These Highlights Do Not Include All The Information Needed To Use Midazolam Injection Safely And Effectively. See Full Prescribing Information For Midazolam Injection
c546cfd4-4f2b-4b94-aa47-995595f1a146
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Personnel and Equipment for Monitoring and Resuscitation Only personnel trained in the administration of procedural sedation, and not involved in the conduct of the diagnostic or therapeutic procedure, should administer Midazolam in 0.9% Sodium Chloride Injection [see Dosage and Administration ( 2.1 ), Warnings and Precautions ( 5.1 )]. Administering personnel must be trained in the detection and management of airway obstruction, hypoventilation, and apnea, including the maintenance of a patent airway, supportive ventilation, and cardiovascular resuscitation [see Dosage and Administration ( 2.1 ) , Warnings and Precautions ( 5.1 )]. Resuscitative drugs, and age- and size-appropriate equipment for bag/valve/mask assisted ventilation must be immediately available during administration of Midazolam in 0.9% Sodium Chloride Injection [see Dosage and Administration ( 2.1 ) , Warnings and Precautions ( 5.1 )]. Continuously monitor vital signs during sedation and during the recovery period [see Dosage and Administration ( 2.1 ) , Warnings and Precautions ( 5.1 )]. Risks from Concomitant Use with Opioid Analgesics and Other Sedative Hypnotics Concomitant use of benzodiazepines, including Midazolam in 0.9% Sodium Chloride Injection, and opioids may result in profound sedation, respiratory depression, coma, and death. Continuously monitor patients for respiratory depression and depth of sedation [see Warnings and Precautions ( 5.2 ) and Drug Interaction ( 7.1 )] .
Indications and Usage
Midazolam in 0.9% Sodium Chloride Injection is indicated: Continuous intravenous infusion for sedation of intubated and mechanically ventilated adult, pediatric, and neonatal patients as a component of anesthesia or during treatment in a critical care setting.
Dosage and Administration
For intravenous injection only. Avoid intra-arterial injection or extravasation. ( 2.1 ) Individualize dosing and titrate to desired clinical response, taking into account patient age, clinical status, and concomitant use of other CNS depressants. ( 2.1 ) See Full Prescribing Information for complete dosage and administration information. ( 2 )
Warnings and Precautions
Cardiorespiratory Adverse Reactions: Serious cardiorespiratory adverse reactions have occurred, sometimes resulting in death or permanent neurologic injury. ( 5.3 ) Paradoxical Behavior: Agitation, involuntary movements (including tonic/clonic movements and muscle tremor), hyperactivity and combativeness have been reported in both adult and pediatric patients. ( 5.4 ) Dependence and Withdrawal with Long-Term Use: Use for several days to weeks may lead to physical dependence to midazolam. Do not abruptly discontinue midazolam. Gradually taper the dosage using a tapering schedule that is individualized to the patient. ( 5.5 ) Debilitation and Comorbid Considerations : Higher risk adult and pediatric surgical patients, elderly patients and debilitated adult and pediatric patients require lower dosages, whether or not concomitant sedating medications have been administered. ( 5.6 ) Risk of Intra-Arterial Injection: There have been limited reports of intra-arterial injection of midazolam. Adverse events have included local reactions, as well as isolated reports of seizure activity in which no clear causal relationship was established. ( 5.7 ) Impaired Cognitive Function : Because of partial or complete impairment of recall, patients should not operate hazardous machinery or a motor vehicle until drug effects have subsided. ( 5.8 ) Hypotension and Seizure in Preterm Infants and Neonates: Avoid rapid injection in the neonatal population. ( 5.9 ) Neonatal Sedation and Withdrawal Syndrome: Receiving Midazolam in 0.9% Sodium Chloride Injection during pregnancy can result in neonatal sedation and/or neonatal withdrawal. ( 5.10 , 8.1 ) Pediatric Neurotoxicity : In developing animals, exposures greater than 3 hours cause neurotoxicity. Weigh benefits against potential risks when considering elective procedures in children under 3 years old. ( 5.11 )
Contraindications
Midazolam in 0.9% Sodium Chloride Injection is contraindicated in patients with: Known hypersensitivity to midazolam Acute narrow-angle glaucoma
Adverse Reactions
Serious cardiorespiratory adverse reactions have occurred after administration of midazolam. These have included respiratory depression, airway obstruction, oxygen desaturation, apnea, respiratory arrest and/or cardiac arrest, sometimes resulting in death or permanent neurologic injury. There have also been rare reports of hypotensive episodes requiring treatment during or after diagnostic or surgical manipulations particularly in adult or pediatric patients with hemodynamic instability. Hypotension occurred more frequently in the sedation studies in patients premedicated with an opioid. Excessive single doses or rapid intravenous administration may result in respiratory depression, airway obstruction and/or arrest. When used for sedation/anxiolysis/amnesia, midazolam should always be titrated slowly in adult or pediatric patients. Adverse hemodynamic events have been reported in pediatric patients with cardiovascular instability; rapid intravenous administration should also be avoided in this population. Continuously monitor patients for early signs of hypoventilation, airway obstruction, and apnea using capnography, pulse oximetry, and clinical assessment [see Dosage and Administration ( 2.2 )] .
Drug Interactions
Opioid Analgesics and Other Sedative Hypnotics: Risk of respiratory depression is increased ( 7.1 ) Cytochrome P450-3A4 Inhibitors: May result in prolonged sedation due to decreased plasma clearance of midazolam. ( 7.2 )
Storage and Handling
Midazolam in 0.9% Sodium Chloride Injection is a clear, colorless solution supplied in single-dose bags with an aluminum overwrap available as: *Partial fill container 50 mL volume in 100 mL container NDC Midazolam in 0.9% Sodium Chloride Injection Package Factor (1 mg per mL) 25021-688-82 *50 mg per 50 mL Single-Dose Bag 10 bags per carton 25021-688-87 100 mg per 100 mL Single-Dose Bag 20 bags per carton Store at 20° to 25°C (68° to 77°F); excursions permitted between 15° and 30°C (59° and 86°F). [See USP Controlled Room Temperature.] Individual containers may be used up to 48 hours after initial penetration. Do not freeze. Discard unused portion. Sterile, Nonpyrogenic, Preservative-free, PVC-free, DEHP-free. The container closure is not made with natural rubber latex.
How Supplied
Midazolam in 0.9% Sodium Chloride Injection is a clear, colorless solution supplied in single-dose bags with an aluminum overwrap available as: *Partial fill container 50 mL volume in 100 mL container NDC Midazolam in 0.9% Sodium Chloride Injection Package Factor (1 mg per mL) 25021-688-82 *50 mg per 50 mL Single-Dose Bag 10 bags per carton 25021-688-87 100 mg per 100 mL Single-Dose Bag 20 bags per carton Store at 20° to 25°C (68° to 77°F); excursions permitted between 15° and 30°C (59° and 86°F). [See USP Controlled Room Temperature.] Individual containers may be used up to 48 hours after initial penetration. Do not freeze. Discard unused portion. Sterile, Nonpyrogenic, Preservative-free, PVC-free, DEHP-free. The container closure is not made with natural rubber latex.
Medication Information
Warnings and Precautions
Cardiorespiratory Adverse Reactions: Serious cardiorespiratory adverse reactions have occurred, sometimes resulting in death or permanent neurologic injury. ( 5.3 ) Paradoxical Behavior: Agitation, involuntary movements (including tonic/clonic movements and muscle tremor), hyperactivity and combativeness have been reported in both adult and pediatric patients. ( 5.4 ) Dependence and Withdrawal with Long-Term Use: Use for several days to weeks may lead to physical dependence to midazolam. Do not abruptly discontinue midazolam. Gradually taper the dosage using a tapering schedule that is individualized to the patient. ( 5.5 ) Debilitation and Comorbid Considerations : Higher risk adult and pediatric surgical patients, elderly patients and debilitated adult and pediatric patients require lower dosages, whether or not concomitant sedating medications have been administered. ( 5.6 ) Risk of Intra-Arterial Injection: There have been limited reports of intra-arterial injection of midazolam. Adverse events have included local reactions, as well as isolated reports of seizure activity in which no clear causal relationship was established. ( 5.7 ) Impaired Cognitive Function : Because of partial or complete impairment of recall, patients should not operate hazardous machinery or a motor vehicle until drug effects have subsided. ( 5.8 ) Hypotension and Seizure in Preterm Infants and Neonates: Avoid rapid injection in the neonatal population. ( 5.9 ) Neonatal Sedation and Withdrawal Syndrome: Receiving Midazolam in 0.9% Sodium Chloride Injection during pregnancy can result in neonatal sedation and/or neonatal withdrawal. ( 5.10 , 8.1 ) Pediatric Neurotoxicity : In developing animals, exposures greater than 3 hours cause neurotoxicity. Weigh benefits against potential risks when considering elective procedures in children under 3 years old. ( 5.11 )
Indications and Usage
Midazolam in 0.9% Sodium Chloride Injection is indicated: Continuous intravenous infusion for sedation of intubated and mechanically ventilated adult, pediatric, and neonatal patients as a component of anesthesia or during treatment in a critical care setting.
Dosage and Administration
For intravenous injection only. Avoid intra-arterial injection or extravasation. ( 2.1 ) Individualize dosing and titrate to desired clinical response, taking into account patient age, clinical status, and concomitant use of other CNS depressants. ( 2.1 ) See Full Prescribing Information for complete dosage and administration information. ( 2 )
Contraindications
Midazolam in 0.9% Sodium Chloride Injection is contraindicated in patients with: Known hypersensitivity to midazolam Acute narrow-angle glaucoma
Adverse Reactions
Serious cardiorespiratory adverse reactions have occurred after administration of midazolam. These have included respiratory depression, airway obstruction, oxygen desaturation, apnea, respiratory arrest and/or cardiac arrest, sometimes resulting in death or permanent neurologic injury. There have also been rare reports of hypotensive episodes requiring treatment during or after diagnostic or surgical manipulations particularly in adult or pediatric patients with hemodynamic instability. Hypotension occurred more frequently in the sedation studies in patients premedicated with an opioid. Excessive single doses or rapid intravenous administration may result in respiratory depression, airway obstruction and/or arrest. When used for sedation/anxiolysis/amnesia, midazolam should always be titrated slowly in adult or pediatric patients. Adverse hemodynamic events have been reported in pediatric patients with cardiovascular instability; rapid intravenous administration should also be avoided in this population. Continuously monitor patients for early signs of hypoventilation, airway obstruction, and apnea using capnography, pulse oximetry, and clinical assessment [see Dosage and Administration ( 2.2 )] .
