Cold Sore Fast-acting Pain Relief
c7511322-dd6c-441c-bf7d-ad4f0c14956a
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Drug Facts
Medication Information
Description
Drug Facts
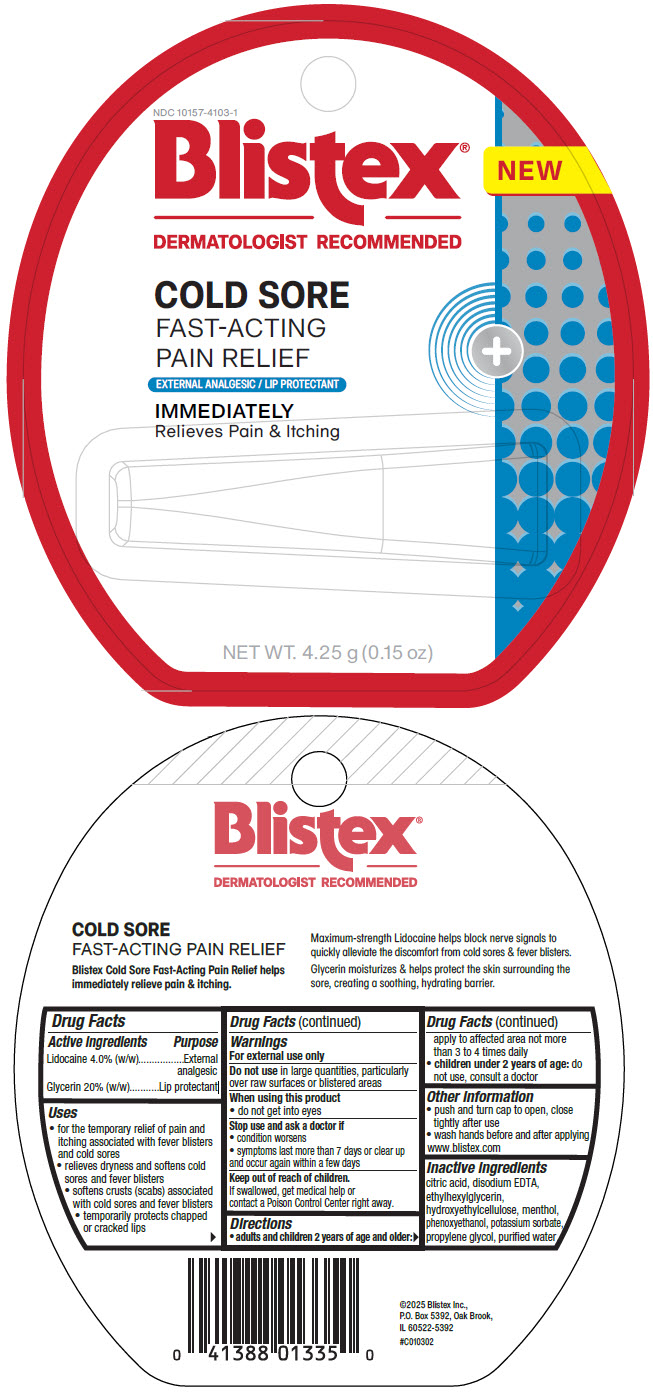
Uses
- for the temporary relief of pain and itching associated with fever blisters and cold sores
- relieves dryness and softens cold sores and fever blisters
- softens crusts (scabs) associated with cold sores and fever blisters
- temporarily protects chapped or cracked lips
Section 42229-5
Drug Facts
Section 50565-1
Keep out of reach of children.
If swallowed, get medical help or contact a Poison Control Center right away.
Section 50570-1
Do not use in large quantities, particularly over raw surfaces or blistered areas
Section 55105-1
| Active ingredients | Purpose |
|---|---|
| Lidocaine 4.0% (w/w) | External analgesic |
| Glycerin 20% (w/w) | Lip protectant |
Warnings
For external use only
Directions
- adults and children 2 years of age and older: apply to affected area not more than 3 to 4 times daily
- children under 2 years of age: do not use, consult a doctor
Other Information
- push and turn cap to open, close tightly after use
- wash hands before and after applying www.blistex.com
Inactive Ingredients
citric acid, disodium EDTA, ethylhexylglycerin, hydroxyethylcellulose, menthol, phenoxyethanol, potassium sorbate, propylene glycol, purified water
When Using This Product
- do not get into eyes
Stop Use and Ask A Doctor If
- condition worsens
- symptoms last more than 7 days or clear up and occur again within a few days
Principal Display Panel 4.25 G Tube Label
NDC 10157-4103-1
Blistex®
DERMATOLOGIST RECOMMENDED
NEW
COLD SORE
FAST-ACTING
PAIN RELIEF
EXTERNAL ANALGESIC / LIP PROTECTANT
IMMEDIATELY
Relieves Pain & Itching
NET WT. 4.25 g (0.15 oz)
Structured Label Content
Uses
- for the temporary relief of pain and itching associated with fever blisters and cold sores
- relieves dryness and softens cold sores and fever blisters
- softens crusts (scabs) associated with cold sores and fever blisters
- temporarily protects chapped or cracked lips
Section 42229-5 (42229-5)
Drug Facts
Section 50565-1 (50565-1)
Keep out of reach of children.
If swallowed, get medical help or contact a Poison Control Center right away.
Section 50570-1 (50570-1)
Do not use in large quantities, particularly over raw surfaces or blistered areas
Section 55105-1 (55105-1)
| Active ingredients | Purpose |
|---|---|
| Lidocaine 4.0% (w/w) | External analgesic |
| Glycerin 20% (w/w) | Lip protectant |
Warnings
For external use only
Directions
- adults and children 2 years of age and older: apply to affected area not more than 3 to 4 times daily
- children under 2 years of age: do not use, consult a doctor
Other Information (Other information)
- push and turn cap to open, close tightly after use
- wash hands before and after applying www.blistex.com
Inactive Ingredients (Inactive ingredients)
citric acid, disodium EDTA, ethylhexylglycerin, hydroxyethylcellulose, menthol, phenoxyethanol, potassium sorbate, propylene glycol, purified water
When Using This Product (When using this product)
- do not get into eyes
Stop Use and Ask A Doctor If (Stop use and ask a doctor if)
- condition worsens
- symptoms last more than 7 days or clear up and occur again within a few days
Principal Display Panel 4.25 G Tube Label (PRINCIPAL DISPLAY PANEL - 4.25 g Tube Label)
NDC 10157-4103-1
Blistex®
DERMATOLOGIST RECOMMENDED
NEW
COLD SORE
FAST-ACTING
PAIN RELIEF
EXTERNAL ANALGESIC / LIP PROTECTANT
IMMEDIATELY
Relieves Pain & Itching
NET WT. 4.25 g (0.15 oz)
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:06:36.763644 · Updated: 2026-03-14T23:09:28.158875