Oxybutynin Chloride Extended-release Tablets
c5950dba-d92b-46a0-993f-af9f9ddb52bf
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Indications and Usage
Oxybutynin chloride extended-release tablets are a muscarinic antagonist indicated for the treatment of overactive bladder with symptoms of urge urinary incontinence, urgency, and frequency. Oxybutynin chloride extended-release tablets are also indicated for the treatment of pediatric patients aged 6 years and older with symptoms of detrusor overactivity associated with a neurological condition (e.g., spina bifida).
Dosage and Administration
Oxybutynin chloride extended-release tablets must be swallowed whole with the aid of liquids, and must not be chewed, divided, or crushed. Oxybutynin chloride extended-release tablets may be administered with or without food.
Contraindications
Oxybutynin chloride extended-release tablets are contraindicated in patients with urinary retention, gastric retention and other severe decreased gastrointestinal motility conditions, uncontrolled narrow-angle glaucoma. Oxybutynin chloride extended-release tablets are also contraindicated in patients who have demonstrated hypersensitivity to the drug substance or other components of the product. There have been reports of hypersensitivity reactions, including anaphylaxis and angioedema.
Warnings and Precautions
Angioedema: Angioedema has been reported with oxybutynin. If symptoms of angioedema occur, discontinue Oxybutynin chloride extended-release tablets immediately and initiate appropriate therapy. ( 5.1 ) Central Nervous System (CNS) effects: CNS effects have been reported with oxybutynin. If patient experiences anticholinergic CNS effects, consider dose adjustment or discontinuation of Oxybutynin chloride extended-release tablets. ( 5.2 ) Use with caution due to aggravation of symptoms: Pre-existing dementia in patients treated with cholinesterase inhibitors (5.2), Parkinson's disease (5.2), Myasthenia gravis (5.3), and Decreased gastrointestinal motility in patients with autonomic neuropathy. (5.4) Urinary Retention: Use with caution in patients with clinically significant bladder outflow obstruction because of the risk of urinary retention (5.5) Gastrointestinal Adverse Reactions: Use with caution in patients with gastrointestinal obstructive disorders or decreased intestinal motility due to risk of gastric retention. Use with caution in patients with gastroesophageal reflux or in patients concurrently taking drugs that can exacerbate esophagitis. (5.6)
Adverse Reactions
Oxybutynin chloride extended-release tablets should be administered with caution to patients with gastrointestinal obstructive disorders because of the risk of gastric retention [see Contraindications (4)] . Oxybutynin chloride extended-release tablets, like other anticholinergic drugs, may decrease gastrointestinal motility and should be used with caution in patients with conditions such as ulcerative colitis and intestinal atony. Oxybutynin chloride extended-release tablets should be used with caution in patients who have gastroesophageal reflux and/or who are concurrently taking drugs (such as bisphosphonates) that can cause or exacerbate esophagitis. As with any other nondeformable material, caution should be used when administering Oxybutynin chloride extended-release tablets to patients with preexisting severe gastrointestinal narrowing (pathologic or iatrogenic). There have been rare reports of obstructive symptoms in patients with known strictures in association with the ingestion of other drugs in nondeformable controlled-release formulations.
Drug Interactions
The concomitant use of oxybutynin with other anticholinergic drugs or with other agents which produce dry mouth, constipation, somnolence (drowsiness), and/or other anticholinergic-like effects may increase the frequency and/or severity of such effects. Anticholinergic agents may potentially alter the absorption of some concomitantly administered drugs due to anticholinergic effects on gastrointestinal motility. This may be of concern for drugs with a narrow therapeutic index. Anticholinergic agents may also antagonize the effects of prokinetic agents, such as metoclopramide. Mean oxybutynin plasma concentrations were approximately 2 fold higher when Oxybutynin chloride extended-release tablets were administered with ketoconazole, a potent CYP3A4 inhibitor. Other inhibitors of the cytochrome P450 3A4 enzyme system, such as antimycotic agents (e.g., itraconazole and miconazole) or macrolide antibiotics (e.g., erythromycin and clarithromycin), may alter oxybutynin mean pharmacokinetic parameters (i.e., C max and AUC). The clinical relevance of such potential interactions is not known. Caution should be used when such drugs are co-administered.
How Supplied
Oxybutynin chloride extended-release tablets, 5 mg are round, biconvex, white coated tablets imprinted in black ink with "270" on one side and "KU" on the other side. They are supplied as follows: Bottles of 100 Tablets NDC 62175-270-37 Bottles of 500 Tablets NDC 62175-270-41 Oxybutynin chloride extended-release tablets, 10 mg are round, biconvex, white coated tablets imprinted in black ink with "271" on one side and "KU" on the other side. They are supplied as follows: Bottles of 100 Tablets NDC 62175-271-37 Bottles of 500 Tablets NDC 62175-271-41 Oxybutynin chloride extended-release tablets, 15 mg are round, biconvex, white coated tablets imprinted in black ink with "272" on one side and "KU" on the other side. They are supplied as follows: Bottles of 100 Tablets NDC 62175-272-37 Bottles of 500 Tablets NDC 62175-272-41 Storage Store at 25°C (77°F); excursions permitted to 15–30°C (59–86°F) [see USP Controlled Room Temperature]. Protect from moisture and humidity. Keep out of reach of children.
Storage and Handling
Oxybutynin chloride extended-release tablets, 5 mg are round, biconvex, white coated tablets imprinted in black ink with "270" on one side and "KU" on the other side. They are supplied as follows: Bottles of 100 Tablets NDC 62175-270-37 Bottles of 500 Tablets NDC 62175-270-41 Oxybutynin chloride extended-release tablets, 10 mg are round, biconvex, white coated tablets imprinted in black ink with "271" on one side and "KU" on the other side. They are supplied as follows: Bottles of 100 Tablets NDC 62175-271-37 Bottles of 500 Tablets NDC 62175-271-41 Oxybutynin chloride extended-release tablets, 15 mg are round, biconvex, white coated tablets imprinted in black ink with "272" on one side and "KU" on the other side. They are supplied as follows: Bottles of 100 Tablets NDC 62175-272-37 Bottles of 500 Tablets NDC 62175-272-41 Storage Store at 25°C (77°F); excursions permitted to 15–30°C (59–86°F) [see USP Controlled Room Temperature]. Protect from moisture and humidity. Keep out of reach of children.
Description
Oxybutynin chloride extended-release tablets are a muscarinic antagonist indicated for the treatment of overactive bladder with symptoms of urge urinary incontinence, urgency, and frequency. Oxybutynin chloride extended-release tablets are also indicated for the treatment of pediatric patients aged 6 years and older with symptoms of detrusor overactivity associated with a neurological condition (e.g., spina bifida).
Medication Information
Warnings and Precautions
Angioedema: Angioedema has been reported with oxybutynin. If symptoms of angioedema occur, discontinue Oxybutynin chloride extended-release tablets immediately and initiate appropriate therapy. ( 5.1 ) Central Nervous System (CNS) effects: CNS effects have been reported with oxybutynin. If patient experiences anticholinergic CNS effects, consider dose adjustment or discontinuation of Oxybutynin chloride extended-release tablets. ( 5.2 ) Use with caution due to aggravation of symptoms: Pre-existing dementia in patients treated with cholinesterase inhibitors (5.2), Parkinson's disease (5.2), Myasthenia gravis (5.3), and Decreased gastrointestinal motility in patients with autonomic neuropathy. (5.4) Urinary Retention: Use with caution in patients with clinically significant bladder outflow obstruction because of the risk of urinary retention (5.5) Gastrointestinal Adverse Reactions: Use with caution in patients with gastrointestinal obstructive disorders or decreased intestinal motility due to risk of gastric retention. Use with caution in patients with gastroesophageal reflux or in patients concurrently taking drugs that can exacerbate esophagitis. (5.6)
Indications and Usage
Oxybutynin chloride extended-release tablets are a muscarinic antagonist indicated for the treatment of overactive bladder with symptoms of urge urinary incontinence, urgency, and frequency. Oxybutynin chloride extended-release tablets are also indicated for the treatment of pediatric patients aged 6 years and older with symptoms of detrusor overactivity associated with a neurological condition (e.g., spina bifida).
Dosage and Administration
Oxybutynin chloride extended-release tablets must be swallowed whole with the aid of liquids, and must not be chewed, divided, or crushed. Oxybutynin chloride extended-release tablets may be administered with or without food.