Drug Interactions
Opioid Analgesics and Other Sedative Hypnotics: Risk of respiratory depression is increased ( 7.1 ) Cytochrome P450-3A4 Inhibitors: May result in prolonged sedation due to decreased plasma clearance of midazolam. ( 7.2 )
Storage and Handling
Midazolam in 0.9% Sodium Chloride Injection is a clear, colorless solution supplied in single-dose bags with an aluminum overwrap available as: *Partial fill container 50 mL volume in 100 mL container NDC Midazolam in 0.9% Sodium Chloride Injection Package Factor (1 mg per mL) 25021-688-82 *50 mg per 50 mL Single-Dose Bag 10 bags per carton 25021-688-87 100 mg per 100 mL Single-Dose Bag 20 bags per carton Store at 20° to 25°C (68° to 77°F); excursions permitted between 15° and 30°C (59° and 86°F). [See USP Controlled Room Temperature.] Individual containers may be used up to 48 hours after initial penetration. Do not freeze. Discard unused portion. Sterile, Nonpyrogenic, Preservative-free, PVC-free, DEHP-free. The container closure is not made with natural rubber latex.
How Supplied
Midazolam in 0.9% Sodium Chloride Injection is a clear, colorless solution supplied in single-dose bags with an aluminum overwrap available as: *Partial fill container 50 mL volume in 100 mL container NDC Midazolam in 0.9% Sodium Chloride Injection Package Factor (1 mg per mL) 25021-688-82 *50 mg per 50 mL Single-Dose Bag 10 bags per carton 25021-688-87 100 mg per 100 mL Single-Dose Bag 20 bags per carton Store at 20° to 25°C (68° to 77°F); excursions permitted between 15° and 30°C (59° and 86°F). [See USP Controlled Room Temperature.] Individual containers may be used up to 48 hours after initial penetration. Do not freeze. Discard unused portion. Sterile, Nonpyrogenic, Preservative-free, PVC-free, DEHP-free. The container closure is not made with natural rubber latex.
Description
Personnel and Equipment for Monitoring and Resuscitation Only personnel trained in the administration of procedural sedation, and not involved in the conduct of the diagnostic or therapeutic procedure, should administer Midazolam in 0.9% Sodium Chloride Injection [see Dosage and Administration ( 2.1 ), Warnings and Precautions ( 5.1 )]. Administering personnel must be trained in the detection and management of airway obstruction, hypoventilation, and apnea, including the maintenance of a patent airway, supportive ventilation, and cardiovascular resuscitation [see Dosage and Administration ( 2.1 ) , Warnings and Precautions ( 5.1 )]. Resuscitative drugs, and age- and size-appropriate equipment for bag/valve/mask assisted ventilation must be immediately available during administration of Midazolam in 0.9% Sodium Chloride Injection [see Dosage and Administration ( 2.1 ) , Warnings and Precautions ( 5.1 )]. Continuously monitor vital signs during sedation and during the recovery period [see Dosage and Administration ( 2.1 ) , Warnings and Precautions ( 5.1 )]. Risks from Concomitant Use with Opioid Analgesics and Other Sedative Hypnotics Concomitant use of benzodiazepines, including Midazolam in 0.9% Sodium Chloride Injection, and opioids may result in profound sedation, respiratory depression, coma, and death. Continuously monitor patients for respiratory depression and depth of sedation [see Warnings and Precautions ( 5.2 ) and Drug Interaction ( 7.1 )] .
Section 42229-5
Adults and Pediatrics:
Sedation guidelines recommend a careful presedation history to determine how a patient's underlying medical conditions or concomitant medications might affect their response to sedation/analgesia as well as a physical examination including a focused examination of the airway for abnormalities. Further recommendations include appropriate presedation fasting.
Section 51945-4
PACKAGE LABEL – PRINCIPAL DISPLAY PANEL – Bag Label
NDC 25021-688-82
Rx only
Midazolam in 0.9% Sodium Chloride Injection
50 mg per 50 mL
(1 mg per mL)
50 mL Single-Dose Container
High Alert Medication
For Intravenous Use Only
Do Not Dilute
9.2 Abuse
Midazolam in 0.9% Sodium Chloride Injection contains the benzodiazepine, midazolam. Benzodiazepines are a class of sedative drugs with a known potential for abuse. Abuse is the intentional, non-therapeutic use of a drug, even once, for its desirable psychological or physiological effects.
Misuse is the intentional use, for therapeutic purposes, of a drug by an individual in a way other than prescribed by a health care provider or for whom it was not prescribed. Drug addiction is a cluster of behavioral, cognitive, and physiological phenomena that may include a strong desire to take the drug, difficulties in controlling drug use (e.g., continuing drug use despite harmful consequences, giving a higher priority to drug use than other activities and obligations), and possible tolerance or physical dependence. Both abuse and misuse may lead to addiction. Midazolam was actively self-administered in primate models used to assess the positive reinforcing effects of psychoactive drugs. Midazolam produced physical dependence of a mild to moderate intensity in cynomolgus monkeys after 5 to 10 weeks of administration. Available data concerning the drug abuse and dependence potential of midazolam suggest that its abuse potential is at least equivalent to that of diazepam.
7.5 Halothane
The intravenous administration of midazolam decreases the minimum alveolar concentration (MAC) of halothane required for general anesthesia. This decrease correlates with the dose of midazolam administered; no similar studies have been carried out in pediatric patients but there is no scientific reason to expect that pediatric patients would respond differently than adults.
11 Description
Midazolam in 0.9% Sodium Chloride Injection is a benzodiazepine available as a sterile, preservative-free, nonpyrogenic solution of midazolam and sodium chloride in water for injection for intravenous use. Each single-dose bag of Midazolam in 0.9% Sodium Chloride Injection contains either 50 mg per 50 mL (1 mg per mL) or 100 mg per 100 mL (1 mg per mL) of midazolam and 9 mg/mL of sodium chloride in water for injection. Midazolam in 0.9% Sodium Chloride Injection may contain hydrochloric acid and/or sodium hydroxide for pH adjustment. The pH is approximately 2.5 to 3.5.
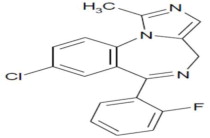
Midazolam is a white or yellowish powder, practically insoluble in water, Chemically, midazolam is 8-chloro-6-(2-fluorophenyl)-1-methyl-4H-imidazo[1,5-a][1,4]-benzodiazepine. Midazolam has the empirical formula C18 H13ClFN3, a calculated molecular weight of 325.8 and the following structural formula:
7.3 Saquinavir
In a placebo-controlled study, saquinavir administered as a 1200 mg dose, tid, for 5 days (n=12), a 56% reduction in the clearance of midazolam following a single 0.05 mg/kg intravenous dose was observed. The half-life was approximately doubled.
7.4 Thiopental
A moderate reduction in induction dosage requirements of thiopental (about 15%) has been noted following use of intramuscular midazolam for premedication in adults.
9.3 Dependence
Midazolam may produce physical dependence after long-term use. Physical dependence is a state that develops as a result of physiological adaptation in response to repeated drug use, manifested by withdrawal signs and symptoms after abrupt discontinuation or a significant dose reduction of a drug. If Midazolam in 0.9% Sodium Chloride Injection is administered long-term (i.e., for several days to weeks), abrupt discontinuation or rapid dosage reduction, or administration of flumazenil, a benzodiazepine antagonist, may precipitate acute withdrawal reactions, including seizures, which can be life-threatening. Patients at an increased risk of withdrawal adverse reactions after benzodiazepine discontinuation or rapid dosage reduction include those who take higher dosages (i.e., higher and/or more frequent doses) and those who have had longer durations of use [see Warnings and Precautions (5.5)].
To reduce the risk of withdrawal reactions, after extended therapy, do not abruptly discontinue Midazolam in 0.9% Sodium Chloride Injection. Gradually taper the dosage using a tapering schedule that is individualized to the patient.
7.6 Pancuronium
Although the possibility of minor interactive effects has not been fully studied, midazolam and pancuronium have been used together in patients without noting clinically significant changes in dosage, onset or duration in adults. Midazolam does not protect against the characteristic circulatory changes noted after administration of succinylcholine or pancuronium and does not protect against the increased intracranial pressure noted following administration of succinylcholine. Midazolam does not cause a clinically significant change in dosage, onset or duration of a single intubating dose of succinylcholine; no similar studies have been carried out in pediatric patients but there is no scientific reason to expect that pediatric patients would respond differently than adults.
8.4 Pediatric Use
The safety and efficacy of midazolam for sedation/anxiolysis/amnesia following continuous infusion have been established in pediatric and neonatal patients. UNLIKE ADULT PATIENTS, PEDIATRIC PATIENTS GENERALLY RECEIVE INCREMENTS OF MIDAZOLAM ON A MG/KG BASIS. As a group, pediatric patients generally require higher dosages of midazolam (mg/kg) than do adults. Younger (less than six years) pediatric patients may require higher dosages (mg/kg) than older pediatric patients, and may require closer monitoring. In obese PEDIATRIC PATIENTS, the dose should be calculated based on ideal body weight. When midazolam is given in conjunction with opioids or other sedatives, the potential for respiratory depression, airway obstruction, or hypoventilation is increased. The health care practitioner who uses this medication in pediatric patients should be aware of and follow accepted professional guidelines for pediatric sedation appropriate to their situation.
Midazolam should not be administered by rapid injection in the neonatal population. Severe hypotension and seizures have been reported following rapid intravenous administration, particularly, with concomitant use of fentanyl.
8.5 Geriatric Use
Because geriatric patients may have altered drug distribution and diminished hepatic and/or renal function, reduced doses of midazolam are recommended. Doses of Midazolam in 0.9% Sodium Chloride Injection should be decreased for elderly and for debilitated patients [see Warnings and Precautions (5.6) and Dosage and Administration (2)] and subjects over 70 years of age may be particularly sensitive. These patients will also probably take longer to recover completely after midazolam administration for the induction of anesthesia. Administration of intravenous midazolam to elderly and/or high-risk surgical patients has been associated with rare reports of death under circumstances compatible with cardiorespiratory depression. In most of these cases, the patients also received other central nervous system depressants capable of depressing respiration, especially opioids [see Dosage and Administration (2)].
Midazolam is known to be substantially excreted by the kidney, and the risk of adverse reactions to this drug may be greater in patients with impaired renal function. Because elderly patients are more likely to have decreased renal function, care should be taken in dose selection, and it may be useful to monitor renal function.
