These Highlights Do Not Include All The Information Needed To Use Memantine Hcl Safely And Effectively. See Full Prescribing Information For Memantine Hcl.
c8f5c20b-4573-44c5-89fc-4da11b2e1fe1
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Memantine hydrochloride is indicated for the treatment of moderate to severe dementia of the Alzheimer's type.
Indications and Usage
Memantine hydrochloride is indicated for the treatment of moderate to severe dementia of the Alzheimer's type.
Dosage and Administration
The recommended starting dose of Memantine HCl is 5 mg once daily. The dose should be increased in 5 mg increments to 10 mg/day (5 mg twice daily), 15 mg/day (5 mg and 10 mg as separate doses), and 20 mg/day (10 mg twice daily). The minimum recommended interval between dose increases is one week. The dosage shown to be effective in controlled clinical trials is 20 mg/day. Memantine HCl can be taken with or without food. If a patient misses a single dose of Memantine HCl, that patient should not double up on the next dose. The next dose should be taken as scheduled. If a patient fails to take Memantine HCl for several days, dosing may need to be resumed at lower doses and retitrated as described above. Do not mix Memantine HCl oral solution with any other liquid. The oral solution is administered with a dosing device that comes with the drug and consists of a syringe, syringe adaptor cap, tubing and other supplies a patient needs to administer the drug. The supplied syringe should be used to withdraw the correct volume of oral solution and the oral solution should be slowly squirted into the corner of the patient's mouth.
Warnings and Precautions
Conditions that raise urine pH may decrease the urinary elimination of memantine, resulting in increased plasma levels of memantine. ( 5.1 , 7.1 )
Contraindications
Memantine hydrochloride is contraindicated in patients with known hypersensitivity to memantine hydrochloride or to any excipients used in the formulation.
Adverse Reactions
Most common adverse reactions (≥ 5 % and greater than placebo) are dizziness, headache, confusion and constipation. ( 6.1 ) To report SUSPECTED ADVERSE REACTIONS, contact Actavis at 1-800-272-5525 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch .
Storage and Handling
5 mg Tablet: Tan, capsule-shaped, film-coated tablets with “5” debossed on one side and FL on the other. Bottle of 60 NDC #0591-3870-60 10 x 10 Unit Dose NDC #0591-3870-44 10 mg Tablet: Gray, capsule-shaped, film-coated tablets with “10” debossed on one side and FL on the other. Bottle of 60 NDC #0591-3875-60 10 x 10 Unit Dose NDC #0591-3875-44 Titration Pack: NDC #0591-3900-87 Blister package containing 49 tablets (28 x 5 mg and 21 x 10 mg tablets). Oral Solution: 2 mg/mL Oral Solution (10 mg = 5 mL) 12 fl. oz. (360 mL) bottle NDC #0591-3011-07 Store Memantine HCl tablets and oral solution at 25°C (77°F); excursions permitted to 15-30°C (59-86°F) [see USP Controlled Room Temperature].
How Supplied
5 mg Tablet: Tan, capsule-shaped, film-coated tablets with “5” debossed on one side and FL on the other. Bottle of 60 NDC #0591-3870-60 10 x 10 Unit Dose NDC #0591-3870-44 10 mg Tablet: Gray, capsule-shaped, film-coated tablets with “10” debossed on one side and FL on the other. Bottle of 60 NDC #0591-3875-60 10 x 10 Unit Dose NDC #0591-3875-44 Titration Pack: NDC #0591-3900-87 Blister package containing 49 tablets (28 x 5 mg and 21 x 10 mg tablets). Oral Solution: 2 mg/mL Oral Solution (10 mg = 5 mL) 12 fl. oz. (360 mL) bottle NDC #0591-3011-07 Store Memantine HCl tablets and oral solution at 25°C (77°F); excursions permitted to 15-30°C (59-86°F) [see USP Controlled Room Temperature].
Medication Information
Warnings and Precautions
Conditions that raise urine pH may decrease the urinary elimination of memantine, resulting in increased plasma levels of memantine. ( 5.1 , 7.1 )
Indications and Usage
Memantine hydrochloride is indicated for the treatment of moderate to severe dementia of the Alzheimer's type.
Dosage and Administration
The recommended starting dose of Memantine HCl is 5 mg once daily. The dose should be increased in 5 mg increments to 10 mg/day (5 mg twice daily), 15 mg/day (5 mg and 10 mg as separate doses), and 20 mg/day (10 mg twice daily). The minimum recommended interval between dose increases is one week. The dosage shown to be effective in controlled clinical trials is 20 mg/day. Memantine HCl can be taken with or without food. If a patient misses a single dose of Memantine HCl, that patient should not double up on the next dose. The next dose should be taken as scheduled. If a patient fails to take Memantine HCl for several days, dosing may need to be resumed at lower doses and retitrated as described above. Do not mix Memantine HCl oral solution with any other liquid. The oral solution is administered with a dosing device that comes with the drug and consists of a syringe, syringe adaptor cap, tubing and other supplies a patient needs to administer the drug. The supplied syringe should be used to withdraw the correct volume of oral solution and the oral solution should be slowly squirted into the corner of the patient's mouth.
Contraindications
Memantine hydrochloride is contraindicated in patients with known hypersensitivity to memantine hydrochloride or to any excipients used in the formulation.
Adverse Reactions
Most common adverse reactions (≥ 5 % and greater than placebo) are dizziness, headache, confusion and constipation. ( 6.1 ) To report SUSPECTED ADVERSE REACTIONS, contact Actavis at 1-800-272-5525 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch .
Storage and Handling
5 mg Tablet: Tan, capsule-shaped, film-coated tablets with “5” debossed on one side and FL on the other. Bottle of 60 NDC #0591-3870-60 10 x 10 Unit Dose NDC #0591-3870-44 10 mg Tablet: Gray, capsule-shaped, film-coated tablets with “10” debossed on one side and FL on the other. Bottle of 60 NDC #0591-3875-60 10 x 10 Unit Dose NDC #0591-3875-44 Titration Pack: NDC #0591-3900-87 Blister package containing 49 tablets (28 x 5 mg and 21 x 10 mg tablets). Oral Solution: 2 mg/mL Oral Solution (10 mg = 5 mL) 12 fl. oz. (360 mL) bottle NDC #0591-3011-07 Store Memantine HCl tablets and oral solution at 25°C (77°F); excursions permitted to 15-30°C (59-86°F) [see USP Controlled Room Temperature].
