These Highlights Do Not Include All The Information Needed To Use Eptifibatide Injection Safely And Effectively. See Full Prescribing Information For Eptifibatide Injection.
c488f1af-a82c-4360-8886-79520635561a
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Eptifibatide injection is a platelet aggregation inhibitor indicated for: Treatment of acute coronary syndrome (ACS) managed medically or with percutaneous coronary intervention (PCI) ( 1.1 ) Treatment of patients undergoing PCI (including intracoronary stenting) ( 1.2 )
Indications and Usage
Eptifibatide injection is a platelet aggregation inhibitor indicated for: Treatment of acute coronary syndrome (ACS) managed medically or with percutaneous coronary intervention (PCI) ( 1.1 ) Treatment of patients undergoing PCI (including intracoronary stenting) ( 1.2 )
Dosage and Administration
Before infusion of eptifibatide injection, the following laboratory tests should be performed to identify pre-existing hemostatic abnormalities: hematocrit or hemoglobin, platelet count, serum creatinine, and PT/aPTT. In patients undergoing PCI, the activated clotting time (ACT) should also be measured. The activated partial thromboplastin time (aPTT) should be maintained between 50 and 70 seconds unless PCI is to be performed. In patients treated with heparin, bleeding can be minimized by close monitoring of the aPTT and ACT.
Warnings and Precautions
Eptifibatide can cause serious bleeding. If bleeding cannot be controlled, discontinue eptifibatide immediately. Minimize vascular and other traumas. If heparin is given concomitantly, monitor aPTT or ACT. ( 5.1 ) Thrombocytopenia: Discontinue eptifibatide and heparin. Monitor and treat condition appropriately. ( 5.2 )
Contraindications
Treatment with eptifibatide is contraindicated in patients with: A history of bleeding diathesis, or evidence of active abnormal bleeding within the previous 30 days Severe hypertension (systolic blood pressure >200 mm Hg or diastolic blood pressure >110 mm Hg) not adequately controlled on antihypertensive therapy Major surgery within the preceding 6 weeks History of stroke within 30 days or any history of hemorrhagic stroke Current or planned administration of another parenteral GP IIb/IIIa inhibitor Dependency on renal dialysis Hypersensitivity to eptifibatide or any component of the product (hypersensitivity reactions that occurred included anaphylaxis and urticaria)
Drug Interactions
Coadministration of antiplatelet agents, thrombolytics, heparin, aspirin, and chronic NSAID use increases the risk of bleeding. Avoid concomitant use with other glycoprotein (GP) IIb/IIIa inhibitors. ( 7.1 )
How Supplied
Eptifibatide injection is supplied as a sterile solution in 10-mL vials containing 20 mg of eptifibatide (NDC 52958-402-01) and 100-mL vials containing 75 mg of eptifibatide (NDC 52958-040-01).
Medication Information
Warnings and Precautions
Eptifibatide can cause serious bleeding. If bleeding cannot be controlled, discontinue eptifibatide immediately. Minimize vascular and other traumas. If heparin is given concomitantly, monitor aPTT or ACT. ( 5.1 ) Thrombocytopenia: Discontinue eptifibatide and heparin. Monitor and treat condition appropriately. ( 5.2 )
Indications and Usage
Eptifibatide injection is a platelet aggregation inhibitor indicated for: Treatment of acute coronary syndrome (ACS) managed medically or with percutaneous coronary intervention (PCI) ( 1.1 ) Treatment of patients undergoing PCI (including intracoronary stenting) ( 1.2 )
Dosage and Administration
Before infusion of eptifibatide injection, the following laboratory tests should be performed to identify pre-existing hemostatic abnormalities: hematocrit or hemoglobin, platelet count, serum creatinine, and PT/aPTT. In patients undergoing PCI, the activated clotting time (ACT) should also be measured. The activated partial thromboplastin time (aPTT) should be maintained between 50 and 70 seconds unless PCI is to be performed. In patients treated with heparin, bleeding can be minimized by close monitoring of the aPTT and ACT.
Contraindications
Treatment with eptifibatide is contraindicated in patients with: A history of bleeding diathesis, or evidence of active abnormal bleeding within the previous 30 days Severe hypertension (systolic blood pressure >200 mm Hg or diastolic blood pressure >110 mm Hg) not adequately controlled on antihypertensive therapy Major surgery within the preceding 6 weeks History of stroke within 30 days or any history of hemorrhagic stroke Current or planned administration of another parenteral GP IIb/IIIa inhibitor Dependency on renal dialysis Hypersensitivity to eptifibatide or any component of the product (hypersensitivity reactions that occurred included anaphylaxis and urticaria)
Drug Interactions
Coadministration of antiplatelet agents, thrombolytics, heparin, aspirin, and chronic NSAID use increases the risk of bleeding. Avoid concomitant use with other glycoprotein (GP) IIb/IIIa inhibitors. ( 7.1 )
How Supplied
Eptifibatide injection is supplied as a sterile solution in 10-mL vials containing 20 mg of eptifibatide (NDC 52958-402-01) and 100-mL vials containing 75 mg of eptifibatide (NDC 52958-040-01).
Description
Eptifibatide injection is a platelet aggregation inhibitor indicated for: Treatment of acute coronary syndrome (ACS) managed medically or with percutaneous coronary intervention (PCI) ( 1.1 ) Treatment of patients undergoing PCI (including intracoronary stenting) ( 1.2 )
Section 42229-5
Use of Thrombolytics, Anticoagulants, and Other Antiplatelet Agents
Risk factors for bleeding include older age, a history of bleeding disorders, and concomitant use of drugs that increase the risk of bleeding (thrombolytics, oral anticoagulants, nonsteroidal anti-inflammatory drugs, and P2Y12 inhibitors). Concomitant treatment with other inhibitors of platelet receptor glycoprotein (GP) IIb/IIIa should be avoided. In patients treated with heparin, bleeding can be minimized by close monitoring of the aPTT and ACT [see Dosage and Administration (2)].
Section 51945-4
NDC 52958-402-01
20 mg/10 mL vial
for weight-adjusted bolus dosing
Eptifibatide
Injection
20 mg/10 mL
(2 mg/mL)
For Intravenous Use Only
1×10 mL Single-dose Vial
Rx only
Shuangcheng
Pharma
16.2 Storage
Vials should be stored refrigerated at 2-8°C (36-46°F). Vials may be transferred to room temperature storage* for a period not to exceed 2 months. Upon transfer, vial cartons must be marked by the dispensing pharmacist with a "DISCARD BY" date (2 months from the transfer date or the labeled expiration date, whichever comes first).
