metformin HCl and 10 mg dapagliflozin treatment. Following the lead-in period, eligible patients were randomized to saxagliptin 5 mg (N = 153) or placebo (N = 162).
The group treated with add-on saxagliptin had statistically significant greater reductions in HbA1c from baseline versus the group treated with placebo (see Table 12).
|
Saxagliptin 5 mg (N = 153) Number of randomized and treated patients.
|
Placebo (N = 162) |
|
|
In combination with Dapagliflozin and Metformin |
||
|
Hemoglobin A1C (%) Analysis of Covariance including all post-baseline data regardless of rescue or treatment discontinuation. Model estimates calculated using multiple imputation to model washout of the treatment effect using placebo data for all patients having missing Week 24 data.
|
||
|
Baseline (mean) |
8.0 |
7.9 |
|
Change from baseline (adjusted mean Least squares mean adjusted for baseline value. )
95% Confidence Interval |
-0.5 (-0.6, -0.4) |
-0.2 (-0.3, -0.1) |
|
Difference from placebo (adjusted mean) 95% Confidence Interval |
-0.4 p-value < 0.0001.
(-0.5, -0.2) |
The known proportion of patients achieving HbA1c < 7% at Week 24 was 35.3% in the saxagliptin-treated group compared to 23.1% in the placebo-treated group.
In Vivo Assessment of Drug Interactions
| ND = not determined; QD = once daily; q6h = every 6 hours; q12h = every 12 hours; BID = twice daily; LA = long acting. | |||||
|
Coadministered Drug |
Dosage of Coadministered Drug Single dose unless otherwise noted. The 10 mg saxagliptin dose is not an approved dosage.
|
Dosage of Saxagliptin |
Geometric Mean Ratio (ratio with/without coadministered drug) No Effect = 1.00 |
||
|
AUC AUC = AUC(INF) for drugs given as single dose and AUC = AUC(TAU) for drugs given in multiple doses.
|
Cmax |
||||
|
No dosing adjustments required for the following: |
|||||
|
Metformin |
1,000 mg |
100 mg |
saxagliptin |
0.98 |
0.79 |
|
5-hydroxy saxagliptin |
0.99 |
0.88 |
|||
|
Glyburide |
5 mg |
10 mg |
saxagliptin |
0.98 |
1.08 |
|
5-hydroxy saxagliptin |
ND |
ND |
|||
|
Pioglitazone Results exclude one patient.
|
45 mg QD for 10 days |
10 mg QD for 5 days |
saxagliptin |
1.11 |
1.11 |
|
5-hydroxy saxagliptin |
ND |
ND |
|||
|
Digoxin |
0.25 mg q6h first day followed by q12h second day followed by QD for 5 days |
10 mg QD for 7 days |
saxagliptin |
1.05 |
0.99 |
|
5-hydroxy saxagliptin |
1.06 |
1.02 |
|||
|
Dapagliflozin |
10 mg single dose |
5 mg single dose |
saxagliptin |
↓ 1% |
↓ 7% |
|
5-hydroxy saxagliptin |
↑ 9% |
↑ 6% |
|||
|
Simvastatin |
40 mg QD for 8 days |
10 mg QD for 4 days |
saxagliptin |
1.12 |
1.21 |
|
5-hydroxy saxagliptin |
1.02 |
1.08 |
|||
|
Diltiazem |
360 mg LA QD for 9 days |
10 mg |
saxagliptin |
2.09 |
1.63 |
|
5-hydroxy saxagliptin |
0.66 |
0.57 |
|||
|
Rifampin The plasma dipeptidyl peptidase-4 (DPP4) activity inhibition over a 24-hour dose interval was not affected by rifampin.
