cae7322c-17de-0d81-e053-2a95a90a08d7
cae7322c-17de-0d81-e053-2a95a90a08d7
34390-5
HUMAN OTC DRUG LABEL
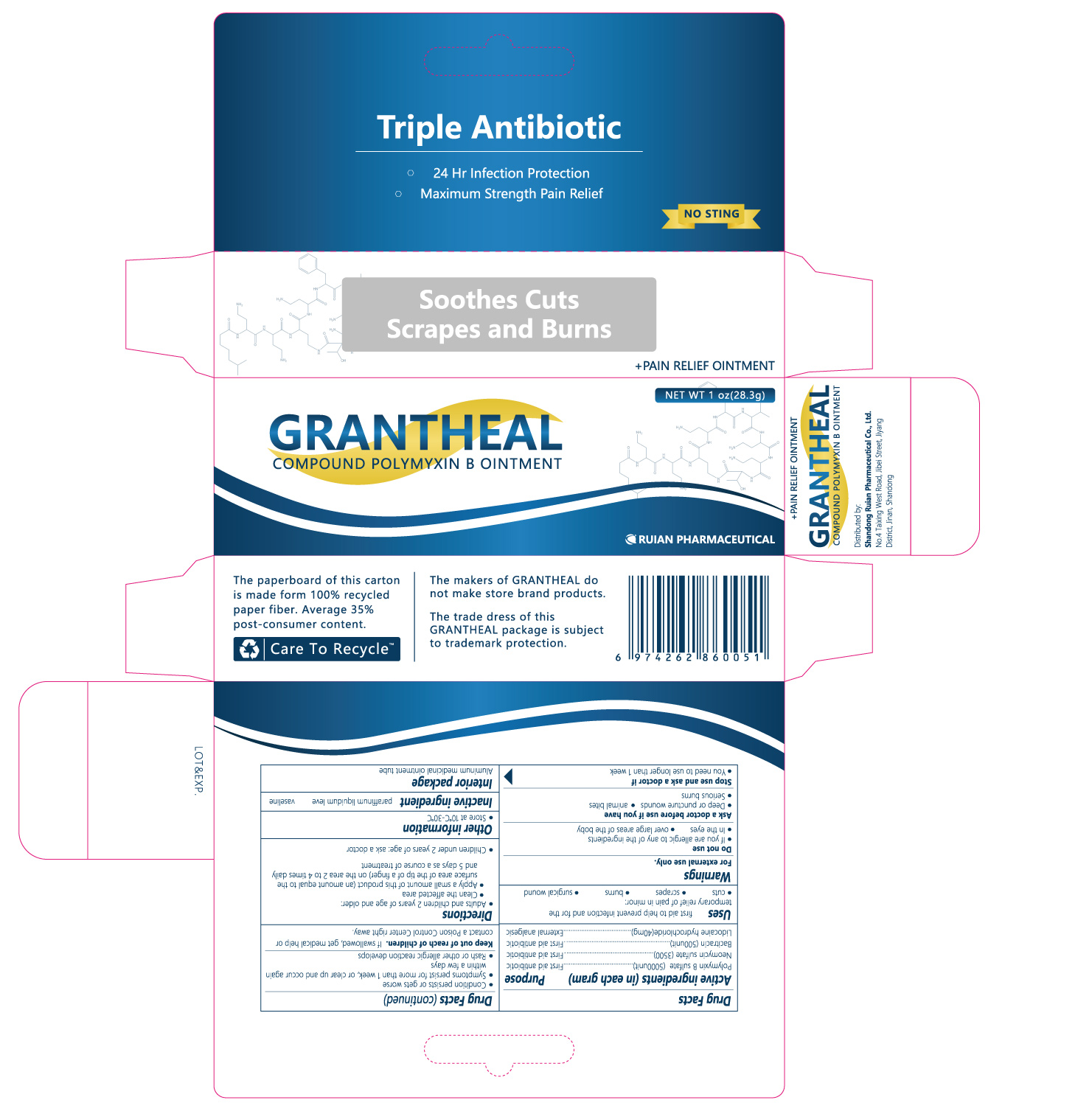
Drug Facts
Composition & Product
Identifiers & Packaging
Description
For external use only.
Medication Information
Warnings and Precautions
For external use only.
Active Ingredient
POLYMYXIN B SULFATE;
NEOMYCIN SULFATE;
BACITRACIN ZINC;
PRAMOXINE HYDROCHLORIDE
Indications and Usage
first aid to help prevent infection and for temporary relief of pain or discomfort in minor:
• cuts
• scrapes
• burns
Dosage and Administration
• adults and children 2 years of age or older:
• clean the affected area
• apply a small amount of this product (an amount equal to the surface area of the tip of a finger) on the area 1 to 3 times daily
• may be covered with a sterile bandage
• children under 2 years of age: ask a doctor
Description
For external use only.
Section 50565-1
If swallowed, get medical help or contact a Poison Control Center right away
Section 50569-3
• deep or puncture wounds
• animal bites
• serious burns
Section 50570-1
• if you are allergic to any of the ingredients
• in the eyes
• over large areas of the body
Section 51727-6
white petrolatum
Section 51945-4
NDC:82199-530-12 28.3 g in 1 TUBE
NDC:82199-530-11 14.2 g in 1 TUBE
Section 55105-1
First aid antibiotic
External analgesic
Section 60561-8
store at 10-30°C
Structured Label Content
Warnings and Precautions (34071-1)
For external use only.
Active Ingredient (55106-9)
POLYMYXIN B SULFATE;
NEOMYCIN SULFATE;
BACITRACIN ZINC;
PRAMOXINE HYDROCHLORIDE
Indications and Usage (34067-9)
first aid to help prevent infection and for temporary relief of pain or discomfort in minor:
• cuts
• scrapes
• burns
Dosage and Administration (34068-7)
• adults and children 2 years of age or older:
• clean the affected area
• apply a small amount of this product (an amount equal to the surface area of the tip of a finger) on the area 1 to 3 times daily
• may be covered with a sterile bandage
• children under 2 years of age: ask a doctor
Section 50565-1 (50565-1)
If swallowed, get medical help or contact a Poison Control Center right away
Section 50569-3 (50569-3)
• deep or puncture wounds
• animal bites
• serious burns
Section 50570-1 (50570-1)
• if you are allergic to any of the ingredients
• in the eyes
• over large areas of the body
Section 51727-6 (51727-6)
white petrolatum
Section 51945-4 (51945-4)
NDC:82199-530-12 28.3 g in 1 TUBE
NDC:82199-530-11 14.2 g in 1 TUBE
Section 55105-1 (55105-1)
First aid antibiotic
External analgesic
Section 60561-8 (60561-8)
store at 10-30°C
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:59:49.510828 · Updated: 2026-03-14T22:59:57.476249