Ertapenem For Injection
c4da28cb-6725-4fa1-a7b0-e84f408fb36b
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Indications and Usage
Ertapenem for injection is a penem antibacterial indicated in adult patients and pediatric patients (3 months of age and older) for the treatment of the following moderate to severe infections caused by susceptible bacteria: Complicated intra-abdominal infections. ( 1.1 ) Complicated skin and skin structure infections, including diabetic foot infections without osteomyelitis. ( 1.2 ) Community-acquired pneumonia. ( 1.3 ) Complicated urinary tract infections including pyelonephritis. ( 1.4 ) Acute pelvic infections including postpartum endomyometritis, septic abortion and post-surgical gynecologic infections. ( 1.5 ) Ertapenem for injection is indicated in adults for the prophylaxis of surgical site infection following elective colorectal surgery. ( 1.6 ) To reduce the development of drug-resistant bacteria and maintain the effectiveness of Ertapenem for injection and other antibacterial drugs, Ertapenem for injection should be used only to treat or prevent infections that are proven or strongly suspected to be caused by susceptible bacteria. ( 1.7 )
Dosage and Administration
Do not mix or co-infuse Ertapenem for injection with other medications. Do not use diluents containing dextrose ( α –D–glucose) . ( 2.1 ) Ertapenem for injection should be infused over 30 minutes in both the Treatment and Prophylactic regimens. ( 2.1 ) Dosing considerations should be made in adults with advanced or end-stage renal impairment and those on hemodialysis. ( 2.4 , 2.5 ) Treatment regimen: Adults and pediatric patients 13 years of age and older. The dosage should be 1 gram once a day intravenously or intramuscularly. ( 2.2 ) Patients 3 months to 12 years of age should be administered 15 mg/kg twice daily (not to exceed 1 g/day intravenously or intramuscularly.) ( 2.2 ) Intravenous infusion may be administered in adults and pediatrics for up to 14 days or intramuscular injection for up to 7 days. ( 2.1 ) Prophylaxis regimen for adults: 1 gram single dose given 1 hour prior to elective colorectal surgery. ( 2.3 )
Contraindications
Ertapenem for injection is contraindicated in patients with known hypersensitivity to any component of this product or to other drugs in the same class or in patients who have demonstrated anaphylactic reactions to beta-lactams. Due to the use of Lidocaine HCl as a diluent, Ertapenem for injection administered intramuscularly is contraindicated in patients with a known hypersensitivity to local anesthetics of the amide type.
Warnings and Precautions
Serious hypersensitivity (anaphylactic) reactions have been reported in patients receiving β-lactams. ( 5.1 ) Seizures and other central nervous system adverse experiences have been reported during treatment. ( 5.2 ) Co-administration of Ertapenem for injection with valproic acid or divalproex sodium reduces the serum concentration of valproic acid potentially increasing the risk of breakthrough seizures. ( 5.3 ) Clostridioides difficile -associated diarrhea (ranging from mild diarrhea to fatal colitis): Evaluate if diarrhea occurs. ( 5.4 ) Caution should be taken when administering Ertapenem for injection intramuscularly to avoid inadvertent injection into a blood vessel. ( 5.5 )
Adverse Reactions
The following are described in greater detail in the Warnings and Precautions section. Hypersensitivity Reactions [see Warnings and Precautions (5.1) ] Seizure Potential [see Warnings and Precautions (5.2) ] Interaction with Valproic Acid [see Warnings and Precautions (5.3) ] Clostridioides difficile -Associated Diarrhea (CDAD) [see Warnings and Precautions (5.4) ] Caution with Intramuscular Administration [see Warnings and Precautions (5.5) ] Development of Drug-Resistant Bacteria [see Warnings and Precautions (5.6) ] Laboratory Tests [see Warnings and Precautions (5.7) ]
Drug Interactions
Co-administration with probenecid inhibits the renal excretion of ertapenem and is therefore not recommended. ( 7.1 ) The concomitant use of ertapenem and valproic acid/divalproex sodium is generally not recommended. Anti-bacterials other than carbapenems should be considered to treat infections in patients whose seizures are well controlled on valproic acid or divalproex sodium. ( 5.2 , 7.2 )
How Supplied
Ertapenem for injection is supplied as a sterile lyophilized powder in single-dose vials containing ertapenem for intravenous infusion or for intramuscular injection as follows: No. 051---1 g ertapenem equivalent NDC 44647-051-01 in 20mL of 1 vial. NDC 44647-051-10 in carton of 10 vials.
Description
Ertapenem for injection is a penem antibacterial indicated in adult patients and pediatric patients (3 months of age and older) for the treatment of the following moderate to severe infections caused by susceptible bacteria: Complicated intra-abdominal infections. ( 1.1 ) Complicated skin and skin structure infections, including diabetic foot infections without osteomyelitis. ( 1.2 ) Community-acquired pneumonia. ( 1.3 ) Complicated urinary tract infections including pyelonephritis. ( 1.4 ) Acute pelvic infections including postpartum endomyometritis, septic abortion and post-surgical gynecologic infections. ( 1.5 ) Ertapenem for injection is indicated in adults for the prophylaxis of surgical site infection following elective colorectal surgery. ( 1.6 ) To reduce the development of drug-resistant bacteria and maintain the effectiveness of Ertapenem for injection and other antibacterial drugs, Ertapenem for injection should be used only to treat or prevent infections that are proven or strongly suspected to be caused by susceptible bacteria. ( 1.7 )
Medication Information
Warnings and Precautions
Serious hypersensitivity (anaphylactic) reactions have been reported in patients receiving β-lactams. ( 5.1 ) Seizures and other central nervous system adverse experiences have been reported during treatment. ( 5.2 ) Co-administration of Ertapenem for injection with valproic acid or divalproex sodium reduces the serum concentration of valproic acid potentially increasing the risk of breakthrough seizures. ( 5.3 ) Clostridioides difficile -associated diarrhea (ranging from mild diarrhea to fatal colitis): Evaluate if diarrhea occurs. ( 5.4 ) Caution should be taken when administering Ertapenem for injection intramuscularly to avoid inadvertent injection into a blood vessel. ( 5.5 )
Indications and Usage
Ertapenem for injection is a penem antibacterial indicated in adult patients and pediatric patients (3 months of age and older) for the treatment of the following moderate to severe infections caused by susceptible bacteria: Complicated intra-abdominal infections. ( 1.1 ) Complicated skin and skin structure infections, including diabetic foot infections without osteomyelitis. ( 1.2 ) Community-acquired pneumonia. ( 1.3 ) Complicated urinary tract infections including pyelonephritis. ( 1.4 ) Acute pelvic infections including postpartum endomyometritis, septic abortion and post-surgical gynecologic infections. ( 1.5 ) Ertapenem for injection is indicated in adults for the prophylaxis of surgical site infection following elective colorectal surgery. ( 1.6 ) To reduce the development of drug-resistant bacteria and maintain the effectiveness of Ertapenem for injection and other antibacterial drugs, Ertapenem for injection should be used only to treat or prevent infections that are proven or strongly suspected to be caused by susceptible bacteria. ( 1.7 )
Dosage and Administration
Do not mix or co-infuse Ertapenem for injection with other medications. Do not use diluents containing dextrose ( α –D–glucose) . ( 2.1 ) Ertapenem for injection should be infused over 30 minutes in both the Treatment and Prophylactic regimens. ( 2.1 ) Dosing considerations should be made in adults with advanced or end-stage renal impairment and those on hemodialysis. ( 2.4 , 2.5 ) Treatment regimen: Adults and pediatric patients 13 years of age and older. The dosage should be 1 gram once a day intravenously or intramuscularly. ( 2.2 ) Patients 3 months to 12 years of age should be administered 15 mg/kg twice daily (not to exceed 1 g/day intravenously or intramuscularly.) ( 2.2 ) Intravenous infusion may be administered in adults and pediatrics for up to 14 days or intramuscular injection for up to 7 days. ( 2.1 ) Prophylaxis regimen for adults: 1 gram single dose given 1 hour prior to elective colorectal surgery. ( 2.3 )
Contraindications
Ertapenem for injection is contraindicated in patients with known hypersensitivity to any component of this product or to other drugs in the same class or in patients who have demonstrated anaphylactic reactions to beta-lactams. Due to the use of Lidocaine HCl as a diluent, Ertapenem for injection administered intramuscularly is contraindicated in patients with a known hypersensitivity to local anesthetics of the amide type.
