These Highlights Do Not Include All The Information Needed To Use Venlafaxine Hydrochloride Extended-release Capsules Safely And Effectively. See Full Prescribing Information For Venlafaxine Hydrochloride Extended-release Capsules.
c37a73fa-ceee-4555-a61b-e1e66ad6e97d
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Antidepressants increased the risk of suicidal thoughts and behavior in pediatric and young adult patients in short-term studies. Closely monitor all antidepressant-treated patients for clinical worsening, and emergence of suicidal thoughts and behaviors [see Warnings and Precautions (5.1) ] . Venlafaxine hydrochloride extended-release capsules are not approved for use in pediatric patient s [see Use in Specific Populations (8.4) ] .
Indications and Usage
Venlafaxine Hydrochloride Extended-Release Capsules, USP are indicated in adults for the treatment of: Major Depressive Disorder (MDD) [see Clinical Studies (14.1) ] Generalized Anxiety Disorder (GAD) [see Clinical Studies (14.2) ] Social Anxiety Disorder (SAD) [see Clinical Studies (14.3) ] Panic Disorder (PD) [see Clinical Studies (14.4) ]
Dosage and Administration
Indication Starting Dose Target Dose Maximum Dose MDD ( 2.2 ) 37.5 to 75 mg/day 75 mg/day 225 mg/day GAD ( 2.3 ) 37.5 to75 mg/day 75 mg/day 225 mg/day SAD ( 2.4 ) 75 mg/day 75 mg/day 75 mg/day PD ( 2.5 ) 37.5 mg/day 75 mg/day 225 mg/day Take once daily with food. Capsules should be taken whole; do not divide, crush, chew, or dissolve ( 2.1 ). When discontinuing treatment, reduce the dose gradually ( 2.10 , 5.7 ). Renal impairment: reduce the total daily dose by 25% to 50% in patients with renal impairment. Reduce the total daily dose by 50% or more in patients undergoing dialysis or with severe renal impairment ( 2.9 ). Hepatic impairment: reduce the daily dose by 50% in patients with mild to moderate hepatic impairment. In patients with severe hepatic impairment or hepatic cirrhosis, it may be necessary to reduce the dose by more than 50% ( 2.8 ).
Warnings and Precautions
Serotonin Syndrome : Increased risk when co-administered with other serotonergic agents, but also when taken alone. If it occurs, discontinue venlafaxine hydrochloride extended-release capsules and serotonergic agents and initiate supportive treatment ( 4 , 5.2 , 7.1 ). Elevated Blood Pressure : Control hypertension before initiating treatment. Monitor blood pressure regularly during treatment ( 5.3 ). Increased Risk of Bleeding : Concomitant use of aspirin, NSAIDs, other antiplatelet drugs, warfarin, and other anticoagulants may increase risk ( 5.4 ). Angle-Closure Glaucoma : Angle-closure glaucoma has occurred in patients with untreated anatomically narrow angles, treated with antidepressants ( 5.5 ). Activation of Mania or Hypomania : Screen patients for bipolar disorder ( 5.6 ). Discontinuation Syndrome : Taper dose and monitor for discontinuation symptoms ( 5.7 ). Seizures : Can occur. Use cautiously in patients with seizure disorder ( 5.8 ). Hyponatremia : Can occur in association with SIADH ( 5.9 ). Interstitial Lung Disease and Eosinophilic Pneumonia : Can occur ( 5.12 ). Sexual Dysfunction : Venlafaxine hydrochloride extended-release capsules may cause symptoms of sexual dysfunction ( 5.13 ).
Contraindications
Venlafaxine Hydrochloride Extended-Release Capsules are contraindicated in patients: with known hypersensitivity to venlafaxine hydrochloride, desvenlafaxine succinate or to any excipients in the formulation [see Adverse Reactions (6.2) ] . taking, or within 14 days of stopping, MAOIs (including the MAOIs linezolid and intravenous methylene blue) because of the risk of serotonin syndrome [see Dosage and Administration (2.11) , Warnings and Precautions (5.2) , and Drug Interactions (7.1) ] .
Adverse Reactions
The following adverse reactions are discussed in more detail in other sections of the labeling: Hypersensitivity [see Contraindications (4) ] Suicidal Thoughts and Behaviors in Adolescents and Young Adults [see Warnings and Precautions (5.1) ] Serotonin Syndrome [see Warnings and Precautions (5.2) ] Elevated Blood Pressure [see Warnings and Precautions (5.3) ] Increased Risk of Bleeding [see Warnings and Precautions (5.4) ] Angle-Closure Glaucoma [see Warnings and Precautions (5.5) ] Activation of Mania/Hypomania [see Warnings and Precautions (5.6) ] Discontinuation Syndrome [see Warnings and Precautions (5.7) ] Seizure [see Warnings and Precautions (5.8) ] Hyponatremia [see Warnings and Precautions (5.9) ] Weight and Height Changes in Pediatric Patients [see Warnings and Precautions (5.10) ] Appetite Changes in Pediatric Patients [see Warnings and Precautions (5.11) ] Interstitial Lung Disease and Eosinophilic Pneumonia [see Warnings and Precautions (5.12) ] Sexual Dysfunction [see Warnings and Precautions (5.13) ]
Storage and Handling
Product: 50090-7323 NDC: 50090-7323-0 30 CAPSULE, EXTENDED RELEASE in a BOTTLE NDC: 50090-7323-1 90 CAPSULE, EXTENDED RELEASE in a BOTTLE
How Supplied
Product: 50090-7323 NDC: 50090-7323-0 30 CAPSULE, EXTENDED RELEASE in a BOTTLE NDC: 50090-7323-1 90 CAPSULE, EXTENDED RELEASE in a BOTTLE
Medication Information
Warnings and Precautions
Serotonin Syndrome : Increased risk when co-administered with other serotonergic agents, but also when taken alone. If it occurs, discontinue venlafaxine hydrochloride extended-release capsules and serotonergic agents and initiate supportive treatment ( 4 , 5.2 , 7.1 ). Elevated Blood Pressure : Control hypertension before initiating treatment. Monitor blood pressure regularly during treatment ( 5.3 ). Increased Risk of Bleeding : Concomitant use of aspirin, NSAIDs, other antiplatelet drugs, warfarin, and other anticoagulants may increase risk ( 5.4 ). Angle-Closure Glaucoma : Angle-closure glaucoma has occurred in patients with untreated anatomically narrow angles, treated with antidepressants ( 5.5 ). Activation of Mania or Hypomania : Screen patients for bipolar disorder ( 5.6 ). Discontinuation Syndrome : Taper dose and monitor for discontinuation symptoms ( 5.7 ). Seizures : Can occur. Use cautiously in patients with seizure disorder ( 5.8 ). Hyponatremia : Can occur in association with SIADH ( 5.9 ). Interstitial Lung Disease and Eosinophilic Pneumonia : Can occur ( 5.12 ). Sexual Dysfunction : Venlafaxine hydrochloride extended-release capsules may cause symptoms of sexual dysfunction ( 5.13 ).
Indications and Usage
Venlafaxine Hydrochloride Extended-Release Capsules, USP are indicated in adults for the treatment of: Major Depressive Disorder (MDD) [see Clinical Studies (14.1) ] Generalized Anxiety Disorder (GAD) [see Clinical Studies (14.2) ] Social Anxiety Disorder (SAD) [see Clinical Studies (14.3) ] Panic Disorder (PD) [see Clinical Studies (14.4) ]
Dosage and Administration
Indication Starting Dose Target Dose Maximum Dose MDD ( 2.2 ) 37.5 to 75 mg/day 75 mg/day 225 mg/day GAD ( 2.3 ) 37.5 to75 mg/day 75 mg/day 225 mg/day SAD ( 2.4 ) 75 mg/day 75 mg/day 75 mg/day PD ( 2.5 ) 37.5 mg/day 75 mg/day 225 mg/day Take once daily with food. Capsules should be taken whole; do not divide, crush, chew, or dissolve ( 2.1 ). When discontinuing treatment, reduce the dose gradually ( 2.10 , 5.7 ). Renal impairment: reduce the total daily dose by 25% to 50% in patients with renal impairment. Reduce the total daily dose by 50% or more in patients undergoing dialysis or with severe renal impairment ( 2.9 ). Hepatic impairment: reduce the daily dose by 50% in patients with mild to moderate hepatic impairment. In patients with severe hepatic impairment or hepatic cirrhosis, it may be necessary to reduce the dose by more than 50% ( 2.8 ).
Contraindications
Venlafaxine Hydrochloride Extended-Release Capsules are contraindicated in patients: with known hypersensitivity to venlafaxine hydrochloride, desvenlafaxine succinate or to any excipients in the formulation [see Adverse Reactions (6.2) ] . taking, or within 14 days of stopping, MAOIs (including the MAOIs linezolid and intravenous methylene blue) because of the risk of serotonin syndrome [see Dosage and Administration (2.11) , Warnings and Precautions (5.2) , and Drug Interactions (7.1) ] .
Adverse Reactions
The following adverse reactions are discussed in more detail in other sections of the labeling: Hypersensitivity [see Contraindications (4) ] Suicidal Thoughts and Behaviors in Adolescents and Young Adults [see Warnings and Precautions (5.1) ] Serotonin Syndrome [see Warnings and Precautions (5.2) ] Elevated Blood Pressure [see Warnings and Precautions (5.3) ] Increased Risk of Bleeding [see Warnings and Precautions (5.4) ] Angle-Closure Glaucoma [see Warnings and Precautions (5.5) ] Activation of Mania/Hypomania [see Warnings and Precautions (5.6) ] Discontinuation Syndrome [see Warnings and Precautions (5.7) ] Seizure [see Warnings and Precautions (5.8) ] Hyponatremia [see Warnings and Precautions (5.9) ] Weight and Height Changes in Pediatric Patients [see Warnings and Precautions (5.10) ] Appetite Changes in Pediatric Patients [see Warnings and Precautions (5.11) ] Interstitial Lung Disease and Eosinophilic Pneumonia [see Warnings and Precautions (5.12) ] Sexual Dysfunction [see Warnings and Precautions (5.13) ]
Storage and Handling
Product: 50090-7323 NDC: 50090-7323-0 30 CAPSULE, EXTENDED RELEASE in a BOTTLE NDC: 50090-7323-1 90 CAPSULE, EXTENDED RELEASE in a BOTTLE
How Supplied
Product: 50090-7323 NDC: 50090-7323-0 30 CAPSULE, EXTENDED RELEASE in a BOTTLE NDC: 50090-7323-1 90 CAPSULE, EXTENDED RELEASE in a BOTTLE
Description
Antidepressants increased the risk of suicidal thoughts and behavior in pediatric and young adult patients in short-term studies. Closely monitor all antidepressant-treated patients for clinical worsening, and emergence of suicidal thoughts and behaviors [see Warnings and Precautions (5.1) ] . Venlafaxine hydrochloride extended-release capsules are not approved for use in pediatric patient s [see Use in Specific Populations (8.4) ] .
Section 42229-5
Weight Changes
The average change in body weight and incidence of weight loss (percentage of patients who lost 3.5% or more) in the placebo-controlled pediatric studies in MDD, GAD, and SAD are shown in Tables 3 and 4.
| Indication
(Duration) |
Venlafaxine Hydrochloride Extended-Release Capsules | Placebo |
|---|---|---|
| MDD and GAD
(4 pooled studies, 8 weeks) |
-0.45 (n=333) | +0.77 (n=333) |
| SAD
(16 weeks) |
-0.75 (n=137) | +0.76 (n=148) |
| aVenlafaxine hydrochloride extended-release capsules are not approved for use in pediatric patients. |
| Indication
(Duration) |
Venlafaxine Hydrochloride Extended-Release Capsules | Placebo |
|---|---|---|
| MDD and GAD
(4 pooled studies, 8 weeks) |
18 b(n=333) | 3.6 (n=333) |
| SAD
(16 weeks) |
47 b(n=137) | 14 (n=148) |
|
aVenlafaxine hydrochloride extended-release capsules are not approved for use in pediatric patients.
bp<0.001 versus placebo |
Weight loss was not limited to patients with anorexia [see Warnings and Precautions (5.11)] .
The risks associated with longer term venlafaxine hydrochloride extended-release capsules use were assessed in an open-label MDD study of children and adolescents who received venlafaxine hydrochloride extended-release capsules for up to six months. The children and adolescents in the study had increases in weight that were less than expected, based on data from age- and sex-matched peers. The difference between observed weight gain and expected weight gain was larger for children (<12 years old) than for adolescents (≥12 years old).
Venlafaxine hydrochloride extended-release capsules are not approved for use in pediatric patients [ Use in Specific Populations (8.4)] .
Section 42231-1
|
Dispense with Medication Guide available at: www.epic-pharma.com/medguide/Venlafaxine-Hydrochloride-ER-Capsule.pdf
MEDICATION GUIDE
Extended-Release Capsules |
||||
|
What is the most important information I should know about venlafaxine hydrochloride extended-release capsules?
Venlafaxine hydrochloride extended-release capsules may cause serious side effects, including:
|
||||
|
|
|||
|
What are venlafaxine hydrochloride extended-release capsules?
Venlafaxine hydrochloride extended-release capsules are a prescription medicine used to treat adults with:
|
||||
Do not take venlafaxine hydrochloride extended-release capsules if you:
Ask your healthcare provider or pharmacist if you are not sure if you take an MAOI, including MAOIs such as linezolid or intravenous methylene blue.
|
||||
Before taking venlafaxine hydrochloride extended-release capsules tell your healthcare provider about all your medical conditions, including if you:
Tell your healthcare provider about all the medicines you take,including prescription and over-the-counter medicines, vitamins, and herbal supplements.
Ask your healthcare provider if you are not sure if you are taking any of these medicines. Your healthcare provider can tell you if it is safe to take venlafaxine hydrochloride extended-release capsules with your other medicines.
|
||||
How should I take venlafaxine hydrochloride extended-release capsules?
|
||||
What should I avoid while taking venlafaxine hydrochloride extended-release capsules?
|
||||
|
What are the possible side effects of venlafaxine hydrochloride extended-release capsules?
venlafaxine hydrochloride extended-release capsules may cause serious side effects, including:
|
||||
|
|
|||
|
||||
|
|
|||
|
||||
|
|
|
||
|
||||
|
Your healthcare provider may tell you to stop taking venlafaxine hydrochloride extended-release capsules if you develop serious side effects during treatment with venlafaxine hydrochloride extended-release capsules. |
||||
| The most common side effects of venlafaxine hydrochloride extended-release capsules include: | ||||
|
|
|||
| These are not all the possible side effects of venlafaxine hydrochloride extended-release capsules.
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
||||
How should I store venlafaxine hydrochloride extended-release capsules?
|
||||
|
General information about the safe and effective use of venlafaxine hydrochloride extended-release capsules.
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use venlafaxine hydrochloride extended-release capsules for a condition for which it was not prescribed. Do not give venlafaxine hydrochloride extended-release capsules to other people, even if they have the same symptoms that you have. It may harm them. You can ask your healthcare provider or pharmacist for information about venlafaxine hydrochloride extended-release capsules that is written for healthcare professionals. |
||||
|
What are the ingredients in venlafaxine hydrochloride extended-release capsules? Active ingredient:venlafaxine hydrochloride, USP Inactive ingredients: empty hard gelatin capsules, ethylcellulose, hypromellose, povidone, sugar spheres (composed of corn starch and sucrose) and talc. The 37.5 mg capsule shell contains red iron oxide, gelatin, and titanium dioxide. The 75 mg capsule shell contains black iron oxide, red iron oxide, gelatin, and titanium dioxide. The 150 mg capsule shell contains FD&C blue 1, FD&C red 3, FD&C yellow 6, gelatin, and titanium dioxide. The imprinting ink contains black iron oxide, potassium hydroxide and shellac. The brands listed are the registered trademarks of their respective owners. This Medication Guide has been approved by the U.S. Food and Drug Administration for all antidepressants.
