These Highlights Do Not Include All The Information Needed To Use Clobazam Tablets Safely And Effectively. See Full Prescribing Information For Clobazam Tablets.
c326d915-5cbb-4f96-ba5d-c2b298e10c5d
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Concomitant use of benzodiazepines and opioids may result in profound sedation, respiratory depression, coma, and death [see Warnings and Precautions (5.1) , Drug Interactions (7.1) ]. Reserve concomitant prescribing of these drugs for use in patients for whom alternative treatment options are inadequate. Limit dosages and durations to the minimum required. Follow patients for signs and symptoms of respiratory depression and sedation.
Indications and Usage
Clobazam tablets are indicated for the adjunctive treatment of seizures associated with Lennox-Gastaut syndrome (LGS) in patients 2 years of age or older.
Dosage and Administration
For doses above 5 mg/day administer in two divided doses ( 2.1 ) Patients ≤30 kg body weight: Initiate at 5 mg daily and titrate as tolerated up to 20 mg daily ( 2.1 ) Patients >30 kg body weight: Initiate at 10 mg daily and titrate as tolerated up to 40 mg daily ( 2.1 ) Dosage adjustment needed in following groups: Geriatric patients ( 2.4 , 8.5 ) Known CYP2C19 poor metabolizers ( 2.5 ) Mild or moderate hepatic impairment; no information for severe hepatic impairment ( 2.7 , 8.8 ) Reduce dose, or discontinue drug gradually ( 2.2 ) Administer whole, broken in half along the score, or crush and mix in applesauce ( 2.3 ) Can be taken with or without food ( 2.3 )
Warnings and Precautions
Somnolence or Sedation: Monitor for central nervous system (CNS) depression. Risk may be increased with concomitant use of other CNS depressants ( 5.2 , 5.3 ) Withdrawal: Symptoms may occur with rapid dose reduction or discontinuation. Discontinue clobazam gradually ( 5.4 ) Serious Dermatological Reactions (including Stevens-Johnson syndrome and toxic epidermal necrolysis): Discontinue clobazam at first sign of rash unless the rash is clearly not drug-related ( 5.5 ) Physical and Psychological Dependence: Monitor patients with a history of substance abuse for signs of habituation and dependence ( 5.6 , 9 ) Suicidal Behavior and Ideation: Monitor for suicidal thoughts or behaviors ( 5.7 )
Contraindications
Clobazam tablets are contraindicated in patients with a history of hypersensitivity to the drug or its ingredients. Hypersensitivity reactions have included serious dermatological reactions [see Warnings and Precautions (5.5) ] .
Adverse Reactions
Clinically significant adverse reactions that appear in other sections of the labeling include the following: Risks from Concomitant Use with Opioids [see Warnings and Precautions (5.1) ] Potentiation of Sedation from Concomitant Use with Central Nervous System Depressants [see Warnings and Precautions (5.2) ] Somnolence or Sedation [see Warnings and Precautions (5.3) ] Withdrawal Symptoms [see Warnings and Precautions (5.4) ] Serious Dermatological Reactions [see Contraindications (4) , Warnings and Precautions (5.5) ] Physical and Psychological Dependence [see Warnings and Precautions (5.6) ] Suicidal Behavior and Ideation [see Warnings and Precautions (5.7) ]
Drug Interactions
Alcohol: Increases blood levels of clobazam by about 50% ( 7.2 ) Drugs metabolized by CYP2D6: Lower doses of these drugs may be required when used concomitantly with clobazam ( 7.3 ) Strong or Moderate CYP2C19 Inhibitors: Dosage adjustment of clobazam may be necessary ( 7.4 )
Storage and Handling
Each clobazam tablet contains 10 mg or 20 mg of clobazam and is a white to off-white, caplet shape tablet functionally-scored on both the sides debossed with "T" over the score and "10" under the score or a "T" over the score and "20" under the score on one side and plain on the other side. NDC 51672-4202-1: 10 mg scored tablet, Bottles of 100 NDC 51672-4203-1: 20 mg scored tablet, Bottles of 100
How Supplied
Each clobazam tablet contains 10 mg or 20 mg of clobazam and is a white to off-white, caplet shape tablet functionally-scored on both the sides debossed with "T" over the score and "10" under the score or a "T" over the score and "20" under the score on one side and plain on the other side. NDC 51672-4202-1: 10 mg scored tablet, Bottles of 100 NDC 51672-4203-1: 20 mg scored tablet, Bottles of 100
