These Highlights Do Not Include All The Information Needed To Use Pantoprazole Sodium For Injection Safely And Effectively. See Full Prescribing Information For Pantoprazole Sodium For Injection.
c39be14b-e881-4586-b77e-4747db494954
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Pantoprazole sodium is a proton pump inhibitor (PPI) indicated in adults for the following: Short-term treatment (7 to 10 days) of gastroesophageal reflux disease (GERD) associated with a history of Erosive Esophagitis (EE). ( 1.1 ) Pathological hypersecretion conditions including Zollinger-Ellison (ZE) Syndrome. ( 1.2 )
Indications and Usage
Pantoprazole sodium is a proton pump inhibitor (PPI) indicated in adults for the following: Short-term treatment (7 to 10 days) of gastroesophageal reflux disease (GERD) associated with a history of Erosive Esophagitis (EE). ( 1.1 ) Pathological hypersecretion conditions including Zollinger-Ellison (ZE) Syndrome. ( 1.2 )
Dosage and Administration
GERD Associated with EE ( 2.1 ) The recommended adult dosage is 40 mg given once daily by intravenous infusion for 7 to 10 days. ( 2.1 ) Pathological Hypersecretion Conditions, Including ZE Syndrome ( 2.3 ): The recommended adult dosage is 80 mg administered every 12 hours by intravenous infusion. For information on how to adjust dosing for individual patient needs, see the full prescribing information. Administration ( 2.2 , 2.4 ): Only for intravenous infusion. The intravenous infusion can be administered over 2 minutes or 15 minutes. For information on how to prepare and administer for each indication, see the full prescribing information.
Warnings and Precautions
Gastric Malignancy : In adults, symptomatic response to therapy with pantoprazole does not preclude the presence of gastric malignancy. Consider additional follow-up and diagnostic testing. ( 5.1 ) Injection Site Reactions : Thrombophlebitis is associated with the administration of intravenous pantoprazole. ( 5.2 ) Potential Exacerbation of Zinc Deficiency : Consider zinc supplementation in patients who are prone to zinc deficiency. Caution should be used when other EDTA containing products are also co-administered intravenously. ( 5.3 ) Acute Tubulointerstitial Nephritis : Discontinue treatment and evaluate patients. ( 5.4 ) Clostridium difficile -Associated Diarrhea : PPI therapy may be associated with increased risk. ( 5.5 ) Bone Fracture : Long term and multiple daily dose PPI therapy may be associated with an increased risk for osteoporosis-related fractures of the hip, wrist or spine. ( 5.6 ) Severe Cutaneous Adverse Reactions : Discontinue at the first signs or symptoms of severe cutaneous adverse reactions or other signs of hypersensitivity and consider further evaluation. ( 5.7 ) Cutaneous and Systemic Lupus Erythematosus : Mostly cutaneous; new onset or exacerbation of existing disease; discontinue pantoprazole sodium for injection and refer to specialist for evaluation. ( 5.8 ) Hepatic Effects : Elevations of transaminases observed. ( 5.9 ) Hypomagnesemia and Mineral Metabolism : Reported rarely with prolonged treatment with PPIs. ( 5.10 ) Fundic Gland Polyps : Risk increases with long-term use, especially beyond one year. Use the shortest duration of therapy. ( 5.11 )
Contraindications
Pantoprazole sodium for injection is contraindicated in patients with known hypersensitivity reactions including anaphylaxis to the formulation or any substituted benzimidazole. Hypersensitivity reactions may include anaphylaxis, anaphylactic shock, angioedema, bronchospasm, acute tubulointerstitial nephritis, and urticaria [see Warnings and Precautions ( 5.2 , 5.4 ), Adverse Reactions ( 6 )] . Proton pump inhibitors (PPIs), including pantoprazole sodium for injection, are contraindicated in patients receiving rilpivirine-containing products [see Drug Interactions ( 7 )] .
Adverse Reactions
The following serious adverse reactions are described below and elsewhere in labeling: Injection Site Reactions [see Warnings and Precautions ( 5.2 )] Potential for Exacerbation of Zinc Deficiency [see Warnings and Precautions ( 5.3 )] Acute Tubulointerstitial Nephritis [see Warnings and Precautions ( 5.4 )] Clostridium difficile -Associated Diarrhea [see Warnings and Precautions ( 5.5 )] Bone Fracture [see Warnings and Precautions ( 5.6 )] Severe Cutaneous Adverse Reactions [see Warnings and Precautions ( 5.7 )] Cutaneous and Systemic Lupus Erythematosus [see Warnings and Precautions ( 5.8 )] Hepatic Effects [see Warnings and Precautions ( 5.9 )] Hypomagnesemia and Mineral Metabolism [see Warnings and Precautions ( 5.10 )] Fundic Gland Polyps [see Warnings and Precautions ( 5.11 )]
Drug Interactions
Table 2 includes drugs with clinically important drug interactions and interaction with diagnostics when administered concomitantly with pantoprazole sodium for injection and instructions for preventing or managing them. Consult the labeling of concomitantly used drugs to obtain further information about interactions with PPIs. Table 2: Clinically Relevant Interactions Affecting Drugs Co-Administered with Pantoprazole Sodium for Injection and Interaction with Diagnostics Antiretrovirals Clinical Impact: The effect of PPIs on antiretroviral drugs is variable. The clinical importance and the mechanisms behind these interactions are not always known. Decreased exposure of some antiretroviral drugs (e.g., rilpivirine atazanavir, and nelfinavir) when used concomitantly with pantoprazole may reduce antiviral effect and promote the development of drug resistance. Increased exposure of other antiretroviral drugs (e.g., saquinavir) when used concomitantly with pantoprazole may increase toxicity of the antiretroviral drugs. There are other antiretroviral drugs which do not result in clinically relevant interactions with pantoprazole. Intervention: Rilpivirine-containing products : Concomitant use with pantoprazole sodium for injection is contraindicated [see Contraindications ( 4 )] . See prescribing information. Atazanavir : See prescribing information for atazanavir for dosing information. Nelfinavir : Avoid concomitant use with pantoprazole sodium for injection See prescribing information for nelfinavir. Saquinavir : See the prescribing information for saquinavir and monitor for potential saquinavir toxicities. Other antiretrovirals : See prescribing information. Warfarin Clinical Impact: Increased INR and prothrombin time in patients receiving PPIs, including pantoprazole, and warfarin concomitantly. Increases in INR and prothrombin time may lead to abnormal bleeding and even death. Intervention: Monitor INR and prothrombin time. Dose adjustment of warfarin may be needed to maintain target INR range. See prescribing information for warfarin. Clopidogrel Clinical Impact: Concomitant administration of pantoprazole and clopidogrel in healthy subjects had no clinically important effect on exposure to the active metabolite of clopidogrel or clopidogrel-induced platelet inhibition [see Clinical Pharmacology ( 12.3 )]. Intervention: No dose adjustment of clopidogrel is necessary when administered with an approved dose of pantoprazole sodium for injection. Methotrexate Clinical Impact: Concomitant use of PPIs with methotrexate (primarily at high dose) may elevate and prolong serum concentrations of methotrexate and/or its metabolite hydroxymethotrexate, possibly leading to methotrexate toxicities. No formal drug interaction studies of high-dose methotrexate with PPIs have been conducted [see Warnings and Precautions ( 5.14 )] . Intervention: A temporary withdrawal of pantoprazole sodium for injection may be considered in some patients receiving high-dose methotrexate. Drugs Dependent on Gastric pH for Absorption (e.g., iron salts, erlotinib, dasatinib, nilotinib, mycophenolate mofetil, ketoconazole/itraconazole) Clinical Impact: Pantoprazole can reduce the absorption of other drugs due to its effect on reducing intragastric acidity. Intervention: Mycophenolate mofetil (MMF): Co-administration of pantoprazole sodium in healthy subjects and in transplant patients receiving MMF has been reported to reduce the exposure to the active metabolite, mycophenolic acid (MPA), possibly due to a decrease in MMF solubility at an increased gastric pH [see Clinical Pharmacology ( 12.3 )] . The clinical relevance of reduced MPA exposure on organ rejection has not been established in transplant patients receiving pantoprazole sodium for injection and MMF. Use pantoprazole sodium for injection with caution in transplant patients receiving MMF. See the prescribing information for other drugs dependent on gastric pH for absorption. Interactions with Investigations of Neuroendocrine Tumors Clinical Impact: CgA levels increase secondary to PPI-induced decreases in gastric acidity. The increased CgA level may cause false positive results in diagnostic investigations for neuroendocrine tumors [see Warnings and Precautions ( 5.12 ), Clinical Pharmacology ( 12.2 )] . Intervention: Temporarily stop pantoprazole sodium for injection treatment at least 14 days before assessing CgA levels and consider repeating the test if initial CgA levels are high. If serial tests are performed (e.g. for monitoring), the same commercial laboratory should be used for testing, as reference ranges between tests may vary. False Positive Urine Tests for THC Clinical Impact: There have been reports of false positive urine screening tests for tetrahydrocannabinol (THC) in patients receiving PPIs [see Warnings and Precautions ( 5.13 )] . Intervention: An alternative confirmatory method should be considered to verify positive results.
Storage and Handling
Pantoprazole Sodium for Injection is available as follows: NDC Pantoprazole Sodium for Injection Package Factor 25021-751-10 40 mg Single-Dose Vial 10 vials per carton 25021-751-11 40 mg Single-Dose Vial 25 vials per carton Pantoprazole Sodium for Injection is a white to off-white freeze-dried powder for reconstitution.
How Supplied
Pantoprazole Sodium for Injection is available as follows: NDC Pantoprazole Sodium for Injection Package Factor 25021-751-10 40 mg Single-Dose Vial 10 vials per carton 25021-751-11 40 mg Single-Dose Vial 25 vials per carton Pantoprazole Sodium for Injection is a white to off-white freeze-dried powder for reconstitution.
Medication Information
Warnings and Precautions
Gastric Malignancy : In adults, symptomatic response to therapy with pantoprazole does not preclude the presence of gastric malignancy. Consider additional follow-up and diagnostic testing. ( 5.1 ) Injection Site Reactions : Thrombophlebitis is associated with the administration of intravenous pantoprazole. ( 5.2 ) Potential Exacerbation of Zinc Deficiency : Consider zinc supplementation in patients who are prone to zinc deficiency. Caution should be used when other EDTA containing products are also co-administered intravenously. ( 5.3 ) Acute Tubulointerstitial Nephritis : Discontinue treatment and evaluate patients. ( 5.4 ) Clostridium difficile -Associated Diarrhea : PPI therapy may be associated with increased risk. ( 5.5 ) Bone Fracture : Long term and multiple daily dose PPI therapy may be associated with an increased risk for osteoporosis-related fractures of the hip, wrist or spine. ( 5.6 ) Severe Cutaneous Adverse Reactions : Discontinue at the first signs or symptoms of severe cutaneous adverse reactions or other signs of hypersensitivity and consider further evaluation. ( 5.7 ) Cutaneous and Systemic Lupus Erythematosus : Mostly cutaneous; new onset or exacerbation of existing disease; discontinue pantoprazole sodium for injection and refer to specialist for evaluation. ( 5.8 ) Hepatic Effects : Elevations of transaminases observed. ( 5.9 ) Hypomagnesemia and Mineral Metabolism : Reported rarely with prolonged treatment with PPIs. ( 5.10 ) Fundic Gland Polyps : Risk increases with long-term use, especially beyond one year. Use the shortest duration of therapy. ( 5.11 )
Indications and Usage
Pantoprazole sodium is a proton pump inhibitor (PPI) indicated in adults for the following: Short-term treatment (7 to 10 days) of gastroesophageal reflux disease (GERD) associated with a history of Erosive Esophagitis (EE). ( 1.1 ) Pathological hypersecretion conditions including Zollinger-Ellison (ZE) Syndrome. ( 1.2 )
Dosage and Administration
GERD Associated with EE ( 2.1 ) The recommended adult dosage is 40 mg given once daily by intravenous infusion for 7 to 10 days. ( 2.1 ) Pathological Hypersecretion Conditions, Including ZE Syndrome ( 2.3 ): The recommended adult dosage is 80 mg administered every 12 hours by intravenous infusion. For information on how to adjust dosing for individual patient needs, see the full prescribing information. Administration ( 2.2 , 2.4 ): Only for intravenous infusion. The intravenous infusion can be administered over 2 minutes or 15 minutes. For information on how to prepare and administer for each indication, see the full prescribing information.
