Proctozone-b Tm
c53ed934-3846-4dc8-b97b-c84171c21ae1
34390-5
HUMAN OTC DRUG LABEL
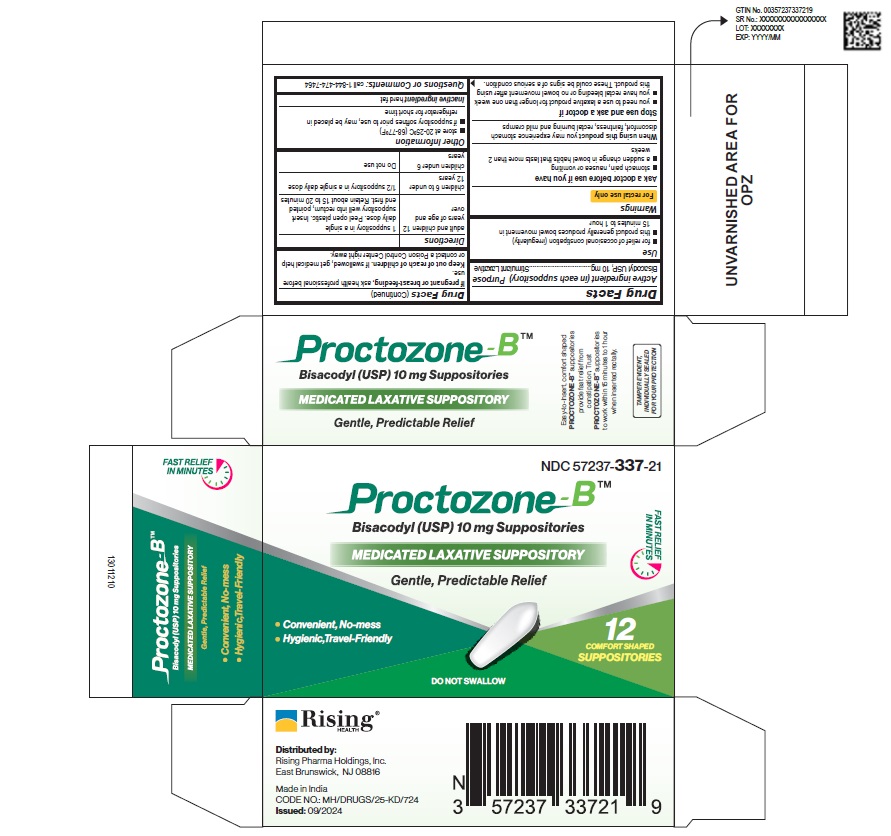
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Drug Facts
Medication Information
Warnings and Precautions
Warnings
For rectal use only
Indications and Usage
Use
- for relief of occasional constipation (irregularity)
- this product generally produces bowel movement in 15 minutes to 1 hour
Dosage and Administration
Directions
|
adults and children 12 years of age and over |
1 suppository in a single daily dose. Peel open plastic. Insert suppository well into rectum, pointed end first. Retain about 15 to 20 minutes |
| children 6 to under 12 years | 1/2 suppository in a single daily dose |
| children under 6 years | Do not use |
Description
Drug Facts
Section 42229-5
Drug Facts
Section 44425-7
Other information
- store at 20-25°C (68-77°F)
- if suppository softens prior to use, may be placed in refrigerator for short time
Section 50565-1
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center right away.
Section 50566-9
Stop use and ask a doctor if
- you need to use a laxative product for longer than one week
- you have rectal bleeding or no bowel movement after using this product. These could be signs of a serious condition.
Section 50567-7
When using this product you may experience stomach discomfort, faintness, rectal burning and mild cramps
Section 50569-3
Ask a doctor before use if you have
- stomach pain, nausea or vomiting
- a sudden change in bowel habits that lasts more than 2 weeks
Section 51727-6
Inactive ingredient hard fat
Section 51945-4
NDC 57237-337-21 (3 blisters of 4 suppositories)
Proctozone-BTM
Bisacodyl (USP) 10 mg Suppositories
MEDICATED LAXATIVE SUPPOSITORY
Gentle, Predictable Relief
DO NOT SWALLOW
NDC 57237-337-04 (4 suppositories in 1 blister pack)
Front Foil
NDC 57237-337-04 (4 suppositories in 1 blister pack)
Back Foil
Section 53413-1
Questions or Comments: Call 1-844-474-7464
Section 53414-9
If pregnant or breast-feeding, ask health professional before use.
Section 55105-1
Purpose
Stimulant Laxative
Section 55106-9
Active ingredient (in each suppository)
Bisacodyl USP, 10 mg
Section 60561-8
TAMPER EVIDENT, INDIVIVIDUALLY SEALED FOR YOUR PROTECTION
Structured Label Content
Indications and Usage (34067-9)
Use
- for relief of occasional constipation (irregularity)
- this product generally produces bowel movement in 15 minutes to 1 hour
Dosage and Administration (34068-7)
Directions
|
adults and children 12 years of age and over |
1 suppository in a single daily dose. Peel open plastic. Insert suppository well into rectum, pointed end first. Retain about 15 to 20 minutes |
| children 6 to under 12 years | 1/2 suppository in a single daily dose |
| children under 6 years | Do not use |
Warnings and Precautions (34071-1)
Warnings
For rectal use only
Section 42229-5 (42229-5)
Drug Facts
Section 44425-7 (44425-7)
Other information
- store at 20-25°C (68-77°F)
- if suppository softens prior to use, may be placed in refrigerator for short time
Section 50565-1 (50565-1)
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center right away.
Section 50566-9 (50566-9)
Stop use and ask a doctor if
- you need to use a laxative product for longer than one week
- you have rectal bleeding or no bowel movement after using this product. These could be signs of a serious condition.
Section 50567-7 (50567-7)
When using this product you may experience stomach discomfort, faintness, rectal burning and mild cramps
Section 50569-3 (50569-3)
Ask a doctor before use if you have
- stomach pain, nausea or vomiting
- a sudden change in bowel habits that lasts more than 2 weeks
Section 51727-6 (51727-6)
Inactive ingredient hard fat
Section 51945-4 (51945-4)
NDC 57237-337-21 (3 blisters of 4 suppositories)
Proctozone-BTM
Bisacodyl (USP) 10 mg Suppositories
MEDICATED LAXATIVE SUPPOSITORY
Gentle, Predictable Relief
DO NOT SWALLOW
NDC 57237-337-04 (4 suppositories in 1 blister pack)
Front Foil
NDC 57237-337-04 (4 suppositories in 1 blister pack)
Back Foil
Section 53413-1 (53413-1)
Questions or Comments: Call 1-844-474-7464
Section 53414-9 (53414-9)
If pregnant or breast-feeding, ask health professional before use.
Section 55105-1 (55105-1)
Purpose
Stimulant Laxative
Section 55106-9 (55106-9)
Active ingredient (in each suppository)
Bisacodyl USP, 10 mg
Section 60561-8 (60561-8)
TAMPER EVIDENT, INDIVIVIDUALLY SEALED FOR YOUR PROTECTION
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:04:34.395168 · Updated: 2026-03-14T23:05:25.370894