Medication Information
Warnings and Precautions
Somnolence or Sedation: Monitor for central nervous system (CNS) depression. Risk may be increased with concomitant use of other CNS depressants ( 5.2 , 5.3 ) Withdrawal: Symptoms may occur with rapid dose reduction or discontinuation. Discontinue clobazam gradually ( 5.4 ) Serious Dermatological Reactions (including Stevens-Johnson syndrome and toxic epidermal necrolysis): Discontinue clobazam at first sign of rash unless the rash is clearly not drug-related ( 5.5 ) Physical and Psychological Dependence: Monitor patients with a history of substance abuse for signs of habituation and dependence ( 5.6 , 9 ) Suicidal Behavior and Ideation: Monitor for suicidal thoughts or behaviors ( 5.7 )
Indications and Usage
Clobazam tablets are indicated for the adjunctive treatment of seizures associated with Lennox-Gastaut syndrome (LGS) in patients 2 years of age or older.
Dosage and Administration
For doses above 5 mg/day administer in two divided doses ( 2.1 ) Patients ≤30 kg body weight: Initiate at 5 mg daily and titrate as tolerated up to 20 mg daily ( 2.1 ) Patients >30 kg body weight: Initiate at 10 mg daily and titrate as tolerated up to 40 mg daily ( 2.1 ) Dosage adjustment needed in following groups: Geriatric patients ( 2.4 , 8.5 ) Known CYP2C19 poor metabolizers ( 2.5 ) Mild or moderate hepatic impairment; no information for severe hepatic impairment ( 2.7 , 8.8 ) Reduce dose, or discontinue drug gradually ( 2.2 ) Administer whole, broken in half along the score, or crush and mix in applesauce ( 2.3 ) Can be taken with or without food ( 2.3 )
Contraindications
Clobazam tablets are contraindicated in patients with a history of hypersensitivity to the drug or its ingredients. Hypersensitivity reactions have included serious dermatological reactions [see Warnings and Precautions (5.5) ] .
Adverse Reactions
Clinically significant adverse reactions that appear in other sections of the labeling include the following: Risks from Concomitant Use with Opioids [see Warnings and Precautions (5.1) ] Potentiation of Sedation from Concomitant Use with Central Nervous System Depressants [see Warnings and Precautions (5.2) ] Somnolence or Sedation [see Warnings and Precautions (5.3) ] Withdrawal Symptoms [see Warnings and Precautions (5.4) ] Serious Dermatological Reactions [see Contraindications (4) , Warnings and Precautions (5.5) ] Physical and Psychological Dependence [see Warnings and Precautions (5.6) ] Suicidal Behavior and Ideation [see Warnings and Precautions (5.7) ]
Drug Interactions
Alcohol: Increases blood levels of clobazam by about 50% ( 7.2 ) Drugs metabolized by CYP2D6: Lower doses of these drugs may be required when used concomitantly with clobazam ( 7.3 ) Strong or Moderate CYP2C19 Inhibitors: Dosage adjustment of clobazam may be necessary ( 7.4 )
Storage and Handling
Each clobazam tablet contains 10 mg or 20 mg of clobazam and is a white to off-white, caplet shape tablet functionally-scored on both the sides debossed with "T" over the score and "10" under the score or a "T" over the score and "20" under the score on one side and plain on the other side. NDC 51672-4202-1: 10 mg scored tablet, Bottles of 100 NDC 51672-4203-1: 20 mg scored tablet, Bottles of 100
How Supplied
Each clobazam tablet contains 10 mg or 20 mg of clobazam and is a white to off-white, caplet shape tablet functionally-scored on both the sides debossed with "T" over the score and "10" under the score or a "T" over the score and "20" under the score on one side and plain on the other side. NDC 51672-4202-1: 10 mg scored tablet, Bottles of 100 NDC 51672-4203-1: 20 mg scored tablet, Bottles of 100
Description
Concomitant use of benzodiazepines and opioids may result in profound sedation, respiratory depression, coma, and death [see Warnings and Precautions (5.1) , Drug Interactions (7.1) ]. Reserve concomitant prescribing of these drugs for use in patients for whom alternative treatment options are inadequate. Limit dosages and durations to the minimum required. Follow patients for signs and symptoms of respiratory depression and sedation.
