These Highlights Do Not Include All The Information Needed To Use Tizanidine Hydrochloride Capsules Safely And Effectively. See Full Prescribing Information For Tizanidine Hydrochloride Capsules.
c4a5b305-4265-4f90-a9ff-c16c0b936a06
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Tizanidine is a central alpha-2-adrenergic agonist indicated for the management of spasticity. Because of the short duration of therapeutic effect, treatment with tizanidine should be reserved for those daily activities and times when relief of spasticity is most important [see Dosage and Administration (2.1) ].
Indications and Usage
Tizanidine is a central alpha-2-adrenergic agonist indicated for the management of spasticity. Because of the short duration of therapeutic effect, treatment with tizanidine should be reserved for those daily activities and times when relief of spasticity is most important [see Dosage and Administration (2.1) ].
Dosage and Administration
Recommended starting dose: 2 mg; dose can be repeated at 6 to 8 hour intervals, up to a maximum of 3 doses in 24 hours ( 2.1 ) Dosage can be increased by 2 mg to 4 mg per dose, wHh 1 to 4 days between increases; total daily dose should not exceed 36 mg ( 2.1 ) Tizanidine pharmacokinetics differs between tablets and capsules, and when taken with or without food. These differences could result in a change in tolerability and control of symptoms ( 2.1 , 12.3 ) To discontinue tizanidine, decrease dose slowly to minimize the risk of withdrawal and rebound hypertension, tachycardia, and hypertonia ( 2.2 )
Warnings and Precautions
Hypotension: monitor for signs and symptoms of hypotension, in particular in patients receiving concurrent anti hypertensives; tizanidine should not be used with other a 2 adrenergic agonists ( 5.1 , 7. 7 ) Risk of liver injury: monitor ALTs; discontinue tizanidine if liver injury occurs ( 5.2 ) Sedation: Tizanidine may interfere with everyday activities; sedative effects of tizanidine, alcohol, and other CNS depressants are additive ( 5.3 , 7.5 , 7.6 ) Hallucinations: consider discontinuation of tizanidine ( 5.4 ) Less potent inhibitors of CYP1 A2: may cause hypotension, bradycardia, or excessive drowsiness, use caution if tizanidine is used with less potent inhibitors of CYP1A2, e.g., zileuton, other fluoroquinolones, antiarrythmics, cimetidine, famotidine, oral contraceptives, acyclovir, and ficlopidine ( 5.5 , 7.3 , 12.3 ) Renal impairment (creatinine clearance< 25 ml/min): use tizanidine with caution, and monitor closely for dry mouth, somnolence, asthenia and dizziness as indicators of potential overdose ( 5.7 )
Contraindications
Tizanidine is contraindicated in patients taking potent inhibitors of CYP1A2, such as fluvoxamine or ciprofloxacin [see Drug Interactions (7.1, 7.2) ].
Adverse Reactions
Tizanidine should be used with caution in patients with renal insufficiency (creatinine clearance < 25 ml/min), as clearance is reduced by more than 50%. In these patients, during titration, the individual doses should be reduced. If higher doses are required, individual doses rather than dosing frequency should be increased. These patients should be monitored closely for the onset or increase in severity of the common adverse events (dry mouth, somnolence, asthenia and dizziness) as indicators of potential overdose. [see Dosage and Administration (2.2) and Use in Specific Populations (8.6) ].
Drug Interactions
Concomitant use of fluvoxamine and tizanidine is contraindicated. Changes in pharmacokinetics of tizanidine when administered with fluvoxamine resulted in significantly decreased blood pressure, increased drowsiness, and increased psychomotor impairment. [see Contraindications (4) and Clinical Pharmacology (12.3) ].
Medication Information
Warnings and Precautions
Hypotension: monitor for signs and symptoms of hypotension, in particular in patients receiving concurrent anti hypertensives; tizanidine should not be used with other a 2 adrenergic agonists ( 5.1 , 7. 7 ) Risk of liver injury: monitor ALTs; discontinue tizanidine if liver injury occurs ( 5.2 ) Sedation: Tizanidine may interfere with everyday activities; sedative effects of tizanidine, alcohol, and other CNS depressants are additive ( 5.3 , 7.5 , 7.6 ) Hallucinations: consider discontinuation of tizanidine ( 5.4 ) Less potent inhibitors of CYP1 A2: may cause hypotension, bradycardia, or excessive drowsiness, use caution if tizanidine is used with less potent inhibitors of CYP1A2, e.g., zileuton, other fluoroquinolones, antiarrythmics, cimetidine, famotidine, oral contraceptives, acyclovir, and ficlopidine ( 5.5 , 7.3 , 12.3 ) Renal impairment (creatinine clearance< 25 ml/min): use tizanidine with caution, and monitor closely for dry mouth, somnolence, asthenia and dizziness as indicators of potential overdose ( 5.7 )
Indications and Usage
Tizanidine is a central alpha-2-adrenergic agonist indicated for the management of spasticity. Because of the short duration of therapeutic effect, treatment with tizanidine should be reserved for those daily activities and times when relief of spasticity is most important [see Dosage and Administration (2.1) ].
Dosage and Administration
Recommended starting dose: 2 mg; dose can be repeated at 6 to 8 hour intervals, up to a maximum of 3 doses in 24 hours ( 2.1 ) Dosage can be increased by 2 mg to 4 mg per dose, wHh 1 to 4 days between increases; total daily dose should not exceed 36 mg ( 2.1 ) Tizanidine pharmacokinetics differs between tablets and capsules, and when taken with or without food. These differences could result in a change in tolerability and control of symptoms ( 2.1 , 12.3 ) To discontinue tizanidine, decrease dose slowly to minimize the risk of withdrawal and rebound hypertension, tachycardia, and hypertonia ( 2.2 )
Contraindications
Tizanidine is contraindicated in patients taking potent inhibitors of CYP1A2, such as fluvoxamine or ciprofloxacin [see Drug Interactions (7.1, 7.2) ].
Adverse Reactions
Tizanidine should be used with caution in patients with renal insufficiency (creatinine clearance < 25 ml/min), as clearance is reduced by more than 50%. In these patients, during titration, the individual doses should be reduced. If higher doses are required, individual doses rather than dosing frequency should be increased. These patients should be monitored closely for the onset or increase in severity of the common adverse events (dry mouth, somnolence, asthenia and dizziness) as indicators of potential overdose. [see Dosage and Administration (2.2) and Use in Specific Populations (8.6) ].
Drug Interactions
Concomitant use of fluvoxamine and tizanidine is contraindicated. Changes in pharmacokinetics of tizanidine when administered with fluvoxamine resulted in significantly decreased blood pressure, increased drowsiness, and increased psychomotor impairment. [see Contraindications (4) and Clinical Pharmacology (12.3) ].
Description
Tizanidine is a central alpha-2-adrenergic agonist indicated for the management of spasticity. Because of the short duration of therapeutic effect, treatment with tizanidine should be reserved for those daily activities and times when relief of spasticity is most important [see Dosage and Administration (2.1) ].
Section 34077-8
Pregnancy Category C
Tizanidine has not been studied in pregnant women. Tizanidine should be given to pregnant women only if the benefit outweighs the risk to the unborn fetus. Reproduction studies performed in rats at a dose of 3 mg/kg, equal to the maximum recommended human dose on a mg/m' basis, and in rabbits at 30 mg/kg, 16 times the maximum recommended human dose on a mg/m2 basis, did not show evidence of teratogeniclty. Tizanidine at doses that are equal to and up to 8 times the maximum recommended human dose on a mg/m2 basis increased gestation duration in rats. Prenatal and postnatal pup loss was increased and developmental retardation occurred. Post-implantation loss was increased in rabbits at doses of 1 mg/kg or greater, equal to or greater than 0.5 times the maximum recommended human dose on a mg/m2 basis.