Distributed by:
ZLN034
|
9.2 Abuse
Abuse is the intentional, non-therapeutic use of a drug, even once, for its desirable psychological or physiological effects.
While venlafaxine has not been systematically studied in clinical studies for its potential for abuse, there was no indication of drug-seeking behavior in the clinical studies. However, it is not possible to predict on the basis of premarketing experience the extent to which a CNS-active drug will be misused, diverted, and/or abused once marketed. Consequently, providers should carefully evaluate patients for history of drug abuse and follow such patients closely, observing them for signs of misuse or abuse of venlafaxine (e.g., development of tolerance, incrementation of dose, drug-seeking behavior).
5.8 Seizures
Cases of seizure have been reported with venlafaxine therapy. Venlafaxine hydrochloride extended-release capsules have not been systematically evaluated in patients with seizure disorder. Venlafaxine hydrochloride extended-release capsules should be prescribed with caution in patients with a seizure disorder.
11 Description
Venlafaxine Hydrochloride Extended-Release Capsules, USP are an extended-release capsule for once-a-day oral administration that contains venlafaxine hydrochloride, a serotonin and norepinephrine reuptake inhibitor (SNRI).
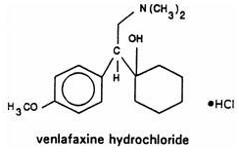
Venlafaxine is designated (R/S)-1-[2-(dimethylamino)-1-(4-methoxyphenyl)ethyl] cyclohexanol hydrochloride or (±)-1-[α-[(dimethylamino)methyl]-p-methoxybenzyl] cyclohexanol hydrochloride and has the empirical formula of C 17H 27NO 2HCl. Its molecular weight is 313.86. The structural formula is shown as follows:
Venlafaxine hydrochloride is a white to off-white crystalline solid, with a solubility of 572 mg/mL in water (adjusted to ionic strength of 0.2 M with sodium chloride). Its octanol: water (0.2 M sodium chloride) partition coefficient is 0.43.
Drug release is controlled by diffusion through the coating membrane on the spheroids and is not pH-dependent. Capsules contain venlafaxine hydrochloride equivalent to 37.5 mg, 75 mg, or 150 mg venlafaxine. Inactive ingredients consist of empty hard gelatin capsules, ethylcellulose, hypromellose, povidone, sugar spheres (composed of corn starch and sucrose) and talc.
The 37.5 mg capsule shell contains red iron oxide, gelatin, and titanium dioxide. The 75 mg capsule shell contains black iron oxide, red iron oxide, gelatin, and titanium dioxide. The 150 mg capsule shell contains FD&C blue 1, FD&C red 3, FD&C yellow 6, gelatin, and titanium dioxide.
The imprinting ink contains black iron oxide, potassium hydroxide and shellac.
FDA approved dissolution test specifications differ from USP.
9.3 Dependence
Physical dependence is a state that develops as a result of physiological adaptation in response to repeated drug use, manifested by withdrawal signs and symptoms after abrupt discontinuation or a significant dose reduction of a drug.
In vitrostudies revealed that venlafaxine has virtually no affinity for opiate, benzodiazepine, phencyclidine (PCP), or N-methyl-D-aspartic acid (NMDA) receptors.
Venlafaxine was not found to have any significant CNS stimulant activity in rodents. In primate drug discrimination studies, venlafaxine showed no significant stimulant or depressant abuse liability.
Discontinuation effects have been reported in patients receiving venlafaxine [see Dosage and Administration (2.10)and Warnings and Precautions (5.7)] .
5.9 Hyponatremia
Hyponatremia can occur as a result of treatment with SNRIs, including venlafaxine hydrochloride extended-release capsules. In many cases, the hyponatremia appears to be the result of the Syndrome of Inappropriate Antidiuretic Hormone (SIADH) secretion. Cases with serum sodium lower than 110 mmol/L have been reported. Elderly patients may be at greater risk of developing hyponatremia with SNRIs. Also, patients taking diuretics, or those who are otherwise volume-depleted, may be at greater risk [see Use in Specific Populations (8.5)and Clinical Pharmacology (12.3)] . Consider discontinuation of venlafaxine hydrochloride extended-release capsules in patients with symptomatic hyponatremia, and institute appropriate medical intervention.
Signs and symptoms of hyponatremia include headache, difficulty concentrating, memory impairment, confusion, weakness, and unsteadiness, which may lead to falls. Signs and symptoms associated with more severe and/or acute cases have included hallucination, syncope, seizure, coma, respiratory arrest, and death.
8.4 Pediatric Use
Safety and effectiveness of venlafaxine hydrochloride extended-release capsules in pediatric patients have not been established.
Two placebo-controlled trials in 766 pediatric patients with MDD and two placebo-controlled trials in 793 pediatric patients with GAD have been conducted with venlafaxine hydrochloride extended-release capsules, and the data were not sufficient to support use in pediatric patients.
In the studies conducted in pediatric patients ages 6 to 17 years, the occurrence of blood pressure and cholesterol increases was considered to be clinically relevant in pediatric patients and was similar to that observed in adult patients [see Warnings and Precautions (5.3), Adverse Reactions (6.1)] . The following adverse reactions were also observed in pediatric patients: abdominal pain, agitation, dyspepsia, ecchymosis, epistaxis, and myalgia.
Although no studies have been designed to primarily assess venlafaxine hydrochloride extended-release capsules' impact on the growth, development, and maturation of children and adolescents, the studies that have been done suggest that venlafaxine hydrochloride extended-release capsules may adversely affect weight and height [see Warnings and Precautions (5.10, 5.11)]. Decreased appetite and weight loss were observed in placebo-controlled studies of pediatric patients 6 to 17 years.
In pediatric clinical studies, the adverse reaction, suicidal ideation, was observed. Antidepressants increased the risk of suicidal thoughts and behaviors in pediatric patients [see Boxed Warning, Warnings and Precautions (5.1)] .
8.5 Geriatric Use
The percentage of patients in clinical studies for venlafaxine hydrochloride extended-release capsules for MDD, GAD, SAD, and PD who were 65 years of age or older are shown in Table 16.
| Indication | Venlafaxine Hydrochloride Extended-Release Capsules |
|---|---|
| MDD | 4 (14/357) |
| GAD | 6 (77/1,381) |
| SAD | 1 (10/819) |
| PD | 2 (16/1,001) |
| aIn addition, in the premarketing assessment of venlafaxine hydrochloride tablets (immediate-release), 12% (357/2,897) of patients were ≥65 years of age. |
No overall differences in effectiveness or safety were observed between geriatric patients and younger patients, and other reported clinical experience generally has not identified differences in response between the elderly and younger patients. However, greater sensitivity of some older individuals cannot be ruled out. SSRIs and SNRIs, including venlafaxine hydrochloride extended-release capsules, have been associated with cases of clinically significant hyponatremia in elderly patients, who may be at greater risk for this adverse event [see Warnings and Precautions (5.9)] .
The pharmacokinetics of venlafaxine and ODV are not substantially altered in the elderly [see Clinical Pharmacology (12.3)] (see Figure 1). No dose adjustment is recommended for the elderly on the basis of age alone, although other clinical circumstances, some of which may be more common in the elderly, such as renal or hepatic impairment, may warrant a dose reduction [see Dosage and Administration (2.8, 2.9)] .
2.5 Panic Disorder
The recommended starting dose is 37.5 mg per day of venlafaxine hydrochloride extended-release capsules for 7 days. Patients not responding to 75 mg per day may benefit from dose increases to a maximum of approximately 225 mg per day. Dose increases should be in increments of up to 75 mg per day, as needed, and should be made at intervals of not less than 7 days.
14.4 Panic Disorder
The efficacy of venlafaxine hydrochloride extended-release capsules as a treatment for Panic Disorder (PD) was established in two double-blind, 12-week, multicenter, placebo-controlled studies in adult outpatients meeting DSM-IV criteria for PD, with or without agoraphobia. Patients received fixed doses of 75 or 150 mg per day in one study (study 1) and 75 or 225 mg per day in the other study (study 2).
Efficacy was assessed on the basis of outcomes in three variables: (1) percentage of patients free of full-symptom panic attacks on the Panic and Anticipatory Anxiety Scale (PAAS); (2) mean change from baseline to endpoint on the Panic Disorder Severity Scale (PDSS) total score; and (3) percentage of patients rated as responders (much improved or very much improved) on the Clinical Global Impressions (CGI) Improvement scale. In these two studies, venlafaxine hydrochloride extended-release capsules were statistically significantly more effective than placebo (for each fixed dose) on all three endpoints, but a dose-response relationship was not clearly established.
Examination of subsets of the population studied did not reveal any differential responsiveness on the basis of gender. There was insufficient information to determine the effect of age or race on outcome in these studies.
In a longer term study (study 3), adult outpatients meeting DSM-IV criteria for PD who had responded during a 12-week open phase with venlafaxine hydrochloride extended-release capsules (75 to 225 mg per day) were randomly assigned to continue the same venlafaxine hydrochloride extended-release capsules dose (75, 150, or 225 mg) or switch to placebo for observation for relapse under double-blind conditions. Response during the open phase was defined as ≤1 full-symptom panic attack per week during the last 2 weeks of the open phase and a CGI Improvement score of 1 (very much improved) or 2 (much improved). Relapse during the double-blind phase was defined as having 2 or more full-symptom panic attacks per week for 2 consecutive weeks or having discontinued due to loss of effectiveness as determined by the investigators during the study. Randomized patients were in response status for a mean time of 34 days prior to being randomized. In the randomized phase following the 12-week open-label period, patients receiving continued venlafaxine hydrochloride extended-release capsules experienced a statistically significantly longer time to relapse.
| Study Treatment Group Number | Primary Efficacy Measure: Whether Free of Full-symptom Panic Attacks | |||
|---|---|---|---|---|
| Percent of patients Free of Full Symptom Panic Attack | Adjusted Odds Ratio ato Placebo | Adjusted Odds Ratio a 95% Confidence Interval | ||
| Study 1 | Venlafaxine Hydrochloride Extended-Release Capsules 75 mg* | 54.1% (85/157) | 2. 268 | (1.43, 3.59) |
| Venlafaxine Hydrochloride Extended-Release Capsules 150 mg* | 61.4% (97/158) | 3.035 | (1.91, 4.82) | |
| Placebo | 34.4% (53/154) | -- | -- | |
| Study 2 | Venlafaxine Hydrochloride Extended-Release Capsules 75 mg* | 64.1% (100/156) | 2.350 | (1.46, 3.78) |
| Venlafaxine Hydrochloride Extended-Release Capsules 225 mg* | 70.0% (112/160) | 2.890 | (1.80, 4.64) | |
| Placebo | 46.5% (73/157) | -- | -- |
aOdds ratio (drug to placebo) in terms of probability of free of full-symptom panic attacks based on logistic regression model.
95% CI: 95% confidence interval without adjusting for multiple dose arms.
*Doses statistically significantly superior to placebo.
4 Contraindications
Venlafaxine Hydrochloride Extended-Release Capsules are contraindicated in patients:
- with known hypersensitivity to venlafaxine hydrochloride, desvenlafaxine succinate or to any excipients in the formulation [see Adverse Reactions (6.2)] .
- taking, or within 14 days of stopping, MAOIs (including the MAOIs linezolid and intravenous methylene blue) because of the risk of serotonin syndrome [see Dosage and Administration (2.11), Warnings and Precautions (5.2), and Drug Interactions (7.1)] .
6 Adverse Reactions
The following adverse reactions are discussed in more detail in other sections of the labeling:
- Hypersensitivity [see Contraindications (4)]
- Suicidal Thoughts and Behaviors in Adolescents and Young Adults [see Warnings and Precautions (5.1)]
- Serotonin Syndrome [see Warnings and Precautions (5.2)]
- Elevated Blood Pressure [see Warnings and Precautions (5.3)]
- Increased Risk of Bleeding [see Warnings and Precautions (5.4)]
- Angle-Closure Glaucoma [see Warnings and Precautions (5.5)]
- Activation of Mania/Hypomania [see Warnings and Precautions (5.6)]
- Discontinuation Syndrome [see Warnings and Precautions (5.7)]
- Seizure [see Warnings and Precautions (5.8)]
- Hyponatremia [see Warnings and Precautions (5.9)]
- Weight and Height Changes in Pediatric Patients [see Warnings and Precautions (5.10)]
- Appetite Changes in Pediatric Patients [see Warnings and Precautions (5.11)]
- Interstitial Lung Disease and Eosinophilic Pneumonia [see Warnings and Precautions (5.12)]
- Sexual Dysfunction [see Warnings and Precautions (5.13)]
8.7 Renal Impairment
Dosage adjustment is recommended in patients with mild (CLcr=60 to 89 mL/min), moderate (CLcr=30 to 59 mL/min), or severe (CLcr<30 mL/min) renal impairment, and in patients undergoing hemodialysis [see Dosage and Administration (2.9)and Clinical Pharmacology (12.3)].
12.2 Pharmacodynamics
In vitrostudies have demonstrated that venlafaxine and its active metabolite, O-desmethylvenlafaxine (ODV), are potent and selective inhibitors of neuronal serotonin and norepinephrine reuptake and weak inhibitors of dopamine reuptake. Venlafaxine and ODV have no significant affinity for muscarinic-cholinergic, H 1-histaminergic, or α 1-adrenergic receptors in vitro. Pharmacologic activity at these receptors is hypothesized to be associated with the various anticholinergic, sedative, and cardiovascular effects seen with other psychotropic drugs. Venlafaxine and ODV do not possess monoamine oxidase (MAO) inhibitory activity.
12.3 Pharmacokinetics
Venlafaxine and ODV steady-state concentrations are reached within 3 days. Venlafaxine and ODV exhibited linear kinetics over the dosage range of 75 to 450 mg per day (0.33 to 2 times the maximum recommended dosage). Time of administration (AM versus PM) did not affect the pharmacokinetics of venlafaxine and ODV from the 75 mg venlafaxine hydrochloride extended-release capsules.
5.2 Serotonin Syndrome
Serotonin-norepinephrine reuptake inhibitors (SNRIs), including venlafaxine hydrochloride extended-release capsules, can precipitate serotonin syndrome, a potentially life-threatening condition. The risk is increased with concomitant use of other serotonergic drugs (including triptans, tricyclic antidepressants, fentanyl, lithium, tramadol, meperidine, methadone, tryptophan, buspirone, amphetamines, and St. John’s Wort) and with drugs that impair metabolism of serotonin, i.e., MAOIs [see Contraindications (4), Drug Interactions (7.1)] . Serotonin syndrome can also occur when these drugs are used alone.
Serotonin syndrome signs and symptoms may include mental status changes (e.g., agitation, hallucinations, delirium, coma), autonomic instability (e.g., tachycardia, labile blood pressure, dizziness, diaphoresis, flushing, hyperthermia), neuromuscular symptoms (e.g., tremor, rigidity, myoclonus, hyperreflexia, incoordination), seizures, and gastrointestinal symptoms (e.g., nausea, vomiting, diarrhea).
The concomitant use of venlafaxine hydrochloride extended-release capsules with MAOIs is contraindicated. In addition, do not initiate venlafaxine hydrochloride extended-release capsules in a patient being treated with MAOIs such as linezolid or intravenous methylene blue. No reports involved the administration of methylene blue by other routes (such as oral tablets or local tissue injection). If it is necessary to initiate treatment with an MAOI such as linezolid or intravenous methylene blue in a patient taking venlafaxine hydrochloride extended-release capsules, discontinue venlafaxine hydrochloride extended-release capsules before initiating treatment with the MAOI [see Contraindications (4), Drug Interactions (7.1)] .