Section 42229-5
Clobazam Tablet Oral Administration
Clobazam tablets can be taken with or without food.
Clobazam tablets can be administered whole, broken in half along the score, or crushed and mixed in applesauce.
Section 42231-1
| This Medication Guide has been approved by the U.S. Food and Drug Administration. | ||
| Revised: June 2018 | 20999-0618-1 | |
|
MEDICATION GUIDE CIV Clobazam (KLOE ba zam) Tablets |
||
|
What is the most important information I should know about clobazam tablets?
Call your healthcare provider right away if you have any of these symptoms, especially if they are new, worse, or worry you: |
||
|
|
|
|
How can I watch for early symptoms of suicidal thoughts and actions?
Call your healthcare provider between visits as needed, especially if you are worried about symptoms. Suicidal thoughts or actions can be caused by things other than medicines. If you have suicidal thoughts or actions, your healthcare provider may check for other causes. |
||
|
What are clobazam tablets? Clobazam tablet is a prescription medicine used along with other medicines to treat seizures associated with Lennox-Gastaut syndrome in people 2 years of age or older. It is not known if clobazam tablets are safe and effective in children less than 2 years old. |
||
|
Do not take clobazam tablets if you:
|
||
|
Before you take clobazam tablets, tell your healthcare provider about all your medical conditions, including if you:
Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. Taking clobazam tablets with certain other medicines can cause side effects or affect how well clobazam tablets or the other medicines work. Do not start or stop other medicines without talking to your healthcare provider. |
||
|
How should I take clobazam tablets?
|
||
|
What should I avoid while taking clobazam tablets?
|
||
|
What are the possible side effects of clobazam tablets? Clobazam tablets may cause serious side effects, including: See "What is the most important information I should know about clobazam tablets?" The most common side effects of clobazam tablets include: |
||
|
|
|
|
These are not all the possible side effects of clobazam tablets. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
||
|
How should I store clobazam tablets?
|
||
|
General information about the safe and effective use of clobazam tablets. Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use clobazam tablets for a condition for which it was not prescribed. Do not give clobazam tablets to other people, even if they have the same symptoms that you have. It may harm them. You can ask your pharmacist or healthcare provider for information about clobazam tablets that is written for health professionals. |
||
|
What are the ingredients in clobazam tablets? Active ingredient: clobazam Inactive ingredients: colloidal silicone dioxide, lactose monohydrate, magnesium stearate, pregelatinized maize starch, and talc. Mfd. by: For more information about clobazam tablets, go to www.taro.com or call Taro Pharmaceuticals U.S.A., Inc. at 1-866-923-4914. |
Section 44425-7
Store tablets at 20°C to 25°C (68°F to 77°F) [See USP Controlled Room Temperature].
9.2 Abuse
Clobazam can be abused in a similar manner as other benzodiazepines, such as diazepam.
The pharmacological profile of clobazam is similar to that of other benzodiazepines listed in Schedule IV of the Controlled Substance Act, particularly in its potentiation of GABAergic transmission through its action on GABAA receptors, which leads to sedation and somnolence.