Retain in carton to protect from light until administration.
* Store at 25oC (77oF); excursions permitted to 15-30oC (59-oF) [see USP Controlled Room Temperature].
5.1 Bleeding
Bleeding is the most common complication encountered during eptifibatide therapy. Administration of eptifibatide is associated with an increase in major and minor bleeding, as classified by the criteria of the Thrombolysis in Myocardial Infarction Study group (TIMI) [see Adverse Reactions (6.1)]. Most major bleeding associated with eptifibatide has been at the arterial access site for cardiac catheterization or from the gastrointestinal or genitourinary tract. Minimize the use of arterial and venous punctures, intramuscular injections, and the use of urinary catheters, nasotracheal intubation, and nasogastric tubes. When obtaining intravenous access, avoid non-compressible sites (e.g., subclavian or jugular veins).
10 Overdosage
There has been only limited experience with overdosage of eptifibatide. There were 8 patients in the IMPACT II study, 9 patients in the PURSUIT study, and no patients in the ESPRIT study who received bolus doses and/or infusion doses more than double those called for in the protocols. None of these patients experienced an intracranial bleed or other major bleeding.
Eptifibatide was not lethal to rats, rabbits, or monkeys when administered by continuous intravenous infusion for 90 minutes at a total dose of 45 mg/kg (about 2 to 5 times the recommended maximum daily human dose on a body surface area basis). Symptoms of acute toxicity were loss of righting reflex, dyspnea, ptosis, and decreased muscle tone in rabbits and petechial hemorrhages in the femoral and abdominal areas of monkeys.
From in vitro studies, eptifibatide is not extensively bound to plasma proteins and thus may be cleared from plasma by dialysis.
8.1 Pregnancy
Risk Summary
Available data on eptifibatide use in pregnant women from published literature and the pharmacovigilance database are insufficient to establish a drug-associated risk of major birth defects, miscarriage or adverse maternal or fetal outcomes. Untreated myocardial infarction can be fatal to the pregnant woman and fetus (see Clinical Considerations). In animal reproduction studies, there was no evidence of adverse developmental effects when eptifibatide was administered intravenously to pregnant rats and rabbits at approximately 4 times the recommended maximum daily human dose.
The estimated background risk of major birth defects and miscarriage for the indicated population is unknown. All pregnancies have a background risk of birth defect, loss, or other adverse outcomes. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2 to 4% and 15 to 20%, respectively.
Clinical Considerations
Disease-associated maternal and/or embryo/fetal risk
Myocardial infarction is a medical emergency in pregnancy which can be fatal to the pregnant woman and fetus if left untreated. Therapy for the pregnant woman should not be withheld because of potential concerns regarding the effects of eptifibatide on the fetus
Data
Animal Data
Embryo-fetal development studies have been performed by continuous intravenous infusion of eptifibatide in pregnant rats during the period of organogenesis at total daily doses of up to 72 mg/kg/day (about 4 times the recommended maximum daily human dose on a body surface area basis) and in pregnant rabbits during the period of organogenesis at total daily doses of up to 36 mg/kg/day (also about 4 times the recommended maximum daily human dose on a body surface area basis). These studies revealed no evidence of harm to the fetus due to eptifibatide.
8.2 Lactation
Risk Summary
There are no available data on the presence of eptifibatide in human milk, the effects on the breastfed infant, or the effects on milk production. As eptifibatide is a peptide, it is likely to be destroyed in the infant’s gastrointestinal tract and not absorbed orally by the breastfed infant.
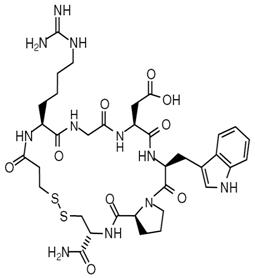
11 Description
Eptifibatide is a cyclic heptapeptide containing 6 amino acids and 1 mercaptopropionyl (des-amino cysteinyl) residue. An interchain disulfide bridge is formed between the cysteine amide and the mercaptopropionyl moieties. Chemically it is N6-(aminoiminomethyl)-N2-(3-mercapto-1-oxopropyl)-L-lysylglycyl-L-α-aspartyl-L-tryptophyl-L-prolyl-L-cysteinamide, cyclic (1→6)-disulfide. Eptifibatide binds to the platelet receptor glycoprotein (GP) IIb/IIIa of human platelets and inhibits platelet aggregation.
The eptifibatide peptide is produced by solid-phase peptide synthesis, and is purified by preparative reverse-phase liquid chromatography and lyophilized. The structural formula is:
Eptifibatide Injection is a clear, colorless, sterile, non-pyrogenic solution for intravenous (IV) use with an empirical formula of C35H49N11O9S2 and a molecular weight of 831.96. Each 10-mL vial contains 2 mg/mL of eptifibatide and each 100-mL vial contains 0.75 mg/mL of eptifibatide. Each vial of either size also contains 5.25 mg/mL citric acid and sodium hydroxide to adjust the pH to 5.35.
16.1 How Supplied
Eptifibatide injection is supplied as a sterile solution in 10-mL vials containing 20 mg of eptifibatide (NDC 52958-402-01) and 100-mL vials containing 75 mg of eptifibatide (NDC 52958-040-01).
8.4 Pediatric Use
Safety and effectiveness of eptifibatide in pediatric patients have not been studied.
8.5 Geriatric Use
The PURSUIT and IMPACT II clinical studies enrolled patients up to the age of 94 years (45% were age 65 and over; 12% were age 75 and older). There was no apparent difference in efficacy between older and younger patients treated with eptifibatide. The incidence of bleeding complications was higher in the elderly in both placebo and eptifibatide groups, and the incremental risk of eptifibatide-associated bleeding was greater in the older patients. No dose adjustment was made for elderly patients, but patients over 75 years of age had to weigh at least 50 kg to be enrolled in the PURSUIT study; no such limitation was stipulated in the ESPRIT study [see Adverse Reactions (6.1)].