Adverse Reactions
The following are described in greater detail in the Warnings and Precautions section. Hypersensitivity Reactions [see Warnings and Precautions (5.1) ] Seizure Potential [see Warnings and Precautions (5.2) ] Interaction with Valproic Acid [see Warnings and Precautions (5.3) ] Clostridioides difficile -Associated Diarrhea (CDAD) [see Warnings and Precautions (5.4) ] Caution with Intramuscular Administration [see Warnings and Precautions (5.5) ] Development of Drug-Resistant Bacteria [see Warnings and Precautions (5.6) ] Laboratory Tests [see Warnings and Precautions (5.7) ]
Drug Interactions
Co-administration with probenecid inhibits the renal excretion of ertapenem and is therefore not recommended. ( 7.1 ) The concomitant use of ertapenem and valproic acid/divalproex sodium is generally not recommended. Anti-bacterials other than carbapenems should be considered to treat infections in patients whose seizures are well controlled on valproic acid or divalproex sodium. ( 5.2 , 7.2 )
How Supplied
Ertapenem for injection is supplied as a sterile lyophilized powder in single-dose vials containing ertapenem for intravenous infusion or for intramuscular injection as follows: No. 051---1 g ertapenem equivalent NDC 44647-051-01 in 20mL of 1 vial. NDC 44647-051-10 in carton of 10 vials.
Description
Ertapenem for injection is a penem antibacterial indicated in adult patients and pediatric patients (3 months of age and older) for the treatment of the following moderate to severe infections caused by susceptible bacteria: Complicated intra-abdominal infections. ( 1.1 ) Complicated skin and skin structure infections, including diabetic foot infections without osteomyelitis. ( 1.2 ) Community-acquired pneumonia. ( 1.3 ) Complicated urinary tract infections including pyelonephritis. ( 1.4 ) Acute pelvic infections including postpartum endomyometritis, septic abortion and post-surgical gynecologic infections. ( 1.5 ) Ertapenem for injection is indicated in adults for the prophylaxis of surgical site infection following elective colorectal surgery. ( 1.6 ) To reduce the development of drug-resistant bacteria and maintain the effectiveness of Ertapenem for injection and other antibacterial drugs, Ertapenem for injection should be used only to treat or prevent infections that are proven or strongly suspected to be caused by susceptible bacteria. ( 1.7 )
Section 34077-8
Risk Summary
Available data from a small number of post-marketing cases with Ertapenem for injection use in pregnancy are insufficient to inform any drug-associated risks for major birth defects, miscarriage, or adverse maternal or fetal outcomes. In animal reproduction studies after intravenous administration of ertapenem during the period of organogenesis, there was no evidence of developmental malformations in rats at systemic exposures (AUC) up to approximately 1.2 times the human exposure at the maximum recommended human dose (MRHD) and in mice at doses up to approximately 3 times the MRHD based on body surface area comparison. In pregnant rats administered ertapenem during organogenesis through lactation, fetal toxicity, developmental delays, and impaired reproduction did not occur in first generation offspring at systemic exposures (AUC) approximately 1.2 times the human exposure at the MRHD (see Data).
The estimated background risk of major birth defects and miscarriage for the indicated population is unknown. All pregnancies have a background risk of birth defect, loss, or other adverse outcomes. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2 to 4% and 15 to 20%, respectively.
Data
Animal Data
In pregnant rats, intravenous administration of ertapenem dosages of up to 700 mg/kg/day (approximately 1.2 times the MRHD based on AUC) during the period of organogenesis (gestation days [GD] 6-20) revealed no maternal or embryofetal effects.
Pregnant mice intravenously administered ertapenem dosages of up to 700 mg/kg/day (approximately 3 times the MRHD based on body surface area comparison) during the period of organogenesis (GD 6-15) showed slight decreases in average fetal weight and an associated decrease in the average number of ossified sacrocaudal vertebrae. There were no maternal effects at any dosage.
In a pre-postnatal study in rats, ertapenem administered to pregnant rats at dosages up to 700 mg/kg/day (approximately 1.2 times the MRHD based on AUC) during organogenesis through lactation, (GD 6 until Lactation Day (LD) 20) did not result in fetal toxicity, developmental delays, or impaired reproduction in first generation offspring, and fetal deaths and malformations were not increased in second generation offspring.
Section 42229-5
Prevention
Ertapenem for injection is indicated in adults for:
1.7 Usage
To reduce the development of drug-resistant bacteria and maintain the effectiveness of Ertapenem for injection and other antibacterial drugs, Ertapenem for injection should be used only to treat or prevent infections that are proven or strongly suspected to be caused by susceptible bacteria. When culture and susceptibility information are available, they should be considered in selecting or modifying antibacterial therapy. In the absence of such data, local epidemiology and susceptibility patterns may contribute to the empiric selection of therapy.
10 Overdosage
No specific information is available on the treatment of overdosage with Ertapenem for injection. Intentional overdosing of Ertapenem for injection is unlikely. Intravenous administration of Ertapenem for injection at a dose of 2 g over 30 min or 3 g over 1-2h in healthy adult volunteers resulted in an increased incidence of nausea. In clinical trials in adults, inadvertent administration of three 1 g doses of Ertapenem for injection in a 24 hour period resulted in diarrhea and transient dizziness in one patient. In pediatric clinical trials, a single intravenous dose of 40 mg/kg up to a maximum of 2 g did not result in toxicity.
In the event of an overdose, Ertapenem for injection should be discontinued and general supportive treatment given until renal elimination takes place.
Ertapenem for injection can be removed by hemodialysis; the plasma clearance of the total fraction of ertapenem was increased 30% in subjects with end-stage renal disease when hemodialysis (4 hour session) was performed immediately following administration. However, no information is available on the use of hemodialysis to treat overdosage.
8.2 Lactation
Risk Summary
Ertapenem is present in human milk (see
Data). There are no data on the effects on the breastfed infant or the effects on milk production. The developmental and health benefits of breastfeeding should be
considered along with the mother’s clinical need for Ertapenem for injection and any potential adverse effects on the breastfed infant from Ertapenem for injection or from the underlying maternal condition.
Data
The concentration of ertapenem in breast milk from 5 lactating women with pelvic infections (5 to 14 days postpartum) measured at random time points daily for 5 consecutive days following the last 1 g dose of intravenous therapy (3 to 10 days of therapy) showed low levels. The concentration of ertapenem in breast milk within 24 hours of the last dose of therapy in all 5 women ranged from (<0.13 (lower limit of quantitation) to 0.38 mcg/ml), although peak concentrations were not assessed. By day 5 after discontinuation of therapy, the level of ertapenem was undetectable in the breast milk of 4 women and below the lower limit of quantitation (<0.13 mcg/mL) in 1 woman. The concentration of ertapenem in transitional milk observed in this study may not reflect the concentration of ertapenem in mature milk.
11 Description
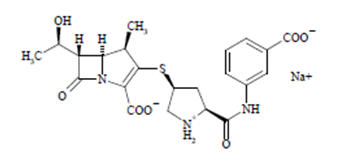
Ertapenem for injection is a sterile, synthetic, parenteral, 1-β methyl-carbapenem that is structurally related to beta-lactam antibiotics.
Chemically, Ertapenem for injection is described as [4R-[3(3S*,5S*),4α,5β,6β(R*)]]-3-[[5-[[(3carboxyphenyl)amino]carbonyl]-3-pyrrolidinyl]thio]-6-(1-hydroxyethyl)-4-methyl-7-oxo-1 azabicyclo[3.2.0]hept-2-ene-2-carboxylic acid monosodium salt. Its molecular weight is 497.50. The empirical formula is C 22H 24N 3O 7SNa, and its structural formula is:
Ertapenem sodium is a white to off-white hygroscopic, weakly crystalline powder. It is soluble in water and 0.9% sodium chloride solution, practically insoluble in ethanol, and insoluble in isopropyl acetate and tetrahydrofuran.
Ertapenem for injection is supplied as sterile lyophilized powder for intravenous infusion after reconstitution with appropriate diluent [see Dosage and Administration (2.7)] and transfer to 50 mL 0.9% Sodium Chloride Injection or for intramuscular injection following reconstitution with 1% lidocaine hydrochloride. Each single-dose vial contains 1 gram ertapenem equivalent to 1.046 grams ertapenem sodium. The sodium content is approximately 137 mg (approximately 6.0 mEq).
Each vial of Ertapenem for injection contains the following inactive ingredients: 175 mg sodium bicarbonate and sodium hydroxide to adjust pH to 7.5.
7.1 Probenecid
Probenecid interferes with the active tubular secretion of ertapenem, resulting in increased plasma concentrations of ertapenem [see Clinical Pharmacology (12.3)] . Co-administration of probenecid with ertapenem is not recommended.
16.1 How Supplied
Ertapenem for injection is supplied as a sterile lyophilized powder in single-dose vials containing ertapenem for intravenous infusion or for intramuscular injection as follows:
No. 051---1 g ertapenem equivalent
NDC 44647-051-01 in 20mL of 1 vial.
NDC 44647-051-10 in carton of 10 vials.
7.2 Valproic Acid
Case reports in the literature have shown that co-administration of carbapenems, including ertapenem, to patients receiving valproic acid or divalproex sodium results in a reduction of valproic acid concentrations. The valproic acid concentrations may drop below the therapeutic range as a result of this interaction, therefore increasing the risk of breakthrough seizures. Although the mechanism of this interaction is unknown, data from in vitro and animal studies suggest that carbapenems may inhibit the hydrolysis of valproic acid's glucuronide metabolite (VPA-g) back to valproic acid, thus decreasing the serum concentrations of valproic acid [see Warnings and Precautions (5.3)].
