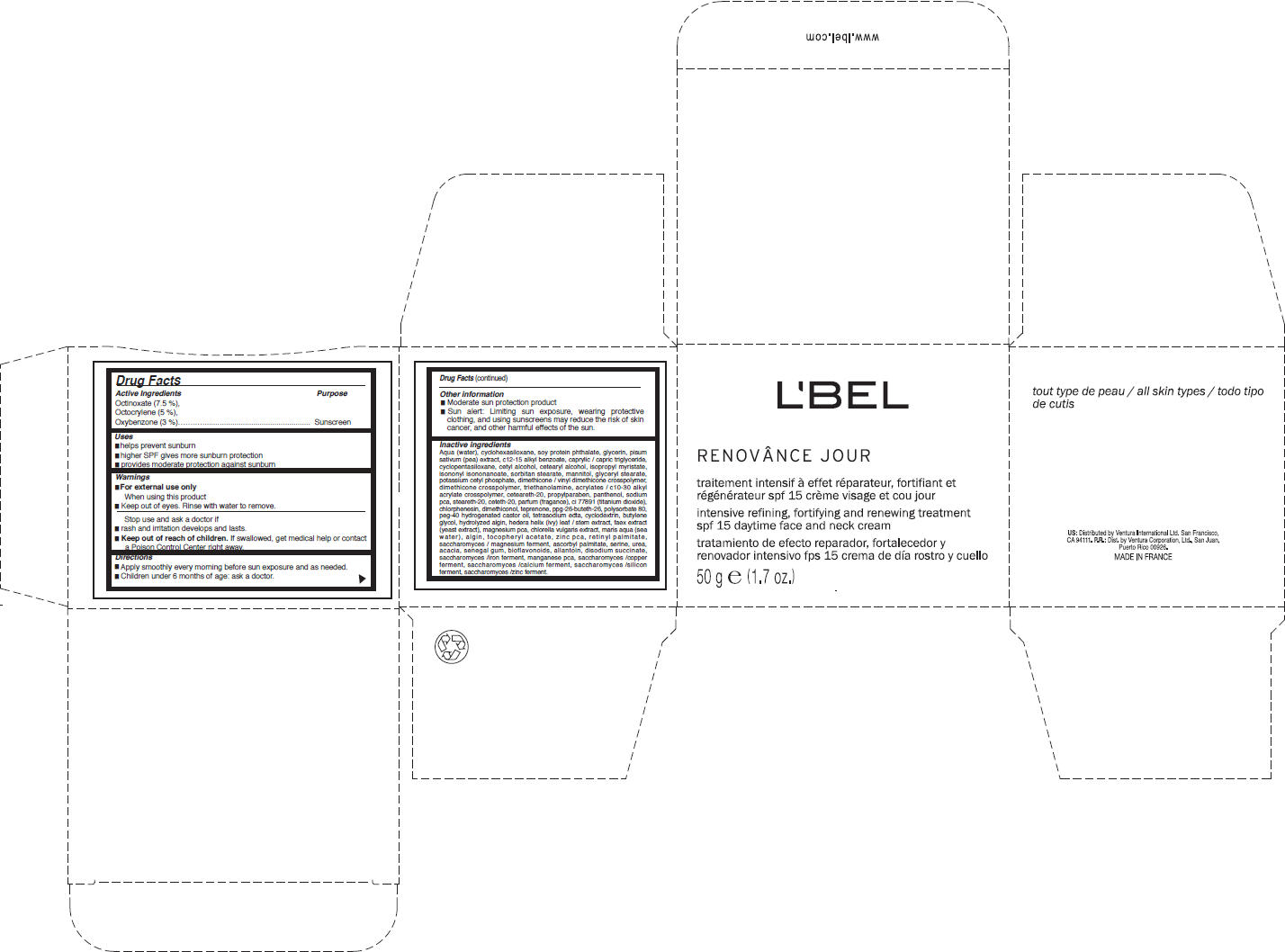
[{"name": "Octinoxate", "unii": "4Y5P7MUD51", "classCode": "ACTIB", "strengthNumerator": "3.75", "strengthDenominator": "50", "strengthNumeratorUnit": "g", "strengthDenominatorUnit": "g"}, {"name": "Octocrylene", "unii": "5A68WGF6WM", "classCode": "ACTIB", "strengthNumerator": "2.5", "strengthDenominator": "50", "strengthNumeratorUnit": "g", "strengthDenominatorUnit": "g"}, {"name": "Oxybenzone", "unii": "95OOS7VE0Y", "classCode": "ACTIB", "strengthNumerator": "1.5", "strengthDenominator": "50", "strengthNumeratorUnit": "g", "strengthDenominatorUnit": "g"}, {"name": "water", "unii": "059QF0KO0R", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "cyclomethicone 6", "unii": "XHK3U310BA", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "glycerin", "unii": "PDC6A3C0OX", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "snow pea", "unii": "84SKC33B1I", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "c12-15 alkyl benzoate", "unii": "A9EJ3J61HQ", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "medium-chain triglycerides", "unii": "C9H2L21V7U", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "cyclomethicone 5", "unii": "0THT5PCI0R", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "cetyl alcohol", "unii": "936JST6JCN", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "cetostearyl alcohol", "unii": "2DMT128M1S", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "isopropyl myristate", "unii": "0RE8K4LNJS", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "isononyl isononanoate", "unii": "S4V5BS6GCX", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "sorbitan monostearate", "unii": "NVZ4I0H58X", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "mannitol", "unii": "3OWL53L36A", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "glyceryl monostearate", "unii": "230OU9XXE4", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "potassium cetyl phosphate", "unii": "03KCY6P7UT", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "trolamine", "unii": "9O3K93S3TK", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "polyoxyl 20 cetostearyl ether", "unii": "YRC528SWUY", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "propylparaben", "unii": "Z8IX2SC1OH", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "panthenol", "unii": "WV9CM0O67Z", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "sodium pyrrolidone carboxylate", "unii": "469OTG57A2", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "steareth-20", "unii": "L0Q8IK9E08", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "ceteth-20", "unii": "I835H2IHHX", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "chlorphenesin", "unii": "I670DAL4SZ", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "titanium dioxide", "unii": "15FIX9V2JP", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "teprenone", "unii": "S8S8451A4O", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "polysorbate 80", "unii": "6OZP39ZG8H", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "polyoxyl 40 hydrogenated castor oil", "unii": "7YC686GQ8F", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "edetate sodium", "unii": "MP1J8420LU", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "butylene glycol", "unii": "3XUS85K0RA", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "kalmia latifolia leaf", "unii": "79N6542N18", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "yeast", "unii": "3NY3SM6B8U", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "vitamin a palmitate", "unii": "1D1K0N0VVC", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "ascorbyl palmitate", "unii": "QN83US2B0N", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "serine", "unii": "452VLY9402", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "urea", "unii": "8W8T17847W", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "acacia", "unii": "5C5403N26O", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "allantoin", "unii": "344S277G0Z", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "sodium succinate