Monitor all patients taking venlafaxine hydrochloride extended-release capsules for the emergence of serotonin syndrome. Discontinue treatment with venlafaxine hydrochloride extended-release capsules and any concomitant serotonergic agents immediately if the above symptoms occur, and initiate supportive symptomatic treatment. If concomitant use of venlafaxine hydrochloride extended-release capsules with other serotonergic drugs is clinically warranted, inform patients of the increased risk for serotonin syndrome and monitor for symptoms.
8.6 Hepatic Impairment
Dosage adjustment is recommended in patients with mild (Child-Pugh Class A), moderate (Child-Pugh Class B), or severe (Child-Pugh Class C) hepatic impairment or hepatic cirrhosis [see Dosage and Administration (2.8)and Clinical Pharmacology (12.3)] .
1 Indications and Usage
Venlafaxine Hydrochloride Extended-Release Capsules, USP are indicated in adults for the treatment of:
- Major Depressive Disorder (MDD) [see Clinical Studies (14.1)]
- Generalized Anxiety Disorder (GAD) [see Clinical Studies (14.2)]
- Social Anxiety Disorder (SAD) [see Clinical Studies (14.3)]
- Panic Disorder (PD) [see Clinical Studies (14.4)]
5.13 Sexual Dysfunction
Use of SNRIs, including venlafaxine hydrochloride extended-release capsules, may cause symptoms of sexual dysfunction [ see Adverse Reactions (6.1)] . In male patients, SNRI use may result in ejaculatory delay or failure, decreased libido, and erectile dysfunction. In female patients, SNRI use may result in decreased libido and delayed or absent orgasm. It is important for prescribers to inquire about sexual function prior to initiation of venlafaxine hydrochloride extended-release capsules and to inquire specifically about changes in sexual function during treatment, because sexual function may not be spontaneously reported. When evaluating changes in sexual function, obtaining a detailed history (including timing of symptom onset) is important because sexual symptoms may have other causes, including the underlying psychiatric disorder. Discuss potential management strategies to support patients in making informed decisions about treatment.
12.1 Mechanism of Action
The mechanism of action of venlafaxine in the treatment of MDD, GAD, SAD, and PD is unclear, but is thought to be related to the potentiation of serotonin and norepinephrine in the central nervous system, through inhibition of their reuptake.
9.1 Controlled Substance
Venlafaxine hydrochloride extended-release capsules contain venlafaxine which is not a controlled substance.
Venlafaxine Hydrochloride
5 Warnings and Precautions
- Serotonin Syndrome: Increased risk when co-administered with other serotonergic agents, but also when taken alone. If it occurs, discontinue venlafaxine hydrochloride extended-release capsules and serotonergic agents and initiate supportive treatment ( 4, 5.2, 7.1).
- Elevated Blood Pressure: Control hypertension before initiating treatment. Monitor blood pressure regularly during treatment ( 5.3).
- Increased Risk of Bleeding: Concomitant use of aspirin, NSAIDs, other antiplatelet drugs, warfarin, and other anticoagulants may increase risk ( 5.4).
- Angle-Closure Glaucoma: Angle-closure glaucoma has occurred in patients with untreated anatomically narrow angles, treated with antidepressants ( 5.5).
- Activation of Mania or Hypomania: Screen patients for bipolar disorder ( 5.6).
- Discontinuation Syndrome: Taper dose and monitor for discontinuation symptoms ( 5.7).
- Seizures: Can occur. Use cautiously in patients with seizure disorder ( 5.8).
- Hyponatremia: Can occur in association with SIADH ( 5.9).
- Interstitial Lung Disease and Eosinophilic Pneumonia: Can occur ( 5.12).
- Sexual Dysfunction: Venlafaxine hydrochloride extended-release capsules may cause symptoms of sexual dysfunction ( 5.13).
5.5 Angle Closure Glaucoma
The pupillary dilation that occurs following use of many antidepressant drugs including venlafaxine hydrochloride extended-release capsules may trigger an angle closure attack in a patient with anatomically narrow angles who does not have a patent iridectomy. Avoid use of antidepressants, including venlafaxine hydrochloride extended-release capsules, in patients with untreated anatomically narrow angles.
2 Dosage and Administration
| Indication | Starting Dose | Target Dose | Maximum Dose |
|---|---|---|---|
| MDD ( 2.2) | 37.5 to 75 mg/day | 75 mg/day | 225 mg/day |
| GAD ( 2.3) | 37.5 to75 mg/day | 75 mg/day | 225 mg/day |
| SAD ( 2.4) | 75 mg/day | 75 mg/day | 75 mg/day |
| PD ( 2.5) | 37.5 mg/day | 75 mg/day | 225 mg/day |
- Take once daily with food. Capsules should be taken whole; do not divide, crush, chew, or dissolve ( 2.1).
- When discontinuing treatment, reduce the dose gradually ( 2.10, 5.7).
- Renal impairment: reduce the total daily dose by 25% to 50% in patients with renal impairment. Reduce the total daily dose by 50% or more in patients undergoing dialysis or with severe renal impairment ( 2.9).
- Hepatic impairment: reduce the daily dose by 50% in patients with mild to moderate hepatic impairment. In patients with severe hepatic impairment or hepatic cirrhosis, it may be necessary to reduce the dose by more than 50% ( 2.8).
5.3 Elevated Blood Pressure
In controlled trials, there were dose-related increases in systolic and diastolic blood pressure, as well as cases of sustained hypertension [see Adverse Reactions (6.1)].
Monitor blood pressure before initiating treatment with venlafaxine hydrochloride extended-release capsules and regularly during treatment. Control pre-existing hypertension before initiating treatment with venlafaxine hydrochloride extended-release capsules. Use caution in treating patients with pre-existing hypertension or cardiovascular or cerebrovascular conditions that might be compromised by increases in blood pressure. Sustained blood pressure elevation can lead to adverse outcomes. Cases of elevated blood pressure requiring immediate treatment have been reported with venlafaxine hydrochloride extended-release capsules. Consider dose reduction or discontinuation of treatment for patients who experience a sustained increase in blood pressure.
Across all clinical studies with venlafaxine hydrochloride tablets, 1.4% of patients in the venlafaxine hydrochloride extended-release capsules treated groups experienced a ≥15 mm Hg increase in supine diastolic blood pressure (SDBP) ≥105 mm Hg, compared to 0.9% of patients in the placebo groups. Similarly, 1% of patients in the venlafaxine hydrochloride extended-release capsules treated groups experienced a ≥20 mm Hg increase in supine systolic blood pressure (SSBP) with blood pressure ≥180 mm Hg, compared to 0.3% of patients in the placebo groups [see Adverse Reactions (6.1)] . Treatment with venlafaxine hydrochloride extended-release capsules was associated with sustained hypertension defined as SDBP ≥90 mm Hg and ≥10 mm Hg above baseline for three consecutive on-therapy visits [see Adverse Reactions (6.1)] . An insufficient number of patients received mean doses of venlafaxine hydrochloride extended-release capsules over 300 mg per day in clinical studies to fully evaluate the incidence of sustained increases in blood pressure at these higher doses.
3 Dosage Forms and Strengths
Venlafaxine Hydrochloride Extended-Release Capsules, USP are available in the following strengths:
- 37.5 mg capsules, opaque flesh cap and opaque flesh body, imprinted with "YH" on cap and "130" on body in black ink.
- 75 mg capsules, opaque light grey cap and opaque flesh body, imprinted with "YH" on cap and "129" on body in black ink.
- 150 mg capsules, opaque caramel cap and body, imprinted with "YH" on cap and "128" on body in black ink.
5.7 Discontinuation Syndrome
Discontinuation symptoms have been systematically evaluated in patients taking venlafaxine, including prospective analyses of clinical studies in GAD and retrospective surveys of studies in MDD and SAD. Abrupt discontinuation or dose reduction of venlafaxine at various doses has been found to be associated with the appearance of new symptoms, the frequency of which increased with increased dose level and with longer duration of treatment. Reported symptoms include agitation, anorexia, anxiety, confusion, impaired coordination and balance, diarrhea, dizziness, dry mouth, dysphoric mood, fasciculation, fatigue, flu-like symptoms, headaches, hypomania, insomnia, nausea, nervousness, nightmares, sensory disturbances (including shock-like electrical sensations), somnolence, sweating, tremor, vertigo, and vomiting.
There have been postmarketing reports of serious discontinuation symptoms which can be protracted and severe. Completed suicide, suicidal thoughts, aggression and violent behavior have been observed in patients during reduction in venlafaxine hydrochloride extended-release capsules dosage, including during discontinuation. Other postmarketing reports describe visual changes (such as blurred vision or trouble focusing) and increased blood pressure after stopping or reducing the dose of venlafaxine hydrochloride extended-release capsules.
During marketing of venlafaxine hydrochloride extended-release capsules, other SNRIs, and SSRIs, there have been reports of adverse events occurring upon discontinuation of these drugs, particularly when abrupt, including the following: irritability, lethargy, emotional lability, tinnitus, and seizures.
Patients should be monitored for these symptoms when discontinuing treatment with venlafaxine hydrochloride extended-release capsules. A gradual reduction in the dose, rather than abrupt cessation, is recommended. If intolerable symptoms occur following a decrease in the dose or upon discontinuation of treatment, then resuming the previously prescribed dose may be considered. Subsequently, the healthcare provider may continue decreasing the dose, but at a more gradual rate. In some patients, discontinuation may need to occur over a period of several months [see Dosage and Administration (2.10)] .
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of venlafaxine hydrochloride extended-release capsules. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Body as a Whole
– Anaphylaxis, angioedema
Cardiovascular System
– QT prolongation, ventricular fibrillation, ventricular tachycardia (including torsade de pointes), takotsubo cardiomyopathy
Digestive System
– Pancreatitis
Hemic/Lymphatic System
– Mucous membrane bleeding
[see
Warnings and Precautions (5.4)]
, blood dyscrasias (including agranulocytosis, aplastic anemia, neutropenia and pancytopenia), prolonged bleeding time, thrombocytopenia
Metabolic/Nutritional
– Hyponatremia
[see
Warnings and Precautions (5.9)]
, Syndrome of Inappropriate Antidiuretic Hormone (SIADH) secretion
[see
Warnings and Precautions (5.9)]
, abnormal liver function tests, hepatitis, prolactin increased
Musculoskeletal
– Rhabdomyolysis
Nervous System
– Neuroleptic Malignant Syndrome (NMS)
[see
Warnings and Precautions (5.2)]
, serotonergic syndrome
[see
Warnings and Precautions (5.2)]
, delirium, extrapyramidal reactions (including dystonia and dyskinesia), impaired coordination and balance, tardive dyskinesia
Respiratory, Thoracic and Mediastinal Disorders
– Anosmia, dyspnea, hyposmia, interstitial lung disease, pulmonary eosinophilia
[see
Warnings and Precautions (5.12)]
Skin and Appendages
– Stevens-Johnson syndrome, toxic epidermal necrolysis, erythema multiforme
Special Senses
– Angle-closure glaucoma
[see
Warnings and Precautions (5.5)]
2.2 Major Depressive Disorder
For most patients, the recommended starting dose for venlafaxine hydrochloride extended-release capsules is 75 mg per day, administered in a single dose. For some patients, it may be desirable to start at 37.5 mg per day for 4 to 7 days to allow new patients to adjust to the medication before increasing to 75 mg per day. Patients not responding to the initial 75 mg per day dose may benefit from dose increases to a maximum of 225 mg per day. Dose increases should be in increments of up to 75 mg per day, as needed, and should be made at intervals of not less than 4 days. In the clinical studies establishing efficacy, upward titration was permitted at intervals of 2 weeks or more.
8 Use in Specific Populations
Pregnancy:Third trimester use may increase risk for symptoms of poor neonatal adaptation (respiratory distress, temperature instability, feeding difficulty, hypotonia, tremor, irritability) in the neonate ( 8.1).
14.1 Major Depressive Disorder
The efficacy of venlafaxine hydrochloride extended-release capsules as a treatment for Major Depressive Disorder (MDD) was established in two placebo-controlled, short-term (8 weeks for study 1; 12 weeks for study 2), flexible-dose studies, with doses starting at 75 mg per day and ranging to 225 mg per day in adult outpatients meeting DSM-III-R or DSM-IV criteria for MDD. In moderately depressed outpatients, the initial dose of venlafaxine was 75 mg per day. In both studies, venlafaxine hydrochloride extended-release capsules demonstrated superiority over placebo on the primary efficacy measure defined as change from baseline in the HAM-D-21 total score to the endpoint visit, venlafaxine hydrochloride extended-release capsules also demonstrated superiority over placebo on the key secondary efficacy endpoint, the Clinical Global Impressions (CGI) Severity of Illness scale. Examination of gender subsets of the population studied did not reveal any differential responsiveness on the basis of gender.
A 4-week study of inpatients meeting DSM-III-R criteria for MDD with melancholia utilizing venlafaxine hydrochloride tablets in a range of 150 to 375 mg per day (divided in a three-times-a-day schedule) demonstrated superiority of venlafaxine hydrochloride tablets over placebo based on the HAM-D-21 total score. The mean dose in completers was 350 mg per day (study 3).
In a longer-term study, adult outpatients with MDD who had responded during an 8-week open-label study on venlafaxine hydrochloride extended-release capsules (75, 150, or 225 mg, once daily every morning) were randomized to continuation of their same venlafaxine hydrochloride extended-release capsules dose or to placebo, for up to 26 weeks of observation for relapse. Response during the open-label phase was defined as a CGI Severity of Illness item score of ≤3 and a HAM-D-21 total score of ≤10 at the day 56 evaluation. Relapse during the double-blind phase was defined as follows: (1) a reappearance of major depressive disorder as defined by DSM-IV criteria and a CGI Severity of Illness item score of ≥4 (moderately ill), (2) 2 consecutive CGI Severity of Illness item scores of ≥4, or (3) a final CGI Severity of Illness item score of ≥4 for any patient who withdrew from the study for any reason. Patients receiving continued venlafaxine hydrochloride extended-release capsules treatment experienced statistically significantly lower relapse rates over the subsequent 26 weeks compared with those receiving placebo (study 4).