8.4 Pediatric Use
Safety and effectiveness of Ertapenem for injection in pediatric patients 3 months to 17 years of age are supported by evidence from adequate and well-controlled trials in adults, pharmacokinetic data in pediatric patients, and additional data from comparator-controlled trials in pediatric patients 3 months to 17 years of age [see Indications and Usage (1.1), (1.2), (1.3), (1.4) and (1.5) and Clinical Studies (14.2)] .
Ertapenem for injection is not recommended in infants under 3 months of age as no data are available.
Ertapenem for injection is not recommended in the treatment of meningitis in the pediatric population due to lack of sufficient CSF penetration.
8.5 Geriatric Use
Of the 1,835 patients in Phase 2b/3 trials treated with Ertapenem for injection, approximately 26 percent were 65 and over, while approximately 12 percent were 75 and over. No overall differences in safety or effectiveness were observed between these patients and younger patients. Other reported clinical experience has not identified differences in responses between the elderly and younger patients, but greater sensitivity of some older individuals cannot be ruled out.
This drug is known to be substantially excreted by the kidney, and the risk of toxic reactions to this drug may be greater in patients with impaired renal function. Because elderly patients are more likely to have decreased renal function, care should be taken in dose selection, and it may be useful to monitor renal function [see Dosage and Administration (2.2)].
4 Contraindications
- Ertapenem for injection is contraindicated in patients with known hypersensitivity to any component of this product or to other drugs in the same class or in patients who have demonstrated anaphylactic reactions to beta-lactams.
- Due to the use of Lidocaine HCl as a diluent, Ertapenem for injection administered intramuscularly is contraindicated in patients with a known hypersensitivity to local anesthetics of the amide type.
6 Adverse Reactions
The following are described in greater detail in the Warnings and Precautions section.
- Hypersensitivity Reactions [see Warnings and Precautions (5.1)]
- Seizure Potential [see Warnings and Precautions (5.2)]
- Interaction with Valproic Acid [see Warnings and Precautions (5.3)]
- Clostridioides difficile-Associated Diarrhea (CDAD) [see Warnings and Precautions (5.4)]
- Caution with Intramuscular Administration [see Warnings and Precautions (5.5)]
- Development of Drug-Resistant Bacteria [see Warnings and Precautions (5.6)]
- Laboratory Tests [see Warnings and Precautions (5.7)]
7 Drug Interactions
- Co-administration with probenecid inhibits the renal excretion of ertapenem and is therefore not recommended. ( 7.1)
- The concomitant use of ertapenem and valproic acid/divalproex sodium is generally not recommended. Anti-bacterials other than carbapenems should be considered to treat infections in patients whose seizures are well controlled on valproic acid or divalproex sodium. ( 5.2, 7.2)
5.7 Laboratory Tests
While Ertapenem for injection possesses toxicity similar to the beta-lactam group of antibiotics, periodic assessment of organ system function, including renal, hepatic, and hematopoietic, is advisable during prolonged therapy.
12.3 Pharmacokinetics
Average plasma concentrations (mcg/mL) of ertapenem following a single 30-minute infusion of a 1 g intravenous (IV) dose and administration of a single 1 g intramuscular (IM) dose in healthy young adults are presented in Table 8.
| Average Plasma Concentrations (mcg/mL) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Dose/Route | 0.5 hr | 1 hr | 2 hr | 4 hr | 6 hr | 8 hr | 12 hr | 18 hr | 24 hr |
| 1 g IV
Infused at a constant rate over 30 minutes
|
155 | 115 | 83 | 48 | 31 | 20 | 9 | 3 | 1 |
| 1 g IM | 33 | 53 | 67 | 57 | 40 | 27 | 13 | 4 | 2 |
The area under the plasma concentration-time curve (AUC) of ertapenem in adults increased less-than dose-proportional based on total ertapenem concentrations over the 0.5 to 2 g dose range, whereas the AUC increased greater-than dose-proportional based on unbound ertapenem concentrations. Ertapenem exhibits non-linear pharmacokinetics due to concentration-dependent plasma protein binding at the proposed therapeutic dose [see Clinical Pharmacology (12.3)] . There is no accumulation of ertapenem following multiple IV or IM 1 g daily doses in healthy adults.
Average plasma concentrations (mcg/mL) of ertapenem in pediatric patients are presented in Table 9.
| Average Group | Dose | Average Plasma Concentrations (mcg/mL) | |||||||
|---|---|---|---|---|---|---|---|---|---|
| 0.5 hr | 1 hr | 2 hr | 4 hr | 6 hr | 8 hr | 12 hr | 24 hr | ||
| 3 to 23 months | |||||||||
| 15 mg/kg
up to a maximum dose of 1 g/day
|
103.8 | 57.3 | 43.6 | 23.7 | 13.5 | 8.2 | 2.5 | - | |
| 20 mg/kg | 126.8 | 87.6 | 58.7 | 28.4 | - | 12.0 | 3.4 | 0.4 | |
| 40 mg/kg
up to a maximum dose of 2 g/day
|
199.1 | 144.1 | 95.7 | 58.0 | - | 20.2 | 7.7 | 0.6 | |
| 2 to 12 years | |||||||||
| 15 mg/kg | 113.2 | 63.9 | 42.1 | 21.9 | 12.8 | 7.6 | 3.0 | - | |
| 20 mg/kg | 147.6 | 97.6 | 63.2 | 34.5 | - | 12.3 | 4.9 | 0.5 | |
| 40 mg/kg | 241.7 | 152.7 | 96.3 | 55.6 | - | 18.8 | 7.2 | 0.6 | |
| 13 to 17 years | |||||||||
| 20 mg/kg | 170.4 | 98.3 | 67.8 | 40.4 | - | 16.0 | 7.0 | 1.1 | |
| 1 g
Based on three patients receiving 1 g ertapenem who volunteered for pharmacokinetic assessment in one of the two safety and efficacy trials
|
155.9 | 110.9 | 74.8 | - | 24.0 | - | 6.2 | - | |
| 40 mg/kg | 255.0 | 188.7 | 127.9 | 76.2 | - | 31.0 | 15.3 | 2.1 |
5.2 Seizure Potential
Seizures and other central nervous system (CNS) adverse experiences have been reported during treatment with Ertapenem for injection [see Adverse Reactions (6.1)] . During clinical investigations in adult patients treated with Ertapenem for injection (1 g once a day), seizures, irrespective of drug relationship, occurred in 0.5% of patients during study therapy plus 14-day follow-up period [see Adverse Reactions (6.1)] . These experiences have occurred most commonly in patients with CNS disorders (e.g., brain lesions or history of seizures) and/or compromised renal function. Close adherence to the recommended dosage regimen is urged, especially in patients with known factors that predispose to convulsive activity. Anticonvulsant therapy should be continued in patients with known seizure disorders. If focal tremors, myoclonus, or seizures occur, patients should be evaluated neurologically, placed on anticonvulsant therapy if not already instituted, and the dosage of Ertapenem for injection re-examined to determine whether it should be decreased or discontinued.
1 Indications and Usage
14.2 Pediatric Patients
Ertapenem was evaluated in pediatric patients 3 months to 17 years of age in two randomized, multicenter clinical trials.
The first trial enrolled 404 patients and compared ertapenem (15 mg/kg intravenous (IV) every 12 hours in patients 3 months to 12 years of age, and 1 g IV once a day in patients 13 to 17 years of age) to ceftriaxone (50 mg/kg/day IV in two divided doses in patients 3 months to 12 years of age and 50 mg/kg/day IV as a single daily dose in patients 13 to 17 years of age) for the treatment of complicated urinary tract infection (UTI), skin and soft tissue infection (SSTI), or community-acquired pneumonia (CAP). Both regimens allowed the option to switch to oral amoxicillin/clavulanate for a total of up to 14 days of treatment (parenteral and oral). The microbiological success rates in the evaluable per protocol (EPP) analysis in patients treated for UTI were 87.0% (40/46) for ertapenem and 90.0% (18/20) for ceftriaxone. The clinical success rates in the EPP analysis in patients treated for SSTI were 95.5% (64/67) for ertapenem and 100% (26/26) for ceftriaxone, and in patients treated for CAP were 96.1% (74/77) for ertapenem and 96.4% (27/28) for ceftriaxone.
The second trial enrolled 112 patients and compared ertapenem (15 mg/kg IV every 12 hours in patients 3 months to 12 years of age, and 1 g IV once a day in patients 13 to 17 years of age) to ticarcillin/clavulanate (50 mg/kg for patients <60 kg or 3.0 g for patients >60 kg, 4 or 6 times a day) up to 14 days for the treatment of complicated intra-abdominal infections (IAI) and acute pelvic infections (API). In patients treated for IAI (primarily patients with perforated or complicated appendicitis), the clinical success rates were 83.7% (36/43) for ertapenem and 63.6% (7/11) for ticarcillin/clavulanate in the EPP analysis. In patients treated for API (post-operative or spontaneous obstetrical endomyometritis, or septic abortion), the clinical success rates were 100% (23/23) for ertapenem and 100% (4/4) for ticarcillin/clavulanate in the EPP analysis.