4 Contraindications
Midazolam in 0.9% Sodium Chloride Injection is contraindicated in patients with:
- Known hypersensitivity to midazolam
- Acute narrow-angle glaucoma
6 Adverse Reactions
The following serious adverse reactions are discussed in greater detail in other sections:
- Cardiorespiratory Adverse Reactions [see Warnings and Precautions (5.3)]
- Paradoxical Behavior [see Warnings and Precautions (5.4)]
- Dependence and Withdrawal [see Warnings and Precautions (5.5)]
- Impaired Cognitive Function [see Warnings and Precautions (5.8)]
- Hypotension and Seizure in Preterm Infants and Neonates [see Warnings and Precautions (5.9)]
- Neonatal Sedation and Withdrawal Syndrome [see Warnings and Precautions (5.10), Use in Specific Populations (8.1)]
- Pediatric Neurotoxicity [see Warnings and Precautions (5.11)]
The following adverse reactions have been identified from literature or postmarketing reports of midazolam. Because some of these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Fluctuations in vital signs were the most frequently seen findings following parenteral administration of midazolam in adults and included decreased tidal volume and/or respiratory rate decrease (23.3% of patients following intravenous administration) and apnea (15.4% of patients following intravenous administration), as well as variations in blood pressure and pulse rate. The majority of serious adverse reactions, particularly those associated with oxygenation and ventilation, have been reported when midazolam is administered with other medications capable of depressing the central nervous system. The incidence of such events is higher in patients undergoing procedures involving the airway without the protective effect of an endotracheal tube, (e.g., upper endoscopy and dental procedures).
7 Drug Interactions
12.2 Pharmacodynamics
The effects of midazolam on the CNS are dependent on the dose administered, the route of administration, and the presence or absence of other medications.
12.3 Pharmacokinetics
Midazolam's activity is primarily due to the parent drug. Elimination of the parent drug takes place via hepatic metabolism of midazolam to hydroxylated metabolites that are conjugated and excreted in the urine. Six single-dose pharmacokinetic studies involving healthy adults yield pharmacokinetic parameters for midazolam in the following ranges: volume of distribution (Vd), 1.0 to 3.1 L/kg; elimination half-life, 1.8 to 6.4 hours (mean approximately 3 hours); total clearance (Cl), 0.25 to 0.54 L/hr/kg. In a parallel group study, there was no difference in the clearance, in subjects administered 0.15 mg/kg (n=4) and 0.30 mg/kg (n=4) intravenous doses indicating linear kinetics. The clearance was successively reduced by approximately 30% at doses of 0.45 mg/kg (n=4) and 0.6 mg/kg (n=5) indicating non-linear kinetics in this dose range.
1 Indications and Usage
Midazolam in 0.9% Sodium Chloride Injection is indicated:
- Continuous intravenous infusion for sedation of intubated and mechanically ventilated adult, pediatric, and neonatal patients as a component of anesthesia or during treatment in a critical care setting.
12.1 Mechanism of Action
Midazolam is a short-acting benzodiazepine central nervous system (CNS) depressant.
9.1 Controlled Substance
Midazolam in 0.9% Sodium Chloride Injection contains midazolam, a Schedule IV controlled substance.
2.3 Dosing Recommendations
Table 1 provides dosing recommendations for adult, pediatric, and neonatal patients.
| ADULT PATIENTS | If a loading dose is necessary to rapidly initiate sedation, 0.01 mg/kg to 0.05 mg/kg (approximately 0.5 mg to 4 mg for a typical adult) may be given slowly or infused over several minutes. This dose may be repeated at 10 to 15 minute intervals until adequate sedation is achieved. For maintenance of sedation, the usual initial infusion rate is 0.02 mg/kg/hr to 0.10 mg/kg/hr (1 mg/hr to 7 mg/hr). Higher loading or maintenance infusion rates may occasionally be required in some patients. Use the lowest recommended doses in patients with residual effects from anesthetic drugs, or in those concurrently receiving other sedatives or opioids. Individual response to midazolam is variable. Titrate the infusion rate to the desired level of sedation, taking into account the patient's age, clinical status and current medications. In general, midazolam should be infused at the lowest rate that produces the desired level of sedation. Assess sedation at regular intervals and adjust the midazolam infusion rate. Finding the minimum effective infusion rate decreases the potential accumulation of midazolam and provides for the most rapid recovery once the infusion is terminated. |
| PEDIATRIC PATIENTS | UNLIKE ADULT PATIENTS, PEDIATRIC PATIENTS GENERALLY RECEIVE INCREMENTS OF MIDAZOLAM ON A MG/KG BASIS. As a group, pediatric patients generally require higher dosages of midazolam (mg/kg) than adults. Younger (less than six years) pediatric patients may require higher dosages (mg/kg) than older pediatric patients. In obese pediatric patients, calculate the dose based on ideal body weight. Titrate the dose to the desired level of sedation. Assess for desired level of sedation and vital signs at regular intervals. |
|
PRETERM AND TERM
NEONATAL PATIENTS |
Based on pharmacokinetic parameters and reported clinical experience in preterm and term neonates WHOSE TRACHEA WAS INTUBATED, initiate continuous intravenous infusions of Midazolam in 0.9% Sodium Chloride Injection at a rate of 0.03 mg/kg/hr (0.5 mcg/kg/min) in neonates <32 weeks and 0.06 mg/kg/hr (1 mcg/kg/min) in neonates >32 weeks. Intravenous loading doses should not be used in neonates, rather the infusion may be run more rapidly for the first several hours to establish therapeutic plasma levels. Frequently assess the rate of infusion, particularly after the first 24 hours so as to administer the lowest possible effective dose and reduce the potential for drug accumulation. Hypotension may be observed in patients who are critically ill and in preterm and term infants, particularly those receiving fentanyl and/or when midazolam is administered rapidly. When sedating preterm and former preterm neonates WHOSE TRACHEA WAS NOT INTUBATED, monitor respiratory parameters due to an increased risk of apnea. |
5 Warnings and Precautions
Cardiorespiratory Adverse Reactions: Serious cardiorespiratory adverse reactions have occurred, sometimes resulting in death or permanent neurologic injury. (5.3)
Paradoxical Behavior: Agitation, involuntary movements (including tonic/clonic movements and muscle tremor), hyperactivity and combativeness have been reported in both adult and pediatric patients. (5.4)
Dependence and Withdrawal with Long-Term Use: Use for several days to weeks may lead to physical dependence to midazolam. Do not abruptly discontinue midazolam. Gradually taper the dosage using a tapering schedule that is individualized to the patient. (5.5)
Debilitation and Comorbid Considerations: Higher risk adult and pediatric surgical patients, elderly patients and debilitated adult and pediatric patients require lower dosages, whether or not concomitant sedating medications have been administered. (5.6)
Risk of Intra-Arterial Injection: There have been limited reports of intra-arterial injection of midazolam. Adverse events have included local reactions, as well as isolated reports of seizure activity in which no clear causal relationship was established. (5.7)
Impaired Cognitive Function: Because of partial or complete impairment of recall, patients should not operate hazardous machinery or a motor vehicle until drug effects have subsided. (5.8)
Hypotension and Seizure in Preterm Infants and Neonates: Avoid rapid injection in the neonatal population. (5.9)
Neonatal Sedation and Withdrawal Syndrome: Receiving Midazolam in 0.9% Sodium Chloride Injection during pregnancy can result in neonatal sedation and/or neonatal withdrawal. (5.10, 8.1)
Pediatric Neurotoxicity: In developing animals, exposures greater than 3 hours cause neurotoxicity. Weigh benefits against potential risks when considering elective procedures in children under 3 years old. (5.11)
2 Dosage and Administration
- For intravenous injection only. Avoid intra-arterial injection or extravasation. (2.1)
- Individualize dosing and titrate to desired clinical response, taking into account patient age, clinical status, and concomitant use of other CNS depressants. (2.1)
- See Full Prescribing Information for complete dosage and administration information. (2)
3 Dosage Forms and Strengths
Midazolam in 0.9% Sodium Chloride Injection, 50 mg per 50 mL (1 mg per mL) and 100 mg per 100 mL (1 mg per mL), is a clear, colorless solution supplied in single-dose bags with an aluminum overwrap.
5.11 Pediatric Neurotoxicity
Published animal studies demonstrate that the administration of anesthetic and sedation drugs that block NMDA receptors and/or potentiate GABA activity increase neuronal apoptosis in the developing brain and result in long-term cognitive deficits when used for longer than 3 hours. The clinical significance of these findings is not clear. However, based on the available data, the window of vulnerability to these changes is believed to correlate with exposures in the third trimester of gestation through the first several months of life, but may extend out to approximately three years of age in humans [see Nonclinical Pharmacology (13.2)].
Some published studies in children suggest that similar deficits may occur after repeated or prolonged exposures to anesthetic agents early in life and may result in adverse cognitive or behavioral effects. These studies have substantial limitations, and it is not clear if the observed effects are due to the anesthetic/sedation drug administration or other factors such as the surgery or underlying illness.
Anesthetic and sedation drugs are a necessary part of the care of children needing surgery, other procedures, or tests that cannot be delayed, and no specific medications have been shown to be safer than any other. Decisions regarding the timing of any elective procedures requiring anesthesia should take into consideration the benefits of the procedure weighed against the potential risks.
8 Use in Specific Populations
Lactation: A lactating woman may pump and discard breast milk for 4 to 8 hours after treatment with midazolam (8.2).
2.2 General Dosing Information
Individualize dosing and titrate to desired clinical response, taking into account patient age, clinical status, and concomitant use of other CNS depressants. Titrate to effect with multiple small doses while continuously monitoring respiratory and cardiac function (i.e., pulse oximetry). To minimize the potential for oversedation, allow adequate time between doses to achieve peak central nervous system effect (3 to 5 minutes).
5.8 Impaired Cognitive Function
Midazolam is associated with a high incidence of partial or complete impairment of recall for the next several hours. The decision as to when patients who have received injectable midazolam, particularly on an outpatient basis, may again engage in activities requiring complete mental alertness, operate hazardous machinery or drive a motor vehicle must be individualized. Gross tests of recovery from the effects of midazolam [see Clinical Pharmacology (12.3)] cannot be relied upon to predict reaction time under stress. It is recommended that no patient operate hazardous machinery or a motor vehicle until the effects of the drug, such as drowsiness, have subsided or until one full day after anesthesia and surgery, whichever is longer. For pediatric patients, particular care should be taken to assure safe ambulation.