How Supplied
5 mg Tablet: Tan, capsule-shaped, film-coated tablets with “5” debossed on one side and FL on the other. Bottle of 60 NDC #0591-3870-60 10 x 10 Unit Dose NDC #0591-3870-44 10 mg Tablet: Gray, capsule-shaped, film-coated tablets with “10” debossed on one side and FL on the other. Bottle of 60 NDC #0591-3875-60 10 x 10 Unit Dose NDC #0591-3875-44 Titration Pack: NDC #0591-3900-87 Blister package containing 49 tablets (28 x 5 mg and 21 x 10 mg tablets). Oral Solution: 2 mg/mL Oral Solution (10 mg = 5 mL) 12 fl. oz. (360 mL) bottle NDC #0591-3011-07 Store Memantine HCl tablets and oral solution at 25°C (77°F); excursions permitted to 15-30°C (59-86°F) [see USP Controlled Room Temperature].
Description
Memantine hydrochloride is indicated for the treatment of moderate to severe dementia of the Alzheimer's type.
Section 42229-5
Special Populations
Section 42230-3
Patient Information
Memantine hydrochloride
Tablets and Oral Solution
Read this Patient Information that comes with Memantine HCl before you start taking it and each time you get a refill. There may be new information. This information does not take the place of talking to your doctor about your medical condition or your treatment.
What is Memantine HCl?
Memantine HCl is a prescription medicine used for the treatment of moderate to severe dementia in people with Alzheimer's disease. Memantine HCl belongs to a class of medicines called NMDA (N-methyl-D-aspartate) inhibitors.
It is not known if Memantine HCl is safe and effective in children.
Who should not take Memantine HCl ?
Do not take Memantine HCl if you are allergic to memantine or any of the ingredients in Memantine HCl. See the end of this leaflet for a complete list of ingredients in Memantine HCl.
What should I tell my doctor before taking Memantine HCl?
Before you take Memantine HCl, tell your doctor if you:
- have or have had seizures
- have or have had problems passing urine
- have or have had bladder or kidney problems
- have liver problems
- have any other medical conditions
- are pregnant or plan to become pregnant. It is not known if Memantine HCl will harm your unborn baby.
- are breastfeeding or plan to breastfeed. It is not known if Memantine HCl passes into your breast milk. You and your doctor should decide if you will take Memantine HCl or breastfeed.
Tell your doctor about all the medicines you take, including prescription and non-prescription medicines, vitamins, and herbal supplements.
Taking Memantine HCl with certain other medicines may affect each other. Taking Memantine HCl with other medicines can cause serious side effects.
Especially tell your doctor if you take:
- other NMDA antagonists such as amantadine, ketamine, and dextromethorphan
- medicines that make your urine alkaline such as carbonic anhydrase inhibitors and sodium bicarbonate
Ask your doctor or pharmacist for a list of these medicines, if you are not sure.
Know the medicines you take. Keep a list of them to show your doctor and pharmacist when you get a new medicine.
How should I take Memantine HCl?
- See the step-by-step instructions for taking Memantine HCl Oral Solution at the end of this Patient Information.
- Your doctor will tell you how much Memantine HCl to take and when to take it.
- Your doctor may change your dose if needed.
- Memantine HCl can be taken with food or without food.
- Do not use any tablets of Memantine HCl that are damaged or show signs of tampering.
- If you forget to take one dose of Memantine HCl, do not double up on the next dose. You should take only the next dose as scheduled.
- If you have forgotten to take Memantine HCl for several days, you should not take the next dose until you talk to your doctor.
- If you take too much Memantine HCl, call your doctor or poison control center at 1-800-222-1222 right away, or go to the nearest hospital emergency room.
What are the possible side effects of Memantine HCl?
Memantine HCl may cause side effects, including:
The most common side effects of Memantine HCl include:
- dizziness
- headache
- confusion
- constipation
These are not all the possible side effects of Memantine HCl. For more information, ask your doctor or pharmacist.
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
How should I store Memantine HCl?
- Store Memantine HCl at room temperature between 59°F to 77°F (15°C to 30°C).
What are the ingredients in Memantine HCl?
Memantine HCl tablets:
Active ingredients: memantine hydrochloride
Inactive ingredients: microcrystalline cellulose/colloidal silicon dioxide, talc, croscarmellose sodium, and magnesium stearate
Inactive ingredients of tablet film coating: hypromellose, titanium dioxide, polyethylene glycol 400, FD&C yellow #6 and FD&C blue #2 (5 mg tablets), and hypromellose, titanium dioxide, macrogol/polyethylene glycol 400 and iron oxide black (10 mg tablets)
Memantine HCl oral solution:
Active ingredients: memantine hydrochloride
Inactive ingredients: sorbitol solution (70%), methyl paraben, propylparaben, propylene glycol, glycerin, natural peppermint flavor #104, citric acid, sodium citrate, and purified water
Keep Memantine HCl and all medicines out of the reach of children.
General information about the safe and effective use of Memantine HCl.
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not take Memantine HCl for a condition for which it was not prescribed. Do not give Memantine HCl to other people, even if they have the same condition. It may harm them.
This Patient Information leaflet summarizes the most important information about Memantine HCl. If you would like more information, talk with your doctor. You can ask your doctor or pharmacist for information about Memantine HCl that was written for healthcare professionals.
For more information about Memantine HCl call Actavis at 1-800-272-5525.
Section 59845-8
INSTRUCTIONS FOR USE
Memantine hydrochloride
Oral Solution
Directions for Using your Memantine HCl Oral Solution
Read these instructions before taking Memantine HCl Oral Solution and each time you get a refill. There may be new information. This information does not take the place of talking to your doctor about your medical condition or your treatment.
Preparing your dose of Memantine HCl Oral Solution.