Contraindications
Pantoprazole sodium for injection is contraindicated in patients with known hypersensitivity reactions including anaphylaxis to the formulation or any substituted benzimidazole. Hypersensitivity reactions may include anaphylaxis, anaphylactic shock, angioedema, bronchospasm, acute tubulointerstitial nephritis, and urticaria [see Warnings and Precautions ( 5.2 , 5.4 ), Adverse Reactions ( 6 )] . Proton pump inhibitors (PPIs), including pantoprazole sodium for injection, are contraindicated in patients receiving rilpivirine-containing products [see Drug Interactions ( 7 )] .
Adverse Reactions
The following serious adverse reactions are described below and elsewhere in labeling: Injection Site Reactions [see Warnings and Precautions ( 5.2 )] Potential for Exacerbation of Zinc Deficiency [see Warnings and Precautions ( 5.3 )] Acute Tubulointerstitial Nephritis [see Warnings and Precautions ( 5.4 )] Clostridium difficile -Associated Diarrhea [see Warnings and Precautions ( 5.5 )] Bone Fracture [see Warnings and Precautions ( 5.6 )] Severe Cutaneous Adverse Reactions [see Warnings and Precautions ( 5.7 )] Cutaneous and Systemic Lupus Erythematosus [see Warnings and Precautions ( 5.8 )] Hepatic Effects [see Warnings and Precautions ( 5.9 )] Hypomagnesemia and Mineral Metabolism [see Warnings and Precautions ( 5.10 )] Fundic Gland Polyps [see Warnings and Precautions ( 5.11 )]
Drug Interactions
Table 2 includes drugs with clinically important drug interactions and interaction with diagnostics when administered concomitantly with pantoprazole sodium for injection and instructions for preventing or managing them. Consult the labeling of concomitantly used drugs to obtain further information about interactions with PPIs. Table 2: Clinically Relevant Interactions Affecting Drugs Co-Administered with Pantoprazole Sodium for Injection and Interaction with Diagnostics Antiretrovirals Clinical Impact: The effect of PPIs on antiretroviral drugs is variable. The clinical importance and the mechanisms behind these interactions are not always known. Decreased exposure of some antiretroviral drugs (e.g., rilpivirine atazanavir, and nelfinavir) when used concomitantly with pantoprazole may reduce antiviral effect and promote the development of drug resistance. Increased exposure of other antiretroviral drugs (e.g., saquinavir) when used concomitantly with pantoprazole may increase toxicity of the antiretroviral drugs. There are other antiretroviral drugs which do not result in clinically relevant interactions with pantoprazole. Intervention: Rilpivirine-containing products : Concomitant use with pantoprazole sodium for injection is contraindicated [see Contraindications ( 4 )] . See prescribing information. Atazanavir : See prescribing information for atazanavir for dosing information. Nelfinavir : Avoid concomitant use with pantoprazole sodium for injection See prescribing information for nelfinavir. Saquinavir : See the prescribing information for saquinavir and monitor for potential saquinavir toxicities. Other antiretrovirals : See prescribing information. Warfarin Clinical Impact: Increased INR and prothrombin time in patients receiving PPIs, including pantoprazole, and warfarin concomitantly. Increases in INR and prothrombin time may lead to abnormal bleeding and even death. Intervention: Monitor INR and prothrombin time. Dose adjustment of warfarin may be needed to maintain target INR range. See prescribing information for warfarin. Clopidogrel Clinical Impact: Concomitant administration of pantoprazole and clopidogrel in healthy subjects had no clinically important effect on exposure to the active metabolite of clopidogrel or clopidogrel-induced platelet inhibition [see Clinical Pharmacology ( 12.3 )]. Intervention: No dose adjustment of clopidogrel is necessary when administered with an approved dose of pantoprazole sodium for injection. Methotrexate Clinical Impact: Concomitant use of PPIs with methotrexate (primarily at high dose) may elevate and prolong serum concentrations of methotrexate and/or its metabolite hydroxymethotrexate, possibly leading to methotrexate toxicities. No formal drug interaction studies of high-dose methotrexate with PPIs have been conducted [see Warnings and Precautions ( 5.14 )] . Intervention: A temporary withdrawal of pantoprazole sodium for injection may be considered in some patients receiving high-dose methotrexate. Drugs Dependent on Gastric pH for Absorption (e.g., iron salts, erlotinib, dasatinib, nilotinib, mycophenolate mofetil, ketoconazole/itraconazole) Clinical Impact: Pantoprazole can reduce the absorption of other drugs due to its effect on reducing intragastric acidity. Intervention: Mycophenolate mofetil (MMF): Co-administration of pantoprazole sodium in healthy subjects and in transplant patients receiving MMF has been reported to reduce the exposure to the active metabolite, mycophenolic acid (MPA), possibly due to a decrease in MMF solubility at an increased gastric pH [see Clinical Pharmacology ( 12.3 )] . The clinical relevance of reduced MPA exposure on organ rejection has not been established in transplant patients receiving pantoprazole sodium for injection and MMF. Use pantoprazole sodium for injection with caution in transplant patients receiving MMF. See the prescribing information for other drugs dependent on gastric pH for absorption. Interactions with Investigations of Neuroendocrine Tumors Clinical Impact: CgA levels increase secondary to PPI-induced decreases in gastric acidity. The increased CgA level may cause false positive results in diagnostic investigations for neuroendocrine tumors [see Warnings and Precautions ( 5.12 ), Clinical Pharmacology ( 12.2 )] . Intervention: Temporarily stop pantoprazole sodium for injection treatment at least 14 days before assessing CgA levels and consider repeating the test if initial CgA levels are high. If serial tests are performed (e.g. for monitoring), the same commercial laboratory should be used for testing, as reference ranges between tests may vary. False Positive Urine Tests for THC Clinical Impact: There have been reports of false positive urine screening tests for tetrahydrocannabinol (THC) in patients receiving PPIs [see Warnings and Precautions ( 5.13 )] . Intervention: An alternative confirmatory method should be considered to verify positive results.
Storage and Handling
Pantoprazole Sodium for Injection is available as follows: NDC Pantoprazole Sodium for Injection Package Factor 25021-751-10 40 mg Single-Dose Vial 10 vials per carton 25021-751-11 40 mg Single-Dose Vial 25 vials per carton Pantoprazole Sodium for Injection is a white to off-white freeze-dried powder for reconstitution.
How Supplied
Pantoprazole Sodium for Injection is available as follows: NDC Pantoprazole Sodium for Injection Package Factor 25021-751-10 40 mg Single-Dose Vial 10 vials per carton 25021-751-11 40 mg Single-Dose Vial 25 vials per carton Pantoprazole Sodium for Injection is a white to off-white freeze-dried powder for reconstitution.
Description
Pantoprazole sodium is a proton pump inhibitor (PPI) indicated in adults for the following: Short-term treatment (7 to 10 days) of gastroesophageal reflux disease (GERD) associated with a history of Erosive Esophagitis (EE). ( 1.1 ) Pathological hypersecretion conditions including Zollinger-Ellison (ZE) Syndrome. ( 1.2 )
Section 42229-5
Fifteen Minute Infusion
- Reconstitute pantoprazole sodium for injection with 10 mL of 0.9% Sodium Chloride Injection, USP.
- Further dilute with 100 mL of 5% Dextrose Injection, USP, 0.9% Sodium Chloride Injection, USP, or Lactated Ringer's Injection, USP, to a final concentration of approximately 0.4 mg per mL.
- Inspect the diluted pantoprazole sodium for injection solution visually for particular matter and discoloration prior to and during administration.
- Administer intravenously over a period of approximately 15 minutes at a rate of approximately 7 mL/min.
Section 44425-7
Storage Conditions
Store at 20° to 25°C (68° to 77°F); excursions permitted to 15° to 30°C (59° to 86°F). [See USP Controlled Room Temperature.]
Protect from light. Retain in carton until time of use.
Discard unused portion.
Sterile, Nonpyrogenic, Preservative-free.
The container closure is not made with natural rubber latex.
Section 51945-4
PACKAGE LABEL – PRINCIPAL DISPLAY PANEL – Vial Label
NDC 25021-751-10
Rx only
Pantoprazole Sodium for Injection
40 mg per vial
For Intravenous Infusion Only
Single-Dose Vial
10 Overdosage
Experience in patients taking very high doses of pantoprazole (greater than 240 mg) is limited. Adverse reactions seen in spontaneous reports of overdose generally reflect the known safety profile of pantoprazole.
Pantoprazole is not removed by hemodialysis. In case of overdose, treatment should be symptomatic and supportive.
Single intravenous doses of pantoprazole at 378, 230, and 266 mg/kg (38, 46, and 177 times the recommended human dose based on body surface area) were lethal to mice, rats and dogs, respectively. The symptoms of acute toxicity were hypoactivity, ataxia, hunched sitting, limb-splay, lateral position, segregation, absence of ear reflex, and tremor.
11 Description
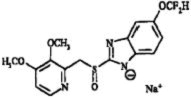
The active ingredient in pantoprazole sodium for injection, a PPI, is a substituted benzimidazole, sodium 5-(difluoromethoxy)-2-[[(3,4-dimethoxy-2-pyridinyl)methyl] sulfinyl]-1H-benzimidazole, a compound that inhibits gastric acid secretion. Its empirical formula is C16H14F2N3NaO4S, with a molecular weight of 405.4. The structural formula is:
Pantoprazole sodium USP is a white to off-white crystalline powder and is racemic. Pantoprazole has weakly basic and acidic properties. Pantoprazole sodium, USP is freely soluble in water, very slightly soluble in phosphate buffer at pH 7.4, and practically insoluble in n-hexane. The stability of the compound in aqueous solution is pH-dependent. The rate of degradation increases with decreasing pH. The reconstituted solution of pantoprazole sodium for injection is in the pH range 9.0 to 10.5.
Pantoprazole sodium for injection is supplied for intravenous administration as a sterile, freeze-dried powder in a single-dose clear glass vial fitted with a rubber stopper and crimp seal. Each vial contains 40 mg pantoprazole (equivalent to 45.1 mg of pantoprazole sodium), edetate disodium (1 mg), and sodium hydroxide to adjust pH.
5.6 Bone Fracture
Several published observational studies suggest that PPI therapy may be associated with an increased risk for osteoporosis-related fractures of the hip, wrist, or spine. The risk of fracture was increased in patients who received high-dose, defined as multiple daily doses, and long-term PPI therapy (a year or longer). Patients should use the lowest dose and shortest duration of PPI therapy appropriate to the condition being treated. Patients at risk for osteoporosis-related fractures should be managed according to established treatment guidelines [see Dosage and Administration (2.2, 2.4), Adverse Reactions (6)].
8.4 Pediatric Use
The safety and effectiveness of pantoprazole sodium for injection have not been established in pediatric patients.