In a second longer term trial, adult outpatients with MDD, recurrent type, who had responded (HAM-D-21 total score ≤12 at the day 56 evaluation) and continued to be improved [defined as the following criteria being met for days 56 through 180: (1) no HAM-D-21 total score ≥20; (2) no more than 2 HAM-D-21 total scores >10, and (3) no single CGI Severity of Illness item score ≥4 (moderately ill)] during an initial 26 weeks of treatment on venlafaxine hydrochloride tablets [100 to 200 mg per day, on a twice daily schedule] were randomized to continuation of their same venlafaxine hydrochloride tablets dose or to placebo. The follow-up period to observe patients for relapse, defined as a CGI Severity of Illness item score ≥4, was for up to 52 weeks. Patients receiving continued venlafaxine hydrochloride tablets treatment experienced statistically significantly lower relapse rates over the subsequent 52 weeks compared with those receiving placebo (study 5).
| Study Number | Treatment Group | Primary Efficacy Measure: HAM-D Score | ||
| Mean Baseline Score (SD) | LS Mean Change from Baseline |
Placebo Subtracted Differencea
(95% CI) |
||
| Study 1 | Venlafaxine Hydrochloride Extended-Release Capsules (75 to 225 mg/day)* | 24.5 | -11.7 | -4.45 (-6.66,-2.25) |
| Placebo | 23.6 | -7.24 | -- | |
| Study 2 | Venlafaxine Hydrochloride Extended-Release Capsules (75 to 225 mg/day)* | 24.5 | -15.11 | -6.40 (-8.45, -4.34) |
| Placebo | 24.9 | -8.71 | -- | |
| Study 3 | Venlafaxine Hydrochloride Extended-Release Capsules (150 to375 mg/day)* | 28.2 (0.5) | -14.9 | -10.2 (-14.4, -6.0) |
| Placebo | 28.6 (0.6) | -4.7 | -- | |
| SD=standard deviation; LS Mean=least-squares mean; CI=confidence interval. | ||||
| aDifference (drug minus placebo) in least-squares mean change from baseline. | ||||
| *Doses statistically significantly superior to placebo. |
5.4 Increased Risk of Bleeding
Drugs that interfere with serotonin reuptake inhibition, including venlafaxine hydrochloride extended-release capsules, may increase the risk of bleeding events, ranging from ecchymoses, hematomas, epistaxis, petechiae, and gastrointestinal hemorrhage to life-threatening hemorrhage. Concomitant use of aspirin, Nonsteroidal Anti-Inflammatory Drugs (NSAIDs), warfarin, and other anti-coagulants or other drugs known to affect platelet function may add to this risk. Case reports and epidemiological studies (case-control and cohort design) have demonstrated an association between use of drugs that interfere with serotonin reuptake and the occurrence of gastrointestinal bleeding. Based on data from the published observational studies, exposure to SNRIs, particularly in the month before delivery, has been associated with a less than 2-fold increase in the risk of postpartum hemorrhage [see Use in Specific Populations ( 8.1)] .
Inform patients about the increased risk of bleeding associated with the concomitant use of venlafaxine hydrochloride extended-release capsules and nonsteroidal anti-inflammatory drugs (NSAIDs), aspirin, or other drugs that affect coagulation. For patients taking warfarin, carefully monitor coagulation indices when initiating, titrating, or discontinuing venlafaxine hydrochloride extended-release capsules.
6.1 Clinical Studies Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical studies of another drug and may not reflect the rates observed in clinical practice.
2.3 Generalized Anxiety Disorder
For most patients, the recommended starting dose for venlafaxine hydrochloride extended-release capsules is 75 mg per day, administered in a single dose. For some patients, it may be desirable to start at 37.5 mg per day for 4 to 7 days to allow new patients to adjust to the medication before increasing to 75 mg per day. Patients not responding to the initial 75 mg per day dose may benefit from dose increases to a maximum of 225 mg per day. Dose increases should be in increments of up to 75 mg per day, as needed, and should be made at intervals of not less than 4 days.
14.2 Generalized Anxiety Disorder
The efficacy of venlafaxine hydrochloride extended-release capsules as a treatment for Generalized Anxiety Disorder (GAD) was established in two 8-week, placebo-controlled, fixed-dose studies (75 to 225 mg per day), one 6-month, placebo-controlled, flexible-dose study (75 to 225 mg per day), and one 6-month, placebo-controlled, fixed-dose study (37.5, 75, and 150 mg per day) in adult outpatients meeting DSM-IV criteria for GAD.
In one 8-week study, venlafaxine hydrochloride extended-release capsules demonstrated superiority over placebo for the 75, 150, and 225 mg per day doses as measured by the Hamilton Rating Scale for Anxiety (HAM-A) total score, both the HAM-A anxiety and tension items, and the Clinical Global Impressions (CGI) scale. However, the 75 and 150 mg per day doses were not as consistently effective as the highest dose (study 1). A second 8-week study evaluating doses of 75 and 150 mg per day and placebo showed that both doses were more effective than placebo on some of these same outcomes; however, the 75 mg per day dose was more consistently effective than the 150 mg per day dose (study 2). A dose-response relationship for effectiveness in GAD was not clearly established in the 75 to 225 mg per day dose range studied.
Two 6-month studies, one evaluating venlafaxine hydrochloride extended-release capsules doses of 37.5, 75, and 150 mg per day (study 3) and the other evaluating venlafaxine hydrochloride extended-release capsules doses of 75 to 225 mg per day (study 4), showed that daily doses of 75 mg or higher were more effective than placebo on the HAM-A total, both the HAM-A anxiety and tension items, and the CGI scale during 6 months of treatment. While there was also evidence for superiority over placebo for the 37.5 mg per day dose, this dose was not as consistently effective as the higher doses.
Examination of gender subsets of the population studied did not reveal any differential responsiveness on the basis of gender.
| Study Number | Treatment Group | Primary Efficacy Measure: HAM-A Score | ||
| Mean Baseline Score (SD) | LS Mean Change from Baseline (SE)* | Placebo Subtracted Difference a(95% CI) | ||
| Study 1 | Venlafaxine Hydrochloride Extended-Release Capsules 75 mg | 24.7 | -11.1 (0.95) | -1.5 (-3.8, 0.8) |
| Venlafaxine Hydrochloride Extended-Release Capsules 150 mg | 24.5 | -11.7 (0.87) | -2.2 (-4.5, 0.1) | |
| Venlafaxine Hydrochloride Extended-Release Capsules 225 mg | 23.6 | -12.1 (0.81) | -2.6 (-4.9, -0.3) | |
| Placebo | 24.1 | -9.5 (0.85) | -- | |
| Study 2 | Venlafaxine Hydrochloride Extended-Release Capsules 75 mg | 23.7 | -10.6 (0.82) | -2.6 (-4.6, -0.5) |
| Venlafaxine Hydrochloride Extended-Release Capsules 150 mg | 23.0 | -9.8 (0.86) | -1.7 (-3.8, 0.3) | |
| Placebo | 23.7 | -8.0 (0.73) | -- | |
| Study 3 | Venlafaxine Hydrochloride Extended-Release Capsules 37.5 mg | 26.6 (0.4) | -13.8 | -2.8 (-5.1, -0.6) |
| Venlafaxine Hydrochloride Extended-Release Capsules 75 mg | 26.3 (0.4) | -15.5 | -4.6 (-6.9, -2.3) | |
| Venlafaxine Hydrochloride Extended-Release Capsules 150 mg | 26.3 (0.4) | -16.4 | -5.5 (-7.8, -3.1) | |
| Placebo | 26.7 (0.5) | -11.0 | -- | |
| Study 4 | Venlafaxine Hydrochloride Extended-Release Capsules 75 to 225 mg | 25.0 | -13.4 (0.79) | -4.7 (-6.6, -2.9) |
| Placebo | 24.9 | -8.7 (0.70) | -- | |
| SD=standard deviation; LS Mean=least-squares mean; CI=confidence interval. | ||||
| aDifference (drug minus placebo) in least-squares mean change from baseline. | ||||
| *Doses statistically significantly superior to placebo. |
17 Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Medication Guide).
16 How Supplied/storage and Handling
Product: 50090-7323
NDC: 50090-7323-0 30 CAPSULE, EXTENDED RELEASE in a BOTTLE
NDC: 50090-7323-1 90 CAPSULE, EXTENDED RELEASE in a BOTTLE
5.6 Activation of Mania Or Hypomania
In patients with bipolar disorder, treating a depressive episode with venlafaxine hydrochloride extended-release capsules or another antidepressant may precipitate a mixed/manic episode. Mania or hypomania was reported in venlafaxine hydrochloride extended-release capsules treated patients in the premarketing studies in MDD, SAD, and PD (see Table 2). Prior to initiating treatment with venlafaxine hydrochloride extended-release capsules, screen for any personal or family history of bipolar disorder, mania, or hypomania.
| Indication | Venlafaxine Hydrochloride Extended-Release Capsules | Placebo |
|---|---|---|
| MDD | 0.3 | 0.0 |
| GAD | 0.0 | 0.2 |
| SAD | 0.2 | 0.0 |
| PD | 0.1 | 0.0 |
2.1 General Administration Information
Administer venlafaxine hydrochloride extended-release capsules as a single dose with food, either in the morning or in the evening at approximately the same time each day [see Clinical Pharmacology (12.3)] . Swallow capsules whole with fluid. Do not divide, crush, chew, or place in water.
The capsule may also be administered by carefully opening the capsule and sprinkling the entire contents on a spoonful of applesauce. This drug/food mixture should be swallowed immediately without chewing and followed with a glass of water to ensure complete swallowing of the pellets (spheroids).
Warning: Suicidal Thoughts and Behaviors
Antidepressants increased the risk of suicidal thoughts and behavior in pediatric and young adult patients in short-term studies. Closely monitor all antidepressant-treated patients for clinical worsening, and emergence of suicidal thoughts and behaviors [see Warnings and Precautions (5.1)] . Venlafaxine hydrochloride extended-release capsules are not approved for use in pediatric patient s [see Use in Specific Populations (8.4)] .
2.4 Social Anxiety Disorder (social Phobia)
The recommended dose is 75 mg per day, administered in a single dose. There was no evidence that higher doses confer any additional benefit.
5.11 Appetite Changes in Pediatric Patients
Decreased appetite (reported as anorexia) was more commonly observed in venlafaxine hydrochloride extended-release capsules treated patients versus placebo-treated patients in the premarketing evaluation of venlafaxine hydrochloride extended-release capsules for MDD, GAD, and SAD (see Table 6).
Venlafaxine hydrochloride extended-release capsules are not approved for use in pediatric patients [see Use in Specific Populations (8.4)] .
| Indication
(Duration) |
Venlafaxine Hydrochloride Extended-Release Capsules Incidence | Discontinuation | Placebo Incidence | Discontinuation |
|---|---|---|---|---|
| MDD and GAD
(pooled, 8 weeks) |
10 | 0.0 | 3 | – |
| SAD
(16 weeks) |
22 | 0.7 | 3 | 0.0 |
|
aThe discontinuation rates for weight loss were 0.7% for patients receiving either venlafaxine hydrochloride extended-release capsules or placebo.
bVenlafaxine hydrochloride extended-release capsules are not approved for use in pediatric patients. |
5.12 Interstitial Lung Disease and Eosinophilic Pneumonia
Interstitial lung disease and eosinophilic pneumonia associated with venlafaxine therapy have been rarely reported. The possibility of these events should be considered in venlafaxine hydrochloride extended-release capsules-treated patients who present with progressive dyspnea, cough or chest discomfort. Such patients should undergo a prompt medical evaluation, and discontinuation of venlafaxine hydrochloride extended-release capsules should be considered.
14.3 Social Anxiety Disorder (also Known As Social Phobia)
The efficacy of venlafaxine hydrochloride extended-release capsules as a treatment for Social Anxiety Disorder (SAD) was established in four double-blind, parallel-group, 12-week, multicenter, placebo-controlled, flexible-dose studies (studies 1 to 4) and one double-blind, parallel-group, 6-month, placebo-controlled, fixed/flexible-dose study, which included doses in a range of 75 to 225 mg per day in adult outpatients meeting DSM-IV criteria for SAD (study 5).
In these five studies, venlafaxine hydrochloride extended-release capsules were statistically significantly more effective than placebo on change from baseline to endpoint on the Liebowitz Social Anxiety Scale (LSAS) total score. There was no evidence for any greater effectiveness of the 150 to 225 mg per day group compared to the 75 mg per day group in the 6-month study.
Examination of subsets of the population studied did not reveal any differential responsiveness on the basis of gender. There was insufficient information to determine the effect of age or race on outcome in these studies.
| Study Number | Treatment Group | Primary Efficacy Measure: LSAS Score | ||
|---|---|---|---|---|
| Mean Baseline Score (SD) | LS Mean Change from Baseline (SE) | Placebo Subtracted Difference a(95% CI) | ||
| Study 1 | Venlafaxine Hydrochloride Extended-Release Capsules (75 to 225 mg)
SD=standard deviation; SE=standard error; LS Mean=least-squares mean; CI=confidence interval.
aDifference (drug minus placebo) in least-squares mean change from baseline.
*Doses statistically significantly superior to placebo.
|
91.1 | -31.0 (2.22) | 11.2 (-5.3, -17.1) |
| Placebo | 86.7 | -19.9 (2.22) | - | |
| Study 2 | Venlafaxine Hydrochloride Extended-Release Capsules (75 to 225 mg) | 90.8 | -32.8 (2.69) | -10.7 (-3.7,-17.6) |
| Placebo | 87.4 | -22.1 (2.66) | - | |
| Study 3 | Venlafaxine Hydrochloride Extended-Release Capsules (75 to 225 mg) | 83.2 | -36.0 (2.35) | -16.9 (-22.6, -11.2) |
| Placebo | 83.6 | -19.1 (2.40) | -12.7 (-6.5, -19.0) | |
| Study 4 | Venlafaxine Hydrochloride Extended-Release Capsules (75 to 225 mg) | 86.2 | -35.0 (2.64) | -14.6 (-21.8, -7.4) |
| Placebo | 86.1 | -22.2 (2.47) | ||
| Study 5 | Venlafaxine Hydrochloride Extended-Release Capsules 75 mg | 91.8 | -38.1 (3.16) | -14.6 (-21.8, -7.4) |
| Venlafaxine Hydrochloride Extended-Release Capsules (150 to 225 mg) | 86.2 | -37.6 (3.05) | -14.1 (-21.3, -6.9) | |
| Placebo | 89.3 | -23.5 (3.08) |
2.9 Dosage Recommendations for Patients With Renal Impairment
Reduce the venlafaxine hydrochloride extended-release capsules total daily dose by 25% to 50% in patients with mild (CLcr 60 to 89 mL/min) or moderate (CLcr 30 to 59 mL/min) renal impairment. Reduce the total daily dose by 50% or more in patients undergoing hemodialysis or with severe renal impairment (CLcr<30 mL/min). Because there was much individual variability in clearance between patients with renal impairment, individualization of dosage is recommended in some patients [see Use in Specific Populations (8.7)] .
2.7 Switching Patients From venlafaxine Hydrochloride Tablets
Patients with depression who are currently being treated with venlafaxine hydrochloride tablets may be switched to venlafaxine hydrochloride extended-release capsules at the nearest equivalent dose (mg per day), e.g., 37.5 mg venlafaxine twice a day to 75 mg venlafaxine hydrochloride extended-release capsules once daily. However, individual dosage adjustments may be necessary.
2.8 Dosage Recommendations for Patients With Hepatic Impairment
Reduce the venlafaxine hydrochloride extended-release capsules total daily dose by 50% in patients with mild (Child-Pugh Class A) to moderate (Child-Pugh Class B) hepatic impairment. Reduce the total daily dose by 50% or more in patients with severe hepatic impairment (Child-Pugh Class C) or hepatic cirrhosis [see Use in Specific Populations (8.6)] .
5.1 Suicidal Thoughts and Behaviors in Adolescents and Young Adults
In pooled analyses of placebo-controlled trials of antidepressant drugs (SSRIs and other antidepressant classes) that included approximately 77,000 adult patients and 4,500 pediatric patients, the incidence of suicidal thoughts and behaviors in antidepressant-treated patients age 24 years and younger was greater than in placebo-treated patients. There was considerable variation in risk of suicidal thoughts and behaviors among drugs, but there was an increased risk identified in young patients for most drugs studied. There were differences in absolute risk of suicidal thoughts and behaviors across the different indications, with the highest incidence in patients with MDD. The drug-placebo differences in the number of cases of suicidal thoughts and behaviors per 1,000 patients treated are provided in Table 1.
| Age Range | Drug-Placebo Difference in Number of Patients of Suicidal Thoughts and Behaviors per 1,000 Patients Treated |
| Increases Compared to Placebo | |
| <18 years old | 14 additional patients |
| 18 to 24 years old | 5 additional patients |
| Decreases Compared to Placebo | |
| 25 to 64 years old | 1 fewer patient |
| ≥65 years old | 6 fewer patients |
| * Venlafaxine hydrochloride extended-release capsules are not approved in pediatric patients. |
It is unknown whether the risk of suicidal thoughts and behaviors in children, adolescents, and young adults extends to longer-term use, i.e., beyond four months. However, there is substantial evidence from placebo-controlled maintenance trials in adults with MDD that antidepressants delay the recurrence of depression and that depression itself is a risk factor for suicidal thoughts and behaviors.