14 Clinical Studies
Eptifibatide was studied in 3 placebo-controlled, randomized studies. PURSUIT evaluated patients with acute coronary syndromes: UA or NSTEMI. Two other studies, ESPRIT and IMPACT II, evaluated patients about to undergo a PCI. Patients underwent primarily balloon angioplasty in IMPACT II and intracoronary stent placement, with or without angioplasty, in ESPRIT.
4 Contraindications
Treatment with eptifibatide is contraindicated in patients with:
- A history of bleeding diathesis, or evidence of active abnormal bleeding within the previous 30 days
- Severe hypertension (systolic blood pressure >200 mm Hg or diastolic blood pressure >110 mm Hg) not adequately controlled on antihypertensive therapy
- Major surgery within the preceding 6 weeks
- History of stroke within 30 days or any history of hemorrhagic stroke
- Current or planned administration of another parenteral GP IIb/IIIa inhibitor
- Dependency on renal dialysis
- Hypersensitivity to eptifibatide or any component of the product (hypersensitivity reactions that occurred included anaphylaxis and urticaria)
7 Drug Interactions
- Coadministration of antiplatelet agents, thrombolytics, heparin, aspirin, and chronic NSAID use increases the risk of bleeding. Avoid concomitant use with other glycoprotein (GP) IIb/IIIa inhibitors. (7.1)
5.2 Thrombocytopenia
There have been reports of acute, profound thrombocytopenia (immune-mediated and non-immune mediated) with eptifibatide. In the event of acute profound thrombocytopenia or a confirmed platelet decrease to <100,000/mm3, discontinue eptifibatide and heparin (unfractionated or low-molecular weight). Monitor serial platelet counts, assess the presence of drug-dependent antibodies, and treat as appropriate [see Adverse Reactions (6.1)].
There has been no clinical experience with eptifibatide initiated in patients with a baseline platelet count <100,000/mm3. If a patient with low platelet counts is receiving eptifibatide, their platelet count should be monitored closely.
8.6 Renal Impairment
Approximately 50% of eptifibatide is cleared by the kidney in patients with normal renal function. Total drug clearance is decreased by approximately 50% and steady-state plasma eptifibatide concentrations are doubled in patients with an estimated CrCl <50 mL/min (using the Cockcroft-Gault equation). Therefore, the infusion dose should be reduced to 1 mcg/kg/min in such patients [see Dosage and Administration (2)]. The safety and efficacy of eptifibatide in patients dependent on dialysis has not been established.
12.2 Pharmacodynamics
Infusion of eptifibatide into baboons caused a dose-dependent inhibition of ex vivo platelet aggregation, with complete inhibition of aggregation achieved at infusion rates greater than 5 mcg/kg/min. In a baboon model that is refractory to aspirin and heparin, doses of eptifibatide that inhibit aggregation prevented acute thrombosis with only a modest prolongation (2- to 3-fold) of the bleeding time. Platelet aggregation in dogs was also inhibited by infusions of eptifibatide, with complete inhibition at 2 mcg/kg/min. This infusion dose completely inhibited canine coronary thrombosis induced by coronary artery injury (Folts model).
Human pharmacodynamic data were obtained in healthy subjects and in patients presenting with UA or NSTEMI and/or undergoing percutaneous coronary intervention. Studies in healthy subjects enrolled only males; patient studies enrolled approximately one-third women. In these studies, eptifibatide inhibited ex vivo platelet aggregation induced by adenosine diphosphate (ADP) and other agonists in a dose- and concentration-dependent manner. The effect of eptifibatide was observed immediately after administration of a 180-mcg/kg intravenous bolus. Table 4 shows the effects of dosing regimens of eptifibatide used in the IMPACT II and PURSUIT studies on ex vivo platelet aggregation induced by 20 µM ADP in PPACK-anticoagulated platelet-rich plasma and on bleeding time. The effects of the dosing regimen used in ESPRIT on platelet aggregation have not been studied.
| PURSUIT 180/2 180-mcg/kg bolus followed by a continuous infusion of 2 mcg/kg/min.
|
|
|---|---|
| Inhibition of platelet aggregation 15 min after bolus | 84% |
| Inhibition of platelet aggregation at steady state | >90% |
| Bleeding-time prolongation at steady state | <5x |
| Inhibition of platelet aggregation 4h after infusion discontinuation | <50% |
| Bleeding-time prolongation 6h after infusion discontinuation | 1.4x |
The eptifibatide dosing regimen used in the ESPRIT study included two 180-mcg/kg bolus doses given 10 minutes apart combined with a continuous 2-mcg/kg/min infusion.
When administered alone, eptifibatide has no measurable effect on PT or aPTT.
There were no important differences between men and women or between age groups in the pharmacodynamic properties of eptifibatide. Differences among ethnic groups have not been assessed.
12.3 Pharmacokinetics
The pharmacokinetics of eptifibatide are linear and dose-proportional for bolus doses ranging from 90 to 250 mcg/kg and infusion rates from 0.5 to 3 mcg/kg/min. Plasma elimination half-life is approximately 2.5 hours. Administration of a single 180-mcg/kg bolus combined with an infusion produces an early peak level, followed by a small decline prior to attaining steady state (within 4-6 hours). This decline can be prevented by administering a second 180-mcg/kg bolus 10 minutes after the first. The extent of eptifibatide binding to human plasma protein is about 25%. Clearance in patients with coronary artery disease is about 55 mL/kg/h. In healthy subjects, renal clearance accounts for approximately 50% of total body clearance, with the majority of the drug excreted in the urine as eptifibatide, deaminated eptifibatide, and other, more polar metabolites. No major metabolites have been detected in human plasma.
1 Indications and Usage
12.1 Mechanism of Action
Eptifibatide reversibly inhibits platelet aggregation by preventing the binding of fibrinogen, von Willebrand factor, and other adhesive ligands to GP IIb/IIIa. When administered intravenously, eptifibatide inhibits ex vivo platelet aggregation in a dose- and concentration-dependent manner. Platelet aggregation inhibition is reversible following cessation of the eptifibatide infusion; this is thought to result from dissociation of eptifibatide from the platelet.
