tobramycin ophthalmic solution usp, 0.3 %
c6435b8f-bb6e-45e0-a52b-3087fd043b5a
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Indications and Usage
Tobramycin ophthalmic solution 0.3% is a topical antibiotic indicated in the treatment of external infections of the eye and its adnexa caused by susceptible bacteria. Appropriate monitoring of bacterial response to topical antibiotic therapy should accompany the use of tobramycin ophthalmic solution 0.3%. Clinical studies have shown tobramycin to be safe and effective for use in children.
Dosage and Administration
In mild to moderate disease, instill 1 or 2 drops into the affected eye(s) every 4 hours. In severe infections, instill 2 drops into the eye(s) hourly until improvement, following which treatment should be reduced prior to discontinuation.
Contraindications
Tobramycin ophthalmic solution 0.3% is contraindicated in patients with known hypersensitivity to any of its components.
Adverse Reactions
The most frequent adverse reactions to tobramycin ophthalmic solution 0.3% are hypersensitivity and localized ocular toxicity, including lid itching and swelling, and conjunctival erythema. These reactions occur in less than three of 100 patients treated with tobramycin ophthalmic solution. Postmarketing Experience: Additional adverse reactions identified from post-marketing use include anaphylactic reaction, Stevens-Johnson syndrome, and erythema multiforme. The following additional adverse reactions have been reported with systemic aminoglycosides: Neurotoxicity, ototoxicity and nephrotoxicity have occurred in patients receiving systemic aminoglycoside therapy. Aminoglycosides may aggravate muscle weakness in patients with known or suspected neuromuscular disorders, such as myasthenia gravis or Parkinson’s disease, because of their potential effect on neuromuscular function. To report SUSPECTED ADVERSE REACTIONS, contact Jvet Pharmaceuticals, LLC at 1-727-292-1166 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
How Supplied
5 mL sterile solution supplied in opaque white low density polyethylene bottle which is closed with natural low density polyethylene nozzle and then with Tan colored high density polyethylene cap as follows: 5 mL containing tobramycin 0.3% (3 mg/mL)……….. NDC 86211-110-01 Storage: Store at 2°C to 25°C (36°F to 77°F). After opening, Tobramycin Ophthalmic Solution, USP 0.3% can be used until the expiration date on the bottle. Distributed by: Jvet Pharmaceuticals LLC 6925112th Circle North, Suite 101 Largo, FL 33773 Revised: May 2025 L110I-VTN R-2505 747 1313001232-00
Description
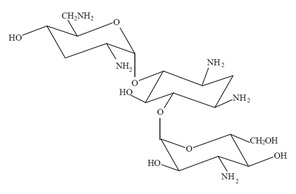
Tobramycin ophthalmic solution USP, 0.3% is a sterile topical ophthalmic antibiotic formulation prepared specifically for topical therapy of external ophthalmic infections. Each mL of tobramycin ophthalmic solution USP, 0.3% contains: Active: tobramycin 0.3 % (3 mg). Preservative: benzalkonium chloride 0.01 % (0.1 mg). Inactives: boric acid, sodium sulfate decahydrate, sodium chloride, tyloxapol, sodium hydroxide and/or sulfuric acid (to adjust pH) and water for injection. Tobramycin ophthalmic solution USP, 0.3% has a pH range between 7.0 and 8.0 and an osmolality of 260-320 mOsm/kg. Tobramycin is a water-soluble aminoglycoside antibiotic active against a wide variety of gram-negative and gram-positive ophthalmic pathogens. The chemical structure of tobramycin is: Molecular Weight = 467.52 Molecular Formula: C 18 H 37 N 5 O 9 Chemical name: O-3-Amino-3-deoxy-α-D-glucopyranosyl-(1→4)-O-[2,6-diamino-2,3,6-trideoxy-α-D-ribo-hexopyranosyl-(1→6)]-2-deoxy-L-streptamine.
Medication Information
Warnings
FOR TOPICAL OPHTHALMIC USE. NOT FOR INJECTION INTO THE EYE. Sensitivity to topically applied aminoglycosides may occur in some patients. Severity of hypersensitivity reactions may vary from local effects to generalized reactions such as erythema, itching, urticaria, skin rash, anaphylaxis, anaphylactoid reactions, or bullous reactions. If a sensitivity reaction to tobramycin ophthalmic solution 0.3% occurs, discontinue use.