Monitor all antidepressant-treated patients for any indication for clinical worsening and emergence of suicidal thoughts and behaviors, especially during the initial few months of drug therapy, and at times of dosage changes. Counsel family members or caregivers of patients to monitor for changes in behavior and to alert the healthcare provider. Consider changing the therapeutic regimen, including possibly discontinuing venlafaxine hydrochloride extended-release capsules, in patients whose depression is persistently worse, or who are experiencing emergent suicidal thoughts or behaviors.
2.10 Discontinuing Treatment With venlafaxine Hydrochloride Extended Release Capsules
A gradual reduction in the dose, rather than abrupt cessation, is recommended when discontinuing therapy with venlafaxine hydrochloride extended-release capsules. In clinical studies with venlafaxine hydrochloride extended-release capsules, tapering was achieved by reducing the daily dose by 75 mg at one-week intervals. Individualization of tapering may be necessary. In some patients, discontinuation may need to occur over a period of several months [see Warnings and Precautions (5.7)] .
2.11 Switching Patients to Or From A Monoamine Oxidase Inhibitor (maoi) Antidepressant
At least 14 days must elapse between discontinuation of an MAOI antidepressant and initiation of venlafaxine hydrochloride extended-release capsules. In addition, at least 7 days must elapse after stopping venlafaxine hydrochloride extended-release capsules before starting an MAOI antidepressant [see Contraindications (4), Warnings and Precautions (5.2), and Drug Interactions (7.1)].
2.6 Screen for Bipolar Disorder Prior to Starting venlafaxine Hydrochloride Extended Release Capsules
Prior to initiating treatment with venlafaxine hydrochloride extended-release capsules, screen patients for a personal or family history of bipolar disorder, mania, or hypomania [see Warnings and Precautions( 5.6)] .
7.1 Drugs Having Clinically Important Interactions With venlafaxine Hydrochloride Extended Release Capsules
| Monoamine Oxidase Inhibitors (MAOI) | |
| Clinical Impact | The concomitant use of SNRIs, including venlafaxine hydrochloride extended-release capsules, with MAOIs increases the risk of serotonin syndrome. |
| Intervention | Concomitant use of venlafaxine hydrochloride extended-release capsules is contraindicated in patients taking MAOIs, including MAOIs such as linezolid or intravenous methylene blue [see Dosage and Administration (2.11), Contraindications (4)and Warnings and Precautions (5.2)]. |
| Other Serotonergic Drugs | |
| Clinical Impact | Concomitant use of venlafaxine hydrochloride extended-release capsules with other serotonergic drugs (including other SNRIs, SSRIs, triptans, tricyclic antidepressants, opioids, lithium, buspirone, amphetamines, tryptophan, and St. John's Wort) increases the risk of serotonin syndrome. |
| Intervention | Monitor for symptoms of serotonin syndrome when venlafaxine hydrochloride extended-release capsules is used concomitantly with other drugs that may affect the serotonergic neurotransmitter systems. If serotonin syndrome occurs, consider discontinuation of venlafaxine hydrochloride extended-release capsules and/or concomitant serotonergic drugs [see Dosage and Administration (2.11)and Warnings and Precautions (5.2)]. |
| Drugs that Interfere with Hemostasis | |
| Clinical Impact | Concomitant use of venlafaxine hydrochloride extended-release capsules with an antiplatelet or anticoagulant drug may potentiate the risk of bleeding. This may be due to the effect of venlafaxine hydrochloride extended-release capsules on the release of serotonin by platelets. |
| Intervention | Closely monitor for bleeding for patients receiving an antiplatelet or anticoagulant drug when venlafaxine hydrochloride extended-release capsules are initiated or discontinued [see Warnings and Precautions (5.4)] . |
| Effect of CYP3A Inhibitors | |
| Clinical Impact | Concomitant use of a CYP3A inhibitor increases the C maxand AUC of venlafaxine and O-desmethylvenlafaxine (ODV) [see Clinical Pharmacology (12.3)] , which may increase the risk of toxicity of venlafaxine hydrochloride extended-release capsules. |
| Intervention | Consider reducing the dose of venlafaxine hydrochloride extended-release capsules. |
| CYP2D6 Substrates | |
| Clinical Impact | Concomitant use of venlafaxine hydrochloride extended-release capsules increases C maxand AUC of a CYP2D6 substrate, which may increase the risk of toxicity of the CYP2D6 substrate [see Clinical Pharmacology (12.3)] . |
| Intervention | Consider reduction in dose of concomitant CYP2D6 substrates. |
Structured Label Content
Section 42229-5 (42229-5)
Weight Changes
The average change in body weight and incidence of weight loss (percentage of patients who lost 3.5% or more) in the placebo-controlled pediatric studies in MDD, GAD, and SAD are shown in Tables 3 and 4.
| Indication
(Duration) |
Venlafaxine Hydrochloride Extended-Release Capsules | Placebo |
|---|---|---|
| MDD and GAD
(4 pooled studies, 8 weeks) |
-0.45 (n=333) | +0.77 (n=333) |
| SAD
(16 weeks) |
-0.75 (n=137) | +0.76 (n=148) |
| aVenlafaxine hydrochloride extended-release capsules are not approved for use in pediatric patients. |
| Indication
(Duration) |
Venlafaxine Hydrochloride Extended-Release Capsules | Placebo |
|---|---|---|
| MDD and GAD
(4 pooled studies, 8 weeks) |
18 b(n=333) | 3.6 (n=333) |
| SAD
(16 weeks) |
47 b(n=137) | 14 (n=148) |
|
aVenlafaxine hydrochloride extended-release capsules are not approved for use in pediatric patients.
bp<0.001 versus placebo |
Weight loss was not limited to patients with anorexia [see Warnings and Precautions (5.11)] .
The risks associated with longer term venlafaxine hydrochloride extended-release capsules use were assessed in an open-label MDD study of children and adolescents who received venlafaxine hydrochloride extended-release capsules for up to six months. The children and adolescents in the study had increases in weight that were less than expected, based on data from age- and sex-matched peers. The difference between observed weight gain and expected weight gain was larger for children (<12 years old) than for adolescents (≥12 years old).
Venlafaxine hydrochloride extended-release capsules are not approved for use in pediatric patients [ Use in Specific Populations (8.4)] .
Section 42231-1 (42231-1)
|
Dispense with Medication Guide available at: www.epic-pharma.com/medguide/Venlafaxine-Hydrochloride-ER-Capsule.pdf
MEDICATION GUIDE
Extended-Release Capsules |
||||
|
What is the most important information I should know about venlafaxine hydrochloride extended-release capsules?
Venlafaxine hydrochloride extended-release capsules may cause serious side effects, including:
|
||||
|
|
|||
|
What are venlafaxine hydrochloride extended-release capsules?
Venlafaxine hydrochloride extended-release capsules are a prescription medicine used to treat adults with:
|
||||
Do not take venlafaxine hydrochloride extended-release capsules if you:
Ask your healthcare provider or pharmacist if you are not sure if you take an MAOI, including MAOIs such as linezolid or intravenous methylene blue.
|
||||
Before taking venlafaxine hydrochloride extended-release capsules tell your healthcare provider about all your medical conditions, including if you:
Tell your healthcare provider about all the medicines you take,including prescription and over-the-counter medicines, vitamins, and herbal supplements.
Ask your healthcare provider if you are not sure if you are taking any of these medicines. Your healthcare provider can tell you if it is safe to take venlafaxine hydrochloride extended-release capsules with your other medicines.
|
||||
How should I take venlafaxine hydrochloride extended-release capsules?
|
||||
What should I avoid while taking venlafaxine hydrochloride extended-release capsules?
|
||||
|
What are the possible side effects of venlafaxine hydrochloride extended-release capsules?
venlafaxine hydrochloride extended-release capsules may cause serious side effects, including:
|
||||
|
|
|||
|
||||
|
|
|||
|
||||
|
|
|
||
|
||||
|
Your healthcare provider may tell you to stop taking venlafaxine hydrochloride extended-release capsules if you develop serious side effects during treatment with venlafaxine hydrochloride extended-release capsules. |
||||
| The most common side effects of venlafaxine hydrochloride extended-release capsules include: | ||||
|
|
|||
| These are not all the possible side effects of venlafaxine hydrochloride extended-release capsules.
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
||||
How should I store venlafaxine hydrochloride extended-release capsules?
|
||||
|
General information about the safe and effective use of venlafaxine hydrochloride extended-release capsules.
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use venlafaxine hydrochloride extended-release capsules for a condition for which it was not prescribed. Do not give venlafaxine hydrochloride extended-release capsules to other people, even if they have the same symptoms that you have. It may harm them. You can ask your healthcare provider or pharmacist for information about venlafaxine hydrochloride extended-release capsules that is written for healthcare professionals. |
||||
|
What are the ingredients in venlafaxine hydrochloride extended-release capsules? Active ingredient:venlafaxine hydrochloride, USP Inactive ingredients: empty hard gelatin capsules, ethylcellulose, hypromellose, povidone, sugar spheres (composed of corn starch and sucrose) and talc. The 37.5 mg capsule shell contains red iron oxide, gelatin, and titanium dioxide. The 75 mg capsule shell contains black iron oxide, red iron oxide, gelatin, and titanium dioxide. The 150 mg capsule shell contains FD&C blue 1, FD&C red 3, FD&C yellow 6, gelatin, and titanium dioxide. The imprinting ink contains black iron oxide, potassium hydroxide and shellac. The brands listed are the registered trademarks of their respective owners. This Medication Guide has been approved by the U.S. Food and Drug Administration for all antidepressants.
Distributed by:
ZLN034
|
9.2 Abuse
Abuse is the intentional, non-therapeutic use of a drug, even once, for its desirable psychological or physiological effects.
While venlafaxine has not been systematically studied in clinical studies for its potential for abuse, there was no indication of drug-seeking behavior in the clinical studies. However, it is not possible to predict on the basis of premarketing experience the extent to which a CNS-active drug will be misused, diverted, and/or abused once marketed. Consequently, providers should carefully evaluate patients for history of drug abuse and follow such patients closely, observing them for signs of misuse or abuse of venlafaxine (e.g., development of tolerance, incrementation of dose, drug-seeking behavior).
5.8 Seizures
Cases of seizure have been reported with venlafaxine therapy. Venlafaxine hydrochloride extended-release capsules have not been systematically evaluated in patients with seizure disorder. Venlafaxine hydrochloride extended-release capsules should be prescribed with caution in patients with a seizure disorder.
11 Description (11 DESCRIPTION)
Venlafaxine Hydrochloride Extended-Release Capsules, USP are an extended-release capsule for once-a-day oral administration that contains venlafaxine hydrochloride, a serotonin and norepinephrine reuptake inhibitor (SNRI).
Venlafaxine is designated (R/S)-1-[2-(dimethylamino)-1-(4-methoxyphenyl)ethyl] cyclohexanol hydrochloride or (±)-1-[α-[(dimethylamino)methyl]-p-methoxybenzyl] cyclohexanol hydrochloride and has the empirical formula of C 17H 27NO 2HCl. Its molecular weight is 313.86. The structural formula is shown as follows:
Venlafaxine hydrochloride is a white to off-white crystalline solid, with a solubility of 572 mg/mL in water (adjusted to ionic strength of 0.2 M with sodium chloride). Its octanol: water (0.2 M sodium chloride) partition coefficient is 0.43.
Drug release is controlled by diffusion through the coating membrane on the spheroids and is not pH-dependent. Capsules contain venlafaxine hydrochloride equivalent to 37.5 mg, 75 mg, or 150 mg venlafaxine. Inactive ingredients consist of empty hard gelatin capsules, ethylcellulose, hypromellose, povidone, sugar spheres (composed of corn starch and sucrose) and talc.
The 37.5 mg capsule shell contains red iron oxide, gelatin, and titanium dioxide. The 75 mg capsule shell contains black iron oxide, red iron oxide, gelatin, and titanium dioxide. The 150 mg capsule shell contains FD&C blue 1, FD&C red 3, FD&C yellow 6, gelatin, and titanium dioxide.
The imprinting ink contains black iron oxide, potassium hydroxide and shellac.
FDA approved dissolution test specifications differ from USP.
9.3 Dependence
Physical dependence is a state that develops as a result of physiological adaptation in response to repeated drug use, manifested by withdrawal signs and symptoms after abrupt discontinuation or a significant dose reduction of a drug.
In vitrostudies revealed that venlafaxine has virtually no affinity for opiate, benzodiazepine, phencyclidine (PCP), or N-methyl-D-aspartic acid (NMDA) receptors.
Venlafaxine was not found to have any significant CNS stimulant activity in rodents. In primate drug discrimination studies, venlafaxine showed no significant stimulant or depressant abuse liability.
Discontinuation effects have been reported in patients receiving venlafaxine [see Dosage and Administration (2.10)and Warnings and Precautions (5.7)] .
5.9 Hyponatremia
Hyponatremia can occur as a result of treatment with SNRIs, including venlafaxine hydrochloride extended-release capsules. In many cases, the hyponatremia appears to be the result of the Syndrome of Inappropriate Antidiuretic Hormone (SIADH) secretion. Cases with serum sodium lower than 110 mmol/L have been reported. Elderly patients may be at greater risk of developing hyponatremia with SNRIs. Also, patients taking diuretics, or those who are otherwise volume-depleted, may be at greater risk [see Use in Specific Populations (8.5)and Clinical Pharmacology (12.3)] . Consider discontinuation of venlafaxine hydrochloride extended-release capsules in patients with symptomatic hyponatremia, and institute appropriate medical intervention.
Signs and symptoms of hyponatremia include headache, difficulty concentrating, memory impairment, confusion, weakness, and unsteadiness, which may lead to falls. Signs and symptoms associated with more severe and/or acute cases have included hallucination, syncope, seizure, coma, respiratory arrest, and death.
8.4 Pediatric Use
Safety and effectiveness of venlafaxine hydrochloride extended-release capsules in pediatric patients have not been established.
Two placebo-controlled trials in 766 pediatric patients with MDD and two placebo-controlled trials in 793 pediatric patients with GAD have been conducted with venlafaxine hydrochloride extended-release capsules, and the data were not sufficient to support use in pediatric patients.
In the studies conducted in pediatric patients ages 6 to 17 years, the occurrence of blood pressure and cholesterol increases was considered to be clinically relevant in pediatric patients and was similar to that observed in adult patients [see Warnings and Precautions (5.3), Adverse Reactions (6.1)] . The following adverse reactions were also observed in pediatric patients: abdominal pain, agitation, dyspepsia, ecchymosis, epistaxis, and myalgia.
Although no studies have been designed to primarily assess venlafaxine hydrochloride extended-release capsules' impact on the growth, development, and maturation of children and adolescents, the studies that have been done suggest that venlafaxine hydrochloride extended-release capsules may adversely affect weight and height [see Warnings and Precautions (5.10, 5.11)]. Decreased appetite and weight loss were observed in placebo-controlled studies of pediatric patients 6 to 17 years.
