These Highlights Do Not Include All The Information Needed To Use
c759668b-5cf1-b237-00c8-a5e4733b6f4c
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
E-Z-DISK is indicated for the evaluation of esophageal patency in adults and pediatric patients aged 12 years and older.
Indications and Usage
E-Z-DISK is indicated for the evaluation of esophageal patency in adults and pediatric patients aged 12 years and older.
Dosage and Administration
The recommended dose of E-Z-DISK in adults and pediatric patients aged 12 years and older is one 700 mg tablet orally during imaging. Swallow one tablet whole with the aid of one or two swallows of water. Do not cut, crush, or chew the tablet. Advise patients to hydrate following the E-Z-DISK imaging procedure [see Warnings and Precautions ( 5.3 )] . E-Z-DISK is formulated to disintegrate within the gastrointestinal (GI) tract. In the event of prolonged retention, consider implementing appropriate interventions.
Warnings and Precautions
Hypersensitivity Reactions: Have emergency equipment and trained personnel immediately available during the procedure. ( 5.1 ) Intra-abdominal Barium Leakage: Barium leakage may occur in conditions such as GI fistula, ulcer, inflammatory bowel disease, appendicitis, diverticulitis, and severe stenosis or obstructing lesions of the GI tract and has been associated with peritonitis and granuloma formation. ( 5.2 ) Baroliths and Bowel Obstruction: Maintain adequate hydration following a barium sulfate procedure and monitor patients at risk for delayed GI transit for development of signs and symptoms of bowel obstruction. ( 5.3 ) Aspiration Pneumonitis: Patients with a history of food aspiration or compromised swallowing mechanism may be at high risk. ( 5.4 )
Contraindications
E-Z-DISK is contraindicated in patients with: Known severe hypersensitivity to barium sulfate or any of the excipients of E-Z-DISK [see Warnings and Precautions ( 5.1 )] Known, suspected, or high risk of perforation of the GI tract such as patients with a recent GI perforation, acute GI hemorrhage or ischemia, toxic megacolon, severe ileus, recent GI surgery or biopsy, acute GI injury, or recent radiotherapy to the pelvis [see Warnings and Precautions ( 5.2 )] Known obstruction of the GI tract [see Warnings and Precautions ( 5.3 )] High risk of aspiration such as patients with known or suspected tracheoesophageal fistula or obtundation [see Warnings and Precautions ( 5.4 )]
Adverse Reactions
The following clinically significant adverse reactions are described elsewhere in the labeling: Hypersensitivity Reactions [see Warnings and Precautions ( 5.1 )] Intra-abdominal Barium Leakage [see Warnings and Precautions ( 5.2 )] Baroliths and Bowel Obstruction [see Warnings and Precautions ( 5.3 )] Aspiration Pneumonitis [see Warnings and Precautions ( 5.4 )] The following adverse reactions associated with the use of E-Z-DISK or other barium sulfate products were identified in postmarketing reports or published clinical studies. Because some of these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or to establish a causal relationship to drug exposure: Cardiovascular disorders : Vasovagal and syncopal episodes Gastrointestinal disorders: Barium sulfate impaction, nausea, vomiting, diarrhea, abdominal cramping Respiratory disorders : Aspiration pneumonitis Adverse Reactions in Pediatric Patients No additional safety signals have been reported in pediatric patients aged 12 years and older.
Storage and Handling
How Supplied E-Z-DISK (barium sulfate) tablets, 700 mg, are white to lightly colored, between 11.5 mm and 13.5 mm (0.45 inch and 0.53 inch) in diameter, flat-sided disks with EZEM inscribed on one side and 778 on the other side supplied in a glass bottle containing 100 tablets (NDC 10361-778-31). Storage and Handling Store at 20°C to 25°C (68°F to 77° F) [see USP controlled room temperature]. Store in original container and protect from moisture.
How Supplied
How Supplied E-Z-DISK (barium sulfate) tablets, 700 mg, are white to lightly colored, between 11.5 mm and 13.5 mm (0.45 inch and 0.53 inch) in diameter, flat-sided disks with EZEM inscribed on one side and 778 on the other side supplied in a glass bottle containing 100 tablets (NDC 10361-778-31). Storage and Handling Store at 20°C to 25°C (68°F to 77° F) [see USP controlled room temperature]. Store in original container and protect from moisture.