5 Warnings and Precautions
- Eptifibatide can cause serious bleeding. If bleeding cannot be controlled, discontinue eptifibatide immediately. Minimize vascular and other traumas. If heparin is given concomitantly, monitor aPTT or ACT. (5.1)
- Thrombocytopenia: Discontinue eptifibatide and heparin. Monitor and treat condition appropriately. (5.2)
2 Dosage and Administration
Before infusion of eptifibatide injection, the following laboratory tests should be performed to identify pre-existing hemostatic abnormalities: hematocrit or hemoglobin, platelet count, serum creatinine, and PT/aPTT. In patients undergoing PCI, the activated clotting time (ACT) should also be measured.
The activated partial thromboplastin time (aPTT) should be maintained between 50 and 70 seconds unless PCI is to be performed. In patients treated with heparin, bleeding can be minimized by close monitoring of the aPTT and ACT.
3 Dosage Forms and Strengths
- Injection: 20 mg of eptifibatide in 10 mL (2 mg/mL), for intravenous bolus.
- Injection: 75 mg of eptifibatide in 100 mL (0.75 mg/mL), for intravenous infusion.
8 Use in Specific Populations
- Geriatric Use: Risk of bleeding increases with age. (8.5)
1.1 Acute Coronary Syndrome (acs)
Eptifibatide injection is indicated to decrease the rate of a combined endpoint of death or new myocardial infarction (MI) in patients with ACS (unstable angina [UA]/non-ST-elevation myocardial infarction [NSTEMI]), including patients who are to be managed medically and those undergoing percutaneous coronary intervention (PCI).
17 Patient Counseling Information
Advise the patient to inform the doctor or healthcare provider about any medical conditions, medications, and allergies.
2.3 Important Administration Instructions
- Inspect eptifibatide injection for particulate matter and discoloration prior to administration, whenever solution and container permit.
- May administer eptifibatide injection in the same intravenous line as alteplase, atropine, dobutamine, heparin, lidocaine, meperidine, metoprolol, midazolam, morphine, nitroglycerin, or verapamil. Do not administer eptifibatide injection through the same intravenous line as furosemide.
- May administer eptifibatide injection in the same IV line with 0.9% NaCl or 0.9% NaCl/5% dextrose. With either vehicle, the infusion may also contain up to 60 mEq/L of potassium chloride.
- Withdraw the bolus dose(s) of eptifibatide injection from the 10-mL vial into a syringe. Administer the bolus dose(s) by IV push.
- Immediately following the bolus dose administration, initiate a continuous infusion of eptifibatide injection. When using an intravenous infusion pump, administer eptifibatide injection undiluted directly from the 100-mL vial. Spike the 100-mL vial with a vented infusion set. Center the spike within the circle on the stopper top.
- Discard any unused portion left in the vial.
Administer eptifibatide injection by volume according to patient weight (see Table 1).
| Patient Weight | 180-mcg/kg Bolus Volume |
2-mcg/kg/min Infusion Volume (CrCl greater than or equal to 50 mL/min) |
1-mcg/kg/min Infusion Volume (CrCl less than 50 mL/min) |
|||
|---|---|---|---|---|---|---|
| (kg) | (lb) | (from 2-mg/mL vial) | (from 2-mg/mL 100-mL vial) | (from 0.75-mg/mL 100-mL vial) | (from 2-mg/mL 100-mL vial) | (from 0.75-mg/mL 100-mL vial) |
| 37-41 | 81-91 | 3.4 mL | 2 mL/h | 6 mL/h | 1 mL/h | 3 mL/h |
| 42-46 | 92-102 | 4 mL | 2.5 mL/h | 7 mL/h | 1.3 mL/h | 3.5 mL/h |
| 47-53 | 103-117 | 4.5 mL | 3 mL/h | 8 mL/h | 1.5 mL/h | 4 mL/h |
| 54-59 | 118-130 | 5 mL | 3.5 mL/h | 9 mL/h | 1.8 mL/h | 4.5 mL/h |
| 60-65 | 131-143 | 5.6 mL | 3.8 mL/h | 10 mL/h | 1.9 mL/h | 5 mL/h |
| 66-71 | 144-157 | 6.2 mL | 4 mL/h | 11 mL/h | 2 mL/h | 5.5 mL/h |
| 72-78 | 158-172 | 6.8 mL | 4.5 mL/h | 12 mL/h | 2.3 mL/h | 6 mL/h |
| 79-84 | 173-185 | 7.3 mL | 5 mL/h | 13 mL/h | 2.5 mL/h | 6.5 mL/h |
| 85-90 | 186-198 | 7.9 mL | 5.3 mL/h | 14 mL/h | 2.7 mL/h | 7 mL/h |
| 91-96 | 199-212 | 8.5 mL | 5.6 mL/h | 15 mL/h | 2.8 mL/h | 7.5 mL/h |
| 97-103 | 213-227 | 9 mL | 6 mL/h | 16 mL/h | 3.0 mL/h | 8 mL/h |
| 104-109 | 228-240 | 9.5 mL | 6.4 mL/h | 17 mL/h | 3.2 mL/h | 8.5 mL/h |
| 110-115 | 241-253 | 10.2 mL | 6.8 mL/h | 18 mL/h | 3.4 mL/h | 9 mL/h |
| 116-121 | 254-267 | 10.7 mL | 7 mL/h | 19 mL/h | 3.5 mL/h | 9.5 mL/h |
| >121 | >267 | 11.3 mL | 7.5 mL/h | 20 mL/h | 3.7 mL/h | 10 mL/h |
2.1 Dosage in Acute Coronary Syndrome (acs)
| Indication | Normal Renal Function | Creatinine Clearance less than 50 mL/min |
|---|---|---|
| Patients with ACS | 180 mcg/kg intravenous (IV) bolus as soon as possible after diagnosis, followed by continuous infusion of 2 mcg/kg/min | 180 mcg/kg IV bolus as soon as possible after diagnosis, followed by continuous infusion of 1 mcg/kg/min |
|
Eptifibatide injection should be given concomitantly with heparin dosed to achieve the following parameters:
During Medical Management: Target aPTT 50 to 70 seconds
- If weight greater than or equal to 70 kg, 5000-unit bolus followed by infusion of 1000 units/h.
- If weight less than 70 kg, 60-units/kg bolus followed by infusion of 12 units/kg/h.
During PCI: Target ACT 200 to 300 seconds
- If heparin is initiated prior to PCI, additional boluses during PCI to maintain an ACT target of 200 to 300 seconds.
- Heparin infusion after the PCI is discouraged.