Section 42229-5
Capsules
2 mg: Light yellow powder filled in size "3" hard gelatin capsule wHh opaque light blue cap imprinted "2 MG" and opaque light blue body
4 mg: Light yellow powder filled in size "3" hard gelatin capsule with opaque blue cap imprinted "4 MG" and opaque white body
6 mg: Light yellow powder filled in size "2" hard gelatin capsule with opaque blue cap imprinted "6 MG" and opaque blue body
Section 44425-7
Store at 25°G (77°F); excursions permitted between 15° to 30°G (59° to 86°F).
[See USP Controlled Room Temperature].
Dispense in containers with child resistant closure.
7. 7 A2
Because hypotensive effects may be cumulative, it is not recommended that tizanidine be used with other a2 -adrenergic agonists. [see Warnings and Precautions (5.1)].
9.2 Abuse
Abuse potential was not evaluated in human studies. Rats were able to distinguish tizanidine from saline in a standard discrimination paradigm, after training, but failed to generalize the effects of morphine, cocaine, diazepam, or phenobarbital to tizanidine.
7.5 Alcohol
Alcohol increases the overall amount of drug in the bloodstream after a dose of tizanidine. This was associated with an increase in adverse reactions of tizanidine. The CNS depressant effects of tizanidine and alcohol are additive. [see Clinical Pharmacology (12.3)].
5.3 Sedation
Tizanidine can cause sedation, which may interfere with everyday activity. In the multiple dose studies, the prevalence of patients with sedation peaked following the first week of titration and then remained stable for the duration of the maintenance phase of the study. The CNS depressant effects of tizanidine with alcohol and other CNS depressants (e.g., benzodiazepines, opioids, tricyclic antidepressants) may be additive. Monitor patients who take tizanidine with another CNS depressant 1or symptoms of excess sedation. [see Drug lnteracllons (7.5, 7.6)]
10 Overddsage
A review of the safety surveillance database revealed cases of intentional and accidental tizanidine overdose. Some of the cases resulted in fatality and many of the intentional overdoses were with multiple drugs including CNS depressants. The clinical manifestations of tizanidine overdose were consistent with its known pharmacology. In the majority of cases a decrease in sensorium was observed including lethargy, somnolence, confusion and coma. Depressed cardiac function is also observed including most often bradycardia and hypotension. Respiratory depression is another common feature of tizanidine overdose.
Should overdose occur, basic steps to ensure the adequacy of an airway and the monitoring of cardiovascular and respiratory systems should be undertaken. Tizanidine is a lipid-soluble drug, which is only slightly soluble in water and methanol. Therefore, dialysis is not likely to be an efficient method of removing drug from the body. In general, symptoms resolve within one to three days following discontinuation of tizanidine and administration of appropriate therapy. Due to the similar mechanism of action, symptoms and management of tizanidine overdose are similar to that following clonidine overdose. For the most recent information concerning the management of overdose, contact a poison control center.
11 Description
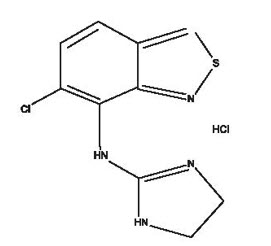
Tizanidine hydrochloride is a central alpha2-adrenergic agonist. Tizanidine HCl is a white to slight⍰ yellow crystalline powder. Tizanidine is soluble in water and methanol. Its chemical name is 5-chloro-4-(2-imidazolin-2-ylamino)-2, 1 ,3-benzothiadiazole hydrochloride. Tizanidine's molecular formula is C9H8CIN5S-HCl, its molecular weight is 290.2 and its structural formula is:
Tizanidine hydrochloride capsules are supplied as 2, 4, and 6 mg capsules for oral administration. Tizanidine hydrochloride capsules contain the active ingredient, tizanidine hydrochloride (2.29 mg equivalent to 2 mg tizanidine base, 4.58 mg equivalent to 4 mg tizanidine base, and 6.87 mg equivalent to 6 mg tizanidine base), and the inactive ingredients, lactose monohydrate, hypromellose, microcrystalline cellulose and colloidal silicon dioxide. Each capsule shell contains FD & C Blue t, FD & C Red 3, titanium dioxide and gelatin. The capsules are printed with SW-0012 while ink which contains shellac, dehydrated alcohol, isopropyl alcohol, butyl alcohol, propylene glycol, strong ammonia solution, potassium hydroxide, and titanium dioxide.
9.3 Dependence
Tizanidine is closely related to clonidine, which is often abused in combination with narcotics and is known to cause symptoms of rebound upon abrupt withdrawal. Three cases of rebound symptoms on sudden withdrawal of tizanidine have been reported. The case reports suggest that these patients were also misusing narcotics. Withdrawal symptoms included hypertension, tachycardia, hypertonia, tremor, and anxiety. Withdrawal symptoms are more likely to occur in cases where high doses are used, especially for prolonged periods, or with concomitant use of narcotics. Lf therapy needs to be discontinued, the dose should be decreased slowly to minimize the risk of withdrawal symptoms [see Dosage and Administration (2.2)].
Monkeys were shown to self-administer tizanidine in a dose-dependent manner, and abrupt cessation of tizanidine produced transient signs of withdrawal at doses > 35 times the maximum recommended human dose on a mg/m2 basis. These transient withdrawal signs (increased locomotion, body twitching, and aversive behavior toward the observer) were not reversed by naloxone administration.
5.1 Hypotension
Tizanidine is an a2-adrenergic agonist that can produce hypotension. Syncope has been reported in the post marketing setting. The chance of significant hypotension may possibly be minimized by titration of the dose and by focusing attention on signs and symptoms of hypotension prior to dose advancement. In addition, patients moving from a supine to fixed upright position may be at increased risk for hypotension and orthostatic effects.
Monitor for hypotension when tizanidine is used in patients receiving concurrent antihypertensive therapy. It is not recommended that tizanidine be used with other a2 -adrenergic agonists. Clinically significant hypotension (decreases in both systolic and diastolic pressure) has been reported with concomitant administration of either fluvoxamine or ciprofloxacin and single doses of 4 mg of tizanidine. Therefore, concomitant use of 1izanidine with fluvoxamine or with ciprofloxacin, potent inhibitors of CYP1A2, is contraindicated [see Contraindications (4) and Drug Interactions (1.1, 7.2)].
7.2 Ciprofloxacin
Concomitant use of ciprofloxacin and tizanidine is contraindicated. Changes in pharmacokinetics of tizanidine when administered with ciprofloxacin resulted in significantly decreased blood pressure, increased drowsiness, and increased psychomotor impairment [See Contraindications (4) and Clinical Pharmacology (12.3)].
8.4 Pediatric Use
Safety and effectiveness in pediatric patients have not been established.
8.5 Geriatric Use
Tizanidine is known to be substantially excreted by the kidney, and the risk of adverse reactions to this drug may be greater in patients with impaired renal function. Because elderly patients are more likely to have decreased renal function, care should be taken in dose selection, and it may be useful to monitor renal function. Clinical studies of tizanidine did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently than younger subjects. Cross-study comparison of pharmacokinetic data following single dose administration of 6 mg tizanidine showed that younger subjects cleared the drug four times faster than the elderly subjects. In elderly patients with renal insufficiency (creatinine clearance <25 mL/min), tizanidine clearance is reduced by more than 50% compared to healthy elderly subjects; this would be expected to lead to a longer duration of clinical effect. During titration, the individual doses should be reduced. If higher doses are required, individual doses rather than dosing frequency should be increased. Monitor elderly patients because they may have an increased risk for adverse reactions associated with tizanidine.
