These Highlights Do Not Include All The Information Needed To Use Pavblu Safely And Effectively. See Full Prescribing Information For Pavblu.
c61062f3-80ec-4a20-8c28-46e468592a06
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
PAVBLU is indicated for the treatment of:
Indications and Usage
PAVBLU is indicated for the treatment of:
Dosage and Administration
Neovascular (Wet) Age-Related Macular Degeneration (AMD): The recommended dose for PAVBLU is 2 mg (0.05 mL of 40 mg/mL solution) administered by intravitreal injection every 4 weeks (approximately every 28 days, monthly) for the first 3 months, followed by 2 mg (0.05 mL of 40 mg/mL solution) via intravitreal injection once every 8 weeks (2 months). ( 2.2 ) Although PAVBLU may be dosed as frequently as 2 mg every 4 weeks (approximately every 25 days, monthly), additional efficacy was not demonstrated in most patients when aflibercept was dosed every 4 weeks compared to every 8 weeks. Some patients may need every 4 week (monthly) dosing after the first 12 weeks (3 months). ( 2.2 ) Although not as effective as the recommended every 8 week dosing regimen, patients may also be treated with one dose every 12 weeks after one year of effective therapy. Patients should be assessed regularly. ( 2.2 ) Macular Edema Following Retinal Vein Occlusion (RVO): The recommended dose for PAVBLU is 2 mg (0.05 mL of 40 mg/mL solution) administered by intravitreal injection once every 4 weeks (approximately every 25 days, monthly). ( 2.3 ) Diabetic Macular Edema (DME) and Diabetic Retinopathy (DR): The recommended dose for PAVBLU is 2 mg (0.05 mL of 40 mg/mL solution) administered by intravitreal injection every 4 weeks (approximately every 28 days, monthly) for the first 5 injections followed by 2 mg (0.05 mL of 40 mg/mL solution) via intravitreal injection once every 8 weeks (2 months). ( 2.4 , 2.5 ) Although PAVBLU may be dosed as frequently as 2 mg every 4 weeks (approximately every 25 days, monthly), additional efficacy was not demonstrated in most patients when aflibercept was dosed every 4 weeks compared to every 8 weeks. Some patients may need every 4 week (monthly) dosing after the first 20 weeks (5 months). ( 2.4 , 2.5 )
Warnings and Precautions
Endophthalmitis, retinal detachments, and retinal vasculitis with or without occlusion may occur following intravitreal injections. Patients and/or caregivers should be instructed to report any signs and/or symptoms suggestive of endophthalmitis, retinal detachment, or retinal vasculitis without delay and should be managed appropriately. ( 5.1 ) Increases in intraocular pressure have been seen within 60 minutes of an intravitreal injection. ( 5.2 ) There is a potential risk of arterial thromboembolic events following intravitreal use of VEGF inhibitors. ( 5.4 )
Contraindications
Ocular or periocular infections ( 4.1 ) Active intraocular inflammation ( 4.2 ) Hypersensitivity ( 4.3 )
Adverse Reactions
The following potentially serious adverse reactions are described elsewhere in the labeling: Hypersensitivity [see Contraindications (4.3) ] Endophthalmitis, retinal detachments, and Retinal Vasculitis with or without Occlusion [see Warnings and Precautions (5.1) ] Increase in intraocular pressure [see Warnings and Precautions (5.2) ] Thromboembolic events [see Warnings and Precautions (5.4) ]
Storage and Handling
Refrigerate PAVBLU at 2°C to 8ºC (36°F to 46ºF). PAVBLU may be kept at room temperature (up to 30° C (86° F)) for a single-period of 3 days. Do not freeze. Do not use beyond the date stamped on the carton and container label. Store in the original carton until time of use to protect from light. Do not open sealed blister tray until time of use.
How Supplied
Each prefilled syringe or vial is for single eye use only. Discard unused portion. PAVBLU injection is a clear to opalescent and colorless to slightly yellow solution supplied in the following presentations: NDC NUMBER CARTON TYPE CARTON CONTENTS 55513-056-01 Prefilled Syringe one blister pack containing one PAVBLU 2 mg (0.05 mL of a 40 mg/mL solution) sterile, single-dose prefilled plastic syringe one Prescribing Information 55513-065-01 Vial Only one PAVBLU 2 mg (0.05 mL of a 40 mg/mL solution) single-dose glass vial one Prescribing Information
Medication Information
Warnings and Precautions
Endophthalmitis, retinal detachments, and retinal vasculitis with or without occlusion may occur following intravitreal injections. Patients and/or caregivers should be instructed to report any signs and/or symptoms suggestive of endophthalmitis, retinal detachment, or retinal vasculitis without delay and should be managed appropriately. ( 5.1 ) Increases in intraocular pressure have been seen within 60 minutes of an intravitreal injection. ( 5.2 ) There is a potential risk of arterial thromboembolic events following intravitreal use of VEGF inhibitors. ( 5.4 )
Indications and Usage
PAVBLU is indicated for the treatment of:
Dosage and Administration
Neovascular (Wet) Age-Related Macular Degeneration (AMD): The recommended dose for PAVBLU is 2 mg (0.05 mL of 40 mg/mL solution) administered by intravitreal injection every 4 weeks (approximately every 28 days, monthly) for the first 3 months, followed by 2 mg (0.05 mL of 40 mg/mL solution) via intravitreal injection once every 8 weeks (2 months). ( 2.2 ) Although PAVBLU may be dosed as frequently as 2 mg every 4 weeks (approximately every 25 days, monthly), additional efficacy was not demonstrated in most patients when aflibercept was dosed every 4 weeks compared to every 8 weeks. Some patients may need every 4 week (monthly) dosing after the first 12 weeks (3 months). ( 2.2 ) Although not as effective as the recommended every 8 week dosing regimen, patients may also be treated with one dose every 12 weeks after one year of effective therapy. Patients should be assessed regularly. ( 2.2 ) Macular Edema Following Retinal Vein Occlusion (RVO): The recommended dose for PAVBLU is 2 mg (0.05 mL of 40 mg/mL solution) administered by intravitreal injection once every 4 weeks (approximately every 25 days, monthly). ( 2.3 ) Diabetic Macular Edema (DME) and Diabetic Retinopathy (DR): The recommended dose for PAVBLU is 2 mg (0.05 mL of 40 mg/mL solution) administered by intravitreal injection every 4 weeks (approximately every 28 days, monthly) for the first 5 injections followed by 2 mg (0.05 mL of 40 mg/mL solution) via intravitreal injection once every 8 weeks (2 months). ( 2.4 , 2.5 ) Although PAVBLU may be dosed as frequently as 2 mg every 4 weeks (approximately every 25 days, monthly), additional efficacy was not demonstrated in most patients when aflibercept was dosed every 4 weeks compared to every 8 weeks. Some patients may need every 4 week (monthly) dosing after the first 20 weeks (5 months). ( 2.4 , 2.5 )
Contraindications
Ocular or periocular infections ( 4.1 ) Active intraocular inflammation ( 4.2 ) Hypersensitivity ( 4.3 )
Adverse Reactions
The following potentially serious adverse reactions are described elsewhere in the labeling: Hypersensitivity [see Contraindications (4.3) ] Endophthalmitis, retinal detachments, and Retinal Vasculitis with or without Occlusion [see Warnings and Precautions (5.1) ] Increase in intraocular pressure [see Warnings and Precautions (5.2) ] Thromboembolic events [see Warnings and Precautions (5.4) ]
Storage and Handling
Refrigerate PAVBLU at 2°C to 8ºC (36°F to 46ºF). PAVBLU may be kept at room temperature (up to 30° C (86° F)) for a single-period of 3 days. Do not freeze. Do not use beyond the date stamped on the carton and container label. Store in the original carton until time of use to protect from light. Do not open sealed blister tray until time of use.
How Supplied
Each prefilled syringe or vial is for single eye use only. Discard unused portion. PAVBLU injection is a clear to opalescent and colorless to slightly yellow solution supplied in the following presentations: NDC NUMBER CARTON TYPE CARTON CONTENTS 55513-056-01 Prefilled Syringe one blister pack containing one PAVBLU 2 mg (0.05 mL of a 40 mg/mL solution) sterile, single-dose prefilled plastic syringe one Prescribing Information 55513-065-01 Vial Only one PAVBLU 2 mg (0.05 mL of a 40 mg/mL solution) single-dose glass vial one Prescribing Information
Description
PAVBLU is indicated for the treatment of:
Section 42229-5
Prefilled syringe: Inject by pressing the plunger carefully and with constant pressure. Do not apply additional pressure once the plunger has reached the bottom of the syringe. A small residual volume may remain in the syringe after a full dose has been injected. This is normal. Do not administer any residual solution observed in the syringe.
Immediately following the intravitreal injection, patients should be monitored for elevation in intraocular pressure. Appropriate monitoring may consist of a check for perfusion of the optic nerve head or tonometry. If required, a sterile paracentesis needle should be available.
Following intravitreal injection, patients and/or caregivers should be instructed to report any signs and/or symptoms suggestive of endophthalmitis or retinal detachment (e.g., eye pain, redness of the eye, photophobia, blurring of vision) without delay [see Patient Counseling Information (17)].
Each sterile, prefilled syringe or vial should only be used for the treatment of a single eye. If the contralateral eye requires treatment, a new sterile, prefilled syringe or vial should be used and the sterile field, syringe, gloves, drapes, eyelid speculum, filter, and injection needles should be changed before PAVBLU is administered to the other eye.
After injection, any unused product must be discarded.
10 Overdosage
Overdosing with increased injection volume may increase intraocular pressure. Therefore, in case of overdosage, intraocular pressure should be monitored and if deemed necessary by the treating physician, adequate treatment should be initiated.
11 Description
Aflibercept-ayyh is a recombinant fusion protein consisting of portions of human VEGF receptors 1 and 2 extracellular domains fused to the Fc portion of human IgG1 formulated as an iso-osmotic solution for intravitreal administration. Aflibercept-ayyh is a dimeric glycoprotein with a protein molecular weight of 97 kilodaltons (kDa) and contains glycosylation, constituting an additional 15% of the total molecular mass, resulting in a total molecular weight of 115 kDa. Aflibercept-ayyh is produced in recombinant Chinese hamster ovary (CHO) cells.
PAVBLU (aflibercept-ayyh) injection is a sterile, clear to opalescent and colorless to slightly yellow solution. PAVBLU does not contain anti-microbial preservative and is supplied as an aqueous solution for intravitreal injection in a single-dose prefilled plastic syringe or a single-dose glass vial designed to deliver 0.05 mL (50 microliters) of solution containing 2 mg of aflibercept-ayyh in polysorbate 80 (0.005 mg), sucrose (2.5 mg), trehalose (1.58 mg) and water for injection with a pH of 6.2.
