These Highlights Do Not Include All The Information Needed To Use Onivyde Safely And Effectively. See Full Prescribing Information For Onivyde
c8b58efa-1820-48a4-b70d-62918fc4abfc
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Neutropenia Severe and life-threatening neutropenia, including fatal neutropenic sepsis and fatal neutropenic fever, has occurred in patients receiving ONIVYDE in combination with oxaliplatin, fluorouracil and leucovorin and in combination with fluorouracil and leucovorin. Withhold ONIVYDE for absolute neutrophil count below 1500/mm 3 or neutropenic fever. Monitor blood cell counts periodically during treatment [see Dosage and Administration (2.2) and Warnings and Precautions (5.1) ]. Diarrhea Severe and life-threatening diarrhea has occurred in patients receiving ONIVYDE in combination with oxaliplatin, fluorouracil and leucovorin and in combination with fluorouracil and leucovorin. Do not administer ONIVYDE to patients with bowel obstruction. Withhold ONIVYDE for diarrhea of Grade 2-4 severity. Administer loperamide for late diarrhea of any severity. Administer atropine, if not contraindicated, for early diarrhea of any severity [see Dosage and Administration (2.2) and Warnings and Precautions (5.2) ].
Indications and Usage
ONIVYDE is indicated, in combination with oxaliplatin, fluorouracil and leucovorin for the first-line treatment of adult patients with metastatic pancreatic adenocarcinoma. ONIVYDE is indicated, in combination with fluorouracil and leucovorin, for the treatment of adult patients with metastatic pancreatic adenocarcinoma after disease progression following gemcitabine-based therapy. Limitations of Use: ONIVYDE is not indicated as a single agent for the treatment of patients with metastatic pancreatic adenocarcinoma. [see Clinical Studies (14) ] .
Dosage and Administration
Do not substitute ONIVYDE for other drugs containing irinotecan HCl. ( 2.1 ) ONIVYDE in combination with oxaliplatin, fluorouracil and leucovorin: Recommended dose of ONIVYDE is 50 mg/m 2 intravenous infusion over 90 minutes every two weeks. ( 2.2 ) Recommended starting dose of ONIVYDE in patients homozygous for UGT1A1*28 is 50 mg/m 2 every two weeks. ( 2.2 ) There is no recommended dose of ONIVYDE for patients with serum bilirubin above the upper limit of normal. ( 2.2 ) ONIVYDE in combination with fluorouracil and leucovorin: Recommended dose of ONIVYDE is 70 mg/m 2 intravenous infusion over 90 minutes every two weeks. ( 2.2 ) Recommended starting dose of ONIVYDE in patients homozygous for UGT1A1*28 is 50 mg/m 2 every two weeks. ( 2.2 ) There is no recommended dose of ONIVYDE for patients with serum bilirubin above the upper limit of normal. ( 2.2 ) Premedicate with a corticosteroid and an anti-emetic 30 minutes prior to ONIVYDE. ( 2.2 )
Warnings and Precautions
Interstitial lung disease (ILD): Fatal ILD has occurred in patients receiving irinotecan including ONIVYDE. Discontinue ONIVYDE if ILD is diagnosed. ( 5.3 ) Severe hypersensitivity reaction: Permanently discontinue ONIVYDE for severe hypersensitivity reactions. ( 5.4 , 4 ) Embryo-fetal toxicity: Can cause fetal harm. Advise females of reproductive potential of the potential risk to a fetus and to use effective contraception. ( 5.5 , 8.1 , 8.3 )
Contraindications
ONIVYDE is contraindicated in patients who have experienced a severe hypersensitivity reaction or anaphylaxis to ONIVYDE or irinotecan HCl. [see Warnings and Precautions (5.4) , Adverse Reactions (6.2) ].
Adverse Reactions
The following adverse drug reactions are discussed in greater detail in other sections of the label: Severe Neutropenia [see Warnings and Precautions (5.1) ] Severe Diarrhea [see Warnings and Precautions (5.2) ] Interstitial Lung Disease [see Warnings and Precautions (5.3) ] Severe Hypersensitivity Reactions [see Warnings and Precautions (5.4) ]
Drug Interactions
Strong CYP3A4 Inducers: Avoid the use of strong CYP3A4 inducers if possible. Substitute non-enzyme inducing therapies at least 2 weeks prior to initiation of ONIVYDE. ( 7.1 ) Strong CYP3A4 Inhibitors: Avoid the use of strong CYP3A4 or UGT1A1 inhibitors, if possible; discontinue strong CYP3A4 inhibitors at least 1 week prior to starting therapy. ( 7.2 )
Medication Information
Warnings and Precautions
Interstitial lung disease (ILD): Fatal ILD has occurred in patients receiving irinotecan including ONIVYDE. Discontinue ONIVYDE if ILD is diagnosed. ( 5.3 ) Severe hypersensitivity reaction: Permanently discontinue ONIVYDE for severe hypersensitivity reactions. ( 5.4 , 4 ) Embryo-fetal toxicity: Can cause fetal harm. Advise females of reproductive potential of the potential risk to a fetus and to use effective contraception. ( 5.5 , 8.1 , 8.3 )
Indications and Usage
ONIVYDE is indicated, in combination with oxaliplatin, fluorouracil and leucovorin for the first-line treatment of adult patients with metastatic pancreatic adenocarcinoma. ONIVYDE is indicated, in combination with fluorouracil and leucovorin, for the treatment of adult patients with metastatic pancreatic adenocarcinoma after disease progression following gemcitabine-based therapy. Limitations of Use: ONIVYDE is not indicated as a single agent for the treatment of patients with metastatic pancreatic adenocarcinoma. [see Clinical Studies (14) ] .
Dosage and Administration
Do not substitute ONIVYDE for other drugs containing irinotecan HCl. ( 2.1 ) ONIVYDE in combination with oxaliplatin, fluorouracil and leucovorin: Recommended dose of ONIVYDE is 50 mg/m 2 intravenous infusion over 90 minutes every two weeks. ( 2.2 ) Recommended starting dose of ONIVYDE in patients homozygous for UGT1A1*28 is 50 mg/m 2 every two weeks. ( 2.2 ) There is no recommended dose of ONIVYDE for patients with serum bilirubin above the upper limit of normal. ( 2.2 ) ONIVYDE in combination with fluorouracil and leucovorin: Recommended dose of ONIVYDE is 70 mg/m 2 intravenous infusion over 90 minutes every two weeks. ( 2.2 ) Recommended starting dose of ONIVYDE in patients homozygous for UGT1A1*28 is 50 mg/m 2 every two weeks. ( 2.2 ) There is no recommended dose of ONIVYDE for patients with serum bilirubin above the upper limit of normal. ( 2.2 ) Premedicate with a corticosteroid and an anti-emetic 30 minutes prior to ONIVYDE. ( 2.2 )
Contraindications
ONIVYDE is contraindicated in patients who have experienced a severe hypersensitivity reaction or anaphylaxis to ONIVYDE or irinotecan HCl. [see Warnings and Precautions (5.4) , Adverse Reactions (6.2) ].