The World Health Organization epidemiology database contains reports of drug abuse, misuse, and overdoses associated with clobazam.
Drug abuse is the intentional non-therapeutic use of a drug, repeatedly or even sporadically, for its rewarding psychological or physiological effects.
7.1 Opioids
The concomitant use of benzodiazepines and opioids increases the risk of respiratory depression because of actions at different receptor sites in the CNS that control respiration. Benzodiazepines interact at GABAA sites, and opioids interact primarily at mu receptors. When benzodiazepines and opioids are combined, the potential for benzodiazepines to significantly worsen opioid-related respiratory depression exists. Limit dosage and duration of concomitant use of benzodiazepines and opioids, and follow patients closely for respiratory depression and sedation [see Warnings and Precautions (5.1)].
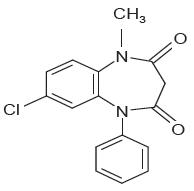
11 Description
| Established Name: | Clobazam Tablets |
| Dosage Form: | Tablet |
| Route of Administration: | Oral |
| Established Pharmacologic Class of Drug: | Benzodiazepine |
| Chemical Name: | 7-Chloro-1-methyl-5-phenyl-1H-1,5 benzodiazepine-2,4(3H,5H)-dione |
| Structural Formula: |
|
Clobazam is a white or almost white, crystalline powder with a slightly bitter taste; is slightly soluble in water, sparingly soluble in ethanol, and freely soluble in methylene chloride. The melting range of clobazam is from 182°C to 185°C. The molecular formula is C16H13O2N2Cl and the molecular weight is 300.7.
Each clobazam tablet contains 10 mg or 20 mg of clobazam. Tablets also contain as inactive ingredients: colloidal silicone dioxide, lactose monohydrate, magnesium stearate, pregelatinized maize starch, and talc.
8.4 Pediatric Use
Safety and effectiveness in patients less than 2 years of age have not been established.
In a study in which clobazam (0 mg/kg/day, 4 mg/kg/day, 36 mg/kg/day, or 120 mg/kg/day) was orally administered to rats during the juvenile period of development (postnatal days 14 to 48), adverse effects on growth (decreased bone density and bone length) and behavior (altered motor activity and auditory startle response; learning deficit) were observed at the high dose. The effect on bone density, but not on behavior, was reversible when drug was discontinued. The no-effect level for juvenile toxicity (36 mg/kg/day) was associated with plasma exposures (AUC) to clobazam and its major active metabolite, N-desmethylclobazam, less than those expected at therapeutic doses in pediatric patients.
8.5 Geriatric Use
Clinical studies of clobazam did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects. However, elderly subjects appear to eliminate clobazam more slowly than younger subjects based on population pharmacokinetic analysis. For these reasons, the initial dose in elderly patients should be 5 mg/day. Patients should be titrated initially to 10 mg/day to 20 mg/day. Patients may be titrated further to a maximum daily dose of 40 mg if tolerated [see Dosage and Administration (2.4), Clinical Pharmacology (12.3)].
14 Clinical Studies
The effectiveness of clobazam for the adjunctive treatment of seizures associated with Lennox-Gastaut syndrome was established in two multicenter controlled studies (Study 1 and Study 2). Both studies were similar in terms of disease characteristics and concomitant AED treatments. The most common concomitant AED treatments at baseline included: valproate, lamotrigine, levetiracetam, and topiramate.
4 Contraindications
Clobazam tablets are contraindicated in patients with a history of hypersensitivity to the drug or its ingredients. Hypersensitivity reactions have included serious dermatological reactions [see Warnings and Precautions (5.5)].
