Hydrocortisone Acetate Suppositories
cb60f2a6-8bab-c0f9-e053-2a95a90a422a
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
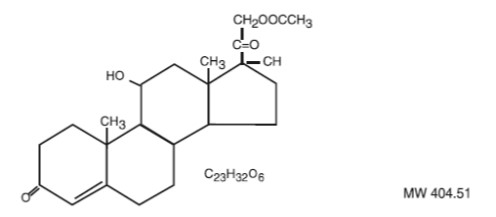
Hydrocortisone Acetate is a corticosteroid designated chemically as pregn-4-ene 3, 20-dione, 21- (acetyloxy)-11,17-dihydroxy-(11ß) with the following structural formula: Each rectal suppository contains hydrocortisone acetate, 25 mg in a specially blended hydrogenated vegetable oil base.
Indications and Usage
Hydrocortisone acetate suppositories are indicated for use in inflamed hemorrhoids, post-irradiation (factitial) proctitis; as an adjunct in the treatment of chronic ulcerative colitis; cryptitis; and other inflammatory conditions of anorectum and pruritus ani.
Dosage and Administration
FOR RECTAL ADMINISTRATION . Detach one suppository from strip of suppositories. Hold suppository upright and carefully separate tabs at top opening and pull downward from the pointed end to expose the suppository. Remove the suppository from the pocket. Avoid excessive handling of suppository which is designed to melt at body temperature. Insert one suppository rectally, pointed end first. Insert one suppository in the rectum twice daily, morning and night for two weeks, in nonspecific proctitis. In more severe cases, one suppository three times a day or two suppositories twice daily. In factitial proctitis, the recommended duration of therapy is six to eight weeks or less, according to the response of the individual case.
Contraindications
Hydrocortisone acetate suppositories are contraindicated in those patients having a history of hypersensitivity to hydrocortisone acetate or any of the components.
Adverse Reactions
The following local adverse reactions have been reported with hydrocortisone acetate suppositories: burning, itching, irritation, dryness, folliculitis, hypopigmentation, allergic contact dermatitis, secondary infection. To report SUSPECTED ADVERSE REACTIONS, contact FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Storage and Handling
Store at 20°-25°C (68°-77°F) [See USP Controlled Room Temperature]. Excursions permitted to 15°- 30°C (59°-86°F). Store away from heat. Protect from freezing. Avoid contact with eyes. KEEP THIS AND ALL DRUGS OUT OF THE REACH OF CHILDREN . In case of accidental ingestion, seek professional assistance or contact a Poison Control Center immediately. PHARMACIST : This product is not an Orange Book rated product, therefore all prescriptions using this product shall be subject to state and federal statutes as applicable. This product has not been subjected to FDA therapeutic or other equivalency testing. There are no claims of bioequivalence or therapeutic equivalence. Each person recommending a prescription substitution using this product shall make such recommendation based on his/her professional knowledge and opinion, upon evaluating the active ingredients, inactive ingredients, excipients and chemical information contained within the enclosed prescribing information.
How Supplied
Boxes of 12 suppositories NDC 24689-102-01 Boxes of 12 suppositories NDC 24689-103-01
Medication Information
Indications and Usage
Hydrocortisone acetate suppositories are indicated for use in inflamed hemorrhoids, post-irradiation (factitial) proctitis; as an adjunct in the treatment of chronic ulcerative colitis; cryptitis; and other inflammatory conditions of anorectum and pruritus ani.
Dosage and Administration
FOR RECTAL ADMINISTRATION. Detach one suppository from strip of suppositories. Hold suppository upright and carefully separate tabs at top opening and pull downward from the pointed end to expose the suppository. Remove the suppository from the pocket. Avoid excessive handling of suppository which is designed to melt at body temperature. Insert one suppository rectally, pointed end first. Insert one suppository in the rectum twice daily, morning and night for two weeks, in nonspecific proctitis. In more severe cases, one suppository three times a day or two suppositories twice daily. In factitial proctitis, the recommended duration of therapy is six to eight weeks or less, according to the response of the individual case.
Contraindications
Hydrocortisone acetate suppositories are contraindicated in those patients having a history of hypersensitivity to hydrocortisone acetate or any of the components.
Adverse Reactions
The following local adverse reactions have been reported with hydrocortisone acetate suppositories: burning, itching, irritation, dryness, folliculitis, hypopigmentation, allergic contact dermatitis, secondary infection.
To report SUSPECTED ADVERSE REACTIONS, contact FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Storage and Handling
Store at 20°-25°C (68°-77°F) [See USP Controlled Room Temperature]. Excursions permitted to 15°- 30°C (59°-86°F).