8.5 Geriatric Use
Of 286 patients in clinical studies of intravenous pantoprazole sodium in patients with GERD and a history of EE, 86 (43%) were 65 years of age and over. No overall differences in safety or effectiveness were observed between these subjects and younger subjects, and other reported clinical experience with oral pantoprazole sodium has not identified differences in responses between the elderly and younger patients, but greater sensitivity of some older individuals cannot be ruled out.
4 Contraindications
- Pantoprazole sodium for injection is contraindicated in patients with known hypersensitivity reactions including anaphylaxis to the formulation or any substituted benzimidazole. Hypersensitivity reactions may include anaphylaxis, anaphylactic shock, angioedema, bronchospasm, acute tubulointerstitial nephritis, and urticaria [see Warnings and Precautions (5.2, 5.4), Adverse Reactions (6)].
- Proton pump inhibitors (PPIs), including pantoprazole sodium for injection, are contraindicated in patients receiving rilpivirine-containing products [see Drug Interactions (7)].
5.9 Hepatic Effects
Mild, transient transaminase elevations have been observed in clinical studies. The clinical significance of this finding in a large population of subjects administered pantoprazole sodium for injection is unknown [see Adverse Reactions (6)].
6 Adverse Reactions
The following serious adverse reactions are described below and elsewhere in labeling:
- Injection Site Reactions [see Warnings and Precautions (5.2)]
- Potential for Exacerbation of Zinc Deficiency [see Warnings and Precautions (5.3)]
- Acute Tubulointerstitial Nephritis [see Warnings and Precautions (5.4)]
- Clostridium difficile-Associated Diarrhea [see Warnings and Precautions (5.5)]
- Bone Fracture [see Warnings and Precautions (5.6)]
- Severe Cutaneous Adverse Reactions [see Warnings and Precautions (5.7)]
- Cutaneous and Systemic Lupus Erythematosus [see Warnings and Precautions (5.8)]
- Hepatic Effects [see Warnings and Precautions (5.9)]
- Hypomagnesemia and Mineral Metabolism [see Warnings and Precautions (5.10)]
- Fundic Gland Polyps [see Warnings and Precautions (5.11)]
7 Drug Interactions
Table 2 includes drugs with clinically important drug interactions and interaction with diagnostics when administered concomitantly with pantoprazole sodium for injection and instructions for preventing or managing them.
Consult the labeling of concomitantly used drugs to obtain further information about interactions with PPIs.
| Antiretrovirals | |
| Clinical Impact: | The effect of PPIs on antiretroviral drugs is variable. The clinical importance and the mechanisms behind these interactions are not always known.
|
| Intervention: |
Rilpivirine-containing products: Concomitant use with pantoprazole sodium for injection is contraindicated [see Contraindications (4)]. See prescribing information. Atazanavir: See prescribing information for atazanavir for dosing information. Nelfinavir: Avoid concomitant use with pantoprazole sodium for injection See prescribing information for nelfinavir. Saquinavir: See the prescribing information for saquinavir and monitor for potential saquinavir toxicities. Other antiretrovirals: See prescribing information. |
| Warfarin | |
| Clinical Impact: | Increased INR and prothrombin time in patients receiving PPIs, including pantoprazole, and warfarin concomitantly. Increases in INR and prothrombin time may lead to abnormal bleeding and even death. |
| Intervention: | Monitor INR and prothrombin time. Dose adjustment of warfarin may be needed to maintain target INR range. See prescribing information for warfarin. |
| Clopidogrel | |
| Clinical Impact: | Concomitant administration of pantoprazole and clopidogrel in healthy subjects had no clinically important effect on exposure to the active metabolite of clopidogrel or clopidogrel-induced platelet inhibition [see Clinical Pharmacology (12.3)]. |
| Intervention: | No dose adjustment of clopidogrel is necessary when administered with an approved dose of pantoprazole sodium for injection. |
| Methotrexate | |
| Clinical Impact: | Concomitant use of PPIs with methotrexate (primarily at high dose) may elevate and prolong serum concentrations of methotrexate and/or its metabolite hydroxymethotrexate, possibly leading to methotrexate toxicities. No formal drug interaction studies of high-dose methotrexate with PPIs have been conducted [see Warnings and Precautions (5.14)]. |
| Intervention: | A temporary withdrawal of pantoprazole sodium for injection may be considered in some patients receiving high-dose methotrexate. |
| Drugs Dependent on Gastric pH for Absorption (e.g., iron salts, erlotinib, dasatinib, nilotinib, mycophenolate mofetil, ketoconazole/itraconazole) | |
| Clinical Impact: | Pantoprazole can reduce the absorption of other drugs due to its effect on reducing intragastric acidity. |
| Intervention: | Mycophenolate mofetil (MMF): Co-administration of pantoprazole sodium in healthy subjects and in transplant patients receiving MMF has been reported to reduce the exposure to the active metabolite, mycophenolic acid (MPA), possibly due to a decrease in MMF solubility at an increased gastric pH [see Clinical Pharmacology (12.3)]. The clinical relevance of reduced MPA exposure on organ rejection has not been established in transplant patients receiving pantoprazole sodium for injection and MMF. Use pantoprazole sodium for injection with caution in transplant patients receiving MMF. See the prescribing information for other drugs dependent on gastric pH for absorption. |
| Interactions with Investigations of Neuroendocrine Tumors | |
| Clinical Impact: | CgA levels increase secondary to PPI-induced decreases in gastric acidity. The increased CgA level may cause false positive results in diagnostic investigations for neuroendocrine tumors [see Warnings and Precautions (5.12), Clinical Pharmacology (12.2)]. |
| Intervention: | Temporarily stop pantoprazole sodium for injection treatment at least 14 days before assessing CgA levels and consider repeating the test if initial CgA levels are high. If serial tests are performed (e.g. for monitoring), the same commercial laboratory should be used for testing, as reference ranges between tests may vary. |
| False Positive Urine Tests for THC | |
| Clinical Impact: | There have been reports of false positive urine screening tests for tetrahydrocannabinol (THC) in patients receiving PPIs [see Warnings and Precautions (5.13)]. |
| Intervention: | An alternative confirmatory method should be considered to verify positive results. |
12.3 Pharmacokinetics
Pantoprazole peak serum concentration (Cmax) and area under the serum concentration-time curve (AUC) increase in a manner proportional to intravenous doses from 10 mg to 80 mg. Pantoprazole does not accumulate and its pharmacokinetics are unaltered with multiple daily dosing. Following the administration of pantoprazole sodium for injection, the serum concentration of pantoprazole declines biexponentially with a terminal elimination half-life of approximately one hour. In CYP2C19 extensive metabolizers [see Clinical Pharmacology (12.5)] with normal liver function receiving a 40 mg dose of pantoprazole sodium for injection by constant rate over 15 minutes, the peak concentration (Cmax) is 5.52 ±1.42 mcg/mL and the total area under the plasma concentration versus time curve (AUC) is 5.4 ±1.5 mcg hr/mL. The total clearance is 7.6 to 14 L/h.
12.5 Pharmacogenomics
CYP2C19 displays a known genetic polymorphism due to its deficiency in some subpopulations (e.g., approximately 3% of Caucasians and African-Americans and 17% to 23% of Asians are poor metabolizers). Although these subpopulations of pantoprazole poor metabolizers have elimination half-life values of 3.5 to 10.0 hours in adults, they still have minimal accumulation (23% or less) with once-daily dosing. For adult patients who are CYP2C19 poor metabolizers, no dosage adjustment is needed.
Similar to adults, pediatric patients who have the poor metabolizer genotype of CYP2C19 (CYP2C19 *2/*2) exhibited greater than a 6-fold increase in AUC compared to pediatric extensive (CYP2C19 *1/*1) and intermediate (CYP2C19 *1/*x) metabolizers. Poor metabolizers exhibited approximately 10-fold lower apparent oral clearance compared to extensive metabolizers.
1 Indications and Usage
Pantoprazole sodium is a proton pump inhibitor (PPI) indicated in adults for the following:
12.1 Mechanism of Action
Pantoprazole is a PPI that suppresses the final step in gastric acid production by covalently binding to the (H+, K+)-ATPase enzyme system at the secretory surface of the gastric parietal cell. This effect leads to inhibition of both basal and stimulated gastric acid secretion irrespective of the stimulus. The binding to the (H+, K+)-ATPase results in a duration of antisecretory effect that persists longer than 24 hours for all doses tested (20 mg to 120 mg).
5.11 Fundic Gland Polyps
PPI use is associated with an increased risk of fundic gland polyps that increases with long-term use, especially beyond one year. Most PPI users who developed fundic gland polyps were asymptomatic and fundic gland polyps were identified incidentally on endoscopy. Use the shortest duration of PPI therapy appropriate to the condition being treated.
5.5 Clostridium Difficile
Published observational studies suggest that PPI therapy like pantoprazole sodium for injection may be associated with an increased risk of Clostridium difficile associated diarrhea, especially in hospitalized patients. This diagnosis should be considered for diarrhea that does not improve [see Adverse Reactions (6.2)].
Patients should use the lowest dose and shortest duration of PPI therapy appropriate to the condition being treated.
5 Warnings and Precautions
-
Gastric Malignancy: In adults, symptomatic response to therapy with pantoprazole does not preclude the presence of gastric malignancy.
Consider additional follow-up and diagnostic testing. (5.1) - Injection Site Reactions: Thrombophlebitis is associated with the administration of intravenous pantoprazole. (5.2)
- Potential Exacerbation of Zinc Deficiency: Consider zinc supplementation in patients who are prone to zinc deficiency. Caution should be used when other EDTA containing products are also co-administered intravenously. (5.3)
- Acute Tubulointerstitial Nephritis: Discontinue treatment and evaluate patients. (5.4)
- Clostridium difficile -Associated Diarrhea: PPI therapy may be associated with increased risk. (5.5)
- Bone Fracture: Long term and multiple daily dose PPI therapy may be associated with an increased risk for osteoporosis-related fractures of the hip, wrist or spine. (5.6)
- Severe Cutaneous Adverse Reactions: Discontinue at the first signs or symptoms of severe cutaneous adverse reactions or other signs of hypersensitivity and consider further evaluation. (5.7)
- Cutaneous and Systemic Lupus Erythematosus: Mostly cutaneous; new onset or exacerbation of existing disease; discontinue pantoprazole sodium for injection and refer to specialist for evaluation. (5.8)
- Hepatic Effects: Elevations of transaminases observed. (5.9)
- Hypomagnesemia and Mineral Metabolism: Reported rarely with prolonged treatment with PPIs. (5.10)
- Fundic Gland Polyps: Risk increases with long-term use, especially beyond one year. Use the shortest duration of therapy. (5.11)
2 Dosage and Administration
GERD Associated with EE (2.1)
- The recommended adult dosage is 40 mg given once daily by intravenous infusion for 7 to 10 days. (2.1)
Pathological Hypersecretion Conditions, Including ZE Syndrome (2.3):
- The recommended adult dosage is 80 mg administered every 12 hours by intravenous infusion. For information on how to adjust dosing for individual patient needs, see the full prescribing information.
- Only for intravenous infusion.
- The intravenous infusion can be administered over 2 minutes or 15 minutes.
- For information on how to prepare and administer for each indication, see the full prescribing information.
3 Dosage Forms and Strengths
For Injection: 40 mg of pantoprazole white to off-white freeze-dried powder in a single-dose vial for reconstitution.
5.2 Injection Site Reactions
Thrombophlebitis was associated with the administration of pantoprazole sodium for injection.