5.4 Risk of Paradoxical Behavior
Reactions such as agitation, involuntary movements (including tonic/clonic movements and muscle tremor), hyperactivity and combativeness have been reported in both adult and pediatric patients. These reactions may be due to inadequate or excessive dosing or improper administration of midazolam; however, consideration should be given to the possibility of cerebral hypoxia or true paradoxical reactions. Should such reactions occur, the response to each dose of midazolam and all other drugs, including local anesthetics, should be evaluated before proceeding. Reversal of such responses with flumazenil has been reported in pediatric patients.
7.2 Cytochrome P450 3a4 Inhibitors
Concomitant administration with drugs that are known to inhibit the P450-3A4 enzyme system, such as cimetidine (not ranitidine), erythromycin, diltiazem, verapamil, ketoconazole and itraconazole, may result in prolonged sedation due to a decrease in plasma clearance of midazolam.
The effect of single oral doses of 800 mg cimetidine and 300 mg ranitidine on steady-state concentrations of midazolam was examined in a randomized crossover study (n=8). Cimetidine increased the mean midazolam steady-state concentration from 57 to 71 ng/mL. Ranitidine increased the mean steady-state concentration to 62 ng/mL. No change in choice reaction time or sedation index was detected after dosing with the H2 receptor antagonists.
In a placebo-controlled study, erythromycin administered as a 500 mg dose, three times a day, for 1 week (n=6), reduced the clearance of midazolam following a single 0.5 mg/kg intravenous dose. The half-life was approximately doubled.
The effects of diltiazem (60 mg three times a day) and verapamil (80 mg three times a day) on the pharmacokinetics and pharmacodynamics of oral midazolam were investigated in a three-way crossover study (n=9). The half-life of midazolam increased from 5 to 7 hours when midazolam was taken in conjunction with verapamil or diltiazem. No interaction was observed in healthy subjects between midazolam and nifedipine.
16 How Supplied/storage and Handling
Midazolam in 0.9% Sodium Chloride Injection is a clear, colorless solution supplied in single-dose bags with an aluminum overwrap available as:
|
*Partial fill container 50 mL volume in 100 mL container |
||
| NDC | Midazolam in 0.9% Sodium Chloride Injection | Package Factor |
| (1 mg per mL) | ||
| 25021-688-82 | *50 mg per 50 mL Single-Dose Bag | 10 bags per carton |
| 25021-688-87 | 100 mg per 100 mL Single-Dose Bag | 20 bags per carton |
Store at 20° to 25°C (68° to 77°F); excursions permitted between 15° and 30°C (59° and 86°F). [See USP Controlled Room Temperature.] Individual containers may be used up to 48 hours after initial penetration.
Do not freeze.
Discard unused portion.
Sterile, Nonpyrogenic, Preservative-free, PVC-free, DEHP-free.
The container closure is not made with natural rubber latex.
5.7 Risk of Intra Arterial Injection
There have been limited reports of intra-arterial injection of midazolam. Adverse events have included local reactions, as well as isolated reports of seizure activity in which no clear causal relationship was established. Precautions against unintended intra-arterial injection should be taken. Extravasation should also be avoided.
The safety and efficacy of Midazolam in 0.9% Sodium Chloride Injection following nonintravenous routes of administration have not been established. Midazolam in 0.9% Sodium Chloride Injection should only be administered intravenously.
7.8 Drug/laboratory Test Interactions
Midazolam has not been shown to interfere with results obtained in clinical laboratory tests.
13.2 Animal Toxicology And/or Pharmacology
Published studies in animals demonstrate that the use of anesthetic agents during the period of rapid brain growth or synaptogenesis results in widespread neuronal and oligodendrocyte cell loss in the developing brain and alterations in synaptic morphology and neurogenesis. Based on comparisons across species, the window of vulnerability to these changes is believed to correlate with exposures in the third trimester through the first several months of life, but may extend out to approximately 3 years of age in humans.
In primates, exposure to 3 hours of an anesthetic regimen that produced a light surgical plane of anesthesia did not increase neuronal cell loss, however, treatment regimens of 5 hours or longer increased neuronal cell loss. Data in rodents and in primates suggest that the neuronal and oligodendrocyte cell losses are associated with subtle but prolonged cognitive deficits in learning and memory. The clinical significance of these nonclinical findings is not known, and health care providers should balance the benefits of appropriate anesthesia in neonates and young children who require procedures against the potential risks suggested by the nonclinical data [see Warnings and Precautions (5.8)].
5.6 Debilitation and Comorbid Considerations
Higher risk adult and pediatric surgical patients, elderly patients and debilitated adult and pediatric patients require lower dosages, whether or not concomitant sedating medications have been administered. Adult or pediatric patients with COPD are unusually sensitive to the respiratory depressant effect of midazolam. Pediatric and adult patients undergoing procedures involving the upper airway such as upper endoscopy or dental care, are particularly vulnerable to episodes of desaturation and hypoventilation due to partial airway obstruction. Adult and pediatric patients with chronic renal failure and patients with congestive heart failure eliminate midazolam more slowly [see Clinical Pharmacology (12.3)]. Because elderly patients frequently have inefficient function of one or more organ systems and because dosage requirements have been shown to decrease with age, reduced initial dosage of midazolam is recommended, and the possibility of profound and/or prolonged effect should be considered.
Do not administer Midazolam in 0.9% Sodium Chloride Injection to adult or pediatric patients in shock or coma, or in acute alcohol intoxication with depression of vital signs. Particular care should be exercised in the use of intravenous midazolam in adult or pediatric patients with uncompensated acute illnesses, such as severe fluid or electrolyte disturbances.
7.7 Other Drugs Used in the Surgical Setting
No significant adverse interactions with commonly used premedications or drugs used during anesthesia and surgery (including atropine, scopolamine, glycopyrrolate, diazepam, hydroxyzine, d-tubocurarine, succinylcholine and other nondepolarizing muscle relaxants) or topical local anesthetics (including lidocaine, dyclonine HCl and Cetacaine) have been observed in adults or pediatric patients. In neonates, however, severe hypotension has been reported with concomitant administration of fentanyl. This effect has been observed in neonates on an infusion of midazolam who received a rapid injection of fentanyl and in patients on an infusion of fentanyl who have received a rapid injection of midazolam.
5.10 Neonatal Sedation and Withdrawal Syndrome
Receiving Midazolam in 0.9% Sodium Chloride Injection late in pregnancy can result in sedation (respiratory depression, lethargy, hypotonia) and/or withdrawal symptoms (hyperreflexia, irritability, restlessness, tremors, inconsolable crying, and feeding difficulties) in the neonate. Monitor neonates exposed to Midazolam in 0.9% Sodium Chloride Injection during pregnancy or labor for signs of sedation and manage these neonates accordingly [see Use in Specific Populations (8.1)].
5.3 Risk of Cardiorespiratory Adverse Reactions
Serious cardiorespiratory adverse reactions have occurred after administration of midazolam. These have included respiratory depression, airway obstruction, oxygen desaturation, apnea, respiratory arrest and/or cardiac arrest, sometimes resulting in death or permanent neurologic injury. There have also been rare reports of hypotensive episodes requiring treatment during or after diagnostic or surgical manipulations particularly in adult or pediatric patients with hemodynamic instability. Hypotension occurred more frequently in the sedation studies in patients premedicated with an opioid. Excessive single doses or rapid intravenous administration may result in respiratory depression, airway obstruction and/or arrest. When used for sedation/anxiolysis/amnesia, midazolam should always be titrated slowly in adult or pediatric patients. Adverse hemodynamic events have been reported in pediatric patients with cardiovascular instability; rapid intravenous administration should also be avoided in this population. Continuously monitor patients for early signs of hypoventilation, airway obstruction, and apnea using capnography, pulse oximetry, and clinical assessment [see Dosage and Administration (2.2)].
7.1 Opioid Analgesics and Other Sedative Hypnotics
The concomitant use of benzodiazepines and opioids increases the risk of respiratory depression because of actions at different receptor sites in the CNS that control respiration. Benzodiazepines interact at GABAA sites and opioids interact primarily at mu receptors. When benzodiazepines and opioids are combined, the potential for benzodiazepines to significantly worsen opioid-related respiratory depression exists. Monitor patients closely for respiratory depression and sedation.
The sedative effect of intravenous midazolam is accentuated by any concomitantly administered medication which depresses the central nervous system, particularly opioids (e.g., morphine, meperidine and fentanyl) and also secobarbital and droperidol. Consequently, the dosage of midazolam should be adjusted according to the type and amount of concomitant medications administered and the desired clinical response [see Dosage and Administration (2)].
2.1 Important Dosage and Administration Instructions
- Midazolam in 0.9% Sodium Chloride Injection should only be administered intravenously. Avoid intra-arterial injection or extravasation [see Warnings and Precautions (5.7)].
- Only personnel trained in the administration of procedural sedation, and not involved in the conduct of the diagnostic or therapeutic procedure, should administer Midazolam in 0.9% Sodium Chloride Injection.
- Administering personnel must be trained in the detection and management of airway obstruction, hypoventilation, and apnea, including the maintenance of a patent airway, supportive ventilation, and cardiovascular resuscitation.
- Supplemental oxygen, resuscitative drugs, and age‑ and size-appropriate equipment for bag/valve/mask assisted ventilation must be immediately available during administration of Midazolam in 0.9% Sodium Chloride Injection. A benzodiazepine reversal agent should be immediately available.
- Continuously monitor vital signs during sedation and through the recovery period [see Warnings and Precautions (5.1)].
Midazolam must never be used without individualization of dosage particularly when used with other medications capable of producing central nervous system depression [see Warnings and Precautions (5.2)].
Midazolam in 0.9% Sodium Chloride Injection can cause respiratory depression. It is a potent sedative agent that requires slow administration and individualization of dosage. Excessive single doses or rapid intravenous administration may result in respiratory depression, airway obstruction and/or arrest. Continuously monitor patients for early signs of hypoventilation, airway obstruction, and apnea using capnography, pulse oximetry, and clinical assessment [see Warnings and Precautions (5.3)].
Reactions such as agitation, involuntary movements, hyperactivity and combativeness have been reported in adult and pediatric patients. Should such reactions occur, the response to each dose of midazolam and all other drugs, including local anesthetics, should be evaluated before proceeding. Reversal of such responses with flumazenil has been reported in pediatric patients [see Warnings and Precautions (5.4)].
Visually inspect parenteral drug products for particulate matter and discoloration prior to administration, whenever solution and container permit. If solution is discolored or particulate matter is present, do not use.