Store away from heat. Protect from freezing. Avoid contact with eyes.
KEEP THIS AND ALL DRUGS OUT OF THE REACH OF CHILDREN. In case of accidental ingestion, seek professional assistance or contact a Poison Control Center immediately.
PHARMACIST: This product is not an Orange Book rated product, therefore all prescriptions using this product shall be subject to state and federal statutes as applicable. This product has not been subjected to FDA therapeutic or other equivalency testing. There are no claims of bioequivalence or therapeutic equivalence. Each person recommending a prescription substitution using this product shall make such recommendation based on his/her professional knowledge and opinion, upon evaluating the active ingredients, inactive ingredients, excipients and chemical information contained within the enclosed prescribing information.
How Supplied
Boxes of 12 suppositories NDC 24689-102-01
Boxes of 12 suppositories NDC 24689-103-01
Description
Hydrocortisone Acetate is a corticosteroid designated chemically as pregn-4-ene 3, 20-dione, 21- (acetyloxy)-11,17-dihydroxy-(11ß)
with the following structural formula:
Each rectal suppository contains hydrocortisone acetate, 25 mg in a specially blended hydrogenated vegetable oil base.
Section 42229-5
Rx Only
Manufactured for:
Apnar Pharma LP
CHINO, CA 91710
Rev. 08/21
Overdosage
If signs and symptoms of systemic overdosage occur, discontinue use.
Precautions
Do not use hydrocortisone acetate suppositories unless adequate proctologic examination is made. If irritation develops, the product should be discontinued and appropriate therapy instituted.
In the presence of an infection, the use of an appropriate antifungal or antibacterial agent should be instituted. If a favorable response does not occur promptly, hydrocortisone acetate should be discontinued until the infection has been adequately controlled.
Carcinogenesis
No long term studies in animals have been performed to evaluate the carcinogenic potential of corticosteroid suppositories.
Pregnancy Category C
In laboratory animals, topical steroids have been associated with an increase in the incidence of fetal abnormalities when gestating females have been exposed to rather low dosage levels. There are no adequate and well controlled studies in pregnant women.
Hydrocortisone acetate suppositories should only be used during pregnancy if the potential benefit justifies the risk to the fetus. Drugs of this class should not be used extensively on pregnant patients, in large amounts, or for prolonged periods of time.
It is not known whether this drug is excreted in human milk and because many drugs are excreted in human milk and because of the potential for serious adverse reactions in nursing infants from hydrocortisone acetate suppositories, a decision should be made whether to discontinue nursing or to discontinue the drug, taking into account the importance of the drug to the mother.
Clinical Pharmacology
In normal subjects, about 26% of hydrocortisone acetate is absorbed when the suppository is applied to the rectum. Absorption of hydrocortisone acetate may vary across abraded or inflamed surfaces. Topical steroids are primarily effective because of their anti- inflammatory, anti-pruritic and vasoconstrictive action.
Information for Patients
Staining of fabric may occur with use of the suppository. Precautionary measures are recommended.
30mg Suppositories Carton
NDC 24689-103-01
Rx Only
Hydrocortisone Acetate
Suppositories
30 mg
FOR RECTAL USE ONLY
APNAR PHARMA LP
12 Suppositories
Unit Dose
Drug Abuse and Dependence
Drug abuse and dependence have not been reported in patients treated with hydrocortisone acetate suppositories.
25mg Suppositories Carton
NDC 24689-102-01
Rx Only
Hydrocortisone Acetate
Suppositories
25 mg
FOR RECTAL USE ONLY
APNAR PHARMA LP
12 Suppositories
Unit Dose
Structured Label Content
Section 42229-5 (42229-5)
Rx Only
Manufactured for:
Apnar Pharma LP
CHINO, CA 91710
Rev. 08/21
Overdosage (OVERDOSAGE)
If signs and symptoms of systemic overdosage occur, discontinue use.
Description (DESCRIPTION)
Hydrocortisone Acetate is a corticosteroid designated chemically as pregn-4-ene 3, 20-dione, 21- (acetyloxy)-11,17-dihydroxy-(11ß)
with the following structural formula:
Each rectal suppository contains hydrocortisone acetate, 25 mg in a specially blended hydrogenated vegetable oil base.
Precautions (PRECAUTIONS)
Do not use hydrocortisone acetate suppositories unless adequate proctologic examination is made. If irritation develops, the product should be discontinued and appropriate therapy instituted.
In the presence of an infection, the use of an appropriate antifungal or antibacterial agent should be instituted. If a favorable response does not occur promptly, hydrocortisone acetate should be discontinued until the infection has been adequately controlled.