Contraindications
Oxybutynin chloride extended-release tablets are contraindicated in patients with urinary retention, gastric retention and other severe decreased gastrointestinal motility conditions, uncontrolled narrow-angle glaucoma. Oxybutynin chloride extended-release tablets are also contraindicated in patients who have demonstrated hypersensitivity to the drug substance or other components of the product. There have been reports of hypersensitivity reactions, including anaphylaxis and angioedema.
Adverse Reactions
Oxybutynin chloride extended-release tablets should be administered with caution to patients with gastrointestinal obstructive disorders because of the risk of gastric retention [see Contraindications (4)] . Oxybutynin chloride extended-release tablets, like other anticholinergic drugs, may decrease gastrointestinal motility and should be used with caution in patients with conditions such as ulcerative colitis and intestinal atony. Oxybutynin chloride extended-release tablets should be used with caution in patients who have gastroesophageal reflux and/or who are concurrently taking drugs (such as bisphosphonates) that can cause or exacerbate esophagitis. As with any other nondeformable material, caution should be used when administering Oxybutynin chloride extended-release tablets to patients with preexisting severe gastrointestinal narrowing (pathologic or iatrogenic). There have been rare reports of obstructive symptoms in patients with known strictures in association with the ingestion of other drugs in nondeformable controlled-release formulations.
Drug Interactions
The concomitant use of oxybutynin with other anticholinergic drugs or with other agents which produce dry mouth, constipation, somnolence (drowsiness), and/or other anticholinergic-like effects may increase the frequency and/or severity of such effects. Anticholinergic agents may potentially alter the absorption of some concomitantly administered drugs due to anticholinergic effects on gastrointestinal motility. This may be of concern for drugs with a narrow therapeutic index. Anticholinergic agents may also antagonize the effects of prokinetic agents, such as metoclopramide. Mean oxybutynin plasma concentrations were approximately 2 fold higher when Oxybutynin chloride extended-release tablets were administered with ketoconazole, a potent CYP3A4 inhibitor. Other inhibitors of the cytochrome P450 3A4 enzyme system, such as antimycotic agents (e.g., itraconazole and miconazole) or macrolide antibiotics (e.g., erythromycin and clarithromycin), may alter oxybutynin mean pharmacokinetic parameters (i.e., C max and AUC). The clinical relevance of such potential interactions is not known. Caution should be used when such drugs are co-administered.
Storage and Handling
Oxybutynin chloride extended-release tablets, 5 mg are round, biconvex, white coated tablets imprinted in black ink with "270" on one side and "KU" on the other side. They are supplied as follows: Bottles of 100 Tablets NDC 62175-270-37 Bottles of 500 Tablets NDC 62175-270-41 Oxybutynin chloride extended-release tablets, 10 mg are round, biconvex, white coated tablets imprinted in black ink with "271" on one side and "KU" on the other side. They are supplied as follows: Bottles of 100 Tablets NDC 62175-271-37 Bottles of 500 Tablets NDC 62175-271-41 Oxybutynin chloride extended-release tablets, 15 mg are round, biconvex, white coated tablets imprinted in black ink with "272" on one side and "KU" on the other side. They are supplied as follows: Bottles of 100 Tablets NDC 62175-272-37 Bottles of 500 Tablets NDC 62175-272-41 Storage Store at 25°C (77°F); excursions permitted to 15–30°C (59–86°F) [see USP Controlled Room Temperature]. Protect from moisture and humidity. Keep out of reach of children.
How Supplied
Oxybutynin chloride extended-release tablets, 5 mg are round, biconvex, white coated tablets imprinted in black ink with "270" on one side and "KU" on the other side. They are supplied as follows: Bottles of 100 Tablets NDC 62175-270-37 Bottles of 500 Tablets NDC 62175-270-41 Oxybutynin chloride extended-release tablets, 10 mg are round, biconvex, white coated tablets imprinted in black ink with "271" on one side and "KU" on the other side. They are supplied as follows: Bottles of 100 Tablets NDC 62175-271-37 Bottles of 500 Tablets NDC 62175-271-41 Oxybutynin chloride extended-release tablets, 15 mg are round, biconvex, white coated tablets imprinted in black ink with "272" on one side and "KU" on the other side. They are supplied as follows: Bottles of 100 Tablets NDC 62175-272-37 Bottles of 500 Tablets NDC 62175-272-41 Storage Store at 25°C (77°F); excursions permitted to 15–30°C (59–86°F) [see USP Controlled Room Temperature]. Protect from moisture and humidity. Keep out of reach of children.
Description
Oxybutynin chloride extended-release tablets are a muscarinic antagonist indicated for the treatment of overactive bladder with symptoms of urge urinary incontinence, urgency, and frequency. Oxybutynin chloride extended-release tablets are also indicated for the treatment of pediatric patients aged 6 years and older with symptoms of detrusor overactivity associated with a neurological condition (e.g., spina bifida).
Section 42229-5
System Components and Performance
Oxybutynin chloride extended-release tablets use osmotic pressure to deliver oxybutynin chloride at a controlled rate over approximately 24 hours. The system, which resembles a conventional tablet in appearance, comprises an osmotically active core surrounded by a semipermeable membrane. The unitary tablet core is composed of the drug and excipients (including the osmotically active components). There is a precision-laser drilled orifice in the semipermeable membrane on the side of the tablet. In an aqueous environment, such as the gastrointestinal tract, water permeates through the membrane into the tablet core, causing the drug to go into suspension and the osmotic components to expand. This expansion pushes the drug out through the orifice. The semipermeable membrane controls the rate at which water permeates into the tablet core, which in turn controls the rate of drug delivery. The controlled rate of drug delivery into the gastrointestinal lumen is thus independent of pH or gastrointestinal motility. The function of Oxybutynin chloride extended-release tablets depends on the existence of an osmotic gradient between the contents of the core and the fluid in the gastrointestinal tract. Since the osmotic gradient remains constant, drug delivery remains essentially constant. The biologically inert components of the tablet remain intact during gastrointestinal transit and are eliminated in the feces as an insoluble shell.
USP Dissolution Test 3.
2.1 Adults
The recommended starting dose of Oxybutynin chloride extended-release tablets is 5 or 10 mg once daily at approximately the same time each day. Dosage may be adjusted in 5-mg increments to achieve a balance of efficacy and tolerability (up to a maximum of 30 mg/day). In general, dosage adjustment may proceed at approximately weekly intervals.
10 Overdosage
The continuous release of oxybutynin from Oxybutynin chloride extended-release tablets should be considered in the treatment of overdosage. Patients should be monitored for at least 24 hours. Treatment should be symptomatic and supportive. A cathartic may be administered.
Overdosage with oxybutynin chloride has been associated with anticholinergic effects including central nervous system excitation, flushing, fever, dehydration, cardiac arrhythmia, vomiting, and urinary retention.
Ingestion of 100 mg oxybutynin chloride in association with alcohol has been reported in a 13-year-old boy who experienced memory loss, and a 34-year-old woman who developed stupor, followed by disorientation and agitation on awakening, dilated pupils, dry skin, cardiac arrhythmia, and retention of urine. Both patients fully recovered with symptomatic treatment.
8.1 Pregnancy
Risk Summary
There are no adequate data on Oxybutynin chloride extended-release tablets use in pregnant women to evaluate for a drug-associated risk of major birth defects, miscarriage or adverse maternal or fetal outcomes.In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2-4% and 15-20%, respectively. The background risk of major birth defects and miscarriage for the indicated population is unknown.
8.2 Lactation
Risk Summary
There are no data on the presence of oxybutynin in human milk, the effects on the breastfed infant, or the effects of Oxybutynin chloride extended-release tablets on milk production. The developmental and health benefits of breastfeeding should be considered along with the mother’s clinical need for Oxybutynin chloride extended-release tablets and any potential adverse effects on the breastfed child from Oxybutynin chloride extended-release tablets or from the underlying maternal condition.
11 Description
Oxybutynin chloride extended-release tablets are an antispasmodic, muscarinic antagonist. Each Oxybutynin chloride extended-release tablet contains 5 mg, 10 mg, or 15 mg of oxybutynin chloride USP, formulated as a once-a-day controlled-release tablet for oral administration. Oxybutynin chloride is administered as a racemate of R- and S-enantiomers.
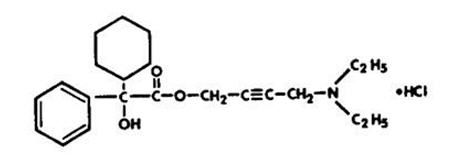
Chemically, oxybutynin chloride is d,l (racemic) 4-diethylamino-2-butynyl phenylcyclohexylglycolate hydrochloride. The empirical formula of oxybutynin chloride is C22H31NO3•HCl.
