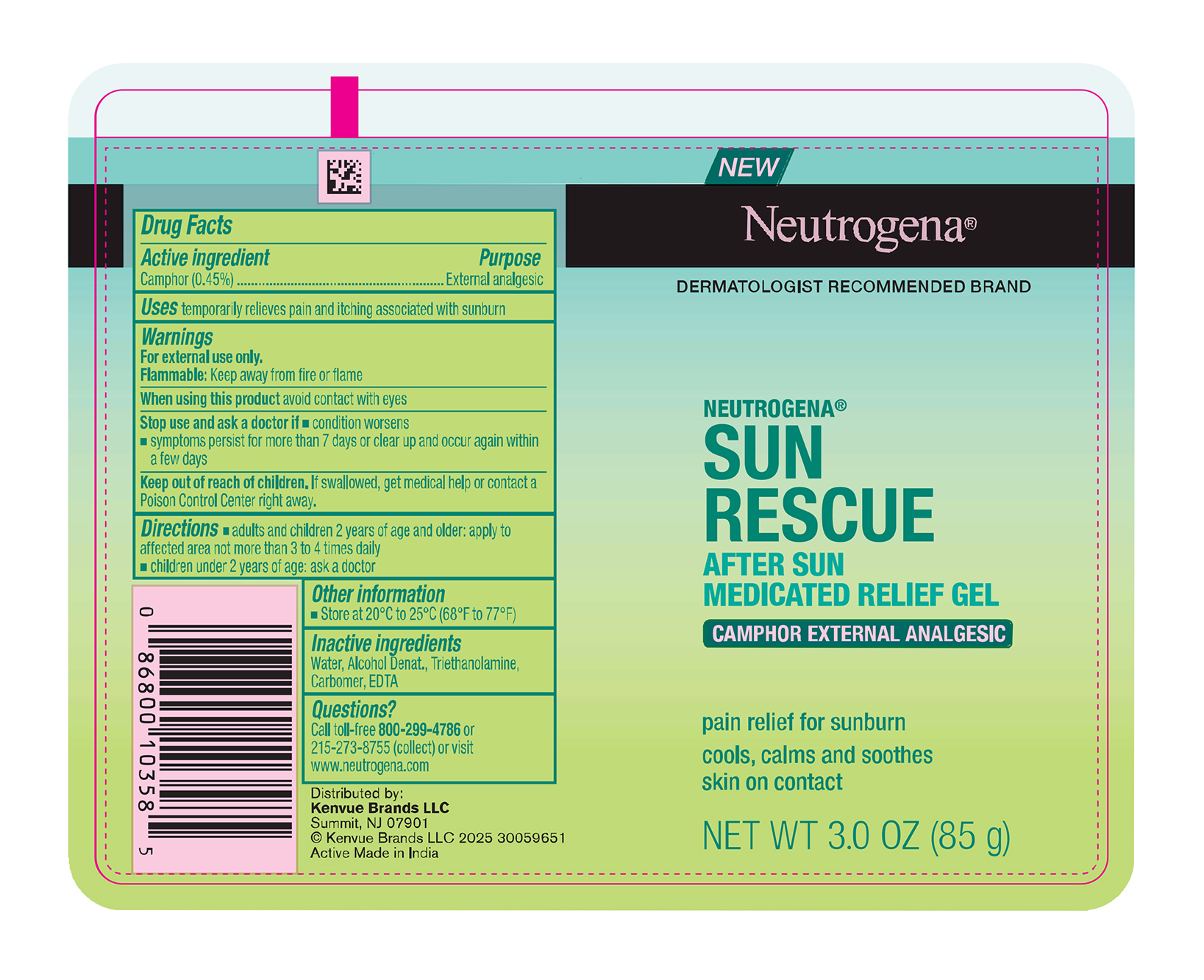
Neutrogena Sun Rescue After Sun Medicated Relief Gel
c95c5b64-b90f-1143-e053-2a95a90aa03c
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Drug Facts
Purpose
External analgesic
Medication Information
Purpose
External analgesic
Description
Drug Facts
Use
temporarily relieves pain and itching associated with sunburn
Section 42229-5
Drug Facts
Section 50565-1
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center right away.
Section 50567-7
When using this product avoid contact with eyes
Warnings
For external use only.
Flammable: Keep away from fire or flame
Directions
- adults and children 2 years of age and older: apply to affected area not more than 3 to 4 times daily
- children under 2 years of age: ask a doctor
Questions?
Call toll-free 800-299-4786 or 215-273-8755 (collect) or visit www.neutrogena.com
Active Ingredient
Camphor 0.45%
Other Information
- Store at 20°C to 25°C (68°F to 77°F)
Inactive Ingredients
Water, Alcohol Denat., Triethanolamine, Carbomer, EDTA
Stop Use and Ask A Doctor If
- condition worsens
- symptoms persist for more than 7 days or clear up and occur again within a few days
Principal Display Panel 85 G Tube Label
NEW
Neutrogena®
DERMATOLOGIST RECOMMENDED BRAND
NEUTROGENA®
SUN
RESCUE
AFTER SUN
MEDICATED RELIEF GEL
CAMPHOR EXTERNAL ANALGESIC
pain relief for sunburn
cools, calms and soothes
skin on contact
NET WT 3.0 OZ (85 g)
Structured Label Content
Use
temporarily relieves pain and itching associated with sunburn
Section 42229-5 (42229-5)
Drug Facts
Section 50565-1 (50565-1)
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center right away.
Section 50567-7 (50567-7)
When using this product avoid contact with eyes
Purpose
External analgesic
Warnings
For external use only.
Flammable: Keep away from fire or flame
Directions
- adults and children 2 years of age and older: apply to affected area not more than 3 to 4 times daily
- children under 2 years of age: ask a doctor
Questions?
Call toll-free 800-299-4786 or 215-273-8755 (collect) or visit www.neutrogena.com
Active Ingredient (Active ingredient)
Camphor 0.45%
Other Information (Other information)
- Store at 20°C to 25°C (68°F to 77°F)
Inactive Ingredients (Inactive ingredients)
Water, Alcohol Denat., Triethanolamine, Carbomer, EDTA
Stop Use and Ask A Doctor If (Stop use and ask a doctor if)
- condition worsens
- symptoms persist for more than 7 days or clear up and occur again within a few days
Principal Display Panel 85 G Tube Label (PRINCIPAL DISPLAY PANEL - 85 g Tube Label)
NEW
Neutrogena®
DERMATOLOGIST RECOMMENDED BRAND
NEUTROGENA®
SUN
RESCUE
AFTER SUN
MEDICATED RELIEF GEL
CAMPHOR EXTERNAL ANALGESIC
pain relief for sunburn
cools, calms and soothes
skin on contact
NET WT 3.0 OZ (85 g)
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:06:29.088080 · Updated: 2026-03-14T23:09:16.713853