You will need the following supplies:
- Memantine HCl Oral Solution bottle with Child-resistant cap
- Green syringe adaptor cap with lid
- Oral dosing syringe
- Plastic tube (in plastic bag)
- Prescribing Information
|
|
1. Remove the oral dosing syringe, the green syringe adaptor cap, and remove the plastic tube from its protective plastic bag. Attach the tube to the green syringe adaptor cap if it is not already attached. |
|
|
2. The bottle comes with a child-resistant cap. To remove the cap, you should push down on the cap and at the same time; turn the cap counter-clockwise (to the left). |
|
|
3. Carefully remove the seal from the bottle and throw away. |
|
|
4. Insert the green syringe adaptor cap, with the attached tube, all the way into the bottle and tightly screw the cap onto the bottle by turning the cap clockwise (to the right). |
| 5. The green syringe adaptor cap has an opening with an attached lid. The adaptor is used to withdraw the correct dose of medicine from the bottle with the syringe. The attached adaptor lid should be closed in between doses. |
|
6. Keep the bottle upright on a table. Open the syringe adaptor lid and insert the tip of syringe into the syringe adaptor opening.
|
|
7. While holding the syringe in place, gently pull the plunger of the syringe until you get to the correct mL (amount) of medicine you need.
|
|
| 8. Remove the syringe from the syringe adaptor cap. | |
|
|
9. Remove the syringe from the bottle and slowly squirt the Memantine HCl Oral Solution into the corner of you or the patient's mouth. Do not mix Memantine HCl Oral Solution with any other liquid. |
| 10. After use, reseal the bottle by snapping the attached syringe adaptor lid closed. | |
| 11. Rinse the empty syringe by inserting the open end of the syringe into a glass of water, pulling the plunger out to draw in water, and pushing the plunger in to remove the water. Repeat several times. Allow the syringe to air dry. | |
|
12. Store the bottle upright. |
This Patient Information and Instructions for Use have been approved by the U.S. Food and Drug Administration.
Distributed by:
Actavis Pharma, Inc.
Parsippany, NJ 07054 USA
Manufactured by:
Forest Laboratories Ireland Ltd
Clonshaugh Business and Technology Park
Clonshaugh, Dublin 17, Ireland
Licensed from Merz Pharmaceuticals GmbH
© 2005-2015 Actavis
Revised April 2015
10 Overdosage
Signs and symptoms most often accompanying memantine overdosage in clinical trials and from worldwide marketing experience, alone or in combination with other drugs and/or alcohol, include agitation, asthenia, bradycardia, confusion, coma, dizziness, ECG changes, increased blood pressure, lethargy, loss of consciousness, psychosis, restlessness, slowed movement, somnolence, stupor, unsteady gait, visual hallucinations, vertigo, vomiting, and weakness. The largest known ingestion of memantine worldwide was 2.0 grams in a patient who took memantine in conjunction with unspecified antidiabetic medications. The patient experienced coma, diplopia, and agitation, but subsequently recovered. Fatal outcome has been very rarely reported with memantine, and the relationship to memantine was unclear.
Because strategies for the management of overdose are continually evolving, it is advisable to contact a poison control center to determine the latest recommendations for the management of an overdose of any drug. As in any cases of overdose, general supportive measures should be utilized, and treatment should be symptomatic. Elimination of memantine can be enhanced by acidification of urine.
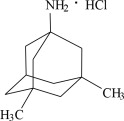
11 Description
Memantine hydrochloride is an orally active NMDA receptor antagonist. The chemical name for memantine hydrochloride is 1-amino-3,5-dimethyladamantane hydrochloride with the following structural formula:
The molecular formula is C12H21N•HCl and the molecular weight is 215.76. Memantine HCl occurs as a fine white to off-white powder and is soluble in water.
Memantine HCl is available as tablets or as an oral solution. Memantine HCl is available for oral administration as capsule-shaped, film-coated tablets containing 5 mg and 10 mg of memantine hydrochloride. The tablets also contain the following inactive ingredients: microcrystalline cellulose/colloidal silicon dioxide, talc, croscarmellose sodium, and magnesium stearate. In addition the following inactive ingredients are also present as components of the film coat: hypromellose, titanium dioxide, polyethylene glycol 400, FD&C yellow #6 and FD&C blue #2 (5 mg tablets), and hypromellose, titanium dioxide, macrogol/polyethylene glycol 400 and iron oxide black (10 mg tablets). Memantine HCl oral solution contains memantine hydrochloride in a strength equivalent to 2 mg of memantine hydrochloride in each mL. The oral solution also contains the following inactive ingredients: sorbitol solution (70%), methylparaben, propylparaben, propylene glycol, glycerin, natural peppermint flavor #104, citric acid, sodium citrate, and purified water.
8.4 Pediatric Use
Safety and effectiveness in pediatric patients have not been established.
8.5 Geriatric Use
The majority of people with Alzheimer's disease are 65 years and older. In the clinical studies of Memantine HCl the mean age of patients was approximately 76; over 90% of patients were 65 years and older, 60% were 75 years and older, and 12% were at or above 85 years of age. The efficacy and safety data presented in the clinical trial sections were obtained from these patients. There were no clinically meaningful differences in most adverse events reported by patient groups ≥65 years old and <65 year old.
14 Clinical Studies
The effectiveness of Memantine HCl as a treatment for patients with moderate to severe Alzheimer's disease was demonstrated in 2 randomized, double-blind, placebo-controlled clinical studies (Studies 1 and 2) conducted in the United States that assessed both cognitive function and day to day function. The mean age of patients participating in these two trials was 76 with a range of 50-93 years. Approximately 66% of patients were female and 91% of patients were Caucasian. A third study (Study 3), carried out in Latvia, enrolled patients with severe dementia, but did not assess cognitive function as a planned endpoint. Study Outcome Measures: In each U.S. study, the effectiveness of Memantine HCl was determined using both an instrument designed to evaluate overall function through caregiver-related assessment, and an instrument that measures cognition. Both studies showed that patients on Memantine HCl experienced significant improvement on both measures compared to placebo.
Day-to-day function was assessed in both studies using the modified Alzheimer's disease Cooperative Study - Activities of Daily Living inventory (ADCS-ADL). The ADCS-ADL consists of a comprehensive battery of ADL questions used to measure the functional capabilities of patients. Each ADL item is rated from the highest level of independent performance to complete loss. The investigator performs the inventory by interviewing a caregiver familiar with the behavior of the patient. A subset of 19 items, including ratings of the patient's ability to eat, dress, bathe, telephone, travel, shop, and perform other household chores has been validated for the assessment of patients with moderate to severe dementia. This is the modified ADCS-ADL, which has a scoring range of 0 to 54, with the lower scores indicating greater functional impairment.
The ability of Memantine HCl to improve cognitive performance was assessed in both studies with the Severe Impairment Battery (SIB), a multi-item instrument that has been validated for the evaluation of cognitive function in patients with moderate to severe dementia. The SIB examines selected aspects of cognitive performance, including elements of attention, orientation, language, memory, visuospatial ability, construction, praxis, and social interaction. The SIB scoring range is from 0 to 100, with lower scores indicating greater cognitive impairment.