16.1 How Supplied
Each prefilled syringe or vial is for single eye use only. Discard unused portion. PAVBLU injection is a clear to opalescent and colorless to slightly yellow solution supplied in the following presentations:
| NDC NUMBER | CARTON TYPE | CARTON CONTENTS |
|---|---|---|
| 55513-056-01 | Prefilled Syringe | one blister pack containing one PAVBLU 2 mg (0.05 mL of a 40 mg/mL solution) sterile, single-dose prefilled plastic syringe one Prescribing Information |
| 55513-065-01 | Vial Only | one PAVBLU 2 mg (0.05 mL of a 40 mg/mL solution) single-dose glass vial one Prescribing Information |
8.4 Pediatric Use
A pediatric assessment for PAVBLU demonstrates that PAVBLU is safe and effective for pediatric patients in an indication for which EYLEA (aflibercept) is approved. However, PAVBLU is not approved for such indication due to marketing exclusivity for EYLEA (aflibercept).
8.5 Geriatric Use
In the clinical studies, approximately 76% (2049/2701) of patients randomized to treatment with aflibercept were ≥65 years of age and approximately 46% (1250/2701) were ≥75 years of age. No significant differences in efficacy or safety were seen with increasing age in these studies.
12.6 Immunogenicity
The observed incidence of anti-drug antibodies is highly dependent on the sensitivity and specificity of the assay. Differences in assay methods preclude meaningful comparisons of the incidence of anti-drug antibodies in the studies described below with the incidence of anti-drug antibodies in other studies, including those of aflibercept or of other aflibercept products.
In the wet AMD, RVO, and DME studies, the pre-treatment incidence of immunoreactivity to aflibercept was approximately 1% to 3% across treatment groups. After dosing with aflibercept for 24-100 weeks, antibodies to aflibercept were detected in a similar percentage range of patients.
There were no differences in efficacy or safety between patients with or without immunoreactivity.
4 Contraindications
4.3 Hypersensitivity
PAVBLU is contraindicated in patients with known hypersensitivity to aflibercept or any of the excipients in PAVBLU. Hypersensitivity reactions may manifest as rash, pruritus, urticaria, severe anaphylactic/anaphylactoid reactions, or severe intraocular inflammation.
6. Adverse Reactions
The following potentially serious adverse reactions are described elsewhere in the labeling:
- Hypersensitivity [see Contraindications (4.3)]
- Endophthalmitis, retinal detachments, and Retinal Vasculitis with or without Occlusion [see Warnings and Precautions (5.1)]
- Increase in intraocular pressure [see Warnings and Precautions (5.2)]
- Thromboembolic events [see Warnings and Precautions (5.4)]
12.3 Pharmacokinetics
Aflibercept is administered intravitreally to exert local effects in the eye. In patients with wet AMD, RVO, or DME, following intravitreal administration of aflibercept, a fraction of the administered dose is expected to bind with endogenous VEGF in the eye to form an inactive aflibercept: VEGF complex. Once absorbed into the systemic circulation, aflibercept presents in the plasma as free aflibercept (unbound to VEGF) and a more predominant stable inactive form with circulating endogenous VEGF (i.e., aflibercept: VEGF complex).
1 Indications and Usage
PAVBLU is indicated for the treatment of:
2.8 Injection Procedure
The intravitreal injection procedure should be carried out under controlled aseptic conditions, which include surgical hand disinfection and the use of sterile gloves, a sterile drape, and a sterile eyelid speculum (or equivalent). Adequate anesthesia and a topical broad−spectrum microbicide should be given prior to the injection.
12.1 Mechanism of Action
Vascular endothelial growth factor-A (VEGF-A) and placental growth factor (PlGF) are members of the VEGF family of angiogenic factors that can act as mitogenic, chemotactic, and vascular permeability factors for endothelial cells. VEGF acts via two receptor tyrosine kinases, VEGFR-1 and VEGFR-2, present on the surface of endothelial cells. PlGF binds only to VEGFR-1, which is also present on the surface of leucocytes. Activation of these receptors by VEGF-A can result in neovascularization and vascular permeability.
Aflibercept products act as a soluble decoy receptor that binds VEGF-A and PlGF, and thereby can inhibit the binding and activation of these cognate VEGF receptors.
16.2 Storage and Handling
Refrigerate PAVBLU at 2°C to 8ºC (36°F to 46ºF). PAVBLU may be kept at room temperature (up to 30° C (86° F)) for a single-period of 3 days. Do not freeze. Do not use beyond the date stamped on the carton and container label. Store in the original carton until time of use to protect from light. Do not open sealed blister tray until time of use.
5.4 Thromboembolic Events
There is a potential risk of arterial thromboembolic events (ATEs) following intravitreal use of VEGF inhibitors, including aflibercept products. ATEs are defined as nonfatal stroke, nonfatal myocardial infarction, or vascular death (including deaths of unknown cause). The incidence of reported thromboembolic events in wet AMD studies during the first year was 1.8% (32 out of 1824) in the combined group of patients treated with aflibercept compared with 1.5% (9 out of 595) in patients treated with ranibizumab; through 96 weeks, the incidence was 3.3% (60 out of 1824) in the aflibercept group compared with 3.2% (19 out of 595) in the ranibizumab group. The incidence in the DME studies from baseline to week 52 was 3.3% (19 out of 578) in the combined group of patients treated with aflibercept compared with 2.8% (8 out of 287) in the control group; from baseline to week 100, the incidence was 6.4% (37 out of 578) in the combined group of patients treated with aflibercept compared with 4.2% (12 out of 287) in the control group. There were no reported thromboembolic events in the patients treated with aflibercept in the first six months of the RVO studies.
5 Warnings and Precautions
- Endophthalmitis, retinal detachments, and retinal vasculitis with or without occlusion may occur following intravitreal injections. Patients and/or caregivers should be instructed to report any signs and/or symptoms suggestive of endophthalmitis, retinal detachment, or retinal vasculitis without delay and should be managed appropriately. (5.1)
- Increases in intraocular pressure have been seen within 60 minutes of an intravitreal injection. (5.2)
- There is a potential risk of arterial thromboembolic events following intravitreal use of VEGF inhibitors. (5.4)
2 Dosage and Administration
Neovascular (Wet) Age-Related Macular Degeneration (AMD):
- The recommended dose for PAVBLU is 2 mg (0.05 mL of 40 mg/mL solution) administered by intravitreal injection every 4 weeks (approximately every 28 days, monthly) for the first 3 months, followed by 2 mg (0.05 mL of 40 mg/mL solution) via intravitreal injection once every 8 weeks (2 months). (2.2)
- Although PAVBLU may be dosed as frequently as 2 mg every 4 weeks (approximately every 25 days, monthly), additional efficacy was not demonstrated in most patients when aflibercept was dosed every 4 weeks compared to every 8 weeks. Some patients may need every 4 week (monthly) dosing after the first 12 weeks (3 months). (2.2)
- Although not as effective as the recommended every 8 week dosing regimen, patients may also be treated with one dose every 12 weeks after one year of effective therapy. Patients should be assessed regularly. (2.2)
Macular Edema Following Retinal Vein Occlusion (RVO):
- The recommended dose for PAVBLU is 2 mg (0.05 mL of 40 mg/mL solution) administered by intravitreal injection once every 4 weeks (approximately every 25 days, monthly). (2.3)
Diabetic Macular Edema (DME) and Diabetic Retinopathy (DR):
- The recommended dose for PAVBLU is 2 mg (0.05 mL of 40 mg/mL solution) administered by intravitreal injection every 4 weeks (approximately every 28 days, monthly) for the first 5 injections followed by 2 mg (0.05 mL of 40 mg/mL solution) via intravitreal injection once every 8 weeks (2 months). (2.4, 2.5)
- Although PAVBLU may be dosed as frequently as 2 mg every 4 weeks (approximately every 25 days, monthly), additional efficacy was not demonstrated in most patients when aflibercept was dosed every 4 weeks compared to every 8 weeks. Some patients may need every 4 week (monthly) dosing after the first 20 weeks (5 months). (2.4, 2.5)
3 Dosage Forms and Strengths
PAVBLU is a clear to opalescent and colorless to slightly yellow solution available as:
- Injection: 2 mg (0.05 mL of 40 mg/mL solution) in a single-dose prefilled plastic syringe
- Injection: 2 mg (0.05 mL of 40 mg/mL solution) in a single-dose glass vial
6.2 Postmarketing Experience
The following adverse reactions have been identified during postapproval use of aflibercept. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Eye disorders: retinal vasculitis and occlusive retinal vasculitis related to intravitreal injection with aflibercept (reported at a rate of 0.6 and 0.2 per 1 million injections, respectively, based on postmarketing experience from November 2011 until November 2023).
1.4 Diabetic Retinopathy (dr)
2.5 Diabetic Retinopathy (dr)
The recommended dose for PAVBLU is 2 mg (0.05 mL of 40 mg/mL solution) administered by intravitreal injection every 4 weeks (approximately every 28 days, monthly) for the first 5 injections, followed by 2 mg (0.05 mL of 40 mg/mL solution) via intravitreal injection once every 8 weeks (2 months). Although PAVBLU may be dosed as frequently as 2 mg every 4 weeks (approximately every 25 days, monthly), additional efficacy was not demonstrated in most patients when aflibercept was dosed every 4 weeks compared to every 8 weeks [see Clinical Studies (14.5)]. Some patients may need every 4 week (monthly) dosing after the first 20 weeks (5 months).
14.5 Diabetic Retinopathy (dr)
Efficacy and safety data of aflibercept in diabetic retinopathy (DR) are derived from the VIVID, VISTA, and PANORAMA studies.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in other clinical trials of the same or another drug and may not reflect the rates observed in practice.
A total of 2980 adult patients treated with aflibercept constituted the safety population in eight phase 3 studies. Among those, 2379 patients were treated with the recommended dose of 2 mg. Serious adverse reactions related to the injection procedure have occurred in <0.1% of intravitreal injections with aflibercept including endophthalmitis and retinal detachment. The most common adverse reactions (≥5%) reported in patients receiving aflibercept were conjunctival hemorrhage, eye pain, cataract, vitreous detachment, vitreous floaters, and intraocular pressure increased.
1.3 Diabetic Macular Edema (dme)
2.4 Diabetic Macular Edema (dme)
The recommended dose for PAVBLU is 2 mg (0.05 mL of 40 mg/mL solution) administered by intravitreal injection every 4 weeks (approximately every 28 days, monthly) for the first 5 injections, followed by 2 mg (0.05 mL of 40 mg/mL solution) via intravitreal injection once every 8 weeks (2 months). Although PAVBLU may be dosed as frequently as 2 mg every 4 weeks (approximately every 25 days, monthly), additional efficacy was not demonstrated in most patients when aflibercept was dosed every 4 weeks compared to every 8 weeks [see Clinical Studies (14.4)]. Some patients may need every 4 week (monthly) dosing after the first 20 weeks (5 months).