Adverse Reactions
The following adverse drug reactions are discussed in greater detail in other sections of the label: Severe Neutropenia [see Warnings and Precautions (5.1) ] Severe Diarrhea [see Warnings and Precautions (5.2) ] Interstitial Lung Disease [see Warnings and Precautions (5.3) ] Severe Hypersensitivity Reactions [see Warnings and Precautions (5.4) ]
Drug Interactions
Strong CYP3A4 Inducers: Avoid the use of strong CYP3A4 inducers if possible. Substitute non-enzyme inducing therapies at least 2 weeks prior to initiation of ONIVYDE. ( 7.1 ) Strong CYP3A4 Inhibitors: Avoid the use of strong CYP3A4 or UGT1A1 inhibitors, if possible; discontinue strong CYP3A4 inhibitors at least 1 week prior to starting therapy. ( 7.2 )
Description
Neutropenia Severe and life-threatening neutropenia, including fatal neutropenic sepsis and fatal neutropenic fever, has occurred in patients receiving ONIVYDE in combination with oxaliplatin, fluorouracil and leucovorin and in combination with fluorouracil and leucovorin. Withhold ONIVYDE for absolute neutrophil count below 1500/mm 3 or neutropenic fever. Monitor blood cell counts periodically during treatment [see Dosage and Administration (2.2) and Warnings and Precautions (5.1) ]. Diarrhea Severe and life-threatening diarrhea has occurred in patients receiving ONIVYDE in combination with oxaliplatin, fluorouracil and leucovorin and in combination with fluorouracil and leucovorin. Do not administer ONIVYDE to patients with bowel obstruction. Withhold ONIVYDE for diarrhea of Grade 2-4 severity. Administer loperamide for late diarrhea of any severity. Administer atropine, if not contraindicated, for early diarrhea of any severity [see Dosage and Administration (2.2) and Warnings and Precautions (5.2) ].
Section 42229-5
In combination with oxaliplatin, fluorouracil and leucovorin for the first-line treatment of patients with metastatic pancreatic adenocarcinoma
Administer ONIVYDE prior to oxaliplatin, fluorouracil and leucovorin [see Clinical Studies (14)].
- The recommended dosage of ONIVYDE regardless of UGT1A1*28 allele genotype is 50 mg/m2 administered by intravenous infusion over 90 minutes every 2 weeks.
- There is no recommended dosage of ONIVYDE for patients with serum bilirubin above the upper limit of normal [see Adverse Reactions (6.1) and Clinical Studies (14)].
Section 43683-2
Section 44425-7
Storage and Handling
Store ONIVYDE at 2ºC to 8ºC (36°F to 46°F). Do NOT freeze. Protect from light.
ONIVYDE is a hazardous drug. Follow applicable special handling and disposal procedures.1
10 Overdosage
There are no treatment interventions known to be effective for management of overdosage of ONIVYDE.
15 References
1. OSHA Hazardous Drugs. OSHA. http://www.osha.gov/SLTC/hazardousdrugs/index.html
11 Description
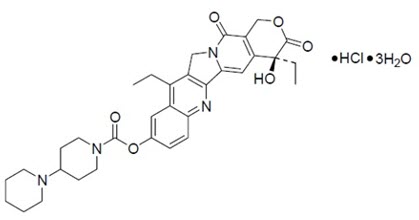
ONIVYDE is formulated with irinotecan hydrochloride trihydrate, a topoisomerase inhibitor, into a liposomal dispersion for intravenous use. The chemical name of irinotecan hydrochloride trihydrate is (S)-4,11-diethyl-3,4,12,14-tetrahydro-4-hydroxy-3,14-dioxo1H-pyrano[3',4':6,7]-indolizino[1,2-b]quinolin-9-yl-[1,4'bipiperidine]-1'-carboxylate, monohydrochloride, trihydrate. The empirical formula is C33H38N4O6∙HCl∙3H2O and the molecular weight is 677.19 g/mole. The molecular structure is:
ONIVYDE is a sterile, white to slightly yellow opaque isotonic liposomal dispersion. Each 10 mL single-dose vial contains 43 mg irinotecan free base at a concentration of 4.3 mg/mL. The liposome is a unilamellar lipid bilayer vesicle, approximately 110 nm in diameter, which encapsulates an aqueous space containing irinotecan in a gelated or precipitated state as the sucrose octasulfate salt. The vesicle is composed of 1,2-distearoyl-sn-glycero-3-phosphocholine (DSPC) 6.81 mg/mL, cholesterol 2.22 mg/mL, and methoxy-terminated polyethylene glycol (MW 2000)-distearoylphosphatidyl ethanolamine (MPEG-2000-DSPE) 0.12 mg/mL. Each mL also contains 2-[4-(2-hydroxyethyl) piperazin-1-yl]ethanesulfonic acid (HEPES) as a buffer 4.05 mg/mL and sodium chloride as an isotonicity reagent 8.42 mg/mL.
8.4 Pediatric Use
Safety and effectiveness of ONIVYDE have not been established in pediatric patients.
8.5 Geriatric Use
Of the 634 patients who received ONIVYDE as a single agent, in combination with FU and leucovorin or in combination with oxaliplatin, FU and leucovorin in NAPOLI-1 and NAPOLI 3, 49% were ≥ 65 years old and 10% were ≥ 75 years old. No overall differences in safety and effectiveness were observed between these patients and younger patients.
4 Contraindications
ONIVYDE is contraindicated in patients who have experienced a severe hypersensitivity reaction or anaphylaxis to ONIVYDE or irinotecan HCl. [see Warnings and Precautions (5.4), Adverse Reactions (6.2)].
5.2 Severe Diarrhea
ONIVYDE can cause severe and life-threatening diarrhea. Do not administer ONIVYDE to patients with a bowel obstruction. Severe or life-threatening diarrhea can follow one of two patterns: late onset diarrhea (onset more than 24 hours following chemotherapy) and early onset diarrhea (onset within 24 hours of chemotherapy, sometimes occurring with other symptoms of cholinergic reaction) [see Adverse Reactions (6.1)]. An individual patient may experience both early and late-onset diarrhea.
In NAPOLI 3, Grade 3 and 4 diarrhea (early and late-onset) occurred in 20% receiving ONIVYDE in combination with oxaliplatin, fluorouracil, and leucovorin (NALIRIFOX). In NAPOLI-1, Grade 3 or 4 diarrhea occurred in 13% receiving ONIVYDE/FU/LV. The incidence of Grade 3 or 4 late onset diarrhea was 9% in patients receiving ONIVYDE/FU/LV. The incidence of Grade 3 or 4 early onset diarrhea was 3% in patients receiving ONIVYDE/FU/LV. Of patients receiving ONIVYDE/FU/LV in NAPOLI-1, 34% received loperamide for late-onset diarrhea and 26% received atropine for early-onset diarrhea.
To reduce the risk of severe diarrhea, patients should stop lactose-containing products, eat a low-fat diet and maintain hydration during treatment with ONIVYDE. Withhold ONIVYDE for Grade 2-4 diarrhea. Administer intravenous or subcutaneous atropine 0.25 to 1 mg (unless clinically contraindicated) for early onset diarrhea of any severity. Initiate loperamide for late onset diarrhea of any severity. Local institutional guidelines should be followed for the treatment of diarrhea that does not improve within 48 hours and may include the addition of diphenoxylate hydrochloride plus atropine sulfate or octreotide. Following recovery to Grade 1 diarrhea, resume ONIVYDE at a reduced dose [see Dosage and Administration (2.3)].