4 Contraindications
Memantine hydrochloride is contraindicated in patients with known hypersensitivity to memantine hydrochloride or to any excipients used in the formulation.
6 Adverse Reactions
Most common adverse reactions (≥ 5 % and greater than placebo) are dizziness, headache, confusion and constipation. (6.1)
To report SUSPECTED ADVERSE REACTIONS, contact Actavis at 1-800-272-5525 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch .
8.3 Nursing Mothers
It is not known whether this drug is excreted in human milk. Because many drugs are excreted in human milk, caution should be exercised when Memantine HCl is administered to a nursing mother.
8.6 Renal Impairment
12.2 Pharmacodynamics
Memantine showed low to negligible affinity for GABA, benzodiazepine, dopamine, adrenergic, histamine and glycine receptors and for voltage-dependent Ca2+, Na+ or K+ channels. Memantine also showed antagonistic effects at the 5HT3 receptor with a potency similar to that for the NMDA receptor and blocked nicotinic acetylcholine receptors with one-sixth to one-tenth the potency.
In vitro studies have shown that memantine does not affect the reversible inhibition of acetylcholinesterase by donepezil, galantamine, or tacrine.
8.7 Hepatic Impairment
1 Indications and Usage
Memantine hydrochloride is indicated for the treatment of moderate to severe dementia of the Alzheimer's type.
Repackaging Information
Please reference the How Supplied section listed above for a description of individual tablets. This drug product has been received by Aphena Pharma - TN in a manufacturer or distributor packaged configuration and repackaged in full compliance with all applicable cGMP regulations. The package configurations available from Aphena are listed below:
| Count | 10 mg |
| 3000 | 71610-011-18 |
Store between 20°-25°C (68°-77°F). See USP Controlled Room Temperature. Dispense in a tight light-resistant container as defined by USP. Keep this and all drugs out of the reach of children.
Repackaged by:
Cookeville, TN 38506
20171130JH
12.1 Mechanism of Action
Persistent activation of central nervous system N-methyl-D-aspartate (NMDA) receptors by the excitatory amino acid glutamate has been hypothesized to contribute to the symptomatology of Alzheimer's disease. Memantine is postulated to exert its therapeutic effect through its action as a low to moderate affinity uncompetitive (open-channel) NMDA receptor antagonist which binds preferentially to the NMDA receptor-operated cation channels. There is no evidence that memantine prevents or slows neurodegeneration in patients with Alzheimer's disease.
5 Warnings and Precautions
2 Dosage and Administration
The recommended starting dose of Memantine HCl is 5 mg once daily. The dose should be increased in 5 mg increments to 10 mg/day (5 mg twice daily), 15 mg/day (5 mg and 10 mg as separate doses), and 20 mg/day (10 mg twice daily). The minimum recommended interval between dose increases is one week. The dosage shown to be effective in controlled clinical trials is 20 mg/day.
Memantine HCl can be taken with or without food. If a patient misses a single dose of Memantine HCl, that patient should not double up on the next dose. The next dose should be taken as scheduled.
If a patient fails to take Memantine HCl for several days, dosing may need to be resumed at lower doses and retitrated as described above.
Do not mix Memantine HCl oral solution with any other liquid. The oral solution is administered with a dosing device that comes with the drug and consists of a syringe, syringe adaptor cap, tubing and other supplies a patient needs to administer the drug. The supplied syringe should be used to withdraw the correct volume of oral solution and the oral solution should be slowly squirted into the corner of the patient's mouth.
3 Dosage Forms and Strengths
Memantine HCl 5 mg tablet: capsule-shaped, film-coated tablets are tan, with the strength (5) debossed on one side and FL on the other.
Memantine HCl 10 mg tablet: capsule-shaped, film-coated tablets are gray, with the strength (10) debossed on one side and FL on the other.
Memantine HCl 2 mg/mL oral solution: clear, alcohol-free, sugar-free, and peppermint flavored.
5.1 Genitourinary Conditions
Conditions that raise urine pH may decrease the urinary elimination of memantine resulting in increased plasma levels of memantine [see Drug Interactions (7.1)].
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of memantine.
Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure. These reactions include:
Blood and Lymphatic System Disorders - agranulocytosis, leukopenia (including neutropenia), pancytopenia, thrombocytopenia, thrombotic thrombocytopenic purpura.
Cardiac Disorders - cardiac failure congestive.
Gastrointestinal Disorders - pancreatitis.
Hepatobiliary Disorders – hepatitis.
Psychiatric Disorders - suicidal ideation.
Renal and Urinary Disorders - acute renal failure (including increased creatinine and renal insufficiency).
Skin Disorders - Stevens Johnson syndrome.
6.1 Clinical Trials Experience
Memantine HCl was evaluated in eight double-blind placebo-controlled trials involving a total of 1862 dementia (Alzheimer's disease, vascular dementia) patients (940 patients treated with Memantine HCl and 922 patients treated with placebo) for a treatment period up to 28 weeks.
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
Principal Display Panel 10mg
NDC 71610-011 - Memantine HCl 10mg - Rx Only
17 Patient Counseling Information
See FDA-approved patient labeling (Patient Information and Instructions for Use).
To assure safe and effective use of Memantine HCl, the following information and instructions provided in the patient information section should be discussed with patients and caregivers.
Patients/caregivers should be instructed to follow the dose titration schedule provided by their physician or healthcare professional for Memantine HCl. They should be warned not to use any tablets of Memantine HCl that are damaged or show signs of tampering.
If a patient misses a single dose of Memantine HCl, that patient should not double up on the next dose. The next dose should be taken as scheduled. If a patient fails to take Memantine HCl for several days, dosing should not be resumed without consulting that patient's healthcare professional.
Patients/caregivers should be instructed on how to use the Memantine HCl Oral Solution dosing device. They should be made aware of the patient instruction sheet that is enclosed with the product. Patients/caregivers should be instructed to address any questions on the usage of the solution to their physician or pharmacist.
Distributed by:
Actavis Pharma, Inc.
Parsippany, NJ 07054 USA
Manufactured by:
Forest Laboratories Ireland Ltd
Clonshaugh Business and Technology Park
Clonshaugh, Dublin 17, Ireland
Licensed from Merz Pharmaceuticals GmbH
© 2005-2015 Actavis
16 How Supplied/storage and Handling
5 mg Tablet:
Tan, capsule-shaped, film-coated tablets with “5” debossed on one side and FL on the other.