14.4 Diabetic Macular Edema (dme)
The safety and efficacy of aflibercept were assessed in two randomized, multi-center, double-masked, controlled studies in patients with DME. A total of 862 randomized and treated patients were evaluable for efficacy. Protocol-specified visits occurred every 28±7 days. Patient ages ranged from 23 to 87 years with a mean of 63 years.
Of those, 576 were randomized to aflibercept groups in the two studies (VIVID and VISTA). In each study, patients were randomly assigned in a 1:1:1 ratio to 1 of 3 dosing regimens: 1) aflibercept administered 2 mg every 8 weeks following 5 initial monthly injections (aflibercept 2Q8); 2) aflibercept administered 2 mg every 4 weeks (aflibercept 2Q4); and 3) macular laser photocoagulation (at baseline and then as needed). Beginning at week 24, patients meeting a pre-specified threshold of vision loss were eligible to receive additional treatment: patients in the aflibercept groups could receive laser and patients in the laser group could receive aflibercept.
In both studies, the primary efficacy endpoint was the mean change from baseline in BCVA at week 52 as measured by ETDRS letter score. Efficacy of both aflibercept 2Q8 and aflibercept 2Q4 groups was statistically superior to the control group. This statistically superior improvement in BCVA was maintained at week 100 in both studies.
Results from the analysis of the VIVID and VISTA studies are shown in Table 7 and Figure 16 below.
| VIVID | VISTA | |||||
|---|---|---|---|---|---|---|
| Aflibercept 2 mg Q8 weeks After treatment initiation with 5 monthly injections
|
Aflibercept 2 mg Q4 weeks |
Control | Aflibercept 2 mg Q8 weeks |
Aflibercept 2 mg Q4 weeks |
Control | |
| Full Analysis Set | N=135 | N=136 | N=132 | N=151 | N=154 | N=154 |
| Efficacy Outcomes at Week 52 | ||||||
| Mean change in BCVA as measured by ETDRS letter score from Baseline (SD) | 10.7 | 10.5 | 1.2 | 10.7 | 12.5 | 0.2 |
| (9.3) | (9.6) | (10.6) | (8.2) | (9.5) | (12.5) | |
| Difference LS mean and CI based on an ANCOVA model with baseline BCVA measurement as a covariate and a factor for treatment group. Additionally, protocol specified stratification factors were included in the model.
,
Difference is aflibercept group minus Control group in LS mean |
9.1 p<0.01 compared with Control
|
9.3 | 10.5 | 12.2 | ||
| (97.5% CI) | (6.3, 11.8) | (6.5, 12.0) | (7.7, 13.2) | (9.4, 15.0) | ||
| Proportion of patients who gained at least 15 letters in BCVA from Baseline (%) | 33.3% | 32.4% | 9.1% | 31.1% | 41.6% | 7.8% |
| Adjusted Difference
,
Difference with confidence interval (CI) and statistical test is calculated using Mantel-Haenszel weighting scheme adjusted by protocol specified stratification factors
(%) |
24.2% | 23.3% | 23.3% | 34.2% | ||
| (97.5% CI) | (13.5, 34.9) | (12.6, 33.9) | (13.5, 33.1) | (24.1, 44.4) | ||
| Efficacy Outcomes at Week 100 | ||||||
| Mean change in BCVA as measured by ETDRS letter score from Baseline (SD) | 9.4 | 11.4 | 0.7 | 11.1 | 11.5 | 0.9 |
| (10.5) | (11.2) | (11.8) | (10.7) | (13.8) | (13.9) | |
| Difference
,
in LS mean |
8.2 | 10.7 | 10.1 | 10.6 | ||
| (97.5% CI) | (5.2, 11.3) | (7.6, 13.8) | (7.0, 13.3) | (7.1, 14.2) | ||
| Proportion of patients who gained at least 15 letters in BCVA from Baseline (%) | 31.1% | 38.2% | 12.1% | 33.1% | 38.3% | 13.0% |
| Adjusted Difference
,
(%) |
19.0% | 26.1% | 20.1% | 25.8% | ||
| (97.5% CI) | (8.0, 29.9) | (14.8, 37.5) | (9.6, 30.6) | (15.1, 36.6) |
|
Figure 16. Mean Change in BCVA as Measured by ETDRS Letter Score from Baseline to Week 100 in VIVID and VISTA Studies
|
Treatment effects in the subgroup of patients who had previously been treated with a VEGF inhibitor prior to study participation were similar to those seen in patients who were VEGF inhibitor naïve prior to study participation.
Treatment effects in evaluable subgroups (e.g., age, gender, race, baseline HbA1c, baseline visual acuity, prior anti-VEGF therapy) in each study were in general consistent with the results in the overall populations.
17 Patient Counseling Information
In the days following PAVBLU administration, patients are at risk of developing endophthalmitis, retinal detachment, or retinal vasculitis with or without occlusion. If the eye becomes red, sensitive to light, painful, or develops a change in vision, advise patients and/or caregivers to seek immediate care from an ophthalmologist [see Warnings and Precautions (5.1)].
Patients may experience temporary visual disturbances after an intravitreal injection with PAVBLU and the associated eye examinations [see Adverse Reactions (6)]. Advise patients not to drive or use machinery until visual function has recovered sufficiently.
4.1 Ocular Or Periocular Infections
PAVBLU is contraindicated in patients with ocular or periocular infections.
4.2 Active Intraocular Inflammation
PAVBLU is contraindicated in patients with active intraocular inflammation.
2.1 Important Injection Instructions
For ophthalmic intravitreal injection. PAVBLU must only be administered by a qualified physician.
Prefilled Syringe: A 30-gauge × ½-inch sterile injection needle is needed but not provided.
Vial: A 5-micron sterile filter needle (18-gauge × 1½-inch), a 1-mL Luer lock syringe and a 30-gauge × ½-inch sterile injection needle are needed but not provided.
PAVBLU is available packaged as follows:
- Prefilled Syringe
- Vial Only
5.2 Increase in Intraocular Pressure
Acute increases in intraocular pressure have been seen within 60 minutes of intravitreal injection, including with aflibercept products [see Adverse Reactions (6.1)]. Sustained increases in intraocular pressure have also been reported after repeated intravitreal dosing with vascular endothelial growth factor (VEGF) inhibitors. Intraocular pressure and the perfusion of the optic nerve head should be monitored and managed appropriately [see Dosage and Administration (2.8)].
2.7 Preparation for Administration Vial
PAVBLU should be inspected visually prior to administration. It is a clear to opalescent and colorless to slightly yellow solution. If particulates, cloudiness, or discoloration are visible, the vial must not be used.
The glass vial is for one-time use in one eye only.
Use aseptic technique to carry out the following preparation steps:
Prepare for intravitreal injection with the following medical devices for single use:
- a 5-micron sterile filter needle 18-gauge × 1½-inch (not provided)
- a 1-mL sterile Luer lock syringe with marking to measure 0.05 mL (not provided)
- a sterile injection needle 30-gauge × ½-inch (not provided)
-
1.Remove the protective plastic cap from the vial (see Figure 6).
Figure 6:
-
2.Clean the top of the vial with an alcohol wipe (see Figure 7).
Figure 7:
-
3.Remove the 18-gauge × 1½-inch, 5-micron, filter needle and the 1-mL syringe from their packaging. Attach the filter needle to the syringe by twisting it onto the Luer lock syringe tip (see Figure 8a and Figure 8b).
| Figure 8a: | Figure 8b: |
|
|
|
Note: When ready to withdraw PAVBLU, remove the plastic needle shield from the needle.
-
4.Push the filter needle into the center of the vial stopper until the needle is completely inserted into the vial and the tip touches the bottom or bottom edge of the vial.
-
5.Using aseptic technique withdraw all of the PAVBLU vial contents into the syringe, keeping the vial in an upright position, slightly inclined to ease complete withdrawal. To deter the introduction of air, ensure the bevel of the filter needle is submerged into the liquid. Continue to tilt the vial during withdrawal keeping the bevel of the filter needle submerged in the liquid (see Figure 9a and Figure 9b).
|
Figure 9a: Figure 9b:
|
-
6.Ensure that the plunger rod is drawn sufficiently back when emptying the vial in order to completely empty the filter needle.
-
7.Remove the filter needle from the syringe and properly dispose of the filter needle. Note: Filter needle is not to be used for intravitreal injection.
-
8.Remove the 30-gauge × ½-inch injection needle from its packaging and attach the injection needle to the syringe by firmly twisting the injection needle onto the Luer lock syringe tip (see Figure 10a and Figure 10b).
|
Figure 10a:
|
Figure 10b:
|
Note: When ready to administer PAVBLU, remove the plastic needle shield from the needle.
-
9.Holding the syringe with the needle pointing up, check the syringe for bubbles. If there are bubbles, gently tap the syringe with your finger until the bubbles rise to the top (see Figure 11).
Figure 11:
-
10.To eliminate all of the bubbles and to expel excess drug, SLOWLY depress the plunger rod so that the plunger edge aligns with the line that marks 0.05 mL on the syringe (see Figure 12a and Figure 12b).
13.2 Animal Toxicology And/or Pharmacology
Erosions and ulcerations of the respiratory epithelium in nasal turbinates in monkeys treated with aflibercept intravitreally were observed at intravitreal doses of 2 or 4 mg per eye. At the NOAEL of 0.5 mg per eye in monkeys, the systemic exposure (AUC) was 56 times higher than the exposure observed in adult patients after an intravitreal dose of 2 mg. Similar effects were not seen in other clinical studies [see Clinical Studies (14)].
Principal Display Panel 0.05 Ml Vial Carton
2 mg /
0.05 mL
NDC 55513-065-01
PAVBLU™
(aflibercept-ayyh)
Injection
2 mg (0.05 mL of
40 mg/mL solution)
For Intravitreal Injection
One Single-Dose Vial
Sterile Solution
Contains no anti-microbial
preservatives.
Keep out of reach of children
Discard unused portion
AMGEN®
Rx Only
Principal Display Panel 0.05 Ml Syringe Carton
2 mg /
0.05 mL
AMGEN®
PAVBLU™
(aflibercept-ayyh)
Injection
NDC 55513-056-01
2 mg (0.05 mL of 40 mg/mL solution)
Carton contents: one blister pack containing one sterile, single-dose prefilled plastic
syringe and package insert.
Prefilled syringe contents:
Each PAVBLU™ single-dose prefilled syringe is designed to deliver 2 mg of aflibercept-ayyh
in 0.05 mL (50 microliters) solution with polysorbate 80 (0.005 mg), sucrose (2.5 mg),
trehalose (1.58 mg) and water for injection with a pH of 6.2.
Recommended Dosage: See prescribing information.
Rx Only
2.6 Preparation for Administration Prefilled Syringe
The PAVBLU prefilled plastic syringe is sterile and for one-time use in one eye only.