5.1 Personnel and Equipment for Monitoring and Resuscitation
- Prior to the intravenous administration of midazolam in any dose, ensure the immediate availability of oxygen, resuscitative drugs, age- and size-appropriate equipment for bag/valve/mask ventilation and intubation, and skilled personnel for the maintenance of a patent airway and support of ventilation.
- Only personnel trained in the administration of procedural sedation, and not involved in the conduct of the diagnostic or therapeutic procedure, should administer Midazolam in 0.9% Sodium Chloride Injection.
- Administering personnel must be trained in the detection and management of airway obstruction, hypoventilation, and apnea, including the maintenance of a patent airway, supportive ventilation, and cardiovascular resuscitation.
- Continuously monitor patients for early signs of hypoventilation, airway obstruction, or apnea, with means readily available (e.g., pulse oximetry). Hypoventilation, airway obstruction, and apnea can lead to hypoxia and/or cardiac arrest unless effective countermeasures are taken immediately.
- A benzodiazepine reversal agent (i.e., flumazenil) should be immediately available during administration of Midazolam in 0.9% Sodium Chloride Injection.
- Continuously monitor vital signs during the recovery period. Because intravenous midazolam can depress respiration [see Clinical Pharmacology (12)], especially when used concomitantly with opioid agonists and other sedatives [see Dosage and Administration (2)], it should be used for sedation/anxiolysis/amnesia only in the presence of personnel skilled in early detection of hypoventilation, maintaining a patent airway, and supporting ventilation.
5.9 Risk of Hypotension and Seizure in Preterm Infants and Neonates
Rapid injection should be avoided in the neonatal population. Midazolam administered rapidly as an intravenous injection (i.e., less than 2 minutes) has been associated with severe hypotension in neonates, particularly when the patient has also received fentanyl. Likewise, severe hypotension has been observed in neonates receiving a continuous infusion of midazolam who then receive a rapid intravenous injection of fentanyl. Seizures have been reported in several neonates following rapid intravenous administration.
The neonate also has reduced and/or immature organ function and is also vulnerable to profound and/or prolonged respiratory effects of midazolam.
5.12 Risk of Increased Intraocular Pressure in Patients With Glaucoma
Benzodiazepines, including Midazolam in 0.9% Sodium Chloride Injection, can increase intraocular pressure in patients with glaucoma. Measurements of intraocular pressure in patients without eye disease show a moderate lowering following induction with midazolam. Midazolam in 0.9% Sodium Chloride Injection may be used in patients with open-angle glaucoma only if they are receiving appropriate therapy. Patients with open-angle glaucoma may need to have their ophthalmologic status evaluated following treatment with Midazolam in 0.9% Sodium Chloride Injection. Midazolam in 0.9% Sodium Chloride Injection is contraindicated in patients with narrow-angle glaucoma.
2.4 Safe Discontinuation of Midazolam in 0.9% Sodium Chloride Injection After Long Term Use
If Midazolam in 0.9% Sodium Chloride Injection is administered long-term (for several days to weeks), do not abruptly discontinue. Gradually taper the dosage in physically-dependent patients using a tapering schedule that is individualized to the patient [see Warnings and Precautions (5.5)].
5.5 Risk of Dependence and Withdrawal With Long Term Use of Midazolam in 0.9% Sodium Chloride Injection
The continued use of benzodiazepines for several days to weeks may lead to clinically significant physical dependence. If used for long-term use (i.e., for several days to weeks), abrupt discontinuation or rapid dosage reduction of midazolam, or administration of flumazenil, a benzodiazepine antagonist, may precipitate acute withdrawal reactions, including seizures, which can be life-threatening.
Patients at an increased risk of withdrawal adverse reactions after benzodiazepine discontinuation or rapid dosage reduction include those who take higher dosages (i.e., higher and/or more frequent doses) and those who have had longer durations of use.
After extended therapy, do not abruptly discontinue Midazolam in 0.9% Sodium Chloride Injection. When discontinuing midazolam in a physically-dependent patient, gradually taper the dosage using a tapering schedule that is individualized to the patient [see Dosage and Administration (2.3), Dependence (9.3)].
5.2 Risks From Concomitant Use With Opioids, Other Sedative Hypnotics, Or Other Central Nervous System Depressants
Concomitant use of benzodiazepines and opioids may result in profound sedation, respiratory depression, coma, and death. Monitor patients for respiratory depression and sedation [see Warnings and Precautions (5.2) and Drug Interactions (7.1)].
Titrate the dose of Midazolam in 0.9% Sodium Chloride Injection when administered with opioid analgesics and sedative-hypnotics to the desired clinical response.
Continuously monitor sedated patients for hypotension, airway obstruction, hypoventilation, apnea, and oxygen desaturation. These cardiorespiratory effects may be more likely to occur in patients with obstructive sleep apnea, the elderly, and ASA‑PS III or IV patients.
Concomitant use of barbiturates, alcohol, or other central nervous system depressants may increase the risk of hypoventilation, airway obstruction, desaturation, or apnea and may contribute to profound and/or prolonged drug effect. Narcotic premedication also depresses the ventilatory response to carbon dioxide stimulation.
Warning: Personnel and Equipment for Monitoring and Resuscitation and Risks From Concomitant Use With Opioid Analgesics and Other Sedative Hypnotics
Personnel and Equipment for Monitoring and Resuscitation
- Only personnel trained in the administration of procedural sedation, and not involved in the conduct of the diagnostic or therapeutic procedure, should administer Midazolam in 0.9% Sodium Chloride Injection [see Dosage and Administration (2.1), Warnings and Precautions (5.1)].
- Administering personnel must be trained in the detection and management of airway obstruction, hypoventilation, and apnea, including the maintenance of a patent airway, supportive ventilation, and cardiovascular resuscitation [see Dosage and Administration (2.1) , Warnings and Precautions (5.1)].
- Resuscitative drugs, and age- and size-appropriate equipment for bag/valve/mask assisted ventilation must be immediately available during administration of Midazolam in 0.9% Sodium Chloride Injection [see Dosage and Administration (2.1) , Warnings and Precautions (5.1)].
- Continuously monitor vital signs during sedation and during the recovery period [see Dosage and Administration (2.1) , Warnings and Precautions (5.1)].
Risks from Concomitant Use with Opioid Analgesics and Other Sedative Hypnotics
Concomitant use of benzodiazepines, including Midazolam in 0.9% Sodium Chloride Injection, and opioids may result in profound sedation, respiratory depression, coma, and death. Continuously monitor patients for respiratory depression and depth of sedation [see Warnings and Precautions (5.2) and Drug Interaction (7.1)].
Structured Label Content
Section 42229-5 (42229-5)
Adults and Pediatrics:
Sedation guidelines recommend a careful presedation history to determine how a patient's underlying medical conditions or concomitant medications might affect their response to sedation/analgesia as well as a physical examination including a focused examination of the airway for abnormalities. Further recommendations include appropriate presedation fasting.
Section 51945-4 (51945-4)
PACKAGE LABEL – PRINCIPAL DISPLAY PANEL – Bag Label
NDC 25021-688-82
Rx only
Midazolam in 0.9% Sodium Chloride Injection
50 mg per 50 mL
(1 mg per mL)
50 mL Single-Dose Container
High Alert Medication
For Intravenous Use Only
Do Not Dilute
9.2 Abuse
Midazolam in 0.9% Sodium Chloride Injection contains the benzodiazepine, midazolam. Benzodiazepines are a class of sedative drugs with a known potential for abuse. Abuse is the intentional, non-therapeutic use of a drug, even once, for its desirable psychological or physiological effects.
Misuse is the intentional use, for therapeutic purposes, of a drug by an individual in a way other than prescribed by a health care provider or for whom it was not prescribed. Drug addiction is a cluster of behavioral, cognitive, and physiological phenomena that may include a strong desire to take the drug, difficulties in controlling drug use (e.g., continuing drug use despite harmful consequences, giving a higher priority to drug use than other activities and obligations), and possible tolerance or physical dependence. Both abuse and misuse may lead to addiction. Midazolam was actively self-administered in primate models used to assess the positive reinforcing effects of psychoactive drugs. Midazolam produced physical dependence of a mild to moderate intensity in cynomolgus monkeys after 5 to 10 weeks of administration. Available data concerning the drug abuse and dependence potential of midazolam suggest that its abuse potential is at least equivalent to that of diazepam.
7.5 Halothane
The intravenous administration of midazolam decreases the minimum alveolar concentration (MAC) of halothane required for general anesthesia. This decrease correlates with the dose of midazolam administered; no similar studies have been carried out in pediatric patients but there is no scientific reason to expect that pediatric patients would respond differently than adults.
11 Description (11 DESCRIPTION)
Midazolam in 0.9% Sodium Chloride Injection is a benzodiazepine available as a sterile, preservative-free, nonpyrogenic solution of midazolam and sodium chloride in water for injection for intravenous use. Each single-dose bag of Midazolam in 0.9% Sodium Chloride Injection contains either 50 mg per 50 mL (1 mg per mL) or 100 mg per 100 mL (1 mg per mL) of midazolam and 9 mg/mL of sodium chloride in water for injection. Midazolam in 0.9% Sodium Chloride Injection may contain hydrochloric acid and/or sodium hydroxide for pH adjustment. The pH is approximately 2.5 to 3.5.
Midazolam is a white or yellowish powder, practically insoluble in water, Chemically, midazolam is 8-chloro-6-(2-fluorophenyl)-1-methyl-4H-imidazo[1,5-a][1,4]-benzodiazepine. Midazolam has the empirical formula C18 H13ClFN3, a calculated molecular weight of 325.8 and the following structural formula:
7.3 Saquinavir
In a placebo-controlled study, saquinavir administered as a 1200 mg dose, tid, for 5 days (n=12), a 56% reduction in the clearance of midazolam following a single 0.05 mg/kg intravenous dose was observed. The half-life was approximately doubled.
7.4 Thiopental
A moderate reduction in induction dosage requirements of thiopental (about 15%) has been noted following use of intramuscular midazolam for premedication in adults.
9.3 Dependence
Midazolam may produce physical dependence after long-term use. Physical dependence is a state that develops as a result of physiological adaptation in response to repeated drug use, manifested by withdrawal signs and symptoms after abrupt discontinuation or a significant dose reduction of a drug. If Midazolam in 0.9% Sodium Chloride Injection is administered long-term (i.e., for several days to weeks), abrupt discontinuation or rapid dosage reduction, or administration of flumazenil, a benzodiazepine antagonist, may precipitate acute withdrawal reactions, including seizures, which can be life-threatening. Patients at an increased risk of withdrawal adverse reactions after benzodiazepine discontinuation or rapid dosage reduction include those who take higher dosages (i.e., higher and/or more frequent doses) and those who have had longer durations of use [see Warnings and Precautions (5.5)].