How Supplied (HOW SUPPLIED)
Boxes of 12 suppositories NDC 24689-102-01
Boxes of 12 suppositories NDC 24689-103-01
Carcinogenesis
No long term studies in animals have been performed to evaluate the carcinogenic potential of corticosteroid suppositories.
Adverse Reactions (ADVERSE REACTIONS)
The following local adverse reactions have been reported with hydrocortisone acetate suppositories: burning, itching, irritation, dryness, folliculitis, hypopigmentation, allergic contact dermatitis, secondary infection.
To report SUSPECTED ADVERSE REACTIONS, contact FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Contraindications (CONTRAINDICATIONS)
Hydrocortisone acetate suppositories are contraindicated in those patients having a history of hypersensitivity to hydrocortisone acetate or any of the components.
Pregnancy Category C (PREGNANCY CATEGORY C)
In laboratory animals, topical steroids have been associated with an increase in the incidence of fetal abnormalities when gestating females have been exposed to rather low dosage levels. There are no adequate and well controlled studies in pregnant women.
Hydrocortisone acetate suppositories should only be used during pregnancy if the potential benefit justifies the risk to the fetus. Drugs of this class should not be used extensively on pregnant patients, in large amounts, or for prolonged periods of time.
It is not known whether this drug is excreted in human milk and because many drugs are excreted in human milk and because of the potential for serious adverse reactions in nursing infants from hydrocortisone acetate suppositories, a decision should be made whether to discontinue nursing or to discontinue the drug, taking into account the importance of the drug to the mother.
Storage and Handling (STORAGE AND HANDLING)
Store at 20°-25°C (68°-77°F) [See USP Controlled Room Temperature]. Excursions permitted to 15°- 30°C (59°-86°F).
Store away from heat. Protect from freezing. Avoid contact with eyes.
KEEP THIS AND ALL DRUGS OUT OF THE REACH OF CHILDREN. In case of accidental ingestion, seek professional assistance or contact a Poison Control Center immediately.
PHARMACIST: This product is not an Orange Book rated product, therefore all prescriptions using this product shall be subject to state and federal statutes as applicable. This product has not been subjected to FDA therapeutic or other equivalency testing. There are no claims of bioequivalence or therapeutic equivalence. Each person recommending a prescription substitution using this product shall make such recommendation based on his/her professional knowledge and opinion, upon evaluating the active ingredients, inactive ingredients, excipients and chemical information contained within the enclosed prescribing information.
Clinical Pharmacology (CLINICAL PHARMACOLOGY)
In normal subjects, about 26% of hydrocortisone acetate is absorbed when the suppository is applied to the rectum. Absorption of hydrocortisone acetate may vary across abraded or inflamed surfaces. Topical steroids are primarily effective because of their anti- inflammatory, anti-pruritic and vasoconstrictive action.
Indications and Usage (INDICATIONS AND USAGE)
Hydrocortisone acetate suppositories are indicated for use in inflamed hemorrhoids, post-irradiation (factitial) proctitis; as an adjunct in the treatment of chronic ulcerative colitis; cryptitis; and other inflammatory conditions of anorectum and pruritus ani.
Information for Patients (INFORMATION FOR PATIENTS)
Staining of fabric may occur with use of the suppository. Precautionary measures are recommended.
30mg Suppositories Carton (30mg-Suppositories Carton)
NDC 24689-103-01
Rx Only
Hydrocortisone Acetate
Suppositories
30 mg
FOR RECTAL USE ONLY
APNAR PHARMA LP
12 Suppositories
Unit Dose
Dosage and Administration (DOSAGE AND ADMINISTRATION)
FOR RECTAL ADMINISTRATION. Detach one suppository from strip of suppositories. Hold suppository upright and carefully separate tabs at top opening and pull downward from the pointed end to expose the suppository. Remove the suppository from the pocket. Avoid excessive handling of suppository which is designed to melt at body temperature. Insert one suppository rectally, pointed end first. Insert one suppository in the rectum twice daily, morning and night for two weeks, in nonspecific proctitis. In more severe cases, one suppository three times a day or two suppositories twice daily. In factitial proctitis, the recommended duration of therapy is six to eight weeks or less, according to the response of the individual case.
Drug Abuse and Dependence (DRUG ABUSE AND DEPENDENCE)
Drug abuse and dependence have not been reported in patients treated with hydrocortisone acetate suppositories.
25mg Suppositories Carton (25mg- Suppositories carton)
NDC 24689-102-01
Rx Only
Hydrocortisone Acetate
Suppositories
25 mg
FOR RECTAL USE ONLY
APNAR PHARMA LP
12 Suppositories
Unit Dose
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:36:44.079767 · Updated: 2026-03-14T21:47:30.128883