6.2 Postmarketing Experience
The following adverse reactions have been identified during postapproval use of pantoprazole and pantoprazole sodium for injection. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
These adverse reactions are listed below by body system:
General Disorders and Administration Conditions: asthenia, fatigue, malaise
Immune System Disorders: anaphylaxis (including anaphylactic shock), systemic lupus erythematosus
Investigations: weight changes
Skin and Subcutaneous Tissue Disorders: severe dermatologic reactions (some fatal), including erythema multiforme, SJS/TEN, DRESS, AGEP, angioedema (Quincke's edema) and cutaneous lupus erythematosus
Musculoskeletal Disorders: rhabdomyolysis, bone fracture
Renal and Genitourinary Disorders: acute tubulointerstitial nephritis, erectile dysfunction
Hepatobiliary Disorders: hepatocellular damage leading to jaundice and hepatic failure
Psychiatric Disorder: hallucinations, confusion, insomnia, somnolence
Metabolism and Nutritional Disorders: hyponatremia, hypomagnesemia, hypocalcemia, hypokalemia, hyponatremia
Infections and Infestations: Clostridium difficile associated diarrhea
Hematologic: pancytopenia, agranulocytosis
Nervous: ageusia, dysgeusia
Gastrointestinal Disorders: fundic gland polyps
2.5 Compatibility Information
- Administer pantoprazole sodium for injection intravenously through a dedicated line or through a Y-site.
- Flush the intravenous line before and after administration of pantoprazole sodium for injection with either 5% Dextrose Injection, USP, 0.9% Sodium Chloride Injection, USP, or Lactated Ringer's Injection, USP.
- When administered through a Y-site, pantoprazole sodium for injection is compatible with the following solutions: 5% Dextrose Injection, USP, 0.9% Sodium Chloride Injection, USP, or Lactated Ringer's Injection, USP.
- Midazolam HCl has been shown to be incompatible with Y-site administration of pantoprazole sodium for injection
- Pantoprazole sodium for injection may not be compatible with products containing zinc [see Warnings and Precautions (5.3)].
- When pantoprazole sodium for injection is administered through a Y-site, immediately stop use if precipitation or discoloration occurs.
8 Use in Specific Populations
Pregnancy: Based on animal data, may cause fetal harm. (8.1)
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
Worldwide, approximately 80,500 patients have been treated with pantoprazole in clinical trials involving various dosages and duration of treatment.
5.1 Presence of Gastric Malignancy
In adults, symptomatic response to therapy with pantoprazole sodium for injection does not preclude the presence of gastric malignancy. Consider additional follow-up and diagnostic testing in adult patients who have a suboptimal response or an early symptomatic relapse after completing treatment with a PPI. In older patients, also consider an endoscopy.
16 How Supplied/storage and Handling
Pantoprazole Sodium for Injection is available as follows:
| NDC | Pantoprazole Sodium for Injection | Package Factor |
| 25021-751-10 | 40 mg Single-Dose Vial | 10 vials per carton |
| 25021-751-11 | 40 mg Single-Dose Vial | 25 vials per carton |
Pantoprazole Sodium for Injection is a white to off-white freeze-dried powder for reconstitution.
5.4 Acute Tubulointerstitial Nephritis
Acute tubulointerstitial nephritis (TIN) has been observed in patients taking PPIs and may occur at any point during PPI therapy. Patients may present with varying signs and symptoms from symptomatic hypersensitivity reactions to non-specific symptoms of decreased renal function (e.g., malaise, nausea, anorexia). In reported case series, some patients were diagnosed on biopsy and in the absence of extra-renal manifestations (e.g., fever, rash or arthralgia). Discontinue pantoprazole sodium for injection and evaluate patients with suspected acute TIN [see Contraindications (4)].
5.7 Severe Cutaneous Adverse Reactions
Severe cutaneous adverse reactions, including erythema multiforme, Stevens-Johnson syndrome (SJS), toxic epidermal necrolysis (TEN), drug reaction with eosinophilia and systemic symptoms (DRESS), and acute generalized exanthematous pustulosis (AGEP) have been reported in association with the use of PPIs [see Adverse Reactions (6.2)]. Discontinue pantoprazole sodium for injection at the first signs or symptoms of severe cutaneous adverse reactions or other signs of hypersensitivity and consider further evaluation.
5.10 Hypomagnesemia and Mineral Metabolism
Hypomagnesemia, symptomatic and asymptomatic, has been reported rarely in patients treated with PPIs for at least three months, and in most cases after a year of therapy. Serious adverse events include tetany, arrhythmias, and seizures. Hypomagnesemia may lead to hypocalcemia and/or hypokalemia and may exacerbate underlying hypocalcemia in at-risk patients. In most patients, treatment of hypomagnesemia required magnesium replacement and discontinuation of the PPI.
For patients expected to be on prolonged treatment or who take PPIs with medications such as digoxin or drugs that may cause hypomagnesemia (e.g., diuretics), health care professionals may consider monitoring magnesium levels prior to initiation of PPI treatment and periodically [see Adverse Reactions (6.2)].
Consider monitoring magnesium and calcium levels prior to initiation of pantoprazole sodium for injection and periodically while on treatment in patients with a preexisting risk of hypocalcemia (e.g., hypoparathyroidism). Supplement with magnesium and/or calcium as necessary. If hypocalcemia is refractory to treatment, consider discontinuing the PPI.
5.13 Interference With Urine Screen for Thc
Pantoprazole sodium may produce false-positive urine screen for THC (tetrahydrocannabinol) [see Drug Interactions (7)].
5.8 Cutaneous and Systemic Lupus Erythematosus
Cutaneous lupus erythematosus (CLE) and systemic lupus erythematosus (SLE) have been reported in patients taking PPIs, including pantoprazole sodium. These events have occurred as both new onset and an exacerbation of existing autoimmune disease. The majority of PPI-induced lupus erythematous cases were CLE.
The most common form of CLE reported in patients treated with PPIs was subacute CLE (SCLE) and occurred within weeks to years after continuous drug therapy in patients ranging from infants to the elderly. Generally, histological findings were observed without organ involvement.
Systemic lupus erythematosus (SLE) is less commonly reported than CLE in patients receiving PPIs. PPI associated SLE is usually milder than non-drug induced SLE. Onset of SLE typically occurred within days to years after initiating treatment primarily in patients ranging from young adults to the elderly. The majority of patients presented with rash; however, arthralgia and cytopenia were also reported.
Avoid administration of PPIs for longer than medically indicated. If signs or symptoms consistent with CLE or SLE are noted in patients receiving pantoprazole sodium for injection, discontinue the drug and refer the patient to the appropriate specialist for evaluation. Most patients improve with discontinuation of the PPI alone in 4 to 12 weeks. Serological testing (e.g., ANA) may be positive and elevated serological test results may take longer to resolve than clinical manifestations.
5.3 Potential for Exacerbation of Zinc Deficiency
Pantoprazole sodium for injection contains edetate disodium (the salt form of EDTA), a chelator of metal ions including zinc. Therefore, zinc supplementation should be considered in patients treated with pantoprazole sodium for injection who are prone to zinc deficiency. Caution should be used when other EDTA containing products are also co-administered intravenously [see Dosage and Administration (2.5)].
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
In a 24-month carcinogenicity study, Sprague-Dawley rats were treated orally with pantoprazole doses of 0.5 to 200 mg/kg/day, about 0.1 to 40 times the exposure on a body surface area basis of a 50-kg person dosed at 40 mg/day. In the gastric fundus, treatment with 0.5 to 200 mg/kg/day produced enterochromaffin-like (ECL) cell hyperplasia and benign and malignant neuroendocrine cell tumors in a dose-related manner. In the forestomach, treatment with 50 and 200 mg/kg/day (about 10 and 40 times the recommended human dose on a body surface area basis) produced benign squamous cell papillomas and malignant squamous cell carcinomas. Rare gastrointestinal tumors associated with pantoprazole treatment included an adenocarcinoma of the duodenum with 50 mg/kg/day and benign polyps and adenocarcinomas of the gastric fundus with 200 mg/kg/day. In the liver, treatment with 0.5 to 200 mg/kg/day produced dose-related increases in the incidences of hepatocellular adenomas and carcinomas. In the thyroid gland, treatment with 200 mg/kg/day produced increased incidences of follicular cell adenomas and carcinomas for both male and female rats.
In a 24-month carcinogenicity study, Fischer 344 rats were treated orally with pantoprazole doses of 5 to 50 mg/kg/day, approximately 1 to 10 times the recommended human dose based on body surface area. In the gastric fundus, treatment with 5 to 50 mg/kg/day produced enterochromaffin-like (ECL) cell hyperplasia and benign and malignant neuroendocrine cell tumors. Dose selection for this study may not have been adequate to comprehensively evaluate the carcinogenic potential of pantoprazole.
In a 24-month carcinogenicity study, B6C3F1 mice were treated orally with pantoprazole doses of 5 to 150 mg/kg/day, 0.5 to 15 times the recommended human dose based on body surface area. In the liver, treatment with 150 mg/kg/day produced increased incidences of hepatocellular adenomas and carcinomas in female mice. Treatment with 5 to 150 mg/kg/day also produced gastric fundic ECL cell hyperplasia.
A 26-week p53 +/- transgenic mouse carcinogenicity study was not positive.
Pantoprazole was positive in the in vitro human lymphocyte chromosomal aberration assays, in one of two mouse micronucleus tests for clastogenic effects, and in the in vitro Chinese hamster ovarian cell/HGPRT forward mutation assay for mutagenic effects. Equivocal results were observed in the in vivo rat liver DNA covalent binding assay. Pantoprazole was negative in the in vitro Ames mutation assay, the in vitro unscheduled DNA synthesis (UDS) assay with rat hepatocytes, the in vitro AS52/GPT mammalian cell-forward gene mutation assay, the in vitro thymidine kinase mutation test with mouse lymphoma L5178Y cells, and the in vivo rat bone marrow cell chromosomal aberration assay.
There were no effects on fertility or reproductive performance when pantoprazole was given at oral doses up to 500 mg/kg/day in male rats (98 times the recommended human dose based on body surface area) and 450 mg/kg/day in female rats (88 times the recommended human dose based on body surface area).
5.12 Interference With Investigations for Neuroendocrine Tumors
Serum chromogranin A (CgA) levels increase secondary to drug-induced decreases in gastric acidity. The increased CgA level may cause false positive results in diagnostic investigations for neuroendocrine tumors. Healthcare providers should temporarily stop pantoprazole sodium for injection treatment at least 14 days before assessing CgA levels and consider repeating the test if initial CgA levels are high. If serial tests are performed (e.g. for monitoring), the same commercial laboratory should be used for testing, as reference ranges between tests may vary [see Clinical Pharmacology (12.2)].
1.2 Pathological Hypersecretion Including Zollinger Ellison Syndrome
Pantoprazole sodium for injection is indicated for the treatment of pathological hypersecretory conditions including Zollinger-Ellison (ZE) Syndrome in adults.
14.2 Pathological Hypersecretion Associated With Zollinger Ellison Syndrome
Two studies measured the pharmacodynamic effects of 6 day treatment with pantoprazole sodium for injection in patients with ZE Syndrome (with and without multiple endocrine neoplasia type I). In one of these studies, an initial treatment with pantoprazole sodium for injection in 21 patients (29 to 75 years; 8 female; 4 Black, 1 Hispanic, 16 White) reduced acid output to the target level (10 mEq/h or less) and significantly reduced H+ concentration and the volume of gastric secretions; target levels were achieved within 45 minutes of drug administration.
In the other study of 14 patients (38 to 67 years; 5 female; 2 Black, 12 White) with ZE Syndrome, treatment was switched from an oral PPI to pantoprazole sodium for injection. Pantoprazole sodium for injection maintained or improved control of gastric acid secretion.