Bottle of 60 NDC #0591-3870-60
10 x 10 Unit Dose NDC #0591-3870-44
10 mg Tablet:
Gray, capsule-shaped, film-coated tablets with “10” debossed on one side and FL on the other.
Bottle of 60 NDC #0591-3875-60
10 x 10 Unit Dose NDC #0591-3875-44
Titration Pack: NDC #0591-3900-87
Blister package containing 49 tablets (28 x 5 mg and 21 x 10 mg tablets).
Oral Solution:
2 mg/mL Oral Solution (10 mg = 5 mL)
12 fl. oz. (360 mL) bottle NDC #0591-3011-07
Store Memantine HCl tablets and oral solution at 25°C (77°F); excursions permitted to 15-30°C (59-86°F) [see USP Controlled Room Temperature].
7.1 Drugs That Make the Urine Alkaline
The clearance of memantine was reduced by about 80% under alkaline urine conditions at pH 8. Therefore, alterations of urine pH towards the alkaline condition may lead to an accumulation of the drug with a possible increase in adverse effects. Urine pH is altered by diet, drugs (e.g. carbonic anhydrase inhibitors, sodium bicarbonate) and clinical state of the patient (e.g. renal tubular acidosis or severe infections of the urinary tract). Hence, memantine should be used with caution under these conditions.
13.2 Animal Toxicology And/or Pharmacology
Memantine induced neuronal lesions (vacuolation and necrosis) in the multipolar and pyramidal cells in cortical layers III and IV of the posterior cingulate and retrosplenial neocortices in rats, similar to those which are known to occur in rodents administered other NMDA receptor antagonists. Lesions were seen after a single dose of memantine. In a study in which rats were given daily oral doses of memantine for 14 days, the no-effect dose for neuronal necrosis was 6 times the maximum recommended human dose of 20 mg/day on a mg/m2 basis
In acute and repeat-dose neurotoxicity studies in female rats, oral administration of memantine and donepezil in combination resulted in increased incidence, severity, and distribution of neurodegeneration compared with memantine alone. The no-effect levels of the combination were associated with clinically relevant plasma memantine and donepezil exposures.
The relevance of these findings to humans is unknown.
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
There was no evidence of carcinogenicity in a 113-week oral study in mice at doses up to 40 mg/kg/day (10 times the maximum recommended human dose [MRHD] on a mg/m2 basis). There was also no evidence of carcinogenicity in rats orally dosed at up to 40 mg/kg/day for 71 weeks followed by 20 mg/kg/day (20 and 10 times the MRHD on a mg/m2 basis, respectively) through 128 weeks.
Memantine produced no evidence of genotoxic potential when evaluated in the in vitro S. typhimurium or E. coli reverse mutation assay, an in vitro chromosomal aberration test in human lymphocytes, an in vivo cytogenetics assay for chromosome damage in rats, and the in vivo mouse micronucleus assay. The results were equivocal in an in vitro gene mutation assay using Chinese hamster V79 cells.
No impairment of fertility or reproductive performance was seen in rats administered up to 18 mg/kg/day (9 times the MRHD on a mg/m2 basis) orally from 14 days prior to mating through gestation and lactation in females, or for 60 days prior to mating in males.
7.2 Use With Other N Methyl D Aspartate (nmda) Antagonists
The combined use of Memantine HCl with other NMDA antagonists (amantadine, ketamine, and dextromethorphan) has not been systematically evaluated and such use should be approached with caution.
Structured Label Content
Section 42229-5 (42229-5)
Special Populations
Section 42230-3 (42230-3)
Patient Information
Memantine hydrochloride
Tablets and Oral Solution
Read this Patient Information that comes with Memantine HCl before you start taking it and each time you get a refill. There may be new information. This information does not take the place of talking to your doctor about your medical condition or your treatment.
What is Memantine HCl?
Memantine HCl is a prescription medicine used for the treatment of moderate to severe dementia in people with Alzheimer's disease. Memantine HCl belongs to a class of medicines called NMDA (N-methyl-D-aspartate) inhibitors.
It is not known if Memantine HCl is safe and effective in children.
Who should not take Memantine HCl ?
Do not take Memantine HCl if you are allergic to memantine or any of the ingredients in Memantine HCl. See the end of this leaflet for a complete list of ingredients in Memantine HCl.
What should I tell my doctor before taking Memantine HCl?
Before you take Memantine HCl, tell your doctor if you:
- have or have had seizures
- have or have had problems passing urine
- have or have had bladder or kidney problems
- have liver problems
- have any other medical conditions
- are pregnant or plan to become pregnant. It is not known if Memantine HCl will harm your unborn baby.
- are breastfeeding or plan to breastfeed. It is not known if Memantine HCl passes into your breast milk. You and your doctor should decide if you will take Memantine HCl or breastfeed.
Tell your doctor about all the medicines you take, including prescription and non-prescription medicines, vitamins, and herbal supplements.
Taking Memantine HCl with certain other medicines may affect each other. Taking Memantine HCl with other medicines can cause serious side effects.
Especially tell your doctor if you take:
- other NMDA antagonists such as amantadine, ketamine, and dextromethorphan
- medicines that make your urine alkaline such as carbonic anhydrase inhibitors and sodium bicarbonate
Ask your doctor or pharmacist for a list of these medicines, if you are not sure.
Know the medicines you take. Keep a list of them to show your doctor and pharmacist when you get a new medicine.
How should I take Memantine HCl?
- See the step-by-step instructions for taking Memantine HCl Oral Solution at the end of this Patient Information.
- Your doctor will tell you how much Memantine HCl to take and when to take it.
- Your doctor may change your dose if needed.
- Memantine HCl can be taken with food or without food.
- Do not use any tablets of Memantine HCl that are damaged or show signs of tampering.
- If you forget to take one dose of Memantine HCl, do not double up on the next dose. You should take only the next dose as scheduled.
- If you have forgotten to take Memantine HCl for several days, you should not take the next dose until you talk to your doctor.
- If you take too much Memantine HCl, call your doctor or poison control center at 1-800-222-1222 right away, or go to the nearest hospital emergency room.
What are the possible side effects of Memantine HCl?
Memantine HCl may cause side effects, including:
The most common side effects of Memantine HCl include:
- dizziness
- headache
- confusion
- constipation
These are not all the possible side effects of Memantine HCl. For more information, ask your doctor or pharmacist.