Medication Information
Warnings and Precautions
Hypersensitivity Reactions: Have emergency equipment and trained personnel immediately available during the procedure. ( 5.1 ) Intra-abdominal Barium Leakage: Barium leakage may occur in conditions such as GI fistula, ulcer, inflammatory bowel disease, appendicitis, diverticulitis, and severe stenosis or obstructing lesions of the GI tract and has been associated with peritonitis and granuloma formation. ( 5.2 ) Baroliths and Bowel Obstruction: Maintain adequate hydration following a barium sulfate procedure and monitor patients at risk for delayed GI transit for development of signs and symptoms of bowel obstruction. ( 5.3 ) Aspiration Pneumonitis: Patients with a history of food aspiration or compromised swallowing mechanism may be at high risk. ( 5.4 )
Indications and Usage
E-Z-DISK is indicated for the evaluation of esophageal patency in adults and pediatric patients aged 12 years and older.
Dosage and Administration
The recommended dose of E-Z-DISK in adults and pediatric patients aged 12 years and older is one 700 mg tablet orally during imaging. Swallow one tablet whole with the aid of one or two swallows of water. Do not cut, crush, or chew the tablet. Advise patients to hydrate following the E-Z-DISK imaging procedure [see Warnings and Precautions ( 5.3 )] . E-Z-DISK is formulated to disintegrate within the gastrointestinal (GI) tract. In the event of prolonged retention, consider implementing appropriate interventions.
Contraindications
E-Z-DISK is contraindicated in patients with: Known severe hypersensitivity to barium sulfate or any of the excipients of E-Z-DISK [see Warnings and Precautions ( 5.1 )] Known, suspected, or high risk of perforation of the GI tract such as patients with a recent GI perforation, acute GI hemorrhage or ischemia, toxic megacolon, severe ileus, recent GI surgery or biopsy, acute GI injury, or recent radiotherapy to the pelvis [see Warnings and Precautions ( 5.2 )] Known obstruction of the GI tract [see Warnings and Precautions ( 5.3 )] High risk of aspiration such as patients with known or suspected tracheoesophageal fistula or obtundation [see Warnings and Precautions ( 5.4 )]
Adverse Reactions
The following clinically significant adverse reactions are described elsewhere in the labeling: Hypersensitivity Reactions [see Warnings and Precautions ( 5.1 )] Intra-abdominal Barium Leakage [see Warnings and Precautions ( 5.2 )] Baroliths and Bowel Obstruction [see Warnings and Precautions ( 5.3 )] Aspiration Pneumonitis [see Warnings and Precautions ( 5.4 )] The following adverse reactions associated with the use of E-Z-DISK or other barium sulfate products were identified in postmarketing reports or published clinical studies. Because some of these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or to establish a causal relationship to drug exposure: Cardiovascular disorders : Vasovagal and syncopal episodes Gastrointestinal disorders: Barium sulfate impaction, nausea, vomiting, diarrhea, abdominal cramping Respiratory disorders : Aspiration pneumonitis Adverse Reactions in Pediatric Patients No additional safety signals have been reported in pediatric patients aged 12 years and older.
Storage and Handling
How Supplied E-Z-DISK (barium sulfate) tablets, 700 mg, are white to lightly colored, between 11.5 mm and 13.5 mm (0.45 inch and 0.53 inch) in diameter, flat-sided disks with EZEM inscribed on one side and 778 on the other side supplied in a glass bottle containing 100 tablets (NDC 10361-778-31). Storage and Handling Store at 20°C to 25°C (68°F to 77° F) [see USP controlled room temperature]. Store in original container and protect from moisture.
How Supplied
How Supplied E-Z-DISK (barium sulfate) tablets, 700 mg, are white to lightly colored, between 11.5 mm and 13.5 mm (0.45 inch and 0.53 inch) in diameter, flat-sided disks with EZEM inscribed on one side and 778 on the other side supplied in a glass bottle containing 100 tablets (NDC 10361-778-31). Storage and Handling Store at 20°C to 25°C (68°F to 77° F) [see USP controlled room temperature]. Store in original container and protect from moisture.