In both studies, total doses of 160 or 240 mg per day of pantoprazole sodium for injection administered in divided doses, maintained basal acid secretion below target levels in all patients. Target levels were 10 mEq/h in patients without prior gastric surgery, and 5 mEq/h in all patients with prior gastric acid-reducing surgery. Once gastric acid secretion was controlled, there was no evidence of tolerance during this 7 day study. Basal acid secretion was maintained below target levels for at least 24 hours in all patients and through the end of treatment in these studies (3 to 7 days) in all but 1 patient who required a dose adjustment guided by acid output measurements until acid control was achieved. In both studies, doses were adjusted to the individual patient need, but gastric acid secretion was controlled in greater than 80% of patients by a starting regimen of 80 mg every 12 hours.
5.14 Concomitant Use of Pantoprazole Sodium for Injection With Methotrexate
Literature suggests that concomitant use of PPIs with methotrexate (primarily at high dose; see methotrexate prescribing information) may elevate and prolong serum levels of methotrexate and/or its metabolite, possibly leading to methotrexate toxicities. In high-dose methotrexate administration, a temporary withdrawal of the PPI may be considered in some patients [see Drug Interactions (7)].
2.3 Dosage for Pathological Hypersecretion Including Zollinger Ellison Syndrome
The recommended adult dosage of pantoprazole sodium for injection is 80 mg intravenously every 12 hours. The frequency of dosing can be adjusted to individual patient needs based on acid output measurements. In those patients who need a higher dosage, 80 mg intravenously every 8 hours is expected to maintain acid output below 10 mEq/h. Daily doses higher than 240 mg or administered for more than 6 days have not been studied [see Clinical Studies (14)]. Transition from oral to intravenous and from intravenous to oral formulations of gastric acid inhibitors should be performed in such a manner to ensure continuity of effect of suppression of acid secretion. Patients with ZE Syndrome may be vulnerable to serious clinical complications of increased acid production even after a short period of loss of effective inhibition.
1.1 Gastroesophageal Reflux Disease Associated With A History of Erosive Esophagitis
Pantoprazole sodium for injection is indicated for short-term treatment (7 to 10 days) of adult patients with gastroesophageal reflux disease (GERD) and a history of erosive esophagitis (EE).
Safety and efficacy of pantoprazole sodium for injection as a treatment of patients with GERD and a history of EE for more than 10 days have not been demonstrated.
14.1 Gastroesophageal Reflux Disease (gerd) Associated With A History of Erosive Esophagitis
A multicenter, double-blind, two-period placebo-controlled study was conducted to assess the ability of pantoprazole sodium for injection to maintain gastric acid suppression in patients switched from Pantoprazole Delayed-Release Tablets to pantoprazole sodium for injection GERD patients (n=65, 26 to 64 years; 35 female; 9 Black, 11 Hispanic, 44 White, 1 other) with a history of EE were randomized to receive either 20 or 40 mg of oral pantoprazole once per day for 10 days (period 1), and then were switched in period 2 to either daily pantoprazole sodium for injection or placebo for 7 days, matching their respective dose level from period 1. Patients were administered all test medication with a light meal. Maximum acid output (MAO) and basal acid output (BAO) were determined 24 hours following the last day of oral medication (day 10), the first day (day 1) of intravenous administration and the last day of intravenous administration (day 7). MAO was estimated from a 1 hour continuous collection of gastric contents following subcutaneous injection of 6.0 mcg/kg of pentagastrin.
This study demonstrated that, after 10 days of repeated oral administration followed by 7 days of intravenous administration, the oral and intravenous dosage forms of pantoprazole 40 mg are similar in their ability to suppress MAO and BAO in patients with GERD and a history of EE (see Table 4). Also, patients on oral pantoprazole who were switched to intravenous placebo experienced a significant increase in acid output within 48 hours of their last oral dose (see Table 4). However, at 48 hours after their last oral dose, patients treated with pantoprazole sodium for injection had a significantly lower mean basal acid output (see Table 4) than those treated with placebo.
|
* p<0.0001 Significantly different from pantoprazole sodium for injection. |
|||
| Parameter |
Pantoprazole Delayed-Release Tablets DAY 10 |
Pantoprazole Sodium for Injection DAY 7 |
Intravenous Placebo DAY 7 |
| Mean maximum acid output |
6.49 n=30 |
6.62 n=23 |
29.19* n=7 |
| Mean basal acid output |
0.80 n=30 |
0.53 n=23 |
4.14* n=7 |
To evaluate the effectiveness of pantoprazole sodium for injection as an initial treatment to suppress gastric acid secretion, two studies were conducted.
Study 1 was a multicenter, double-blind, placebo-controlled, study of the pharmacodynamic effects of pantoprazole sodium for injection and Pantoprazole Delayed-Release Tablets. Patients with GERD and a history of EE (n=78, 20 to 67 years; 39 females; 7 Black, 19 Hispanic, 52 White) were randomized to receive either 40 mg pantoprazole sodium for injection, 40 mg pantoprazole Delayed-Release Tablets, or placebo once daily for 7 days. Following an overnight fast, test medication was administered and patients were given a light meal within 15 minutes. MAO and BAO were determined 24 hours following the last day of study medication. MAO was estimated from a 1 hour continuous collection of gastric contents following subcutaneous injection of 6.0 mcg/kg of pentagastrin to stimulate acid secretion. This study demonstrated that, after treatment for 7 days, patients treated with pantoprazole sodium for injection had a significantly lower MAO and BAO than those treated with placebo (p<0.001), and results were comparable to those of patients treated with Pantoprazole Delayed-Release Tablets (see Table 5).
|
* p<0.001 Significantly different from pantoprazole sodium for injection. |
|||
| Parameter |
Pantoprazole Sodium for Injection DAY 7 |
Pantoprazole Delayed-Release Tablets DAY 7 |
Placebo DAY 7 |
| Maximum acid output (mean ± SD) |
8.4 ± 5.9 n=25 |
6.3 ± 6.6 n=22 |
20.9 ± 14.5* n=24 |
| Basal acid output (mean ± SD) |
0.4 ± 0.5 n=25 |
0.6 ± 0.8 n=22 |
2.8 ± 3.0* n=23 |
Study 2 was a single-center, double-blind, parallel-group study to compare the clinical effects of pantoprazole sodium for injection and Pantoprazole Delayed-Release Tablets. Patients (n=45, median age 56 years, 21 males and 24 females) with acute endoscopically proven reflux esophagitis (Savary/Miller Stage II or III) with at least 1 of 3 symptoms typical for reflux esophagitis (acid eructation, heartburn, or pain on swallowing) were randomized to receive either 40 mg pantoprazole sodium for injection or 40 mg Pantoprazole Delayed-Release Tablets once daily for 5 days. After the initial 5 days, all patients were treated with 40 mg oral pantoprazole daily to complete a total of 8 weeks of treatment. Symptom relief was assessed by calculating the daily mean of the sums of the average scores for these 3 symptoms and the daily mean of the average score for each of the symptoms separately. There was no significant difference in symptom relief between pantoprazole sodium for injection and Pantoprazole Delayed-Release Tablets within the first 5 days. A repeat endoscopy after 8 weeks of treatment revealed that 20 out of 23 (87%) patients treated with pantoprazole sodium for injection plus Pantoprazole Delayed-Release Tablets and 19 out of 22 (86%) of the patients treated with Pantoprazole Delayed-Release Tablets had endoscopically proven healing of their esophageal lesions.
Data comparing pantoprazole sodium for injection to other PPIs (oral or intravenous) or H2-receptor antagonists (oral or intravenous) are limited, and therefore, are inadequate to support any conclusions regarding comparative efficacy.
2.1 Dosage for Gastroesophageal Reflux Disease Associated With A History of Erosive Esophagitis
The recommended adult dosage of pantoprazole sodium for injection is 40 mg given once daily by intravenous infusion for 7 to 10 days.
Discontinue treatment with pantoprazole sodium for injection as soon as the patient is able to receive treatment with Pantoprazole Delayed-Release Tablets or Oral Suspension.
Data on the safe and effective dosing for conditions other than those described [see Indications and Usage (1)] such as life-threatening upper gastrointestinal bleeds, are not available. Pantoprazole sodium for injection 40 mg once daily does not raise gastric pH to levels sufficient to contribute to the treatment of such life-threatening conditions.
2.4 Preparation and Administration Instructions for Pathological Hypersecretion Including Zollinger Ellison Syndrome
Only for intravenous infusion; other parenteral routes of administration are not recommended.
Fifteen Minute Infusion
- Reconstitute each vial of pantoprazole sodium for injection with 10 mL of 0.9% Sodium Chloride Injection, USP.
- Combine the contents of the two vials and further dilute with 80 mL of 5% Dextrose Injection, USP, 0.9% Sodium Chloride Injection, USP, or Lactated Ringer's Injection, USP, to a total volume of 100 mL with a final concentration of approximately 0.8 mg per mL.
- Inspect the diluted pantoprazole sodium for injection solution visually for particular matter and discoloration prior to and during administration.
- Administer intravenously over a period of approximately 15 minutes at a rate of approximately 7 mL/min.
2.2 Preparation and Administration Instructions for Gastroesophageal Reflux Disease Associated With A History of Erosive Esophagitis
Only for intravenous infusion; other parenteral routes of administration are not recommended.
Structured Label Content
Section 42229-5 (42229-5)
Fifteen Minute Infusion
- Reconstitute pantoprazole sodium for injection with 10 mL of 0.9% Sodium Chloride Injection, USP.
- Further dilute with 100 mL of 5% Dextrose Injection, USP, 0.9% Sodium Chloride Injection, USP, or Lactated Ringer's Injection, USP, to a final concentration of approximately 0.4 mg per mL.
- Inspect the diluted pantoprazole sodium for injection solution visually for particular matter and discoloration prior to and during administration.
- Administer intravenously over a period of approximately 15 minutes at a rate of approximately 7 mL/min.
Section 44425-7 (44425-7)
Storage Conditions
Store at 20° to 25°C (68° to 77°F); excursions permitted to 15° to 30°C (59° to 86°F). [See USP Controlled Room Temperature.]
Protect from light. Retain in carton until time of use.
Discard unused portion.
Sterile, Nonpyrogenic, Preservative-free.
The container closure is not made with natural rubber latex.
Section 51945-4 (51945-4)
PACKAGE LABEL – PRINCIPAL DISPLAY PANEL – Vial Label
NDC 25021-751-10
Rx only
Pantoprazole Sodium for Injection
40 mg per vial
For Intravenous Infusion Only
Single-Dose Vial
10 Overdosage (10 OVERDOSAGE)
Experience in patients taking very high doses of pantoprazole (greater than 240 mg) is limited. Adverse reactions seen in spontaneous reports of overdose generally reflect the known safety profile of pantoprazole.
Pantoprazole is not removed by hemodialysis. In case of overdose, treatment should be symptomatic and supportive.
Single intravenous doses of pantoprazole at 378, 230, and 266 mg/kg (38, 46, and 177 times the recommended human dose based on body surface area) were lethal to mice, rats and dogs, respectively. The symptoms of acute toxicity were hypoactivity, ataxia, hunched sitting, limb-splay, lateral position, segregation, absence of ear reflex, and tremor.
11 Description (11 DESCRIPTION)
The active ingredient in pantoprazole sodium for injection, a PPI, is a substituted benzimidazole, sodium 5-(difluoromethoxy)-2-[[(3,4-dimethoxy-2-pyridinyl)methyl] sulfinyl]-1H-benzimidazole, a compound that inhibits gastric acid secretion. Its empirical formula is C16H14F2N3NaO4S, with a molecular weight of 405.4. The structural formula is:
Pantoprazole sodium USP is a white to off-white crystalline powder and is racemic. Pantoprazole has weakly basic and acidic properties. Pantoprazole sodium, USP is freely soluble in water, very slightly soluble in phosphate buffer at pH 7.4, and practically insoluble in n-hexane. The stability of the compound in aqueous solution is pH-dependent. The rate of degradation increases with decreasing pH. The reconstituted solution of pantoprazole sodium for injection is in the pH range 9.0 to 10.5.