12.1 Mechanism of Action
Ertapenem sodium is a carbapenem antibiotic [see Clinical Pharmacology (12.4)].
5 Warnings and Precautions
- Serious hypersensitivity (anaphylactic) reactions have been reported in patients receiving β-lactams. ( 5.1)
- Seizures and other central nervous system adverse experiences have been reported during treatment. ( 5.2)
- Co-administration of Ertapenem for injection with valproic acid or divalproex sodium reduces the serum concentration of valproic acid potentially increasing the risk of breakthrough seizures. ( 5.3)
- Clostridioides difficile-associated diarrhea (ranging from mild diarrhea to fatal colitis): Evaluate if diarrhea occurs. ( 5.4)
- Caution should be taken when administering Ertapenem for injection intramuscularly to avoid inadvertent injection into a blood vessel. ( 5.5)
2 Dosage and Administration
Do not mix or co-infuse Ertapenem for injection with other medications. Do not use diluents containing dextrose (α –D–glucose). ( 2.1)
Ertapenem for injection should be infused over 30 minutes in both the Treatment and Prophylactic regimens. ( 2.1)
Dosing considerations should be made in adults with advanced or end-stage renal impairment and those on hemodialysis. ( 2.4, 2.5)
Treatment regimen:
- Adults and pediatric patients 13 years of age and older. The dosage should be 1 gram once a day intravenously or intramuscularly. ( 2.2)
- Patients 3 months to 12 years of age should be administered 15 mg/kg twice daily (not to exceed 1 g/day intravenously or intramuscularly.) ( 2.2)
- Intravenous infusion may be administered in adults and pediatrics for up to 14 days or intramuscular injection for up to 7 days. ( 2.1)
Prophylaxis regimen for adults:
- 1 gram single dose given 1 hour prior to elective colorectal surgery. ( 2.3)
2.5 Patients On Hemodialysis
When adult patients on hemodialysis are given the recommended daily dose of 500 mg of Ertapenem for injection within 6 hours prior to hemodialysis, a supplementary dose of 150 mg is recommended following the hemodialysis session. If Ertapenem for injection is given at least 6 hours prior to hemodialysis, no supplementary dose is needed. There are no data in patients undergoing peritoneal dialysis or hemofiltration. There are no data in pediatric patients on hemodialysis.
When only the serum creatinine is available, the following formula
| Males: | (weight in kg) × (140-age in years) |
| (72) × serum creatine (mg/100 mL) | |
| Females: | (0.85) × (value calculated for males) |
3 Dosage Forms and Strengths
- For injection: Single-dose vial 1 gram. ( 3)
5.4 Clostridioides Difficile
CDAD has been reported with use of nearly all antibacterial agents, including ertapenem, and may range in severity from mild diarrhea to fatal colitis. Treatment with antibacterial agents alters the normal flora of the colon leading to overgrowth of Clostridioides difficile.
Clostridioides difficile produces toxins A and B which contribute to the development of CDAD. Hypertoxin producing strains of Clostridioides difficile cause increased morbidity and mortality, as these infections can be refractory to antimicrobial therapy and may require colectomy. CDAD must be considered in all patients who present with diarrhea following antibiotic use. Careful medical history is necessary since CDAD has been reported to occur over two months after the administration of antibacterial agents.
If CDAD is suspected or confirmed, ongoing antibiotic use not directed against Clostridioides difficile may need to be discontinued. Appropriate fluid and electrolyte management, protein supplementation, antibiotic treatment of Clostridioides difficile, and surgical evaluation should be instituted as clinically indicated.
6.2 Post Marketing Experience
The following additional adverse reactions have been identified during the post-approval use of Ertapenem for injection. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Gastrointestinal Disorders: teeth staining
Immune System Disorders: anaphylaxis including anaphylactoid reactions
Musculoskeletal and Connective Tissue Disorders: muscular weakness
Nervous System Disorders: coordination abnormal, depressed level of consciousness, dyskinesia, gait disturbance, myoclonus, tremor, encephalopathy (recovery was prolonged in patients with renal impairment)
Psychiatric Disorders: altered mental status (including aggression, delirium), hallucinations
Skin and Subcutaneous Tissue Disorders: Acute Generalized Exanthematous Pustulosis (AGEP), Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS syndrome), hypersensitivity vasculitis
8 Use in Specific Populations
17.1 Instructions for Patients
Patients should be advised that allergic reactions, including serious allergic reactions could occur and that serious reactions may require immediate treatment. Advise patients to report any previous hypersensitivity reactions to Ertapenem for injection, other beta-lactams or other allergens.
Patients should be counseled to inform their physician if they are taking valproic acid or divalproex sodium. Valproic acid concentrations in the blood may drop below the therapeutic range upon co-administration with Ertapenem for injection. If treatment with Ertapenem for injection is necessary and continued, alternative or supplemental anti-convulsant medication to prevent and/or treat seizures may be needed.
Patients should be counseled that antibacterial drugs including Ertapenem for injection should only be used to treat bacterial infections. They do not treat viral infections (e.g., the common cold). When Ertapenem for injection is prescribed to treat a bacterial infection, patients should be told that although it is common to feel better early in the course of therapy, the medication should be taken exactly as directed. Skipping doses or not completing the full course of therapy may (1) decrease the effectiveness of the immediate treatment and (2) increase the likelihood that bacteria will develop resistance and will not be treatable by Ertapenem for injection or other antibacterial drugs in the future.
Diarrhea is a common problem caused by antibiotics which usually ends when the antibiotic is discontinued. Sometimes after starting treatment with antibiotics, patients can develop watery and bloody stools (with or without stomach cramps and fever) even as late as two or more months after having taken the last dose of the antibiotic. If this occurs, patients should contact their physician as soon as possible.
5.1 Hypersensitivity Reactions
Serious and occasionally fatal hypersensitivity (anaphylactic) reactions have been reported in patients receiving therapy with beta-lactams. These reactions are more likely to occur in individuals with a history of sensitivity to multiple allergens. There have been reports of individuals with a history of penicillin hypersensitivity who have experienced severe hypersensitivity reactions when treated with another betalactam. Before initiating therapy with Ertapenem for injection, careful inquiry should be made concerning previous hypersensitivity reactions to penicillins, cephalosporins, other beta-lactams and other allergens. If an allergic reaction to Ertapenem for injection occurs, discontinue the drug immediately. Serious anaphylactic reactions require immediate emergency treatment as clinically indicated.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
1.3 Community Acquired Pneumonia
Ertapenem for injection is indicated for the treatment of adult patients and pediatric patients (3 months of age and older) with community acquired pneumonia due to Streptococcus pneumoniae (penicillin susceptible isolates only) including cases with concurrent bacteremia, Haemophilus influenzae (beta-lactamase negative isolates only), or Moraxella catarrhalis.
2.3 Prophylactic Regimen in Adults
Table 2 presents prophylaxis guidelines for Ertapenem for injection.
| Indication | Daily Dose
(IV) Adults |
Recommended Duration of Total Antimicrobial Treatment |
|---|---|---|
| Prophylaxis of surgical site infection following elective colorectal surgery | 1 g | Single intravenous dose given 1 hour prior to surgical incision |
2.4 Patients With Renal Impairment
Ertapenem for injection may be used for the treatment of infections in adult patients with renal impairment. In patients whose creatinine clearance is >30 mL/min/1.73 m 2, no dosage adjustment is necessary. Adult patients with severe renal impairment (creatinine clearance ≤30 mL/min/1.73 m 2) and end-stage renal disease (creatinine clearance ≤10 mL/min/1.73 m 2) should receive 500 mg daily. A supplementary dose of 150 mg is recommended if ertapenem is administered within 6 hours prior to hemodialysis. There are no data in pediatric patients with renal impairment.
5.3 Interaction With Valproic Acid
Case reports in the literature have shown that co-administration of carbapenems, including ertapenem, to patients receiving valproic acid or divalproex sodium results in a reduction in valproic acid concentrations. The valproic acid concentrations may drop below the therapeutic range as a result of this interaction, therefore increasing the risk of breakthrough seizures. Increasing the dose of valproic acid or divalproex sodium may not be sufficient to overcome this interaction. The concomitant use of ertapenem and valproic acid/divalproex sodium is generally not recommended. Anti-bacterials other than carbapenems should be considered to treat infections in patients whose seizures are well controlled on valproic acid or divalproex sodium. If administration of Ertapenem for injection is necessary, supplemental anticonvulsant therapy should be considered [see Drug Interactions (7.2)].
8.6 Patients With Renal Impairment
Dosage adjustment is necessary in patients with creatinine clearance 30 mL/min or less [see Dosage and Administration (2.4) and Clinical Pharmacology (12.3)] .