6 Adverse Reactions
The following adverse drug reactions are discussed in greater detail in other sections of the label:
- Severe Neutropenia [see Warnings and Precautions (5.1)]
- Severe Diarrhea [see Warnings and Precautions (5.2)]
- Interstitial Lung Disease [see Warnings and Precautions (5.3)]
- Severe Hypersensitivity Reactions [see Warnings and Precautions (5.4)]
7 Drug Interactions
- Strong CYP3A4 Inducers: Avoid the use of strong CYP3A4 inducers if possible. Substitute non-enzyme inducing therapies at least 2 weeks prior to initiation of ONIVYDE. (7.1)
- Strong CYP3A4 Inhibitors: Avoid the use of strong CYP3A4 or UGT1A1 inhibitors, if possible; discontinue strong CYP3A4 inhibitors at least 1 week prior to starting therapy. (7.2)
12.3 Pharmacokinetics
The plasma pharmacokinetics of total irinotecan and total SN-38 were evaluated in patients with cancer who received ONIVYDE, as a single agent or as part of combination chemotherapy, at doses between 35 mg/m2 and 155 mg/m2 and concentration proportional to dose was observed.
The pharmacokinetic parameters of total irinotecan and total SN-38 following the administration of ONIVYDE 70 mg/m2 as a single agent or part of combination chemotherapy are presented in Table 7.
| Dose (mg/m2) |
Descriptive Statistics | Total Irinotecan | Total SN-38 | |||||
|---|---|---|---|---|---|---|---|---|
| Cmax
[µg/mL] |
AUCSS
[day∙µg/mL] |
t1/2
[day] |
V [L] |
Cmax
[ng/mL] |
AUCSS
[day∙ng/mL] |
V [L] |
||
| AUCSS: Area under the plasma concentration curve at steady-state | ||||||||
| t1/2: Terminal elimination half-life | ||||||||
| V: Volume of distribution | ||||||||
| 50 | Geometric Mean | 25.1 | 37.8 | 1.93 | 3.63 | 2.09 | 12.1 | 3.46 |
| CV (%) | 18.5 | 73.6 | 14 | 33.5 | 42.1 | 46.6 | 35.5 | |
| 70 | Geometric Mean | 30.8 | 50.4 | 1.87 | 4.23 | 2.64 | 14.7 | 4.06 |
| CV (%) | 19.7 | 75.3 | 26.4 | 28.1 | 64.5 | 58 | 29.4 |
12.5 Pharmacogenomics
Individuals who are homozygous for the UGT1A1*28 allele are at increased risk for neutropenia from irinotecan HCl. In NAPOLI-1, patients homozygous for the UGT1A1*28 allele (N=7) initiated ONIVYDE at a reduced dose of 50 mg/m2 in combination with FU/LV. The frequency of Grade 3 or 4 neutropenia in these patients [2 of 7 (28.6%)] was similar to the frequency in patients not homozygous for the UGT1A1*28 allele who received a starting dose of ONIVYDE of 70 mg/m2 [30 of 110 (27.3%)]. In NAPOLI-3, patients homozygous for the UGT1A1*28 allele (N = 39) initiated ONIVYDE at the same starting dose of 50 mg/m2 as patients not homozygous for the UGT1A1*28 allele (N = 328). The frequency of Grade 3 or 4 neutropenia was 23% in patients homozygous for the UGT1A1*28 allele and 13% in patients not homozygous for the UGT1A1*28 allele. The frequency of dose reduction of ONIVYDE due to treatment-emergent adverse effects was 59% versus 51% in patients homozygous versus non-homozygous for the UGT1A1*28 allele.
5.1 Severe Neutropenia
ONIVYDE can cause severe or life-threatening neutropenia and fatal neutropenic sepsis.
In NAPOLI 3, Grade 3 and 4 neutropenia occurred in 26% of patients receiving ONIVYDE in combination with oxaliplatin, fluorouracil, and leucovorin (NALIRIFOX) and fatal neutropenic fever in 0.3% of patients [see Adverse Reactions (6.1)]. In NAPOLI-1, Grade 3 and 4 neutropenia occurred in 20% of patients receiving ONIVYDE in combination with fluorouracil and leucovorin (ONIVYDE/FU/LV). Neutropenic sepsis occurred in 3% and fatal neutropenic sepsis in 0.8% [see Adverse Reactions (6.1)].
In NAPOLI 3, the incidence of Grade 3 or 4 neutropenia was similar among Asian patients [6 of 20 (30%)] compared to White patients [76 of 289 (26%)] receiving ONIVYDE in combination with oxaliplatin, fluorouracil, and leucovorin. Neutropenic fever was reported in 5% of Asian patients (1 of 20) compared to 2.3% of White patients (7 of 306). In NAPOLI-1, the incidence of Grade 3 or 4 neutropenia was higher among Asian patients [18 of 33 (55%)] compared to White patients [13 of 73 (18%)] receiving ONIVYDE/FU/LV. Neutropenic fever/neutropenic sepsis was reported in 6% of Asian patients compared to 1% of White patients [see Clinical Pharmacology (12.3)].
Monitor complete blood cell counts on Days 1 and 8 of every cycle and more frequently if clinically indicated. Withhold ONIVYDE if the absolute neutrophil count (ANC) is below 1500/mm3 or if neutropenic fever occurs. Resume ONIVYDE when the ANC is 1500/mm3 or above. Reduce ONIVYDE dose for Grade 3-4 neutropenia or neutropenic fever following recovery in subsequent cycles [see Dosage and Administration (2.2)].
1 Indications and Usage
- ONIVYDE is indicated, in combination with oxaliplatin, fluorouracil and leucovorin for the first-line treatment of adult patients with metastatic pancreatic adenocarcinoma.
- ONIVYDE is indicated, in combination with fluorouracil and leucovorin, for the treatment of adult patients with metastatic pancreatic adenocarcinoma after disease progression following gemcitabine-based therapy.
Limitations of Use: ONIVYDE is not indicated as a single agent for the treatment of patients with metastatic pancreatic adenocarcinoma. [see Clinical Studies (14)].
12.1 Mechanism of Action
Irinotecan liposome injection is a topoisomerase 1 inhibitor encapsulated in a lipid bilayer vesicle or liposome. Topoisomerase 1 relieves torsional strain in DNA by inducing single-strand breaks. Irinotecan and its active metabolite SN-38 bind reversibly to the topoisomerase 1-DNA complex and prevent re-ligation of the single-strand breaks, leading to exposure time-dependent double-strand DNA damage and cell death. In mice bearing human tumor xenografts, irinotecan liposome administered at irinotecan HCl-equivalent doses 5-fold lower than irinotecan HCl achieved similar intratumoral exposure of SN-38.
5.5 Embryo Fetal Toxicity
Based on animal data with irinotecan HCl and the mechanism of action of ONIVYDE, ONIVYDE can cause fetal harm when administered to a pregnant woman. Embryotoxicity and teratogenicity were observed following treatment with irinotecan HCl, at doses resulting in irinotecan exposures lower than those achieved with ONIVYDE 70 mg/m2 in humans, administered to pregnant rats and rabbits during organogenesis. Advise pregnant women of the potential risk to a fetus. Advise females of reproductive potential to use effective contraception during treatment with ONIVYDE and for seven months following the last dose [see Use in Specific Populations (8.1, 8.3), Clinical Pharmacology (12.1)].