The prefilled syringe should be inspected visually prior to administration. It is a clear to opalescent and colorless to slightly yellow solution. Do not use if particulates, cloudiness, or discoloration are visible, or if the package is open or damaged. The appearance of the syringe cap on the prefilled syringe may vary (for example, color and design).
Do not use if any part of the prefilled syringe is damaged or if the syringe cap is detached from the Luer lock.
The intravitreal injection should be performed with a 30-gauge × ½-inch injection needle (not provided).
The prefilled syringe contains more than the recommended dose of 2 mg aflibercept-ayyh (equivalent to 50 microliters). The excess volume must be discarded prior to the administration.
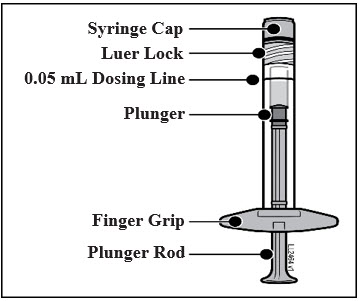
PREFILLED SYRINGE DESCRIPTION - Figure 1:
Use aseptic technique to carry out the following steps:
-
1.PREPARE
When ready to administer PAVBLU, open the carton and remove sterilized blister pack. Carefully peel open the sterilized blister pack ensuring the sterility of its contents. Keep the syringe in the sterile tray until you are ready for assembly.
-
2.REMOVE SYRINGE
Using aseptic technique, remove the syringe from the sterilized blister pack.
-
3.TWIST OFF SYRINGE CAP
Twist off (do not snap off) the syringe cap by holding the syringe in one hand and the syringe cap with the thumb and forefinger of the other hand (see Figure 2).
Figure 2:
-
4.ATTACH NEEDLE
Using aseptic technique, firmly twist a 30-gauge × ½-inch injection needle onto the Luer lock syringe tip (see Figure 3).
Figure 3:
Note: When ready to administer PAVBLU, remove the plastic needle shield from the needle.
-
5.DISLODGE AIR BUBBLES
Holding the syringe with the needle pointing up, check the syringe for bubbles. If there are bubbles, gently tap the syringe with your finger until the bubbles rise to the top (see Figure 4).
Figure 4:
-
6.EXPEL AIR AND SET THE DOSE
To eliminate all bubbles and to expel excess drug, slowly depress the plunger rod to align the plunger dome edge (see Figure 5a) with the black dosing line on the syringe (equivalent to 50 microliters) (see Figure 5b).
|
Figure 5a:
|
Figure 5b:
|
-
7.The prefilled syringe is for one-time use in one eye only. After injection any unused product must be discarded.
1.2 Macular Edema Following Retinal Vein Occlusion (rvo)
2.3 Macular Edema Following Retinal Vein Occlusion (rvo)
The recommended dose for PAVBLU is 2 mg (0.05 mL of 40 mg/mL solution) administered by intravitreal injection once every 4 weeks (approximately every 25 days, monthly) [see Clinical Studies (14.2), (14.3)].
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
No studies have been conducted on the mutagenic or carcinogenic potential of aflibercept products. Effects on male and female fertility were assessed as part of a 6-month study in monkeys with intravenous administration of aflibercept at weekly doses ranging from 3 to 30 mg per kg. Absent or irregular menses associated with alterations in female reproductive hormone levels and changes in sperm morphology and motility were observed at all dose levels. In addition, females showed decreased ovarian and uterine weight accompanied by compromised luteal development and reduction of maturing follicles. These changes correlated with uterine and vaginal atrophy. A No Observed Adverse Effect Level (NOAEL) was not identified. Intravenous administration of the lowest dose of aflibercept assessed in monkeys (3 mg per kg) resulted in systemic exposure (AUC) for free aflibercept that was approximately 1500 times higher than the systemic exposure observed in adult patients after an intravitreal dose of 2 mg. All changes were reversible within 20 weeks after cessation of treatment.
1.1 Neovascular (wet) Age Related Macular Degeneration (amd)
2.2 Neovascular (wet) Age Related Macular Degeneration (amd)
The recommended dose for PAVBLU is 2 mg (0.05 mL of 40 mg/mL solution) administered by intravitreal injection every 4 weeks (approximately every 28 days, monthly) for the first 12 weeks (3 months), followed by 2 mg (0.05 mL of 40 mg/mL solution) via intravitreal injection once every 8 weeks (2 months). Although PAVBLU may be dosed as frequently as 2 mg every 4 weeks (approximately every 25 days, monthly), additional efficacy was not demonstrated in most patients when aflibercept was dosed every 4 weeks compared to every 8 weeks [see Clinical Studies (14.1)]. Some patients may need every 4 week (monthly) dosing after the first 12 weeks (3 months). Although not as effective as the recommended every 8 week dosing regimen, patients may also be treated with one dose every 12 weeks after one year of effective therapy. Patients should be assessed regularly.
14.1 Neovascular (wet) Age Related Macular Degeneration (amd)
The safety and efficacy of aflibercept were assessed in two randomized, multi-center, double-masked, active-controlled studies in patients with wet AMD. A total of 2412 patients were treated and evaluable for efficacy (1817 with aflibercept) in the two studies (VIEW1 and VIEW2). In each study, up to week 52, patients were randomly assigned in a 1:1:1:1 ratio to 1 of 4 dosing regimens: 1) aflibercept administered 2 mg every 8 weeks following 3 initial monthly doses (aflibercept 2Q8); 2) aflibercept administered 2 mg every 4 weeks (aflibercept 2Q4); 3) aflibercept 0.5 mg administered every 4 weeks (aflibercept 0.5Q4); and 4) ranibizumab administered 0.5 mg every 4 weeks (ranibizumab 0.5 mg Q4). Protocol-specified visits occurred every 28±3 days. Patient ages ranged from 49 to 99 years with a mean of 76 years.
In both studies, the primary efficacy endpoint was the proportion of patients who maintained vision, defined as losing fewer than 15 letters of visual acuity at week 52 compared to baseline. Both aflibercept 2Q8 and aflibercept 2Q4 groups were shown to have efficacy that was clinically equivalent to the ranibizumab 0.5 mg Q4 group in year 1.
Detailed results from the analysis of the VIEW1 and VIEW2 studies are shown in Table 4 and Figure 13 below.
| VIEW 1 | VIEW 2 | |||||
|---|---|---|---|---|---|---|
| Aflibercept 2 mg Q8 weeks After treatment initiation with 3 monthly doses
|
Aflibercept 2 mg Q4 weeks |
Ranibizumab 0.5 mg Q4 weeks |
Aflibercept 2 mg Q8 weeks |
Aflibercept 2 mg Q4 weeks |
Ranibizumab 0.5 mg Q4 weeks |
|
| Full Analysis Set | N=301 | N=304 | N=304 | N=306 | N=309 | N=291 |
| BCVA = Best Corrected Visual Acuity; CI = Confidence Interval; ETDRS = Early Treatment Diabetic Retinopathy Study; LOCF = Last Observation Carried Forward (baseline values are not carried forward); 95.1% confidence intervals were presented to adjust for safety assessment conducted during the study | ||||||
| Efficacy Outcomes | ||||||
| Proportion of patients who maintained visual acuity (%) (<15 letters of BCVA loss) |
94% | 95% | 94% | 95% | 95% | 95% |
| Difference Aflibercept group minus the ranibizumab group (%)(95.1% CI) |
0.6 (-3.2, 4.4) |
1.3 (-2.4, 5.0) |
0.6 (-2.9, 4.0) |
-0.3 (-4.0, 3.3) |
||
| Mean change in BCVA as measured by ETDRS letter score from Baseline | 7.9 | 10.9 | 8.1 | 8.9 | 7.6 | 9.4 |
| Difference in LS mean (95.1% CI) |
0.3 (-2.0, 2.5) |
3.2 (0.9, 5.4) |
-0.9 (-3.1, 1.3) |
-2.0 (-4.1, 0.2) |
||
| Number of patients who gained at least 15 letters of vision from Baseline (%) | 92 (31%) |
114 (38%) |
94 (31%) |
96 (31%) |
91 (29%) |
99 (34%) |
| Difference (%) (95.1% CI) |
-0.4 (-7.7, 7.0) |
6.6 (-1.0, 14.1) |
-2.6 (-10.2, 4.9) |
-4.6 (-12.1, 2.9) |
Treatment effects in evaluable subgroups (e.g., age, gender, race, baseline visual acuity) in each study were in general consistent with the results in the overall populations.
|
Figure 13. Mean Change in Visual Acuity from Baseline to Week 96 Patient dosing schedules were individualized from weeks 52 to 96 using a modified 12-week dosing regimen. in VIEW1 and VIEW2 Studies
|
VIEW1 and VIEW2 studies were both 96 weeks in duration. However after 52 weeks patients no longer followed a fixed dosing schedule. Between week 52 and week 96, patients continued to receive the drug and dosage strength to which they were initially randomized on a modified 12 week dosing schedule (doses at least every 12 weeks and additional doses as needed). Therefore, during the second year of these studies there was no active control comparison arm.
14.3 Macular Edema Following Branch Retinal Vein Occlusion (brvo)
The safety and efficacy of aflibercept were assessed in a 24-week, randomized, multi-center, double-masked, controlled study in patients with macular edema following BRVO. A total of 181 patients were treated and evaluable for efficacy (91 with aflibercept) in the VIBRANT study. In the study, patients were randomly assigned in a 1:1 ratio to either 2 mg aflibercept administered every 4 weeks (2Q4) or laser photocoagulation administered at baseline and subsequently as needed (control group). Protocol-specified visits occurred every 28±7 days. Patient ages ranged from 42 to 94 years with a mean of 65 years.
In the VIBRANT study, the primary efficacy endpoint was the proportion of patients who gained at least 15 letters in BCVA at week 24 compared to baseline. At week 24, the aflibercept 2 mg Q4 group was superior to the control group for the primary endpoint.
Detailed results from the analysis of the VIBRANT study are shown in Table 6 and Figure 15 below.
| VIBRANT | ||
|---|---|---|
| Control | Aflibercept 2 mg Q4 weeks |
|
| N=90 | N=91 | |
| Efficacy Outcomes | ||
| Proportion of patients who gained at least 15 letters in BCVA from Baseline (%) | 26.7% | 52.7% |
| Weighted Difference Difference is aflibercept 2 mg Q4 weeks minus Control
,
Difference and CI are calculated using Mantel-Haenszel weighting scheme adjusted for region (North America vs. Japan) and baseline BCVA category (> 20/200 and ≤ 20/200) (%) |
26.6% p<0.01 compared with Control
|
|
| (95% CI) | (13.0, 40.1) | |
| Mean change in BCVA as measured by ETDRS letter score from Baseline (SD) | 6.9 | 17.0 |
| (12.9) | (11.9) | |
| Difference in LS mean
,
LS mean and CI based on an ANCOVA model
|
10.5 | |
| (95.1% CI) | (7.1, 14.0) |
|
Figure 15. Mean Change in BCVA as Measured by ETDRS Letter Score from Baseline to Week 24 in VIBRANT Study
|
Treatment effects in evaluable subgroups (e.g., age, gender, and baseline retinal perfusion status) in the study were in general consistent with the results in the overall populations.