Pantoprazole sodium for injection is supplied for intravenous administration as a sterile, freeze-dried powder in a single-dose clear glass vial fitted with a rubber stopper and crimp seal. Each vial contains 40 mg pantoprazole (equivalent to 45.1 mg of pantoprazole sodium), edetate disodium (1 mg), and sodium hydroxide to adjust pH.
5.6 Bone Fracture
Several published observational studies suggest that PPI therapy may be associated with an increased risk for osteoporosis-related fractures of the hip, wrist, or spine. The risk of fracture was increased in patients who received high-dose, defined as multiple daily doses, and long-term PPI therapy (a year or longer). Patients should use the lowest dose and shortest duration of PPI therapy appropriate to the condition being treated. Patients at risk for osteoporosis-related fractures should be managed according to established treatment guidelines [see Dosage and Administration (2.2, 2.4), Adverse Reactions (6)].
8.4 Pediatric Use
The safety and effectiveness of pantoprazole sodium for injection have not been established in pediatric patients.
8.5 Geriatric Use
Of 286 patients in clinical studies of intravenous pantoprazole sodium in patients with GERD and a history of EE, 86 (43%) were 65 years of age and over. No overall differences in safety or effectiveness were observed between these subjects and younger subjects, and other reported clinical experience with oral pantoprazole sodium has not identified differences in responses between the elderly and younger patients, but greater sensitivity of some older individuals cannot be ruled out.
4 Contraindications (4 CONTRAINDICATIONS)
- Pantoprazole sodium for injection is contraindicated in patients with known hypersensitivity reactions including anaphylaxis to the formulation or any substituted benzimidazole. Hypersensitivity reactions may include anaphylaxis, anaphylactic shock, angioedema, bronchospasm, acute tubulointerstitial nephritis, and urticaria [see Warnings and Precautions (5.2, 5.4), Adverse Reactions (6)].
- Proton pump inhibitors (PPIs), including pantoprazole sodium for injection, are contraindicated in patients receiving rilpivirine-containing products [see Drug Interactions (7)].
5.9 Hepatic Effects
Mild, transient transaminase elevations have been observed in clinical studies. The clinical significance of this finding in a large population of subjects administered pantoprazole sodium for injection is unknown [see Adverse Reactions (6)].
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following serious adverse reactions are described below and elsewhere in labeling:
- Injection Site Reactions [see Warnings and Precautions (5.2)]
- Potential for Exacerbation of Zinc Deficiency [see Warnings and Precautions (5.3)]
- Acute Tubulointerstitial Nephritis [see Warnings and Precautions (5.4)]
- Clostridium difficile-Associated Diarrhea [see Warnings and Precautions (5.5)]
- Bone Fracture [see Warnings and Precautions (5.6)]
- Severe Cutaneous Adverse Reactions [see Warnings and Precautions (5.7)]
- Cutaneous and Systemic Lupus Erythematosus [see Warnings and Precautions (5.8)]
- Hepatic Effects [see Warnings and Precautions (5.9)]
- Hypomagnesemia and Mineral Metabolism [see Warnings and Precautions (5.10)]
- Fundic Gland Polyps [see Warnings and Precautions (5.11)]
7 Drug Interactions (7 DRUG INTERACTIONS)
Table 2 includes drugs with clinically important drug interactions and interaction with diagnostics when administered concomitantly with pantoprazole sodium for injection and instructions for preventing or managing them.
Consult the labeling of concomitantly used drugs to obtain further information about interactions with PPIs.
| Antiretrovirals | |
| Clinical Impact: | The effect of PPIs on antiretroviral drugs is variable. The clinical importance and the mechanisms behind these interactions are not always known.
|
| Intervention: |
Rilpivirine-containing products: Concomitant use with pantoprazole sodium for injection is contraindicated [see Contraindications (4)]. See prescribing information. Atazanavir: See prescribing information for atazanavir for dosing information. Nelfinavir: Avoid concomitant use with pantoprazole sodium for injection See prescribing information for nelfinavir. Saquinavir: See the prescribing information for saquinavir and monitor for potential saquinavir toxicities. Other antiretrovirals: See prescribing information. |
| Warfarin | |
| Clinical Impact: | Increased INR and prothrombin time in patients receiving PPIs, including pantoprazole, and warfarin concomitantly. Increases in INR and prothrombin time may lead to abnormal bleeding and even death. |
| Intervention: | Monitor INR and prothrombin time. Dose adjustment of warfarin may be needed to maintain target INR range. See prescribing information for warfarin. |
| Clopidogrel | |
| Clinical Impact: | Concomitant administration of pantoprazole and clopidogrel in healthy subjects had no clinically important effect on exposure to the active metabolite of clopidogrel or clopidogrel-induced platelet inhibition [see Clinical Pharmacology (12.3)]. |
| Intervention: | No dose adjustment of clopidogrel is necessary when administered with an approved dose of pantoprazole sodium for injection. |
| Methotrexate | |
| Clinical Impact: | Concomitant use of PPIs with methotrexate (primarily at high dose) may elevate and prolong serum concentrations of methotrexate and/or its metabolite hydroxymethotrexate, possibly leading to methotrexate toxicities. No formal drug interaction studies of high-dose methotrexate with PPIs have been conducted [see Warnings and Precautions (5.14)]. |
| Intervention: | A temporary withdrawal of pantoprazole sodium for injection may be considered in some patients receiving high-dose methotrexate. |
| Drugs Dependent on Gastric pH for Absorption (e.g., iron salts, erlotinib, dasatinib, nilotinib, mycophenolate mofetil, ketoconazole/itraconazole) | |
| Clinical Impact: | Pantoprazole can reduce the absorption of other drugs due to its effect on reducing intragastric acidity. |
| Intervention: | Mycophenolate mofetil (MMF): Co-administration of pantoprazole sodium in healthy subjects and in transplant patients receiving MMF has been reported to reduce the exposure to the active metabolite, mycophenolic acid (MPA), possibly due to a decrease in MMF solubility at an increased gastric pH [see Clinical Pharmacology (12.3)]. The clinical relevance of reduced MPA exposure on organ rejection has not been established in transplant patients receiving pantoprazole sodium for injection and MMF. Use pantoprazole sodium for injection with caution in transplant patients receiving MMF. See the prescribing information for other drugs dependent on gastric pH for absorption. |
| Interactions with Investigations of Neuroendocrine Tumors | |
| Clinical Impact: | CgA levels increase secondary to PPI-induced decreases in gastric acidity. The increased CgA level may cause false positive results in diagnostic investigations for neuroendocrine tumors [see Warnings and Precautions (5.12), Clinical Pharmacology (12.2)]. |
| Intervention: | Temporarily stop pantoprazole sodium for injection treatment at least 14 days before assessing CgA levels and consider repeating the test if initial CgA levels are high. If serial tests are performed (e.g. for monitoring), the same commercial laboratory should be used for testing, as reference ranges between tests may vary. |
| False Positive Urine Tests for THC | |
| Clinical Impact: | There have been reports of false positive urine screening tests for tetrahydrocannabinol (THC) in patients receiving PPIs [see Warnings and Precautions (5.13)]. |
| Intervention: | An alternative confirmatory method should be considered to verify positive results. |
12.3 Pharmacokinetics
Pantoprazole peak serum concentration (Cmax) and area under the serum concentration-time curve (AUC) increase in a manner proportional to intravenous doses from 10 mg to 80 mg. Pantoprazole does not accumulate and its pharmacokinetics are unaltered with multiple daily dosing. Following the administration of pantoprazole sodium for injection, the serum concentration of pantoprazole declines biexponentially with a terminal elimination half-life of approximately one hour. In CYP2C19 extensive metabolizers [see Clinical Pharmacology (12.5)] with normal liver function receiving a 40 mg dose of pantoprazole sodium for injection by constant rate over 15 minutes, the peak concentration (Cmax) is 5.52 ±1.42 mcg/mL and the total area under the plasma concentration versus time curve (AUC) is 5.4 ±1.5 mcg hr/mL. The total clearance is 7.6 to 14 L/h.
12.5 Pharmacogenomics
CYP2C19 displays a known genetic polymorphism due to its deficiency in some subpopulations (e.g., approximately 3% of Caucasians and African-Americans and 17% to 23% of Asians are poor metabolizers). Although these subpopulations of pantoprazole poor metabolizers have elimination half-life values of 3.5 to 10.0 hours in adults, they still have minimal accumulation (23% or less) with once-daily dosing. For adult patients who are CYP2C19 poor metabolizers, no dosage adjustment is needed.
Similar to adults, pediatric patients who have the poor metabolizer genotype of CYP2C19 (CYP2C19 *2/*2) exhibited greater than a 6-fold increase in AUC compared to pediatric extensive (CYP2C19 *1/*1) and intermediate (CYP2C19 *1/*x) metabolizers. Poor metabolizers exhibited approximately 10-fold lower apparent oral clearance compared to extensive metabolizers.
1 Indications and Usage (1 INDICATIONS AND USAGE)
Pantoprazole sodium is a proton pump inhibitor (PPI) indicated in adults for the following:
12.1 Mechanism of Action
Pantoprazole is a PPI that suppresses the final step in gastric acid production by covalently binding to the (H+, K+)-ATPase enzyme system at the secretory surface of the gastric parietal cell. This effect leads to inhibition of both basal and stimulated gastric acid secretion irrespective of the stimulus. The binding to the (H+, K+)-ATPase results in a duration of antisecretory effect that persists longer than 24 hours for all doses tested (20 mg to 120 mg).
5.11 Fundic Gland Polyps
PPI use is associated with an increased risk of fundic gland polyps that increases with long-term use, especially beyond one year. Most PPI users who developed fundic gland polyps were asymptomatic and fundic gland polyps were identified incidentally on endoscopy. Use the shortest duration of PPI therapy appropriate to the condition being treated.
5.5 Clostridium Difficile (5.5 Clostridium difficile)
Published observational studies suggest that PPI therapy like pantoprazole sodium for injection may be associated with an increased risk of Clostridium difficile associated diarrhea, especially in hospitalized patients. This diagnosis should be considered for diarrhea that does not improve [see Adverse Reactions (6.2)].
Patients should use the lowest dose and shortest duration of PPI therapy appropriate to the condition being treated.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
-
Gastric Malignancy: In adults, symptomatic response to therapy with pantoprazole does not preclude the presence of gastric malignancy.
Consider additional follow-up and diagnostic testing. (5.1) - Injection Site Reactions: Thrombophlebitis is associated with the administration of intravenous pantoprazole. (5.2)
- Potential Exacerbation of Zinc Deficiency: Consider zinc supplementation in patients who are prone to zinc deficiency. Caution should be used when other EDTA containing products are also co-administered intravenously. (5.3)
- Acute Tubulointerstitial Nephritis: Discontinue treatment and evaluate patients. (5.4)
- Clostridium difficile -Associated Diarrhea: PPI therapy may be associated with increased risk. (5.5)
- Bone Fracture: Long term and multiple daily dose PPI therapy may be associated with an increased risk for osteoporosis-related fractures of the hip, wrist or spine. (5.6)
- Severe Cutaneous Adverse Reactions: Discontinue at the first signs or symptoms of severe cutaneous adverse reactions or other signs of hypersensitivity and consider further evaluation. (5.7)
- Cutaneous and Systemic Lupus Erythematosus: Mostly cutaneous; new onset or exacerbation of existing disease; discontinue pantoprazole sodium for injection and refer to specialist for evaluation. (5.8)
- Hepatic Effects: Elevations of transaminases observed. (5.9)
- Hypomagnesemia and Mineral Metabolism: Reported rarely with prolonged treatment with PPIs. (5.10)
- Fundic Gland Polyps: Risk increases with long-term use, especially beyond one year. Use the shortest duration of therapy. (5.11)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
GERD Associated with EE (2.1)
- The recommended adult dosage is 40 mg given once daily by intravenous infusion for 7 to 10 days. (2.1)
Pathological Hypersecretion Conditions, Including ZE Syndrome (2.3):
- The recommended adult dosage is 80 mg administered every 12 hours by intravenous infusion. For information on how to adjust dosing for individual patient needs, see the full prescribing information.