anhydrous", "unii": "V8ZGC8ISR3", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "Octinoxate", "unii": "4Y5P7MUD51", "classCode": "ACTIB", "strengthNumerator": "0.375", "strengthDenominator": "5", "strengthNumeratorUnit": "g", "strengthDenominatorUnit": "g"}, {"name": "Octocrylene", "unii": "5A68WGF6WM", "classCode": "ACTIB", "strengthNumerator": "0.25", "strengthDenominator": "5", "strengthNumeratorUnit": "g", "strengthDenominatorUnit": "g"}, {"name": "Oxybenzone", "unii": "95OOS7VE0Y", "classCode": "ACTIB", "strengthNumerator": "0.15", "strengthDenominator": "5", "strengthNumeratorUnit": "g", "strengthDenominatorUnit": "g"}, {"name": "water", "unii": "059QF0KO0R", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "cyclomethicone 6", "unii": "XHK3U310BA", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "glycerin", "unii": "PDC6A3C0OX", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "snow pea", "unii": "84SKC33B1I", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "c12-15 alkyl benzoate", "unii": "A9EJ3J61HQ", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "medium-chain triglycerides", "unii": "C9H2L21V7U", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "cyclomethicone 5", "unii": "0THT5PCI0R", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "cetyl alcohol", "unii": "936JST6JCN", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "cetostearyl alcohol", "unii": "2DMT128M1S", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "isopropyl myristate", "unii": "0RE8K4LNJS", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "isononyl isononanoate", "unii": "S4V5BS6GCX", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "sorbitan monostearate", "unii": "NVZ4I0H58X", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "mannitol", "unii": "3OWL53L36A", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "glyceryl monostearate", "unii": "230OU9XXE4", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "potassium cetyl phosphate", "unii": "03KCY6P7UT", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "trolamine", "unii": "9O3K93S3TK", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "polyoxyl 20 cetostearyl ether", "unii": "YRC528SWUY", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "propylparaben", "unii": "Z8IX2SC1OH", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "panthenol", "unii": "WV9CM0O67Z", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "sodium pyrrolidone carboxylate", "unii": "469OTG57A2", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "steareth-20", "unii": "L0Q8IK9E08", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "ceteth-20", "unii": "I835H2IHHX", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "chlorphenesin", "unii": "I670DAL4SZ", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "titanium dioxide", "unii": "15FIX9V2JP", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "teprenone", "unii": "S8S8451A4O", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "polysorbate 80", "unii": "6OZP39ZG8H", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "polyoxyl 40 hydrogenated castor oil", "unii": "7YC686GQ8F", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "edetate sodium", "unii": "MP1J8420LU", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "butylene glycol", "unii": "3XUS85K0RA", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "kalmia latifolia leaf", "unii": "79N6542N18", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "yeast", "unii": "3NY3SM6B8U", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "vitamin a palmitate", "unii": "1D1K0N0VVC", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "ascorbyl palmitate", "unii": "QN83US2B0N", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "serine", "unii": "452VLY9402", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "urea", "unii": "8W8T17847W", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "acacia", "unii": "5C5403N26O", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "allantoin", "unii": "344S277G0Z", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "sodium succinate