Description
E-Z-DISK is indicated for the evaluation of esophageal patency in adults and pediatric patients aged 12 years and older.
Section 51945-4
E-Z-Disk Tablets NDC: 10361-778-3
8.1 pregnancy
Risk Summary
Barium sulfate is not absorbed systemically following oral administration, and maternal use is not expected to result in fetal exposure to E-Z-DISK [see Clinical Pharmacology (12.3)]
8.2 lactation
Risk Summary
Barium sulfate is not absorbed systemically by the mother following oral administration, and breastfeeding is not expected to result in exposure of the infant to E-Z-DISK.
11 description
E-Z-DISK (barium sulfate) tablet is a radiographic contrast agent for oral use.
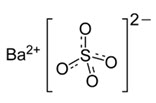
Barium sulfate is designated chemically as BaSO4 with molecular weight of 233.4 g/mol, density of 4.5 g/cm3, and the following chemical structure:
E-Z-DISK is a white to lightly colored, flat-sided disk, between 11.5 mm and 13.5 mm (0.45 inch and 0.53 inch) in diameter. Each tablet contains 700 mg barium sulfate and the following inactive ingredients: confectioner’s sugar, microcrystalline cellulose, corn starch, povidone, croscarmellose sodium, and magnesium stearate.
8.4 pediatric Use
The safety and effectiveness of E-Z-DISK for use in radiographic evaluation of esophageal patency have been established in pediatric patients 12 years and older. Use of E-Z-DISK in this age group for this indication is supported by effectiveness established in studies of adults and pediatric safety data from other barium sulfate products [see Adverse Reactions (6)].
8.5 geriatric Use
Reported clinical experience has not identified differences between elderly and younger patients.
4 contraindications
E-Z-DISK is contraindicated in patients with:
- Known severe hypersensitivity to barium sulfate or any of the excipients of E-Z-DISK [see Warnings and Precautions (5.1)]
- Known, suspected, or high risk of perforation of the GI tract such as patients with a recent GI perforation, acute GI hemorrhage or ischemia, toxic megacolon, severe ileus, recent GI surgery or biopsy, acute GI injury, or recent radiotherapy to the pelvis [see Warnings and Precautions (5.2)]
- Known obstruction of the GI tract [see Warnings and Precautions (5.3)]
- High risk of aspiration such as patients with known or suspected tracheoesophageal fistula or obtundation [see Warnings and Precautions (5.4)]
6 adverse Reactions
The following clinically significant adverse reactions are described elsewhere in the labeling:
- Hypersensitivity Reactions [see Warnings and Precautions (5.1)]
- Intra-abdominal Barium Leakage [see Warnings and Precautions (5.2)]
- Baroliths and Bowel Obstruction [see Warnings and Precautions (5.3)]
- Aspiration Pneumonitis [see Warnings and Precautions (5.4)]
The following adverse reactions associated with the use of E-Z-DISK or other barium sulfate products were identified in postmarketing reports or published clinical studies. Because some of these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or to establish a causal relationship to drug exposure:
Cardiovascular disorders: Vasovagal and syncopal episodes
Gastrointestinal disorders: Barium sulfate impaction, nausea, vomiting, diarrhea, abdominal cramping
Respiratory disorders: Aspiration pneumonitis
Adverse Reactions in Pediatric Patients
No additional safety signals have been reported in pediatric patients aged 12 years and older.
12.2 pharmacodynamics
Barium sulfate has no pharmacological effects.
12.3 pharmacokinetics
Orally administered barium sulfate passes through the gastrointestinal tract in an unchanged form and is absorbed only in insignificant amounts.
1 indications and Usage
E-Z-DISK is indicated for the evaluation of esophageal patency in adults and pediatric patients aged 12 years and older.