Its structural formula is:
Oxybutynin chloride is a white crystalline solid with a molecular weight of 393.9. It is readily soluble in water and acids, but relatively insoluble in alkalis.
Oxybutynin chloride extended-release tablets also contain the following inert ingredients: black iron oxide, cellulose acetate, colloidal silicon dioxide, dextrose, hypromellose, lactose, magnesium stearate, mannitol, polyethylene glycol, propylene glycol, tartaric acid, titanium dioxide, triacetin.
5.1 Angioedema
Angioedema of the face, lips, tongue and/or larynx has been reported with oxybutynin. In some cases, angioedema occurred after the first dose. Angioedema associated with upper airway swelling may be life-threatening. If involvement of the tongue, hypopharynx, or larynx occurs, oxybutynin should be promptly discontinued and appropriate therapy and/or measures necessary to ensure a patent airway should be promptly provided.
8.4 Pediatric Use
The safety and efficacy of Oxybutynin chloride extended-release tablets were studied in 60 children in a 24-week, open-label, non-randomized trial. Patients were aged 6 to 15 years, all had symptoms of detrusor overactivity in association with a neurological condition (e.g., spina bifida), all used clean intermittent catheterization, and all were current users of oxybutynin chloride. Study results demonstrated that administration of Oxybutynin chloride extended-release tablets 5 to 20 mg/day was associated with an increase from baseline in mean urine volume per catheterization from 108 mL to 136 mL, an increase from baseline in mean urine volume after morning awakening from 148 mL to 189 mL, and an increase from baseline in the mean percentage of catheterizations without a leaking episode from 34% to 51%.
Urodynamic results were consistent with clinical results. Administration of Oxybutynin chloride extended-release tablets resulted in an increase from baseline in mean maximum cystometric capacity from 185 mL to 254 mL, a decrease from baseline in mean detrusor pressure at maximum cystometric capacity from 44 cm H2O to 33 cm H2O, and a reduction in the percentage of patients demonstrating uninhibited detrusor contractions (of at least 15 cm H2O) from 60% to 28%.
The pharmacokinetics of Oxybutynin chloride extended-release tablets in these patients were consistent with those reported for adults [see Clinical Pharmacology (12.3)] .
Oxybutynin chloride extended-release tablets are not recommended in pediatric patients who cannot swallow the tablet whole without chewing, dividing, or crushing, or in children under the age of 6.
8.5 Geriatric Use
The rate and severity of anticholinergic effects reported by patients less than 65 years old and those 65 years and older were similar. The pharmacokinetics of Oxybutynin chloride extended-release tablets were similar in all patients studied (up to 78 years of age).
14 Clinical Studies
Oxybutynin chloride extended-release tablets were evaluated for the treatment of patients with overactive bladder with symptoms of urge urinary incontinence, urgency, and frequency in three controlled efficacy studies. The majority of patients were Caucasian (89.0%) and female (91.9%) with a mean age of 59 years (range, 18 to 98 years). Entry criteria required that patients have urge or mixed incontinence (with a predominance of urge) as evidenced by ≥ 6 urge incontinence episodes per week and ≥ 10 micturitions per day. Study 1 was a fixed-dose escalation design, whereas the other two studies used a dose-adjustment design in which each patient's final dose was adjusted to a balance between improvement of incontinence symptoms and tolerability of side effects. All three studies included patients known to be responsive to oxybutynin or other anticholinergic medications, and these patients were maintained on a final dose for up to 2 weeks.
The efficacy results for the three controlled trials are presented in the following Tables 4, 5, and 6 and Figures 3, 4, and 5.
Table 4: Number of Urge Urinary Incontinence Episodes Per Week
| Study 1 | n | Oxybutynin chloride extended-release tablets | n | Placebo |
|---|---|---|---|---|
| Mean Baseline | 34 | 15.9 | 16 | 20.9 |
| Mean (SD) Change from Baseline† | 34 | -15.8 (8.9) | 16 | -7.6 (8.6) |
| 95% Confidence Interval for Difference | (-13.6, -2.8)* | |||
| (Oxybutynin chloride extended-release tablets- Placebo) |
* The difference between Oxybutynin chloride extended-release tablets and placebo was statistically significant.
† Covariate adjusted mean with missing observations set to baseline values
Figure 3: Mean Change (±SD) in Urge Incontinence Episodes Per Week from Baseline (Study 1)
*The difference between Oxybutynin chloride extended-release tablets and placebo was statistically significant.
Table 5: Number of Urge Urinary Incontinence Episodes Per Week (Study 2)
| Study 2 | n | Oxybutynin chloride extended-release tablets | n | oxybutynin |
|---|---|---|---|---|
| Mean Baseline | 53 | 27.6 | 52 | 23.0 |
| Mean (SD) Change from Baseline† | 53 | -17.6 (11.9) | 52 | -19.4 (11.9) |
| 95% Confidence Interval for Difference | (-2.8, 6.5) | |||
| (Oxybutynin chloride extended-release tablets- oxybutynin) |
† Covariate adjusted mean with missing observations set to baseline values
Figure 4: Mean Change (±SD) in Urge Urinary Incontinence Episodes Per Week from Baseline (Study 2)
Table 6: Number of Urge Urinary Incontinence Episodes Per Week (Study 3)
| Study 3 | n | Oxybutynin chloride extended-release tablets | n | oxybutynin |
|---|---|---|---|---|
| Mean Baseline | 111 | 18.9 | 115 | 19.5 |
| Mean (SD) Change from Baseline† | 111 | -14.5 (8.7) | 115 | -13.8 (8.6) |
| 95% Confidence Interval for Difference | (-3.0, 1.6)** | |||
| (Oxybutynin chloride extended-release tablets- oxybutynin) |
** The difference between Oxybutynin chloride extended-release tablets and oxybutynin fulfilled the criteria for comparable efficacy.
† Covariate adjusted mean with missing observations set to baseline values
Figure 5: Mean Change (±SD) in Urge Urinary Incontinence Episodes Per Week from Baseline (Study 3)
** The difference between Oxybutynin chloride extended-release tablets and oxybutynin fulfilled the criteria for comparable efficacy.
4 Contraindications
Oxybutynin chloride extended-release tablets are contraindicated in patients with urinary retention, gastric retention and other severe decreased gastrointestinal motility conditions, uncontrolled narrow-angle glaucoma.
Oxybutynin chloride extended-release tablets are also contraindicated in patients who have demonstrated hypersensitivity to the drug substance or other components of the product. There have been reports of hypersensitivity reactions, including anaphylaxis and angioedema.
6 Adverse Reactions
The most common (incidence ≥5%) adverse reactions were dry mouth, constipation, diarrhea, headache, somnolence, and dizziness. (6)
To report SUSPECTED ADVERSE REACTIONS, contact Lannett Company, Inc. at 1-844-834-0530 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
7 Drug Interactions
The concomitant use of oxybutynin with other anticholinergic drugs or with other agents which produce dry mouth, constipation, somnolence (drowsiness), and/or other anticholinergic-like effects may increase the frequency and/or severity of such effects.
Anticholinergic agents may potentially alter the absorption of some concomitantly administered drugs due to anticholinergic effects on gastrointestinal motility. This may be of concern for drugs with a narrow therapeutic index. Anticholinergic agents may also antagonize the effects of prokinetic agents, such as metoclopramide.
Mean oxybutynin plasma concentrations were approximately 2 fold higher when Oxybutynin chloride extended-release tablets were administered with ketoconazole, a potent CYP3A4 inhibitor. Other inhibitors of the cytochrome P450 3A4 enzyme system, such as antimycotic agents (e.g., itraconazole and miconazole) or macrolide antibiotics (e.g., erythromycin and clarithromycin), may alter oxybutynin mean pharmacokinetic parameters (i.e., Cmax and AUC). The clinical relevance of such potential interactions is not known. Caution should be used when such drugs are co-administered.
8.6 Renal Impairment
There were no studies conducted with Oxybutynin chloride extended-release tablets in patients with renal impairment.
12.2 Pharmacodynamics
In patients with conditions characterized by involuntary bladder contractions, cystometric studies have demonstrated that oxybutynin increases bladder (vesical) capacity, diminishes the frequency of uninhibited contractions of the detrusor muscle, and delays the initial desire to void.