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
How should I store Memantine HCl?
- Store Memantine HCl at room temperature between 59°F to 77°F (15°C to 30°C).
What are the ingredients in Memantine HCl?
Memantine HCl tablets:
Active ingredients: memantine hydrochloride
Inactive ingredients: microcrystalline cellulose/colloidal silicon dioxide, talc, croscarmellose sodium, and magnesium stearate
Inactive ingredients of tablet film coating: hypromellose, titanium dioxide, polyethylene glycol 400, FD&C yellow #6 and FD&C blue #2 (5 mg tablets), and hypromellose, titanium dioxide, macrogol/polyethylene glycol 400 and iron oxide black (10 mg tablets)
Memantine HCl oral solution:
Active ingredients: memantine hydrochloride
Inactive ingredients: sorbitol solution (70%), methyl paraben, propylparaben, propylene glycol, glycerin, natural peppermint flavor #104, citric acid, sodium citrate, and purified water
Keep Memantine HCl and all medicines out of the reach of children.
General information about the safe and effective use of Memantine HCl.
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not take Memantine HCl for a condition for which it was not prescribed. Do not give Memantine HCl to other people, even if they have the same condition. It may harm them.
This Patient Information leaflet summarizes the most important information about Memantine HCl. If you would like more information, talk with your doctor. You can ask your doctor or pharmacist for information about Memantine HCl that was written for healthcare professionals.
For more information about Memantine HCl call Actavis at 1-800-272-5525.
Section 59845-8 (59845-8)
INSTRUCTIONS FOR USE
Memantine hydrochloride
Oral Solution
Directions for Using your Memantine HCl Oral Solution
Read these instructions before taking Memantine HCl Oral Solution and each time you get a refill. There may be new information. This information does not take the place of talking to your doctor about your medical condition or your treatment.
Preparing your dose of Memantine HCl Oral Solution.
You will need the following supplies:
- Memantine HCl Oral Solution bottle with Child-resistant cap
- Green syringe adaptor cap with lid
- Oral dosing syringe
- Plastic tube (in plastic bag)
- Prescribing Information
|
|
1. Remove the oral dosing syringe, the green syringe adaptor cap, and remove the plastic tube from its protective plastic bag. Attach the tube to the green syringe adaptor cap if it is not already attached. |
|
|
2. The bottle comes with a child-resistant cap. To remove the cap, you should push down on the cap and at the same time; turn the cap counter-clockwise (to the left). |
|
|
3. Carefully remove the seal from the bottle and throw away. |
|
|
4. Insert the green syringe adaptor cap, with the attached tube, all the way into the bottle and tightly screw the cap onto the bottle by turning the cap clockwise (to the right). |
| 5. The green syringe adaptor cap has an opening with an attached lid. The adaptor is used to withdraw the correct dose of medicine from the bottle with the syringe. The attached adaptor lid should be closed in between doses. |
|
6. Keep the bottle upright on a table. Open the syringe adaptor lid and insert the tip of syringe into the syringe adaptor opening.
|
|
7. While holding the syringe in place, gently pull the plunger of the syringe until you get to the correct mL (amount) of medicine you need.
|
|
| 8. Remove the syringe from the syringe adaptor cap. | |
|
|
9. Remove the syringe from the bottle and slowly squirt the Memantine HCl Oral Solution into the corner of you or the patient's mouth. Do not mix Memantine HCl Oral Solution with any other liquid. |
| 10. After use, reseal the bottle by snapping the attached syringe adaptor lid closed. | |
| 11. Rinse the empty syringe by inserting the open end of the syringe into a glass of water, pulling the plunger out to draw in water, and pushing the plunger in to remove the water. Repeat several times. Allow the syringe to air dry. | |
|
12. Store the bottle upright. |
This Patient Information and Instructions for Use have been approved by the U.S. Food and Drug Administration.
Distributed by:
Actavis Pharma, Inc.
Parsippany, NJ 07054 USA
Manufactured by:
Forest Laboratories Ireland Ltd
Clonshaugh Business and Technology Park
Clonshaugh, Dublin 17, Ireland
Licensed from Merz Pharmaceuticals GmbH
© 2005-2015 Actavis
Revised April 2015
10 Overdosage (10 OVERDOSAGE)
Signs and symptoms most often accompanying memantine overdosage in clinical trials and from worldwide marketing experience, alone or in combination with other drugs and/or alcohol, include agitation, asthenia, bradycardia, confusion, coma, dizziness, ECG changes, increased blood pressure, lethargy, loss of consciousness, psychosis, restlessness, slowed movement, somnolence, stupor, unsteady gait, visual hallucinations, vertigo, vomiting, and weakness. The largest known ingestion of memantine worldwide was 2.0 grams in a patient who took memantine in conjunction with unspecified antidiabetic medications. The patient experienced coma, diplopia, and agitation, but subsequently recovered. Fatal outcome has been very rarely reported with memantine, and the relationship to memantine was unclear.
Because strategies for the management of overdose are continually evolving, it is advisable to contact a poison control center to determine the latest recommendations for the management of an overdose of any drug. As in any cases of overdose, general supportive measures should be utilized, and treatment should be symptomatic. Elimination of memantine can be enhanced by acidification of urine.
11 Description (11 DESCRIPTION)
Memantine hydrochloride is an orally active NMDA receptor antagonist. The chemical name for memantine hydrochloride is 1-amino-3,5-dimethyladamantane hydrochloride with the following structural formula:
The molecular formula is C12H21N•HCl and the molecular weight is 215.76. Memantine HCl occurs as a fine white to off-white powder and is soluble in water.
Memantine HCl is available as tablets or as an oral solution. Memantine HCl is available for oral administration as capsule-shaped, film-coated tablets containing 5 mg and 10 mg of memantine hydrochloride. The tablets also contain the following inactive ingredients: microcrystalline cellulose/colloidal silicon dioxide, talc, croscarmellose sodium, and magnesium stearate. In addition the following inactive ingredients are also present as components of the film coat: hypromellose, titanium dioxide, polyethylene glycol 400, FD&C yellow #6 and FD&C blue #2 (5 mg tablets), and hypromellose, titanium dioxide, macrogol/polyethylene glycol 400 and iron oxide black (10 mg tablets). Memantine HCl oral solution contains memantine hydrochloride in a strength equivalent to 2 mg of memantine hydrochloride in each mL. The oral solution also contains the following inactive ingredients: sorbitol solution (70%), methylparaben, propylparaben, propylene glycol, glycerin, natural peppermint flavor #104, citric acid, sodium citrate, and purified water.