14 Clinical Studies
Tizanidine's capacity to reduce increased muscle tone associated with spasticity was demonstrated in two adequate and well controlled studies in patients with multiple sclerosis or spinal cord injury (Studies t and 2).
4 Contraindications
Tizanidine is contraindicated in patients taking potent inhibitors of CYP1A2, such as fluvoxamine or ciprofloxacin [see Drug Interactions (7.1, 7.2)].
6 Adverse Reactions
The following adverse reactions are described elsewhere in other sections of the prescribing information:
- Hypotension [see Warnings and Precaullons (5.1)]
- Liver Injury [see Warnings and Precautions (5.2)]
- Sedation [see Wamings and Precautions (5.3)]
- Hallucinosis/Psychotic-Like Symptoms [see Warnings and Precautions (5.4)]
- Hypersensitivity Reactions [see Warnings and Precautions (5.6)]
7 Drug Interactions
Concomitant use of fluvoxamine and tizanidine is contraindicated. Changes in pharmacokinetics of tizanidine when administered with fluvoxamine resulted in significantly decreased blood pressure, increased drowsiness, and increased psychomotor impairment. [see Contraindications (4) and Clinical Pharmacology (12.3)].
8.3 Nursing Mothers
It is not known whether this drug is excreted in human milk. Because many drugs are excreted in human milk, caution should be exercised when tizanidine is administered to a nursing woman.
2.1 Dosing Lnformation
Tizanidine hydrochloride capsules may be prescribed with or without food. Once the formulation has been selected and the decision to take with or without food has been made, this regimen should not be altered.
Food has complex effects on tizanidine pharmacokinetics, which differ with the different formulations. Tizanidine hydrochloride capsules and tizanidine hydrochloride tablets are bioequivalent to each other under fasting conditions (more than 3 hours after a meal), but not under fed conditions (within 30 minutes of a meal). These pharmacokinetic differences may result in clinically significant differences when switching administration of tablet and capsules and when switching administration between the fed or fasted state.
These changes may result in increased adverse events, or delayed or more rapid onset of activity, depending upon the nature of the switch. For this reason, the prescriber should be thoroughly familiar with the changes in kinetics associated with these different conditions [see Clinical Pharmacology (12.3)].
The recommended starting dose is 2 mg. Because the effect of tizanidine peaks at approximately 1 to 2 hours post-dose and dissipates between 3 to 6 hours post-dose, treatment can be repeated at 6 to 8 hour intervals, as needed, to a maximum of three doses in 24 hours.
Dosage can be gradually increased by 2 mg to 4 mg at each dose, with 1 to 4 days between dosage increases, until a satisfactory reduction of muscle tone is achieved. The total daily dose should not exceed 36 mg. Single doses greater than 16 mg have not been studied.
1 Indications and Usage
Tizanidine is a central alpha-2-adrenergic agonist indicated for the management of spasticity. Because of the short duration of therapeutic effect, treatment with tizanidine should be reserved for those daily activities and times when relief of spasticity is most important [see Dosage and Administration (2.1)].
7.4 Oral Contraceptives
Concomitant use of tizanidine with oral contraceptives is not recommended. However, if concomitant use is clinically necessary, initiate tizanidine with a single 2 mg dose and increase in 2-4 mg steps daily based on patient response to therapy. 11 adverse reactions such as hypotension, bradycardia, or excessive drowsiness occur, reduce or discontinue tizanidine therapy. [see Clinical Pharmacology (12.3)].
12.1 Mechanism of Action
Tizanidine is a central alpha-2-adrenergic receptor agonist and presumably reduces spasticity by increasing presynaptic inhibition of motor neurons. The effects of tizanidine are greatest on polysynaptic pathways. The overall effect of these actions is thought to reduce facilitation of spinal motor neurons.
2.4 Drug Discontinuation
If therapy needs to be discontinued, particularly in patients who have been receiving high doses (20 mg to 36 mg daily) for long periods (9 weeks or more) or who may be on concomitant treatment with narcotics, the dose should be decreased slowly (2 mg to 4 mg per day) to minimize the risk of withdrawal and rebound hypertension, tachycardia, and hypertonia [see Drug Abuse and Dependence (9.3)].
5.2 Risk of Liver Injury
Tizanidine may cause hepatocellular liver injury. Tizanidine should be used with caution in patients with any hepatic impairment. Monitoring of aminotransferase levels is recommended for baseline and 1 month after maximum dose is achieved, or if hepatic injury is suspected. [see Dosage and Administration (2.3) and Use in Specific Populations (8. 7)].
7.6 Other Cns Depressants
The sedative effects of tizanidine with CNS depressants (e.g., benzodiazepines, opioids, tricyclic antidepressants) may be additive. Monitor patients who take tizanidine with another CNS depressant for symptoms of excess sedation. [see Clinical Pharmacology (12.3)].
5 Warnings and Precautions
- Hypotension: monitor for signs and symptoms of hypotension, in particular in patients receiving concurrent anti hypertensives; tizanidine should not be used with other a2 adrenergic agonists (5.1, 7. 7)
- Risk of liver injury: monitor ALTs; discontinue tizanidine if liver injury occurs (5.2)
- Sedation: Tizanidine may interfere with everyday activities; sedative effects of tizanidine, alcohol, and other CNS depressants are additive (5.3, 7.5, 7.6)
- Hallucinations: consider discontinuation of tizanidine (5.4)
- Less potent inhibitors of CYP1 A2: may cause hypotension, bradycardia, or excessive drowsiness, use caution if tizanidine is used with less potent inhibitors of CYP1A2, e.g., zileuton, other fluoroquinolones, antiarrythmics, cimetidine, famotidine, oral contraceptives, acyclovir, and ficlopidine (5.5, 7.3, 12.3)
- Renal impairment (creatinine clearance< 25 ml/min): use tizanidine with caution, and monitor closely for dry mouth, somnolence, asthenia and dizziness as indicators of potential overdose (5.7)
2 Dosage and Administration
- Recommended starting dose: 2 mg; dose can be repeated at 6 to 8 hour intervals, up to a maximum of 3 doses in 24 hours (2.1)
- Dosage can be increased by 2 mg to 4 mg per dose, wHh 1 to 4 days between increases; total daily dose should not exceed 36 mg (2.1)
- Tizanidine pharmacokinetics differs between tablets and capsules, and when taken with or without food. These differences could result in a change in tolerability and control of symptoms (2.1, 12.3)
- To discontinue tizanidine, decrease dose slowly to minimize the risk of withdrawal and rebound hypertension, tachycardia, and hypertonia (2.2)
8.6 Impaired Renal Function
Tizanidine is known to be substantially excreted by the kidney, and the risk of adverse reactions to this drug may be greater in patients with impaired renal function. In patients with renal insufficiency (creatinine clearance < 25 ml/min) clearance was reduced by more than 50%. In these patients, during titration, the individual doses should be reduced. If higher doses are required, individual doses rather than dosing frequency should be increased. These patients should be monitored closely for the onset or increase in severity of the common adverse events (dry mouth, somnolence, asthenia and dizziness) as indicators of potential overdose. [see Dosage and Administration (2.2), Warnings and Precautions (5. 7) and Clinical Pharmacology (12.3)].