5.5 Urinary Retention
Oxybutynin chloride extended-release tablets should be administered with caution to patients with clinically significant bladder outflow obstruction because of the risk of urinary retention [see Contraindications (4)].
8.7 Hepatic Impairment
There were no studies conducted with Oxybutynin chloride extended-release tablets in patients with hepatic impairment.
1 Indications and Usage
Oxybutynin chloride extended-release tablets are a muscarinic antagonist indicated for the treatment of overactive bladder with symptoms of urge urinary incontinence, urgency, and frequency.
Oxybutynin chloride extended-release tablets are also indicated for the treatment of pediatric patients aged 6 years and older with symptoms of detrusor overactivity associated with a neurological condition (e.g., spina bifida).
12.1 Mechanism of Action
Oxybutynin relaxes bladder smooth muscle. Oxybutynin chloride exerts a direct antispasmodic effect on smooth muscle and inhibits the muscarinic action of acetylcholine on smooth muscle. No blocking effects occur at skeletal neuromuscular junctions or autonomic ganglia (antinicotinic effects).
Antimuscarinic activity resides predominantly in the R-isomer. A metabolite, desethyloxybutynin, has pharmacological activity similar to that of oxybutynin in in vitro studies.
5 Warnings and Precautions
- Angioedema: Angioedema has been reported with oxybutynin. If symptoms of angioedema occur, discontinue Oxybutynin chloride extended-release tablets immediately and initiate appropriate therapy. (5.1)
- Central Nervous System (CNS) effects: CNS effects have been reported with oxybutynin. If patient experiences anticholinergic CNS effects, consider dose adjustment or discontinuation of Oxybutynin chloride extended-release tablets. (5.2)
- Use with caution due to aggravation of symptoms:
- Pre-existing dementia in patients treated with cholinesterase inhibitors (5.2),
- Parkinson's disease (5.2),
- Myasthenia gravis (5.3), and
- Decreased gastrointestinal motility in patients with autonomic neuropathy. (5.4)
- Urinary Retention: Use with caution in patients with clinically significant bladder outflow obstruction because of the risk of urinary retention (5.5)
- Gastrointestinal Adverse Reactions: Use with caution in patients with gastrointestinal obstructive disorders or decreased intestinal motility due to risk of gastric retention. Use with caution in patients with gastroesophageal reflux or in patients concurrently taking drugs that can exacerbate esophagitis. (5.6)
2 Dosage and Administration
Oxybutynin chloride extended-release tablets must be swallowed whole with the aid of liquids, and must not be chewed, divided, or crushed.
Oxybutynin chloride extended-release tablets may be administered with or without food.
3 Dosage Forms and Strengths
Oxybutynin chloride extended-release tablets are available as 5, 10 and 15 mg tablets for oral use:
5 mg: White, round, biconvex tablet with "270" printed on one side and "KU" printed on the other side with black ink.
10 mg: White, round, biconvex tablet with "271" printed on one side and "KU" printed on the other side with black ink.
15 mg: White, round, biconvex tablet with "272" printed on one side and "KU" printed on the other side with black ink.
6.2 Postmarketing Experience
The following additional adverse reactions have been reported from worldwide postmarketing experience with Oxybutynin chloride extended-release tablets. Because postmarketing reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Infections and Infestations: Urinary tract infection; Psychiatric Disorders: psychotic disorder, agitation, confusional state, hallucinations, memory impairment, abnormal behavior; Nervous System Disorders: convulsions; Eye Disorders: glaucoma; Respiratory, Thoracic and Mediastinal Disorders: nasal congestion; Cardiac Disorders: arrhythmia, tachycardia, palpitations, QT interval prolongation; Vascular Disorders: flushing, hypertension; Skin and Subcutaneous Tissue Disorders: rash; Renal and Urinary Disorders: impotence; General Disorders and Administration Site Conditions: hypersensitivity reactions, including angioedema with airway obstruction, urticaria, and face edema; anaphylactic reactions requiring hospitalization for emergency treatment; Injury, poisoning and procedural complications: fall.
Additional adverse events reported with some other oxybutynin chloride formulations include: cycloplegia, mydriasis, and suppression of lactation. In one reported case, concomitant use of oxybutynin with carbamazepine and dantrolene was associated with adverse events of vomiting, drowsiness, confusion, unsteadiness, slurred speech and nystagmus, suggestive of carbamazepine toxicity.
8 Use in Specific Populations
- Pediatric Use: Oxybutynin chloride extended-release tablets are not recommended in pediatric patients who cannot swallow the tablet whole without chewing, dividing or crushing, or in children under the age of 6 years. (8.4)
- Renal or Hepatic Impairment: There have been no studies conducted in patients with renal or hepatic impairment. (8.6, 8.7)
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, the adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
The safety and efficacy of Oxybutynin chloride extended-release tablets (5 to 30 mg/day) was evaluated in 774 adult subjects who participated in five double-blind, controlled clinical trials. In four of the five studies, Oxybutynin chloride IR (5 to 20 mg/day in 199 subjects) was an active comparator. Adverse reactions reported by ≥ 1% of subjects are shown in Table 1.
| System/Organ Class Preferred Term |
Oxybutynin chloride extended-release tablets 5 to 30 mg/day n = 774 % |
Oxybutynin chloride IR IR = immediate release
5 to 20 mg/day n = 199 % |
|---|---|---|
| Psychiatric Disorders | ||
| Insomnia | 3.0 | 5.5 |
| Nervous System Disorders | ||
| Headache | 7.5 | 8.0 |
| Somnolence | 5.6 | 14.1 |
| Dizziness | 5.0 | 16.6 |
| Dysgeusia | 1.6 | 1.5 |
| Eye Disorders | ||
| Vision blurred | 4.3 | 9.6 |
| Dry eye | 3.1 | 2.5 |
| Respiratory, Thoracic and Mediastinal Disorders | ||
| Cough | 1.9 | 3.0 |
| Oropharyngeal pain | 1.9 | 1.5 |
| Dry throat | 1.7 | 2.5 |
| Nasal dryness | 1.7 | 4.5 |
| Gastrointestinal Disorders | ||
| Dry mouth | 34.9 | 72.4 |
| Constipation | 8.7 | 15.1 |
| Diarrhea | 7.9 | 6.5 |
| Dyspepsia | 4.5 | 6.0 |
| Nausea | 4.5 | 11.6 |
| Abdominal pain | 1.6 | 2.0 |
| Vomiting | 1.3 | 1.5 |
| Flatulence | 1.2 | 2.5 |
| Gastro-esophageal reflux disease | 1.0 | 0.5 |
| Skin and Subcutaneous Tissue Disorders | ||
| Dry skin | 1.8 | 2.5 |
| Pruritus | 1.3 | 1.5 |
| Renal and Urinary Disorders | ||
| Dysuria | 1.9 | 2.0 |
| Urinary hesitation | 1.9 | 8.5 |
| Urinary retention | 1.2 | 3.0 |
| General Disorders and Administration Site Conditions | ||
| Fatigue | 2.6 | 3.0 |
| Investigations | ||
| Residual urine volume The bundled term residual urine volume consists of the preferred terms residual urine volume and residual urine volume increased.
|
2.3 | 3.5 |
The discontinuation rate due to adverse reactions was 4.4% with Oxybutynin chloride extended-release tablets compared to 0% with Oxybutynin chloride IR. The most frequent adverse reaction causing discontinuation of study medication was dry mouth (0.7%).
The following adverse reactions were reported by <1% of Oxybutynin chloride extended-release tablets-treated patients and at a higher incidence than placebo in clinical trials: Metabolism and Nutrition Disorders: anorexia, fluid retention; Vascular disorders: hot flush; Respiratory, thoracic and mediastinal disorders: dysphonia; Gastrointestinal Disorders: dysphagia, frequent bowel movements; General disorders and administration site conditions: chest discomfort, thirst.
17 Patient Counseling Information
- Patients should be informed that oxybutynin may produce angioedema that could result in life threatening airway obstruction. Patients should be advised to promptly discontinue oxybutynin therapy and seek immediate medical attention if they experience swelling of the tongue, edema of the laryngopharynx, or difficulty breathing.
- Patients should be informed that anticholinergic (antimuscarinic) agents such as Oxybutynin chloride extended-release tablets, may produce clinically significant adverse reactions related to anticholinergic activity such as:
- Urinary retention and constipation
- Heat prostration due to decreased sweating. Heat prostration can occur when anticholinergic medicines are administered in the presence of high environmental temperature.