14.2 Macular Edema Following Central Retinal Vein Occlusion (crvo)
The safety and efficacy of aflibercept were assessed in two randomized, multi-center, double-masked, sham-controlled studies in patients with macular edema following CRVO. A total of 358 patients were treated and evaluable for efficacy (217 with aflibercept) in the two studies (COPERNICUS and GALILEO). In both studies, patients were randomly assigned in a 3:2 ratio to either 2 mg aflibercept administered every 4 weeks (2Q4), or sham injections (control group) administered every 4 weeks for a total of 6 injections. Protocol-specified visits occurred every 28±7 days. Patient ages ranged from 22 to 89 years with a mean of 64 years.
In both studies, the primary efficacy endpoint was the proportion of patients who gained at least 15 letters in BCVA compared to baseline. At week 24, the aflibercept 2 mg Q4 group was superior to the control group for the primary endpoint.
Results from the analysis of the COPERNICUS and GALILEO studies are shown in Table 5 and Figure 14 below.
| COPERNICUS | GALILEO | |||
|---|---|---|---|---|
| Control | Aflibercept 2 mg Q4 weeks |
Control | Aflibercept 2 mg Q4 weeks |
|
| N=73 | N=114 | N=68 | N=103 | |
| Efficacy Outcomes | ||||
| Proportion of patients who gained at least 15 letters in BCVA from Baseline (%) | 12% | 56% | 22% | 60% |
| Weighted Difference Difference is aflibercept 2 mg Q4 weeks minus Control
,
Difference and CI are calculated using Cochran-Mantel-Haenszel (CMH) test adjusted for baseline factors; 95.1% confidence intervals were presented to adjust for the multiple assessments conducted during the study (%) |
44.8% p<0.01 compared with Control
|
38.3% | ||
| (95.1% CI) | (32.9, 56.6) | (24.4, 52.1) | ||
| Mean change in BCVA as measured by ETDRS letter score from Baseline (SD) | -4.0 | 17.3 | 3.3 | 18.0 |
| (18.0) | (12.8) | (14.1) | (12.2) | |
| Difference in LS mean
,
LS mean and CI based on an ANCOVA model
|
21.7 | 14.7 | ||
| (95.1% CI) | (17.3, 26.1) | (10.7, 18.7) |
|
Figure 14. Mean Change in BCVA as Measured by ETDRS Letter Score from Baseline to Week 24 in COPERNICUS and GALILEO Studies
|
Treatment effects in evaluable subgroups (e.g., age, gender, race, baseline visual acuity, retinal perfusion status, and CRVO duration) in each study and in the combined analysis were in general consistent with the results in the overall populations.
5.1 Endophthalmitis, Retinal Detachments, and Retinal Vasculitis With Or Without Occlusion
Intravitreal injections, including those with aflibercept products, have been associated with endophthalmitis and retinal detachments [see Adverse Reactions (6.1)] and, more rarely, retinal vasculitis with or without occlusion [see Adverse Reactions (6.2)]. Proper aseptic injection technique must always be used when administering PAVBLU. Patients and/or caregivers should be instructed to report any signs and/or symptoms suggestive of endophthalmitis, retinal detachment, or retinal vasculitis without delay and should be managed appropriately [see Dosage and Administration (2.8) and Patient Counseling Information (17)].
Structured Label Content
Section 42229-5 (42229-5)
Prefilled syringe: Inject by pressing the plunger carefully and with constant pressure. Do not apply additional pressure once the plunger has reached the bottom of the syringe. A small residual volume may remain in the syringe after a full dose has been injected. This is normal. Do not administer any residual solution observed in the syringe.
Immediately following the intravitreal injection, patients should be monitored for elevation in intraocular pressure. Appropriate monitoring may consist of a check for perfusion of the optic nerve head or tonometry. If required, a sterile paracentesis needle should be available.
Following intravitreal injection, patients and/or caregivers should be instructed to report any signs and/or symptoms suggestive of endophthalmitis or retinal detachment (e.g., eye pain, redness of the eye, photophobia, blurring of vision) without delay [see Patient Counseling Information (17)].
Each sterile, prefilled syringe or vial should only be used for the treatment of a single eye. If the contralateral eye requires treatment, a new sterile, prefilled syringe or vial should be used and the sterile field, syringe, gloves, drapes, eyelid speculum, filter, and injection needles should be changed before PAVBLU is administered to the other eye.
After injection, any unused product must be discarded.
10 Overdosage (10 OVERDOSAGE)
Overdosing with increased injection volume may increase intraocular pressure. Therefore, in case of overdosage, intraocular pressure should be monitored and if deemed necessary by the treating physician, adequate treatment should be initiated.
11 Description (11 DESCRIPTION)
Aflibercept-ayyh is a recombinant fusion protein consisting of portions of human VEGF receptors 1 and 2 extracellular domains fused to the Fc portion of human IgG1 formulated as an iso-osmotic solution for intravitreal administration. Aflibercept-ayyh is a dimeric glycoprotein with a protein molecular weight of 97 kilodaltons (kDa) and contains glycosylation, constituting an additional 15% of the total molecular mass, resulting in a total molecular weight of 115 kDa. Aflibercept-ayyh is produced in recombinant Chinese hamster ovary (CHO) cells.
PAVBLU (aflibercept-ayyh) injection is a sterile, clear to opalescent and colorless to slightly yellow solution. PAVBLU does not contain anti-microbial preservative and is supplied as an aqueous solution for intravitreal injection in a single-dose prefilled plastic syringe or a single-dose glass vial designed to deliver 0.05 mL (50 microliters) of solution containing 2 mg of aflibercept-ayyh in polysorbate 80 (0.005 mg), sucrose (2.5 mg), trehalose (1.58 mg) and water for injection with a pH of 6.2.
16.1 How Supplied
Each prefilled syringe or vial is for single eye use only. Discard unused portion. PAVBLU injection is a clear to opalescent and colorless to slightly yellow solution supplied in the following presentations:
| NDC NUMBER | CARTON TYPE | CARTON CONTENTS |
|---|---|---|
| 55513-056-01 | Prefilled Syringe | one blister pack containing one PAVBLU 2 mg (0.05 mL of a 40 mg/mL solution) sterile, single-dose prefilled plastic syringe one Prescribing Information |
| 55513-065-01 | Vial Only | one PAVBLU 2 mg (0.05 mL of a 40 mg/mL solution) single-dose glass vial one Prescribing Information |
8.4 Pediatric Use
A pediatric assessment for PAVBLU demonstrates that PAVBLU is safe and effective for pediatric patients in an indication for which EYLEA (aflibercept) is approved. However, PAVBLU is not approved for such indication due to marketing exclusivity for EYLEA (aflibercept).
8.5 Geriatric Use
In the clinical studies, approximately 76% (2049/2701) of patients randomized to treatment with aflibercept were ≥65 years of age and approximately 46% (1250/2701) were ≥75 years of age. No significant differences in efficacy or safety were seen with increasing age in these studies.
12.6 Immunogenicity
The observed incidence of anti-drug antibodies is highly dependent on the sensitivity and specificity of the assay. Differences in assay methods preclude meaningful comparisons of the incidence of anti-drug antibodies in the studies described below with the incidence of anti-drug antibodies in other studies, including those of aflibercept or of other aflibercept products.
In the wet AMD, RVO, and DME studies, the pre-treatment incidence of immunoreactivity to aflibercept was approximately 1% to 3% across treatment groups. After dosing with aflibercept for 24-100 weeks, antibodies to aflibercept were detected in a similar percentage range of patients.
There were no differences in efficacy or safety between patients with or without immunoreactivity.
4 Contraindications (4 CONTRAINDICATIONS)
4.3 Hypersensitivity
PAVBLU is contraindicated in patients with known hypersensitivity to aflibercept or any of the excipients in PAVBLU. Hypersensitivity reactions may manifest as rash, pruritus, urticaria, severe anaphylactic/anaphylactoid reactions, or severe intraocular inflammation.
6. Adverse Reactions (6. ADVERSE REACTIONS)
The following potentially serious adverse reactions are described elsewhere in the labeling:
- Hypersensitivity [see Contraindications (4.3)]
- Endophthalmitis, retinal detachments, and Retinal Vasculitis with or without Occlusion [see Warnings and Precautions (5.1)]
- Increase in intraocular pressure [see Warnings and Precautions (5.2)]
- Thromboembolic events [see Warnings and Precautions (5.4)]
12.3 Pharmacokinetics
Aflibercept is administered intravitreally to exert local effects in the eye. In patients with wet AMD, RVO, or DME, following intravitreal administration of aflibercept, a fraction of the administered dose is expected to bind with endogenous VEGF in the eye to form an inactive aflibercept: VEGF complex. Once absorbed into the systemic circulation, aflibercept presents in the plasma as free aflibercept (unbound to VEGF) and a more predominant stable inactive form with circulating endogenous VEGF (i.e., aflibercept: VEGF complex).
1 Indications and Usage (1 INDICATIONS AND USAGE)
PAVBLU is indicated for the treatment of:
2.8 Injection Procedure
The intravitreal injection procedure should be carried out under controlled aseptic conditions, which include surgical hand disinfection and the use of sterile gloves, a sterile drape, and a sterile eyelid speculum (or equivalent). Adequate anesthesia and a topical broad−spectrum microbicide should be given prior to the injection.
12.1 Mechanism of Action
Vascular endothelial growth factor-A (VEGF-A) and placental growth factor (PlGF) are members of the VEGF family of angiogenic factors that can act as mitogenic, chemotactic, and vascular permeability factors for endothelial cells. VEGF acts via two receptor tyrosine kinases, VEGFR-1 and VEGFR-2, present on the surface of endothelial cells. PlGF binds only to VEGFR-1, which is also present on the surface of leucocytes. Activation of these receptors by VEGF-A can result in neovascularization and vascular permeability.
Aflibercept products act as a soluble decoy receptor that binds VEGF-A and PlGF, and thereby can inhibit the binding and activation of these cognate VEGF receptors.
16.2 Storage and Handling
Refrigerate PAVBLU at 2°C to 8ºC (36°F to 46ºF). PAVBLU may be kept at room temperature (up to 30° C (86° F)) for a single-period of 3 days. Do not freeze. Do not use beyond the date stamped on the carton and container label. Store in the original carton until time of use to protect from light. Do not open sealed blister tray until time of use.
