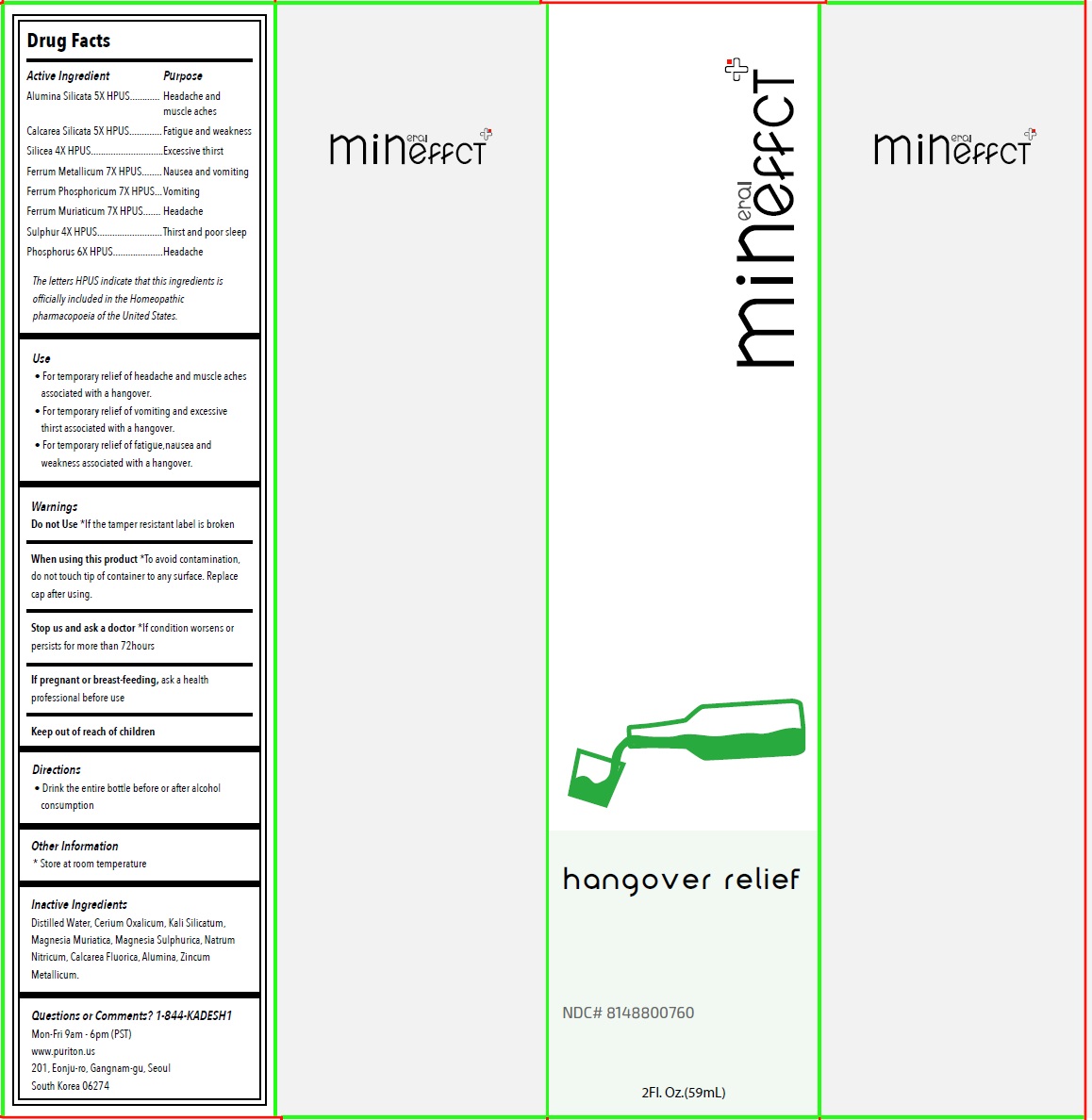
Mineffect Hangover Relief
c9821e87-1e8d-48ab-8821-32e7e660309a
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Alumina Silicata 5X HPUS Calcarea Silicata 5X HPUS Silicea 4X HPUS Ferrum Metallicum 7X HPUS Ferrum Phosphoricum 7X HPUS Ferrum Muriaticum 7X HPUS Sulphur 4X HPUS Phosphorus 6X HPUS The letters HPUS indicate that this ingredients is officially included in the Homeopathic pharmacopoeia of the United States.