- Patients should be informed that anticholinergic medicines such as Oxybutynin chloride extended-release tablets may produce drowsiness (somnolence), dizziness or blurred vision. Patients should be advised to exercise caution in decisions to engage in potentially dangerous activities until Oxybutynin chloride extended-release tablets effects have been determined.
- Patients should be informed that alcohol may enhance the drowsiness caused by anticholinergic agents such as Oxybutynin chloride extended-release tablets.
- Patients should be informed that Oxybutynin chloride extended-release tablets should be swallowed whole with the aid of liquids. Patients should not chew, divide, or crush tablets. The medication is contained within a nonabsorbable shell designed to release the drug at a controlled rate. The tablet shell is eliminated from the body; patients should not be concerned if they occasionally notice in their stool something that looks like a tablet.
- Oxybutynin chloride extended-release tablets should be taken at approximately the same time each day.
For more information call 1-844-834-0530.
5.2 Central Nervous System Effects
Oxybutynin is associated with anticholinergic central nervous system (CNS) effects [see Adverse Reactions (6)] . A variety of CNS anticholinergic effects have been reported, including hallucinations, agitation, confusion and somnolence. Patients should be monitored for signs of anticholinergic CNS effects, particularly in the first few months after beginning treatment or increasing the dose. Advise patients not to drive or operate heavy machinery until they know how Oxybutynin chloride extended-release tablets affect them. If a patient experiences anticholinergic CNS effects, dose reduction or drug discontinuation should be considered.
Oxybutynin chloride extended-release tablets should be used with caution in patients with preexisting dementia treated with cholinesterase inhibitors due to the risk of aggravation of symptoms.
Oxybutynin chloride extended-release tablets should be used with caution in patients with Parkinson's disease due to the risk of aggravation of symptoms.
16 How Supplied/storage and Handling
Oxybutynin chloride extended-release tablets, 5 mg are round, biconvex, white coated tablets imprinted in black ink with "270" on one side and "KU" on the other side.
They are supplied as follows:
Bottles of 100 Tablets NDC 62175-270-37
Bottles of 500 Tablets NDC 62175-270-41
Oxybutynin chloride extended-release tablets, 10 mg are round, biconvex, white coated tablets imprinted in black ink with "271" on one side and "KU" on the other side.
They are supplied as follows:
Bottles of 100 Tablets NDC 62175-271-37
Bottles of 500 Tablets NDC 62175-271-41
Oxybutynin chloride extended-release tablets, 15 mg are round, biconvex, white coated tablets imprinted in black ink with "272" on one side and "KU" on the other side.
They are supplied as follows:
Bottles of 100 Tablets NDC 62175-272-37
Bottles of 500 Tablets NDC 62175-272-41
Storage
Store at 25°C (77°F); excursions permitted to 15–30°C (59–86°F) [see USP Controlled Room Temperature]. Protect from moisture and humidity.
Keep out of reach of children.
5.6 Gastrointestinal Adverse Reactions
Oxybutynin chloride extended-release tablets should be administered with caution to patients with gastrointestinal obstructive disorders because of the risk of gastric retention [see Contraindications (4)] .
Oxybutynin chloride extended-release tablets, like other anticholinergic drugs, may decrease gastrointestinal motility and should be used with caution in patients with conditions such as ulcerative colitis and intestinal atony.
Oxybutynin chloride extended-release tablets should be used with caution in patients who have gastroesophageal reflux and/or who are concurrently taking drugs (such as bisphosphonates) that can cause or exacerbate esophagitis.
As with any other nondeformable material, caution should be used when administering Oxybutynin chloride extended-release tablets to patients with preexisting severe gastrointestinal narrowing (pathologic or iatrogenic). There have been rare reports of obstructive symptoms in patients with known strictures in association with the ingestion of other drugs in nondeformable controlled-release formulations.
5.3 Worsening of Symptoms of Myasthenia Gravis
Oxybutynin chloride extended-release tablets should be used with caution in patients with myasthenia gravis due to the risk of aggravation of symptoms.
2.2 Pediatric Patients Aged 6 Years of Age and Older
The recommended starting dose of Oxybutynin chloride extended-release tablets is 5 mg once daily at approximately the same time each day. Dosage may be adjusted in 5-mg increments to achieve a balance of efficacy and tolerability (up to a maximum of 20 mg/day).
Principal Display Panel 5 Mg 500 Tablet Bottle Label
NDC 62175-270-41
500 tablets
Oxybutynin chloride
Extended-release tablets
5 mg
Rx only
Principal Display Panel 10 Mg 500 Tablet Bottle Label
NDC 62175-271-41
500 tablets
Oxybutynin chloride
Extended-release tablets
10 mg
Rx only
Principal Display Panel 5 Mg 100 Tablet Bottle Label
NDC 62175-270-37
100 tablets
Oxybutynin chloride
Extended-release tablets
5 mg
Rx only
Principal Display Panel 10 Mg 100 Tablet Bottle Label
NDC 62175-271-37
100 tablets
Oxybutynin chloride
Extended-release tablets
10 mg
Rx only
Principal Display Panel 15 Mg 100 tablet Bottle Label
NDC 62175-272-37
100 tablets
Oxybutynin chloride
Extended-release tablets
15 mg
Rx only
Principal Display Panel 15 Mg 500 tablet Bottle Label
NDC 62175-272-41
500 tablets
Oxybutynin chloride
Extended-release tablets
15 mg
Rx only
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenesis
A 24-month study in rats at dosages of oxybutynin chloride of 20, 80, and 160 mg/kg/day showed no evidence of carcinogenicity. These doses are approximately 6, 25, and 50 times the maximum human exposure, based on a human equivalent dose taking into account normalization of body surface area.
Mutagenesis
Oxybutynin chloride showed no increase of mutagenic activity when tested in Schizosaccharomyces pompholiciformis, Saccharomyces cerevisiae, and Salmonella typhimurium test systems.
Impairment of Fertility
No impairment of fertility was seen in rats at dosages up to 75 mg/kg/day (24 times the MRHD on a mg/m2 basis) when administered for 2 weeks prior to mating in females and for 9 weeks prior to mating in males.
5.4 Worsening of Symptoms of Decreased Gastrointestinal Motility in Patients With Autonomic Neuropathy
Oxybutynin chloride extended-release tablets should be used with caution in patients with autonomic neuropathy due to the risk of aggravation of symptoms of decreased gastrointestinal motility.
Structured Label Content
Section 42229-5 (42229-5)
System Components and Performance
Oxybutynin chloride extended-release tablets use osmotic pressure to deliver oxybutynin chloride at a controlled rate over approximately 24 hours. The system, which resembles a conventional tablet in appearance, comprises an osmotically active core surrounded by a semipermeable membrane. The unitary tablet core is composed of the drug and excipients (including the osmotically active components). There is a precision-laser drilled orifice in the semipermeable membrane on the side of the tablet. In an aqueous environment, such as the gastrointestinal tract, water permeates through the membrane into the tablet core, causing the drug to go into suspension and the osmotic components to expand. This expansion pushes the drug out through the orifice. The semipermeable membrane controls the rate at which water permeates into the tablet core, which in turn controls the rate of drug delivery. The controlled rate of drug delivery into the gastrointestinal lumen is thus independent of pH or gastrointestinal motility. The function of Oxybutynin chloride extended-release tablets depends on the existence of an osmotic gradient between the contents of the core and the fluid in the gastrointestinal tract. Since the osmotic gradient remains constant, drug delivery remains essentially constant. The biologically inert components of the tablet remain intact during gastrointestinal transit and are eliminated in the feces as an insoluble shell.
USP Dissolution Test 3.
2.1 Adults
The recommended starting dose of Oxybutynin chloride extended-release tablets is 5 or 10 mg once daily at approximately the same time each day. Dosage may be adjusted in 5-mg increments to achieve a balance of efficacy and tolerability (up to a maximum of 30 mg/day). In general, dosage adjustment may proceed at approximately weekly intervals.
10 Overdosage (10 OVERDOSAGE)
The continuous release of oxybutynin from Oxybutynin chloride extended-release tablets should be considered in the treatment of overdosage. Patients should be monitored for at least 24 hours. Treatment should be symptomatic and supportive. A cathartic may be administered.
Overdosage with oxybutynin chloride has been associated with anticholinergic effects including central nervous system excitation, flushing, fever, dehydration, cardiac arrhythmia, vomiting, and urinary retention.
