c73953ab-09fa-4116-82c8-8f9d6f9d679e
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
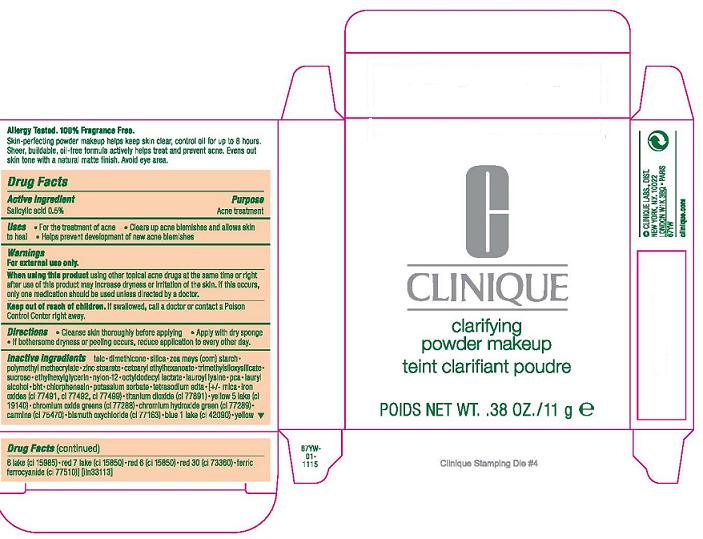
ACTIVE INGREDIENT : SALICYLIC ACID 0.5%
Medication Information
Warnings and Precautions
WARNINGS: FOR EXTERNAL USE ONLY.
Indications and Usage
INDICATIONS: FOR THE TREATMENT OF ACNE
Dosage and Administration
DIRECTIONS
- CLEANSE SKIN THOROUGHLY BEFORE APPLYING
- COVER THE ENTIRE AFFECTED AREA WITH ONE APPLICATION
- GRADUALLY INCREASE TO 2 OR 3 TIMES DAILY IF NEEDED OR AS DIRECTED BY A DOCTOR
- IF BOTHERSOME DRYNESS OR PEELING OCCURS, REDUCE APPLICATION TO EVERY OTHER DAY.
Description
ACTIVE INGREDIENT : SALICYLIC ACID 0.5%
Section 50565-1
KEEP OUT OF REACH OF CHILDREN. IF SWALLOWED, GET MEDICAL HELP OR CONTACT A POISON CONTROL CENTER RIGHT AWAY.
Section 50567-7
WHEN USING THIS PRODUCT: USING OTHER TOPICAL ACNE DRUGS AT THE SAME TIME OR RIGHT AFTER USE OF THIS PRODUCT MAY INCREASE DRYNESS OR IRRITATION OF THE SKIN. IF THIS OCCURS, ONLY ONE MEDICATION SHOULD BE USED UNLESS DIRECTED BY A DOCTOR.
Section 51727-6
INACTIVE INGREDIENTS:
talc [] dimethicone [] silica [] zea mays (corn) starch [] polymethyl methacrylate [] zinc stearate [] cetearyl ethylhexanoate [] trimethylsiloxysilicate [] sucrose [] ethylhexylglycerin [] nylon-12 [] octyldodecyl lactate [] lauroyl lysine [] pca [] lauryl alcohol [] bht [] chlorphenesin [] potassium sorbate [] tetrasodium edta [] [+/- mica [] iron oxides (ci 77491, ci 77492, ci 77499) [] titanium dioxide (ci 77891) [] yellow 5 lake (ci 19140) [] chromium oxide greens (ci 77288) [] chromium hydroxide green (ci 77289) [] carmine (ci 75470) [] bismuth oxychloride (ci 77163) [] blue 1 lake (ci 42090) [] yellow 6 lake (ci 15985) [] red 7 lake (ci 15850) [] red 6 (ci 15850) [] red 30 (ci 73360) [] ferric ferrocyanide (ci 77510
Section 51945-4
PRINCIPLE DISPLAY PANEL:
CLINIQUE
CLARIFYING POWDER MAKEUP
NET WT. .38 OZ/11 g
clinique labs dist
new york, ny 10022
Section 55105-1
PURPOSE: ACNE TREATMENT
USE: FOR THE TREATMENT OF ACNE
CLEARS UP ACNE BLEMISHES AND ALLOWS SKIN TO HEAL
HELPS PREVENT DEVELOPMENT OF NEW ACNE BLEMISHES
Section 55106-9
ACTIVE INGREDIENT: SALICYLIC ACID 0.5%
Structured Label Content
Indications and Usage (34067-9)
INDICATIONS: FOR THE TREATMENT OF ACNE
Dosage and Administration (34068-7)
DIRECTIONS
- CLEANSE SKIN THOROUGHLY BEFORE APPLYING
- COVER THE ENTIRE AFFECTED AREA WITH ONE APPLICATION
- GRADUALLY INCREASE TO 2 OR 3 TIMES DAILY IF NEEDED OR AS DIRECTED BY A DOCTOR
- IF BOTHERSOME DRYNESS OR PEELING OCCURS, REDUCE APPLICATION TO EVERY OTHER DAY.
Warnings and Precautions (34071-1)
WARNINGS: FOR EXTERNAL USE ONLY.
Section 50565-1 (50565-1)
KEEP OUT OF REACH OF CHILDREN. IF SWALLOWED, GET MEDICAL HELP OR CONTACT A POISON CONTROL CENTER RIGHT AWAY.
Section 50567-7 (50567-7)
WHEN USING THIS PRODUCT: USING OTHER TOPICAL ACNE DRUGS AT THE SAME TIME OR RIGHT AFTER USE OF THIS PRODUCT MAY INCREASE DRYNESS OR IRRITATION OF THE SKIN. IF THIS OCCURS, ONLY ONE MEDICATION SHOULD BE USED UNLESS DIRECTED BY A DOCTOR.
Section 51727-6 (51727-6)
INACTIVE INGREDIENTS:
talc [] dimethicone [] silica [] zea mays (corn) starch [] polymethyl methacrylate [] zinc stearate [] cetearyl ethylhexanoate [] trimethylsiloxysilicate [] sucrose [] ethylhexylglycerin [] nylon-12 [] octyldodecyl lactate [] lauroyl lysine [] pca [] lauryl alcohol [] bht [] chlorphenesin [] potassium sorbate [] tetrasodium edta [] [+/- mica [] iron oxides (ci 77491, ci 77492, ci 77499) [] titanium dioxide (ci 77891) [] yellow 5 lake (ci 19140) [] chromium oxide greens (ci 77288) [] chromium hydroxide green (ci 77289) [] carmine (ci 75470) [] bismuth oxychloride (ci 77163) [] blue 1 lake (ci 42090) [] yellow 6 lake (ci 15985) [] red 7 lake (ci 15850) [] red 6 (ci 15850) [] red 30 (ci 73360) [] ferric ferrocyanide (ci 77510
Section 51945-4 (51945-4)
PRINCIPLE DISPLAY PANEL:
CLINIQUE
CLARIFYING POWDER MAKEUP
NET WT. .38 OZ/11 g
clinique labs dist
new york, ny 10022
Section 55105-1 (55105-1)
PURPOSE: ACNE TREATMENT
USE: FOR THE TREATMENT OF ACNE
CLEARS UP ACNE BLEMISHES AND ALLOWS SKIN TO HEAL
HELPS PREVENT DEVELOPMENT OF NEW ACNE BLEMISHES
Section 55106-9 (55106-9)
ACTIVE INGREDIENT: SALICYLIC ACID 0.5%
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:55:12.138759 · Updated: 2026-03-14T22:52:34.752454