5.4 Thromboembolic Events
There is a potential risk of arterial thromboembolic events (ATEs) following intravitreal use of VEGF inhibitors, including aflibercept products. ATEs are defined as nonfatal stroke, nonfatal myocardial infarction, or vascular death (including deaths of unknown cause). The incidence of reported thromboembolic events in wet AMD studies during the first year was 1.8% (32 out of 1824) in the combined group of patients treated with aflibercept compared with 1.5% (9 out of 595) in patients treated with ranibizumab; through 96 weeks, the incidence was 3.3% (60 out of 1824) in the aflibercept group compared with 3.2% (19 out of 595) in the ranibizumab group. The incidence in the DME studies from baseline to week 52 was 3.3% (19 out of 578) in the combined group of patients treated with aflibercept compared with 2.8% (8 out of 287) in the control group; from baseline to week 100, the incidence was 6.4% (37 out of 578) in the combined group of patients treated with aflibercept compared with 4.2% (12 out of 287) in the control group. There were no reported thromboembolic events in the patients treated with aflibercept in the first six months of the RVO studies.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Endophthalmitis, retinal detachments, and retinal vasculitis with or without occlusion may occur following intravitreal injections. Patients and/or caregivers should be instructed to report any signs and/or symptoms suggestive of endophthalmitis, retinal detachment, or retinal vasculitis without delay and should be managed appropriately. (5.1)
- Increases in intraocular pressure have been seen within 60 minutes of an intravitreal injection. (5.2)
- There is a potential risk of arterial thromboembolic events following intravitreal use of VEGF inhibitors. (5.4)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
Neovascular (Wet) Age-Related Macular Degeneration (AMD):
- The recommended dose for PAVBLU is 2 mg (0.05 mL of 40 mg/mL solution) administered by intravitreal injection every 4 weeks (approximately every 28 days, monthly) for the first 3 months, followed by 2 mg (0.05 mL of 40 mg/mL solution) via intravitreal injection once every 8 weeks (2 months). (2.2)
- Although PAVBLU may be dosed as frequently as 2 mg every 4 weeks (approximately every 25 days, monthly), additional efficacy was not demonstrated in most patients when aflibercept was dosed every 4 weeks compared to every 8 weeks. Some patients may need every 4 week (monthly) dosing after the first 12 weeks (3 months). (2.2)
- Although not as effective as the recommended every 8 week dosing regimen, patients may also be treated with one dose every 12 weeks after one year of effective therapy. Patients should be assessed regularly. (2.2)
Macular Edema Following Retinal Vein Occlusion (RVO):
- The recommended dose for PAVBLU is 2 mg (0.05 mL of 40 mg/mL solution) administered by intravitreal injection once every 4 weeks (approximately every 25 days, monthly). (2.3)
Diabetic Macular Edema (DME) and Diabetic Retinopathy (DR):
- The recommended dose for PAVBLU is 2 mg (0.05 mL of 40 mg/mL solution) administered by intravitreal injection every 4 weeks (approximately every 28 days, monthly) for the first 5 injections followed by 2 mg (0.05 mL of 40 mg/mL solution) via intravitreal injection once every 8 weeks (2 months). (2.4, 2.5)
- Although PAVBLU may be dosed as frequently as 2 mg every 4 weeks (approximately every 25 days, monthly), additional efficacy was not demonstrated in most patients when aflibercept was dosed every 4 weeks compared to every 8 weeks. Some patients may need every 4 week (monthly) dosing after the first 20 weeks (5 months). (2.4, 2.5)
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
PAVBLU is a clear to opalescent and colorless to slightly yellow solution available as:
- Injection: 2 mg (0.05 mL of 40 mg/mL solution) in a single-dose prefilled plastic syringe
- Injection: 2 mg (0.05 mL of 40 mg/mL solution) in a single-dose glass vial
6.2 Postmarketing Experience
The following adverse reactions have been identified during postapproval use of aflibercept. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Eye disorders: retinal vasculitis and occlusive retinal vasculitis related to intravitreal injection with aflibercept (reported at a rate of 0.6 and 0.2 per 1 million injections, respectively, based on postmarketing experience from November 2011 until November 2023).
1.4 Diabetic Retinopathy (dr) (1.4 Diabetic Retinopathy (DR))
2.5 Diabetic Retinopathy (dr) (2.5 Diabetic Retinopathy (DR))
The recommended dose for PAVBLU is 2 mg (0.05 mL of 40 mg/mL solution) administered by intravitreal injection every 4 weeks (approximately every 28 days, monthly) for the first 5 injections, followed by 2 mg (0.05 mL of 40 mg/mL solution) via intravitreal injection once every 8 weeks (2 months). Although PAVBLU may be dosed as frequently as 2 mg every 4 weeks (approximately every 25 days, monthly), additional efficacy was not demonstrated in most patients when aflibercept was dosed every 4 weeks compared to every 8 weeks [see Clinical Studies (14.5)]. Some patients may need every 4 week (monthly) dosing after the first 20 weeks (5 months).
14.5 Diabetic Retinopathy (dr) (14.5 Diabetic Retinopathy (DR))
Efficacy and safety data of aflibercept in diabetic retinopathy (DR) are derived from the VIVID, VISTA, and PANORAMA studies.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in other clinical trials of the same or another drug and may not reflect the rates observed in practice.
A total of 2980 adult patients treated with aflibercept constituted the safety population in eight phase 3 studies. Among those, 2379 patients were treated with the recommended dose of 2 mg. Serious adverse reactions related to the injection procedure have occurred in <0.1% of intravitreal injections with aflibercept including endophthalmitis and retinal detachment. The most common adverse reactions (≥5%) reported in patients receiving aflibercept were conjunctival hemorrhage, eye pain, cataract, vitreous detachment, vitreous floaters, and intraocular pressure increased.
1.3 Diabetic Macular Edema (dme) (1.3 Diabetic Macular Edema (DME))
2.4 Diabetic Macular Edema (dme) (2.4 Diabetic Macular Edema (DME))
The recommended dose for PAVBLU is 2 mg (0.05 mL of 40 mg/mL solution) administered by intravitreal injection every 4 weeks (approximately every 28 days, monthly) for the first 5 injections, followed by 2 mg (0.05 mL of 40 mg/mL solution) via intravitreal injection once every 8 weeks (2 months). Although PAVBLU may be dosed as frequently as 2 mg every 4 weeks (approximately every 25 days, monthly), additional efficacy was not demonstrated in most patients when aflibercept was dosed every 4 weeks compared to every 8 weeks [see Clinical Studies (14.4)]. Some patients may need every 4 week (monthly) dosing after the first 20 weeks (5 months).
14.4 Diabetic Macular Edema (dme) (14.4 Diabetic Macular Edema (DME))
The safety and efficacy of aflibercept were assessed in two randomized, multi-center, double-masked, controlled studies in patients with DME. A total of 862 randomized and treated patients were evaluable for efficacy. Protocol-specified visits occurred every 28±7 days. Patient ages ranged from 23 to 87 years with a mean of 63 years.
Of those, 576 were randomized to aflibercept groups in the two studies (VIVID and VISTA). In each study, patients were randomly assigned in a 1:1:1 ratio to 1 of 3 dosing regimens: 1) aflibercept administered 2 mg every 8 weeks following 5 initial monthly injections (aflibercept 2Q8); 2) aflibercept administered 2 mg every 4 weeks (aflibercept 2Q4); and 3) macular laser photocoagulation (at baseline and then as needed). Beginning at week 24, patients meeting a pre-specified threshold of vision loss were eligible to receive additional treatment: patients in the aflibercept groups could receive laser and patients in the laser group could receive aflibercept.
In both studies, the primary efficacy endpoint was the mean change from baseline in BCVA at week 52 as measured by ETDRS letter score. Efficacy of both aflibercept 2Q8 and aflibercept 2Q4 groups was statistically superior to the control group. This statistically superior improvement in BCVA was maintained at week 100 in both studies.
Results from the analysis of the VIVID and VISTA studies are shown in Table 7 and Figure 16 below.
| VIVID | VISTA | |||||
|---|---|---|---|---|---|---|
| Aflibercept 2 mg Q8 weeks After treatment initiation with 5 monthly injections
|
Aflibercept 2 mg Q4 weeks |
Control | Aflibercept 2 mg Q8 weeks |
Aflibercept 2 mg Q4 weeks |
Control | |
| Full Analysis Set | N=135 | N=136 | N=132 | N=151 | N=154 | N=154 |
| Efficacy Outcomes at Week 52 | ||||||
| Mean change in BCVA as measured by ETDRS letter score from Baseline (SD) | 10.7 | 10.5 | 1.2 | 10.7 | 12.5 | 0.2 |
| (9.3) | (9.6) | (10.6) | (8.2) | (9.5) | (12.5) | |
| Difference LS mean and CI based on an ANCOVA model with baseline BCVA measurement as a covariate and a factor for treatment group. Additionally, protocol specified stratification factors were included in the model.
,
Difference is aflibercept group minus Control group in LS mean |
9.1 p<0.01 compared with Control
|
9.3 | 10.5 | 12.2 | ||
| (97.5% CI) | (6.3, 11.8) | (6.5, 12.0) | (7.7, 13.2) | (9.4, 15.0) | ||
| Proportion of patients who gained at least 15 letters in BCVA from Baseline (%) | 33.3% | 32.4% | 9.1% | 31.1% | 41.6% | 7.8% |
| Adjusted Difference
,
Difference with confidence interval (CI) and statistical test is calculated using Mantel-Haenszel weighting scheme adjusted by protocol specified stratification factors
(%) |
24.2% | 23.3% | 23.3% | 34.2% | ||
| (97.5% CI) | (13.5, 34.9) | (12.6, 33.9) | (13.5, 33.1) | (24.1, 44.4) | ||
| Efficacy Outcomes at Week 100 | ||||||
| Mean change in BCVA as measured by ETDRS letter score from Baseline (SD) | 9.4 | 11.4 | 0.7 | 11.1 | 11.5 | 0.9 |
| (10.5) | (11.2) | (11.8) | (10.7) | (13.8) | (13.9) | |
| Difference
,
in LS mean |
8.2 | 10.7 | 10.1 | 10.6 | ||
| (97.5% CI) | (5.2, 11.3) | (7.6, 13.8) | (7.0, 13.3) | (7.1, 14.2) | ||
| Proportion of patients who gained at least 15 letters in BCVA from Baseline (%) | 31.1% | 38.2% | 12.1% | 33.1% | 38.3% | 13.0% |
| Adjusted Difference
,
(%) |
19.0% | 26.1% | 20.1% | 25.8% | ||
| (97.5% CI) | (8.0, 29.9) | (14.8, 37.5) | (9.6, 30.6) | (15.1, 36.6) |
|
Figure 16. Mean Change in BCVA as Measured by ETDRS Letter Score from Baseline to Week 100 in VIVID and VISTA Studies
|
Treatment effects in the subgroup of patients who had previously been treated with a VEGF inhibitor prior to study participation were similar to those seen in patients who were VEGF inhibitor naïve prior to study participation.