Ingestion of 100 mg oxybutynin chloride in association with alcohol has been reported in a 13-year-old boy who experienced memory loss, and a 34-year-old woman who developed stupor, followed by disorientation and agitation on awakening, dilated pupils, dry skin, cardiac arrhythmia, and retention of urine. Both patients fully recovered with symptomatic treatment.
8.1 Pregnancy
Risk Summary
There are no adequate data on Oxybutynin chloride extended-release tablets use in pregnant women to evaluate for a drug-associated risk of major birth defects, miscarriage or adverse maternal or fetal outcomes.In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2-4% and 15-20%, respectively. The background risk of major birth defects and miscarriage for the indicated population is unknown.
8.2 Lactation
Risk Summary
There are no data on the presence of oxybutynin in human milk, the effects on the breastfed infant, or the effects of Oxybutynin chloride extended-release tablets on milk production. The developmental and health benefits of breastfeeding should be considered along with the mother’s clinical need for Oxybutynin chloride extended-release tablets and any potential adverse effects on the breastfed child from Oxybutynin chloride extended-release tablets or from the underlying maternal condition.
11 Description (11 DESCRIPTION)
Oxybutynin chloride extended-release tablets are an antispasmodic, muscarinic antagonist. Each Oxybutynin chloride extended-release tablet contains 5 mg, 10 mg, or 15 mg of oxybutynin chloride USP, formulated as a once-a-day controlled-release tablet for oral administration. Oxybutynin chloride is administered as a racemate of R- and S-enantiomers.
Chemically, oxybutynin chloride is d,l (racemic) 4-diethylamino-2-butynyl phenylcyclohexylglycolate hydrochloride. The empirical formula of oxybutynin chloride is C22H31NO3•HCl.
Its structural formula is:
Oxybutynin chloride is a white crystalline solid with a molecular weight of 393.9. It is readily soluble in water and acids, but relatively insoluble in alkalis.
Oxybutynin chloride extended-release tablets also contain the following inert ingredients: black iron oxide, cellulose acetate, colloidal silicon dioxide, dextrose, hypromellose, lactose, magnesium stearate, mannitol, polyethylene glycol, propylene glycol, tartaric acid, titanium dioxide, triacetin.
5.1 Angioedema
Angioedema of the face, lips, tongue and/or larynx has been reported with oxybutynin. In some cases, angioedema occurred after the first dose. Angioedema associated with upper airway swelling may be life-threatening. If involvement of the tongue, hypopharynx, or larynx occurs, oxybutynin should be promptly discontinued and appropriate therapy and/or measures necessary to ensure a patent airway should be promptly provided.
8.4 Pediatric Use
The safety and efficacy of Oxybutynin chloride extended-release tablets were studied in 60 children in a 24-week, open-label, non-randomized trial. Patients were aged 6 to 15 years, all had symptoms of detrusor overactivity in association with a neurological condition (e.g., spina bifida), all used clean intermittent catheterization, and all were current users of oxybutynin chloride. Study results demonstrated that administration of Oxybutynin chloride extended-release tablets 5 to 20 mg/day was associated with an increase from baseline in mean urine volume per catheterization from 108 mL to 136 mL, an increase from baseline in mean urine volume after morning awakening from 148 mL to 189 mL, and an increase from baseline in the mean percentage of catheterizations without a leaking episode from 34% to 51%.
Urodynamic results were consistent with clinical results. Administration of Oxybutynin chloride extended-release tablets resulted in an increase from baseline in mean maximum cystometric capacity from 185 mL to 254 mL, a decrease from baseline in mean detrusor pressure at maximum cystometric capacity from 44 cm H2O to 33 cm H2O, and a reduction in the percentage of patients demonstrating uninhibited detrusor contractions (of at least 15 cm H2O) from 60% to 28%.
The pharmacokinetics of Oxybutynin chloride extended-release tablets in these patients were consistent with those reported for adults [see Clinical Pharmacology (12.3)] .
Oxybutynin chloride extended-release tablets are not recommended in pediatric patients who cannot swallow the tablet whole without chewing, dividing, or crushing, or in children under the age of 6.
8.5 Geriatric Use
The rate and severity of anticholinergic effects reported by patients less than 65 years old and those 65 years and older were similar. The pharmacokinetics of Oxybutynin chloride extended-release tablets were similar in all patients studied (up to 78 years of age).
14 Clinical Studies (14 CLINICAL STUDIES)
Oxybutynin chloride extended-release tablets were evaluated for the treatment of patients with overactive bladder with symptoms of urge urinary incontinence, urgency, and frequency in three controlled efficacy studies. The majority of patients were Caucasian (89.0%) and female (91.9%) with a mean age of 59 years (range, 18 to 98 years). Entry criteria required that patients have urge or mixed incontinence (with a predominance of urge) as evidenced by ≥ 6 urge incontinence episodes per week and ≥ 10 micturitions per day. Study 1 was a fixed-dose escalation design, whereas the other two studies used a dose-adjustment design in which each patient's final dose was adjusted to a balance between improvement of incontinence symptoms and tolerability of side effects. All three studies included patients known to be responsive to oxybutynin or other anticholinergic medications, and these patients were maintained on a final dose for up to 2 weeks.
The efficacy results for the three controlled trials are presented in the following Tables 4, 5, and 6 and Figures 3, 4, and 5.
Table 4: Number of Urge Urinary Incontinence Episodes Per Week
| Study 1 | n | Oxybutynin chloride extended-release tablets | n | Placebo |
|---|---|---|---|---|
| Mean Baseline | 34 | 15.9 | 16 | 20.9 |
| Mean (SD) Change from Baseline† | 34 | -15.8 (8.9) | 16 | -7.6 (8.6) |
| 95% Confidence Interval for Difference | (-13.6, -2.8)* | |||
| (Oxybutynin chloride extended-release tablets- Placebo) |
* The difference between Oxybutynin chloride extended-release tablets and placebo was statistically significant.
† Covariate adjusted mean with missing observations set to baseline values
Figure 3: Mean Change (±SD) in Urge Incontinence Episodes Per Week from Baseline (Study 1)
*The difference between Oxybutynin chloride extended-release tablets and placebo was statistically significant.
Table 5: Number of Urge Urinary Incontinence Episodes Per Week (Study 2)
| Study 2 | n | Oxybutynin chloride extended-release tablets | n | oxybutynin |
|---|---|---|---|---|
| Mean Baseline | 53 | 27.6 | 52 | 23.0 |
| Mean (SD) Change from Baseline† | 53 | -17.6 (11.9) | 52 | -19.4 (11.9) |
| 95% Confidence Interval for Difference | (-2.8, 6.5) | |||
| (Oxybutynin chloride extended-release tablets- oxybutynin) |
† Covariate adjusted mean with missing observations set to baseline values
Figure 4: Mean Change (±SD) in Urge Urinary Incontinence Episodes Per Week from Baseline (Study 2)
Table 6: Number of Urge Urinary Incontinence Episodes Per Week (Study 3)
| Study 3 | n | Oxybutynin chloride extended-release tablets | n | oxybutynin |
|---|---|---|---|---|
| Mean Baseline | 111 | 18.9 | 115 | 19.5 |
| Mean (SD) Change from Baseline† | 111 | -14.5 (8.7) | 115 | -13.8 (8.6) |
| 95% Confidence Interval for Difference | (-3.0, 1.6)** | |||
| (Oxybutynin chloride extended-release tablets- oxybutynin) |
** The difference between Oxybutynin chloride extended-release tablets and oxybutynin fulfilled the criteria for comparable efficacy.
† Covariate adjusted mean with missing observations set to baseline values
Figure 5: Mean Change (±SD) in Urge Urinary Incontinence Episodes Per Week from Baseline (Study 3)
** The difference between Oxybutynin chloride extended-release tablets and oxybutynin fulfilled the criteria for comparable efficacy.
4 Contraindications (4 CONTRAINDICATIONS)
Oxybutynin chloride extended-release tablets are contraindicated in patients with urinary retention, gastric retention and other severe decreased gastrointestinal motility conditions, uncontrolled narrow-angle glaucoma.
Oxybutynin chloride extended-release tablets are also contraindicated in patients who have demonstrated hypersensitivity to the drug substance or other components of the product. There have been reports of hypersensitivity reactions, including anaphylaxis and angioedema.
6 Adverse Reactions (6 ADVERSE REACTIONS)
The most common (incidence ≥5%) adverse reactions were dry mouth, constipation, diarrhea, headache, somnolence, and dizziness. (6)
To report SUSPECTED ADVERSE REACTIONS, contact Lannett Company, Inc. at 1-844-834-0530 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
7 Drug Interactions (7 DRUG INTERACTIONS)
The concomitant use of oxybutynin with other anticholinergic drugs or with other agents which produce dry mouth, constipation, somnolence (drowsiness), and/or other anticholinergic-like effects may increase the frequency and/or severity of such effects.