3 Dosage Forms and Strengths
- Capsules: 2 mg, 4 mg or 6 mg (3)
6.2 Post Marketing Experience
The following adverse reactions have been identified during post approval use of tizanidine. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Certain events, such as somnolence, dry mouth, hypotension, decreased blood pressure, bradycardia, dizziness, weakness or asthenia, muscle spasms, hallucinations, fatigue, liver function test abnormality and hepatotoxicity, have been observed in post marketing and clinical trials and are discussed in previous sections of this document.
The following adverse reactions have been identified as occurring in the post marketing experience of 1izanidine. Based on the information provided regarding these reactions, a causal relationship with tizanidine cannot be entirely excluded. The events are listed in order of decreasing clinical significance; severity in the post marketing setting is not reported.
- Stevens Johnson Syndrome
- Anaphylactic Reaction
- Exfoliative Dermatitis
- Ventricular Tachycardia
- Hepatitis
- Convulsion
- Depression
- Arthralgia
- Paresthesia
- Rash
- Tremor
8 Use in Specific Populations
5.6 Hypersensitivity Reactions
Tizanidine can cause anaphylaxis. Signs and symptoms including respiratory compromise, urticaria, and angioedema of the throat and tongue have been reported. Patients should be informed of the signs and symptoms of severe allergic reactions and instructed to discontinue tizanidine and seek immediate medical care should these signs and symptoms occur. [see Contraindications (4)].
6.1 Clinical Trials Experience
Because clinical studies are conducted under widely varying conditions, adverse reaction rates observed in the clinical studies of a drug cannot be directly compared to rates in the clinical studies of another drug and may not reflect the rates observed in clinical practice.
Three double-blind, randomized, placebo controlled -clinical studies were conducted to evaluate the effect of tizanidine on spasticity control. Two studies were conducted in patients with multiple sclerosis and one in patients with spinal cord injury. Each study had a 13-week active treatment period which included a 3-week titration phase to the maximum tolerated dose up to 36 mg/day in three divided doses, a 9-week plateau phase where the dose of tizanidine was held constant and a 1- week dose tapering. In all, 264 patients received tizanidine and 261 patients received placebo. Across the three studies patient ages ranged from 15-69 years and 51.4 percent were women. The median dose during the plateau phase ranged from 20-28 mg/day.
The most frequent adverse reactions reported in multiple dose, placebo-controlled clinical studies involving 264 patients with spasticity were dry mouth, somnolence/sedation, asthenia (weakness, fatigue and/or tiredness) and dizziness. Three-quarters of the patients rated the events as mild to moderate and one-quarter of the patients rated the events as being severe. These events appeared to be dose related.
Table 1 lists signs and symptoms that were reported in greater than 2% of patients in three muffiple dose, placebo-controlled studies who received tizanidine where the frequency in the tizanidine group was greater than the placebo group. For comparison purposes, the corresponding frequency of the event (per 100 patients) among placebo treated patients is also provided.
| Event | Placebo N = 261 % |
Tizanidine Tablet N = 264 % |
|---|---|---|
| Dry mouth | 10 | 49 |
| Somnolence | 10 | 48 |
| Asthenia
(weakness, fatigue, and/or tiredness)
|
16 | 41 |
| Dizziness | 4 | 16 |
| UTI | 7 | 10 |
| Infection | 5 | 6 |
| Constipation | 1 | 4 |
| Liver test abnormality | 2 | 6 |
| Vomiting | 0 | 3 |
| Speech disorder | 0 | 3 |
| Amblyopia (blurred vision) | <1 | 3 |
| Urinary frequency | 2 | 3 |
| Flu syndrome | 2 | 3 |
| Dyskinesia | 0 | 3 |
| Nervousness | <1 | 3 |
| Pharyngitis | 1 | 3 |
| Rhinitis | 2 | 3 |
In the single dose, placebo-controlled study involving 142 patients with spasticity due to multiple sclerosis (Study 1) [see Clinical Studies (14)], the patients were specifically asked if they had experienced any of the 1our most common adverse reactions: dry mouth, somnolence (drowsiness), asthenia (weakness, fatigue and/or tiredness) and dizziness. In addition, hypotension and bradycardia were observed. The occurrence of these reactions is summarized in Table 2. Other events were, in general, reported at a rate of 2% or less.
| Event | Placebo N = 48 % | Tizanidine Tablet, 8mg, N = 45 % | Tizanidine Tablet, 16 mg, N = 49 % |
|---|---|---|---|
| Somnolence | 31 | 78 | 92 |
| Dry mouth | 35 | 76 | 88 |
| Asthenia
(weakness, 1atigue, and/or tiredness)
|
40 | 67 | 78 |
| Dizziness | 4 | 22 | 45 |
| Hypotension | 0 | 16 | 33 |
| Bradycardia | 0 | 2 | 10 |
8. 7 Impaired Hepatic Function
The influence of hepatic impairment on the pharmacokinetics of tizanidine has not been evaluated. Because tizanidine is extensively metabolized in the liver, hepatic impairment would be expected to have significant effects on pharmacokinetics of tizanidine. /see Dosing and Administration (2.3), Warnings and Precautions (5.2), and Clinical Pharmacology (12.3)].
5.8 Withdrawal Adverse Reactions
Withdrawal adverse reactions include rebound hypertension, tachycardia, and hypertonia. To minimize the risk of these reactions, particularly in patients who have been receiving high doses (20 to 28 mg daily) for long periods of time (9 weeks or more) or who may be on concomitant treatment with narcotics, the dose should be decreased slowly (2 to 4 mg per day). [see Dosage and Administration (2.2)].
16.1 Tizanidine Hydrochloride Capsules
Tizanidine hydrochloride capsules are available in three strengths as two-piece hard gelatin capsules containing tizanidine hydrochloride 2.29 mg, 4.58 mg and 6.87 mg, equivalent to 2 mg, 4 mg and 6 mg tizanidine base.
- The 2 mg capsules have a light yellow powder filled in size "3" hard gelatin capsule with opaque light blue cap imprinted "2 MG" and opaque light blue body: bottle of 150 capsules (NDC 75834-207-15)
- The 4 mg capsules have a light yellow powder filled in size "3" hard gelatin capsule with opaque blue cap imprinted "4 MG" and opaque white body: bottle of 150 capsules (NDC 75834-208-15)
- The 6 mg capsules have a light yellow powder filled in size "2" hard gelatin capsule with opaque blue cap imprinted "6 MG" and opaque blue body: bottle of 150 capsules (NDC 75834-209-15)
5.5 Lnteraclion With Cyp1a2 Inhibitors
Because of potential drug interactions, tizanidine is contraindicated in patients taking potent CYP1A2 inhibitors, such as fluvoxamine or ciprofloxacin. Adverse reactions such as hypotension, bradycardia, or excessive drowsiness can occur when tizanidine is taken with other CYP1A2 inhibitors, such as zileuton, fluoroquinolones other than ciprofloxacin (which is contraindicated), antiarrythmics (amiodarone, mexiletine, propafenone), cimetidine, famotidine, oral contraceptives, acyclovir, and ticlopidine). Concomitant use should be avoided unless the necessity for tizanidine therapy is clinically evident. In such a case, use with caution. [see Drug Interactions (7.3) and Clinical Pharmacology (12.3)].
5.4 Hallucinosis/psychotic Like Symptoms
Tizanidine use has been associated with hallucinations. Formed, visual hallucinations or delusions have been reported in 5 of 170 patients (3%) in two North American controlled clinical studies. Most of the patients were aware that the events were unreal. One patient developed psychosis in association with the hallucinations. One patient among these 5 continued to have problems for at least 2 weeks following discontinuation of tizanidine. Consider discontinuing tizanidine in patients who develop hallucinations.