anhydrous", "unii": "V8ZGC8ISR3", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "Octinoxate", "unii": "4Y5P7MUD51", "classCode": "ACTIB", "strengthNumerator": "0.075", "strengthDenominator": "1", "strengthNumeratorUnit": "g", "strengthDenominatorUnit": "g"}, {"name": "Octocrylene", "unii": "5A68WGF6WM", "classCode": "ACTIB", "strengthNumerator": "0.05", "strengthDenominator": "1", "strengthNumeratorUnit": "g", "strengthDenominatorUnit": "g"}, {"name": "Oxybenzone", "unii": "95OOS7VE0Y", "classCode": "ACTIB", "strengthNumerator": "0.03", "strengthDenominator": "1", "strengthNumeratorUnit": "g", "strengthDenominatorUnit": "g"}, {"name": "water", "unii": "059QF0KO0R", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "cyclomethicone 6", "unii": "XHK3U310BA", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "glycerin", "unii": "PDC6A3C0OX", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "snow pea", "unii": "84SKC33B1I", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "c12-15 alkyl benzoate", "unii": "A9EJ3J61HQ", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "medium-chain triglycerides", "unii": "C9H2L21V7U", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "cyclomethicone 5", "unii": "0THT5PCI0R", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "cetyl alcohol", "unii": "936JST6JCN", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "cetostearyl alcohol", "unii": "2DMT128M1S", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "isopropyl myristate", "unii": "0RE8K4LNJS", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "isononyl isononanoate", "unii": "S4V5BS6GCX", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "sorbitan monostearate", "unii": "NVZ4I0H58X", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "mannitol", "unii": "3OWL53L36A", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "glyceryl monostearate", "unii": "230OU9XXE4", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "potassium cetyl phosphate", "unii": "03KCY6P7UT", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "trolamine", "unii": "9O3K93S3TK", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "polyoxyl 20 cetostearyl ether", "unii": "YRC528SWUY", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "propylparaben", "unii": "Z8IX2SC1OH", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "panthenol", "unii": "WV9CM0O67Z", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "sodium pyrrolidone carboxylate", "unii": "469OTG57A2", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "steareth-20", "unii": "L0Q8IK9E08", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "ceteth-20", "unii": "I835H2IHHX", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "chlorphenesin", "unii": "I670DAL4SZ", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "titanium dioxide", "unii": "15FIX9V2JP", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "teprenone", "unii": "S8S8451A4O", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "polysorbate 80", "unii": "6OZP39ZG8H", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "polyoxyl 40 hydrogenated castor oil", "unii": "7YC686GQ8F", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "edetate sodium", "unii": "MP1J8420LU", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "butylene glycol", "unii": "3XUS85K0RA", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "kalmia latifolia leaf", "unii": "79N6542N18", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "yeast", "unii": "3NY3SM6B8U", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "vitamin a palmitate", "unii": "1D1K0N0VVC", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "ascorbyl palmitate", "unii": "QN83US2B0N", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "serine", "unii": "452VLY9402", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "urea", "unii": "8W8T17847W", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "acacia", "unii": "5C5403N26O", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "allantoin", "unii": "344S277G0Z", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "sodium succinate anhydrous", "unii": "V8ZGC8ISR3", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}]