To reduce the risk of withdrawal reactions, after extended therapy, do not abruptly discontinue Midazolam in 0.9% Sodium Chloride Injection. Gradually taper the dosage using a tapering schedule that is individualized to the patient.
7.6 Pancuronium
Although the possibility of minor interactive effects has not been fully studied, midazolam and pancuronium have been used together in patients without noting clinically significant changes in dosage, onset or duration in adults. Midazolam does not protect against the characteristic circulatory changes noted after administration of succinylcholine or pancuronium and does not protect against the increased intracranial pressure noted following administration of succinylcholine. Midazolam does not cause a clinically significant change in dosage, onset or duration of a single intubating dose of succinylcholine; no similar studies have been carried out in pediatric patients but there is no scientific reason to expect that pediatric patients would respond differently than adults.
8.4 Pediatric Use
The safety and efficacy of midazolam for sedation/anxiolysis/amnesia following continuous infusion have been established in pediatric and neonatal patients. UNLIKE ADULT PATIENTS, PEDIATRIC PATIENTS GENERALLY RECEIVE INCREMENTS OF MIDAZOLAM ON A MG/KG BASIS. As a group, pediatric patients generally require higher dosages of midazolam (mg/kg) than do adults. Younger (less than six years) pediatric patients may require higher dosages (mg/kg) than older pediatric patients, and may require closer monitoring. In obese PEDIATRIC PATIENTS, the dose should be calculated based on ideal body weight. When midazolam is given in conjunction with opioids or other sedatives, the potential for respiratory depression, airway obstruction, or hypoventilation is increased. The health care practitioner who uses this medication in pediatric patients should be aware of and follow accepted professional guidelines for pediatric sedation appropriate to their situation.
Midazolam should not be administered by rapid injection in the neonatal population. Severe hypotension and seizures have been reported following rapid intravenous administration, particularly, with concomitant use of fentanyl.
8.5 Geriatric Use
Because geriatric patients may have altered drug distribution and diminished hepatic and/or renal function, reduced doses of midazolam are recommended. Doses of Midazolam in 0.9% Sodium Chloride Injection should be decreased for elderly and for debilitated patients [see Warnings and Precautions (5.6) and Dosage and Administration (2)] and subjects over 70 years of age may be particularly sensitive. These patients will also probably take longer to recover completely after midazolam administration for the induction of anesthesia. Administration of intravenous midazolam to elderly and/or high-risk surgical patients has been associated with rare reports of death under circumstances compatible with cardiorespiratory depression. In most of these cases, the patients also received other central nervous system depressants capable of depressing respiration, especially opioids [see Dosage and Administration (2)].
Midazolam is known to be substantially excreted by the kidney, and the risk of adverse reactions to this drug may be greater in patients with impaired renal function. Because elderly patients are more likely to have decreased renal function, care should be taken in dose selection, and it may be useful to monitor renal function.
4 Contraindications (4 CONTRAINDICATIONS)
Midazolam in 0.9% Sodium Chloride Injection is contraindicated in patients with:
- Known hypersensitivity to midazolam
- Acute narrow-angle glaucoma
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following serious adverse reactions are discussed in greater detail in other sections:
- Cardiorespiratory Adverse Reactions [see Warnings and Precautions (5.3)]
- Paradoxical Behavior [see Warnings and Precautions (5.4)]
- Dependence and Withdrawal [see Warnings and Precautions (5.5)]
- Impaired Cognitive Function [see Warnings and Precautions (5.8)]
- Hypotension and Seizure in Preterm Infants and Neonates [see Warnings and Precautions (5.9)]
- Neonatal Sedation and Withdrawal Syndrome [see Warnings and Precautions (5.10), Use in Specific Populations (8.1)]
- Pediatric Neurotoxicity [see Warnings and Precautions (5.11)]
The following adverse reactions have been identified from literature or postmarketing reports of midazolam. Because some of these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Fluctuations in vital signs were the most frequently seen findings following parenteral administration of midazolam in adults and included decreased tidal volume and/or respiratory rate decrease (23.3% of patients following intravenous administration) and apnea (15.4% of patients following intravenous administration), as well as variations in blood pressure and pulse rate. The majority of serious adverse reactions, particularly those associated with oxygenation and ventilation, have been reported when midazolam is administered with other medications capable of depressing the central nervous system. The incidence of such events is higher in patients undergoing procedures involving the airway without the protective effect of an endotracheal tube, (e.g., upper endoscopy and dental procedures).
7 Drug Interactions (7 DRUG INTERACTIONS)
12.2 Pharmacodynamics
The effects of midazolam on the CNS are dependent on the dose administered, the route of administration, and the presence or absence of other medications.
12.3 Pharmacokinetics
Midazolam's activity is primarily due to the parent drug. Elimination of the parent drug takes place via hepatic metabolism of midazolam to hydroxylated metabolites that are conjugated and excreted in the urine. Six single-dose pharmacokinetic studies involving healthy adults yield pharmacokinetic parameters for midazolam in the following ranges: volume of distribution (Vd), 1.0 to 3.1 L/kg; elimination half-life, 1.8 to 6.4 hours (mean approximately 3 hours); total clearance (Cl), 0.25 to 0.54 L/hr/kg. In a parallel group study, there was no difference in the clearance, in subjects administered 0.15 mg/kg (n=4) and 0.30 mg/kg (n=4) intravenous doses indicating linear kinetics. The clearance was successively reduced by approximately 30% at doses of 0.45 mg/kg (n=4) and 0.6 mg/kg (n=5) indicating non-linear kinetics in this dose range.
1 Indications and Usage (1 INDICATIONS AND USAGE)
Midazolam in 0.9% Sodium Chloride Injection is indicated:
- Continuous intravenous infusion for sedation of intubated and mechanically ventilated adult, pediatric, and neonatal patients as a component of anesthesia or during treatment in a critical care setting.
12.1 Mechanism of Action
Midazolam is a short-acting benzodiazepine central nervous system (CNS) depressant.
9.1 Controlled Substance
Midazolam in 0.9% Sodium Chloride Injection contains midazolam, a Schedule IV controlled substance.
2.3 Dosing Recommendations
Table 1 provides dosing recommendations for adult, pediatric, and neonatal patients.
| ADULT PATIENTS | If a loading dose is necessary to rapidly initiate sedation, 0.01 mg/kg to 0.05 mg/kg (approximately 0.5 mg to 4 mg for a typical adult) may be given slowly or infused over several minutes. This dose may be repeated at 10 to 15 minute intervals until adequate sedation is achieved. For maintenance of sedation, the usual initial infusion rate is 0.02 mg/kg/hr to 0.10 mg/kg/hr (1 mg/hr to 7 mg/hr). Higher loading or maintenance infusion rates may occasionally be required in some patients. Use the lowest recommended doses in patients with residual effects from anesthetic drugs, or in those concurrently receiving other sedatives or opioids. Individual response to midazolam is variable. Titrate the infusion rate to the desired level of sedation, taking into account the patient's age, clinical status and current medications. In general, midazolam should be infused at the lowest rate that produces the desired level of sedation. Assess sedation at regular intervals and adjust the midazolam infusion rate. Finding the minimum effective infusion rate decreases the potential accumulation of midazolam and provides for the most rapid recovery once the infusion is terminated. |
| PEDIATRIC PATIENTS | UNLIKE ADULT PATIENTS, PEDIATRIC PATIENTS GENERALLY RECEIVE INCREMENTS OF MIDAZOLAM ON A MG/KG BASIS. As a group, pediatric patients generally require higher dosages of midazolam (mg/kg) than adults. Younger (less than six years) pediatric patients may require higher dosages (mg/kg) than older pediatric patients. In obese pediatric patients, calculate the dose based on ideal body weight. Titrate the dose to the desired level of sedation. Assess for desired level of sedation and vital signs at regular intervals. |
|
PRETERM AND TERM
NEONATAL PATIENTS |
Based on pharmacokinetic parameters and reported clinical experience in preterm and term neonates WHOSE TRACHEA WAS INTUBATED, initiate continuous intravenous infusions of Midazolam in 0.9% Sodium Chloride Injection at a rate of 0.03 mg/kg/hr (0.5 mcg/kg/min) in neonates <32 weeks and 0.06 mg/kg/hr (1 mcg/kg/min) in neonates >32 weeks. Intravenous loading doses should not be used in neonates, rather the infusion may be run more rapidly for the first several hours to establish therapeutic plasma levels. Frequently assess the rate of infusion, particularly after the first 24 hours so as to administer the lowest possible effective dose and reduce the potential for drug accumulation. Hypotension may be observed in patients who are critically ill and in preterm and term infants, particularly those receiving fentanyl and/or when midazolam is administered rapidly. When sedating preterm and former preterm neonates WHOSE TRACHEA WAS NOT INTUBATED, monitor respiratory parameters due to an increased risk of apnea. |
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
Cardiorespiratory Adverse Reactions: Serious cardiorespiratory adverse reactions have occurred, sometimes resulting in death or permanent neurologic injury. (5.3)
Paradoxical Behavior: Agitation, involuntary movements (including tonic/clonic movements and muscle tremor), hyperactivity and combativeness have been reported in both adult and pediatric patients. (5.4)
Dependence and Withdrawal with Long-Term Use: Use for several days to weeks may lead to physical dependence to midazolam. Do not abruptly discontinue midazolam. Gradually taper the dosage using a tapering schedule that is individualized to the patient. (5.5)
Debilitation and Comorbid Considerations: Higher risk adult and pediatric surgical patients, elderly patients and debilitated adult and pediatric patients require lower dosages, whether or not concomitant sedating medications have been administered. (5.6)
Risk of Intra-Arterial Injection: There have been limited reports of intra-arterial injection of midazolam. Adverse events have included local reactions, as well as isolated reports of seizure activity in which no clear causal relationship was established. (5.7)
Impaired Cognitive Function: Because of partial or complete impairment of recall, patients should not operate hazardous machinery or a motor vehicle until drug effects have subsided. (5.8)
Hypotension and Seizure in Preterm Infants and Neonates: Avoid rapid injection in the neonatal population. (5.9)
Neonatal Sedation and Withdrawal Syndrome: Receiving Midazolam in 0.9% Sodium Chloride Injection during pregnancy can result in neonatal sedation and/or neonatal withdrawal. (5.10, 8.1)
Pediatric Neurotoxicity: In developing animals, exposures greater than 3 hours cause neurotoxicity. Weigh benefits against potential risks when considering elective procedures in children under 3 years old. (5.11)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
- For intravenous injection only. Avoid intra-arterial injection or extravasation. (2.1)
- Individualize dosing and titrate to desired clinical response, taking into account patient age, clinical status, and concomitant use of other CNS depressants. (2.1)
- See Full Prescribing Information for complete dosage and administration information. (2)
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Midazolam in 0.9% Sodium Chloride Injection, 50 mg per 50 mL (1 mg per mL) and 100 mg per 100 mL (1 mg per mL), is a clear, colorless solution supplied in single-dose bags with an aluminum overwrap.