- Only for intravenous infusion.
- The intravenous infusion can be administered over 2 minutes or 15 minutes.
- For information on how to prepare and administer for each indication, see the full prescribing information.
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
For Injection: 40 mg of pantoprazole white to off-white freeze-dried powder in a single-dose vial for reconstitution.
5.2 Injection Site Reactions
Thrombophlebitis was associated with the administration of pantoprazole sodium for injection.
6.2 Postmarketing Experience
The following adverse reactions have been identified during postapproval use of pantoprazole and pantoprazole sodium for injection. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
These adverse reactions are listed below by body system:
General Disorders and Administration Conditions: asthenia, fatigue, malaise
Immune System Disorders: anaphylaxis (including anaphylactic shock), systemic lupus erythematosus
Investigations: weight changes
Skin and Subcutaneous Tissue Disorders: severe dermatologic reactions (some fatal), including erythema multiforme, SJS/TEN, DRESS, AGEP, angioedema (Quincke's edema) and cutaneous lupus erythematosus
Musculoskeletal Disorders: rhabdomyolysis, bone fracture
Renal and Genitourinary Disorders: acute tubulointerstitial nephritis, erectile dysfunction
Hepatobiliary Disorders: hepatocellular damage leading to jaundice and hepatic failure
Psychiatric Disorder: hallucinations, confusion, insomnia, somnolence
Metabolism and Nutritional Disorders: hyponatremia, hypomagnesemia, hypocalcemia, hypokalemia, hyponatremia
Infections and Infestations: Clostridium difficile associated diarrhea
Hematologic: pancytopenia, agranulocytosis
Nervous: ageusia, dysgeusia
Gastrointestinal Disorders: fundic gland polyps
2.5 Compatibility Information
- Administer pantoprazole sodium for injection intravenously through a dedicated line or through a Y-site.
- Flush the intravenous line before and after administration of pantoprazole sodium for injection with either 5% Dextrose Injection, USP, 0.9% Sodium Chloride Injection, USP, or Lactated Ringer's Injection, USP.
- When administered through a Y-site, pantoprazole sodium for injection is compatible with the following solutions: 5% Dextrose Injection, USP, 0.9% Sodium Chloride Injection, USP, or Lactated Ringer's Injection, USP.
- Midazolam HCl has been shown to be incompatible with Y-site administration of pantoprazole sodium for injection
- Pantoprazole sodium for injection may not be compatible with products containing zinc [see Warnings and Precautions (5.3)].
- When pantoprazole sodium for injection is administered through a Y-site, immediately stop use if precipitation or discoloration occurs.
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
Pregnancy: Based on animal data, may cause fetal harm. (8.1)
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
Worldwide, approximately 80,500 patients have been treated with pantoprazole in clinical trials involving various dosages and duration of treatment.
5.1 Presence of Gastric Malignancy
In adults, symptomatic response to therapy with pantoprazole sodium for injection does not preclude the presence of gastric malignancy. Consider additional follow-up and diagnostic testing in adult patients who have a suboptimal response or an early symptomatic relapse after completing treatment with a PPI. In older patients, also consider an endoscopy.
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
Pantoprazole Sodium for Injection is available as follows:
| NDC | Pantoprazole Sodium for Injection | Package Factor |
| 25021-751-10 | 40 mg Single-Dose Vial | 10 vials per carton |
| 25021-751-11 | 40 mg Single-Dose Vial | 25 vials per carton |
Pantoprazole Sodium for Injection is a white to off-white freeze-dried powder for reconstitution.
5.4 Acute Tubulointerstitial Nephritis
Acute tubulointerstitial nephritis (TIN) has been observed in patients taking PPIs and may occur at any point during PPI therapy. Patients may present with varying signs and symptoms from symptomatic hypersensitivity reactions to non-specific symptoms of decreased renal function (e.g., malaise, nausea, anorexia). In reported case series, some patients were diagnosed on biopsy and in the absence of extra-renal manifestations (e.g., fever, rash or arthralgia). Discontinue pantoprazole sodium for injection and evaluate patients with suspected acute TIN [see Contraindications (4)].
5.7 Severe Cutaneous Adverse Reactions
Severe cutaneous adverse reactions, including erythema multiforme, Stevens-Johnson syndrome (SJS), toxic epidermal necrolysis (TEN), drug reaction with eosinophilia and systemic symptoms (DRESS), and acute generalized exanthematous pustulosis (AGEP) have been reported in association with the use of PPIs [see Adverse Reactions (6.2)]. Discontinue pantoprazole sodium for injection at the first signs or symptoms of severe cutaneous adverse reactions or other signs of hypersensitivity and consider further evaluation.
5.10 Hypomagnesemia and Mineral Metabolism
Hypomagnesemia, symptomatic and asymptomatic, has been reported rarely in patients treated with PPIs for at least three months, and in most cases after a year of therapy. Serious adverse events include tetany, arrhythmias, and seizures. Hypomagnesemia may lead to hypocalcemia and/or hypokalemia and may exacerbate underlying hypocalcemia in at-risk patients. In most patients, treatment of hypomagnesemia required magnesium replacement and discontinuation of the PPI.
For patients expected to be on prolonged treatment or who take PPIs with medications such as digoxin or drugs that may cause hypomagnesemia (e.g., diuretics), health care professionals may consider monitoring magnesium levels prior to initiation of PPI treatment and periodically [see Adverse Reactions (6.2)].
Consider monitoring magnesium and calcium levels prior to initiation of pantoprazole sodium for injection and periodically while on treatment in patients with a preexisting risk of hypocalcemia (e.g., hypoparathyroidism). Supplement with magnesium and/or calcium as necessary. If hypocalcemia is refractory to treatment, consider discontinuing the PPI.
5.13 Interference With Urine Screen for Thc (5.13 Interference with Urine Screen for THC)
Pantoprazole sodium may produce false-positive urine screen for THC (tetrahydrocannabinol) [see Drug Interactions (7)].
5.8 Cutaneous and Systemic Lupus Erythematosus
Cutaneous lupus erythematosus (CLE) and systemic lupus erythematosus (SLE) have been reported in patients taking PPIs, including pantoprazole sodium. These events have occurred as both new onset and an exacerbation of existing autoimmune disease. The majority of PPI-induced lupus erythematous cases were CLE.
The most common form of CLE reported in patients treated with PPIs was subacute CLE (SCLE) and occurred within weeks to years after continuous drug therapy in patients ranging from infants to the elderly. Generally, histological findings were observed without organ involvement.
Systemic lupus erythematosus (SLE) is less commonly reported than CLE in patients receiving PPIs. PPI associated SLE is usually milder than non-drug induced SLE. Onset of SLE typically occurred within days to years after initiating treatment primarily in patients ranging from young adults to the elderly. The majority of patients presented with rash; however, arthralgia and cytopenia were also reported.
Avoid administration of PPIs for longer than medically indicated. If signs or symptoms consistent with CLE or SLE are noted in patients receiving pantoprazole sodium for injection, discontinue the drug and refer the patient to the appropriate specialist for evaluation. Most patients improve with discontinuation of the PPI alone in 4 to 12 weeks. Serological testing (e.g., ANA) may be positive and elevated serological test results may take longer to resolve than clinical manifestations.
5.3 Potential for Exacerbation of Zinc Deficiency
Pantoprazole sodium for injection contains edetate disodium (the salt form of EDTA), a chelator of metal ions including zinc. Therefore, zinc supplementation should be considered in patients treated with pantoprazole sodium for injection who are prone to zinc deficiency. Caution should be used when other EDTA containing products are also co-administered intravenously [see Dosage and Administration (2.5)].
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
In a 24-month carcinogenicity study, Sprague-Dawley rats were treated orally with pantoprazole doses of 0.5 to 200 mg/kg/day, about 0.1 to 40 times the exposure on a body surface area basis of a 50-kg person dosed at 40 mg/day. In the gastric fundus, treatment with 0.5 to 200 mg/kg/day produced enterochromaffin-like (ECL) cell hyperplasia and benign and malignant neuroendocrine cell tumors in a dose-related manner. In the forestomach, treatment with 50 and 200 mg/kg/day (about 10 and 40 times the recommended human dose on a body surface area basis) produced benign squamous cell papillomas and malignant squamous cell carcinomas. Rare gastrointestinal tumors associated with pantoprazole treatment included an adenocarcinoma of the duodenum with 50 mg/kg/day and benign polyps and adenocarcinomas of the gastric fundus with 200 mg/kg/day. In the liver, treatment with 0.5 to 200 mg/kg/day produced dose-related increases in the incidences of hepatocellular adenomas and carcinomas. In the thyroid gland, treatment with 200 mg/kg/day produced increased incidences of follicular cell adenomas and carcinomas for both male and female rats.
In a 24-month carcinogenicity study, Fischer 344 rats were treated orally with pantoprazole doses of 5 to 50 mg/kg/day, approximately 1 to 10 times the recommended human dose based on body surface area. In the gastric fundus, treatment with 5 to 50 mg/kg/day produced enterochromaffin-like (ECL) cell hyperplasia and benign and malignant neuroendocrine cell tumors. Dose selection for this study may not have been adequate to comprehensively evaluate the carcinogenic potential of pantoprazole.
In a 24-month carcinogenicity study, B6C3F1 mice were treated orally with pantoprazole doses of 5 to 150 mg/kg/day, 0.5 to 15 times the recommended human dose based on body surface area. In the liver, treatment with 150 mg/kg/day produced increased incidences of hepatocellular adenomas and carcinomas in female mice. Treatment with 5 to 150 mg/kg/day also produced gastric fundic ECL cell hyperplasia.
A 26-week p53 +/- transgenic mouse carcinogenicity study was not positive.
Pantoprazole was positive in the in vitro human lymphocyte chromosomal aberration assays, in one of two mouse micronucleus tests for clastogenic effects, and in the in vitro Chinese hamster ovarian cell/HGPRT forward mutation assay for mutagenic effects. Equivocal results were observed in the in vivo rat liver DNA covalent binding assay. Pantoprazole was negative in the in vitro Ames mutation assay, the in vitro unscheduled DNA synthesis (UDS) assay with rat hepatocytes, the in vitro AS52/GPT mammalian cell-forward gene mutation assay, the in vitro thymidine kinase mutation test with mouse lymphoma L5178Y cells, and the in vivo rat bone marrow cell chromosomal aberration assay.
There were no effects on fertility or reproductive performance when pantoprazole was given at oral doses up to 500 mg/kg/day in male rats (98 times the recommended human dose based on body surface area) and 450 mg/kg/day in female rats (88 times the recommended human dose based on body surface area).
