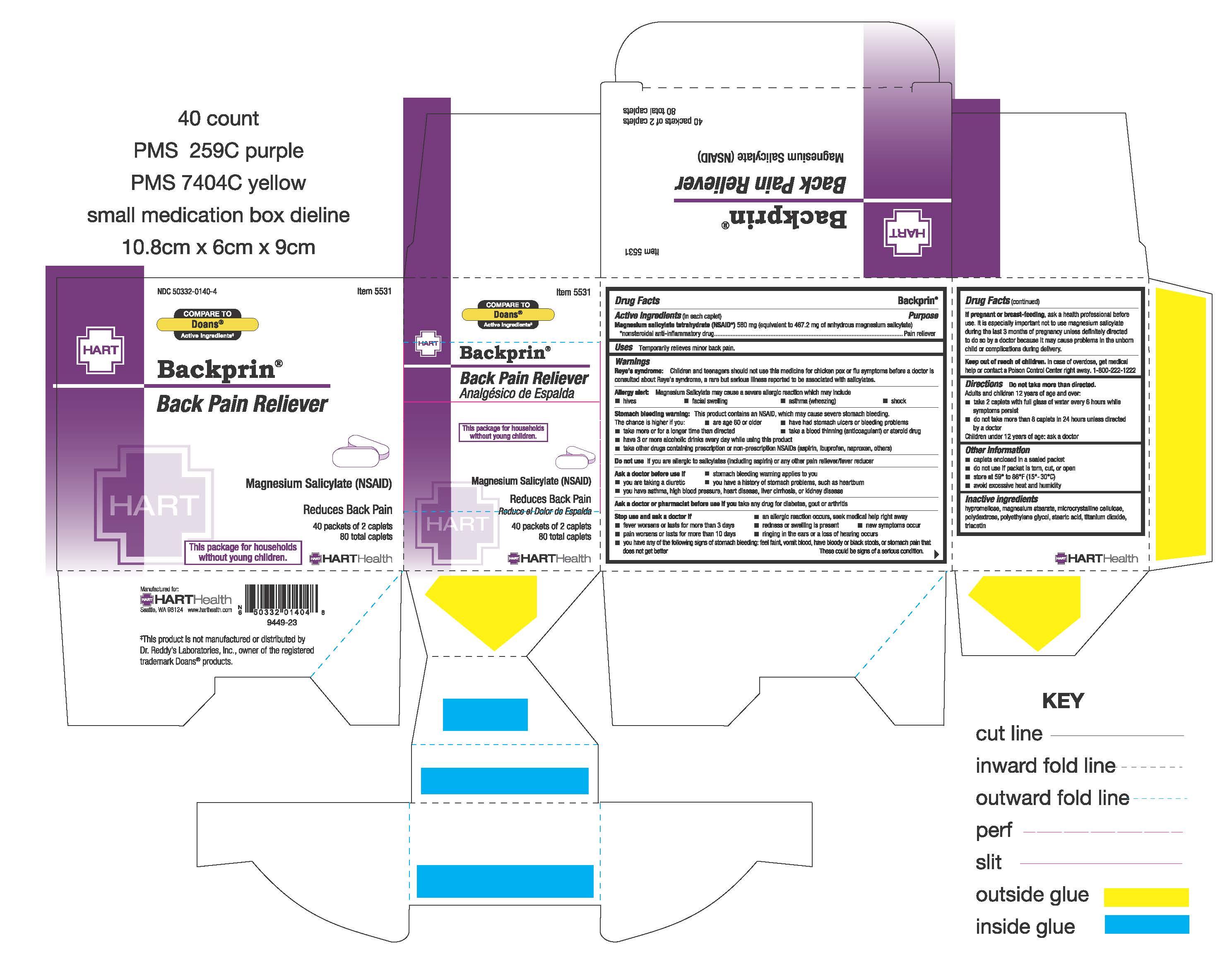
Backprin
c8d1cffc-9483-4462-9f92-c0c2fbfc2b7c
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Active ingredient (in each caplet) Magnesium Salicylate Tetrahydrate (NSAID*) 580 mg (equivalent to 467.2 mg of anhydrous magnesium salicylate) *nonsteroidal anti-inflammatory drug
Medication Information
Warnings and Precautions
Warnings
Reye's syndrome:
Children and teenagers should not use this medicine for chicken pox or flu symptoms before a doctor is consulted about Reye's Syndrome, a rare but serious illness reported to be associated with salicylates.
Allergy alert:.
Magnesium salicylate may cause a severe allergic reaction which may include:
■ hives
■ facial swelling
■ asthma (wheezing)
■ shock
Stomach bleeding warning:
This product contains an NSAID, which may cause severe stomach bleeding. The chance is higher if you:
■ are age 60 or older
■ have had stomach ulcers or bleeding problems
■ take a blood thinning (anticoagulant) or steroid drug
■ take other drugs containing prescription or nonprescription NSAIDs (aspirin, ibuprofen, naproxen, or others)
■ have 3 or more alcoholic drinks every day while using this product
■ take more or for a longer time than directed
Indications and Usage
Uses
Temporarily relieves minor back pain
Dosage and Administration
Directions
Do not take more than directed.