5.11 Pediatric Neurotoxicity
Published animal studies demonstrate that the administration of anesthetic and sedation drugs that block NMDA receptors and/or potentiate GABA activity increase neuronal apoptosis in the developing brain and result in long-term cognitive deficits when used for longer than 3 hours. The clinical significance of these findings is not clear. However, based on the available data, the window of vulnerability to these changes is believed to correlate with exposures in the third trimester of gestation through the first several months of life, but may extend out to approximately three years of age in humans [see Nonclinical Pharmacology (13.2)].
Some published studies in children suggest that similar deficits may occur after repeated or prolonged exposures to anesthetic agents early in life and may result in adverse cognitive or behavioral effects. These studies have substantial limitations, and it is not clear if the observed effects are due to the anesthetic/sedation drug administration or other factors such as the surgery or underlying illness.
Anesthetic and sedation drugs are a necessary part of the care of children needing surgery, other procedures, or tests that cannot be delayed, and no specific medications have been shown to be safer than any other. Decisions regarding the timing of any elective procedures requiring anesthesia should take into consideration the benefits of the procedure weighed against the potential risks.
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
Lactation: A lactating woman may pump and discard breast milk for 4 to 8 hours after treatment with midazolam (8.2).
2.2 General Dosing Information
Individualize dosing and titrate to desired clinical response, taking into account patient age, clinical status, and concomitant use of other CNS depressants. Titrate to effect with multiple small doses while continuously monitoring respiratory and cardiac function (i.e., pulse oximetry). To minimize the potential for oversedation, allow adequate time between doses to achieve peak central nervous system effect (3 to 5 minutes).
5.8 Impaired Cognitive Function
Midazolam is associated with a high incidence of partial or complete impairment of recall for the next several hours. The decision as to when patients who have received injectable midazolam, particularly on an outpatient basis, may again engage in activities requiring complete mental alertness, operate hazardous machinery or drive a motor vehicle must be individualized. Gross tests of recovery from the effects of midazolam [see Clinical Pharmacology (12.3)] cannot be relied upon to predict reaction time under stress. It is recommended that no patient operate hazardous machinery or a motor vehicle until the effects of the drug, such as drowsiness, have subsided or until one full day after anesthesia and surgery, whichever is longer. For pediatric patients, particular care should be taken to assure safe ambulation.
5.4 Risk of Paradoxical Behavior
Reactions such as agitation, involuntary movements (including tonic/clonic movements and muscle tremor), hyperactivity and combativeness have been reported in both adult and pediatric patients. These reactions may be due to inadequate or excessive dosing or improper administration of midazolam; however, consideration should be given to the possibility of cerebral hypoxia or true paradoxical reactions. Should such reactions occur, the response to each dose of midazolam and all other drugs, including local anesthetics, should be evaluated before proceeding. Reversal of such responses with flumazenil has been reported in pediatric patients.
7.2 Cytochrome P450 3a4 Inhibitors (7.2 Cytochrome P450-3A4 Inhibitors)
Concomitant administration with drugs that are known to inhibit the P450-3A4 enzyme system, such as cimetidine (not ranitidine), erythromycin, diltiazem, verapamil, ketoconazole and itraconazole, may result in prolonged sedation due to a decrease in plasma clearance of midazolam.
The effect of single oral doses of 800 mg cimetidine and 300 mg ranitidine on steady-state concentrations of midazolam was examined in a randomized crossover study (n=8). Cimetidine increased the mean midazolam steady-state concentration from 57 to 71 ng/mL. Ranitidine increased the mean steady-state concentration to 62 ng/mL. No change in choice reaction time or sedation index was detected after dosing with the H2 receptor antagonists.
In a placebo-controlled study, erythromycin administered as a 500 mg dose, three times a day, for 1 week (n=6), reduced the clearance of midazolam following a single 0.5 mg/kg intravenous dose. The half-life was approximately doubled.
The effects of diltiazem (60 mg three times a day) and verapamil (80 mg three times a day) on the pharmacokinetics and pharmacodynamics of oral midazolam were investigated in a three-way crossover study (n=9). The half-life of midazolam increased from 5 to 7 hours when midazolam was taken in conjunction with verapamil or diltiazem. No interaction was observed in healthy subjects between midazolam and nifedipine.
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
Midazolam in 0.9% Sodium Chloride Injection is a clear, colorless solution supplied in single-dose bags with an aluminum overwrap available as:
|
*Partial fill container 50 mL volume in 100 mL container |
||
| NDC | Midazolam in 0.9% Sodium Chloride Injection | Package Factor |
| (1 mg per mL) | ||
| 25021-688-82 | *50 mg per 50 mL Single-Dose Bag | 10 bags per carton |
| 25021-688-87 | 100 mg per 100 mL Single-Dose Bag | 20 bags per carton |
Store at 20° to 25°C (68° to 77°F); excursions permitted between 15° and 30°C (59° and 86°F). [See USP Controlled Room Temperature.] Individual containers may be used up to 48 hours after initial penetration.
Do not freeze.
Discard unused portion.
Sterile, Nonpyrogenic, Preservative-free, PVC-free, DEHP-free.
The container closure is not made with natural rubber latex.
5.7 Risk of Intra Arterial Injection (5.7 Risk of Intra-Arterial Injection)
There have been limited reports of intra-arterial injection of midazolam. Adverse events have included local reactions, as well as isolated reports of seizure activity in which no clear causal relationship was established. Precautions against unintended intra-arterial injection should be taken. Extravasation should also be avoided.
The safety and efficacy of Midazolam in 0.9% Sodium Chloride Injection following nonintravenous routes of administration have not been established. Midazolam in 0.9% Sodium Chloride Injection should only be administered intravenously.
7.8 Drug/laboratory Test Interactions (7.8 Drug/Laboratory Test Interactions)
Midazolam has not been shown to interfere with results obtained in clinical laboratory tests.
13.2 Animal Toxicology And/or Pharmacology (13.2 Animal Toxicology and/or Pharmacology)
Published studies in animals demonstrate that the use of anesthetic agents during the period of rapid brain growth or synaptogenesis results in widespread neuronal and oligodendrocyte cell loss in the developing brain and alterations in synaptic morphology and neurogenesis. Based on comparisons across species, the window of vulnerability to these changes is believed to correlate with exposures in the third trimester through the first several months of life, but may extend out to approximately 3 years of age in humans.
In primates, exposure to 3 hours of an anesthetic regimen that produced a light surgical plane of anesthesia did not increase neuronal cell loss, however, treatment regimens of 5 hours or longer increased neuronal cell loss. Data in rodents and in primates suggest that the neuronal and oligodendrocyte cell losses are associated with subtle but prolonged cognitive deficits in learning and memory. The clinical significance of these nonclinical findings is not known, and health care providers should balance the benefits of appropriate anesthesia in neonates and young children who require procedures against the potential risks suggested by the nonclinical data [see Warnings and Precautions (5.8)].
5.6 Debilitation and Comorbid Considerations
Higher risk adult and pediatric surgical patients, elderly patients and debilitated adult and pediatric patients require lower dosages, whether or not concomitant sedating medications have been administered. Adult or pediatric patients with COPD are unusually sensitive to the respiratory depressant effect of midazolam. Pediatric and adult patients undergoing procedures involving the upper airway such as upper endoscopy or dental care, are particularly vulnerable to episodes of desaturation and hypoventilation due to partial airway obstruction. Adult and pediatric patients with chronic renal failure and patients with congestive heart failure eliminate midazolam more slowly [see Clinical Pharmacology (12.3)]. Because elderly patients frequently have inefficient function of one or more organ systems and because dosage requirements have been shown to decrease with age, reduced initial dosage of midazolam is recommended, and the possibility of profound and/or prolonged effect should be considered.
Do not administer Midazolam in 0.9% Sodium Chloride Injection to adult or pediatric patients in shock or coma, or in acute alcohol intoxication with depression of vital signs. Particular care should be exercised in the use of intravenous midazolam in adult or pediatric patients with uncompensated acute illnesses, such as severe fluid or electrolyte disturbances.
7.7 Other Drugs Used in the Surgical Setting
No significant adverse interactions with commonly used premedications or drugs used during anesthesia and surgery (including atropine, scopolamine, glycopyrrolate, diazepam, hydroxyzine, d-tubocurarine, succinylcholine and other nondepolarizing muscle relaxants) or topical local anesthetics (including lidocaine, dyclonine HCl and Cetacaine) have been observed in adults or pediatric patients. In neonates, however, severe hypotension has been reported with concomitant administration of fentanyl. This effect has been observed in neonates on an infusion of midazolam who received a rapid injection of fentanyl and in patients on an infusion of fentanyl who have received a rapid injection of midazolam.
5.10 Neonatal Sedation and Withdrawal Syndrome
Receiving Midazolam in 0.9% Sodium Chloride Injection late in pregnancy can result in sedation (respiratory depression, lethargy, hypotonia) and/or withdrawal symptoms (hyperreflexia, irritability, restlessness, tremors, inconsolable crying, and feeding difficulties) in the neonate. Monitor neonates exposed to Midazolam in 0.9% Sodium Chloride Injection during pregnancy or labor for signs of sedation and manage these neonates accordingly [see Use in Specific Populations (8.1)].
5.3 Risk of Cardiorespiratory Adverse Reactions
Serious cardiorespiratory adverse reactions have occurred after administration of midazolam. These have included respiratory depression, airway obstruction, oxygen desaturation, apnea, respiratory arrest and/or cardiac arrest, sometimes resulting in death or permanent neurologic injury. There have also been rare reports of hypotensive episodes requiring treatment during or after diagnostic or surgical manipulations particularly in adult or pediatric patients with hemodynamic instability. Hypotension occurred more frequently in the sedation studies in patients premedicated with an opioid. Excessive single doses or rapid intravenous administration may result in respiratory depression, airway obstruction and/or arrest. When used for sedation/anxiolysis/amnesia, midazolam should always be titrated slowly in adult or pediatric patients. Adverse hemodynamic events have been reported in pediatric patients with cardiovascular instability; rapid intravenous administration should also be avoided in this population. Continuously monitor patients for early signs of hypoventilation, airway obstruction, and apnea using capnography, pulse oximetry, and clinical assessment [see Dosage and Administration (2.2)].