5 Warnings and Precautions
- Interstitial lung disease (ILD): Fatal ILD has occurred in patients receiving irinotecan including ONIVYDE. Discontinue ONIVYDE if ILD is diagnosed. (5.3)
- Severe hypersensitivity reaction: Permanently discontinue ONIVYDE for severe hypersensitivity reactions. (5.4, 4)
- Embryo-fetal toxicity: Can cause fetal harm. Advise females of reproductive potential of the potential risk to a fetus and to use effective contraception. (5.5, 8.1, 8.3)
7.1 Strong Cyp3a4 Inducers
Following administration of non-liposomal irinotecan (i.e., irinotecan HCl), exposure to irinotecan or its active metabolite, SN-38, is substantially reduced in adult and pediatric patients concomitantly receiving the CYP3A4 enzyme-inducing anticonvulsants phenytoin and strong CYP3A4 inducers. Avoid the use of strong CYP3A4 inducers (e.g., rifampin, phenytoin, carbamazepine, rifabutin, rifapentine, phenobarbital, St. John's wort) if possible. Substitute non-enzyme inducing therapies at least 2 weeks prior to initiation of ONIVYDE therapy [see Clinical Pharmacology (12.3)].
2 Dosage and Administration
- Do not substitute ONIVYDE for other drugs containing irinotecan HCl. (2.1)
- ONIVYDE in combination with oxaliplatin, fluorouracil and leucovorin:
- Recommended dose of ONIVYDE is 50 mg/m2 intravenous infusion over 90 minutes every two weeks. (2.2)
- Recommended starting dose of ONIVYDE in patients homozygous for UGT1A1*28 is 50 mg/m2 every two weeks. (2.2)
- There is no recommended dose of ONIVYDE for patients with serum bilirubin above the upper limit of normal. (2.2)
- ONIVYDE in combination with fluorouracil and leucovorin:
- Recommended dose of ONIVYDE is 70 mg/m2 intravenous infusion over 90 minutes every two weeks. (2.2)
- Recommended starting dose of ONIVYDE in patients homozygous for UGT1A1*28 is 50 mg/m2 every two weeks. (2.2)
- There is no recommended dose of ONIVYDE for patients with serum bilirubin above the upper limit of normal. (2.2)
- Premedicate with a corticosteroid and an anti-emetic 30 minutes prior to ONIVYDE. (2.2)
3 Dosage Forms and Strengths
Injection: 43 mg/10 mL irinotecan free base as a white to slightly yellow, opaque, liposomal dispersion in a single-dose vial.
6.2 Postmarketing Experience
The following adverse reactions have been identified during post approval use of ONIVYDE. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Immune system disorders: Hypersensitivity (including Anaphylactic reaction and Angioedema)
2.1 Important Use Information
DO NOT SUBSTITUTE ONIVYDE for other drugs containing irinotecan HCl.
5.3 Interstitial Lung Disease
ONIVYDE can cause severe and fatal interstitial lung disease (ILD), including pneumonitis. Postmarketing cases of severe and fatal ILD have been reported with ONIVYDE. Risk factors include pre-existing lung disease, use of pneumotoxic medicinal products, colony stimulating factors or having previously received radiation therapy. Patients with risk factors should be closely monitored for respiratory symptoms before and during ONIVYDE therapy. Withhold ONIVYDE in patients with new or progressive dyspnea, cough, and fever, pending diagnostic evaluation. Discontinue ONIVYDE in patients with a confirmed diagnosis of ILD.
8 Use in Specific Populations
- Lactation: Do not breastfeed. (8.2)
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in clinical trials of ONIVYDE cannot be directly compared to rates in clinical trials of other drugs and may not reflect the rates observed in practice.
17 Patient Counseling Information
Advise patients of the following:
2.4 Preparation and Administration
ONIVYDE is a hazardous drug. Follow applicable special handling and disposal procedures.1
5.4 Severe Hypersensitivity Reaction
Irinotecan including ONIVYDE can cause severe hypersensitivity reactions, including anaphylactic reactions. Permanently discontinue ONIVYDE in patients who experience a severe hypersensitivity reaction [see Contraindications (4), Adverse reactions (6.2)].
7.2 Strong Cyp3a4 Or Ugt1a1 Inhibitors
Following administration of non-liposomal irinotecan (i.e., irinotecan HCl), patients receiving concomitant ketoconazole, a CYP3A4 and UGT1A1 inhibitor, have increased exposure to irinotecan and its active metabolite SN-38. Co-administration of ONIVYDE with other inhibitors of CYP3A4 (e.g., clarithromycin, indinavir, itraconazole, lopinavir, nefazodone, nelfinavir, ritonavir, saquinavir, telaprevir, voriconazole) or UGT1A1 (e.g., atazanavir, gemfibrozil, indinavir) may increase systemic exposure to irinotecan or SN-38. Avoid the use of strong CYP3A4 or UGT1A1 inhibitors if possible. Discontinue strong CYP3A4 inhibitors at least 1 week prior to starting ONIVYDE therapy [see Clinical Pharmacology (12.3)].
2.3 Dosage Modifications for Adverse Reactions
Recommended dosage modifications for ONIVYDE are in Table 1 and Table 2.
| Toxicity Toxicity grading per NCI CTCAE v5.0.
|
Occurrence | ONIVYDE adjustment in patients receiving 50mg/m2 |
|---|---|---|
|
Grade 3 or 4 Adverse reactions No dosage modification is necessary for asthenia, alopecia and Grade 3 anorexia.
|
Withhold ONIVYDE Upon recovery to ≤ Grade 1 Do not resume until the absolute neutrophil count is ≥2000/mm3 (2×109/L) and the platelet count is ≥100,000/mm3 (100×109/L). ,For Grade ≥3 nausea and vomiting, reduce dose only if occurs despite optimal anti-emetic therapy. ,Refer to the Full Prescribing Information of fluorouracil and oxaliplatin. When ONIVYDE dose is reduced for adverse reactions, reduce fluorouracil (FU) and oxaliplatin doses: for first occurrence, reduce dose to 80% of original dose; for second occurrence, reduce dose to 65% of original dose; for third occurrence, reduce dose to 50% of original dose; discontinue therapy for fourth occurrence. Oxaliplatin may be discontinued if not well tolerated and treatment with ONIVYDE + FU/LV can continue. Maintain original dose level of leucovorin for first, second and third occurrence of toxicity. , resume ONIVYDE at: |
|
| First | 40 mg/m2 | |
| Second | 32.5 mg/m2 | |
| Third | 25 mg/m2 | |
| Fourth | Discontinue ONIVYDE | |
| Grade 3 or 4 Hand foot syndrome | First | Discontinue ONIVYDE |
| Any grade neurocerebellar toxicity | First | Discontinue ONIVYDE |
| Grade ≥ 2 cardiac toxicity | First | Discontinue ONIVYDE |
| Interstitial lung disease | First | Discontinue ONIVYDE |
| Anaphylactic reaction | First | Discontinue ONIVYDE |
| Toxicity NCI CTCAE v4.0 NCI CTCAE v 4.0=National Cancer Institute Common Toxicity Criteria for Adverse Events version 4.0
|
Occurrence | ONIVYDE adjustment in patients receiving 70 mg/m2 | Patients homozygous for UGT1A1*28 without previous increase to 70 mg/m2 |
|---|---|---|---|
| Grade 3 or 4 adverse reactions | Withhold ONIVYDE. Upon recovery to ≤ Grade 1, resume ONIVYDE at: |
||
| First | 50 mg/m2 | 43 mg/m2 | |
| Second | 43 mg/m2 | 35 mg/m2 | |
| Third | Discontinue ONIVYDE | Discontinue ONIVYDE | |
| Interstitial Lung Disease | First | Discontinue ONIVYDE | Discontinue ONIVYDE |
| Anaphylactic Reaction | First | Discontinue ONIVYDE | Discontinue ONIVYDE |
For recommended dose modifications of fluorouracil (FU) or leucovorin (LV), refer to the Full Prescribing Information; refer to Clinical Studies (14).
