c886cfd2-ea8a-47ae-b276-623dfaf498fc
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
OCTINOXATE 7.5%, OCTISALATE 4.0%, ZINC OXIDE 3.5%, OXYBENZONE 2.5%, TITANIUM DIOXIDE 1.1%
Medication Information
Warnings and Precautions
Keep out of eyes. Stop use if irritation occurs. Keep out of reach of children.
Description
OCTINOXATE 7.5%, OCTISALATE 4.0%, ZINC OXIDE 3.5%, OXYBENZONE 2.5%, TITANIUM DIOXIDE 1.1%
Section 50567-7
Apply daily all over face.
Section 51945-4
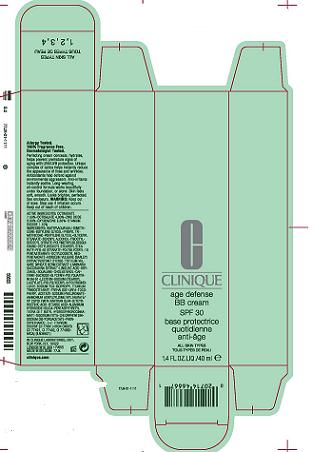
PRINCIPAL DISPLAY PANEL:
Clinique
Age Defense BB Cream
SPF 30
40ml 1.4Floz
Clinique Laboratories, Dist.
New York, NY 1002277jxcarton.jpg
Section 55106-9
OCTINOXATE 7.5%, OCTISALATE 4.0%, ZINC OXIDE 3.5%, OXYBENZONE 2.5%, TITANIUM DIOXIDE 1.1%
Structured Label Content
Warnings and Precautions (34071-1)
Keep out of eyes. Stop use if irritation occurs. Keep out of reach of children.
Section 50567-7 (50567-7)
Apply daily all over face.
Section 51945-4 (51945-4)
PRINCIPAL DISPLAY PANEL:
Clinique
Age Defense BB Cream
SPF 30
40ml 1.4Floz
Clinique Laboratories, Dist.
New York, NY 1002277jxcarton.jpg
Section 55106-9 (55106-9)
OCTINOXATE 7.5%, OCTISALATE 4.0%, ZINC OXIDE 3.5%, OXYBENZONE 2.5%, TITANIUM DIOXIDE 1.1%
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:55:07.162138 · Updated: 2026-03-14T22:52:27.184723