Indications and Usage
Tobramycin ophthalmic solution 0.3% is a topical antibiotic indicated in the treatment of external infections of the eye and its adnexa caused by susceptible bacteria. Appropriate monitoring of bacterial response to topical antibiotic therapy should accompany the use of tobramycin ophthalmic solution 0.3%. Clinical studies have shown tobramycin to be safe and effective for use in children.
Dosage and Administration
In mild to moderate disease, instill 1 or 2 drops into the affected eye(s) every 4 hours. In severe infections, instill 2 drops into the eye(s) hourly until improvement, following which treatment should be reduced prior to discontinuation.
Contraindications
Tobramycin ophthalmic solution 0.3% is contraindicated in patients with known hypersensitivity to any of its components.
Adverse Reactions
The most frequent adverse reactions to tobramycin ophthalmic solution 0.3% are hypersensitivity and localized ocular toxicity, including lid itching and swelling, and conjunctival erythema. These reactions occur in less than three of 100 patients treated with tobramycin ophthalmic solution.
Postmarketing Experience: Additional adverse reactions identified from post-marketing use include anaphylactic reaction, Stevens-Johnson syndrome, and erythema multiforme.
The following additional adverse reactions have been reported with systemic aminoglycosides: Neurotoxicity, ototoxicity and nephrotoxicity have occurred in patients receiving systemic aminoglycoside therapy. Aminoglycosides may aggravate muscle weakness in patients with known or suspected neuromuscular disorders, such as myasthenia gravis or Parkinson’s disease, because of their potential effect on neuromuscular function.
To report SUSPECTED ADVERSE REACTIONS, contact Jvet Pharmaceuticals, LLC at 1-727-292-1166 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
How Supplied
5 mL sterile solution supplied in opaque white low density polyethylene bottle which is closed with natural low density polyethylene nozzle and then with Tan colored high density polyethylene cap as follows:
5 mL containing tobramycin 0.3% (3 mg/mL)……….. NDC 86211-110-01
Storage: Store at 2°C to 25°C (36°F to 77°F).
After opening, Tobramycin Ophthalmic Solution, USP 0.3% can be used until the expiration date on the bottle.
Distributed by:
Jvet Pharmaceuticals LLC
6925112th Circle North, Suite 101
Largo, FL 33773
Revised: May 2025
L110I-VTN R-2505
747
1313001232-00
Description
Tobramycin ophthalmic solution USP, 0.3% is a sterile topical ophthalmic antibiotic formulation prepared specifically for topical therapy of external ophthalmic infections.
Each mL of tobramycin ophthalmic solution USP, 0.3% contains: Active: tobramycin 0.3 % (3 mg). Preservative: benzalkonium chloride 0.01 % (0.1 mg). Inactives: boric acid, sodium sulfate decahydrate, sodium chloride, tyloxapol, sodium hydroxide and/or sulfuric acid (to adjust pH) and water for injection. Tobramycin ophthalmic solution USP, 0.3% has a pH range between 7.0 and 8.0 and an osmolality of 260-320 mOsm/kg.
Tobramycin is a water-soluble aminoglycoside antibiotic active against a wide variety of gram-negative and gram-positive ophthalmic pathogens.
The chemical structure of tobramycin is:
Molecular Weight = 467.52
Molecular Formula: C18H37N5O9
Chemical name:
O-3-Amino-3-deoxy-α-D-glucopyranosyl-(1→4)-O-[2,6-diamino-2,3,6-trideoxy-α-D-ribo-hexopyranosyl-(1→6)]-2-deoxy-L-streptamine.
Precautions
General: As with other antibiotic preparations, prolonged use may result in overgrowth of nonsusceptible organisms, including fungi. If superinfection occurs, appropriate therapy should be initiated.
Cross-sensitivity to other aminoglycoside antibiotics may occur; if hypersensitivity develops with this product, discontinue use and institute appropriate therapy. Patients should be advised not to wear contact lenses if they have signs and symptoms of bacterial ocular infection.
Information for Patients: Do not touch dropper tip to any surface, as this may contaminate the solution.
Pregnancy: Reproduction studies in 3 types of animals at doses up to 33 times the normal human systemic dose have revealed no evidence of impaired fertility or harm to the fetus due to tobramycin. There are, however, no adequate and well-controlled studies in pregnant women. Because animal studies are not always predictive of human response, this drug should be used during pregnancy only if clearly needed.