Warning: Severe Neutropenia and Severe Diarrhea
Neutropenia
- Severe and life-threatening neutropenia, including fatal neutropenic sepsis and fatal neutropenic fever, has occurred in patients receiving ONIVYDE in combination with oxaliplatin, fluorouracil and leucovorin and in combination with fluorouracil and leucovorin. Withhold ONIVYDE for absolute neutrophil count below 1500/mm3 or neutropenic fever. Monitor blood cell counts periodically during treatment [see Dosage and Administration (2.2) and Warnings and Precautions (5.1)].
Diarrhea
- Severe and life-threatening diarrhea has occurred in patients receiving ONIVYDE in combination with oxaliplatin, fluorouracil and leucovorin and in combination with fluorouracil and leucovorin. Do not administer ONIVYDE to patients with bowel obstruction. Withhold ONIVYDE for diarrhea of Grade 2-4 severity. Administer loperamide for late diarrhea of any severity. Administer atropine, if not contraindicated, for early diarrhea of any severity [see Dosage and Administration (2.2) and Warnings and Precautions (5.2)].
Principal Display Panel 43 Mg/10 Ml Vial Label
NDC 15054-0043-1
Rx Only
Onivyde®
(irinotecan liposome injection)
43 mg/10 mL
(4.3 mg/mL)
For Intravenous Infusion After Dilution
Single Dose Vial
Discard Unused Portion
Liposomal Formulation
Do Not Substitute For
Irinotecan Hydrochloride
Principal Display Panel 43 Mg/10 Ml Vial Carton
NDC 15054-0043-1
Rx Only
onivyde®
(irinotecan liposome injection)
43 mg/10 mL
(4.3 mg/mL)
For Intravenous Infusion After Dilution
Warning: Hazardous drug
Single Dose Vial
Discard Unused Portion
Liposomal Formulation
Do Not Substitute For Irinotecan Hydrochloride
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
No studies have been performed to assess the potential of irinotecan liposome for carcinogenicity, genotoxicity or impairment of fertility. Intravenous administration of irinotecan hydrochloride to rats once weekly for 13 weeks followed by a 91-week recovery period resulted in a significant linear trend between irinotecan HCl dosage and the incidence of combined uterine horn endometrial stromal polyps and endometrial stromal sarcomas. Irinotecan HCl was clastogenic both in vitro (chromosome aberrations in Chinese hamster ovary cells) and in vivo (micronucleus test in mice). Neither irinotecan nor its active metabolite, SN-38, was mutagenic in the in vitro Ames assay.
Dedicated fertility studies have not been performed with irinotecan liposome injection. Atrophy of male and female reproductive organs was observed in dogs receiving irinotecan liposome injection every 3 weeks at doses equal to or greater than 15 mg/kg, (approximately 3 times the clinical exposure of irinotecan following administration to ONIVYDE dosed at 70 mg/m2) for a total of 6 doses. No significant adverse effects on fertility and general reproductive performance were observed after intravenous administration of irinotecan HCl in doses of up to 6 mg/kg/day to rats; however, atrophy of male reproductive organs was observed after multiple daily irinotecan HCl doses both in rodents at 20 mg/kg (approximately 0.007 times the clinical irinotecan exposure following ONIVYDE administration at 70 mg/m2) and in dogs at 0.4 mg/kg (0.0007 times the clinical exposure to irinotecan following administration of ONIVYDE).
Structured Label Content
Section 42229-5 (42229-5)
In combination with oxaliplatin, fluorouracil and leucovorin for the first-line treatment of patients with metastatic pancreatic adenocarcinoma
Administer ONIVYDE prior to oxaliplatin, fluorouracil and leucovorin [see Clinical Studies (14)].
- The recommended dosage of ONIVYDE regardless of UGT1A1*28 allele genotype is 50 mg/m2 administered by intravenous infusion over 90 minutes every 2 weeks.
- There is no recommended dosage of ONIVYDE for patients with serum bilirubin above the upper limit of normal [see Adverse Reactions (6.1) and Clinical Studies (14)].
Section 43683-2 (43683-2)
Section 44425-7 (44425-7)
Storage and Handling
Store ONIVYDE at 2ºC to 8ºC (36°F to 46°F). Do NOT freeze. Protect from light.
ONIVYDE is a hazardous drug. Follow applicable special handling and disposal procedures.1
10 Overdosage (10 OVERDOSAGE)
There are no treatment interventions known to be effective for management of overdosage of ONIVYDE.
15 References (15 REFERENCES)
1. OSHA Hazardous Drugs. OSHA. http://www.osha.gov/SLTC/hazardousdrugs/index.html
11 Description (11 DESCRIPTION)
ONIVYDE is formulated with irinotecan hydrochloride trihydrate, a topoisomerase inhibitor, into a liposomal dispersion for intravenous use. The chemical name of irinotecan hydrochloride trihydrate is (S)-4,11-diethyl-3,4,12,14-tetrahydro-4-hydroxy-3,14-dioxo1H-pyrano[3',4':6,7]-indolizino[1,2-b]quinolin-9-yl-[1,4'bipiperidine]-1'-carboxylate, monohydrochloride, trihydrate. The empirical formula is C33H38N4O6∙HCl∙3H2O and the molecular weight is 677.19 g/mole. The molecular structure is:
ONIVYDE is a sterile, white to slightly yellow opaque isotonic liposomal dispersion. Each 10 mL single-dose vial contains 43 mg irinotecan free base at a concentration of 4.3 mg/mL. The liposome is a unilamellar lipid bilayer vesicle, approximately 110 nm in diameter, which encapsulates an aqueous space containing irinotecan in a gelated or precipitated state as the sucrose octasulfate salt. The vesicle is composed of 1,2-distearoyl-sn-glycero-3-phosphocholine (DSPC) 6.81 mg/mL, cholesterol 2.22 mg/mL, and methoxy-terminated polyethylene glycol (MW 2000)-distearoylphosphatidyl ethanolamine (MPEG-2000-DSPE) 0.12 mg/mL. Each mL also contains 2-[4-(2-hydroxyethyl) piperazin-1-yl]ethanesulfonic acid (HEPES) as a buffer 4.05 mg/mL and sodium chloride as an isotonicity reagent 8.42 mg/mL.
8.4 Pediatric Use
Safety and effectiveness of ONIVYDE have not been established in pediatric patients.