8.4 Pediatric Use
Safety and effectiveness in pediatric patients have not been established.
8.5 Geriatric Use
The majority of people with Alzheimer's disease are 65 years and older. In the clinical studies of Memantine HCl the mean age of patients was approximately 76; over 90% of patients were 65 years and older, 60% were 75 years and older, and 12% were at or above 85 years of age. The efficacy and safety data presented in the clinical trial sections were obtained from these patients. There were no clinically meaningful differences in most adverse events reported by patient groups ≥65 years old and <65 year old.
14 Clinical Studies (14 CLINICAL STUDIES)
The effectiveness of Memantine HCl as a treatment for patients with moderate to severe Alzheimer's disease was demonstrated in 2 randomized, double-blind, placebo-controlled clinical studies (Studies 1 and 2) conducted in the United States that assessed both cognitive function and day to day function. The mean age of patients participating in these two trials was 76 with a range of 50-93 years. Approximately 66% of patients were female and 91% of patients were Caucasian. A third study (Study 3), carried out in Latvia, enrolled patients with severe dementia, but did not assess cognitive function as a planned endpoint. Study Outcome Measures: In each U.S. study, the effectiveness of Memantine HCl was determined using both an instrument designed to evaluate overall function through caregiver-related assessment, and an instrument that measures cognition. Both studies showed that patients on Memantine HCl experienced significant improvement on both measures compared to placebo.
Day-to-day function was assessed in both studies using the modified Alzheimer's disease Cooperative Study - Activities of Daily Living inventory (ADCS-ADL). The ADCS-ADL consists of a comprehensive battery of ADL questions used to measure the functional capabilities of patients. Each ADL item is rated from the highest level of independent performance to complete loss. The investigator performs the inventory by interviewing a caregiver familiar with the behavior of the patient. A subset of 19 items, including ratings of the patient's ability to eat, dress, bathe, telephone, travel, shop, and perform other household chores has been validated for the assessment of patients with moderate to severe dementia. This is the modified ADCS-ADL, which has a scoring range of 0 to 54, with the lower scores indicating greater functional impairment.
The ability of Memantine HCl to improve cognitive performance was assessed in both studies with the Severe Impairment Battery (SIB), a multi-item instrument that has been validated for the evaluation of cognitive function in patients with moderate to severe dementia. The SIB examines selected aspects of cognitive performance, including elements of attention, orientation, language, memory, visuospatial ability, construction, praxis, and social interaction. The SIB scoring range is from 0 to 100, with lower scores indicating greater cognitive impairment.
4 Contraindications (4 CONTRAINDICATIONS)
Memantine hydrochloride is contraindicated in patients with known hypersensitivity to memantine hydrochloride or to any excipients used in the formulation.
6 Adverse Reactions (6 ADVERSE REACTIONS)
Most common adverse reactions (≥ 5 % and greater than placebo) are dizziness, headache, confusion and constipation. (6.1)
To report SUSPECTED ADVERSE REACTIONS, contact Actavis at 1-800-272-5525 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch .
8.3 Nursing Mothers
It is not known whether this drug is excreted in human milk. Because many drugs are excreted in human milk, caution should be exercised when Memantine HCl is administered to a nursing mother.
8.6 Renal Impairment
12.2 Pharmacodynamics
Memantine showed low to negligible affinity for GABA, benzodiazepine, dopamine, adrenergic, histamine and glycine receptors and for voltage-dependent Ca2+, Na+ or K+ channels. Memantine also showed antagonistic effects at the 5HT3 receptor with a potency similar to that for the NMDA receptor and blocked nicotinic acetylcholine receptors with one-sixth to one-tenth the potency.
In vitro studies have shown that memantine does not affect the reversible inhibition of acetylcholinesterase by donepezil, galantamine, or tacrine.
8.7 Hepatic Impairment
1 Indications and Usage (1 INDICATIONS AND USAGE)
Memantine hydrochloride is indicated for the treatment of moderate to severe dementia of the Alzheimer's type.
Repackaging Information
Please reference the How Supplied section listed above for a description of individual tablets. This drug product has been received by Aphena Pharma - TN in a manufacturer or distributor packaged configuration and repackaged in full compliance with all applicable cGMP regulations. The package configurations available from Aphena are listed below:
| Count | 10 mg |
| 3000 | 71610-011-18 |
Store between 20°-25°C (68°-77°F). See USP Controlled Room Temperature. Dispense in a tight light-resistant container as defined by USP. Keep this and all drugs out of the reach of children.
Repackaged by:
Cookeville, TN 38506
20171130JH
12.1 Mechanism of Action
Persistent activation of central nervous system N-methyl-D-aspartate (NMDA) receptors by the excitatory amino acid glutamate has been hypothesized to contribute to the symptomatology of Alzheimer's disease. Memantine is postulated to exert its therapeutic effect through its action as a low to moderate affinity uncompetitive (open-channel) NMDA receptor antagonist which binds preferentially to the NMDA receptor-operated cation channels. There is no evidence that memantine prevents or slows neurodegeneration in patients with Alzheimer's disease.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
The recommended starting dose of Memantine HCl is 5 mg once daily. The dose should be increased in 5 mg increments to 10 mg/day (5 mg twice daily), 15 mg/day (5 mg and 10 mg as separate doses), and 20 mg/day (10 mg twice daily). The minimum recommended interval between dose increases is one week. The dosage shown to be effective in controlled clinical trials is 20 mg/day.
Memantine HCl can be taken with or without food. If a patient misses a single dose of Memantine HCl, that patient should not double up on the next dose. The next dose should be taken as scheduled.
If a patient fails to take Memantine HCl for several days, dosing may need to be resumed at lower doses and retitrated as described above.
Do not mix Memantine HCl oral solution with any other liquid. The oral solution is administered with a dosing device that comes with the drug and consists of a syringe, syringe adaptor cap, tubing and other supplies a patient needs to administer the drug. The supplied syringe should be used to withdraw the correct volume of oral solution and the oral solution should be slowly squirted into the corner of the patient's mouth.
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Memantine HCl 5 mg tablet: capsule-shaped, film-coated tablets are tan, with the strength (5) debossed on one side and FL on the other.