5.12 Interference With Investigations for Neuroendocrine Tumors (5.12 Interference with Investigations for Neuroendocrine Tumors)
Serum chromogranin A (CgA) levels increase secondary to drug-induced decreases in gastric acidity. The increased CgA level may cause false positive results in diagnostic investigations for neuroendocrine tumors. Healthcare providers should temporarily stop pantoprazole sodium for injection treatment at least 14 days before assessing CgA levels and consider repeating the test if initial CgA levels are high. If serial tests are performed (e.g. for monitoring), the same commercial laboratory should be used for testing, as reference ranges between tests may vary [see Clinical Pharmacology (12.2)].
1.2 Pathological Hypersecretion Including Zollinger Ellison Syndrome (1.2 Pathological Hypersecretion Including Zollinger-Ellison Syndrome)
Pantoprazole sodium for injection is indicated for the treatment of pathological hypersecretory conditions including Zollinger-Ellison (ZE) Syndrome in adults.
14.2 Pathological Hypersecretion Associated With Zollinger Ellison Syndrome (14.2 Pathological Hypersecretion Associated with Zollinger-Ellison Syndrome)
Two studies measured the pharmacodynamic effects of 6 day treatment with pantoprazole sodium for injection in patients with ZE Syndrome (with and without multiple endocrine neoplasia type I). In one of these studies, an initial treatment with pantoprazole sodium for injection in 21 patients (29 to 75 years; 8 female; 4 Black, 1 Hispanic, 16 White) reduced acid output to the target level (10 mEq/h or less) and significantly reduced H+ concentration and the volume of gastric secretions; target levels were achieved within 45 minutes of drug administration.
In the other study of 14 patients (38 to 67 years; 5 female; 2 Black, 12 White) with ZE Syndrome, treatment was switched from an oral PPI to pantoprazole sodium for injection. Pantoprazole sodium for injection maintained or improved control of gastric acid secretion.
In both studies, total doses of 160 or 240 mg per day of pantoprazole sodium for injection administered in divided doses, maintained basal acid secretion below target levels in all patients. Target levels were 10 mEq/h in patients without prior gastric surgery, and 5 mEq/h in all patients with prior gastric acid-reducing surgery. Once gastric acid secretion was controlled, there was no evidence of tolerance during this 7 day study. Basal acid secretion was maintained below target levels for at least 24 hours in all patients and through the end of treatment in these studies (3 to 7 days) in all but 1 patient who required a dose adjustment guided by acid output measurements until acid control was achieved. In both studies, doses were adjusted to the individual patient need, but gastric acid secretion was controlled in greater than 80% of patients by a starting regimen of 80 mg every 12 hours.
5.14 Concomitant Use of Pantoprazole Sodium for Injection With Methotrexate (5.14 Concomitant Use of Pantoprazole Sodium for Injection with Methotrexate)
Literature suggests that concomitant use of PPIs with methotrexate (primarily at high dose; see methotrexate prescribing information) may elevate and prolong serum levels of methotrexate and/or its metabolite, possibly leading to methotrexate toxicities. In high-dose methotrexate administration, a temporary withdrawal of the PPI may be considered in some patients [see Drug Interactions (7)].
2.3 Dosage for Pathological Hypersecretion Including Zollinger Ellison Syndrome (2.3 Dosage for Pathological Hypersecretion Including Zollinger-Ellison Syndrome)
The recommended adult dosage of pantoprazole sodium for injection is 80 mg intravenously every 12 hours. The frequency of dosing can be adjusted to individual patient needs based on acid output measurements. In those patients who need a higher dosage, 80 mg intravenously every 8 hours is expected to maintain acid output below 10 mEq/h. Daily doses higher than 240 mg or administered for more than 6 days have not been studied [see Clinical Studies (14)]. Transition from oral to intravenous and from intravenous to oral formulations of gastric acid inhibitors should be performed in such a manner to ensure continuity of effect of suppression of acid secretion. Patients with ZE Syndrome may be vulnerable to serious clinical complications of increased acid production even after a short period of loss of effective inhibition.
1.1 Gastroesophageal Reflux Disease Associated With A History of Erosive Esophagitis (1.1 Gastroesophageal Reflux Disease Associated with a History of Erosive Esophagitis)
Pantoprazole sodium for injection is indicated for short-term treatment (7 to 10 days) of adult patients with gastroesophageal reflux disease (GERD) and a history of erosive esophagitis (EE).
Safety and efficacy of pantoprazole sodium for injection as a treatment of patients with GERD and a history of EE for more than 10 days have not been demonstrated.
14.1 Gastroesophageal Reflux Disease (gerd) Associated With A History of Erosive Esophagitis (14.1 Gastroesophageal Reflux Disease (GERD) Associated with a History of Erosive Esophagitis)
A multicenter, double-blind, two-period placebo-controlled study was conducted to assess the ability of pantoprazole sodium for injection to maintain gastric acid suppression in patients switched from Pantoprazole Delayed-Release Tablets to pantoprazole sodium for injection GERD patients (n=65, 26 to 64 years; 35 female; 9 Black, 11 Hispanic, 44 White, 1 other) with a history of EE were randomized to receive either 20 or 40 mg of oral pantoprazole once per day for 10 days (period 1), and then were switched in period 2 to either daily pantoprazole sodium for injection or placebo for 7 days, matching their respective dose level from period 1. Patients were administered all test medication with a light meal. Maximum acid output (MAO) and basal acid output (BAO) were determined 24 hours following the last day of oral medication (day 10), the first day (day 1) of intravenous administration and the last day of intravenous administration (day 7). MAO was estimated from a 1 hour continuous collection of gastric contents following subcutaneous injection of 6.0 mcg/kg of pentagastrin.
This study demonstrated that, after 10 days of repeated oral administration followed by 7 days of intravenous administration, the oral and intravenous dosage forms of pantoprazole 40 mg are similar in their ability to suppress MAO and BAO in patients with GERD and a history of EE (see Table 4). Also, patients on oral pantoprazole who were switched to intravenous placebo experienced a significant increase in acid output within 48 hours of their last oral dose (see Table 4). However, at 48 hours after their last oral dose, patients treated with pantoprazole sodium for injection had a significantly lower mean basal acid output (see Table 4) than those treated with placebo.
|
* p<0.0001 Significantly different from pantoprazole sodium for injection. |
|||
| Parameter |
Pantoprazole Delayed-Release Tablets DAY 10 |
Pantoprazole Sodium for Injection DAY 7 |
Intravenous Placebo DAY 7 |
| Mean maximum acid output |
6.49 n=30 |
6.62 n=23 |
29.19* n=7 |
| Mean basal acid output |
0.80 n=30 |
0.53 n=23 |
4.14* n=7 |
To evaluate the effectiveness of pantoprazole sodium for injection as an initial treatment to suppress gastric acid secretion, two studies were conducted.
Study 1 was a multicenter, double-blind, placebo-controlled, study of the pharmacodynamic effects of pantoprazole sodium for injection and Pantoprazole Delayed-Release Tablets. Patients with GERD and a history of EE (n=78, 20 to 67 years; 39 females; 7 Black, 19 Hispanic, 52 White) were randomized to receive either 40 mg pantoprazole sodium for injection, 40 mg pantoprazole Delayed-Release Tablets, or placebo once daily for 7 days. Following an overnight fast, test medication was administered and patients were given a light meal within 15 minutes. MAO and BAO were determined 24 hours following the last day of study medication. MAO was estimated from a 1 hour continuous collection of gastric contents following subcutaneous injection of 6.0 mcg/kg of pentagastrin to stimulate acid secretion. This study demonstrated that, after treatment for 7 days, patients treated with pantoprazole sodium for injection had a significantly lower MAO and BAO than those treated with placebo (p<0.001), and results were comparable to those of patients treated with Pantoprazole Delayed-Release Tablets (see Table 5).
|
* p<0.001 Significantly different from pantoprazole sodium for injection. |
|||
| Parameter |
Pantoprazole Sodium for Injection DAY 7 |
Pantoprazole Delayed-Release Tablets DAY 7 |
Placebo DAY 7 |
| Maximum acid output (mean ± SD) |
8.4 ± 5.9 n=25 |
6.3 ± 6.6 n=22 |
20.9 ± 14.5* n=24 |
| Basal acid output (mean ± SD) |
0.4 ± 0.5 n=25 |
0.6 ± 0.8 n=22 |
2.8 ± 3.0* n=23 |
Study 2 was a single-center, double-blind, parallel-group study to compare the clinical effects of pantoprazole sodium for injection and Pantoprazole Delayed-Release Tablets. Patients (n=45, median age 56 years, 21 males and 24 females) with acute endoscopically proven reflux esophagitis (Savary/Miller Stage II or III) with at least 1 of 3 symptoms typical for reflux esophagitis (acid eructation, heartburn, or pain on swallowing) were randomized to receive either 40 mg pantoprazole sodium for injection or 40 mg Pantoprazole Delayed-Release Tablets once daily for 5 days. After the initial 5 days, all patients were treated with 40 mg oral pantoprazole daily to complete a total of 8 weeks of treatment. Symptom relief was assessed by calculating the daily mean of the sums of the average scores for these 3 symptoms and the daily mean of the average score for each of the symptoms separately. There was no significant difference in symptom relief between pantoprazole sodium for injection and Pantoprazole Delayed-Release Tablets within the first 5 days. A repeat endoscopy after 8 weeks of treatment revealed that 20 out of 23 (87%) patients treated with pantoprazole sodium for injection plus Pantoprazole Delayed-Release Tablets and 19 out of 22 (86%) of the patients treated with Pantoprazole Delayed-Release Tablets had endoscopically proven healing of their esophageal lesions.
Data comparing pantoprazole sodium for injection to other PPIs (oral or intravenous) or H2-receptor antagonists (oral or intravenous) are limited, and therefore, are inadequate to support any conclusions regarding comparative efficacy.
2.1 Dosage for Gastroesophageal Reflux Disease Associated With A History of Erosive Esophagitis (2.1 Dosage for Gastroesophageal Reflux Disease Associated With a History of Erosive Esophagitis)
The recommended adult dosage of pantoprazole sodium for injection is 40 mg given once daily by intravenous infusion for 7 to 10 days.
Discontinue treatment with pantoprazole sodium for injection as soon as the patient is able to receive treatment with Pantoprazole Delayed-Release Tablets or Oral Suspension.
Data on the safe and effective dosing for conditions other than those described [see Indications and Usage (1)] such as life-threatening upper gastrointestinal bleeds, are not available. Pantoprazole sodium for injection 40 mg once daily does not raise gastric pH to levels sufficient to contribute to the treatment of such life-threatening conditions.
2.4 Preparation and Administration Instructions for Pathological Hypersecretion Including Zollinger Ellison Syndrome (2.4 Preparation and Administration Instructions for Pathological Hypersecretion Including Zollinger-Ellison Syndrome)
Only for intravenous infusion; other parenteral routes of administration are not recommended.
Fifteen Minute Infusion
- Reconstitute each vial of pantoprazole sodium for injection with 10 mL of 0.9% Sodium Chloride Injection, USP.
- Combine the contents of the two vials and further dilute with 80 mL of 5% Dextrose Injection, USP, 0.9% Sodium Chloride Injection, USP, or Lactated Ringer's Injection, USP, to a total volume of 100 mL with a final concentration of approximately 0.8 mg per mL.
- Inspect the diluted pantoprazole sodium for injection solution visually for particular matter and discoloration prior to and during administration.
- Administer intravenously over a period of approximately 15 minutes at a rate of approximately 7 mL/min.
2.2 Preparation and Administration Instructions for Gastroesophageal Reflux Disease Associated With A History of Erosive Esophagitis (2.2 Preparation and Administration Instructions for Gastroesophageal Reflux Disease Associated With a History of Erosive Esophagitis)
Only for intravenous infusion; other parenteral routes of administration are not recommended.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:44:11.615063 · Updated: 2026-03-14T22:19:05.102582