2.6 Patients With Hepatic Impairment
No dose adjustment recommendations can be made in patients with hepatic impairment [see Use in Specific Populations (8.7) and Clinical Pharmacology (12.3)].
8.7 Patients With Hepatic Impairment
The pharmacokinetics of ertapenem in patients with hepatic impairment have not been established. Of the total number of patients in clinical trials, 37 patients receiving ertapenem 1 g daily and 36 patients receiving comparator drugs were considered to have Child-Pugh Class A, B, or C liver impairment. The incidence of adverse experiences in patients with hepatic impairment was similar between the ertapenem group and the comparator groups.
Principal Display Panel 1 G Vial Label
NDC 44647-051-01
Ertapenem
for Injection
1 gram/vial
For Intravenous
or Intramuscular Use
Rx only
Principal Display Panel 10 Vial Carton
NDC 44647-051-10
Ertapenem for Injection
1 gram/vial
For Intravenous Or Intramuscular Use
R x Only
10 x 1 g Single dose vials
Each vial contains:
1.046 grams ertapenem sodium, equiv. to 1 gram ertapenem.
Prior to Constitution: Store lyophilized powder below 25°C (77°F).
1.1 Complicated Intra Abdominal Infections
Ertapenem for injection is indicated for the treatment of adult patients and pediatric patients (3 months of age and older) with complicated intra-abdominal infections due to Escherichia coli, Clostridium clostridioforme, Eubacterium lentum, Peptostreptococcus species, Bacteroides fragilis, Bacteroides distasonis, Bacteroides ovatus, Bacteroides thetaiotaomicron, or Bacteroides uniformis.
13.2 Animal Toxicology And/or Pharmacology
In repeat-dose studies in rats, treatment-related neutropenia occurred at every dose-level tested, including the lowest dose of 2 mg/kg (approximately 2% of the human dose on a body surface area basis).
Studies in rabbits and Rhesus monkeys were inconclusive with regard to the effect on neutrophil counts.
5.6 Development of Drug Resistant Bacteria
As with other antibiotics, prolonged use of Ertapenem for injection may result in overgrowth of non-susceptible organisms. Repeated evaluation of the patient's condition is essential. If superinfection occurs during therapy, appropriate measures should be taken.
Prescribing Ertapenem for injection in the absence of a proven or strongly suspected bacterial infection or a prophylactic indication is unlikely to provide benefit to the patient and increases the risk of the development of drug-resistant bacteria.
5.5 Caution With Intramuscular Administration
Caution should be taken when administering Ertapenem for injection intramuscularly to avoid inadvertent injection into a blood vessel [see Dosage and Administration (2.7)] .
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenesis and Mutagenesis
No long-term studies in animals have been performed to evaluate the carcinogenic potential of ertapenem.
Ertapenem was not genotoxic in in vitro or in vivo assays, including: alkaline elution/rat hepatocyte assay, chromosomal aberration assay in Chinese hamster ovary cells, a TK6 human lymphoblastoid cell mutagenesis assay and a mouse micronucleus assay.
Impairment of Fertility
In rats, intravenous dosages up to 700 mg/kg/day (approximately 1.2 times the human exposure at the human recommended human dose of 1 g based on plasma AUC) did not impair fertility.
1.4 Complicated Urinary Tract Infections Including Pyelonephritis
Ertapenem for injection is indicated for the treatment of adult patients and pediatic patients (3 months of age and older) with complicated urinary tract infections including pyelonephritis due to Escherichia coli, including cases with concurrent bacteremia, or Klebsiella pneumoniae.
1.6 Prophylaxis of Surgical Site Infection Following Elective Colorectal Surgery
Ertapenem for injection is indicated in adults for the prevention of surgical site infection following elective colorectal surgery.
1.2 Complicated Skin and Skin Structure Infections, Including Diabetic Foot Infections Without Osteomyelitis
Ertapenem for injection is indicated for the treatment of adult patients and pediatric patients (3 months of age and older) with complicated skin and skin structure infections, including diabetic foot infections without osteomyelitis due to Staphylococcus aureus (methicillin susceptible isolates only), Streptococcus agalactiae, Streptococcus pyogenes, Escherichia coli, Klebsiella pneumoniae, Proteus mirabilis, Bacteroides fragilis, Peptostreptococcus species, Porphyromonas asaccharolytica, or Prevotella bivia. Ertapenem for injection has not been studied in diabetic foot infections with concomitant osteomyelitis [see Clinical Studies (14)].
1.5 Acute Pelvic Infections Including Postpartum Endomyometritis, Septic Abortion and Post Surgical Gynecologic Infections
Ertapenem for injection is indicated for the treatment of adult patients and pediatric patients (3 months of age and older) with acute pelvic infections including postpartum endomyometritis, septic abortion and post-surgical gynecological infections due to Streptococcus agalactiae, Escherichia coli, Bacteroides fragilis, Porphyromonas asaccharolytica, Peptostreptococcus species, or Prevotella bivia.
Structured Label Content
Section 34077-8 (34077-8)
Risk Summary
Available data from a small number of post-marketing cases with Ertapenem for injection use in pregnancy are insufficient to inform any drug-associated risks for major birth defects, miscarriage, or adverse maternal or fetal outcomes. In animal reproduction studies after intravenous administration of ertapenem during the period of organogenesis, there was no evidence of developmental malformations in rats at systemic exposures (AUC) up to approximately 1.2 times the human exposure at the maximum recommended human dose (MRHD) and in mice at doses up to approximately 3 times the MRHD based on body surface area comparison. In pregnant rats administered ertapenem during organogenesis through lactation, fetal toxicity, developmental delays, and impaired reproduction did not occur in first generation offspring at systemic exposures (AUC) approximately 1.2 times the human exposure at the MRHD (see Data).
The estimated background risk of major birth defects and miscarriage for the indicated population is unknown. All pregnancies have a background risk of birth defect, loss, or other adverse outcomes. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2 to 4% and 15 to 20%, respectively.
Data
Animal Data
In pregnant rats, intravenous administration of ertapenem dosages of up to 700 mg/kg/day (approximately 1.2 times the MRHD based on AUC) during the period of organogenesis (gestation days [GD] 6-20) revealed no maternal or embryofetal effects.
Pregnant mice intravenously administered ertapenem dosages of up to 700 mg/kg/day (approximately 3 times the MRHD based on body surface area comparison) during the period of organogenesis (GD 6-15) showed slight decreases in average fetal weight and an associated decrease in the average number of ossified sacrocaudal vertebrae. There were no maternal effects at any dosage.
In a pre-postnatal study in rats, ertapenem administered to pregnant rats at dosages up to 700 mg/kg/day (approximately 1.2 times the MRHD based on AUC) during organogenesis through lactation, (GD 6 until Lactation Day (LD) 20) did not result in fetal toxicity, developmental delays, or impaired reproduction in first generation offspring, and fetal deaths and malformations were not increased in second generation offspring.
Section 42229-5 (42229-5)
Prevention
Ertapenem for injection is indicated in adults for:
1.7 Usage
To reduce the development of drug-resistant bacteria and maintain the effectiveness of Ertapenem for injection and other antibacterial drugs, Ertapenem for injection should be used only to treat or prevent infections that are proven or strongly suspected to be caused by susceptible bacteria. When culture and susceptibility information are available, they should be considered in selecting or modifying antibacterial therapy. In the absence of such data, local epidemiology and susceptibility patterns may contribute to the empiric selection of therapy.
10 Overdosage (10 OVERDOSAGE)
No specific information is available on the treatment of overdosage with Ertapenem for injection. Intentional overdosing of Ertapenem for injection is unlikely. Intravenous administration of Ertapenem for injection at a dose of 2 g over 30 min or 3 g over 1-2h in healthy adult volunteers resulted in an increased incidence of nausea. In clinical trials in adults, inadvertent administration of three 1 g doses of Ertapenem for injection in a 24 hour period resulted in diarrhea and transient dizziness in one patient. In pediatric clinical trials, a single intravenous dose of 40 mg/kg up to a maximum of 2 g did not result in toxicity.
In the event of an overdose, Ertapenem for injection should be discontinued and general supportive treatment given until renal elimination takes place.
Ertapenem for injection can be removed by hemodialysis; the plasma clearance of the total fraction of ertapenem was increased 30% in subjects with end-stage renal disease when hemodialysis (4 hour session) was performed immediately following administration. However, no information is available on the use of hemodialysis to treat overdosage.
8.2 Lactation
Risk Summary
Ertapenem is present in human milk (see
Data). There are no data on the effects on the breastfed infant or the effects on milk production. The developmental and health benefits of breastfeeding should be
considered along with the mother’s clinical need for Ertapenem for injection and any potential adverse effects on the breastfed infant from Ertapenem for injection or from the underlying maternal condition.