Memantine HCl 10 mg tablet: capsule-shaped, film-coated tablets are gray, with the strength (10) debossed on one side and FL on the other.
Memantine HCl 2 mg/mL oral solution: clear, alcohol-free, sugar-free, and peppermint flavored.
5.1 Genitourinary Conditions
Conditions that raise urine pH may decrease the urinary elimination of memantine resulting in increased plasma levels of memantine [see Drug Interactions (7.1)].
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of memantine.
Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure. These reactions include:
Blood and Lymphatic System Disorders - agranulocytosis, leukopenia (including neutropenia), pancytopenia, thrombocytopenia, thrombotic thrombocytopenic purpura.
Cardiac Disorders - cardiac failure congestive.
Gastrointestinal Disorders - pancreatitis.
Hepatobiliary Disorders – hepatitis.
Psychiatric Disorders - suicidal ideation.
Renal and Urinary Disorders - acute renal failure (including increased creatinine and renal insufficiency).
Skin Disorders - Stevens Johnson syndrome.
6.1 Clinical Trials Experience
Memantine HCl was evaluated in eight double-blind placebo-controlled trials involving a total of 1862 dementia (Alzheimer's disease, vascular dementia) patients (940 patients treated with Memantine HCl and 922 patients treated with placebo) for a treatment period up to 28 weeks.
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
Principal Display Panel 10mg (PRINCIPAL DISPLAY PANEL - 10mg)
NDC 71610-011 - Memantine HCl 10mg - Rx Only
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
See FDA-approved patient labeling (Patient Information and Instructions for Use).
To assure safe and effective use of Memantine HCl, the following information and instructions provided in the patient information section should be discussed with patients and caregivers.
Patients/caregivers should be instructed to follow the dose titration schedule provided by their physician or healthcare professional for Memantine HCl. They should be warned not to use any tablets of Memantine HCl that are damaged or show signs of tampering.
If a patient misses a single dose of Memantine HCl, that patient should not double up on the next dose. The next dose should be taken as scheduled. If a patient fails to take Memantine HCl for several days, dosing should not be resumed without consulting that patient's healthcare professional.
Patients/caregivers should be instructed on how to use the Memantine HCl Oral Solution dosing device. They should be made aware of the patient instruction sheet that is enclosed with the product. Patients/caregivers should be instructed to address any questions on the usage of the solution to their physician or pharmacist.
Distributed by:
Actavis Pharma, Inc.
Parsippany, NJ 07054 USA
Manufactured by:
Forest Laboratories Ireland Ltd
Clonshaugh Business and Technology Park
Clonshaugh, Dublin 17, Ireland
Licensed from Merz Pharmaceuticals GmbH
© 2005-2015 Actavis
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
5 mg Tablet:
Tan, capsule-shaped, film-coated tablets with “5” debossed on one side and FL on the other.
Bottle of 60 NDC #0591-3870-60
10 x 10 Unit Dose NDC #0591-3870-44
10 mg Tablet:
Gray, capsule-shaped, film-coated tablets with “10” debossed on one side and FL on the other.
Bottle of 60 NDC #0591-3875-60
10 x 10 Unit Dose NDC #0591-3875-44
Titration Pack: NDC #0591-3900-87
Blister package containing 49 tablets (28 x 5 mg and 21 x 10 mg tablets).
Oral Solution:
2 mg/mL Oral Solution (10 mg = 5 mL)
12 fl. oz. (360 mL) bottle NDC #0591-3011-07
Store Memantine HCl tablets and oral solution at 25°C (77°F); excursions permitted to 15-30°C (59-86°F) [see USP Controlled Room Temperature].
7.1 Drugs That Make the Urine Alkaline (7.1 Drugs that Make the Urine Alkaline)
The clearance of memantine was reduced by about 80% under alkaline urine conditions at pH 8. Therefore, alterations of urine pH towards the alkaline condition may lead to an accumulation of the drug with a possible increase in adverse effects. Urine pH is altered by diet, drugs (e.g. carbonic anhydrase inhibitors, sodium bicarbonate) and clinical state of the patient (e.g. renal tubular acidosis or severe infections of the urinary tract). Hence, memantine should be used with caution under these conditions.
13.2 Animal Toxicology And/or Pharmacology (13.2 Animal Toxicology and/or Pharmacology)
Memantine induced neuronal lesions (vacuolation and necrosis) in the multipolar and pyramidal cells in cortical layers III and IV of the posterior cingulate and retrosplenial neocortices in rats, similar to those which are known to occur in rodents administered other NMDA receptor antagonists. Lesions were seen after a single dose of memantine. In a study in which rats were given daily oral doses of memantine for 14 days, the no-effect dose for neuronal necrosis was 6 times the maximum recommended human dose of 20 mg/day on a mg/m2 basis
In acute and repeat-dose neurotoxicity studies in female rats, oral administration of memantine and donepezil in combination resulted in increased incidence, severity, and distribution of neurodegeneration compared with memantine alone. The no-effect levels of the combination were associated with clinically relevant plasma memantine and donepezil exposures.
The relevance of these findings to humans is unknown.
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
There was no evidence of carcinogenicity in a 113-week oral study in mice at doses up to 40 mg/kg/day (10 times the maximum recommended human dose [MRHD] on a mg/m2 basis). There was also no evidence of carcinogenicity in rats orally dosed at up to 40 mg/kg/day for 71 weeks followed by 20 mg/kg/day (20 and 10 times the MRHD on a mg/m2 basis, respectively) through 128 weeks.
Memantine produced no evidence of genotoxic potential when evaluated in the in vitro S. typhimurium or E. coli reverse mutation assay, an in vitro chromosomal aberration test in human lymphocytes, an in vivo cytogenetics assay for chromosome damage in rats, and the in vivo mouse micronucleus assay. The results were equivocal in an in vitro gene mutation assay using Chinese hamster V79 cells.
No impairment of fertility or reproductive performance was seen in rats administered up to 18 mg/kg/day (9 times the MRHD on a mg/m2 basis) orally from 14 days prior to mating through gestation and lactation in females, or for 60 days prior to mating in males.
7.2 Use With Other N Methyl D Aspartate (nmda) Antagonists (7.2 Use with Other N-methyl-D-aspartate (NMDA) Antagonists)
The combined use of Memantine HCl with other NMDA antagonists (amantadine, ketamine, and dextromethorphan) has not been systematically evaluated and such use should be approached with caution.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:34:09.395989 · Updated: 2026-03-14T21:39:42.717550