Treatment effects in evaluable subgroups (e.g., age, gender, race, baseline HbA1c, baseline visual acuity, prior anti-VEGF therapy) in each study were in general consistent with the results in the overall populations.
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
In the days following PAVBLU administration, patients are at risk of developing endophthalmitis, retinal detachment, or retinal vasculitis with or without occlusion. If the eye becomes red, sensitive to light, painful, or develops a change in vision, advise patients and/or caregivers to seek immediate care from an ophthalmologist [see Warnings and Precautions (5.1)].
Patients may experience temporary visual disturbances after an intravitreal injection with PAVBLU and the associated eye examinations [see Adverse Reactions (6)]. Advise patients not to drive or use machinery until visual function has recovered sufficiently.
4.1 Ocular Or Periocular Infections (4.1 Ocular or Periocular Infections)
PAVBLU is contraindicated in patients with ocular or periocular infections.
4.2 Active Intraocular Inflammation
PAVBLU is contraindicated in patients with active intraocular inflammation.
2.1 Important Injection Instructions
For ophthalmic intravitreal injection. PAVBLU must only be administered by a qualified physician.
Prefilled Syringe: A 30-gauge × ½-inch sterile injection needle is needed but not provided.
Vial: A 5-micron sterile filter needle (18-gauge × 1½-inch), a 1-mL Luer lock syringe and a 30-gauge × ½-inch sterile injection needle are needed but not provided.
PAVBLU is available packaged as follows:
- Prefilled Syringe
- Vial Only
5.2 Increase in Intraocular Pressure
Acute increases in intraocular pressure have been seen within 60 minutes of intravitreal injection, including with aflibercept products [see Adverse Reactions (6.1)]. Sustained increases in intraocular pressure have also been reported after repeated intravitreal dosing with vascular endothelial growth factor (VEGF) inhibitors. Intraocular pressure and the perfusion of the optic nerve head should be monitored and managed appropriately [see Dosage and Administration (2.8)].
2.7 Preparation for Administration Vial (2.7 Preparation for Administration - Vial)
PAVBLU should be inspected visually prior to administration. It is a clear to opalescent and colorless to slightly yellow solution. If particulates, cloudiness, or discoloration are visible, the vial must not be used.
The glass vial is for one-time use in one eye only.
Use aseptic technique to carry out the following preparation steps:
Prepare for intravitreal injection with the following medical devices for single use:
- a 5-micron sterile filter needle 18-gauge × 1½-inch (not provided)
- a 1-mL sterile Luer lock syringe with marking to measure 0.05 mL (not provided)
- a sterile injection needle 30-gauge × ½-inch (not provided)
-
1.Remove the protective plastic cap from the vial (see Figure 6).
Figure 6:
-
2.Clean the top of the vial with an alcohol wipe (see Figure 7).
Figure 7:
-
3.Remove the 18-gauge × 1½-inch, 5-micron, filter needle and the 1-mL syringe from their packaging. Attach the filter needle to the syringe by twisting it onto the Luer lock syringe tip (see Figure 8a and Figure 8b).
| Figure 8a: | Figure 8b: |
|
|
|
Note: When ready to withdraw PAVBLU, remove the plastic needle shield from the needle.
-
4.Push the filter needle into the center of the vial stopper until the needle is completely inserted into the vial and the tip touches the bottom or bottom edge of the vial.
-
5.Using aseptic technique withdraw all of the PAVBLU vial contents into the syringe, keeping the vial in an upright position, slightly inclined to ease complete withdrawal. To deter the introduction of air, ensure the bevel of the filter needle is submerged into the liquid. Continue to tilt the vial during withdrawal keeping the bevel of the filter needle submerged in the liquid (see Figure 9a and Figure 9b).
|
Figure 9a: Figure 9b:
|
-
6.Ensure that the plunger rod is drawn sufficiently back when emptying the vial in order to completely empty the filter needle.
-
7.Remove the filter needle from the syringe and properly dispose of the filter needle. Note: Filter needle is not to be used for intravitreal injection.
-
8.Remove the 30-gauge × ½-inch injection needle from its packaging and attach the injection needle to the syringe by firmly twisting the injection needle onto the Luer lock syringe tip (see Figure 10a and Figure 10b).
|
Figure 10a:
|
Figure 10b:
|
Note: When ready to administer PAVBLU, remove the plastic needle shield from the needle.
-
9.Holding the syringe with the needle pointing up, check the syringe for bubbles. If there are bubbles, gently tap the syringe with your finger until the bubbles rise to the top (see Figure 11).
Figure 11:
-
10.To eliminate all of the bubbles and to expel excess drug, SLOWLY depress the plunger rod so that the plunger edge aligns with the line that marks 0.05 mL on the syringe (see Figure 12a and Figure 12b).
13.2 Animal Toxicology And/or Pharmacology (13.2 Animal Toxicology and/or Pharmacology)
Erosions and ulcerations of the respiratory epithelium in nasal turbinates in monkeys treated with aflibercept intravitreally were observed at intravitreal doses of 2 or 4 mg per eye. At the NOAEL of 0.5 mg per eye in monkeys, the systemic exposure (AUC) was 56 times higher than the exposure observed in adult patients after an intravitreal dose of 2 mg. Similar effects were not seen in other clinical studies [see Clinical Studies (14)].
Principal Display Panel 0.05 Ml Vial Carton (PRINCIPAL DISPLAY PANEL - 0.05 mL Vial Carton)
2 mg /
0.05 mL
NDC 55513-065-01
PAVBLU™
(aflibercept-ayyh)
Injection
2 mg (0.05 mL of
40 mg/mL solution)
For Intravitreal Injection
One Single-Dose Vial
Sterile Solution
Contains no anti-microbial
preservatives.
Keep out of reach of children
Discard unused portion
AMGEN®
Rx Only
Principal Display Panel 0.05 Ml Syringe Carton (PRINCIPAL DISPLAY PANEL - 0.05 mL Syringe Carton)
2 mg /
0.05 mL
AMGEN®
PAVBLU™
(aflibercept-ayyh)
Injection
NDC 55513-056-01
2 mg (0.05 mL of 40 mg/mL solution)
Carton contents: one blister pack containing one sterile, single-dose prefilled plastic
syringe and package insert.
Prefilled syringe contents:
Each PAVBLU™ single-dose prefilled syringe is designed to deliver 2 mg of aflibercept-ayyh
in 0.05 mL (50 microliters) solution with polysorbate 80 (0.005 mg), sucrose (2.5 mg),
trehalose (1.58 mg) and water for injection with a pH of 6.2.
Recommended Dosage: See prescribing information.
Rx Only
2.6 Preparation for Administration Prefilled Syringe (2.6 Preparation for Administration - Prefilled Syringe)
The PAVBLU prefilled plastic syringe is sterile and for one-time use in one eye only.
The prefilled syringe should be inspected visually prior to administration. It is a clear to opalescent and colorless to slightly yellow solution. Do not use if particulates, cloudiness, or discoloration are visible, or if the package is open or damaged. The appearance of the syringe cap on the prefilled syringe may vary (for example, color and design).
Do not use if any part of the prefilled syringe is damaged or if the syringe cap is detached from the Luer lock.
The intravitreal injection should be performed with a 30-gauge × ½-inch injection needle (not provided).
The prefilled syringe contains more than the recommended dose of 2 mg aflibercept-ayyh (equivalent to 50 microliters). The excess volume must be discarded prior to the administration.
PREFILLED SYRINGE DESCRIPTION - Figure 1:
Use aseptic technique to carry out the following steps:
-
1.PREPARE
When ready to administer PAVBLU, open the carton and remove sterilized blister pack. Carefully peel open the sterilized blister pack ensuring the sterility of its contents. Keep the syringe in the sterile tray until you are ready for assembly.
-
2.REMOVE SYRINGE
Using aseptic technique, remove the syringe from the sterilized blister pack.
-
3.TWIST OFF SYRINGE CAP
Twist off (do not snap off) the syringe cap by holding the syringe in one hand and the syringe cap with the thumb and forefinger of the other hand (see Figure 2).
Figure 2:
-
4.ATTACH NEEDLE
Using aseptic technique, firmly twist a 30-gauge × ½-inch injection needle onto the Luer lock syringe tip (see Figure 3).
Figure 3:
Note: When ready to administer PAVBLU, remove the plastic needle shield from the needle.
-
5.DISLODGE AIR BUBBLES
Holding the syringe with the needle pointing up, check the syringe for bubbles. If there are bubbles, gently tap the syringe with your finger until the bubbles rise to the top (see Figure 4).
Figure 4:
-
6.EXPEL AIR AND SET THE DOSE
To eliminate all bubbles and to expel excess drug, slowly depress the plunger rod to align the plunger dome edge (see Figure 5a) with the black dosing line on the syringe (equivalent to 50 microliters) (see Figure 5b).
|
Figure 5a:
|
Figure 5b:
|
-
7.The prefilled syringe is for one-time use in one eye only. After injection any unused product must be discarded.
1.2 Macular Edema Following Retinal Vein Occlusion (rvo) (1.2 Macular Edema Following Retinal Vein Occlusion (RVO))
2.3 Macular Edema Following Retinal Vein Occlusion (rvo) (2.3 Macular Edema Following Retinal Vein Occlusion (RVO))
The recommended dose for PAVBLU is 2 mg (0.05 mL of 40 mg/mL solution) administered by intravitreal injection once every 4 weeks (approximately every 25 days, monthly) [see Clinical Studies (14.2), (14.3)].
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
No studies have been conducted on the mutagenic or carcinogenic potential of aflibercept products. Effects on male and female fertility were assessed as part of a 6-month study in monkeys with intravenous administration of aflibercept at weekly doses ranging from 3 to 30 mg per kg. Absent or irregular menses associated with alterations in female reproductive hormone levels and changes in sperm morphology and motility were observed at all dose levels. In addition, females showed decreased ovarian and uterine weight accompanied by compromised luteal development and reduction of maturing follicles. These changes correlated with uterine and vaginal atrophy. A No Observed Adverse Effect Level (NOAEL) was not identified. Intravenous administration of the lowest dose of aflibercept assessed in monkeys (3 mg per kg) resulted in systemic exposure (AUC) for free aflibercept that was approximately 1500 times higher than the systemic exposure observed in adult patients after an intravitreal dose of 2 mg. All changes were reversible within 20 weeks after cessation of treatment.