|
600 mg QD for 6 days |
5 mg |
saxagliptin |
0.24 |
0.47 |
|
5-hydroxy saxagliptin |
1.03 |
1.39 |
|||
|
Omeprazole |
40 mg QD for 5 days |
10 mg |
saxagliptin |
1.13 |
0.98 |
|
5-hydroxy saxagliptin |
ND |
ND |
|||
|
Aluminum hydroxide + magnesium hydroxide + simethicone |
aluminum hydroxide: 2400 mg magnesium hydroxide: 2400 mg simethicone: 240 mg |
10 mg |
saxagliptin |
0.97 |
0.74 |
|
5-hydroxy saxagliptin |
ND |
ND |
|||
|
Famotidine |
40 mg |
10 mg |
saxagliptin |
1.03 |
1.14 |
|
5-hydroxy saxagliptin |
ND |
ND |
|||
|
Limit saxagliptin and metformin hydrochloride extended-release tablet dose to 2.5 mg/1,000 mg once daily when coadministered with strong CYP3A4/5 inhibitors [see Drug Interactions (7.1) and Dosage and Administration (2.2) ]: |
|||||
|
Ketoconazole |
200 mg BID for 9 days |
100 mg |
saxagliptin |
2.45 |
1.62 |
|
5-hydroxy saxagliptin |
0.12 |
0.05 |
|||
|
Ketoconazole |
200 mg BID for 7 days |
20 mg |
saxagliptin |
3.67 |
2.44 |
|
5-hydroxy saxagliptin |
ND |
ND |
| ND = not determined; QD = once daily; q6h = every 6 hours; q12h = every 12 hours; BID = twice daily; LA = long acting. | |||||
|
Coadministered Drug |
Dosage of Coadministered Drug Single dose unless otherwise noted. The 10 mg saxagliptin dose is not an approved dosage.
|
Dosage of Saxagliptin |
Geometric Mean Ratio (ratio with/without saxagliptin) No Effect = 1.00 |
||
|
AUC AUC = AUC(INF) for drugs given as single dose and AUC = AUC(TAU) for drugs given in multiple doses.
|
Cmax |
||||
|
No dosing adjustments required for the following: |
|||||
|
Metformin |
1,000 mg |
100 mg |
metformin |
1.20 |
1.09 |
|
Glyburide |
5 mg |
10 mg |
glyburide |
1.06 |
1.16 |
|
Pioglitazone Results include all patients.
|
45 mg QD for 10 days |
10 mg QD for 5 days |
pioglitazone |
1.08 |
1.14 |
|
hydroxy-pioglitazone |
ND |
ND |
|||
|
Digoxin |
0.25 mg q6h first day followed by q12h second day followed by QD for 5 days |
10 mg QD for 7 days |
digoxin |
1.06 |
1.09 |
|
Simvastatin |
40 mg QD for 8 days |
10 mg QD for 4 days |
simvastatin |
1.04 |
0.88 |
|
simvastatin acid |
1.16 |
1.00 |
|||
|
Diltiazem |
360 mg LA QD for 9 days |
10 mg |
diltiazem |
1.10 |
1.16 |
|
Ketoconazole |
200 mg BID for 9 days |
100 mg |
ketoconazole |
0.87 |
0.84 |
|
Ethinyl estradiol and norgestimate |
ethinyl estradiol 0.035 mg and norgestimate 0.250 mg for 21 days |
5 mg QD for 21 days |
ethinyl estradiol |
1.07 |
0.98 |
|
norelgestromin |
1.10 |
1.09 |
|||
|
norgestrel |
1.13 |
1.17 |
|
Coadministered Drug |
Dose of Coadministered Drug All metformin and coadministered drugs were given as single doses.
|
Dose of Metformin |
Geometric Mean Ratio (ratio with/without coadministered drug) No Effect = 1.00 |
||
|
AUC AUC = AUC(INF).
|
Cmax |
||||
|
No dosing adjustments required for the following: |
|||||
|
Glyburide |
5 mg |
850 mg |
metformin |
0.91 Ratio of arithmetic means.
|
0.93 |
|
Furosemide |
40 mg |
850 mg |
metformin |
1.09 |
1.22 |
|
Nifedipine |
10 mg |
850 mg |
metformin |
1.16 |
1.21 |
|
Propranolol |
40 mg |
850 mg |
metformin |
0.90 |
0.94 |
|
Ibuprofen |
400 mg |
850 mg |
metformin |
1.05 |
1.07 |
|
Drugs that are eliminated by renal tubular secretion may increase the accumulation of metformin [see Drug Interactions (7.3) ]. |
|||||
|
Cimetidine |
400 mg |
850 mg |
metformin |
1.40 |
1.61 |
|
Coadministered |
Dose of All metformin and coadministered drugs were given as single doses.
|
Dose of |
Geometric Mean Ratio (ratio with/without metformin) No Effect = 1.00 |
||
|
AUC AUC = AUC(INF) unless otherwise noted.