12.1 mechanism of Action
E-Z-DISK is formulated to pass through the esophagus into the stomach when the esophageal lumen is greater than 11.5 mm to 13.5 mm (0.45 inch to 0.53 inch) in diameter. Due to its high atomic number, barium is opaque to X-rays and therefore acts as a positive contrast agent for radiographic studies.
5.5 systemic Embolization
Barium sulfate from orally administered E-Z-DISK may intravasate into the venous drainage of the GI tract and enter the circulation as a "barium embolus" leading to potentially fatal complications, which include systemic and pulmonary embolism, disseminated intravascular coagulation, septicemia, and prolonged severe hypotension.
5.4 aspiration Pneumonitis
Oral administration of barium is associated with aspiration pneumonitis, especially in patients with a history of food aspiration or with compromised swallowing mechanism. Vomiting following oral administration of barium sulfate may lead to aspiration pneumonitis. E-Z-DISK is contraindicated in patients with high risk of aspiration such as known or suspected tracheoesophageal fistula or obtundation [see Contraindications (4)].
5 warnings and Precautions
- Hypersensitivity Reactions: Have emergency equipment and trained personnel immediately available during the procedure. (5.1)
- Intra-abdominal Barium Leakage: Barium leakage may occur in conditions such as GI fistula, ulcer, inflammatory bowel disease, appendicitis, diverticulitis, and severe stenosis or obstructing lesions of the GI tract and has been associated with peritonitis and granuloma formation. (5.2)
- Baroliths and Bowel Obstruction: Maintain adequate hydration following a barium sulfate procedure and monitor patients at risk for delayed GI transit for development of signs and symptoms of bowel obstruction. (5.3)
- Aspiration Pneumonitis: Patients with a history of food aspiration or compromised swallowing mechanism may be at high risk. (5.4)
2 dosage and Administration
The recommended dose of E-Z-DISK in adults and pediatric patients aged 12 years and older is one 700 mg tablet orally during imaging.
Swallow one tablet whole with the aid of one or two swallows of water. Do not cut, crush, or chew the tablet.
Advise patients to hydrate following the E-Z-DISK imaging procedure [see Warnings and Precautions (5.3)].
E-Z-DISK is formulated to disintegrate within the gastrointestinal (GI) tract. In the event of prolonged retention, consider implementing appropriate interventions.
3 dosage Forms and Strengths
Tablets: 700 mg of barium sulfate as a white to lightly colored, between 11.5 mm and 13.5 mm (0.45 inch and 0.53 inch) in diameter, flat-sided disk with EZEM inscribed on one side and 778 on the other side.
5.1 hypersensitivity Reactions
E-Z-DISK may induce serious hypersensitivity reactions with manifestations including hypotension, bronchospasm and other respiratory impairments, and dermal reactions including rashes, urticaria and itching. A history of bronchial asthma, atopy, food allergies, or a reaction to a contrast agent may increase the risk for hypersensitivity reactions. E-Z-DISK is contraindicated in patients with known severe hypersensitivity to barium sulfate or any of the excipients of E-Z-DISK [see Contraindications (4)]. Have emergency equipment and trained personnel immediately available during the procedure.
17 patient Counseling Information
Administration Instructions
Instruct patients to swallow E-Z-DISK as a whole tablet with the aid of one or two swallows of water (do not cut, crush, or chew) [see Dosage and Administration (2.2)].
Hypersensitivity Reactions
Advise patients to seek medical attention for any delayed onset of hypersensitivity such as rash, urticaria, or respiratory difficulty [see Warnings and Precautions (5.1)].
Baroliths and Bowel Obstruction
Advise patients to drink a sufficient amount of water to maintain adequate hydration following the E-Z-DISK procedure and to seek medical attention for signs and symptoms of bowel obstruction [see Warnings and Precautions (5.3)].
Manufactured for
Bracco Diagnostics Inc.
Princeton, NJ 08540
by
Confab Laboratories Inc.