7.1 Opioid Analgesics and Other Sedative Hypnotics
The concomitant use of benzodiazepines and opioids increases the risk of respiratory depression because of actions at different receptor sites in the CNS that control respiration. Benzodiazepines interact at GABAA sites and opioids interact primarily at mu receptors. When benzodiazepines and opioids are combined, the potential for benzodiazepines to significantly worsen opioid-related respiratory depression exists. Monitor patients closely for respiratory depression and sedation.
The sedative effect of intravenous midazolam is accentuated by any concomitantly administered medication which depresses the central nervous system, particularly opioids (e.g., morphine, meperidine and fentanyl) and also secobarbital and droperidol. Consequently, the dosage of midazolam should be adjusted according to the type and amount of concomitant medications administered and the desired clinical response [see Dosage and Administration (2)].
2.1 Important Dosage and Administration Instructions
- Midazolam in 0.9% Sodium Chloride Injection should only be administered intravenously. Avoid intra-arterial injection or extravasation [see Warnings and Precautions (5.7)].
- Only personnel trained in the administration of procedural sedation, and not involved in the conduct of the diagnostic or therapeutic procedure, should administer Midazolam in 0.9% Sodium Chloride Injection.
- Administering personnel must be trained in the detection and management of airway obstruction, hypoventilation, and apnea, including the maintenance of a patent airway, supportive ventilation, and cardiovascular resuscitation.
- Supplemental oxygen, resuscitative drugs, and age‑ and size-appropriate equipment for bag/valve/mask assisted ventilation must be immediately available during administration of Midazolam in 0.9% Sodium Chloride Injection. A benzodiazepine reversal agent should be immediately available.
- Continuously monitor vital signs during sedation and through the recovery period [see Warnings and Precautions (5.1)].
Midazolam must never be used without individualization of dosage particularly when used with other medications capable of producing central nervous system depression [see Warnings and Precautions (5.2)].
Midazolam in 0.9% Sodium Chloride Injection can cause respiratory depression. It is a potent sedative agent that requires slow administration and individualization of dosage. Excessive single doses or rapid intravenous administration may result in respiratory depression, airway obstruction and/or arrest. Continuously monitor patients for early signs of hypoventilation, airway obstruction, and apnea using capnography, pulse oximetry, and clinical assessment [see Warnings and Precautions (5.3)].
Reactions such as agitation, involuntary movements, hyperactivity and combativeness have been reported in adult and pediatric patients. Should such reactions occur, the response to each dose of midazolam and all other drugs, including local anesthetics, should be evaluated before proceeding. Reversal of such responses with flumazenil has been reported in pediatric patients [see Warnings and Precautions (5.4)].
Visually inspect parenteral drug products for particulate matter and discoloration prior to administration, whenever solution and container permit. If solution is discolored or particulate matter is present, do not use.
5.1 Personnel and Equipment for Monitoring and Resuscitation
- Prior to the intravenous administration of midazolam in any dose, ensure the immediate availability of oxygen, resuscitative drugs, age- and size-appropriate equipment for bag/valve/mask ventilation and intubation, and skilled personnel for the maintenance of a patent airway and support of ventilation.
- Only personnel trained in the administration of procedural sedation, and not involved in the conduct of the diagnostic or therapeutic procedure, should administer Midazolam in 0.9% Sodium Chloride Injection.
- Administering personnel must be trained in the detection and management of airway obstruction, hypoventilation, and apnea, including the maintenance of a patent airway, supportive ventilation, and cardiovascular resuscitation.
- Continuously monitor patients for early signs of hypoventilation, airway obstruction, or apnea, with means readily available (e.g., pulse oximetry). Hypoventilation, airway obstruction, and apnea can lead to hypoxia and/or cardiac arrest unless effective countermeasures are taken immediately.
- A benzodiazepine reversal agent (i.e., flumazenil) should be immediately available during administration of Midazolam in 0.9% Sodium Chloride Injection.
- Continuously monitor vital signs during the recovery period. Because intravenous midazolam can depress respiration [see Clinical Pharmacology (12)], especially when used concomitantly with opioid agonists and other sedatives [see Dosage and Administration (2)], it should be used for sedation/anxiolysis/amnesia only in the presence of personnel skilled in early detection of hypoventilation, maintaining a patent airway, and supporting ventilation.
5.9 Risk of Hypotension and Seizure in Preterm Infants and Neonates
Rapid injection should be avoided in the neonatal population. Midazolam administered rapidly as an intravenous injection (i.e., less than 2 minutes) has been associated with severe hypotension in neonates, particularly when the patient has also received fentanyl. Likewise, severe hypotension has been observed in neonates receiving a continuous infusion of midazolam who then receive a rapid intravenous injection of fentanyl. Seizures have been reported in several neonates following rapid intravenous administration.
The neonate also has reduced and/or immature organ function and is also vulnerable to profound and/or prolonged respiratory effects of midazolam.
5.12 Risk of Increased Intraocular Pressure in Patients With Glaucoma (5.12 Risk of Increased Intraocular Pressure in Patients with Glaucoma)
Benzodiazepines, including Midazolam in 0.9% Sodium Chloride Injection, can increase intraocular pressure in patients with glaucoma. Measurements of intraocular pressure in patients without eye disease show a moderate lowering following induction with midazolam. Midazolam in 0.9% Sodium Chloride Injection may be used in patients with open-angle glaucoma only if they are receiving appropriate therapy. Patients with open-angle glaucoma may need to have their ophthalmologic status evaluated following treatment with Midazolam in 0.9% Sodium Chloride Injection. Midazolam in 0.9% Sodium Chloride Injection is contraindicated in patients with narrow-angle glaucoma.
2.4 Safe Discontinuation of Midazolam in 0.9% Sodium Chloride Injection After Long Term Use (2.4 Safe Discontinuation of Midazolam in 0.9% Sodium Chloride Injection After Long-Term Use)
If Midazolam in 0.9% Sodium Chloride Injection is administered long-term (for several days to weeks), do not abruptly discontinue. Gradually taper the dosage in physically-dependent patients using a tapering schedule that is individualized to the patient [see Warnings and Precautions (5.5)].
5.5 Risk of Dependence and Withdrawal With Long Term Use of Midazolam in 0.9% Sodium Chloride Injection (5.5 Risk of Dependence and Withdrawal with Long-Term Use of Midazolam in 0.9% Sodium Chloride Injection)
The continued use of benzodiazepines for several days to weeks may lead to clinically significant physical dependence. If used for long-term use (i.e., for several days to weeks), abrupt discontinuation or rapid dosage reduction of midazolam, or administration of flumazenil, a benzodiazepine antagonist, may precipitate acute withdrawal reactions, including seizures, which can be life-threatening.
Patients at an increased risk of withdrawal adverse reactions after benzodiazepine discontinuation or rapid dosage reduction include those who take higher dosages (i.e., higher and/or more frequent doses) and those who have had longer durations of use.
After extended therapy, do not abruptly discontinue Midazolam in 0.9% Sodium Chloride Injection. When discontinuing midazolam in a physically-dependent patient, gradually taper the dosage using a tapering schedule that is individualized to the patient [see Dosage and Administration (2.3), Dependence (9.3)].
5.2 Risks From Concomitant Use With Opioids, Other Sedative Hypnotics, Or Other Central Nervous System Depressants (5.2 Risks from Concomitant Use with Opioids, Other Sedative Hypnotics, or Other Central Nervous System Depressants)
Concomitant use of benzodiazepines and opioids may result in profound sedation, respiratory depression, coma, and death. Monitor patients for respiratory depression and sedation [see Warnings and Precautions (5.2) and Drug Interactions (7.1)].
Titrate the dose of Midazolam in 0.9% Sodium Chloride Injection when administered with opioid analgesics and sedative-hypnotics to the desired clinical response.
Continuously monitor sedated patients for hypotension, airway obstruction, hypoventilation, apnea, and oxygen desaturation. These cardiorespiratory effects may be more likely to occur in patients with obstructive sleep apnea, the elderly, and ASA‑PS III or IV patients.
Concomitant use of barbiturates, alcohol, or other central nervous system depressants may increase the risk of hypoventilation, airway obstruction, desaturation, or apnea and may contribute to profound and/or prolonged drug effect. Narcotic premedication also depresses the ventilatory response to carbon dioxide stimulation.
Warning: Personnel and Equipment for Monitoring and Resuscitation and Risks From Concomitant Use With Opioid Analgesics and Other Sedative Hypnotics (WARNING: PERSONNEL AND EQUIPMENT FOR MONITORING AND RESUSCITATION AND RISKS FROM CONCOMITANT USE WITH OPIOID ANALGESICS AND OTHER SEDATIVE-HYPNOTICS)
Personnel and Equipment for Monitoring and Resuscitation
- Only personnel trained in the administration of procedural sedation, and not involved in the conduct of the diagnostic or therapeutic procedure, should administer Midazolam in 0.9% Sodium Chloride Injection [see Dosage and Administration (2.1), Warnings and Precautions (5.1)].
- Administering personnel must be trained in the detection and management of airway obstruction, hypoventilation, and apnea, including the maintenance of a patent airway, supportive ventilation, and cardiovascular resuscitation [see Dosage and Administration (2.1) , Warnings and Precautions (5.1)].
- Resuscitative drugs, and age- and size-appropriate equipment for bag/valve/mask assisted ventilation must be immediately available during administration of Midazolam in 0.9% Sodium Chloride Injection [see Dosage and Administration (2.1) , Warnings and Precautions (5.1)].
- Continuously monitor vital signs during sedation and during the recovery period [see Dosage and Administration (2.1) , Warnings and Precautions (5.1)].
Risks from Concomitant Use with Opioid Analgesics and Other Sedative Hypnotics
Concomitant use of benzodiazepines, including Midazolam in 0.9% Sodium Chloride Injection, and opioids may result in profound sedation, respiratory depression, coma, and death. Continuously monitor patients for respiratory depression and depth of sedation [see Warnings and Precautions (5.2) and Drug Interaction (7.1)].
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:46:40.554174 · Updated: 2026-03-14T22:25:27.698826