In pediatric clinical studies, the adverse reaction, suicidal ideation, was observed. Antidepressants increased the risk of suicidal thoughts and behaviors in pediatric patients [see Boxed Warning, Warnings and Precautions (5.1)] .
8.5 Geriatric Use
The percentage of patients in clinical studies for venlafaxine hydrochloride extended-release capsules for MDD, GAD, SAD, and PD who were 65 years of age or older are shown in Table 16.
| Indication | Venlafaxine Hydrochloride Extended-Release Capsules |
|---|---|
| MDD | 4 (14/357) |
| GAD | 6 (77/1,381) |
| SAD | 1 (10/819) |
| PD | 2 (16/1,001) |
| aIn addition, in the premarketing assessment of venlafaxine hydrochloride tablets (immediate-release), 12% (357/2,897) of patients were ≥65 years of age. |
No overall differences in effectiveness or safety were observed between geriatric patients and younger patients, and other reported clinical experience generally has not identified differences in response between the elderly and younger patients. However, greater sensitivity of some older individuals cannot be ruled out. SSRIs and SNRIs, including venlafaxine hydrochloride extended-release capsules, have been associated with cases of clinically significant hyponatremia in elderly patients, who may be at greater risk for this adverse event [see Warnings and Precautions (5.9)] .
The pharmacokinetics of venlafaxine and ODV are not substantially altered in the elderly [see Clinical Pharmacology (12.3)] (see Figure 1). No dose adjustment is recommended for the elderly on the basis of age alone, although other clinical circumstances, some of which may be more common in the elderly, such as renal or hepatic impairment, may warrant a dose reduction [see Dosage and Administration (2.8, 2.9)] .
2.5 Panic Disorder
The recommended starting dose is 37.5 mg per day of venlafaxine hydrochloride extended-release capsules for 7 days. Patients not responding to 75 mg per day may benefit from dose increases to a maximum of approximately 225 mg per day. Dose increases should be in increments of up to 75 mg per day, as needed, and should be made at intervals of not less than 7 days.
14.4 Panic Disorder
The efficacy of venlafaxine hydrochloride extended-release capsules as a treatment for Panic Disorder (PD) was established in two double-blind, 12-week, multicenter, placebo-controlled studies in adult outpatients meeting DSM-IV criteria for PD, with or without agoraphobia. Patients received fixed doses of 75 or 150 mg per day in one study (study 1) and 75 or 225 mg per day in the other study (study 2).
Efficacy was assessed on the basis of outcomes in three variables: (1) percentage of patients free of full-symptom panic attacks on the Panic and Anticipatory Anxiety Scale (PAAS); (2) mean change from baseline to endpoint on the Panic Disorder Severity Scale (PDSS) total score; and (3) percentage of patients rated as responders (much improved or very much improved) on the Clinical Global Impressions (CGI) Improvement scale. In these two studies, venlafaxine hydrochloride extended-release capsules were statistically significantly more effective than placebo (for each fixed dose) on all three endpoints, but a dose-response relationship was not clearly established.
Examination of subsets of the population studied did not reveal any differential responsiveness on the basis of gender. There was insufficient information to determine the effect of age or race on outcome in these studies.
In a longer term study (study 3), adult outpatients meeting DSM-IV criteria for PD who had responded during a 12-week open phase with venlafaxine hydrochloride extended-release capsules (75 to 225 mg per day) were randomly assigned to continue the same venlafaxine hydrochloride extended-release capsules dose (75, 150, or 225 mg) or switch to placebo for observation for relapse under double-blind conditions. Response during the open phase was defined as ≤1 full-symptom panic attack per week during the last 2 weeks of the open phase and a CGI Improvement score of 1 (very much improved) or 2 (much improved). Relapse during the double-blind phase was defined as having 2 or more full-symptom panic attacks per week for 2 consecutive weeks or having discontinued due to loss of effectiveness as determined by the investigators during the study. Randomized patients were in response status for a mean time of 34 days prior to being randomized. In the randomized phase following the 12-week open-label period, patients receiving continued venlafaxine hydrochloride extended-release capsules experienced a statistically significantly longer time to relapse.
| Study Treatment Group Number | Primary Efficacy Measure: Whether Free of Full-symptom Panic Attacks | |||
|---|---|---|---|---|
| Percent of patients Free of Full Symptom Panic Attack | Adjusted Odds Ratio ato Placebo | Adjusted Odds Ratio a 95% Confidence Interval | ||
| Study 1 | Venlafaxine Hydrochloride Extended-Release Capsules 75 mg* | 54.1% (85/157) | 2. 268 | (1.43, 3.59) |
| Venlafaxine Hydrochloride Extended-Release Capsules 150 mg* | 61.4% (97/158) | 3.035 | (1.91, 4.82) | |
| Placebo | 34.4% (53/154) | -- | -- | |
| Study 2 | Venlafaxine Hydrochloride Extended-Release Capsules 75 mg* | 64.1% (100/156) | 2.350 | (1.46, 3.78) |
| Venlafaxine Hydrochloride Extended-Release Capsules 225 mg* | 70.0% (112/160) | 2.890 | (1.80, 4.64) | |
| Placebo | 46.5% (73/157) | -- | -- |
aOdds ratio (drug to placebo) in terms of probability of free of full-symptom panic attacks based on logistic regression model.
95% CI: 95% confidence interval without adjusting for multiple dose arms.
*Doses statistically significantly superior to placebo.
4 Contraindications (4 CONTRAINDICATIONS)
Venlafaxine Hydrochloride Extended-Release Capsules are contraindicated in patients:
- with known hypersensitivity to venlafaxine hydrochloride, desvenlafaxine succinate or to any excipients in the formulation [see Adverse Reactions (6.2)] .
- taking, or within 14 days of stopping, MAOIs (including the MAOIs linezolid and intravenous methylene blue) because of the risk of serotonin syndrome [see Dosage and Administration (2.11), Warnings and Precautions (5.2), and Drug Interactions (7.1)] .
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following adverse reactions are discussed in more detail in other sections of the labeling:
- Hypersensitivity [see Contraindications (4)]
- Suicidal Thoughts and Behaviors in Adolescents and Young Adults [see Warnings and Precautions (5.1)]
- Serotonin Syndrome [see Warnings and Precautions (5.2)]
- Elevated Blood Pressure [see Warnings and Precautions (5.3)]
- Increased Risk of Bleeding [see Warnings and Precautions (5.4)]
- Angle-Closure Glaucoma [see Warnings and Precautions (5.5)]
- Activation of Mania/Hypomania [see Warnings and Precautions (5.6)]
- Discontinuation Syndrome [see Warnings and Precautions (5.7)]
- Seizure [see Warnings and Precautions (5.8)]
- Hyponatremia [see Warnings and Precautions (5.9)]
- Weight and Height Changes in Pediatric Patients [see Warnings and Precautions (5.10)]
- Appetite Changes in Pediatric Patients [see Warnings and Precautions (5.11)]
- Interstitial Lung Disease and Eosinophilic Pneumonia [see Warnings and Precautions (5.12)]
- Sexual Dysfunction [see Warnings and Precautions (5.13)]
8.7 Renal Impairment
Dosage adjustment is recommended in patients with mild (CLcr=60 to 89 mL/min), moderate (CLcr=30 to 59 mL/min), or severe (CLcr<30 mL/min) renal impairment, and in patients undergoing hemodialysis [see Dosage and Administration (2.9)and Clinical Pharmacology (12.3)].
12.2 Pharmacodynamics
In vitrostudies have demonstrated that venlafaxine and its active metabolite, O-desmethylvenlafaxine (ODV), are potent and selective inhibitors of neuronal serotonin and norepinephrine reuptake and weak inhibitors of dopamine reuptake. Venlafaxine and ODV have no significant affinity for muscarinic-cholinergic, H 1-histaminergic, or α 1-adrenergic receptors in vitro. Pharmacologic activity at these receptors is hypothesized to be associated with the various anticholinergic, sedative, and cardiovascular effects seen with other psychotropic drugs. Venlafaxine and ODV do not possess monoamine oxidase (MAO) inhibitory activity.
12.3 Pharmacokinetics
Venlafaxine and ODV steady-state concentrations are reached within 3 days. Venlafaxine and ODV exhibited linear kinetics over the dosage range of 75 to 450 mg per day (0.33 to 2 times the maximum recommended dosage). Time of administration (AM versus PM) did not affect the pharmacokinetics of venlafaxine and ODV from the 75 mg venlafaxine hydrochloride extended-release capsules.
5.2 Serotonin Syndrome
Serotonin-norepinephrine reuptake inhibitors (SNRIs), including venlafaxine hydrochloride extended-release capsules, can precipitate serotonin syndrome, a potentially life-threatening condition. The risk is increased with concomitant use of other serotonergic drugs (including triptans, tricyclic antidepressants, fentanyl, lithium, tramadol, meperidine, methadone, tryptophan, buspirone, amphetamines, and St. John’s Wort) and with drugs that impair metabolism of serotonin, i.e., MAOIs [see Contraindications (4), Drug Interactions (7.1)] . Serotonin syndrome can also occur when these drugs are used alone.
Serotonin syndrome signs and symptoms may include mental status changes (e.g., agitation, hallucinations, delirium, coma), autonomic instability (e.g., tachycardia, labile blood pressure, dizziness, diaphoresis, flushing, hyperthermia), neuromuscular symptoms (e.g., tremor, rigidity, myoclonus, hyperreflexia, incoordination), seizures, and gastrointestinal symptoms (e.g., nausea, vomiting, diarrhea).
The concomitant use of venlafaxine hydrochloride extended-release capsules with MAOIs is contraindicated. In addition, do not initiate venlafaxine hydrochloride extended-release capsules in a patient being treated with MAOIs such as linezolid or intravenous methylene blue. No reports involved the administration of methylene blue by other routes (such as oral tablets or local tissue injection). If it is necessary to initiate treatment with an MAOI such as linezolid or intravenous methylene blue in a patient taking venlafaxine hydrochloride extended-release capsules, discontinue venlafaxine hydrochloride extended-release capsules before initiating treatment with the MAOI [see Contraindications (4), Drug Interactions (7.1)] .
Monitor all patients taking venlafaxine hydrochloride extended-release capsules for the emergence of serotonin syndrome. Discontinue treatment with venlafaxine hydrochloride extended-release capsules and any concomitant serotonergic agents immediately if the above symptoms occur, and initiate supportive symptomatic treatment. If concomitant use of venlafaxine hydrochloride extended-release capsules with other serotonergic drugs is clinically warranted, inform patients of the increased risk for serotonin syndrome and monitor for symptoms.
8.6 Hepatic Impairment
Dosage adjustment is recommended in patients with mild (Child-Pugh Class A), moderate (Child-Pugh Class B), or severe (Child-Pugh Class C) hepatic impairment or hepatic cirrhosis [see Dosage and Administration (2.8)and Clinical Pharmacology (12.3)] .
1 Indications and Usage (1 INDICATIONS AND USAGE)
Venlafaxine Hydrochloride Extended-Release Capsules, USP are indicated in adults for the treatment of:
- Major Depressive Disorder (MDD) [see Clinical Studies (14.1)]
- Generalized Anxiety Disorder (GAD) [see Clinical Studies (14.2)]
- Social Anxiety Disorder (SAD) [see Clinical Studies (14.3)]
- Panic Disorder (PD) [see Clinical Studies (14.4)]
5.13 Sexual Dysfunction
Use of SNRIs, including venlafaxine hydrochloride extended-release capsules, may cause symptoms of sexual dysfunction [ see Adverse Reactions (6.1)] . In male patients, SNRI use may result in ejaculatory delay or failure, decreased libido, and erectile dysfunction. In female patients, SNRI use may result in decreased libido and delayed or absent orgasm. It is important for prescribers to inquire about sexual function prior to initiation of venlafaxine hydrochloride extended-release capsules and to inquire specifically about changes in sexual function during treatment, because sexual function may not be spontaneously reported. When evaluating changes in sexual function, obtaining a detailed history (including timing of symptom onset) is important because sexual symptoms may have other causes, including the underlying psychiatric disorder. Discuss potential management strategies to support patients in making informed decisions about treatment.
12.1 Mechanism of Action
The mechanism of action of venlafaxine in the treatment of MDD, GAD, SAD, and PD is unclear, but is thought to be related to the potentiation of serotonin and norepinephrine in the central nervous system, through inhibition of their reuptake.
9.1 Controlled Substance
Venlafaxine hydrochloride extended-release capsules contain venlafaxine which is not a controlled substance.
Venlafaxine Hydrochloride
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Serotonin Syndrome: Increased risk when co-administered with other serotonergic agents, but also when taken alone. If it occurs, discontinue venlafaxine hydrochloride extended-release capsules and serotonergic agents and initiate supportive treatment ( 4, 5.2, 7.1).
- Elevated Blood Pressure: Control hypertension before initiating treatment. Monitor blood pressure regularly during treatment ( 5.3).
- Increased Risk of Bleeding: Concomitant use of aspirin, NSAIDs, other antiplatelet drugs, warfarin, and other anticoagulants may increase risk ( 5.4).
- Angle-Closure Glaucoma: Angle-closure glaucoma has occurred in patients with untreated anatomically narrow angles, treated with antidepressants ( 5.5).
- Activation of Mania or Hypomania: Screen patients for bipolar disorder ( 5.6).
- Discontinuation Syndrome: Taper dose and monitor for discontinuation symptoms ( 5.7).
- Seizures: Can occur. Use cautiously in patients with seizure disorder ( 5.8).
- Hyponatremia: Can occur in association with SIADH ( 5.9).
- Interstitial Lung Disease and Eosinophilic Pneumonia: Can occur ( 5.12).
- Sexual Dysfunction: Venlafaxine hydrochloride extended-release capsules may cause symptoms of sexual dysfunction ( 5.13).
5.5 Angle Closure Glaucoma (5.5 Angle-Closure Glaucoma)
The pupillary dilation that occurs following use of many antidepressant drugs including venlafaxine hydrochloride extended-release capsules may trigger an angle closure attack in a patient with anatomically narrow angles who does not have a patent iridectomy. Avoid use of antidepressants, including venlafaxine hydrochloride extended-release capsules, in patients with untreated anatomically narrow angles.
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
| Indication | Starting Dose | Target Dose | Maximum Dose |
|---|---|---|---|
| MDD ( 2.2) | 37.5 to 75 mg/day | 75 mg/day | 225 mg/day |
| GAD ( 2.3) | 37.5 to75 mg/day | 75 mg/day | 225 mg/day |
| SAD ( 2.4) | 75 mg/day | 75 mg/day | 75 mg/day |
| PD ( 2.5) | 37.5 mg/day | 75 mg/day | 225 mg/day |
- Take once daily with food. Capsules should be taken whole; do not divide, crush, chew, or dissolve ( 2.1).
- When discontinuing treatment, reduce the dose gradually ( 2.10, 5.7).
- Renal impairment: reduce the total daily dose by 25% to 50% in patients with renal impairment. Reduce the total daily dose by 50% or more in patients undergoing dialysis or with severe renal impairment ( 2.9).
- Hepatic impairment: reduce the daily dose by 50% in patients with mild to moderate hepatic impairment. In patients with severe hepatic impairment or hepatic cirrhosis, it may be necessary to reduce the dose by more than 50% ( 2.8).
5.3 Elevated Blood Pressure
In controlled trials, there were dose-related increases in systolic and diastolic blood pressure, as well as cases of sustained hypertension [see Adverse Reactions (6.1)].