Saint Hubert (Quebec) Canada J3Y 3X3
rev. 08/25 301871-02
5.2 intra Abdominal Barium Leakage
Barium leakage from the GI tract has been associated with peritonitis and granuloma formation. Barium sulfate from orally administered E-Z-DISK may leak in the presence of conditions such as carcinomas, GI fistula, inflammatory bowel disease, gastric or duodenal ulcer, appendicitis, or diverticulitis, and in patients with severe stenosis of the GI tract, especially if it is distal to the stomach. E-Z-DISK is contraindicated in patients with known, suspected, or high risk of perforation of the GI tract [see Contraindications (4)].
5.3 baroliths and Bowel Obstruction
Barium sulfate from orally administered E-Z-DISK may accumulate in the GI tract, causing obstruction or impaction with development of baroliths (inspissated barium associated with feces) and may lead to abdominal pain, appendicitis, or perforation. Patients with the following are at higher risk for developing obstruction or baroliths: severe stenosis at any level of the GI tract, impaired GI motility, electrolyte imbalance, dehydration, low residue diet, medications that delay GI motility, constipation, pediatric patients with cystic fibrosis or Hirschsprung disease, and advanced age. E-Z-DISK is contraindicated in patients with known obstruction of the GI tract [see Contraindications (4)]. To reduce the risk of delayed GI transit and obstruction, maintain adequate hydration after the E-Z-DISK procedure. Monitor patients at risk for delayed gastrointestinal transit for development of signs and symptoms of bowel obstruction.
16 how Supplied/storage and Handling
How Supplied
E-Z-DISK (barium sulfate) tablets, 700 mg, are white to lightly colored, between 11.5 mm and 13.5 mm (0.45 inch and 0.53 inch) in diameter, flat-sided disks with EZEM inscribed on one side and 778 on the other side supplied in a glass bottle containing 100 tablets (NDC 10361-778-31).
Storage and Handling
Store at 20°C to 25°C (68°F to 77° F) [see USP controlled room temperature]. Store in original container and protect from moisture.
13.1 carcinogenesis, Mutagenesis, Impairment of Fertility
No animal studies have been performed to evaluate the carcinogenic potential of barium sulfate or potential effects on fertility.
Structured Label Content
Section 51945-4 (51945-4)
E-Z-Disk Tablets NDC: 10361-778-3
8.1 pregnancy (8.1 Pregnancy)
Risk Summary
Barium sulfate is not absorbed systemically following oral administration, and maternal use is not expected to result in fetal exposure to E-Z-DISK [see Clinical Pharmacology (12.3)]
8.2 lactation (8.2 Lactation)
Risk Summary
Barium sulfate is not absorbed systemically by the mother following oral administration, and breastfeeding is not expected to result in exposure of the infant to E-Z-DISK.
11 description (11 DESCRIPTION)
E-Z-DISK (barium sulfate) tablet is a radiographic contrast agent for oral use.
Barium sulfate is designated chemically as BaSO4 with molecular weight of 233.4 g/mol, density of 4.5 g/cm3, and the following chemical structure:
E-Z-DISK is a white to lightly colored, flat-sided disk, between 11.5 mm and 13.5 mm (0.45 inch and 0.53 inch) in diameter. Each tablet contains 700 mg barium sulfate and the following inactive ingredients: confectioner’s sugar, microcrystalline cellulose, corn starch, povidone, croscarmellose sodium, and magnesium stearate.
8.4 pediatric Use (8.4 Pediatric Use)
The safety and effectiveness of E-Z-DISK for use in radiographic evaluation of esophageal patency have been established in pediatric patients 12 years and older. Use of E-Z-DISK in this age group for this indication is supported by effectiveness established in studies of adults and pediatric safety data from other barium sulfate products [see Adverse Reactions (6)].
8.5 geriatric Use (8.5 Geriatric use)
Reported clinical experience has not identified differences between elderly and younger patients.