2.2 Dosing in Patients With Renal Impairment
Tizanidine should be used with caution in patients with renal insufficiency (creatinine clearance < 25 ml/min), as clearance is reduced by more than 50%. In these patients, during titration, the individual doses should be reduced. 11 higher doses are required, individual doses rather than dosing frequency should be increased [see Warnings and Precautions (5. 7)].
2.3 Dosing in Patients With Hepatic Impairment
Tizanidine should be used with caution in patients with any hepatic impairment. In these patients, during titration, the individual doses should be reduced. If higher doses are required, individual doses rather than dosing frequency should be increased. Monitoring of aminotransferase levels is recommended for baseline and 1 month after maximum dose is achieved, or if hepatic injury is suspected. [see Use in Specific Populations (8. 7)].
Principal Display Panel 2 Mg Capsule Bottle Label
NDC 75834-207-15
Tizanidine
Hydrochloride
Capsules
2 mg*
150 CAPSULES
STOP
not interchangeable with tizanidine
tablets or Zanaflex® tablets.
NIVAGEN
Rx only
Principal Display Panel 4 Mg Capsule Bottle Label
NDC 75834-208-15
Tizanidine
Hydrochloride
Capsules
4 mg*
150 CAPSULES
STOP
not interchangeable with tizanidine
tablets or Zanaflex® tablets.
NIVAGEN
Rx only
Principal Display Panel 6 Mg Capsule Bottle Label
NDC 75834-209-15
Tizanidine
Hydrochloride
Capsules
6 mg*
150 CAPSULES
STOP
not interchangeable with tizanidine
tablets or Zanaflex® tablets.
NIVAGEN
Rx only
7.3 Cyp1a2 Inhibitors Other Than Fluvoxamine and Ciprolloxacin
Because of potential drug interactions, concomitant use of tizanidine with other CYP1 A2 inhibitors, such as zileuton, fluoroquinolones other than strong CYP1A2 inhibitors (which are contraindicated), antiarrythmics (amiodarone, mexiletine, propafenone, and verapamil), cimetidine, famo1idine, oral contraceptives, acyclovir, and ticlopidine) should be avoided. If their use is clinically necessary, therapy should be initiated with 2 mg dose and increased in 2-4 mg steps daily based on patient response to therapy. H adverse reactions such as hypotension, bradycardia, or excessive drowsiness occur, reduce or discontinue 1izanidine therapy. [see Warnings and Precautions (5.5) and Clinical Pharmacology (12.3)].
5.7 Increased Risk of Adverse Reactions in Patients With Renal Impairment
Tizanidine should be used with caution in patients with renal insufficiency (creatinine clearance < 25 ml/min), as clearance is reduced by more than 50%. In these patients, during titration, the individual doses should be reduced. If higher doses are required, individual doses rather than dosing frequency should be increased. These patients should be monitored closely for the onset or increase in severity of the common adverse events (dry mouth, somnolence, asthenia and dizziness) as indicators of potential overdose. [see Dosage and Administration (2.2) and Use in Specific Populations (8.6)].
Structured Label Content
Section 34077-8 (34077-8)
Pregnancy Category C
Tizanidine has not been studied in pregnant women. Tizanidine should be given to pregnant women only if the benefit outweighs the risk to the unborn fetus. Reproduction studies performed in rats at a dose of 3 mg/kg, equal to the maximum recommended human dose on a mg/m' basis, and in rabbits at 30 mg/kg, 16 times the maximum recommended human dose on a mg/m2 basis, did not show evidence of teratogeniclty. Tizanidine at doses that are equal to and up to 8 times the maximum recommended human dose on a mg/m2 basis increased gestation duration in rats. Prenatal and postnatal pup loss was increased and developmental retardation occurred. Post-implantation loss was increased in rabbits at doses of 1 mg/kg or greater, equal to or greater than 0.5 times the maximum recommended human dose on a mg/m2 basis.
Section 42229-5 (42229-5)
Capsules
2 mg: Light yellow powder filled in size "3" hard gelatin capsule wHh opaque light blue cap imprinted "2 MG" and opaque light blue body
4 mg: Light yellow powder filled in size "3" hard gelatin capsule with opaque blue cap imprinted "4 MG" and opaque white body
6 mg: Light yellow powder filled in size "2" hard gelatin capsule with opaque blue cap imprinted "6 MG" and opaque blue body
Section 44425-7 (44425-7)
Store at 25°G (77°F); excursions permitted between 15° to 30°G (59° to 86°F).
[See USP Controlled Room Temperature].
Dispense in containers with child resistant closure.
7. 7 A2 (7. 7 a2)
Because hypotensive effects may be cumulative, it is not recommended that tizanidine be used with other a2 -adrenergic agonists. [see Warnings and Precautions (5.1)].
9.2 Abuse
Abuse potential was not evaluated in human studies. Rats were able to distinguish tizanidine from saline in a standard discrimination paradigm, after training, but failed to generalize the effects of morphine, cocaine, diazepam, or phenobarbital to tizanidine.
7.5 Alcohol
Alcohol increases the overall amount of drug in the bloodstream after a dose of tizanidine. This was associated with an increase in adverse reactions of tizanidine. The CNS depressant effects of tizanidine and alcohol are additive. [see Clinical Pharmacology (12.3)].
5.3 Sedation
Tizanidine can cause sedation, which may interfere with everyday activity. In the multiple dose studies, the prevalence of patients with sedation peaked following the first week of titration and then remained stable for the duration of the maintenance phase of the study. The CNS depressant effects of tizanidine with alcohol and other CNS depressants (e.g., benzodiazepines, opioids, tricyclic antidepressants) may be additive. Monitor patients who take tizanidine with another CNS depressant 1or symptoms of excess sedation. [see Drug lnteracllons (7.5, 7.6)]
10 Overddsage (10 OVERDDSAGE)
A review of the safety surveillance database revealed cases of intentional and accidental tizanidine overdose. Some of the cases resulted in fatality and many of the intentional overdoses were with multiple drugs including CNS depressants. The clinical manifestations of tizanidine overdose were consistent with its known pharmacology. In the majority of cases a decrease in sensorium was observed including lethargy, somnolence, confusion and coma. Depressed cardiac function is also observed including most often bradycardia and hypotension. Respiratory depression is another common feature of tizanidine overdose.
Should overdose occur, basic steps to ensure the adequacy of an airway and the monitoring of cardiovascular and respiratory systems should be undertaken. Tizanidine is a lipid-soluble drug, which is only slightly soluble in water and methanol. Therefore, dialysis is not likely to be an efficient method of removing drug from the body. In general, symptoms resolve within one to three days following discontinuation of tizanidine and administration of appropriate therapy. Due to the similar mechanism of action, symptoms and management of tizanidine overdose are similar to that following clonidine overdose. For the most recent information concerning the management of overdose, contact a poison control center.
11 Description (11 DESCRIPTION)
Tizanidine hydrochloride is a central alpha2-adrenergic agonist. Tizanidine HCl is a white to slight⍰ yellow crystalline powder. Tizanidine is soluble in water and methanol. Its chemical name is 5-chloro-4-(2-imidazolin-2-ylamino)-2, 1 ,3-benzothiadiazole hydrochloride. Tizanidine's molecular formula is C9H8CIN5S-HCl, its molecular weight is 290.2 and its structural formula is:
Tizanidine hydrochloride capsules are supplied as 2, 4, and 6 mg capsules for oral administration. Tizanidine hydrochloride capsules contain the active ingredient, tizanidine hydrochloride (2.29 mg equivalent to 2 mg tizanidine base, 4.58 mg equivalent to 4 mg tizanidine base, and 6.87 mg equivalent to 6 mg tizanidine base), and the inactive ingredients, lactose monohydrate, hypromellose, microcrystalline cellulose and colloidal silicon dioxide. Each capsule shell contains FD & C Blue t, FD & C Red 3, titanium dioxide and gelatin. The capsules are printed with SW-0012 while ink which contains shellac, dehydrated alcohol, isopropyl alcohol, butyl alcohol, propylene glycol, strong ammonia solution, potassium hydroxide, and titanium dioxide.