Data
The concentration of ertapenem in breast milk from 5 lactating women with pelvic infections (5 to 14 days postpartum) measured at random time points daily for 5 consecutive days following the last 1 g dose of intravenous therapy (3 to 10 days of therapy) showed low levels. The concentration of ertapenem in breast milk within 24 hours of the last dose of therapy in all 5 women ranged from (<0.13 (lower limit of quantitation) to 0.38 mcg/ml), although peak concentrations were not assessed. By day 5 after discontinuation of therapy, the level of ertapenem was undetectable in the breast milk of 4 women and below the lower limit of quantitation (<0.13 mcg/mL) in 1 woman. The concentration of ertapenem in transitional milk observed in this study may not reflect the concentration of ertapenem in mature milk.
11 Description (11 DESCRIPTION)
Ertapenem for injection is a sterile, synthetic, parenteral, 1-β methyl-carbapenem that is structurally related to beta-lactam antibiotics.
Chemically, Ertapenem for injection is described as [4R-[3(3S*,5S*),4α,5β,6β(R*)]]-3-[[5-[[(3carboxyphenyl)amino]carbonyl]-3-pyrrolidinyl]thio]-6-(1-hydroxyethyl)-4-methyl-7-oxo-1 azabicyclo[3.2.0]hept-2-ene-2-carboxylic acid monosodium salt. Its molecular weight is 497.50. The empirical formula is C 22H 24N 3O 7SNa, and its structural formula is:
Ertapenem sodium is a white to off-white hygroscopic, weakly crystalline powder. It is soluble in water and 0.9% sodium chloride solution, practically insoluble in ethanol, and insoluble in isopropyl acetate and tetrahydrofuran.
Ertapenem for injection is supplied as sterile lyophilized powder for intravenous infusion after reconstitution with appropriate diluent [see Dosage and Administration (2.7)] and transfer to 50 mL 0.9% Sodium Chloride Injection or for intramuscular injection following reconstitution with 1% lidocaine hydrochloride. Each single-dose vial contains 1 gram ertapenem equivalent to 1.046 grams ertapenem sodium. The sodium content is approximately 137 mg (approximately 6.0 mEq).
Each vial of Ertapenem for injection contains the following inactive ingredients: 175 mg sodium bicarbonate and sodium hydroxide to adjust pH to 7.5.
7.1 Probenecid
Probenecid interferes with the active tubular secretion of ertapenem, resulting in increased plasma concentrations of ertapenem [see Clinical Pharmacology (12.3)] . Co-administration of probenecid with ertapenem is not recommended.
16.1 How Supplied
Ertapenem for injection is supplied as a sterile lyophilized powder in single-dose vials containing ertapenem for intravenous infusion or for intramuscular injection as follows:
No. 051---1 g ertapenem equivalent
NDC 44647-051-01 in 20mL of 1 vial.
NDC 44647-051-10 in carton of 10 vials.
7.2 Valproic Acid
Case reports in the literature have shown that co-administration of carbapenems, including ertapenem, to patients receiving valproic acid or divalproex sodium results in a reduction of valproic acid concentrations. The valproic acid concentrations may drop below the therapeutic range as a result of this interaction, therefore increasing the risk of breakthrough seizures. Although the mechanism of this interaction is unknown, data from in vitro and animal studies suggest that carbapenems may inhibit the hydrolysis of valproic acid's glucuronide metabolite (VPA-g) back to valproic acid, thus decreasing the serum concentrations of valproic acid [see Warnings and Precautions (5.3)].
8.4 Pediatric Use
Safety and effectiveness of Ertapenem for injection in pediatric patients 3 months to 17 years of age are supported by evidence from adequate and well-controlled trials in adults, pharmacokinetic data in pediatric patients, and additional data from comparator-controlled trials in pediatric patients 3 months to 17 years of age [see Indications and Usage (1.1), (1.2), (1.3), (1.4) and (1.5) and Clinical Studies (14.2)] .
Ertapenem for injection is not recommended in infants under 3 months of age as no data are available.
Ertapenem for injection is not recommended in the treatment of meningitis in the pediatric population due to lack of sufficient CSF penetration.
8.5 Geriatric Use
Of the 1,835 patients in Phase 2b/3 trials treated with Ertapenem for injection, approximately 26 percent were 65 and over, while approximately 12 percent were 75 and over. No overall differences in safety or effectiveness were observed between these patients and younger patients. Other reported clinical experience has not identified differences in responses between the elderly and younger patients, but greater sensitivity of some older individuals cannot be ruled out.
This drug is known to be substantially excreted by the kidney, and the risk of toxic reactions to this drug may be greater in patients with impaired renal function. Because elderly patients are more likely to have decreased renal function, care should be taken in dose selection, and it may be useful to monitor renal function [see Dosage and Administration (2.2)].
4 Contraindications (4 CONTRAINDICATIONS)
- Ertapenem for injection is contraindicated in patients with known hypersensitivity to any component of this product or to other drugs in the same class or in patients who have demonstrated anaphylactic reactions to beta-lactams.
- Due to the use of Lidocaine HCl as a diluent, Ertapenem for injection administered intramuscularly is contraindicated in patients with a known hypersensitivity to local anesthetics of the amide type.
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following are described in greater detail in the Warnings and Precautions section.
- Hypersensitivity Reactions [see Warnings and Precautions (5.1)]
- Seizure Potential [see Warnings and Precautions (5.2)]
- Interaction with Valproic Acid [see Warnings and Precautions (5.3)]
- Clostridioides difficile-Associated Diarrhea (CDAD) [see Warnings and Precautions (5.4)]
- Caution with Intramuscular Administration [see Warnings and Precautions (5.5)]
- Development of Drug-Resistant Bacteria [see Warnings and Precautions (5.6)]
- Laboratory Tests [see Warnings and Precautions (5.7)]
7 Drug Interactions (7 DRUG INTERACTIONS)
- Co-administration with probenecid inhibits the renal excretion of ertapenem and is therefore not recommended. ( 7.1)
- The concomitant use of ertapenem and valproic acid/divalproex sodium is generally not recommended. Anti-bacterials other than carbapenems should be considered to treat infections in patients whose seizures are well controlled on valproic acid or divalproex sodium. ( 5.2, 7.2)
5.7 Laboratory Tests
While Ertapenem for injection possesses toxicity similar to the beta-lactam group of antibiotics, periodic assessment of organ system function, including renal, hepatic, and hematopoietic, is advisable during prolonged therapy.
12.3 Pharmacokinetics
Average plasma concentrations (mcg/mL) of ertapenem following a single 30-minute infusion of a 1 g intravenous (IV) dose and administration of a single 1 g intramuscular (IM) dose in healthy young adults are presented in Table 8.
| Average Plasma Concentrations (mcg/mL) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Dose/Route | 0.5 hr | 1 hr | 2 hr | 4 hr | 6 hr | 8 hr | 12 hr | 18 hr | 24 hr |
| 1 g IV
Infused at a constant rate over 30 minutes
|
155 | 115 | 83 | 48 | 31 | 20 | 9 | 3 | 1 |
| 1 g IM | 33 | 53 | 67 | 57 | 40 | 27 | 13 | 4 | 2 |
The area under the plasma concentration-time curve (AUC) of ertapenem in adults increased less-than dose-proportional based on total ertapenem concentrations over the 0.5 to 2 g dose range, whereas the AUC increased greater-than dose-proportional based on unbound ertapenem concentrations. Ertapenem exhibits non-linear pharmacokinetics due to concentration-dependent plasma protein binding at the proposed therapeutic dose [see Clinical Pharmacology (12.3)] . There is no accumulation of ertapenem following multiple IV or IM 1 g daily doses in healthy adults.
Average plasma concentrations (mcg/mL) of ertapenem in pediatric patients are presented in Table 9.
| Average Group | Dose | Average Plasma Concentrations (mcg/mL) | |||||||
|---|---|---|---|---|---|---|---|---|---|
| 0.5 hr | 1 hr | 2 hr | 4 hr | 6 hr | 8 hr | 12 hr | 24 hr | ||
| 3 to 23 months | |||||||||
| 15 mg/kg
up to a maximum dose of 1 g/day
|
103.8 | 57.3 | 43.6 | 23.7 | 13.5 | 8.2 | 2.5 | - | |
| 20 mg/kg | 126.8 | 87.6 | 58.7 | 28.4 | - | 12.0 | 3.4 | 0.4 | |
| 40 mg/kg
up to a maximum dose of 2 g/day
|
199.1 | 144.1 | 95.7 | 58.0 | - | 20.2 | 7.7 | 0.6 | |
| 2 to 12 years | |||||||||
| 15 mg/kg | 113.2 | 63.9 | 42.1 | 21.9 | 12.8 | 7.6 | 3.0 | - | |
| 20 mg/kg | 147.6 | 97.6 | 63.2 | 34.5 | - | 12.3 | 4.9 | 0.5 | |
| 40 mg/kg | 241.7 | 152.7 | 96.3 | 55.6 | - | 18.8 | 7.2 | 0.6 | |
| 13 to 17 years | |||||||||
| 20 mg/kg | 170.4 | 98.3 | 67.8 | 40.4 | - | 16.0 | 7.0 | 1.1 | |
| 1 g
Based on three patients receiving 1 g ertapenem who volunteered for pharmacokinetic assessment in one of the two safety and efficacy trials
|
155.9 | 110.9 | 74.8 | - | 24.0 | - | 6.2 | - | |
| 40 mg/kg | 255.0 | 188.7 | 127.9 | 76.2 | - | 31.0 | 15.3 | 2.1 |
5.2 Seizure Potential
Seizures and other central nervous system (CNS) adverse experiences have been reported during treatment with Ertapenem for injection [see Adverse Reactions (6.1)] . During clinical investigations in adult patients treated with Ertapenem for injection (1 g once a day), seizures, irrespective of drug relationship, occurred in 0.5% of patients during study therapy plus 14-day follow-up period [see Adverse Reactions (6.1)] . These experiences have occurred most commonly in patients with CNS disorders (e.g., brain lesions or history of seizures) and/or compromised renal function. Close adherence to the recommended dosage regimen is urged, especially in patients with known factors that predispose to convulsive activity. Anticonvulsant therapy should be continued in patients with known seizure disorders. If focal tremors, myoclonus, or seizures occur, patients should be evaluated neurologically, placed on anticonvulsant therapy if not already instituted, and the dosage of Ertapenem for injection re-examined to determine whether it should be decreased or discontinued.