1.1 Neovascular (wet) Age Related Macular Degeneration (amd) (1.1 Neovascular (Wet) Age-Related Macular Degeneration (AMD))
2.2 Neovascular (wet) Age Related Macular Degeneration (amd) (2.2 Neovascular (Wet) Age-Related Macular Degeneration (AMD))
The recommended dose for PAVBLU is 2 mg (0.05 mL of 40 mg/mL solution) administered by intravitreal injection every 4 weeks (approximately every 28 days, monthly) for the first 12 weeks (3 months), followed by 2 mg (0.05 mL of 40 mg/mL solution) via intravitreal injection once every 8 weeks (2 months). Although PAVBLU may be dosed as frequently as 2 mg every 4 weeks (approximately every 25 days, monthly), additional efficacy was not demonstrated in most patients when aflibercept was dosed every 4 weeks compared to every 8 weeks [see Clinical Studies (14.1)]. Some patients may need every 4 week (monthly) dosing after the first 12 weeks (3 months). Although not as effective as the recommended every 8 week dosing regimen, patients may also be treated with one dose every 12 weeks after one year of effective therapy. Patients should be assessed regularly.
14.1 Neovascular (wet) Age Related Macular Degeneration (amd) (14.1 Neovascular (Wet) Age-Related Macular Degeneration (AMD))
The safety and efficacy of aflibercept were assessed in two randomized, multi-center, double-masked, active-controlled studies in patients with wet AMD. A total of 2412 patients were treated and evaluable for efficacy (1817 with aflibercept) in the two studies (VIEW1 and VIEW2). In each study, up to week 52, patients were randomly assigned in a 1:1:1:1 ratio to 1 of 4 dosing regimens: 1) aflibercept administered 2 mg every 8 weeks following 3 initial monthly doses (aflibercept 2Q8); 2) aflibercept administered 2 mg every 4 weeks (aflibercept 2Q4); 3) aflibercept 0.5 mg administered every 4 weeks (aflibercept 0.5Q4); and 4) ranibizumab administered 0.5 mg every 4 weeks (ranibizumab 0.5 mg Q4). Protocol-specified visits occurred every 28±3 days. Patient ages ranged from 49 to 99 years with a mean of 76 years.
In both studies, the primary efficacy endpoint was the proportion of patients who maintained vision, defined as losing fewer than 15 letters of visual acuity at week 52 compared to baseline. Both aflibercept 2Q8 and aflibercept 2Q4 groups were shown to have efficacy that was clinically equivalent to the ranibizumab 0.5 mg Q4 group in year 1.
Detailed results from the analysis of the VIEW1 and VIEW2 studies are shown in Table 4 and Figure 13 below.
| VIEW 1 | VIEW 2 | |||||
|---|---|---|---|---|---|---|
| Aflibercept 2 mg Q8 weeks After treatment initiation with 3 monthly doses
|
Aflibercept 2 mg Q4 weeks |
Ranibizumab 0.5 mg Q4 weeks |
Aflibercept 2 mg Q8 weeks |
Aflibercept 2 mg Q4 weeks |
Ranibizumab 0.5 mg Q4 weeks |
|
| Full Analysis Set | N=301 | N=304 | N=304 | N=306 | N=309 | N=291 |
| BCVA = Best Corrected Visual Acuity; CI = Confidence Interval; ETDRS = Early Treatment Diabetic Retinopathy Study; LOCF = Last Observation Carried Forward (baseline values are not carried forward); 95.1% confidence intervals were presented to adjust for safety assessment conducted during the study | ||||||
| Efficacy Outcomes | ||||||
| Proportion of patients who maintained visual acuity (%) (<15 letters of BCVA loss) |
94% | 95% | 94% | 95% | 95% | 95% |
| Difference Aflibercept group minus the ranibizumab group (%)(95.1% CI) |
0.6 (-3.2, 4.4) |
1.3 (-2.4, 5.0) |
0.6 (-2.9, 4.0) |
-0.3 (-4.0, 3.3) |
||
| Mean change in BCVA as measured by ETDRS letter score from Baseline | 7.9 | 10.9 | 8.1 | 8.9 | 7.6 | 9.4 |
| Difference in LS mean (95.1% CI) |
0.3 (-2.0, 2.5) |
3.2 (0.9, 5.4) |
-0.9 (-3.1, 1.3) |
-2.0 (-4.1, 0.2) |
||
| Number of patients who gained at least 15 letters of vision from Baseline (%) | 92 (31%) |
114 (38%) |
94 (31%) |
96 (31%) |
91 (29%) |
99 (34%) |
| Difference (%) (95.1% CI) |
-0.4 (-7.7, 7.0) |
6.6 (-1.0, 14.1) |
-2.6 (-10.2, 4.9) |
-4.6 (-12.1, 2.9) |
Treatment effects in evaluable subgroups (e.g., age, gender, race, baseline visual acuity) in each study were in general consistent with the results in the overall populations.
|
Figure 13. Mean Change in Visual Acuity from Baseline to Week 96 Patient dosing schedules were individualized from weeks 52 to 96 using a modified 12-week dosing regimen. in VIEW1 and VIEW2 Studies
|
VIEW1 and VIEW2 studies were both 96 weeks in duration. However after 52 weeks patients no longer followed a fixed dosing schedule. Between week 52 and week 96, patients continued to receive the drug and dosage strength to which they were initially randomized on a modified 12 week dosing schedule (doses at least every 12 weeks and additional doses as needed). Therefore, during the second year of these studies there was no active control comparison arm.
14.3 Macular Edema Following Branch Retinal Vein Occlusion (brvo) (14.3 Macular Edema Following Branch Retinal Vein Occlusion (BRVO))
The safety and efficacy of aflibercept were assessed in a 24-week, randomized, multi-center, double-masked, controlled study in patients with macular edema following BRVO. A total of 181 patients were treated and evaluable for efficacy (91 with aflibercept) in the VIBRANT study. In the study, patients were randomly assigned in a 1:1 ratio to either 2 mg aflibercept administered every 4 weeks (2Q4) or laser photocoagulation administered at baseline and subsequently as needed (control group). Protocol-specified visits occurred every 28±7 days. Patient ages ranged from 42 to 94 years with a mean of 65 years.
In the VIBRANT study, the primary efficacy endpoint was the proportion of patients who gained at least 15 letters in BCVA at week 24 compared to baseline. At week 24, the aflibercept 2 mg Q4 group was superior to the control group for the primary endpoint.
Detailed results from the analysis of the VIBRANT study are shown in Table 6 and Figure 15 below.
| VIBRANT | ||
|---|---|---|
| Control | Aflibercept 2 mg Q4 weeks |
|
| N=90 | N=91 | |
| Efficacy Outcomes | ||
| Proportion of patients who gained at least 15 letters in BCVA from Baseline (%) | 26.7% | 52.7% |
| Weighted Difference Difference is aflibercept 2 mg Q4 weeks minus Control
,
Difference and CI are calculated using Mantel-Haenszel weighting scheme adjusted for region (North America vs. Japan) and baseline BCVA category (> 20/200 and ≤ 20/200) (%) |
26.6% p<0.01 compared with Control
|
|
| (95% CI) | (13.0, 40.1) | |
| Mean change in BCVA as measured by ETDRS letter score from Baseline (SD) | 6.9 | 17.0 |
| (12.9) | (11.9) | |
| Difference in LS mean
,
LS mean and CI based on an ANCOVA model
|
10.5 | |
| (95.1% CI) | (7.1, 14.0) |
|
Figure 15. Mean Change in BCVA as Measured by ETDRS Letter Score from Baseline to Week 24 in VIBRANT Study
|
Treatment effects in evaluable subgroups (e.g., age, gender, and baseline retinal perfusion status) in the study were in general consistent with the results in the overall populations.
14.2 Macular Edema Following Central Retinal Vein Occlusion (crvo) (14.2 Macular Edema Following Central Retinal Vein Occlusion (CRVO))
The safety and efficacy of aflibercept were assessed in two randomized, multi-center, double-masked, sham-controlled studies in patients with macular edema following CRVO. A total of 358 patients were treated and evaluable for efficacy (217 with aflibercept) in the two studies (COPERNICUS and GALILEO). In both studies, patients were randomly assigned in a 3:2 ratio to either 2 mg aflibercept administered every 4 weeks (2Q4), or sham injections (control group) administered every 4 weeks for a total of 6 injections. Protocol-specified visits occurred every 28±7 days. Patient ages ranged from 22 to 89 years with a mean of 64 years.
In both studies, the primary efficacy endpoint was the proportion of patients who gained at least 15 letters in BCVA compared to baseline. At week 24, the aflibercept 2 mg Q4 group was superior to the control group for the primary endpoint.
Results from the analysis of the COPERNICUS and GALILEO studies are shown in Table 5 and Figure 14 below.
| COPERNICUS | GALILEO | |||
|---|---|---|---|---|
| Control | Aflibercept 2 mg Q4 weeks |
Control | Aflibercept 2 mg Q4 weeks |
|
| N=73 | N=114 | N=68 | N=103 | |
| Efficacy Outcomes | ||||
| Proportion of patients who gained at least 15 letters in BCVA from Baseline (%) | 12% | 56% | 22% | 60% |
| Weighted Difference Difference is aflibercept 2 mg Q4 weeks minus Control
,
Difference and CI are calculated using Cochran-Mantel-Haenszel (CMH) test adjusted for baseline factors; 95.1% confidence intervals were presented to adjust for the multiple assessments conducted during the study (%) |
44.8% p<0.01 compared with Control
|
38.3% | ||
| (95.1% CI) | (32.9, 56.6) | (24.4, 52.1) | ||
| Mean change in BCVA as measured by ETDRS letter score from Baseline (SD) | -4.0 | 17.3 | 3.3 | 18.0 |
| (18.0) | (12.8) | (14.1) | (12.2) | |
| Difference in LS mean
,
LS mean and CI based on an ANCOVA model
|
21.7 | 14.7 | ||
| (95.1% CI) | (17.3, 26.1) | (10.7, 18.7) |
|
Figure 14. Mean Change in BCVA as Measured by ETDRS Letter Score from Baseline to Week 24 in COPERNICUS and GALILEO Studies
|
Treatment effects in evaluable subgroups (e.g., age, gender, race, baseline visual acuity, retinal perfusion status, and CRVO duration) in each study and in the combined analysis were in general consistent with the results in the overall populations.
5.1 Endophthalmitis, Retinal Detachments, and Retinal Vasculitis With Or Without Occlusion (5.1 Endophthalmitis, Retinal Detachments, and Retinal Vasculitis with or without Occlusion)
Intravitreal injections, including those with aflibercept products, have been associated with endophthalmitis and retinal detachments [see Adverse Reactions (6.1)] and, more rarely, retinal vasculitis with or without occlusion [see Adverse Reactions (6.2)]. Proper aseptic injection technique must always be used when administering PAVBLU. Patients and/or caregivers should be instructed to report any signs and/or symptoms suggestive of endophthalmitis, retinal detachment, or retinal vasculitis without delay and should be managed appropriately [see Dosage and Administration (2.8) and Patient Counseling Information (17)].
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:46:03.405942 · Updated: 2026-03-14T22:23:51.806226