9.3 Dependence
Tizanidine is closely related to clonidine, which is often abused in combination with narcotics and is known to cause symptoms of rebound upon abrupt withdrawal. Three cases of rebound symptoms on sudden withdrawal of tizanidine have been reported. The case reports suggest that these patients were also misusing narcotics. Withdrawal symptoms included hypertension, tachycardia, hypertonia, tremor, and anxiety. Withdrawal symptoms are more likely to occur in cases where high doses are used, especially for prolonged periods, or with concomitant use of narcotics. Lf therapy needs to be discontinued, the dose should be decreased slowly to minimize the risk of withdrawal symptoms [see Dosage and Administration (2.2)].
Monkeys were shown to self-administer tizanidine in a dose-dependent manner, and abrupt cessation of tizanidine produced transient signs of withdrawal at doses > 35 times the maximum recommended human dose on a mg/m2 basis. These transient withdrawal signs (increased locomotion, body twitching, and aversive behavior toward the observer) were not reversed by naloxone administration.
5.1 Hypotension
Tizanidine is an a2-adrenergic agonist that can produce hypotension. Syncope has been reported in the post marketing setting. The chance of significant hypotension may possibly be minimized by titration of the dose and by focusing attention on signs and symptoms of hypotension prior to dose advancement. In addition, patients moving from a supine to fixed upright position may be at increased risk for hypotension and orthostatic effects.
Monitor for hypotension when tizanidine is used in patients receiving concurrent antihypertensive therapy. It is not recommended that tizanidine be used with other a2 -adrenergic agonists. Clinically significant hypotension (decreases in both systolic and diastolic pressure) has been reported with concomitant administration of either fluvoxamine or ciprofloxacin and single doses of 4 mg of tizanidine. Therefore, concomitant use of 1izanidine with fluvoxamine or with ciprofloxacin, potent inhibitors of CYP1A2, is contraindicated [see Contraindications (4) and Drug Interactions (1.1, 7.2)].
7.2 Ciprofloxacin
Concomitant use of ciprofloxacin and tizanidine is contraindicated. Changes in pharmacokinetics of tizanidine when administered with ciprofloxacin resulted in significantly decreased blood pressure, increased drowsiness, and increased psychomotor impairment [See Contraindications (4) and Clinical Pharmacology (12.3)].
8.4 Pediatric Use (8.4 Pediatric use)
Safety and effectiveness in pediatric patients have not been established.
8.5 Geriatric Use (8.5 Geriatric use)
Tizanidine is known to be substantially excreted by the kidney, and the risk of adverse reactions to this drug may be greater in patients with impaired renal function. Because elderly patients are more likely to have decreased renal function, care should be taken in dose selection, and it may be useful to monitor renal function. Clinical studies of tizanidine did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently than younger subjects. Cross-study comparison of pharmacokinetic data following single dose administration of 6 mg tizanidine showed that younger subjects cleared the drug four times faster than the elderly subjects. In elderly patients with renal insufficiency (creatinine clearance <25 mL/min), tizanidine clearance is reduced by more than 50% compared to healthy elderly subjects; this would be expected to lead to a longer duration of clinical effect. During titration, the individual doses should be reduced. If higher doses are required, individual doses rather than dosing frequency should be increased. Monitor elderly patients because they may have an increased risk for adverse reactions associated with tizanidine.
14 Clinical Studies (14 CLINICAL STUDIES)
Tizanidine's capacity to reduce increased muscle tone associated with spasticity was demonstrated in two adequate and well controlled studies in patients with multiple sclerosis or spinal cord injury (Studies t and 2).
4 Contraindications (4 CONTRAINDICATIONS)
Tizanidine is contraindicated in patients taking potent inhibitors of CYP1A2, such as fluvoxamine or ciprofloxacin [see Drug Interactions (7.1, 7.2)].
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following adverse reactions are described elsewhere in other sections of the prescribing information:
- Hypotension [see Warnings and Precaullons (5.1)]
- Liver Injury [see Warnings and Precautions (5.2)]
- Sedation [see Wamings and Precautions (5.3)]
- Hallucinosis/Psychotic-Like Symptoms [see Warnings and Precautions (5.4)]
- Hypersensitivity Reactions [see Warnings and Precautions (5.6)]
7 Drug Interactions (7 DRUG INTERACTIONS)
Concomitant use of fluvoxamine and tizanidine is contraindicated. Changes in pharmacokinetics of tizanidine when administered with fluvoxamine resulted in significantly decreased blood pressure, increased drowsiness, and increased psychomotor impairment. [see Contraindications (4) and Clinical Pharmacology (12.3)].
8.3 Nursing Mothers (8.3 Nursing mothers)
It is not known whether this drug is excreted in human milk. Because many drugs are excreted in human milk, caution should be exercised when tizanidine is administered to a nursing woman.
2.1 Dosing Lnformation (2.1 Dosing lnformation)
Tizanidine hydrochloride capsules may be prescribed with or without food. Once the formulation has been selected and the decision to take with or without food has been made, this regimen should not be altered.
Food has complex effects on tizanidine pharmacokinetics, which differ with the different formulations. Tizanidine hydrochloride capsules and tizanidine hydrochloride tablets are bioequivalent to each other under fasting conditions (more than 3 hours after a meal), but not under fed conditions (within 30 minutes of a meal). These pharmacokinetic differences may result in clinically significant differences when switching administration of tablet and capsules and when switching administration between the fed or fasted state.
These changes may result in increased adverse events, or delayed or more rapid onset of activity, depending upon the nature of the switch. For this reason, the prescriber should be thoroughly familiar with the changes in kinetics associated with these different conditions [see Clinical Pharmacology (12.3)].
The recommended starting dose is 2 mg. Because the effect of tizanidine peaks at approximately 1 to 2 hours post-dose and dissipates between 3 to 6 hours post-dose, treatment can be repeated at 6 to 8 hour intervals, as needed, to a maximum of three doses in 24 hours.
Dosage can be gradually increased by 2 mg to 4 mg at each dose, with 1 to 4 days between dosage increases, until a satisfactory reduction of muscle tone is achieved. The total daily dose should not exceed 36 mg. Single doses greater than 16 mg have not been studied.
1 Indications and Usage (1 INDICATIONS AND USAGE)
Tizanidine is a central alpha-2-adrenergic agonist indicated for the management of spasticity. Because of the short duration of therapeutic effect, treatment with tizanidine should be reserved for those daily activities and times when relief of spasticity is most important [see Dosage and Administration (2.1)].
7.4 Oral Contraceptives
Concomitant use of tizanidine with oral contraceptives is not recommended. However, if concomitant use is clinically necessary, initiate tizanidine with a single 2 mg dose and increase in 2-4 mg steps daily based on patient response to therapy. 11 adverse reactions such as hypotension, bradycardia, or excessive drowsiness occur, reduce or discontinue tizanidine therapy. [see Clinical Pharmacology (12.3)].