8.5 Geriatric Use
Of the 634 patients who received ONIVYDE as a single agent, in combination with FU and leucovorin or in combination with oxaliplatin, FU and leucovorin in NAPOLI-1 and NAPOLI 3, 49% were ≥ 65 years old and 10% were ≥ 75 years old. No overall differences in safety and effectiveness were observed between these patients and younger patients.
4 Contraindications (4 CONTRAINDICATIONS)
ONIVYDE is contraindicated in patients who have experienced a severe hypersensitivity reaction or anaphylaxis to ONIVYDE or irinotecan HCl. [see Warnings and Precautions (5.4), Adverse Reactions (6.2)].
5.2 Severe Diarrhea
ONIVYDE can cause severe and life-threatening diarrhea. Do not administer ONIVYDE to patients with a bowel obstruction. Severe or life-threatening diarrhea can follow one of two patterns: late onset diarrhea (onset more than 24 hours following chemotherapy) and early onset diarrhea (onset within 24 hours of chemotherapy, sometimes occurring with other symptoms of cholinergic reaction) [see Adverse Reactions (6.1)]. An individual patient may experience both early and late-onset diarrhea.
In NAPOLI 3, Grade 3 and 4 diarrhea (early and late-onset) occurred in 20% receiving ONIVYDE in combination with oxaliplatin, fluorouracil, and leucovorin (NALIRIFOX). In NAPOLI-1, Grade 3 or 4 diarrhea occurred in 13% receiving ONIVYDE/FU/LV. The incidence of Grade 3 or 4 late onset diarrhea was 9% in patients receiving ONIVYDE/FU/LV. The incidence of Grade 3 or 4 early onset diarrhea was 3% in patients receiving ONIVYDE/FU/LV. Of patients receiving ONIVYDE/FU/LV in NAPOLI-1, 34% received loperamide for late-onset diarrhea and 26% received atropine for early-onset diarrhea.
To reduce the risk of severe diarrhea, patients should stop lactose-containing products, eat a low-fat diet and maintain hydration during treatment with ONIVYDE. Withhold ONIVYDE for Grade 2-4 diarrhea. Administer intravenous or subcutaneous atropine 0.25 to 1 mg (unless clinically contraindicated) for early onset diarrhea of any severity. Initiate loperamide for late onset diarrhea of any severity. Local institutional guidelines should be followed for the treatment of diarrhea that does not improve within 48 hours and may include the addition of diphenoxylate hydrochloride plus atropine sulfate or octreotide. Following recovery to Grade 1 diarrhea, resume ONIVYDE at a reduced dose [see Dosage and Administration (2.3)].
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following adverse drug reactions are discussed in greater detail in other sections of the label:
- Severe Neutropenia [see Warnings and Precautions (5.1)]
- Severe Diarrhea [see Warnings and Precautions (5.2)]
- Interstitial Lung Disease [see Warnings and Precautions (5.3)]
- Severe Hypersensitivity Reactions [see Warnings and Precautions (5.4)]
7 Drug Interactions (7 DRUG INTERACTIONS)
- Strong CYP3A4 Inducers: Avoid the use of strong CYP3A4 inducers if possible. Substitute non-enzyme inducing therapies at least 2 weeks prior to initiation of ONIVYDE. (7.1)
- Strong CYP3A4 Inhibitors: Avoid the use of strong CYP3A4 or UGT1A1 inhibitors, if possible; discontinue strong CYP3A4 inhibitors at least 1 week prior to starting therapy. (7.2)
12.3 Pharmacokinetics
The plasma pharmacokinetics of total irinotecan and total SN-38 were evaluated in patients with cancer who received ONIVYDE, as a single agent or as part of combination chemotherapy, at doses between 35 mg/m2 and 155 mg/m2 and concentration proportional to dose was observed.
The pharmacokinetic parameters of total irinotecan and total SN-38 following the administration of ONIVYDE 70 mg/m2 as a single agent or part of combination chemotherapy are presented in Table 7.
| Dose (mg/m2) |
Descriptive Statistics | Total Irinotecan | Total SN-38 | |||||
|---|---|---|---|---|---|---|---|---|
| Cmax
[µg/mL] |
AUCSS
[day∙µg/mL] |
t1/2
[day] |
V [L] |
Cmax
[ng/mL] |
AUCSS
[day∙ng/mL] |
V [L] |
||
| AUCSS: Area under the plasma concentration curve at steady-state | ||||||||
| t1/2: Terminal elimination half-life | ||||||||
| V: Volume of distribution | ||||||||
| 50 | Geometric Mean | 25.1 | 37.8 | 1.93 | 3.63 | 2.09 | 12.1 | 3.46 |
| CV (%) | 18.5 | 73.6 | 14 | 33.5 | 42.1 | 46.6 | 35.5 | |
| 70 | Geometric Mean | 30.8 | 50.4 | 1.87 | 4.23 | 2.64 | 14.7 | 4.06 |
| CV (%) | 19.7 | 75.3 | 26.4 | 28.1 | 64.5 | 58 | 29.4 |
12.5 Pharmacogenomics
Individuals who are homozygous for the UGT1A1*28 allele are at increased risk for neutropenia from irinotecan HCl. In NAPOLI-1, patients homozygous for the UGT1A1*28 allele (N=7) initiated ONIVYDE at a reduced dose of 50 mg/m2 in combination with FU/LV. The frequency of Grade 3 or 4 neutropenia in these patients [2 of 7 (28.6%)] was similar to the frequency in patients not homozygous for the UGT1A1*28 allele who received a starting dose of ONIVYDE of 70 mg/m2 [30 of 110 (27.3%)]. In NAPOLI-3, patients homozygous for the UGT1A1*28 allele (N = 39) initiated ONIVYDE at the same starting dose of 50 mg/m2 as patients not homozygous for the UGT1A1*28 allele (N = 328). The frequency of Grade 3 or 4 neutropenia was 23% in patients homozygous for the UGT1A1*28 allele and 13% in patients not homozygous for the UGT1A1*28 allele. The frequency of dose reduction of ONIVYDE due to treatment-emergent adverse effects was 59% versus 51% in patients homozygous versus non-homozygous for the UGT1A1*28 allele.
5.1 Severe Neutropenia
ONIVYDE can cause severe or life-threatening neutropenia and fatal neutropenic sepsis.
In NAPOLI 3, Grade 3 and 4 neutropenia occurred in 26% of patients receiving ONIVYDE in combination with oxaliplatin, fluorouracil, and leucovorin (NALIRIFOX) and fatal neutropenic fever in 0.3% of patients [see Adverse Reactions (6.1)]. In NAPOLI-1, Grade 3 and 4 neutropenia occurred in 20% of patients receiving ONIVYDE in combination with fluorouracil and leucovorin (ONIVYDE/FU/LV). Neutropenic sepsis occurred in 3% and fatal neutropenic sepsis in 0.8% [see Adverse Reactions (6.1)].