4 contraindications (4 CONTRAINDICATIONS)
E-Z-DISK is contraindicated in patients with:
- Known severe hypersensitivity to barium sulfate or any of the excipients of E-Z-DISK [see Warnings and Precautions (5.1)]
- Known, suspected, or high risk of perforation of the GI tract such as patients with a recent GI perforation, acute GI hemorrhage or ischemia, toxic megacolon, severe ileus, recent GI surgery or biopsy, acute GI injury, or recent radiotherapy to the pelvis [see Warnings and Precautions (5.2)]
- Known obstruction of the GI tract [see Warnings and Precautions (5.3)]
- High risk of aspiration such as patients with known or suspected tracheoesophageal fistula or obtundation [see Warnings and Precautions (5.4)]
6 adverse Reactions (6 ADVERSE REACTIONS)
The following clinically significant adverse reactions are described elsewhere in the labeling:
- Hypersensitivity Reactions [see Warnings and Precautions (5.1)]
- Intra-abdominal Barium Leakage [see Warnings and Precautions (5.2)]
- Baroliths and Bowel Obstruction [see Warnings and Precautions (5.3)]
- Aspiration Pneumonitis [see Warnings and Precautions (5.4)]
The following adverse reactions associated with the use of E-Z-DISK or other barium sulfate products were identified in postmarketing reports or published clinical studies. Because some of these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or to establish a causal relationship to drug exposure:
Cardiovascular disorders: Vasovagal and syncopal episodes
Gastrointestinal disorders: Barium sulfate impaction, nausea, vomiting, diarrhea, abdominal cramping
Respiratory disorders: Aspiration pneumonitis
Adverse Reactions in Pediatric Patients
No additional safety signals have been reported in pediatric patients aged 12 years and older.
12.2 pharmacodynamics (12.2 Pharmacodynamics)
Barium sulfate has no pharmacological effects.
12.3 pharmacokinetics (12.3 Pharmacokinetics)
Orally administered barium sulfate passes through the gastrointestinal tract in an unchanged form and is absorbed only in insignificant amounts.
1 indications and Usage (1 INDICATIONS AND USAGE)
E-Z-DISK is indicated for the evaluation of esophageal patency in adults and pediatric patients aged 12 years and older.
12.1 mechanism of Action (12.1 Mechanism of Action)
E-Z-DISK is formulated to pass through the esophagus into the stomach when the esophageal lumen is greater than 11.5 mm to 13.5 mm (0.45 inch to 0.53 inch) in diameter. Due to its high atomic number, barium is opaque to X-rays and therefore acts as a positive contrast agent for radiographic studies.
5.5 systemic Embolization (5.5 Systemic Embolization)
Barium sulfate from orally administered E-Z-DISK may intravasate into the venous drainage of the GI tract and enter the circulation as a "barium embolus" leading to potentially fatal complications, which include systemic and pulmonary embolism, disseminated intravascular coagulation, septicemia, and prolonged severe hypotension.
5.4 aspiration Pneumonitis (5.4 Aspiration Pneumonitis)
Oral administration of barium is associated with aspiration pneumonitis, especially in patients with a history of food aspiration or with compromised swallowing mechanism. Vomiting following oral administration of barium sulfate may lead to aspiration pneumonitis. E-Z-DISK is contraindicated in patients with high risk of aspiration such as known or suspected tracheoesophageal fistula or obtundation [see Contraindications (4)].
5 warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Hypersensitivity Reactions: Have emergency equipment and trained personnel immediately available during the procedure. (5.1)
- Intra-abdominal Barium Leakage: Barium leakage may occur in conditions such as GI fistula, ulcer, inflammatory bowel disease, appendicitis, diverticulitis, and severe stenosis or obstructing lesions of the GI tract and has been associated with peritonitis and granuloma formation. (5.2)
- Baroliths and Bowel Obstruction: Maintain adequate hydration following a barium sulfate procedure and monitor patients at risk for delayed GI transit for development of signs and symptoms of bowel obstruction. (5.3)
- Aspiration Pneumonitis: Patients with a history of food aspiration or compromised swallowing mechanism may be at high risk. (5.4)
2 dosage and Administration (2 DOSAGE AND ADMINISTRATION)
The recommended dose of E-Z-DISK in adults and pediatric patients aged 12 years and older is one 700 mg tablet orally during imaging.
Swallow one tablet whole with the aid of one or two swallows of water. Do not cut, crush, or chew the tablet.
Advise patients to hydrate following the E-Z-DISK imaging procedure [see Warnings and Precautions (5.3)].