Anticholinergic agents may potentially alter the absorption of some concomitantly administered drugs due to anticholinergic effects on gastrointestinal motility. This may be of concern for drugs with a narrow therapeutic index. Anticholinergic agents may also antagonize the effects of prokinetic agents, such as metoclopramide.
Mean oxybutynin plasma concentrations were approximately 2 fold higher when Oxybutynin chloride extended-release tablets were administered with ketoconazole, a potent CYP3A4 inhibitor. Other inhibitors of the cytochrome P450 3A4 enzyme system, such as antimycotic agents (e.g., itraconazole and miconazole) or macrolide antibiotics (e.g., erythromycin and clarithromycin), may alter oxybutynin mean pharmacokinetic parameters (i.e., Cmax and AUC). The clinical relevance of such potential interactions is not known. Caution should be used when such drugs are co-administered.
8.6 Renal Impairment
There were no studies conducted with Oxybutynin chloride extended-release tablets in patients with renal impairment.
12.2 Pharmacodynamics
In patients with conditions characterized by involuntary bladder contractions, cystometric studies have demonstrated that oxybutynin increases bladder (vesical) capacity, diminishes the frequency of uninhibited contractions of the detrusor muscle, and delays the initial desire to void.
5.5 Urinary Retention
Oxybutynin chloride extended-release tablets should be administered with caution to patients with clinically significant bladder outflow obstruction because of the risk of urinary retention [see Contraindications (4)].
8.7 Hepatic Impairment
There were no studies conducted with Oxybutynin chloride extended-release tablets in patients with hepatic impairment.
1 Indications and Usage (1 INDICATIONS AND USAGE)
Oxybutynin chloride extended-release tablets are a muscarinic antagonist indicated for the treatment of overactive bladder with symptoms of urge urinary incontinence, urgency, and frequency.
Oxybutynin chloride extended-release tablets are also indicated for the treatment of pediatric patients aged 6 years and older with symptoms of detrusor overactivity associated with a neurological condition (e.g., spina bifida).
12.1 Mechanism of Action
Oxybutynin relaxes bladder smooth muscle. Oxybutynin chloride exerts a direct antispasmodic effect on smooth muscle and inhibits the muscarinic action of acetylcholine on smooth muscle. No blocking effects occur at skeletal neuromuscular junctions or autonomic ganglia (antinicotinic effects).
Antimuscarinic activity resides predominantly in the R-isomer. A metabolite, desethyloxybutynin, has pharmacological activity similar to that of oxybutynin in in vitro studies.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Angioedema: Angioedema has been reported with oxybutynin. If symptoms of angioedema occur, discontinue Oxybutynin chloride extended-release tablets immediately and initiate appropriate therapy. (5.1)
- Central Nervous System (CNS) effects: CNS effects have been reported with oxybutynin. If patient experiences anticholinergic CNS effects, consider dose adjustment or discontinuation of Oxybutynin chloride extended-release tablets. (5.2)
- Use with caution due to aggravation of symptoms:
- Pre-existing dementia in patients treated with cholinesterase inhibitors (5.2),
- Parkinson's disease (5.2),
- Myasthenia gravis (5.3), and
- Decreased gastrointestinal motility in patients with autonomic neuropathy. (5.4)
- Urinary Retention: Use with caution in patients with clinically significant bladder outflow obstruction because of the risk of urinary retention (5.5)
- Gastrointestinal Adverse Reactions: Use with caution in patients with gastrointestinal obstructive disorders or decreased intestinal motility due to risk of gastric retention. Use with caution in patients with gastroesophageal reflux or in patients concurrently taking drugs that can exacerbate esophagitis. (5.6)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
Oxybutynin chloride extended-release tablets must be swallowed whole with the aid of liquids, and must not be chewed, divided, or crushed.
Oxybutynin chloride extended-release tablets may be administered with or without food.
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Oxybutynin chloride extended-release tablets are available as 5, 10 and 15 mg tablets for oral use:
5 mg: White, round, biconvex tablet with "270" printed on one side and "KU" printed on the other side with black ink.
10 mg: White, round, biconvex tablet with "271" printed on one side and "KU" printed on the other side with black ink.
15 mg: White, round, biconvex tablet with "272" printed on one side and "KU" printed on the other side with black ink.
6.2 Postmarketing Experience
The following additional adverse reactions have been reported from worldwide postmarketing experience with Oxybutynin chloride extended-release tablets. Because postmarketing reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Infections and Infestations: Urinary tract infection; Psychiatric Disorders: psychotic disorder, agitation, confusional state, hallucinations, memory impairment, abnormal behavior; Nervous System Disorders: convulsions; Eye Disorders: glaucoma; Respiratory, Thoracic and Mediastinal Disorders: nasal congestion; Cardiac Disorders: arrhythmia, tachycardia, palpitations, QT interval prolongation; Vascular Disorders: flushing, hypertension; Skin and Subcutaneous Tissue Disorders: rash; Renal and Urinary Disorders: impotence; General Disorders and Administration Site Conditions: hypersensitivity reactions, including angioedema with airway obstruction, urticaria, and face edema; anaphylactic reactions requiring hospitalization for emergency treatment; Injury, poisoning and procedural complications: fall.
Additional adverse events reported with some other oxybutynin chloride formulations include: cycloplegia, mydriasis, and suppression of lactation. In one reported case, concomitant use of oxybutynin with carbamazepine and dantrolene was associated with adverse events of vomiting, drowsiness, confusion, unsteadiness, slurred speech and nystagmus, suggestive of carbamazepine toxicity.
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
- Pediatric Use: Oxybutynin chloride extended-release tablets are not recommended in pediatric patients who cannot swallow the tablet whole without chewing, dividing or crushing, or in children under the age of 6 years. (8.4)
- Renal or Hepatic Impairment: There have been no studies conducted in patients with renal or hepatic impairment. (8.6, 8.7)
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, the adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
The safety and efficacy of Oxybutynin chloride extended-release tablets (5 to 30 mg/day) was evaluated in 774 adult subjects who participated in five double-blind, controlled clinical trials. In four of the five studies, Oxybutynin chloride IR (5 to 20 mg/day in 199 subjects) was an active comparator. Adverse reactions reported by ≥ 1% of subjects are shown in Table 1.
| System/Organ Class Preferred Term |
Oxybutynin chloride extended-release tablets 5 to 30 mg/day n = 774 % |
Oxybutynin chloride IR IR = immediate release
5 to 20 mg/day n = 199 % |
|---|---|---|
| Psychiatric Disorders | ||
| Insomnia | 3.0 | 5.5 |
| Nervous System Disorders | ||
| Headache | 7.5 | 8.0 |
| Somnolence | 5.6 | 14.1 |
| Dizziness | 5.0 | 16.6 |
| Dysgeusia | 1.6 | 1.5 |
| Eye Disorders | ||
| Vision blurred | 4.3 | 9.6 |
| Dry eye | 3.1 | 2.5 |
| Respiratory, Thoracic and Mediastinal Disorders | ||
| Cough | 1.9 | 3.0 |
| Oropharyngeal pain | 1.9 | 1.5 |
| Dry throat | 1.7 | 2.5 |
| Nasal dryness | 1.7 | 4.5 |
| Gastrointestinal Disorders | ||
| Dry mouth | 34.9 | 72.4 |
| Constipation | 8.7 | 15.1 |
| Diarrhea | 7.9 | 6.5 |
| Dyspepsia | 4.5 | 6.0 |
| Nausea | 4.5 | 11.6 |
| Abdominal pain | 1.6 | 2.0 |
| Vomiting | 1.3 | 1.5 |
| Flatulence | 1.2 | 2.5 |
| Gastro-esophageal reflux disease | 1.0 | 0.5 |
| Skin and Subcutaneous Tissue Disorders | ||
| Dry skin | 1.8 | 2.5 |
| Pruritus | 1.3 | 1.5 |
| Renal and Urinary Disorders | ||
| Dysuria | 1.9 | 2.0 |
| Urinary hesitation | 1.9 | 8.5 |
| Urinary retention | 1.2 | 3.0 |
| General Disorders and Administration Site Conditions | ||
| Fatigue | 2.6 | 3.0 |
| Investigations | ||
| Residual urine volume The bundled term residual urine volume consists of the preferred terms residual urine volume and residual urine volume increased.
|
2.3 | 3.5 |
The discontinuation rate due to adverse reactions was 4.4% with Oxybutynin chloride extended-release tablets compared to 0% with Oxybutynin chloride IR. The most frequent adverse reaction causing discontinuation of study medication was dry mouth (0.7%).
