Drug Facts
c69b563d-ea9d-4ebc-b9ca-b84bd4e0de85
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Ethyl Alcohol 62% w/w (equivalent to 70% v/v)
Purpose
Antiseptic handwash
Medication Information
Purpose
Antiseptic handwash
Description
Ethyl Alcohol 62% w/w (equivalent to 70% v/v)
Uses
- For handwashing to decrease bacteria on skin
Section 50565-1
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center right away.
Section 51727-6
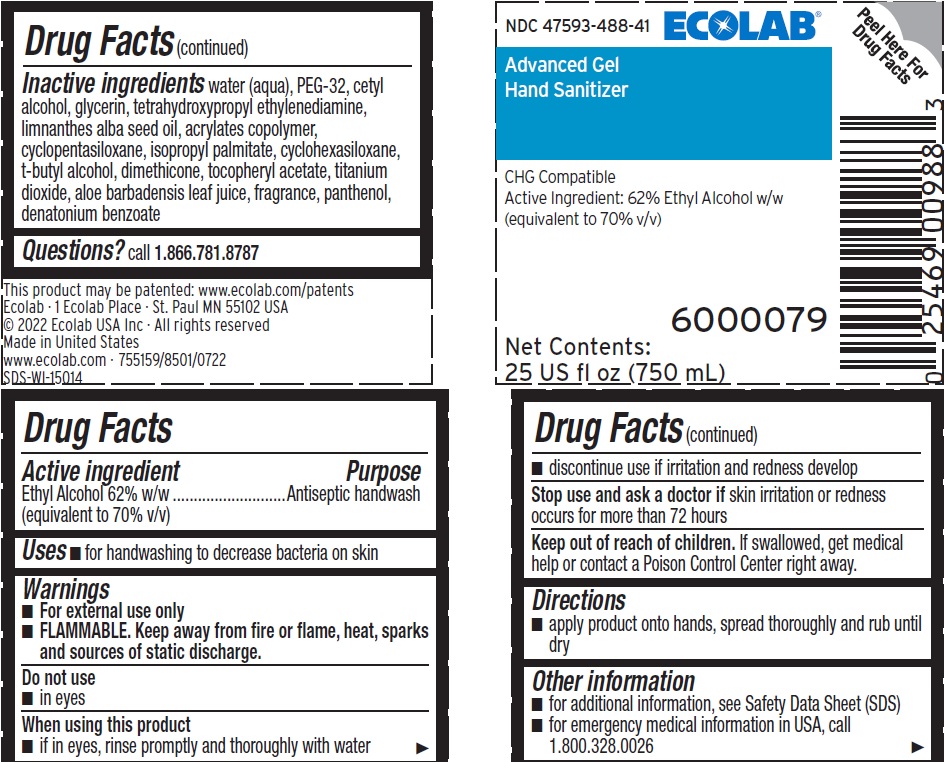
Inactive ingredients Water (Aqua), PEG-32, Cetyl Alcohol, Glycerin, Tetrahydroxypropyl Ethylenediamine, Limnanthes Alba Seed Oil , Acrylates Copolymer, Cyclopentasiloxane, Isopropyl Palmitate, Cyclohexasiloxane, t-Butyl Alcohol, Dimethicone, Tocopheryl Acetate, Titanium Dioxide, Aloe Barbadensis Leaf Juice, fragrance, Panthenol, Denatonium benzoate
Section 53413-1
Questions? call 1.866.781.8787
Warnings
- For external use only
- FLAMMABLE. Keep away from fire or flame, heat, sparks and sources of static discharge.
Directions
- Apply product onto hands, spread thoroughly and rub until dry
Do Not Use
- In eyes
Active Ingredient
Ethyl Alcohol 62% w/w (equivalent to 70% v/v)
Other Information
- for additional information, see Safety Data Sheet (SDS)
- for emergency medical information in USA, call 1.800.328.0026
When Using This Product
- If in eyes, rinse promptly and thoroughly with water
- Discontinue use if irritation and redness develop
Stop Use and Ask A Doctor If
Skin irritation or redness occurs for more than 72 hours
Representative Label and Principal Display Panel
NDC 47593-488-41 ECOLAB
Advanced Gel
Hand Sanitizer
CHG Compatible
Active Ingredient: 62% Ethyl Alcohol w/w
(equivalent to 70% v/v)
6000079
Net Contents:
25 US fl oz (750 mL)
This product may be patented: www.ecolab.com/patents
Ecolab · 1 Ecolab Place · St. Paul MN 55102 USA
© 2022 Ecolab USA Inc · All rights reserved
Made in United States
www.ecolab.com · 755159/8501/0722
SDS-WI-15014
Structured Label Content
Uses
- For handwashing to decrease bacteria on skin
Section 50565-1 (50565-1)
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center right away.
Section 51727-6 (51727-6)
Inactive ingredients Water (Aqua), PEG-32, Cetyl Alcohol, Glycerin, Tetrahydroxypropyl Ethylenediamine, Limnanthes Alba Seed Oil , Acrylates Copolymer, Cyclopentasiloxane, Isopropyl Palmitate, Cyclohexasiloxane, t-Butyl Alcohol, Dimethicone, Tocopheryl Acetate, Titanium Dioxide, Aloe Barbadensis Leaf Juice, fragrance, Panthenol, Denatonium benzoate
Section 53413-1 (53413-1)
Questions? call 1.866.781.8787
Purpose
Antiseptic handwash
Warnings
- For external use only
- FLAMMABLE. Keep away from fire or flame, heat, sparks and sources of static discharge.
Directions
- Apply product onto hands, spread thoroughly and rub until dry
Do Not Use (Do not use)
- In eyes
Active Ingredient (Active ingredient)
Ethyl Alcohol 62% w/w (equivalent to 70% v/v)
Other Information (Other information)
- for additional information, see Safety Data Sheet (SDS)
- for emergency medical information in USA, call 1.800.328.0026
When Using This Product (When using this product)
- If in eyes, rinse promptly and thoroughly with water
- Discontinue use if irritation and redness develop
Stop Use and Ask A Doctor If (Stop use and ask a doctor if)
Skin irritation or redness occurs for more than 72 hours
Representative Label and Principal Display Panel (Representative label and principal display panel)
NDC 47593-488-41 ECOLAB
Advanced Gel
Hand Sanitizer
CHG Compatible
Active Ingredient: 62% Ethyl Alcohol w/w
(equivalent to 70% v/v)
6000079
Net Contents:
25 US fl oz (750 mL)
This product may be patented: www.ecolab.com/patents
Ecolab · 1 Ecolab Place · St. Paul MN 55102 USA
© 2022 Ecolab USA Inc · All rights reserved
Made in United States
www.ecolab.com · 755159/8501/0722
SDS-WI-15014
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:07:35.416818 · Updated: 2026-03-14T23:11:11.874697