Purpose
Headache and muscle aches Fatigue and weakness Excessive thirst Nausea and vomiting Vomiting Headache Thirst and poor sleep Headache
Medication Information
Purpose
Headache and muscle aches
Fatigue and weakness
Excessive thirst
Nausea and vomiting
Vomiting
Headache
Thirst and poor sleep
Headache
Description
Alumina Silicata 5X HPUS Calcarea Silicata 5X HPUS Silicea 4X HPUS Ferrum Metallicum 7X HPUS Ferrum Phosphoricum 7X HPUS Ferrum Muriaticum 7X HPUS Sulphur 4X HPUS Phosphorus 6X HPUS The letters HPUS indicate that this ingredients is officially included in the Homeopathic pharmacopoeia of the United States.
Use
• For temporary relief of headache and muscle aches associated with a hangover.
• For temporary relief of vomiting and excessive thirst associated with a hangover.
• For temporary relief of fatigue,nausea and weakness associated with a hangover.
Section 42229-5
mineffCT
Section 50565-1
Keep out of reach of children
Warnings
Do not Use*If the tamper resistant label is broken
When using this product*To avoid contamination, do not touch tip of container to any surface. Replace cap after using.
Stop us and ask a doctor*If condition worsens or persists for more than 72hours
If pregnant or breast-feeding, ask a health professional before use
Packaging
Directions
• Drink the entire bottle before or after alcohol consumption
Drug Facts
Active Ingredient
Alumina Silicata 5X HPUS
Calcarea Silicata 5X HPUS
Silicea 4X HPUS
Ferrum Metallicum 7X HPUS
Ferrum Phosphoricum 7X HPUS
Ferrum Muriaticum 7X HPUS
Sulphur 4X HPUS
Phosphorus 6X HPUS
The letters HPUS indicate that this ingredients is officially included in the Homeopathic pharmacopoeia of the United States.
Other Information
* Store at room temperature
Inactive Ingredients
Distilled Water, Cerium Oxalicum, Kali Silicatum, Magnesia Muriatica, Magnesia Sulphurica, Natrum Nitricum, Calcarea Fluorica, Alumina, Zincum Metallicum.
Questions Or Comments? 1 844 Kadesh1
Mon-Fri 9am - 6pm (PST)
www.puriton.us
201, Eonju-ro, Gangnam-gu, Seoul
South Korea 06274
Structured Label Content
Use
• For temporary relief of headache and muscle aches associated with a hangover.
• For temporary relief of vomiting and excessive thirst associated with a hangover.
• For temporary relief of fatigue,nausea and weakness associated with a hangover.
Section 42229-5 (42229-5)
mineffCT
Section 50565-1 (50565-1)
Keep out of reach of children
Purpose
Headache and muscle aches
Fatigue and weakness
Excessive thirst
Nausea and vomiting
Vomiting
Headache
Thirst and poor sleep
Headache
Warnings
Do not Use*If the tamper resistant label is broken
When using this product*To avoid contamination, do not touch tip of container to any surface. Replace cap after using.
Stop us and ask a doctor*If condition worsens or persists for more than 72hours
If pregnant or breast-feeding, ask a health professional before use
Packaging
Directions
• Drink the entire bottle before or after alcohol consumption
Drug Facts
Active Ingredient
Alumina Silicata 5X HPUS
Calcarea Silicata 5X HPUS
Silicea 4X HPUS
Ferrum Metallicum 7X HPUS
Ferrum Phosphoricum 7X HPUS
Ferrum Muriaticum 7X HPUS
Sulphur 4X HPUS
Phosphorus 6X HPUS
The letters HPUS indicate that this ingredients is officially included in the Homeopathic pharmacopoeia of the United States.
Other Information
* Store at room temperature
Inactive Ingredients
Distilled Water, Cerium Oxalicum, Kali Silicatum, Magnesia Muriatica, Magnesia Sulphurica, Natrum Nitricum, Calcarea Fluorica, Alumina, Zincum Metallicum.
Questions Or Comments? 1 844 Kadesh1 (Questions or Comments? 1-844-KADESH1)
Mon-Fri 9am - 6pm (PST)
www.puriton.us
201, Eonju-ro, Gangnam-gu, Seoul
South Korea 06274
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:11:18.978964 · Updated: 2026-03-14T23:16:32.315166