The following adverse reactions were reported by <1% of Oxybutynin chloride extended-release tablets-treated patients and at a higher incidence than placebo in clinical trials: Metabolism and Nutrition Disorders: anorexia, fluid retention; Vascular disorders: hot flush; Respiratory, thoracic and mediastinal disorders: dysphonia; Gastrointestinal Disorders: dysphagia, frequent bowel movements; General disorders and administration site conditions: chest discomfort, thirst.
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
- Patients should be informed that oxybutynin may produce angioedema that could result in life threatening airway obstruction. Patients should be advised to promptly discontinue oxybutynin therapy and seek immediate medical attention if they experience swelling of the tongue, edema of the laryngopharynx, or difficulty breathing.
- Patients should be informed that anticholinergic (antimuscarinic) agents such as Oxybutynin chloride extended-release tablets, may produce clinically significant adverse reactions related to anticholinergic activity such as:
- Urinary retention and constipation
- Heat prostration due to decreased sweating. Heat prostration can occur when anticholinergic medicines are administered in the presence of high environmental temperature.
- Patients should be informed that anticholinergic medicines such as Oxybutynin chloride extended-release tablets may produce drowsiness (somnolence), dizziness or blurred vision. Patients should be advised to exercise caution in decisions to engage in potentially dangerous activities until Oxybutynin chloride extended-release tablets effects have been determined.
- Patients should be informed that alcohol may enhance the drowsiness caused by anticholinergic agents such as Oxybutynin chloride extended-release tablets.
- Patients should be informed that Oxybutynin chloride extended-release tablets should be swallowed whole with the aid of liquids. Patients should not chew, divide, or crush tablets. The medication is contained within a nonabsorbable shell designed to release the drug at a controlled rate. The tablet shell is eliminated from the body; patients should not be concerned if they occasionally notice in their stool something that looks like a tablet.
- Oxybutynin chloride extended-release tablets should be taken at approximately the same time each day.
For more information call 1-844-834-0530.
5.2 Central Nervous System Effects
Oxybutynin is associated with anticholinergic central nervous system (CNS) effects [see Adverse Reactions (6)] . A variety of CNS anticholinergic effects have been reported, including hallucinations, agitation, confusion and somnolence. Patients should be monitored for signs of anticholinergic CNS effects, particularly in the first few months after beginning treatment or increasing the dose. Advise patients not to drive or operate heavy machinery until they know how Oxybutynin chloride extended-release tablets affect them. If a patient experiences anticholinergic CNS effects, dose reduction or drug discontinuation should be considered.
Oxybutynin chloride extended-release tablets should be used with caution in patients with preexisting dementia treated with cholinesterase inhibitors due to the risk of aggravation of symptoms.
Oxybutynin chloride extended-release tablets should be used with caution in patients with Parkinson's disease due to the risk of aggravation of symptoms.
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
Oxybutynin chloride extended-release tablets, 5 mg are round, biconvex, white coated tablets imprinted in black ink with "270" on one side and "KU" on the other side.
They are supplied as follows:
Bottles of 100 Tablets NDC 62175-270-37
Bottles of 500 Tablets NDC 62175-270-41
Oxybutynin chloride extended-release tablets, 10 mg are round, biconvex, white coated tablets imprinted in black ink with "271" on one side and "KU" on the other side.
They are supplied as follows:
Bottles of 100 Tablets NDC 62175-271-37
Bottles of 500 Tablets NDC 62175-271-41
Oxybutynin chloride extended-release tablets, 15 mg are round, biconvex, white coated tablets imprinted in black ink with "272" on one side and "KU" on the other side.
They are supplied as follows:
Bottles of 100 Tablets NDC 62175-272-37
Bottles of 500 Tablets NDC 62175-272-41
Storage
Store at 25°C (77°F); excursions permitted to 15–30°C (59–86°F) [see USP Controlled Room Temperature]. Protect from moisture and humidity.
Keep out of reach of children.
5.6 Gastrointestinal Adverse Reactions
Oxybutynin chloride extended-release tablets should be administered with caution to patients with gastrointestinal obstructive disorders because of the risk of gastric retention [see Contraindications (4)] .
Oxybutynin chloride extended-release tablets, like other anticholinergic drugs, may decrease gastrointestinal motility and should be used with caution in patients with conditions such as ulcerative colitis and intestinal atony.
Oxybutynin chloride extended-release tablets should be used with caution in patients who have gastroesophageal reflux and/or who are concurrently taking drugs (such as bisphosphonates) that can cause or exacerbate esophagitis.
As with any other nondeformable material, caution should be used when administering Oxybutynin chloride extended-release tablets to patients with preexisting severe gastrointestinal narrowing (pathologic or iatrogenic). There have been rare reports of obstructive symptoms in patients with known strictures in association with the ingestion of other drugs in nondeformable controlled-release formulations.
5.3 Worsening of Symptoms of Myasthenia Gravis
Oxybutynin chloride extended-release tablets should be used with caution in patients with myasthenia gravis due to the risk of aggravation of symptoms.
2.2 Pediatric Patients Aged 6 Years of Age and Older
The recommended starting dose of Oxybutynin chloride extended-release tablets is 5 mg once daily at approximately the same time each day. Dosage may be adjusted in 5-mg increments to achieve a balance of efficacy and tolerability (up to a maximum of 20 mg/day).
Principal Display Panel 5 Mg 500 Tablet Bottle Label (PRINCIPAL DISPLAY PANEL - 5 mg 500 Tablet Bottle Label)
NDC 62175-270-41
500 tablets
Oxybutynin chloride
Extended-release tablets
5 mg
Rx only
Principal Display Panel 10 Mg 500 Tablet Bottle Label (PRINCIPAL DISPLAY PANEL - 10 mg 500 Tablet Bottle Label)
NDC 62175-271-41
500 tablets
Oxybutynin chloride
Extended-release tablets
10 mg
Rx only
Principal Display Panel 5 Mg 100 Tablet Bottle Label (PRINCIPAL DISPLAY PANEL - 5 mg 100 Tablet Bottle Label)
NDC 62175-270-37
100 tablets
Oxybutynin chloride
Extended-release tablets
5 mg
Rx only
Principal Display Panel 10 Mg 100 Tablet Bottle Label (PRINCIPAL DISPLAY PANEL - 10 mg 100 Tablet Bottle Label)
NDC 62175-271-37
100 tablets
Oxybutynin chloride
Extended-release tablets
10 mg
Rx only
Principal Display Panel 15 Mg 100 tablet Bottle Label (PRINCIPAL DISPLAY PANEL - 15 mg 100 Tablet Bottle Label)
NDC 62175-272-37
100 tablets
Oxybutynin chloride
Extended-release tablets
15 mg
Rx only
Principal Display Panel 15 Mg 500 tablet Bottle Label (PRINCIPAL DISPLAY PANEL - 15 mg 500 Tablet Bottle Label)
NDC 62175-272-41
500 tablets
Oxybutynin chloride
Extended-release tablets
15 mg
Rx only
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenesis
A 24-month study in rats at dosages of oxybutynin chloride of 20, 80, and 160 mg/kg/day showed no evidence of carcinogenicity. These doses are approximately 6, 25, and 50 times the maximum human exposure, based on a human equivalent dose taking into account normalization of body surface area.
Mutagenesis
Oxybutynin chloride showed no increase of mutagenic activity when tested in Schizosaccharomyces pompholiciformis, Saccharomyces cerevisiae, and Salmonella typhimurium test systems.
Impairment of Fertility
No impairment of fertility was seen in rats at dosages up to 75 mg/kg/day (24 times the MRHD on a mg/m2 basis) when administered for 2 weeks prior to mating in females and for 9 weeks prior to mating in males.
5.4 Worsening of Symptoms of Decreased Gastrointestinal Motility in Patients With Autonomic Neuropathy (5.4 Worsening of Symptoms of Decreased Gastrointestinal Motility in Patients with Autonomic Neuropathy)
Oxybutynin chloride extended-release tablets should be used with caution in patients with autonomic neuropathy due to the risk of aggravation of symptoms of decreased gastrointestinal motility.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:38:34.221386 · Updated: 2026-03-14T21:55:04.881225