E-Z-DISK is formulated to disintegrate within the gastrointestinal (GI) tract. In the event of prolonged retention, consider implementing appropriate interventions.
3 dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Tablets: 700 mg of barium sulfate as a white to lightly colored, between 11.5 mm and 13.5 mm (0.45 inch and 0.53 inch) in diameter, flat-sided disk with EZEM inscribed on one side and 778 on the other side.
5.1 hypersensitivity Reactions (5.1 Hypersensitivity Reactions)
E-Z-DISK may induce serious hypersensitivity reactions with manifestations including hypotension, bronchospasm and other respiratory impairments, and dermal reactions including rashes, urticaria and itching. A history of bronchial asthma, atopy, food allergies, or a reaction to a contrast agent may increase the risk for hypersensitivity reactions. E-Z-DISK is contraindicated in patients with known severe hypersensitivity to barium sulfate or any of the excipients of E-Z-DISK [see Contraindications (4)]. Have emergency equipment and trained personnel immediately available during the procedure.
17 patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Administration Instructions
Instruct patients to swallow E-Z-DISK as a whole tablet with the aid of one or two swallows of water (do not cut, crush, or chew) [see Dosage and Administration (2.2)].
Hypersensitivity Reactions
Advise patients to seek medical attention for any delayed onset of hypersensitivity such as rash, urticaria, or respiratory difficulty [see Warnings and Precautions (5.1)].
Baroliths and Bowel Obstruction
Advise patients to drink a sufficient amount of water to maintain adequate hydration following the E-Z-DISK procedure and to seek medical attention for signs and symptoms of bowel obstruction [see Warnings and Precautions (5.3)].
Manufactured for
Bracco Diagnostics Inc.
Princeton, NJ 08540
by
Confab Laboratories Inc.
Saint Hubert (Quebec) Canada J3Y 3X3
rev. 08/25 301871-02
5.2 intra Abdominal Barium Leakage (5.2 Intra-abdominal Barium Leakage)
Barium leakage from the GI tract has been associated with peritonitis and granuloma formation. Barium sulfate from orally administered E-Z-DISK may leak in the presence of conditions such as carcinomas, GI fistula, inflammatory bowel disease, gastric or duodenal ulcer, appendicitis, or diverticulitis, and in patients with severe stenosis of the GI tract, especially if it is distal to the stomach. E-Z-DISK is contraindicated in patients with known, suspected, or high risk of perforation of the GI tract [see Contraindications (4)].
5.3 baroliths and Bowel Obstruction (5.3 Baroliths and Bowel Obstruction)
Barium sulfate from orally administered E-Z-DISK may accumulate in the GI tract, causing obstruction or impaction with development of baroliths (inspissated barium associated with feces) and may lead to abdominal pain, appendicitis, or perforation. Patients with the following are at higher risk for developing obstruction or baroliths: severe stenosis at any level of the GI tract, impaired GI motility, electrolyte imbalance, dehydration, low residue diet, medications that delay GI motility, constipation, pediatric patients with cystic fibrosis or Hirschsprung disease, and advanced age. E-Z-DISK is contraindicated in patients with known obstruction of the GI tract [see Contraindications (4)]. To reduce the risk of delayed GI transit and obstruction, maintain adequate hydration after the E-Z-DISK procedure. Monitor patients at risk for delayed gastrointestinal transit for development of signs and symptoms of bowel obstruction.
16 how Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
How Supplied
E-Z-DISK (barium sulfate) tablets, 700 mg, are white to lightly colored, between 11.5 mm and 13.5 mm (0.45 inch and 0.53 inch) in diameter, flat-sided disks with EZEM inscribed on one side and 778 on the other side supplied in a glass bottle containing 100 tablets (NDC 10361-778-31).
Storage and Handling
Store at 20°C to 25°C (68°F to 77° F) [see USP controlled room temperature]. Store in original container and protect from moisture.
13.1 carcinogenesis, Mutagenesis, Impairment of Fertility (13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility)
No animal studies have been performed to evaluate the carcinogenic potential of barium sulfate or potential effects on fertility.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:50:01.314380 · Updated: 2026-03-14T22:35:35.043059