1.2 Percutaneous Coronary Intervention (pci)
Eptifibatide injection is indicated to decrease the rate of a combined endpoint of death, new MI, or need for urgent intervention in patients undergoing PCI, including those undergoing intracoronary stenting [see Clinical Studies (14.1, 14.2)].
14.1 Non St Segment Elevation Acute Coronary Syndrome
Non-ST-segment elevation acute coronary syndrome is defined as prolonged (≥10 minutes) symptoms of cardiac ischemia within the previous 24 hours associated with either ST-segment changes (elevations between 0.6 mm and 1 mm or depression >0.5 mm), T-wave inversion (>1 mm), or positive CK-MB. This definition includes "unstable angina" and "NSTEMI" but excludes MI that is associated with Q waves or greater degrees of ST-segment elevation.
2.2 Dosage in Percutaneous Coronary Intervention (pci)
| Indication | Normal Renal Function | Creatinine Clearance less than 50 mL/min |
|---|---|---|
| Patients with PCI | 180 mcg/kg IV bolus immediately before PCI followed by continuous infusion of 2 mcg/kg/min and a second bolus of 180 mcg/kg (given 10 minutes after the first bolus) | 180 mcg/kg IV bolus immediately before PCI followed by continuous infusion of 1 mcg/kg/min and a second bolus of 180 mcg/kg (given 10 minutes after the first bolus) |
|
- Eptifibatide injection should be given concomitantly with heparin to achieve a target ACT of 200 to 300 seconds. Administer 60-units/kg bolus initially in patients not treated with heparin within 6 hours prior to PCI.
- Additional boluses during PCI to maintain ACT within target.
- Heparin infusion after the PCI is strongly discouraged.
Patients requiring thrombolytic therapy should discontinue eptifibatide injection.
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
No long-term studies in animals have been performed to evaluate the carcinogenic potential of eptifibatide. Eptifibatide was not genotoxic in the Ames test, the mouse lymphoma cell (L 5178Y, TK+/-) forward mutation test, the human lymphocyte chromosome aberration test, or the mouse micronucleus test. Administered by continuous intravenous infusion at total daily doses up to 72 mg/kg/day (about 4 times the recommended maximum daily human dose on a body surface area basis), eptifibatide had no effect on fertility and reproductive performance of male and female rats.
7.1 Use of Thrombolytics, Anticoagulants, and Other Antiplatelet Agents
Coadministration of antiplatelet agents, thrombolytics, heparin, aspirin, and chronic NSAID use increases the risk of bleeding. Concomitant treatment with other inhibitors of platelet receptor GP IIb/IIIa should be avoided.
Structured Label Content
Section 42229-5 (42229-5)
Use of Thrombolytics, Anticoagulants, and Other Antiplatelet Agents
Risk factors for bleeding include older age, a history of bleeding disorders, and concomitant use of drugs that increase the risk of bleeding (thrombolytics, oral anticoagulants, nonsteroidal anti-inflammatory drugs, and P2Y12 inhibitors). Concomitant treatment with other inhibitors of platelet receptor glycoprotein (GP) IIb/IIIa should be avoided. In patients treated with heparin, bleeding can be minimized by close monitoring of the aPTT and ACT [see Dosage and Administration (2)].
Section 51945-4 (51945-4)
NDC 52958-402-01
20 mg/10 mL vial
for weight-adjusted bolus dosing
Eptifibatide
Injection
20 mg/10 mL
(2 mg/mL)
For Intravenous Use Only
1×10 mL Single-dose Vial
Rx only
Shuangcheng
Pharma
16.2 Storage
Vials should be stored refrigerated at 2-8°C (36-46°F). Vials may be transferred to room temperature storage* for a period not to exceed 2 months. Upon transfer, vial cartons must be marked by the dispensing pharmacist with a "DISCARD BY" date (2 months from the transfer date or the labeled expiration date, whichever comes first).
Retain in carton to protect from light until administration.
* Store at 25oC (77oF); excursions permitted to 15-30oC (59-oF) [see USP Controlled Room Temperature].
5.1 Bleeding
Bleeding is the most common complication encountered during eptifibatide therapy. Administration of eptifibatide is associated with an increase in major and minor bleeding, as classified by the criteria of the Thrombolysis in Myocardial Infarction Study group (TIMI) [see Adverse Reactions (6.1)]. Most major bleeding associated with eptifibatide has been at the arterial access site for cardiac catheterization or from the gastrointestinal or genitourinary tract. Minimize the use of arterial and venous punctures, intramuscular injections, and the use of urinary catheters, nasotracheal intubation, and nasogastric tubes. When obtaining intravenous access, avoid non-compressible sites (e.g., subclavian or jugular veins).
10 Overdosage (10 OVERDOSAGE)
There has been only limited experience with overdosage of eptifibatide. There were 8 patients in the IMPACT II study, 9 patients in the PURSUIT study, and no patients in the ESPRIT study who received bolus doses and/or infusion doses more than double those called for in the protocols. None of these patients experienced an intracranial bleed or other major bleeding.
Eptifibatide was not lethal to rats, rabbits, or monkeys when administered by continuous intravenous infusion for 90 minutes at a total dose of 45 mg/kg (about 2 to 5 times the recommended maximum daily human dose on a body surface area basis). Symptoms of acute toxicity were loss of righting reflex, dyspnea, ptosis, and decreased muscle tone in rabbits and petechial hemorrhages in the femoral and abdominal areas of monkeys.
From in vitro studies, eptifibatide is not extensively bound to plasma proteins and thus may be cleared from plasma by dialysis.
8.1 Pregnancy
Risk Summary
Available data on eptifibatide use in pregnant women from published literature and the pharmacovigilance database are insufficient to establish a drug-associated risk of major birth defects, miscarriage or adverse maternal or fetal outcomes. Untreated myocardial infarction can be fatal to the pregnant woman and fetus (see Clinical Considerations). In animal reproduction studies, there was no evidence of adverse developmental effects when eptifibatide was administered intravenously to pregnant rats and rabbits at approximately 4 times the recommended maximum daily human dose.
The estimated background risk of major birth defects and miscarriage for the indicated population is unknown. All pregnancies have a background risk of birth defect, loss, or other adverse outcomes. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2 to 4% and 15 to 20%, respectively.