Monitor blood pressure before initiating treatment with venlafaxine hydrochloride extended-release capsules and regularly during treatment. Control pre-existing hypertension before initiating treatment with venlafaxine hydrochloride extended-release capsules. Use caution in treating patients with pre-existing hypertension or cardiovascular or cerebrovascular conditions that might be compromised by increases in blood pressure. Sustained blood pressure elevation can lead to adverse outcomes. Cases of elevated blood pressure requiring immediate treatment have been reported with venlafaxine hydrochloride extended-release capsules. Consider dose reduction or discontinuation of treatment for patients who experience a sustained increase in blood pressure.
Across all clinical studies with venlafaxine hydrochloride tablets, 1.4% of patients in the venlafaxine hydrochloride extended-release capsules treated groups experienced a ≥15 mm Hg increase in supine diastolic blood pressure (SDBP) ≥105 mm Hg, compared to 0.9% of patients in the placebo groups. Similarly, 1% of patients in the venlafaxine hydrochloride extended-release capsules treated groups experienced a ≥20 mm Hg increase in supine systolic blood pressure (SSBP) with blood pressure ≥180 mm Hg, compared to 0.3% of patients in the placebo groups [see Adverse Reactions (6.1)] . Treatment with venlafaxine hydrochloride extended-release capsules was associated with sustained hypertension defined as SDBP ≥90 mm Hg and ≥10 mm Hg above baseline for three consecutive on-therapy visits [see Adverse Reactions (6.1)] . An insufficient number of patients received mean doses of venlafaxine hydrochloride extended-release capsules over 300 mg per day in clinical studies to fully evaluate the incidence of sustained increases in blood pressure at these higher doses.
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Venlafaxine Hydrochloride Extended-Release Capsules, USP are available in the following strengths:
- 37.5 mg capsules, opaque flesh cap and opaque flesh body, imprinted with "YH" on cap and "130" on body in black ink.
- 75 mg capsules, opaque light grey cap and opaque flesh body, imprinted with "YH" on cap and "129" on body in black ink.
- 150 mg capsules, opaque caramel cap and body, imprinted with "YH" on cap and "128" on body in black ink.
5.7 Discontinuation Syndrome
Discontinuation symptoms have been systematically evaluated in patients taking venlafaxine, including prospective analyses of clinical studies in GAD and retrospective surveys of studies in MDD and SAD. Abrupt discontinuation or dose reduction of venlafaxine at various doses has been found to be associated with the appearance of new symptoms, the frequency of which increased with increased dose level and with longer duration of treatment. Reported symptoms include agitation, anorexia, anxiety, confusion, impaired coordination and balance, diarrhea, dizziness, dry mouth, dysphoric mood, fasciculation, fatigue, flu-like symptoms, headaches, hypomania, insomnia, nausea, nervousness, nightmares, sensory disturbances (including shock-like electrical sensations), somnolence, sweating, tremor, vertigo, and vomiting.
There have been postmarketing reports of serious discontinuation symptoms which can be protracted and severe. Completed suicide, suicidal thoughts, aggression and violent behavior have been observed in patients during reduction in venlafaxine hydrochloride extended-release capsules dosage, including during discontinuation. Other postmarketing reports describe visual changes (such as blurred vision or trouble focusing) and increased blood pressure after stopping or reducing the dose of venlafaxine hydrochloride extended-release capsules.
During marketing of venlafaxine hydrochloride extended-release capsules, other SNRIs, and SSRIs, there have been reports of adverse events occurring upon discontinuation of these drugs, particularly when abrupt, including the following: irritability, lethargy, emotional lability, tinnitus, and seizures.
Patients should be monitored for these symptoms when discontinuing treatment with venlafaxine hydrochloride extended-release capsules. A gradual reduction in the dose, rather than abrupt cessation, is recommended. If intolerable symptoms occur following a decrease in the dose or upon discontinuation of treatment, then resuming the previously prescribed dose may be considered. Subsequently, the healthcare provider may continue decreasing the dose, but at a more gradual rate. In some patients, discontinuation may need to occur over a period of several months [see Dosage and Administration (2.10)] .
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of venlafaxine hydrochloride extended-release capsules. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Body as a Whole
– Anaphylaxis, angioedema
Cardiovascular System
– QT prolongation, ventricular fibrillation, ventricular tachycardia (including torsade de pointes), takotsubo cardiomyopathy
Digestive System
– Pancreatitis
Hemic/Lymphatic System
– Mucous membrane bleeding
[see
Warnings and Precautions (5.4)]
, blood dyscrasias (including agranulocytosis, aplastic anemia, neutropenia and pancytopenia), prolonged bleeding time, thrombocytopenia
Metabolic/Nutritional
– Hyponatremia
[see
Warnings and Precautions (5.9)]
, Syndrome of Inappropriate Antidiuretic Hormone (SIADH) secretion
[see
Warnings and Precautions (5.9)]
, abnormal liver function tests, hepatitis, prolactin increased
Musculoskeletal
– Rhabdomyolysis
Nervous System
– Neuroleptic Malignant Syndrome (NMS)
[see
Warnings and Precautions (5.2)]
, serotonergic syndrome
[see
Warnings and Precautions (5.2)]
, delirium, extrapyramidal reactions (including dystonia and dyskinesia), impaired coordination and balance, tardive dyskinesia
Respiratory, Thoracic and Mediastinal Disorders
– Anosmia, dyspnea, hyposmia, interstitial lung disease, pulmonary eosinophilia
[see
Warnings and Precautions (5.12)]
Skin and Appendages
– Stevens-Johnson syndrome, toxic epidermal necrolysis, erythema multiforme
Special Senses
– Angle-closure glaucoma
[see
Warnings and Precautions (5.5)]
2.2 Major Depressive Disorder
For most patients, the recommended starting dose for venlafaxine hydrochloride extended-release capsules is 75 mg per day, administered in a single dose. For some patients, it may be desirable to start at 37.5 mg per day for 4 to 7 days to allow new patients to adjust to the medication before increasing to 75 mg per day. Patients not responding to the initial 75 mg per day dose may benefit from dose increases to a maximum of 225 mg per day. Dose increases should be in increments of up to 75 mg per day, as needed, and should be made at intervals of not less than 4 days. In the clinical studies establishing efficacy, upward titration was permitted at intervals of 2 weeks or more.
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
Pregnancy:Third trimester use may increase risk for symptoms of poor neonatal adaptation (respiratory distress, temperature instability, feeding difficulty, hypotonia, tremor, irritability) in the neonate ( 8.1).
14.1 Major Depressive Disorder
The efficacy of venlafaxine hydrochloride extended-release capsules as a treatment for Major Depressive Disorder (MDD) was established in two placebo-controlled, short-term (8 weeks for study 1; 12 weeks for study 2), flexible-dose studies, with doses starting at 75 mg per day and ranging to 225 mg per day in adult outpatients meeting DSM-III-R or DSM-IV criteria for MDD. In moderately depressed outpatients, the initial dose of venlafaxine was 75 mg per day. In both studies, venlafaxine hydrochloride extended-release capsules demonstrated superiority over placebo on the primary efficacy measure defined as change from baseline in the HAM-D-21 total score to the endpoint visit, venlafaxine hydrochloride extended-release capsules also demonstrated superiority over placebo on the key secondary efficacy endpoint, the Clinical Global Impressions (CGI) Severity of Illness scale. Examination of gender subsets of the population studied did not reveal any differential responsiveness on the basis of gender.
A 4-week study of inpatients meeting DSM-III-R criteria for MDD with melancholia utilizing venlafaxine hydrochloride tablets in a range of 150 to 375 mg per day (divided in a three-times-a-day schedule) demonstrated superiority of venlafaxine hydrochloride tablets over placebo based on the HAM-D-21 total score. The mean dose in completers was 350 mg per day (study 3).
In a longer-term study, adult outpatients with MDD who had responded during an 8-week open-label study on venlafaxine hydrochloride extended-release capsules (75, 150, or 225 mg, once daily every morning) were randomized to continuation of their same venlafaxine hydrochloride extended-release capsules dose or to placebo, for up to 26 weeks of observation for relapse. Response during the open-label phase was defined as a CGI Severity of Illness item score of ≤3 and a HAM-D-21 total score of ≤10 at the day 56 evaluation. Relapse during the double-blind phase was defined as follows: (1) a reappearance of major depressive disorder as defined by DSM-IV criteria and a CGI Severity of Illness item score of ≥4 (moderately ill), (2) 2 consecutive CGI Severity of Illness item scores of ≥4, or (3) a final CGI Severity of Illness item score of ≥4 for any patient who withdrew from the study for any reason. Patients receiving continued venlafaxine hydrochloride extended-release capsules treatment experienced statistically significantly lower relapse rates over the subsequent 26 weeks compared with those receiving placebo (study 4).
In a second longer term trial, adult outpatients with MDD, recurrent type, who had responded (HAM-D-21 total score ≤12 at the day 56 evaluation) and continued to be improved [defined as the following criteria being met for days 56 through 180: (1) no HAM-D-21 total score ≥20; (2) no more than 2 HAM-D-21 total scores >10, and (3) no single CGI Severity of Illness item score ≥4 (moderately ill)] during an initial 26 weeks of treatment on venlafaxine hydrochloride tablets [100 to 200 mg per day, on a twice daily schedule] were randomized to continuation of their same venlafaxine hydrochloride tablets dose or to placebo. The follow-up period to observe patients for relapse, defined as a CGI Severity of Illness item score ≥4, was for up to 52 weeks. Patients receiving continued venlafaxine hydrochloride tablets treatment experienced statistically significantly lower relapse rates over the subsequent 52 weeks compared with those receiving placebo (study 5).
| Study Number | Treatment Group | Primary Efficacy Measure: HAM-D Score | ||
| Mean Baseline Score (SD) | LS Mean Change from Baseline |
Placebo Subtracted Differencea
(95% CI) |
||
| Study 1 | Venlafaxine Hydrochloride Extended-Release Capsules (75 to 225 mg/day)* | 24.5 | -11.7 | -4.45 (-6.66,-2.25) |
| Placebo | 23.6 | -7.24 | -- | |
| Study 2 | Venlafaxine Hydrochloride Extended-Release Capsules (75 to 225 mg/day)* | 24.5 | -15.11 | -6.40 (-8.45, -4.34) |
| Placebo | 24.9 | -8.71 | -- | |
| Study 3 | Venlafaxine Hydrochloride Extended-Release Capsules (150 to375 mg/day)* | 28.2 (0.5) | -14.9 | -10.2 (-14.4, -6.0) |
| Placebo | 28.6 (0.6) | -4.7 | -- | |
| SD=standard deviation; LS Mean=least-squares mean; CI=confidence interval. | ||||
| aDifference (drug minus placebo) in least-squares mean change from baseline. | ||||
| *Doses statistically significantly superior to placebo. |
5.4 Increased Risk of Bleeding
Drugs that interfere with serotonin reuptake inhibition, including venlafaxine hydrochloride extended-release capsules, may increase the risk of bleeding events, ranging from ecchymoses, hematomas, epistaxis, petechiae, and gastrointestinal hemorrhage to life-threatening hemorrhage. Concomitant use of aspirin, Nonsteroidal Anti-Inflammatory Drugs (NSAIDs), warfarin, and other anti-coagulants or other drugs known to affect platelet function may add to this risk. Case reports and epidemiological studies (case-control and cohort design) have demonstrated an association between use of drugs that interfere with serotonin reuptake and the occurrence of gastrointestinal bleeding. Based on data from the published observational studies, exposure to SNRIs, particularly in the month before delivery, has been associated with a less than 2-fold increase in the risk of postpartum hemorrhage [see Use in Specific Populations ( 8.1)] .
Inform patients about the increased risk of bleeding associated with the concomitant use of venlafaxine hydrochloride extended-release capsules and nonsteroidal anti-inflammatory drugs (NSAIDs), aspirin, or other drugs that affect coagulation. For patients taking warfarin, carefully monitor coagulation indices when initiating, titrating, or discontinuing venlafaxine hydrochloride extended-release capsules.
6.1 Clinical Studies Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical studies of another drug and may not reflect the rates observed in clinical practice.
2.3 Generalized Anxiety Disorder
For most patients, the recommended starting dose for venlafaxine hydrochloride extended-release capsules is 75 mg per day, administered in a single dose. For some patients, it may be desirable to start at 37.5 mg per day for 4 to 7 days to allow new patients to adjust to the medication before increasing to 75 mg per day. Patients not responding to the initial 75 mg per day dose may benefit from dose increases to a maximum of 225 mg per day. Dose increases should be in increments of up to 75 mg per day, as needed, and should be made at intervals of not less than 4 days.
14.2 Generalized Anxiety Disorder
The efficacy of venlafaxine hydrochloride extended-release capsules as a treatment for Generalized Anxiety Disorder (GAD) was established in two 8-week, placebo-controlled, fixed-dose studies (75 to 225 mg per day), one 6-month, placebo-controlled, flexible-dose study (75 to 225 mg per day), and one 6-month, placebo-controlled, fixed-dose study (37.5, 75, and 150 mg per day) in adult outpatients meeting DSM-IV criteria for GAD.
In one 8-week study, venlafaxine hydrochloride extended-release capsules demonstrated superiority over placebo for the 75, 150, and 225 mg per day doses as measured by the Hamilton Rating Scale for Anxiety (HAM-A) total score, both the HAM-A anxiety and tension items, and the Clinical Global Impressions (CGI) scale. However, the 75 and 150 mg per day doses were not as consistently effective as the highest dose (study 1). A second 8-week study evaluating doses of 75 and 150 mg per day and placebo showed that both doses were more effective than placebo on some of these same outcomes; however, the 75 mg per day dose was more consistently effective than the 150 mg per day dose (study 2). A dose-response relationship for effectiveness in GAD was not clearly established in the 75 to 225 mg per day dose range studied.
Two 6-month studies, one evaluating venlafaxine hydrochloride extended-release capsules doses of 37.5, 75, and 150 mg per day (study 3) and the other evaluating venlafaxine hydrochloride extended-release capsules doses of 75 to 225 mg per day (study 4), showed that daily doses of 75 mg or higher were more effective than placebo on the HAM-A total, both the HAM-A anxiety and tension items, and the CGI scale during 6 months of treatment. While there was also evidence for superiority over placebo for the 37.5 mg per day dose, this dose was not as consistently effective as the higher doses.
Examination of gender subsets of the population studied did not reveal any differential responsiveness on the basis of gender.
| Study Number | Treatment Group | Primary Efficacy Measure: HAM-A Score | ||
| Mean Baseline Score (SD) | LS Mean Change from Baseline (SE)* | Placebo Subtracted Difference a(95% CI) | ||
| Study 1 | Venlafaxine Hydrochloride Extended-Release Capsules 75 mg | 24.7 | -11.1 (0.95) | -1.5 (-3.8, 0.8) |
| Venlafaxine Hydrochloride Extended-Release Capsules 150 mg | 24.5 | -11.7 (0.87) | -2.2 (-4.5, 0.1) | |
| Venlafaxine Hydrochloride Extended-Release Capsules 225 mg | 23.6 | -12.1 (0.81) | -2.6 (-4.9, -0.3) | |
| Placebo | 24.1 | -9.5 (0.85) | -- | |
| Study 2 | Venlafaxine Hydrochloride Extended-Release Capsules 75 mg | 23.7 | -10.6 (0.82) | -2.6 (-4.6, -0.5) |
| Venlafaxine Hydrochloride Extended-Release Capsules 150 mg | 23.0 | -9.8 (0.86) | -1.7 (-3.8, 0.3) | |
| Placebo | 23.7 | -8.0 (0.73) | -- | |
| Study 3 | Venlafaxine Hydrochloride Extended-Release Capsules 37.5 mg | 26.6 (0.4) | -13.8 | -2.8 (-5.1, -0.6) |
| Venlafaxine Hydrochloride Extended-Release Capsules 75 mg | 26.3 (0.4) | -15.5 | -4.6 (-6.9, -2.3) | |
| Venlafaxine Hydrochloride Extended-Release Capsules 150 mg | 26.3 (0.4) | -16.4 | -5.5 (-7.8, -3.1) | |
| Placebo | 26.7 (0.5) | -11.0 | -- | |
| Study 4 | Venlafaxine Hydrochloride Extended-Release Capsules 75 to 225 mg | 25.0 | -13.4 (0.79) | -4.7 (-6.6, -2.9) |
| Placebo | 24.9 | -8.7 (0.70) | -- | |
| SD=standard deviation; LS Mean=least-squares mean; CI=confidence interval. | ||||
| aDifference (drug minus placebo) in least-squares mean change from baseline. | ||||
| *Doses statistically significantly superior to placebo. |
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient to read the FDA-approved patient labeling (Medication Guide).