In NAPOLI 3, the incidence of Grade 3 or 4 neutropenia was similar among Asian patients [6 of 20 (30%)] compared to White patients [76 of 289 (26%)] receiving ONIVYDE in combination with oxaliplatin, fluorouracil, and leucovorin. Neutropenic fever was reported in 5% of Asian patients (1 of 20) compared to 2.3% of White patients (7 of 306). In NAPOLI-1, the incidence of Grade 3 or 4 neutropenia was higher among Asian patients [18 of 33 (55%)] compared to White patients [13 of 73 (18%)] receiving ONIVYDE/FU/LV. Neutropenic fever/neutropenic sepsis was reported in 6% of Asian patients compared to 1% of White patients [see Clinical Pharmacology (12.3)].
Monitor complete blood cell counts on Days 1 and 8 of every cycle and more frequently if clinically indicated. Withhold ONIVYDE if the absolute neutrophil count (ANC) is below 1500/mm3 or if neutropenic fever occurs. Resume ONIVYDE when the ANC is 1500/mm3 or above. Reduce ONIVYDE dose for Grade 3-4 neutropenia or neutropenic fever following recovery in subsequent cycles [see Dosage and Administration (2.2)].
1 Indications and Usage (1 INDICATIONS AND USAGE)
- ONIVYDE is indicated, in combination with oxaliplatin, fluorouracil and leucovorin for the first-line treatment of adult patients with metastatic pancreatic adenocarcinoma.
- ONIVYDE is indicated, in combination with fluorouracil and leucovorin, for the treatment of adult patients with metastatic pancreatic adenocarcinoma after disease progression following gemcitabine-based therapy.
Limitations of Use: ONIVYDE is not indicated as a single agent for the treatment of patients with metastatic pancreatic adenocarcinoma. [see Clinical Studies (14)].
12.1 Mechanism of Action
Irinotecan liposome injection is a topoisomerase 1 inhibitor encapsulated in a lipid bilayer vesicle or liposome. Topoisomerase 1 relieves torsional strain in DNA by inducing single-strand breaks. Irinotecan and its active metabolite SN-38 bind reversibly to the topoisomerase 1-DNA complex and prevent re-ligation of the single-strand breaks, leading to exposure time-dependent double-strand DNA damage and cell death. In mice bearing human tumor xenografts, irinotecan liposome administered at irinotecan HCl-equivalent doses 5-fold lower than irinotecan HCl achieved similar intratumoral exposure of SN-38.
5.5 Embryo Fetal Toxicity (5.5 Embryo-Fetal Toxicity)
Based on animal data with irinotecan HCl and the mechanism of action of ONIVYDE, ONIVYDE can cause fetal harm when administered to a pregnant woman. Embryotoxicity and teratogenicity were observed following treatment with irinotecan HCl, at doses resulting in irinotecan exposures lower than those achieved with ONIVYDE 70 mg/m2 in humans, administered to pregnant rats and rabbits during organogenesis. Advise pregnant women of the potential risk to a fetus. Advise females of reproductive potential to use effective contraception during treatment with ONIVYDE and for seven months following the last dose [see Use in Specific Populations (8.1, 8.3), Clinical Pharmacology (12.1)].
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Interstitial lung disease (ILD): Fatal ILD has occurred in patients receiving irinotecan including ONIVYDE. Discontinue ONIVYDE if ILD is diagnosed. (5.3)
- Severe hypersensitivity reaction: Permanently discontinue ONIVYDE for severe hypersensitivity reactions. (5.4, 4)
- Embryo-fetal toxicity: Can cause fetal harm. Advise females of reproductive potential of the potential risk to a fetus and to use effective contraception. (5.5, 8.1, 8.3)
7.1 Strong Cyp3a4 Inducers (7.1 Strong CYP3A4 Inducers)
Following administration of non-liposomal irinotecan (i.e., irinotecan HCl), exposure to irinotecan or its active metabolite, SN-38, is substantially reduced in adult and pediatric patients concomitantly receiving the CYP3A4 enzyme-inducing anticonvulsants phenytoin and strong CYP3A4 inducers. Avoid the use of strong CYP3A4 inducers (e.g., rifampin, phenytoin, carbamazepine, rifabutin, rifapentine, phenobarbital, St. John's wort) if possible. Substitute non-enzyme inducing therapies at least 2 weeks prior to initiation of ONIVYDE therapy [see Clinical Pharmacology (12.3)].
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
- Do not substitute ONIVYDE for other drugs containing irinotecan HCl. (2.1)
- ONIVYDE in combination with oxaliplatin, fluorouracil and leucovorin:
- Recommended dose of ONIVYDE is 50 mg/m2 intravenous infusion over 90 minutes every two weeks. (2.2)
- Recommended starting dose of ONIVYDE in patients homozygous for UGT1A1*28 is 50 mg/m2 every two weeks. (2.2)
- There is no recommended dose of ONIVYDE for patients with serum bilirubin above the upper limit of normal. (2.2)
- ONIVYDE in combination with fluorouracil and leucovorin:
- Recommended dose of ONIVYDE is 70 mg/m2 intravenous infusion over 90 minutes every two weeks. (2.2)
- Recommended starting dose of ONIVYDE in patients homozygous for UGT1A1*28 is 50 mg/m2 every two weeks. (2.2)
- There is no recommended dose of ONIVYDE for patients with serum bilirubin above the upper limit of normal. (2.2)
- Premedicate with a corticosteroid and an anti-emetic 30 minutes prior to ONIVYDE. (2.2)
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Injection: 43 mg/10 mL irinotecan free base as a white to slightly yellow, opaque, liposomal dispersion in a single-dose vial.
6.2 Postmarketing Experience
The following adverse reactions have been identified during post approval use of ONIVYDE. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Immune system disorders: Hypersensitivity (including Anaphylactic reaction and Angioedema)
2.1 Important Use Information
DO NOT SUBSTITUTE ONIVYDE for other drugs containing irinotecan HCl.
5.3 Interstitial Lung Disease
ONIVYDE can cause severe and fatal interstitial lung disease (ILD), including pneumonitis. Postmarketing cases of severe and fatal ILD have been reported with ONIVYDE. Risk factors include pre-existing lung disease, use of pneumotoxic medicinal products, colony stimulating factors or having previously received radiation therapy. Patients with risk factors should be closely monitored for respiratory symptoms before and during ONIVYDE therapy. Withhold ONIVYDE in patients with new or progressive dyspnea, cough, and fever, pending diagnostic evaluation. Discontinue ONIVYDE in patients with a confirmed diagnosis of ILD.
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
- Lactation: Do not breastfeed. (8.2)
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in clinical trials of ONIVYDE cannot be directly compared to rates in clinical trials of other drugs and may not reflect the rates observed in practice.
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise patients of the following:
2.4 Preparation and Administration
ONIVYDE is a hazardous drug. Follow applicable special handling and disposal procedures.1
5.4 Severe Hypersensitivity Reaction
Irinotecan including ONIVYDE can cause severe hypersensitivity reactions, including anaphylactic reactions. Permanently discontinue ONIVYDE in patients who experience a severe hypersensitivity reaction [see Contraindications (4), Adverse reactions (6.2)].