|
Cmax |
||||
|
No dosing adjustments required for the following: |
|||||
|
Glyburide |
5 mg |
850 mg |
glyburide |
0.78 Ratio of arithmetic means, p-value of difference < 0.05.
|
0.63 |
|
Furosemide |
40 mg |
850 mg |
furosemide |
0.87 |
0.69 |
|
Nifedipine |
10 mg |
850 mg |
nifedipine |
1.10 AUC(0-24 hr) reported.
|
1.08 |
|
Propranolol |
40 mg |
850 mg |
propranolol |
1.01 |
1.02 |
|
Ibuprofen |
400 mg |
850 mg |
ibuprofen |
0.97 Ratio of arithmetic means.
|
1.01 |
|
Cimetidine |
400 mg |
850 mg |
cimetidine |
0.95 |
1.01 |
Principal Display Panel – 5 Mg/1000mg (PRINCIPAL DISPLAY PANEL – 5 mg/1000mg)
NDC 0378-8177-93
Saxagliptin and
Metformin HCl
Extended-Release
Tablets
5 mg/1000 mg
PHARMACIST: Dispense the accompanying
Medication Guide to each patient.
Do not crush, cut, or chew tablets.
Tablets must be swallowed whole.
Rx only 30 Tablets
Each film-coated extended-release tablet
contains 6.149 mg saxagliptin hydrochloride
dihydrate equivalent to 5 mg saxagliptin and
1000 mg metformin hydrochloride, USP.
Usual Dosage: See accompanying
prescribing information.
Keep this and all medication out of
the reach of children.
Store at 20° to 25°C (68° to 77°F).
[See USP Controlled Room
Temperature.]
Manufactured for:
Mylan Pharmaceuticals Inc.
Morgantown, WV 26505 U.S.A.
Made in India
Mylan.com
RMXA8177H1
Dispense in a tight, light-resistant
container as defined in the USP
using a child-resistant closure.
Keep container tightly closed.
Code No.: MH/DRUGS/AD/089
Principal Display Panel – 5 Mg/500 Mg (PRINCIPAL DISPLAY PANEL – 5 mg/500 mg)
NDC 0378-8175-93
Saxagliptin and
Metformin HCl
Extended-Release
Tablets
5 mg/500 mg
PHARMACIST: Dispense the accompanying
Medication Guide to each patient.
Do not crush, cut, or chew tablets.
Tablets must be swallowed whole.
Rx only 30 Tablets
Each film-coated extended-release tablet
contains 6.149 mg saxagliptin hydrochloride
dihydrate equivalent to 5 mg saxagliptin and
500 mg metformin hydrochloride, USP.
Usual Dosage: See accompanying
prescribing information.
Keep this and all medication out of
the reach of children.
Store at 20° to 25°C (68° to 77°F). [See
USP Controlled Room Temperature.]
Manufactured for:
Mylan Pharmaceuticals Inc.
Morgantown, WV 26505 U.S.A.
Made in India
Mylan.com
RMXA8175H1
Dispense in a tight, light-resistant
container as defined in the USP
using a child-resistant closure.
Keep container tightly closed.
Code No.: MH/DRUGS/AD/089
In Vitro Assessment of Drug Interactions
In in vitro studies, saxagliptin and its active metabolite did not inhibit CYP1A2, 2A6, 2B6, 2C9, 2C19, 2D6, 2E1, or 3A4, or induce CYP1A2, 2B6, 2C9, or 3A4. Therefore, saxagliptin is not expected to alter the metabolic clearance of coadministered drugs that are metabolized by these enzymes. Saxagliptin is a P-glycoprotein (P-gp) substrate, but is not a significant inhibitor or inducer of P-gp.
2.1 Recommended Dosage and Administration
Individualize the starting dosage of saxagliptin and metformin hydrochloride extended-release tablets based on the patient’s current regimen and the available strengths of saxagliptin and metformin hydrochloride extended-release tablets [see Dosage Forms and Strengths (3) ].
Administer saxagliptin and metformin hydrochloride extended-release tablets once daily with the evening meal, with gradual dose titration to reduce the gastrointestinal side effects associated with metformin HCl [see Adverse Reactions (6.1) ].
The recommended starting dosage of saxagliptin and metformin hydrochloride extended-release tablets in patients who need 5 mg of saxagliptin and who are not currently treated with metformin HCl is one saxagliptin and metformin hydrochloride extended-release tablet containing 5 mg saxagliptin and 500 mg metformin HCl extended-release once daily with gradual dose escalation to reduce the gastrointestinal side effects due to metformin HCl.
In patients treated with metformin HCl, the recommended starting dosage of saxagliptin and metformin hydrochloride extended-release tablets should provide metformin HCl at the dose already being taken, or the nearest therapeutically appropriate dose. Following a switch from metformin HCl immediate-release to saxagliptin and metformin hydrochloride extended-release tablets, closely monitor glycemic control and adjust the dosage accordingly.