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
Product: 50090-7323
NDC: 50090-7323-0 30 CAPSULE, EXTENDED RELEASE in a BOTTLE
NDC: 50090-7323-1 90 CAPSULE, EXTENDED RELEASE in a BOTTLE
5.6 Activation of Mania Or Hypomania (5.6 Activation of Mania or Hypomania)
In patients with bipolar disorder, treating a depressive episode with venlafaxine hydrochloride extended-release capsules or another antidepressant may precipitate a mixed/manic episode. Mania or hypomania was reported in venlafaxine hydrochloride extended-release capsules treated patients in the premarketing studies in MDD, SAD, and PD (see Table 2). Prior to initiating treatment with venlafaxine hydrochloride extended-release capsules, screen for any personal or family history of bipolar disorder, mania, or hypomania.
| Indication | Venlafaxine Hydrochloride Extended-Release Capsules | Placebo |
|---|---|---|
| MDD | 0.3 | 0.0 |
| GAD | 0.0 | 0.2 |
| SAD | 0.2 | 0.0 |
| PD | 0.1 | 0.0 |
2.1 General Administration Information
Administer venlafaxine hydrochloride extended-release capsules as a single dose with food, either in the morning or in the evening at approximately the same time each day [see Clinical Pharmacology (12.3)] . Swallow capsules whole with fluid. Do not divide, crush, chew, or place in water.
The capsule may also be administered by carefully opening the capsule and sprinkling the entire contents on a spoonful of applesauce. This drug/food mixture should be swallowed immediately without chewing and followed with a glass of water to ensure complete swallowing of the pellets (spheroids).
Warning: Suicidal Thoughts and Behaviors (WARNING: SUICIDAL THOUGHTS AND BEHAVIORS)
Antidepressants increased the risk of suicidal thoughts and behavior in pediatric and young adult patients in short-term studies. Closely monitor all antidepressant-treated patients for clinical worsening, and emergence of suicidal thoughts and behaviors [see Warnings and Precautions (5.1)] . Venlafaxine hydrochloride extended-release capsules are not approved for use in pediatric patient s [see Use in Specific Populations (8.4)] .
2.4 Social Anxiety Disorder (social Phobia) (2.4 Social Anxiety Disorder (Social Phobia))
The recommended dose is 75 mg per day, administered in a single dose. There was no evidence that higher doses confer any additional benefit.
5.11 Appetite Changes in Pediatric Patients
Decreased appetite (reported as anorexia) was more commonly observed in venlafaxine hydrochloride extended-release capsules treated patients versus placebo-treated patients in the premarketing evaluation of venlafaxine hydrochloride extended-release capsules for MDD, GAD, and SAD (see Table 6).
Venlafaxine hydrochloride extended-release capsules are not approved for use in pediatric patients [see Use in Specific Populations (8.4)] .
| Indication
(Duration) |
Venlafaxine Hydrochloride Extended-Release Capsules Incidence | Discontinuation | Placebo Incidence | Discontinuation |
|---|---|---|---|---|
| MDD and GAD
(pooled, 8 weeks) |
10 | 0.0 | 3 | – |
| SAD
(16 weeks) |
22 | 0.7 | 3 | 0.0 |
|
aThe discontinuation rates for weight loss were 0.7% for patients receiving either venlafaxine hydrochloride extended-release capsules or placebo.
bVenlafaxine hydrochloride extended-release capsules are not approved for use in pediatric patients. |
5.12 Interstitial Lung Disease and Eosinophilic Pneumonia
Interstitial lung disease and eosinophilic pneumonia associated with venlafaxine therapy have been rarely reported. The possibility of these events should be considered in venlafaxine hydrochloride extended-release capsules-treated patients who present with progressive dyspnea, cough or chest discomfort. Such patients should undergo a prompt medical evaluation, and discontinuation of venlafaxine hydrochloride extended-release capsules should be considered.
14.3 Social Anxiety Disorder (also Known As Social Phobia) (14.3 Social Anxiety Disorder (Also Known as Social Phobia))
The efficacy of venlafaxine hydrochloride extended-release capsules as a treatment for Social Anxiety Disorder (SAD) was established in four double-blind, parallel-group, 12-week, multicenter, placebo-controlled, flexible-dose studies (studies 1 to 4) and one double-blind, parallel-group, 6-month, placebo-controlled, fixed/flexible-dose study, which included doses in a range of 75 to 225 mg per day in adult outpatients meeting DSM-IV criteria for SAD (study 5).
In these five studies, venlafaxine hydrochloride extended-release capsules were statistically significantly more effective than placebo on change from baseline to endpoint on the Liebowitz Social Anxiety Scale (LSAS) total score. There was no evidence for any greater effectiveness of the 150 to 225 mg per day group compared to the 75 mg per day group in the 6-month study.
Examination of subsets of the population studied did not reveal any differential responsiveness on the basis of gender. There was insufficient information to determine the effect of age or race on outcome in these studies.
| Study Number | Treatment Group | Primary Efficacy Measure: LSAS Score | ||
|---|---|---|---|---|
| Mean Baseline Score (SD) | LS Mean Change from Baseline (SE) | Placebo Subtracted Difference a(95% CI) | ||
| Study 1 | Venlafaxine Hydrochloride Extended-Release Capsules (75 to 225 mg)
SD=standard deviation; SE=standard error; LS Mean=least-squares mean; CI=confidence interval.
aDifference (drug minus placebo) in least-squares mean change from baseline.
*Doses statistically significantly superior to placebo.
|
91.1 | -31.0 (2.22) | 11.2 (-5.3, -17.1) |
| Placebo | 86.7 | -19.9 (2.22) | - | |
| Study 2 | Venlafaxine Hydrochloride Extended-Release Capsules (75 to 225 mg) | 90.8 | -32.8 (2.69) | -10.7 (-3.7,-17.6) |
| Placebo | 87.4 | -22.1 (2.66) | - | |
| Study 3 | Venlafaxine Hydrochloride Extended-Release Capsules (75 to 225 mg) | 83.2 | -36.0 (2.35) | -16.9 (-22.6, -11.2) |
| Placebo | 83.6 | -19.1 (2.40) | -12.7 (-6.5, -19.0) | |
| Study 4 | Venlafaxine Hydrochloride Extended-Release Capsules (75 to 225 mg) | 86.2 | -35.0 (2.64) | -14.6 (-21.8, -7.4) |
| Placebo | 86.1 | -22.2 (2.47) | ||
| Study 5 | Venlafaxine Hydrochloride Extended-Release Capsules 75 mg | 91.8 | -38.1 (3.16) | -14.6 (-21.8, -7.4) |
| Venlafaxine Hydrochloride Extended-Release Capsules (150 to 225 mg) | 86.2 | -37.6 (3.05) | -14.1 (-21.3, -6.9) | |
| Placebo | 89.3 | -23.5 (3.08) |
2.9 Dosage Recommendations for Patients With Renal Impairment (2.9 Dosage Recommendations for Patients with Renal Impairment)
Reduce the venlafaxine hydrochloride extended-release capsules total daily dose by 25% to 50% in patients with mild (CLcr 60 to 89 mL/min) or moderate (CLcr 30 to 59 mL/min) renal impairment. Reduce the total daily dose by 50% or more in patients undergoing hemodialysis or with severe renal impairment (CLcr<30 mL/min). Because there was much individual variability in clearance between patients with renal impairment, individualization of dosage is recommended in some patients [see Use in Specific Populations (8.7)] .
2.7 Switching Patients From venlafaxine Hydrochloride Tablets (2.7 Switching Patients from Venlafaxine Hydrochloride Tablets)
Patients with depression who are currently being treated with venlafaxine hydrochloride tablets may be switched to venlafaxine hydrochloride extended-release capsules at the nearest equivalent dose (mg per day), e.g., 37.5 mg venlafaxine twice a day to 75 mg venlafaxine hydrochloride extended-release capsules once daily. However, individual dosage adjustments may be necessary.
2.8 Dosage Recommendations for Patients With Hepatic Impairment (2.8 Dosage Recommendations for Patients with Hepatic Impairment)
Reduce the venlafaxine hydrochloride extended-release capsules total daily dose by 50% in patients with mild (Child-Pugh Class A) to moderate (Child-Pugh Class B) hepatic impairment. Reduce the total daily dose by 50% or more in patients with severe hepatic impairment (Child-Pugh Class C) or hepatic cirrhosis [see Use in Specific Populations (8.6)] .
5.1 Suicidal Thoughts and Behaviors in Adolescents and Young Adults
In pooled analyses of placebo-controlled trials of antidepressant drugs (SSRIs and other antidepressant classes) that included approximately 77,000 adult patients and 4,500 pediatric patients, the incidence of suicidal thoughts and behaviors in antidepressant-treated patients age 24 years and younger was greater than in placebo-treated patients. There was considerable variation in risk of suicidal thoughts and behaviors among drugs, but there was an increased risk identified in young patients for most drugs studied. There were differences in absolute risk of suicidal thoughts and behaviors across the different indications, with the highest incidence in patients with MDD. The drug-placebo differences in the number of cases of suicidal thoughts and behaviors per 1,000 patients treated are provided in Table 1.
| Age Range | Drug-Placebo Difference in Number of Patients of Suicidal Thoughts and Behaviors per 1,000 Patients Treated |
| Increases Compared to Placebo | |
| <18 years old | 14 additional patients |
| 18 to 24 years old | 5 additional patients |
| Decreases Compared to Placebo | |
| 25 to 64 years old | 1 fewer patient |
| ≥65 years old | 6 fewer patients |
| * Venlafaxine hydrochloride extended-release capsules are not approved in pediatric patients. |
It is unknown whether the risk of suicidal thoughts and behaviors in children, adolescents, and young adults extends to longer-term use, i.e., beyond four months. However, there is substantial evidence from placebo-controlled maintenance trials in adults with MDD that antidepressants delay the recurrence of depression and that depression itself is a risk factor for suicidal thoughts and behaviors.
Monitor all antidepressant-treated patients for any indication for clinical worsening and emergence of suicidal thoughts and behaviors, especially during the initial few months of drug therapy, and at times of dosage changes. Counsel family members or caregivers of patients to monitor for changes in behavior and to alert the healthcare provider. Consider changing the therapeutic regimen, including possibly discontinuing venlafaxine hydrochloride extended-release capsules, in patients whose depression is persistently worse, or who are experiencing emergent suicidal thoughts or behaviors.
2.10 Discontinuing Treatment With venlafaxine Hydrochloride Extended Release Capsules (2.10 Discontinuing Treatment with Venlafaxine Hydrochloride Extended-Release Capsules)
A gradual reduction in the dose, rather than abrupt cessation, is recommended when discontinuing therapy with venlafaxine hydrochloride extended-release capsules. In clinical studies with venlafaxine hydrochloride extended-release capsules, tapering was achieved by reducing the daily dose by 75 mg at one-week intervals. Individualization of tapering may be necessary. In some patients, discontinuation may need to occur over a period of several months [see Warnings and Precautions (5.7)] .
2.11 Switching Patients to Or From A Monoamine Oxidase Inhibitor (maoi) Antidepressant (2.11 Switching Patients to or from a Monoamine Oxidase Inhibitor (MAOI) Antidepressant)
At least 14 days must elapse between discontinuation of an MAOI antidepressant and initiation of venlafaxine hydrochloride extended-release capsules. In addition, at least 7 days must elapse after stopping venlafaxine hydrochloride extended-release capsules before starting an MAOI antidepressant [see Contraindications (4), Warnings and Precautions (5.2), and Drug Interactions (7.1)].
2.6 Screen for Bipolar Disorder Prior to Starting venlafaxine Hydrochloride Extended Release Capsules (2.6 Screen for Bipolar Disorder Prior to Starting Venlafaxine Hydrochloride Extended-Release Capsules)
Prior to initiating treatment with venlafaxine hydrochloride extended-release capsules, screen patients for a personal or family history of bipolar disorder, mania, or hypomania [see Warnings and Precautions( 5.6)] .
7.1 Drugs Having Clinically Important Interactions With venlafaxine Hydrochloride Extended Release Capsules (7.1 Drugs Having Clinically Important Interactions with Venlafaxine Hydrochloride Extended-Release Capsules)
| Monoamine Oxidase Inhibitors (MAOI) | |
| Clinical Impact | The concomitant use of SNRIs, including venlafaxine hydrochloride extended-release capsules, with MAOIs increases the risk of serotonin syndrome. |
| Intervention | Concomitant use of venlafaxine hydrochloride extended-release capsules is contraindicated in patients taking MAOIs, including MAOIs such as linezolid or intravenous methylene blue [see Dosage and Administration (2.11), Contraindications (4)and Warnings and Precautions (5.2)]. |
| Other Serotonergic Drugs | |
| Clinical Impact | Concomitant use of venlafaxine hydrochloride extended-release capsules with other serotonergic drugs (including other SNRIs, SSRIs, triptans, tricyclic antidepressants, opioids, lithium, buspirone, amphetamines, tryptophan, and St. John's Wort) increases the risk of serotonin syndrome. |
| Intervention | Monitor for symptoms of serotonin syndrome when venlafaxine hydrochloride extended-release capsules is used concomitantly with other drugs that may affect the serotonergic neurotransmitter systems. If serotonin syndrome occurs, consider discontinuation of venlafaxine hydrochloride extended-release capsules and/or concomitant serotonergic drugs [see Dosage and Administration (2.11)and Warnings and Precautions (5.2)]. |
| Drugs that Interfere with Hemostasis | |
| Clinical Impact | Concomitant use of venlafaxine hydrochloride extended-release capsules with an antiplatelet or anticoagulant drug may potentiate the risk of bleeding. This may be due to the effect of venlafaxine hydrochloride extended-release capsules on the release of serotonin by platelets. |
| Intervention | Closely monitor for bleeding for patients receiving an antiplatelet or anticoagulant drug when venlafaxine hydrochloride extended-release capsules are initiated or discontinued [see Warnings and Precautions (5.4)] . |
| Effect of CYP3A Inhibitors | |
| Clinical Impact | Concomitant use of a CYP3A inhibitor increases the C maxand AUC of venlafaxine and O-desmethylvenlafaxine (ODV) [see Clinical Pharmacology (12.3)] , which may increase the risk of toxicity of venlafaxine hydrochloride extended-release capsules. |
| Intervention | Consider reducing the dose of venlafaxine hydrochloride extended-release capsules. |
| CYP2D6 Substrates | |
| Clinical Impact | Concomitant use of venlafaxine hydrochloride extended-release capsules increases C maxand AUC of a CYP2D6 substrate, which may increase the risk of toxicity of the CYP2D6 substrate [see Clinical Pharmacology (12.3)] . |
| Intervention | Consider reduction in dose of concomitant CYP2D6 substrates. |
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:50:39.111303 · Updated: 2026-03-14T22:38:11.072710