1 Indications and Usage (1 INDICATIONS AND USAGE)
14.2 Pediatric Patients
Ertapenem was evaluated in pediatric patients 3 months to 17 years of age in two randomized, multicenter clinical trials.
The first trial enrolled 404 patients and compared ertapenem (15 mg/kg intravenous (IV) every 12 hours in patients 3 months to 12 years of age, and 1 g IV once a day in patients 13 to 17 years of age) to ceftriaxone (50 mg/kg/day IV in two divided doses in patients 3 months to 12 years of age and 50 mg/kg/day IV as a single daily dose in patients 13 to 17 years of age) for the treatment of complicated urinary tract infection (UTI), skin and soft tissue infection (SSTI), or community-acquired pneumonia (CAP). Both regimens allowed the option to switch to oral amoxicillin/clavulanate for a total of up to 14 days of treatment (parenteral and oral). The microbiological success rates in the evaluable per protocol (EPP) analysis in patients treated for UTI were 87.0% (40/46) for ertapenem and 90.0% (18/20) for ceftriaxone. The clinical success rates in the EPP analysis in patients treated for SSTI were 95.5% (64/67) for ertapenem and 100% (26/26) for ceftriaxone, and in patients treated for CAP were 96.1% (74/77) for ertapenem and 96.4% (27/28) for ceftriaxone.
The second trial enrolled 112 patients and compared ertapenem (15 mg/kg IV every 12 hours in patients 3 months to 12 years of age, and 1 g IV once a day in patients 13 to 17 years of age) to ticarcillin/clavulanate (50 mg/kg for patients <60 kg or 3.0 g for patients >60 kg, 4 or 6 times a day) up to 14 days for the treatment of complicated intra-abdominal infections (IAI) and acute pelvic infections (API). In patients treated for IAI (primarily patients with perforated or complicated appendicitis), the clinical success rates were 83.7% (36/43) for ertapenem and 63.6% (7/11) for ticarcillin/clavulanate in the EPP analysis. In patients treated for API (post-operative or spontaneous obstetrical endomyometritis, or septic abortion), the clinical success rates were 100% (23/23) for ertapenem and 100% (4/4) for ticarcillin/clavulanate in the EPP analysis.
12.1 Mechanism of Action
Ertapenem sodium is a carbapenem antibiotic [see Clinical Pharmacology (12.4)].
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Serious hypersensitivity (anaphylactic) reactions have been reported in patients receiving β-lactams. ( 5.1)
- Seizures and other central nervous system adverse experiences have been reported during treatment. ( 5.2)
- Co-administration of Ertapenem for injection with valproic acid or divalproex sodium reduces the serum concentration of valproic acid potentially increasing the risk of breakthrough seizures. ( 5.3)
- Clostridioides difficile-associated diarrhea (ranging from mild diarrhea to fatal colitis): Evaluate if diarrhea occurs. ( 5.4)
- Caution should be taken when administering Ertapenem for injection intramuscularly to avoid inadvertent injection into a blood vessel. ( 5.5)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
Do not mix or co-infuse Ertapenem for injection with other medications. Do not use diluents containing dextrose (α –D–glucose). ( 2.1)
Ertapenem for injection should be infused over 30 minutes in both the Treatment and Prophylactic regimens. ( 2.1)
Dosing considerations should be made in adults with advanced or end-stage renal impairment and those on hemodialysis. ( 2.4, 2.5)
Treatment regimen:
- Adults and pediatric patients 13 years of age and older. The dosage should be 1 gram once a day intravenously or intramuscularly. ( 2.2)
- Patients 3 months to 12 years of age should be administered 15 mg/kg twice daily (not to exceed 1 g/day intravenously or intramuscularly.) ( 2.2)
- Intravenous infusion may be administered in adults and pediatrics for up to 14 days or intramuscular injection for up to 7 days. ( 2.1)
Prophylaxis regimen for adults:
- 1 gram single dose given 1 hour prior to elective colorectal surgery. ( 2.3)
2.5 Patients On Hemodialysis (2.5 Patients on Hemodialysis)
When adult patients on hemodialysis are given the recommended daily dose of 500 mg of Ertapenem for injection within 6 hours prior to hemodialysis, a supplementary dose of 150 mg is recommended following the hemodialysis session. If Ertapenem for injection is given at least 6 hours prior to hemodialysis, no supplementary dose is needed. There are no data in patients undergoing peritoneal dialysis or hemofiltration. There are no data in pediatric patients on hemodialysis.
When only the serum creatinine is available, the following formula
| Males: | (weight in kg) × (140-age in years) |
| (72) × serum creatine (mg/100 mL) | |
| Females: | (0.85) × (value calculated for males) |
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
- For injection: Single-dose vial 1 gram. ( 3)
5.4 Clostridioides Difficile (5.4 Clostridioides difficile)
CDAD has been reported with use of nearly all antibacterial agents, including ertapenem, and may range in severity from mild diarrhea to fatal colitis. Treatment with antibacterial agents alters the normal flora of the colon leading to overgrowth of Clostridioides difficile.
Clostridioides difficile produces toxins A and B which contribute to the development of CDAD. Hypertoxin producing strains of Clostridioides difficile cause increased morbidity and mortality, as these infections can be refractory to antimicrobial therapy and may require colectomy. CDAD must be considered in all patients who present with diarrhea following antibiotic use. Careful medical history is necessary since CDAD has been reported to occur over two months after the administration of antibacterial agents.
If CDAD is suspected or confirmed, ongoing antibiotic use not directed against Clostridioides difficile may need to be discontinued. Appropriate fluid and electrolyte management, protein supplementation, antibiotic treatment of Clostridioides difficile, and surgical evaluation should be instituted as clinically indicated.
6.2 Post Marketing Experience (6.2 Post-Marketing Experience)
The following additional adverse reactions have been identified during the post-approval use of Ertapenem for injection. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Gastrointestinal Disorders: teeth staining
Immune System Disorders: anaphylaxis including anaphylactoid reactions
Musculoskeletal and Connective Tissue Disorders: muscular weakness
Nervous System Disorders: coordination abnormal, depressed level of consciousness, dyskinesia, gait disturbance, myoclonus, tremor, encephalopathy (recovery was prolonged in patients with renal impairment)
Psychiatric Disorders: altered mental status (including aggression, delirium), hallucinations
Skin and Subcutaneous Tissue Disorders: Acute Generalized Exanthematous Pustulosis (AGEP), Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS syndrome), hypersensitivity vasculitis
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
17.1 Instructions for Patients
Patients should be advised that allergic reactions, including serious allergic reactions could occur and that serious reactions may require immediate treatment. Advise patients to report any previous hypersensitivity reactions to Ertapenem for injection, other beta-lactams or other allergens.
Patients should be counseled to inform their physician if they are taking valproic acid or divalproex sodium. Valproic acid concentrations in the blood may drop below the therapeutic range upon co-administration with Ertapenem for injection. If treatment with Ertapenem for injection is necessary and continued, alternative or supplemental anti-convulsant medication to prevent and/or treat seizures may be needed.
Patients should be counseled that antibacterial drugs including Ertapenem for injection should only be used to treat bacterial infections. They do not treat viral infections (e.g., the common cold). When Ertapenem for injection is prescribed to treat a bacterial infection, patients should be told that although it is common to feel better early in the course of therapy, the medication should be taken exactly as directed. Skipping doses or not completing the full course of therapy may (1) decrease the effectiveness of the immediate treatment and (2) increase the likelihood that bacteria will develop resistance and will not be treatable by Ertapenem for injection or other antibacterial drugs in the future.