Clinical Considerations
Disease-associated maternal and/or embryo/fetal risk
Myocardial infarction is a medical emergency in pregnancy which can be fatal to the pregnant woman and fetus if left untreated. Therapy for the pregnant woman should not be withheld because of potential concerns regarding the effects of eptifibatide on the fetus
Data
Animal Data
Embryo-fetal development studies have been performed by continuous intravenous infusion of eptifibatide in pregnant rats during the period of organogenesis at total daily doses of up to 72 mg/kg/day (about 4 times the recommended maximum daily human dose on a body surface area basis) and in pregnant rabbits during the period of organogenesis at total daily doses of up to 36 mg/kg/day (also about 4 times the recommended maximum daily human dose on a body surface area basis). These studies revealed no evidence of harm to the fetus due to eptifibatide.
8.2 Lactation
Risk Summary
There are no available data on the presence of eptifibatide in human milk, the effects on the breastfed infant, or the effects on milk production. As eptifibatide is a peptide, it is likely to be destroyed in the infant’s gastrointestinal tract and not absorbed orally by the breastfed infant.
11 Description (11 DESCRIPTION)
Eptifibatide is a cyclic heptapeptide containing 6 amino acids and 1 mercaptopropionyl (des-amino cysteinyl) residue. An interchain disulfide bridge is formed between the cysteine amide and the mercaptopropionyl moieties. Chemically it is N6-(aminoiminomethyl)-N2-(3-mercapto-1-oxopropyl)-L-lysylglycyl-L-α-aspartyl-L-tryptophyl-L-prolyl-L-cysteinamide, cyclic (1→6)-disulfide. Eptifibatide binds to the platelet receptor glycoprotein (GP) IIb/IIIa of human platelets and inhibits platelet aggregation.
The eptifibatide peptide is produced by solid-phase peptide synthesis, and is purified by preparative reverse-phase liquid chromatography and lyophilized. The structural formula is:
Eptifibatide Injection is a clear, colorless, sterile, non-pyrogenic solution for intravenous (IV) use with an empirical formula of C35H49N11O9S2 and a molecular weight of 831.96. Each 10-mL vial contains 2 mg/mL of eptifibatide and each 100-mL vial contains 0.75 mg/mL of eptifibatide. Each vial of either size also contains 5.25 mg/mL citric acid and sodium hydroxide to adjust the pH to 5.35.
16.1 How Supplied
Eptifibatide injection is supplied as a sterile solution in 10-mL vials containing 20 mg of eptifibatide (NDC 52958-402-01) and 100-mL vials containing 75 mg of eptifibatide (NDC 52958-040-01).
8.4 Pediatric Use
Safety and effectiveness of eptifibatide in pediatric patients have not been studied.
8.5 Geriatric Use
The PURSUIT and IMPACT II clinical studies enrolled patients up to the age of 94 years (45% were age 65 and over; 12% were age 75 and older). There was no apparent difference in efficacy between older and younger patients treated with eptifibatide. The incidence of bleeding complications was higher in the elderly in both placebo and eptifibatide groups, and the incremental risk of eptifibatide-associated bleeding was greater in the older patients. No dose adjustment was made for elderly patients, but patients over 75 years of age had to weigh at least 50 kg to be enrolled in the PURSUIT study; no such limitation was stipulated in the ESPRIT study [see Adverse Reactions (6.1)].
14 Clinical Studies (14 CLINICAL STUDIES)
Eptifibatide was studied in 3 placebo-controlled, randomized studies. PURSUIT evaluated patients with acute coronary syndromes: UA or NSTEMI. Two other studies, ESPRIT and IMPACT II, evaluated patients about to undergo a PCI. Patients underwent primarily balloon angioplasty in IMPACT II and intracoronary stent placement, with or without angioplasty, in ESPRIT.
4 Contraindications (4 CONTRAINDICATIONS)
Treatment with eptifibatide is contraindicated in patients with:
- A history of bleeding diathesis, or evidence of active abnormal bleeding within the previous 30 days
- Severe hypertension (systolic blood pressure >200 mm Hg or diastolic blood pressure >110 mm Hg) not adequately controlled on antihypertensive therapy
- Major surgery within the preceding 6 weeks
- History of stroke within 30 days or any history of hemorrhagic stroke
- Current or planned administration of another parenteral GP IIb/IIIa inhibitor
- Dependency on renal dialysis
- Hypersensitivity to eptifibatide or any component of the product (hypersensitivity reactions that occurred included anaphylaxis and urticaria)
7 Drug Interactions (7 DRUG INTERACTIONS)
- Coadministration of antiplatelet agents, thrombolytics, heparin, aspirin, and chronic NSAID use increases the risk of bleeding. Avoid concomitant use with other glycoprotein (GP) IIb/IIIa inhibitors. (7.1)
5.2 Thrombocytopenia
There have been reports of acute, profound thrombocytopenia (immune-mediated and non-immune mediated) with eptifibatide. In the event of acute profound thrombocytopenia or a confirmed platelet decrease to <100,000/mm3, discontinue eptifibatide and heparin (unfractionated or low-molecular weight). Monitor serial platelet counts, assess the presence of drug-dependent antibodies, and treat as appropriate [see Adverse Reactions (6.1)].
There has been no clinical experience with eptifibatide initiated in patients with a baseline platelet count <100,000/mm3. If a patient with low platelet counts is receiving eptifibatide, their platelet count should be monitored closely.
8.6 Renal Impairment
Approximately 50% of eptifibatide is cleared by the kidney in patients with normal renal function. Total drug clearance is decreased by approximately 50% and steady-state plasma eptifibatide concentrations are doubled in patients with an estimated CrCl <50 mL/min (using the Cockcroft-Gault equation). Therefore, the infusion dose should be reduced to 1 mcg/kg/min in such patients [see Dosage and Administration (2)]. The safety and efficacy of eptifibatide in patients dependent on dialysis has not been established.
12.2 Pharmacodynamics
Infusion of eptifibatide into baboons caused a dose-dependent inhibition of ex vivo platelet aggregation, with complete inhibition of aggregation achieved at infusion rates greater than 5 mcg/kg/min. In a baboon model that is refractory to aspirin and heparin, doses of eptifibatide that inhibit aggregation prevented acute thrombosis with only a modest prolongation (2- to 3-fold) of the bleeding time. Platelet aggregation in dogs was also inhibited by infusions of eptifibatide, with complete inhibition at 2 mcg/kg/min. This infusion dose completely inhibited canine coronary thrombosis induced by coronary artery injury (Folts model).