7.2 Strong Cyp3a4 Or Ugt1a1 Inhibitors (7.2 Strong CYP3A4 or UGT1A1 Inhibitors)
Following administration of non-liposomal irinotecan (i.e., irinotecan HCl), patients receiving concomitant ketoconazole, a CYP3A4 and UGT1A1 inhibitor, have increased exposure to irinotecan and its active metabolite SN-38. Co-administration of ONIVYDE with other inhibitors of CYP3A4 (e.g., clarithromycin, indinavir, itraconazole, lopinavir, nefazodone, nelfinavir, ritonavir, saquinavir, telaprevir, voriconazole) or UGT1A1 (e.g., atazanavir, gemfibrozil, indinavir) may increase systemic exposure to irinotecan or SN-38. Avoid the use of strong CYP3A4 or UGT1A1 inhibitors if possible. Discontinue strong CYP3A4 inhibitors at least 1 week prior to starting ONIVYDE therapy [see Clinical Pharmacology (12.3)].
2.3 Dosage Modifications for Adverse Reactions
Recommended dosage modifications for ONIVYDE are in Table 1 and Table 2.
| Toxicity Toxicity grading per NCI CTCAE v5.0.
|
Occurrence | ONIVYDE adjustment in patients receiving 50mg/m2 |
|---|---|---|
|
Grade 3 or 4 Adverse reactions No dosage modification is necessary for asthenia, alopecia and Grade 3 anorexia.
|
Withhold ONIVYDE Upon recovery to ≤ Grade 1 Do not resume until the absolute neutrophil count is ≥2000/mm3 (2×109/L) and the platelet count is ≥100,000/mm3 (100×109/L). ,For Grade ≥3 nausea and vomiting, reduce dose only if occurs despite optimal anti-emetic therapy. ,Refer to the Full Prescribing Information of fluorouracil and oxaliplatin. When ONIVYDE dose is reduced for adverse reactions, reduce fluorouracil (FU) and oxaliplatin doses: for first occurrence, reduce dose to 80% of original dose; for second occurrence, reduce dose to 65% of original dose; for third occurrence, reduce dose to 50% of original dose; discontinue therapy for fourth occurrence. Oxaliplatin may be discontinued if not well tolerated and treatment with ONIVYDE + FU/LV can continue. Maintain original dose level of leucovorin for first, second and third occurrence of toxicity. , resume ONIVYDE at: |
|
| First | 40 mg/m2 | |
| Second | 32.5 mg/m2 | |
| Third | 25 mg/m2 | |
| Fourth | Discontinue ONIVYDE | |
| Grade 3 or 4 Hand foot syndrome | First | Discontinue ONIVYDE |
| Any grade neurocerebellar toxicity | First | Discontinue ONIVYDE |
| Grade ≥ 2 cardiac toxicity | First | Discontinue ONIVYDE |
| Interstitial lung disease | First | Discontinue ONIVYDE |
| Anaphylactic reaction | First | Discontinue ONIVYDE |
| Toxicity NCI CTCAE v4.0 NCI CTCAE v 4.0=National Cancer Institute Common Toxicity Criteria for Adverse Events version 4.0
|
Occurrence | ONIVYDE adjustment in patients receiving 70 mg/m2 | Patients homozygous for UGT1A1*28 without previous increase to 70 mg/m2 |
|---|---|---|---|
| Grade 3 or 4 adverse reactions | Withhold ONIVYDE. Upon recovery to ≤ Grade 1, resume ONIVYDE at: |
||
| First | 50 mg/m2 | 43 mg/m2 | |
| Second | 43 mg/m2 | 35 mg/m2 | |
| Third | Discontinue ONIVYDE | Discontinue ONIVYDE | |
| Interstitial Lung Disease | First | Discontinue ONIVYDE | Discontinue ONIVYDE |
| Anaphylactic Reaction | First | Discontinue ONIVYDE | Discontinue ONIVYDE |
For recommended dose modifications of fluorouracil (FU) or leucovorin (LV), refer to the Full Prescribing Information; refer to Clinical Studies (14).
Warning: Severe Neutropenia and Severe Diarrhea (WARNING: SEVERE NEUTROPENIA AND SEVERE DIARRHEA)
Neutropenia
- Severe and life-threatening neutropenia, including fatal neutropenic sepsis and fatal neutropenic fever, has occurred in patients receiving ONIVYDE in combination with oxaliplatin, fluorouracil and leucovorin and in combination with fluorouracil and leucovorin. Withhold ONIVYDE for absolute neutrophil count below 1500/mm3 or neutropenic fever. Monitor blood cell counts periodically during treatment [see Dosage and Administration (2.2) and Warnings and Precautions (5.1)].
Diarrhea
- Severe and life-threatening diarrhea has occurred in patients receiving ONIVYDE in combination with oxaliplatin, fluorouracil and leucovorin and in combination with fluorouracil and leucovorin. Do not administer ONIVYDE to patients with bowel obstruction. Withhold ONIVYDE for diarrhea of Grade 2-4 severity. Administer loperamide for late diarrhea of any severity. Administer atropine, if not contraindicated, for early diarrhea of any severity [see Dosage and Administration (2.2) and Warnings and Precautions (5.2)].
Principal Display Panel 43 Mg/10 Ml Vial Label (PRINCIPAL DISPLAY PANEL - 43 mg/10 mL Vial Label)
NDC 15054-0043-1
Rx Only
Onivyde®
(irinotecan liposome injection)
43 mg/10 mL
(4.3 mg/mL)
For Intravenous Infusion After Dilution
Single Dose Vial
Discard Unused Portion
Liposomal Formulation
Do Not Substitute For
Irinotecan Hydrochloride
Principal Display Panel 43 Mg/10 Ml Vial Carton (PRINCIPAL DISPLAY PANEL - 43 mg/10 mL Vial Carton)
NDC 15054-0043-1
Rx Only
onivyde®
(irinotecan liposome injection)
43 mg/10 mL
(4.3 mg/mL)
For Intravenous Infusion After Dilution
Warning: Hazardous drug
Single Dose Vial
Discard Unused Portion
Liposomal Formulation
Do Not Substitute For Irinotecan Hydrochloride
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
No studies have been performed to assess the potential of irinotecan liposome for carcinogenicity, genotoxicity or impairment of fertility. Intravenous administration of irinotecan hydrochloride to rats once weekly for 13 weeks followed by a 91-week recovery period resulted in a significant linear trend between irinotecan HCl dosage and the incidence of combined uterine horn endometrial stromal polyps and endometrial stromal sarcomas. Irinotecan HCl was clastogenic both in vitro (chromosome aberrations in Chinese hamster ovary cells) and in vivo (micronucleus test in mice). Neither irinotecan nor its active metabolite, SN-38, was mutagenic in the in vitro Ames assay.
Dedicated fertility studies have not been performed with irinotecan liposome injection. Atrophy of male and female reproductive organs was observed in dogs receiving irinotecan liposome injection every 3 weeks at doses equal to or greater than 15 mg/kg, (approximately 3 times the clinical exposure of irinotecan following administration to ONIVYDE dosed at 70 mg/m2) for a total of 6 doses. No significant adverse effects on fertility and general reproductive performance were observed after intravenous administration of irinotecan HCl in doses of up to 6 mg/kg/day to rats; however, atrophy of male reproductive organs was observed after multiple daily irinotecan HCl doses both in rodents at 20 mg/kg (approximately 0.007 times the clinical irinotecan exposure following ONIVYDE administration at 70 mg/m2) and in dogs at 0.4 mg/kg (0.0007 times the clinical exposure to irinotecan following administration of ONIVYDE).
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:45:59.888275 · Updated: 2026-03-14T22:23:45.136619