12.1 Mechanism of Action (12.1 Mechanism of action)
Tizanidine is a central alpha-2-adrenergic receptor agonist and presumably reduces spasticity by increasing presynaptic inhibition of motor neurons. The effects of tizanidine are greatest on polysynaptic pathways. The overall effect of these actions is thought to reduce facilitation of spinal motor neurons.
2.4 Drug Discontinuation
If therapy needs to be discontinued, particularly in patients who have been receiving high doses (20 mg to 36 mg daily) for long periods (9 weeks or more) or who may be on concomitant treatment with narcotics, the dose should be decreased slowly (2 mg to 4 mg per day) to minimize the risk of withdrawal and rebound hypertension, tachycardia, and hypertonia [see Drug Abuse and Dependence (9.3)].
5.2 Risk of Liver Injury
Tizanidine may cause hepatocellular liver injury. Tizanidine should be used with caution in patients with any hepatic impairment. Monitoring of aminotransferase levels is recommended for baseline and 1 month after maximum dose is achieved, or if hepatic injury is suspected. [see Dosage and Administration (2.3) and Use in Specific Populations (8. 7)].
7.6 Other Cns Depressants (7.6 Other CNS Depressants)
The sedative effects of tizanidine with CNS depressants (e.g., benzodiazepines, opioids, tricyclic antidepressants) may be additive. Monitor patients who take tizanidine with another CNS depressant for symptoms of excess sedation. [see Clinical Pharmacology (12.3)].
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Hypotension: monitor for signs and symptoms of hypotension, in particular in patients receiving concurrent anti hypertensives; tizanidine should not be used with other a2 adrenergic agonists (5.1, 7. 7)
- Risk of liver injury: monitor ALTs; discontinue tizanidine if liver injury occurs (5.2)
- Sedation: Tizanidine may interfere with everyday activities; sedative effects of tizanidine, alcohol, and other CNS depressants are additive (5.3, 7.5, 7.6)
- Hallucinations: consider discontinuation of tizanidine (5.4)
- Less potent inhibitors of CYP1 A2: may cause hypotension, bradycardia, or excessive drowsiness, use caution if tizanidine is used with less potent inhibitors of CYP1A2, e.g., zileuton, other fluoroquinolones, antiarrythmics, cimetidine, famotidine, oral contraceptives, acyclovir, and ficlopidine (5.5, 7.3, 12.3)
- Renal impairment (creatinine clearance< 25 ml/min): use tizanidine with caution, and monitor closely for dry mouth, somnolence, asthenia and dizziness as indicators of potential overdose (5.7)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
- Recommended starting dose: 2 mg; dose can be repeated at 6 to 8 hour intervals, up to a maximum of 3 doses in 24 hours (2.1)
- Dosage can be increased by 2 mg to 4 mg per dose, wHh 1 to 4 days between increases; total daily dose should not exceed 36 mg (2.1)
- Tizanidine pharmacokinetics differs between tablets and capsules, and when taken with or without food. These differences could result in a change in tolerability and control of symptoms (2.1, 12.3)
- To discontinue tizanidine, decrease dose slowly to minimize the risk of withdrawal and rebound hypertension, tachycardia, and hypertonia (2.2)
8.6 Impaired Renal Function
Tizanidine is known to be substantially excreted by the kidney, and the risk of adverse reactions to this drug may be greater in patients with impaired renal function. In patients with renal insufficiency (creatinine clearance < 25 ml/min) clearance was reduced by more than 50%. In these patients, during titration, the individual doses should be reduced. If higher doses are required, individual doses rather than dosing frequency should be increased. These patients should be monitored closely for the onset or increase in severity of the common adverse events (dry mouth, somnolence, asthenia and dizziness) as indicators of potential overdose. [see Dosage and Administration (2.2), Warnings and Precautions (5. 7) and Clinical Pharmacology (12.3)].
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
- Capsules: 2 mg, 4 mg or 6 mg (3)
6.2 Post Marketing Experience (6.2 Post-Marketing Experience)
The following adverse reactions have been identified during post approval use of tizanidine. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Certain events, such as somnolence, dry mouth, hypotension, decreased blood pressure, bradycardia, dizziness, weakness or asthenia, muscle spasms, hallucinations, fatigue, liver function test abnormality and hepatotoxicity, have been observed in post marketing and clinical trials and are discussed in previous sections of this document.
The following adverse reactions have been identified as occurring in the post marketing experience of 1izanidine. Based on the information provided regarding these reactions, a causal relationship with tizanidine cannot be entirely excluded. The events are listed in order of decreasing clinical significance; severity in the post marketing setting is not reported.
- Stevens Johnson Syndrome
- Anaphylactic Reaction
- Exfoliative Dermatitis
- Ventricular Tachycardia
- Hepatitis
- Convulsion
- Depression
- Arthralgia
- Paresthesia
- Rash
- Tremor
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
5.6 Hypersensitivity Reactions
Tizanidine can cause anaphylaxis. Signs and symptoms including respiratory compromise, urticaria, and angioedema of the throat and tongue have been reported. Patients should be informed of the signs and symptoms of severe allergic reactions and instructed to discontinue tizanidine and seek immediate medical care should these signs and symptoms occur. [see Contraindications (4)].
6.1 Clinical Trials Experience
Because clinical studies are conducted under widely varying conditions, adverse reaction rates observed in the clinical studies of a drug cannot be directly compared to rates in the clinical studies of another drug and may not reflect the rates observed in clinical practice.
Three double-blind, randomized, placebo controlled -clinical studies were conducted to evaluate the effect of tizanidine on spasticity control. Two studies were conducted in patients with multiple sclerosis and one in patients with spinal cord injury. Each study had a 13-week active treatment period which included a 3-week titration phase to the maximum tolerated dose up to 36 mg/day in three divided doses, a 9-week plateau phase where the dose of tizanidine was held constant and a 1- week dose tapering. In all, 264 patients received tizanidine and 261 patients received placebo. Across the three studies patient ages ranged from 15-69 years and 51.4 percent were women. The median dose during the plateau phase ranged from 20-28 mg/day.
The most frequent adverse reactions reported in multiple dose, placebo-controlled clinical studies involving 264 patients with spasticity were dry mouth, somnolence/sedation, asthenia (weakness, fatigue and/or tiredness) and dizziness. Three-quarters of the patients rated the events as mild to moderate and one-quarter of the patients rated the events as being severe. These events appeared to be dose related.
Table 1 lists signs and symptoms that were reported in greater than 2% of patients in three muffiple dose, placebo-controlled studies who received tizanidine where the frequency in the tizanidine group was greater than the placebo group. For comparison purposes, the corresponding frequency of the event (per 100 patients) among placebo treated patients is also provided.
| Event | Placebo N = 261 % |
Tizanidine Tablet N = 264 % |
|---|---|---|
| Dry mouth | 10 | 49 |
| Somnolence | 10 | 48 |
| Asthenia
(weakness, fatigue, and/or tiredness)
|
16 | 41 |
| Dizziness | 4 | 16 |
| UTI | 7 | 10 |
| Infection | 5 | 6 |
| Constipation | 1 | 4 |
| Liver test abnormality | 2 | 6 |
| Vomiting | 0 | 3 |
| Speech disorder | 0 | 3 |
| Amblyopia (blurred vision) | <1 | 3 |
| Urinary frequency | 2 | 3 |
| Flu syndrome | 2 | 3 |
| Dyskinesia | 0 | 3 |
| Nervousness | <1 | 3 |
| Pharyngitis | 1 | 3 |
| Rhinitis | 2 | 3 |
In the single dose, placebo-controlled study involving 142 patients with spasticity due to multiple sclerosis (Study 1) [see Clinical Studies (14)], the patients were specifically asked if they had experienced any of the 1our most common adverse reactions: dry mouth, somnolence (drowsiness), asthenia (weakness, fatigue and/or tiredness) and dizziness. In addition, hypotension and bradycardia were observed. The occurrence of these reactions is summarized in Table 2. Other events were, in general, reported at a rate of 2% or less.