Nursing Mothers: Because of the potential for adverse reactions in nursing infants from tobramycin ophthalmic solution, a decision should be made whether to discontinue nursing the infant or discontinue the drug, taking into account the importance of the drug to the mother.
Pediatric Use: Safety and effectiveness in pediatric patients below the age of 2 months has not been established.
Geriatric Use: No overall differences in safety or effectiveness have been observed between elderly and younger patients.
Clinical Pharmacology
In Vitro Data: In vitro studies have demonstrated tobramycin is active against susceptible strains of the following microorganisms: Staphylococci, including S. aureus and S. epidermidis (coagulase-positive and coagulase-negative), including penicillin-resistant strains.
Streptococci, including some of the Group A-beta-hemolytic species, some nonhemolytic species, and some Streptococcus pneumoniae.
Pseudomonas aeruginosa, Escherichia coli, Klebsiella pneumoniae, Enterobacter aerogenes, Proteus mirabilis, Morganella morganii, most Proteus vulgaris strains, Haemophilus influenzae and H. aegyptius, Moraxella lacunata, Acinetobacter calcoaceticus and some Neisseria species. Bacterial susceptibility studies demonstrate that in some cases, microorganisms resistant to gentamicin retain susceptibility to tobramycin.
Package Label.principal Display Panel
Bottle Label:
NDC 86211-110-01
Tobramycin Ophthalmic solution, USP 0.3%
Sterile 5 mL Rx Only
Carton Label:
NDC 86211-110-01
Tobramycin Ophthalmic solution, USP 0.3%
5 mL Sterile Rx Only
Structured Label Content
Warnings (WARNINGS)
FOR TOPICAL OPHTHALMIC USE. NOT FOR INJECTION INTO THE EYE. Sensitivity to topically applied aminoglycosides may occur in some patients. Severity of hypersensitivity reactions may vary from local effects to generalized reactions such as erythema, itching, urticaria, skin rash, anaphylaxis, anaphylactoid reactions, or bullous reactions. If a sensitivity reaction to tobramycin ophthalmic solution 0.3% occurs, discontinue use.
Indications and Usage (INDICATIONS AND USAGE)
Tobramycin ophthalmic solution 0.3% is a topical antibiotic indicated in the treatment of external infections of the eye and its adnexa caused by susceptible bacteria. Appropriate monitoring of bacterial response to topical antibiotic therapy should accompany the use of tobramycin ophthalmic solution 0.3%. Clinical studies have shown tobramycin to be safe and effective for use in children.
Dosage and Administration (DOSAGE AND ADMINISTRATION)
In mild to moderate disease, instill 1 or 2 drops into the affected eye(s) every 4 hours. In severe infections, instill 2 drops into the eye(s) hourly until improvement, following which treatment should be reduced prior to discontinuation.
Contraindications (CONTRAINDICATIONS)
Tobramycin ophthalmic solution 0.3% is contraindicated in patients with known hypersensitivity to any of its components.
Adverse Reactions (ADVERSE REACTIONS)
The most frequent adverse reactions to tobramycin ophthalmic solution 0.3% are hypersensitivity and localized ocular toxicity, including lid itching and swelling, and conjunctival erythema. These reactions occur in less than three of 100 patients treated with tobramycin ophthalmic solution.
Postmarketing Experience: Additional adverse reactions identified from post-marketing use include anaphylactic reaction, Stevens-Johnson syndrome, and erythema multiforme.
The following additional adverse reactions have been reported with systemic aminoglycosides: Neurotoxicity, ototoxicity and nephrotoxicity have occurred in patients receiving systemic aminoglycoside therapy. Aminoglycosides may aggravate muscle weakness in patients with known or suspected neuromuscular disorders, such as myasthenia gravis or Parkinson’s disease, because of their potential effect on neuromuscular function.
To report SUSPECTED ADVERSE REACTIONS, contact Jvet Pharmaceuticals, LLC at 1-727-292-1166 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
How Supplied (HOW SUPPLIED)
5 mL sterile solution supplied in opaque white low density polyethylene bottle which is closed with natural low density polyethylene nozzle and then with Tan colored high density polyethylene cap as follows:
5 mL containing tobramycin 0.3% (3 mg/mL)……….. NDC 86211-110-01
Storage: Store at 2°C to 25°C (36°F to 77°F).