Patients who need 2.5 mg saxagliptin in combination with metformin HCl extended-release may be treated with saxagliptin and metformin hydrochloride extended-release tablets 2.5 mg/1,000 mg. Patients who need 2.5 mg saxagliptin who are either metformin HCl naive or who require a dose of metformin HCl higher than 1,000 mg should use the individual components.
Gradually titrate the dosage of saxagliptin and metformin hydrochloride extended-release tablets, as needed, after assessing therapeutic response and tolerability, up to a maximum recommended dosage of saxagliptin and metformin hydrochloride extended-release tablets (5 mg for saxagliptin and 2,000 mg for metformin HCl extended-release orally once daily).
Inform patients that saxagliptin and metformin hydrochloride extended-release tablets must be swallowed whole and never crushed, cut, or chewed. Occasionally, the inactive ingredients of saxagliptin and metformin hydrochloride extended-release tablets will be eliminated in the feces as a soft, hydrated mass that may resemble the original tablet.
If a dose is missed, advise patients not to take an extra dose. Resume treatment with the next dose.
7.1 Strong Inhibitors of Cyp3a4/5 Enzymes (7.1 Strong Inhibitors of CYP3A4/5 Enzymes)
Ketoconazole significantly increased saxagliptin exposure. Similar significant increases in plasma concentrations of saxagliptin are anticipated with other strong CYP3A4/5 inhibitors (e.g., atazanavir, clarithromycin, indinavir, itraconazole, nefazodone, nelfinavir, ritonavir, saquinavir, and telithromycin). The dose of saxagliptin and metformin hydrochloride extended-release tablets should be limited to 2.5 mg of saxagliptin when coadministered with a strong CYP3A4/5 inhibitor [see Dosage and Administration (2.3) and Clinical Pharmacology (12.3) ].
7.3 Drugs That Reduce Metformin Clearance (7.3 Drugs that Reduce Metformin Clearance)
Concomitant use of drugs that interfere with common renal tubular transport systems involved in the renal elimination of metformin (e.g., organic cationic transporter-2 [OCT2]/multidrug and toxin extrusion [MATE] inhibitors such as ranolazine, vandetanib, dolutegravir, and cimetidine) could increase systemic exposure to metformin and may increase the risk for lactic acidosis [see Clinical Pharmacology (12.3) ]. Consider the benefits and risks of concomitant use.
Principal Display Panel – 2.5 Mg/1000 Mg (PRINCIPAL DISPLAY PANEL – 2.5 mg/1000 mg)
NDC 0378-8176-91
Saxagliptin and
Metformin HCl
Extended-Release
Tablets
2.5 mg/1000 mg
PHARMACIST: Dispense the accompanying
Medication Guide to each patient.
Do not crush, cut, or chew tablets.
Tablets must be swallowed whole.
Rx only 60 Tablets
Each film-coated extended-release tablet
contains 3.075 mg saxagliptin hydrochloride
dihydrate equivalent to 2.5 mg saxagliptin and
1000 mg metformin hydrochloride, USP.
Usual Dosage: See accompanying
prescribing information.
Keep this and all medication out of
the reach of children.
Store at 20° to 25°C (68° to 77°F).
[See USP Controlled Room
Temperature.]
Manufactured for:
Mylan Pharmaceuticals Inc.
Morgantown, WV 26505 U.S.A.
Made in India
Mylan.com
RMXA8176D1
Dispense in a tight, light-resistant
container as defined in the USP
using a child-resistant closure.
Keep container tightly closed.
Code No.: MH/DRUGS/AD/089
Adverse Reactions With Concomitant Use With Insulin (Adverse Reactions with Concomitant Use with Insulin)
In the add-on to insulin trial [see Clinical Studies (14.1) ], the incidence of adverse events, including serious adverse events and discontinuations due to adverse events, was similar between saxagliptin and placebo, except for confirmed hypoglycemia [see Adverse Reactions (6.1) ].
Disease Associated Maternal And/or Embryo/fetal Risk (Disease-associated maternal and/or embryo/fetal risk)
Poorly controlled diabetes in pregnancy increases the maternal risk for diabetic ketoacidosis, preeclampsia, spontaneous abortions, preterm delivery, still birth and delivery complications. Poorly controlled diabetes increases the fetal risk for major birth defects, stillbirth, and macrosomia related morbidity.