Human pharmacodynamic data were obtained in healthy subjects and in patients presenting with UA or NSTEMI and/or undergoing percutaneous coronary intervention. Studies in healthy subjects enrolled only males; patient studies enrolled approximately one-third women. In these studies, eptifibatide inhibited ex vivo platelet aggregation induced by adenosine diphosphate (ADP) and other agonists in a dose- and concentration-dependent manner. The effect of eptifibatide was observed immediately after administration of a 180-mcg/kg intravenous bolus. Table 4 shows the effects of dosing regimens of eptifibatide used in the IMPACT II and PURSUIT studies on ex vivo platelet aggregation induced by 20 µM ADP in PPACK-anticoagulated platelet-rich plasma and on bleeding time. The effects of the dosing regimen used in ESPRIT on platelet aggregation have not been studied.
| PURSUIT 180/2 180-mcg/kg bolus followed by a continuous infusion of 2 mcg/kg/min.
|
|
|---|---|
| Inhibition of platelet aggregation 15 min after bolus | 84% |
| Inhibition of platelet aggregation at steady state | >90% |
| Bleeding-time prolongation at steady state | <5x |
| Inhibition of platelet aggregation 4h after infusion discontinuation | <50% |
| Bleeding-time prolongation 6h after infusion discontinuation | 1.4x |
The eptifibatide dosing regimen used in the ESPRIT study included two 180-mcg/kg bolus doses given 10 minutes apart combined with a continuous 2-mcg/kg/min infusion.
When administered alone, eptifibatide has no measurable effect on PT or aPTT.
There were no important differences between men and women or between age groups in the pharmacodynamic properties of eptifibatide. Differences among ethnic groups have not been assessed.
12.3 Pharmacokinetics
The pharmacokinetics of eptifibatide are linear and dose-proportional for bolus doses ranging from 90 to 250 mcg/kg and infusion rates from 0.5 to 3 mcg/kg/min. Plasma elimination half-life is approximately 2.5 hours. Administration of a single 180-mcg/kg bolus combined with an infusion produces an early peak level, followed by a small decline prior to attaining steady state (within 4-6 hours). This decline can be prevented by administering a second 180-mcg/kg bolus 10 minutes after the first. The extent of eptifibatide binding to human plasma protein is about 25%. Clearance in patients with coronary artery disease is about 55 mL/kg/h. In healthy subjects, renal clearance accounts for approximately 50% of total body clearance, with the majority of the drug excreted in the urine as eptifibatide, deaminated eptifibatide, and other, more polar metabolites. No major metabolites have been detected in human plasma.
1 Indications and Usage (1 INDICATIONS AND USAGE)
12.1 Mechanism of Action
Eptifibatide reversibly inhibits platelet aggregation by preventing the binding of fibrinogen, von Willebrand factor, and other adhesive ligands to GP IIb/IIIa. When administered intravenously, eptifibatide inhibits ex vivo platelet aggregation in a dose- and concentration-dependent manner. Platelet aggregation inhibition is reversible following cessation of the eptifibatide infusion; this is thought to result from dissociation of eptifibatide from the platelet.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Eptifibatide can cause serious bleeding. If bleeding cannot be controlled, discontinue eptifibatide immediately. Minimize vascular and other traumas. If heparin is given concomitantly, monitor aPTT or ACT. (5.1)
- Thrombocytopenia: Discontinue eptifibatide and heparin. Monitor and treat condition appropriately. (5.2)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
Before infusion of eptifibatide injection, the following laboratory tests should be performed to identify pre-existing hemostatic abnormalities: hematocrit or hemoglobin, platelet count, serum creatinine, and PT/aPTT. In patients undergoing PCI, the activated clotting time (ACT) should also be measured.
The activated partial thromboplastin time (aPTT) should be maintained between 50 and 70 seconds unless PCI is to be performed. In patients treated with heparin, bleeding can be minimized by close monitoring of the aPTT and ACT.
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
- Injection: 20 mg of eptifibatide in 10 mL (2 mg/mL), for intravenous bolus.
- Injection: 75 mg of eptifibatide in 100 mL (0.75 mg/mL), for intravenous infusion.
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
- Geriatric Use: Risk of bleeding increases with age. (8.5)
1.1 Acute Coronary Syndrome (acs) (1.1 Acute Coronary Syndrome (ACS))
Eptifibatide injection is indicated to decrease the rate of a combined endpoint of death or new myocardial infarction (MI) in patients with ACS (unstable angina [UA]/non-ST-elevation myocardial infarction [NSTEMI]), including patients who are to be managed medically and those undergoing percutaneous coronary intervention (PCI).
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient to inform the doctor or healthcare provider about any medical conditions, medications, and allergies.
2.3 Important Administration Instructions
- Inspect eptifibatide injection for particulate matter and discoloration prior to administration, whenever solution and container permit.
- May administer eptifibatide injection in the same intravenous line as alteplase, atropine, dobutamine, heparin, lidocaine, meperidine, metoprolol, midazolam, morphine, nitroglycerin, or verapamil. Do not administer eptifibatide injection through the same intravenous line as furosemide.
- May administer eptifibatide injection in the same IV line with 0.9% NaCl or 0.9% NaCl/5% dextrose. With either vehicle, the infusion may also contain up to 60 mEq/L of potassium chloride.
- Withdraw the bolus dose(s) of eptifibatide injection from the 10-mL vial into a syringe. Administer the bolus dose(s) by IV push.
- Immediately following the bolus dose administration, initiate a continuous infusion of eptifibatide injection. When using an intravenous infusion pump, administer eptifibatide injection undiluted directly from the 100-mL vial. Spike the 100-mL vial with a vented infusion set. Center the spike within the circle on the stopper top.
- Discard any unused portion left in the vial.