After opening, Tobramycin Ophthalmic Solution, USP 0.3% can be used until the expiration date on the bottle.
Distributed by:
Jvet Pharmaceuticals LLC
6925112th Circle North, Suite 101
Largo, FL 33773
Revised: May 2025
L110I-VTN R-2505
747
1313001232-00
Description (DESCRIPTION)
Tobramycin ophthalmic solution USP, 0.3% is a sterile topical ophthalmic antibiotic formulation prepared specifically for topical therapy of external ophthalmic infections.
Each mL of tobramycin ophthalmic solution USP, 0.3% contains: Active: tobramycin 0.3 % (3 mg). Preservative: benzalkonium chloride 0.01 % (0.1 mg). Inactives: boric acid, sodium sulfate decahydrate, sodium chloride, tyloxapol, sodium hydroxide and/or sulfuric acid (to adjust pH) and water for injection. Tobramycin ophthalmic solution USP, 0.3% has a pH range between 7.0 and 8.0 and an osmolality of 260-320 mOsm/kg.
Tobramycin is a water-soluble aminoglycoside antibiotic active against a wide variety of gram-negative and gram-positive ophthalmic pathogens.
The chemical structure of tobramycin is:
Molecular Weight = 467.52
Molecular Formula: C18H37N5O9
Chemical name:
O-3-Amino-3-deoxy-α-D-glucopyranosyl-(1→4)-O-[2,6-diamino-2,3,6-trideoxy-α-D-ribo-hexopyranosyl-(1→6)]-2-deoxy-L-streptamine.
Precautions (PRECAUTIONS)
General: As with other antibiotic preparations, prolonged use may result in overgrowth of nonsusceptible organisms, including fungi. If superinfection occurs, appropriate therapy should be initiated.
Cross-sensitivity to other aminoglycoside antibiotics may occur; if hypersensitivity develops with this product, discontinue use and institute appropriate therapy. Patients should be advised not to wear contact lenses if they have signs and symptoms of bacterial ocular infection.
Information for Patients: Do not touch dropper tip to any surface, as this may contaminate the solution.
Pregnancy: Reproduction studies in 3 types of animals at doses up to 33 times the normal human systemic dose have revealed no evidence of impaired fertility or harm to the fetus due to tobramycin. There are, however, no adequate and well-controlled studies in pregnant women. Because animal studies are not always predictive of human response, this drug should be used during pregnancy only if clearly needed.
Nursing Mothers: Because of the potential for adverse reactions in nursing infants from tobramycin ophthalmic solution, a decision should be made whether to discontinue nursing the infant or discontinue the drug, taking into account the importance of the drug to the mother.
Pediatric Use: Safety and effectiveness in pediatric patients below the age of 2 months has not been established.
Geriatric Use: No overall differences in safety or effectiveness have been observed between elderly and younger patients.
Clinical Pharmacology (CLINICAL PHARMACOLOGY)
In Vitro Data: In vitro studies have demonstrated tobramycin is active against susceptible strains of the following microorganisms: Staphylococci, including S. aureus and S. epidermidis (coagulase-positive and coagulase-negative), including penicillin-resistant strains.
Streptococci, including some of the Group A-beta-hemolytic species, some nonhemolytic species, and some Streptococcus pneumoniae.
Pseudomonas aeruginosa, Escherichia coli, Klebsiella pneumoniae, Enterobacter aerogenes, Proteus mirabilis, Morganella morganii, most Proteus vulgaris strains, Haemophilus influenzae and H. aegyptius, Moraxella lacunata, Acinetobacter calcoaceticus and some Neisseria species. Bacterial susceptibility studies demonstrate that in some cases, microorganisms resistant to gentamicin retain susceptibility to tobramycin.
Package Label.principal Display Panel (PACKAGE LABEL.PRINCIPAL DISPLAY PANEL)
Bottle Label:
NDC 86211-110-01
Tobramycin Ophthalmic solution, USP 0.3%
Sterile 5 mL Rx Only
Carton Label:
NDC 86211-110-01
Tobramycin Ophthalmic solution, USP 0.3%
5 mL Sterile Rx Only
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:52:47.581068 · Updated: 2026-03-14T22:44:38.896877