Adults and children 12 years of age and over:
■ take 2 caplets with a full glass of water every 6 hours while symptoms persist
■ do not take more than 8 caplets in 24 hours unless directed by a doctor
Children under 12 years: ask a doctor
Description
Active ingredient (in each caplet) Magnesium Salicylate Tetrahydrate (NSAID*) 580 mg (equivalent to 467.2 mg of anhydrous magnesium salicylate) *nonsteroidal anti-inflammatory drug
Section 50565-1
Keep out of reach of children. In case of overdose, get medical help or contact a Poison Control Center right away. 1-800-222-1222
Section 50566-9
Stop use and ask a doctor if
■ an allergic reaction occurs, seek medical help right away
■ fever worsens or lasts more than 3 days
■ pain worsens or lasts more than 10 days
■ you experience any of the following signs of stomach bleeding: feel faint, vomit blood, have bloody or black stools, or stomach pain that does not get better
■ ringing in the ears or loss of hearing occurs
■ new symptoms occur
■ redness or swelling is present
These could be signs of a serious condition.
Section 50568-5
Ask a doctor or pharmacist before use if you take any drug for diabetes, gout or arthritis
Section 50569-3
Ask a doctor before use if
■ stomach bleeding warning applies to you
■ you have a history of stomach problems, such as heartburn
■ you have asthma, high blood pressure, heart disease, liver cirrhosis, or kidney disease
■ you are taking a diuretic
Section 50570-1
Do not use if you are allergic to salicylates (including aspirin) or any other pain reliever/fever reducer
Section 51727-6
Inactive ingredients
hypromellose, magnesium stearate, microcrystalline cellulose, polydextrose, polyethylene glycol, stearic acid, titanium dioxide, triacetin
Section 51945-4
Section 53414-9
If pregnant or breast-feeding, ask a health professional before use. It is especially important not to use magnesium salicylate at 20 weeks or later in pregnancy unless definitely directed to do so by a doctor because it may cause problems in the unborn child or complications during delivery.
Section 55105-1
Purpose
Pain reliever
Section 55106-9
Active ingredient (in each caplet)
Magnesium Salicylate Tetrahydrate (NSAID*) 580 mg
(equivalent to 467.2 mg of anhydrous magnesium salicylate)
*nonsteroidal anti-inflammatory drug
Section 60561-8
Other information
■ caplets enclosed in a sealed packet
■ do not use if packet is torn, cut or open
■ store at 59° to 86°F (15°-30°C)
■ avoid excessive heat and humidity
Structured Label Content
Indications and Usage (34067-9)
Uses
Temporarily relieves minor back pain
Dosage and Administration (34068-7)
Directions
Do not take more than directed.
Adults and children 12 years of age and over:
■ take 2 caplets with a full glass of water every 6 hours while symptoms persist
■ do not take more than 8 caplets in 24 hours unless directed by a doctor
Children under 12 years: ask a doctor
Warnings and Precautions (34071-1)
Warnings
Reye's syndrome:
Children and teenagers should not use this medicine for chicken pox or flu symptoms before a doctor is consulted about Reye's Syndrome, a rare but serious illness reported to be associated with salicylates.
Allergy alert:.
Magnesium salicylate may cause a severe allergic reaction which may include:
■ hives
■ facial swelling
■ asthma (wheezing)
■ shock
Stomach bleeding warning:
This product contains an NSAID, which may cause severe stomach bleeding. The chance is higher if you:
■ are age 60 or older
■ have had stomach ulcers or bleeding problems
■ take a blood thinning (anticoagulant) or steroid drug
■ take other drugs containing prescription or nonprescription NSAIDs (aspirin, ibuprofen, naproxen, or others)
■ have 3 or more alcoholic drinks every day while using this product
■ take more or for a longer time than directed
Section 50565-1 (50565-1)
Keep out of reach of children. In case of overdose, get medical help or contact a Poison Control Center right away. 1-800-222-1222
Section 50566-9 (50566-9)
Stop use and ask a doctor if
■ an allergic reaction occurs, seek medical help right away
■ fever worsens or lasts more than 3 days
■ pain worsens or lasts more than 10 days
■ you experience any of the following signs of stomach bleeding: feel faint, vomit blood, have bloody or black stools, or stomach pain that does not get better
■ ringing in the ears or loss of hearing occurs
■ new symptoms occur
■ redness or swelling is present
These could be signs of a serious condition.
Section 50568-5 (50568-5)
Ask a doctor or pharmacist before use if you take any drug for diabetes, gout or arthritis
Section 50569-3 (50569-3)
Ask a doctor before use if
■ stomach bleeding warning applies to you
■ you have a history of stomach problems, such as heartburn
■ you have asthma, high blood pressure, heart disease, liver cirrhosis, or kidney disease
■ you are taking a diuretic
Section 50570-1 (50570-1)
Do not use if you are allergic to salicylates (including aspirin) or any other pain reliever/fever reducer
Section 51727-6 (51727-6)
Inactive ingredients
hypromellose, magnesium stearate, microcrystalline cellulose, polydextrose, polyethylene glycol, stearic acid, titanium dioxide, triacetin
Section 51945-4 (51945-4)
Section 53414-9 (53414-9)
If pregnant or breast-feeding, ask a health professional before use. It is especially important not to use magnesium salicylate at 20 weeks or later in pregnancy unless definitely directed to do so by a doctor because it may cause problems in the unborn child or complications during delivery.
Section 55105-1 (55105-1)
Purpose
Pain reliever
Section 55106-9 (55106-9)
Active ingredient (in each caplet)
Magnesium Salicylate Tetrahydrate (NSAID*) 580 mg
(equivalent to 467.2 mg of anhydrous magnesium salicylate)
*nonsteroidal anti-inflammatory drug
Section 60561-8 (60561-8)
Other information
■ caplets enclosed in a sealed packet
■ do not use if packet is torn, cut or open
■ store at 59° to 86°F (15°-30°C)
■ avoid excessive heat and humidity
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:04:36.773921 · Updated: 2026-03-14T23:05:28.061539