| Event | Placebo N = 48 % | Tizanidine Tablet, 8mg, N = 45 % | Tizanidine Tablet, 16 mg, N = 49 % |
|---|---|---|---|
| Somnolence | 31 | 78 | 92 |
| Dry mouth | 35 | 76 | 88 |
| Asthenia
(weakness, 1atigue, and/or tiredness)
|
40 | 67 | 78 |
| Dizziness | 4 | 22 | 45 |
| Hypotension | 0 | 16 | 33 |
| Bradycardia | 0 | 2 | 10 |
8. 7 Impaired Hepatic Function
The influence of hepatic impairment on the pharmacokinetics of tizanidine has not been evaluated. Because tizanidine is extensively metabolized in the liver, hepatic impairment would be expected to have significant effects on pharmacokinetics of tizanidine. /see Dosing and Administration (2.3), Warnings and Precautions (5.2), and Clinical Pharmacology (12.3)].
5.8 Withdrawal Adverse Reactions
Withdrawal adverse reactions include rebound hypertension, tachycardia, and hypertonia. To minimize the risk of these reactions, particularly in patients who have been receiving high doses (20 to 28 mg daily) for long periods of time (9 weeks or more) or who may be on concomitant treatment with narcotics, the dose should be decreased slowly (2 to 4 mg per day). [see Dosage and Administration (2.2)].
16.1 Tizanidine Hydrochloride Capsules
Tizanidine hydrochloride capsules are available in three strengths as two-piece hard gelatin capsules containing tizanidine hydrochloride 2.29 mg, 4.58 mg and 6.87 mg, equivalent to 2 mg, 4 mg and 6 mg tizanidine base.
- The 2 mg capsules have a light yellow powder filled in size "3" hard gelatin capsule with opaque light blue cap imprinted "2 MG" and opaque light blue body: bottle of 150 capsules (NDC 75834-207-15)
- The 4 mg capsules have a light yellow powder filled in size "3" hard gelatin capsule with opaque blue cap imprinted "4 MG" and opaque white body: bottle of 150 capsules (NDC 75834-208-15)
- The 6 mg capsules have a light yellow powder filled in size "2" hard gelatin capsule with opaque blue cap imprinted "6 MG" and opaque blue body: bottle of 150 capsules (NDC 75834-209-15)
5.5 Lnteraclion With Cyp1a2 Inhibitors (5.5 lnteraclion with CYP1A2 Inhibitors)
Because of potential drug interactions, tizanidine is contraindicated in patients taking potent CYP1A2 inhibitors, such as fluvoxamine or ciprofloxacin. Adverse reactions such as hypotension, bradycardia, or excessive drowsiness can occur when tizanidine is taken with other CYP1A2 inhibitors, such as zileuton, fluoroquinolones other than ciprofloxacin (which is contraindicated), antiarrythmics (amiodarone, mexiletine, propafenone), cimetidine, famotidine, oral contraceptives, acyclovir, and ticlopidine). Concomitant use should be avoided unless the necessity for tizanidine therapy is clinically evident. In such a case, use with caution. [see Drug Interactions (7.3) and Clinical Pharmacology (12.3)].
5.4 Hallucinosis/psychotic Like Symptoms (5.4 Hallucinosis/Psychotic-Like Symptoms)
Tizanidine use has been associated with hallucinations. Formed, visual hallucinations or delusions have been reported in 5 of 170 patients (3%) in two North American controlled clinical studies. Most of the patients were aware that the events were unreal. One patient developed psychosis in association with the hallucinations. One patient among these 5 continued to have problems for at least 2 weeks following discontinuation of tizanidine. Consider discontinuing tizanidine in patients who develop hallucinations.
2.2 Dosing in Patients With Renal Impairment (2.2 Dosing in Patients with Renal Impairment)
Tizanidine should be used with caution in patients with renal insufficiency (creatinine clearance < 25 ml/min), as clearance is reduced by more than 50%. In these patients, during titration, the individual doses should be reduced. 11 higher doses are required, individual doses rather than dosing frequency should be increased [see Warnings and Precautions (5. 7)].
2.3 Dosing in Patients With Hepatic Impairment (2.3 Dosing in Patients with Hepatic Impairment)
Tizanidine should be used with caution in patients with any hepatic impairment. In these patients, during titration, the individual doses should be reduced. If higher doses are required, individual doses rather than dosing frequency should be increased. Monitoring of aminotransferase levels is recommended for baseline and 1 month after maximum dose is achieved, or if hepatic injury is suspected. [see Use in Specific Populations (8. 7)].
Principal Display Panel 2 Mg Capsule Bottle Label (PRINCIPAL DISPLAY PANEL - 2 mg Capsule Bottle Label)
NDC 75834-207-15
Tizanidine
Hydrochloride
Capsules
2 mg*
150 CAPSULES
STOP
not interchangeable with tizanidine
tablets or Zanaflex® tablets.
NIVAGEN
Rx only
Principal Display Panel 4 Mg Capsule Bottle Label (PRINCIPAL DISPLAY PANEL - 4 mg Capsule Bottle Label)
NDC 75834-208-15
Tizanidine
Hydrochloride
Capsules
4 mg*
150 CAPSULES
STOP
not interchangeable with tizanidine
tablets or Zanaflex® tablets.
NIVAGEN
Rx only
Principal Display Panel 6 Mg Capsule Bottle Label (PRINCIPAL DISPLAY PANEL - 6 mg Capsule Bottle Label)
NDC 75834-209-15
Tizanidine
Hydrochloride
Capsules
6 mg*
150 CAPSULES
STOP
not interchangeable with tizanidine
tablets or Zanaflex® tablets.
NIVAGEN
Rx only
7.3 Cyp1a2 Inhibitors Other Than Fluvoxamine and Ciprolloxacin (7.3 CYP1A2 Inhibitors other than Fluvoxamine and Ciprolloxacin)
Because of potential drug interactions, concomitant use of tizanidine with other CYP1 A2 inhibitors, such as zileuton, fluoroquinolones other than strong CYP1A2 inhibitors (which are contraindicated), antiarrythmics (amiodarone, mexiletine, propafenone, and verapamil), cimetidine, famo1idine, oral contraceptives, acyclovir, and ticlopidine) should be avoided. If their use is clinically necessary, therapy should be initiated with 2 mg dose and increased in 2-4 mg steps daily based on patient response to therapy. H adverse reactions such as hypotension, bradycardia, or excessive drowsiness occur, reduce or discontinue 1izanidine therapy. [see Warnings and Precautions (5.5) and Clinical Pharmacology (12.3)].
5.7 Increased Risk of Adverse Reactions in Patients With Renal Impairment (5.7 Increased Risk of Adverse Reactions In Patients with Renal Impairment)
Tizanidine should be used with caution in patients with renal insufficiency (creatinine clearance < 25 ml/min), as clearance is reduced by more than 50%. In these patients, during titration, the individual doses should be reduced. If higher doses are required, individual doses rather than dosing frequency should be increased. These patients should be monitored closely for the onset or increase in severity of the common adverse events (dry mouth, somnolence, asthenia and dizziness) as indicators of potential overdose. [see Dosage and Administration (2.2) and Use in Specific Populations (8.6)].
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:35:47.902459 · Updated: 2026-03-14T21:44:26.969907