2.4 Discontinuation for Iodinated Contrast Imaging Procedures
Discontinue saxagliptin and metformin hydrochloride extended-release tablets at the time of, or prior to, an iodinated contrast imaging procedure in patients with an eGFR less than 60 mL/min/1.73 m2; a history of liver disease, alcoholism or heart failure; or in any patient who will be administered intra-arterial iodinated contrast. Re-evaluate eGFR 48 hours after the imaging procedure; restart saxagliptin and metformin hydrochloride extended-release tablets if renal function is stable [see Warnings and Precautions (5.1) ].
Saxagliptin and Metformin Hydrochloride Extended Release Tablets (Saxagliptin and Metformin Hydrochloride Extended-Release Tablets)
Elderly patients are more likely to have decreased renal function. Assess renal function more frequently in the elderly [see Warnings and Precautions (5.1) and Clinical Pharmacology (12.3) ].
2.2 Recommendations for Dosage and Administration in Renal Impairment
Assess renal function prior to initiation of saxagliptin and metformin hydrochloride extended-release tablets and then as clinically indicated [see Use in Specific Populations (8.6) ].
The recommended dosage of saxagliptin and metformin hydrochloride extended-release tablets in patients with an estimated glomerular filtration rate (eGFR) greater than or equal to 45 mL/minute/1.73 m2 is the same as the recommended dosage in patients with normal renal function [see Dosage and Administration (2.1) ].
In patients taking saxagliptin and metformin hydrochloride extended-release tablets whose eGFR later falls below 45 mL/minute/1.73 m2, assess the benefit risk of continuing therapy and limit dose of the saxagliptin component to 2.5 mg once daily.
Initiation of saxagliptin and metformin hydrochloride extended-release tablets in patients with an eGFR between 30 – 45 mL/minute/1.73 m2 is not recommended.
Saxagliptin and metformin hydrochloride extended-release tablets are contraindicated in patients with an eGFR below 30 mL/minute/1.73 m2.
Discontinue saxagliptin and metformin hydrochloride extended-release tablets if the patient’s eGFR later falls below 30 mL/minute/1.73 m2 [see Contraindications (4) and Warnings and Precautions (5.1) ].
2.3 Dosage Modifications With Concomitant Use of Strong Cyp3a4/5 Inhibitors (2.3 Dosage Modifications with Concomitant Use of Strong CYP3A4/5 Inhibitors)
The maximum recommended dosage of saxagliptin and metformin hydrochloride extended-release tablets is 2.5 mg of saxagliptin and 1,000 mg of metformin HCl given orally once daily when used concomitantly with strong cytochrome P450 3A4/5 (CYP3A4/5) inhibitors (e.g., ketoconazole, atazanavir, clarithromycin, indinavir, itraconazole, nefazodone, nelfinavir, ritonavir, saquinavir, and telithromycin) [see Dosage and Administration (2.1) , Drug Interactions (7.1), and Clinical Pharmacology (12.3) ].
Adverse Reactions Associated With Saxagliptin Coadministered With Metformin Hcl Immediate Release in Treatment Naive Patients With Type 2 Diabetes Mellitus (Adverse Reactions Associated with Saxagliptin Coadministered with Metformin HCl Immediate-Release in Treatment-Naive Patients with Type 2 Diabetes Mellitus)
Table 2 shows the adverse reactions reported (regardless of investigator assessment of causality) in ≥ 5% of patients participating in an additional 24-week, active-controlled trial of coadministered saxagliptin and metformin HCl in treatment-naive patients.
|
Number (%) of Patients |
||
|
Saxagliptin 5 mg + Metformin HCl Metformin HCl immediate-release was initiated at a starting dose of 500 mg daily and titrated up to a maximum of 2,000 mg daily.
N = 320 |
Placebo + Metformin HCl N = 328 |
|
|
Headache |
24 (7.5) |
17 (5.2) |
|
Nasopharyngitis |
22 (6.9) |
13 (4.0) |
In patients treated with the combination of saxagliptin and metformin HCl immediate-release, either as saxagliptin add-on to metformin HCl immediate-release therapy or as coadministration in treatment-naive patients, diarrhea was the only gastrointestinal-related event that occurred with an incidence ≥ 5% in any treatment group in both trials. In the saxagliptin add-on to metformin HCl immediate-release trial, the incidence of diarrhea was 9.9%, 5.8%, and 11.2% in the saxagliptin 2.5 mg, 5 mg, and placebo groups, respectively. When saxagliptin and metformin HCl immediate-release were coadministered in treatment-naive patients, the incidence of diarrhea was 6.9% in the saxagliptin 5 mg + metformin HCl immediate-release group and 7.3% in the placebo + metformin HCl immediate-release group.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:46:23.468325 · Updated: 2026-03-14T22:24:42.571850