Administer eptifibatide injection by volume according to patient weight (see Table 1).
| Patient Weight | 180-mcg/kg Bolus Volume |
2-mcg/kg/min Infusion Volume (CrCl greater than or equal to 50 mL/min) |
1-mcg/kg/min Infusion Volume (CrCl less than 50 mL/min) |
|||
|---|---|---|---|---|---|---|
| (kg) | (lb) | (from 2-mg/mL vial) | (from 2-mg/mL 100-mL vial) | (from 0.75-mg/mL 100-mL vial) | (from 2-mg/mL 100-mL vial) | (from 0.75-mg/mL 100-mL vial) |
| 37-41 | 81-91 | 3.4 mL | 2 mL/h | 6 mL/h | 1 mL/h | 3 mL/h |
| 42-46 | 92-102 | 4 mL | 2.5 mL/h | 7 mL/h | 1.3 mL/h | 3.5 mL/h |
| 47-53 | 103-117 | 4.5 mL | 3 mL/h | 8 mL/h | 1.5 mL/h | 4 mL/h |
| 54-59 | 118-130 | 5 mL | 3.5 mL/h | 9 mL/h | 1.8 mL/h | 4.5 mL/h |
| 60-65 | 131-143 | 5.6 mL | 3.8 mL/h | 10 mL/h | 1.9 mL/h | 5 mL/h |
| 66-71 | 144-157 | 6.2 mL | 4 mL/h | 11 mL/h | 2 mL/h | 5.5 mL/h |
| 72-78 | 158-172 | 6.8 mL | 4.5 mL/h | 12 mL/h | 2.3 mL/h | 6 mL/h |
| 79-84 | 173-185 | 7.3 mL | 5 mL/h | 13 mL/h | 2.5 mL/h | 6.5 mL/h |
| 85-90 | 186-198 | 7.9 mL | 5.3 mL/h | 14 mL/h | 2.7 mL/h | 7 mL/h |
| 91-96 | 199-212 | 8.5 mL | 5.6 mL/h | 15 mL/h | 2.8 mL/h | 7.5 mL/h |
| 97-103 | 213-227 | 9 mL | 6 mL/h | 16 mL/h | 3.0 mL/h | 8 mL/h |
| 104-109 | 228-240 | 9.5 mL | 6.4 mL/h | 17 mL/h | 3.2 mL/h | 8.5 mL/h |
| 110-115 | 241-253 | 10.2 mL | 6.8 mL/h | 18 mL/h | 3.4 mL/h | 9 mL/h |
| 116-121 | 254-267 | 10.7 mL | 7 mL/h | 19 mL/h | 3.5 mL/h | 9.5 mL/h |
| >121 | >267 | 11.3 mL | 7.5 mL/h | 20 mL/h | 3.7 mL/h | 10 mL/h |
2.1 Dosage in Acute Coronary Syndrome (acs) (2.1 Dosage in Acute Coronary Syndrome (ACS))
| Indication | Normal Renal Function | Creatinine Clearance less than 50 mL/min |
|---|---|---|
| Patients with ACS | 180 mcg/kg intravenous (IV) bolus as soon as possible after diagnosis, followed by continuous infusion of 2 mcg/kg/min | 180 mcg/kg IV bolus as soon as possible after diagnosis, followed by continuous infusion of 1 mcg/kg/min |
|
Eptifibatide injection should be given concomitantly with heparin dosed to achieve the following parameters:
During Medical Management: Target aPTT 50 to 70 seconds
- If weight greater than or equal to 70 kg, 5000-unit bolus followed by infusion of 1000 units/h.
- If weight less than 70 kg, 60-units/kg bolus followed by infusion of 12 units/kg/h.
During PCI: Target ACT 200 to 300 seconds
- If heparin is initiated prior to PCI, additional boluses during PCI to maintain an ACT target of 200 to 300 seconds.
- Heparin infusion after the PCI is discouraged.
1.2 Percutaneous Coronary Intervention (pci) (1.2 Percutaneous Coronary Intervention (PCI))
Eptifibatide injection is indicated to decrease the rate of a combined endpoint of death, new MI, or need for urgent intervention in patients undergoing PCI, including those undergoing intracoronary stenting [see Clinical Studies (14.1, 14.2)].
14.1 Non St Segment Elevation Acute Coronary Syndrome (14.1 Non-ST-Segment Elevation Acute Coronary Syndrome)
Non-ST-segment elevation acute coronary syndrome is defined as prolonged (≥10 minutes) symptoms of cardiac ischemia within the previous 24 hours associated with either ST-segment changes (elevations between 0.6 mm and 1 mm or depression >0.5 mm), T-wave inversion (>1 mm), or positive CK-MB. This definition includes "unstable angina" and "NSTEMI" but excludes MI that is associated with Q waves or greater degrees of ST-segment elevation.
2.2 Dosage in Percutaneous Coronary Intervention (pci) (2.2 Dosage in Percutaneous Coronary Intervention (PCI))
| Indication | Normal Renal Function | Creatinine Clearance less than 50 mL/min |
|---|---|---|
| Patients with PCI | 180 mcg/kg IV bolus immediately before PCI followed by continuous infusion of 2 mcg/kg/min and a second bolus of 180 mcg/kg (given 10 minutes after the first bolus) | 180 mcg/kg IV bolus immediately before PCI followed by continuous infusion of 1 mcg/kg/min and a second bolus of 180 mcg/kg (given 10 minutes after the first bolus) |
|
- Eptifibatide injection should be given concomitantly with heparin to achieve a target ACT of 200 to 300 seconds. Administer 60-units/kg bolus initially in patients not treated with heparin within 6 hours prior to PCI.
- Additional boluses during PCI to maintain ACT within target.
- Heparin infusion after the PCI is strongly discouraged.
Patients requiring thrombolytic therapy should discontinue eptifibatide injection.
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
No long-term studies in animals have been performed to evaluate the carcinogenic potential of eptifibatide. Eptifibatide was not genotoxic in the Ames test, the mouse lymphoma cell (L 5178Y, TK+/-) forward mutation test, the human lymphocyte chromosome aberration test, or the mouse micronucleus test. Administered by continuous intravenous infusion at total daily doses up to 72 mg/kg/day (about 4 times the recommended maximum daily human dose on a body surface area basis), eptifibatide had no effect on fertility and reproductive performance of male and female rats.
7.1 Use of Thrombolytics, Anticoagulants, and Other Antiplatelet Agents
Coadministration of antiplatelet agents, thrombolytics, heparin, aspirin, and chronic NSAID use increases the risk of bleeding. Concomitant treatment with other inhibitors of platelet receptor GP IIb/IIIa should be avoided.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:50:49.402612 · Updated: 2026-03-14T22:38:43.060348