Diarrhea is a common problem caused by antibiotics which usually ends when the antibiotic is discontinued. Sometimes after starting treatment with antibiotics, patients can develop watery and bloody stools (with or without stomach cramps and fever) even as late as two or more months after having taken the last dose of the antibiotic. If this occurs, patients should contact their physician as soon as possible.
5.1 Hypersensitivity Reactions
Serious and occasionally fatal hypersensitivity (anaphylactic) reactions have been reported in patients receiving therapy with beta-lactams. These reactions are more likely to occur in individuals with a history of sensitivity to multiple allergens. There have been reports of individuals with a history of penicillin hypersensitivity who have experienced severe hypersensitivity reactions when treated with another betalactam. Before initiating therapy with Ertapenem for injection, careful inquiry should be made concerning previous hypersensitivity reactions to penicillins, cephalosporins, other beta-lactams and other allergens. If an allergic reaction to Ertapenem for injection occurs, discontinue the drug immediately. Serious anaphylactic reactions require immediate emergency treatment as clinically indicated.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
1.3 Community Acquired Pneumonia
Ertapenem for injection is indicated for the treatment of adult patients and pediatric patients (3 months of age and older) with community acquired pneumonia due to Streptococcus pneumoniae (penicillin susceptible isolates only) including cases with concurrent bacteremia, Haemophilus influenzae (beta-lactamase negative isolates only), or Moraxella catarrhalis.
2.3 Prophylactic Regimen in Adults
Table 2 presents prophylaxis guidelines for Ertapenem for injection.
| Indication | Daily Dose
(IV) Adults |
Recommended Duration of Total Antimicrobial Treatment |
|---|---|---|
| Prophylaxis of surgical site infection following elective colorectal surgery | 1 g | Single intravenous dose given 1 hour prior to surgical incision |
2.4 Patients With Renal Impairment (2.4 Patients with Renal Impairment)
Ertapenem for injection may be used for the treatment of infections in adult patients with renal impairment. In patients whose creatinine clearance is >30 mL/min/1.73 m 2, no dosage adjustment is necessary. Adult patients with severe renal impairment (creatinine clearance ≤30 mL/min/1.73 m 2) and end-stage renal disease (creatinine clearance ≤10 mL/min/1.73 m 2) should receive 500 mg daily. A supplementary dose of 150 mg is recommended if ertapenem is administered within 6 hours prior to hemodialysis. There are no data in pediatric patients with renal impairment.
5.3 Interaction With Valproic Acid (5.3 Interaction with Valproic Acid)
Case reports in the literature have shown that co-administration of carbapenems, including ertapenem, to patients receiving valproic acid or divalproex sodium results in a reduction in valproic acid concentrations. The valproic acid concentrations may drop below the therapeutic range as a result of this interaction, therefore increasing the risk of breakthrough seizures. Increasing the dose of valproic acid or divalproex sodium may not be sufficient to overcome this interaction. The concomitant use of ertapenem and valproic acid/divalproex sodium is generally not recommended. Anti-bacterials other than carbapenems should be considered to treat infections in patients whose seizures are well controlled on valproic acid or divalproex sodium. If administration of Ertapenem for injection is necessary, supplemental anticonvulsant therapy should be considered [see Drug Interactions (7.2)].
8.6 Patients With Renal Impairment (8.6 Patients with Renal Impairment)
Dosage adjustment is necessary in patients with creatinine clearance 30 mL/min or less [see Dosage and Administration (2.4) and Clinical Pharmacology (12.3)] .
2.6 Patients With Hepatic Impairment (2.6 Patients with Hepatic Impairment)
No dose adjustment recommendations can be made in patients with hepatic impairment [see Use in Specific Populations (8.7) and Clinical Pharmacology (12.3)].
8.7 Patients With Hepatic Impairment (8.7 Patients with Hepatic Impairment)
The pharmacokinetics of ertapenem in patients with hepatic impairment have not been established. Of the total number of patients in clinical trials, 37 patients receiving ertapenem 1 g daily and 36 patients receiving comparator drugs were considered to have Child-Pugh Class A, B, or C liver impairment. The incidence of adverse experiences in patients with hepatic impairment was similar between the ertapenem group and the comparator groups.
Principal Display Panel 1 G Vial Label (PRINCIPAL DISPLAY PANEL - 1 g Vial Label)
NDC 44647-051-01
Ertapenem
for Injection
1 gram/vial
For Intravenous
or Intramuscular Use
Rx only
Principal Display Panel 10 Vial Carton (PRINCIPAL DISPLAY PANEL - 10 Vial Carton)
NDC 44647-051-10
Ertapenem for Injection
1 gram/vial
For Intravenous Or Intramuscular Use
R x Only
10 x 1 g Single dose vials
Each vial contains:
1.046 grams ertapenem sodium, equiv. to 1 gram ertapenem.
Prior to Constitution: Store lyophilized powder below 25°C (77°F).
1.1 Complicated Intra Abdominal Infections (1.1 Complicated Intra-Abdominal Infections)
Ertapenem for injection is indicated for the treatment of adult patients and pediatric patients (3 months of age and older) with complicated intra-abdominal infections due to Escherichia coli, Clostridium clostridioforme, Eubacterium lentum, Peptostreptococcus species, Bacteroides fragilis, Bacteroides distasonis, Bacteroides ovatus, Bacteroides thetaiotaomicron, or Bacteroides uniformis.
13.2 Animal Toxicology And/or Pharmacology (13.2 Animal Toxicology and/or Pharmacology)
In repeat-dose studies in rats, treatment-related neutropenia occurred at every dose-level tested, including the lowest dose of 2 mg/kg (approximately 2% of the human dose on a body surface area basis).
Studies in rabbits and Rhesus monkeys were inconclusive with regard to the effect on neutrophil counts.
5.6 Development of Drug Resistant Bacteria (5.6 Development of Drug-Resistant Bacteria)
As with other antibiotics, prolonged use of Ertapenem for injection may result in overgrowth of non-susceptible organisms. Repeated evaluation of the patient's condition is essential. If superinfection occurs during therapy, appropriate measures should be taken.
Prescribing Ertapenem for injection in the absence of a proven or strongly suspected bacterial infection or a prophylactic indication is unlikely to provide benefit to the patient and increases the risk of the development of drug-resistant bacteria.
5.5 Caution With Intramuscular Administration (5.5 Caution with Intramuscular Administration)
Caution should be taken when administering Ertapenem for injection intramuscularly to avoid inadvertent injection into a blood vessel [see Dosage and Administration (2.7)] .
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenesis and Mutagenesis
No long-term studies in animals have been performed to evaluate the carcinogenic potential of ertapenem.
Ertapenem was not genotoxic in in vitro or in vivo assays, including: alkaline elution/rat hepatocyte assay, chromosomal aberration assay in Chinese hamster ovary cells, a TK6 human lymphoblastoid cell mutagenesis assay and a mouse micronucleus assay.
Impairment of Fertility
In rats, intravenous dosages up to 700 mg/kg/day (approximately 1.2 times the human exposure at the human recommended human dose of 1 g based on plasma AUC) did not impair fertility.
1.4 Complicated Urinary Tract Infections Including Pyelonephritis
Ertapenem for injection is indicated for the treatment of adult patients and pediatic patients (3 months of age and older) with complicated urinary tract infections including pyelonephritis due to Escherichia coli, including cases with concurrent bacteremia, or Klebsiella pneumoniae.
1.6 Prophylaxis of Surgical Site Infection Following Elective Colorectal Surgery
Ertapenem for injection is indicated in adults for the prevention of surgical site infection following elective colorectal surgery.
1.2 Complicated Skin and Skin Structure Infections, Including Diabetic Foot Infections Without Osteomyelitis (1.2 Complicated Skin and Skin Structure Infections, Including Diabetic Foot Infections without Osteomyelitis)
Ertapenem for injection is indicated for the treatment of adult patients and pediatric patients (3 months of age and older) with complicated skin and skin structure infections, including diabetic foot infections without osteomyelitis due to Staphylococcus aureus (methicillin susceptible isolates only), Streptococcus agalactiae, Streptococcus pyogenes, Escherichia coli, Klebsiella pneumoniae, Proteus mirabilis, Bacteroides fragilis, Peptostreptococcus species, Porphyromonas asaccharolytica, or Prevotella bivia. Ertapenem for injection has not been studied in diabetic foot infections with concomitant osteomyelitis [see Clinical Studies (14)].
1.5 Acute Pelvic Infections Including Postpartum Endomyometritis, Septic Abortion and Post Surgical Gynecologic Infections (1.5 Acute Pelvic Infections Including Postpartum Endomyometritis, Septic Abortion and Post-Surgical Gynecologic Infections)
Ertapenem for injection is indicated for the treatment of adult patients and pediatric patients (3 months of age and older) with acute pelvic infections including postpartum endomyometritis, septic abortion and post-surgical gynecological infections due to Streptococcus agalactiae, Escherichia coli, Bacteroides fragilis, Porphyromonas asaccharolytica, Peptostreptococcus species, or Prevotella bivia.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:39:25.848489 · Updated: 2026-03-14T21:57:49.801735