6 Adverse Reactions
Clinically significant adverse reactions that appear in other sections of the labeling include the following:
- Risks from Concomitant Use with Opioids [see Warnings and Precautions (5.1)]
- Potentiation of Sedation from Concomitant Use with Central Nervous System Depressants [see Warnings and Precautions (5.2)]
- Somnolence or Sedation [see Warnings and Precautions (5.3)]
- Withdrawal Symptoms [see Warnings and Precautions (5.4)]
- Serious Dermatological Reactions [see Contraindications (4), Warnings and Precautions (5.5)]
- Physical and Psychological Dependence [see Warnings and Precautions (5.6)]
- Suicidal Behavior and Ideation [see Warnings and Precautions (5.7)]
7 Drug Interactions
8.7 Renal Impairment
The pharmacokinetics of clobazam were evaluated in patients with mild and moderate renal impairment. There were no significant differences in systemic exposure (AUC and Cmax) between patients with mild or moderate renal impairment and healthy subjects. No dose adjustment is required for patients with mild and moderate renal impairment. There is essentially no experience with clobazam in patients with severe renal impairment or ESRD. It is not known if clobazam or its active metabolite, N-desmethylclobazam, is dialyzable [see Dosage and Administration (2.6), Clinical Pharmacology (12.3)].
12.3 Pharmacokinetics
The peak plasma levels (Cmax) and the area under the curve (AUC) of clobazam are dose-proportional over the dose range of 10 mg to 80 mg following single- or multiple-dose administration of clobazam. Based on a population pharmacokinetic analysis, the pharmacokinetics of clobazam are linear from 5 mg/day to 160 mg/day. Clobazam is converted to N-desmethylclobazam which has about 1/5 the activity of clobazam. The estimated mean elimination half-lives (t½) of clobazam and N-desmethylclobazam were 36 hours to 42 hours and 71 hours to 82 hours, respectively.
12.5 Pharmacogenomics
The polymorphic CYP2C19 is the main enzyme that metabolizes the pharmacologically active N-desmethylclobazam. Compared to CYP2C19 extensive metabolizers, N-desmethylclobazam AUC and Cmax are approximately 3 times to 5 times higher in poor metabolizers (e.g., subjects with *2/*2 genotype) and 2 times higher in intermediate metabolizers (e.g., subjects with *1/*2 genotype). The prevalence of CYP2C19 poor metabolism differs depending on racial/ethnic background. Dosage in patients who are known CYP2C19 poor metabolizers may need to be adjusted [see Dosage and Administration (2.5)].
The systemic exposure of clobazam is similar for both CYP2C19 poor and extensive metabolizers.
2.1 Dosing Information
A daily dose of clobazam tablets greater than 5 mg should be administered in divided doses twice daily; a 5 mg daily dose can be administered as a single dose. Dose patients according to body weight. Individualize dosing within each body weight group, based on clinical efficacy and tolerability. Each dose in Table 1 (e.g., 5 mg to 20 mg in ≤30 kg weight group) has been shown to be effective, although effectiveness increases with increasing dose [see Clinical Studies (14)]. Do not proceed with dose escalation more rapidly than weekly, because serum concentrations of clobazam and its active metabolite require 5 and 9 days, respectively, to reach steady-state.
| ≤30 kg Body Weight | >30 kg Body Weight | |
|---|---|---|
| Starting Dose | 5 mg | 10 mg |
| Starting Day 7 | 10 mg | 20 mg |
| Starting Day 14 | 20 mg | 40 mg |
2.2 Gradual Withdrawal
As with all antiepileptic drugs and benzodiazepines, withdraw clobazam tablets gradually. Taper by decreasing the total daily dose by 5 mg/day to 10 mg/day on a weekly basis until discontinued [see Warnings and Precautions (5.4)].
8.8 Hepatic Impairment
Clobazam is hepatically metabolized; however, there are limited data to characterize the effect of hepatic impairment on the pharmacokinetics of clobazam. For this reason, dosage adjustment is recommended in patients with mild to moderate hepatic impairment (Child-Pugh score 5 to 9). There is inadequate information about metabolism of clobazam in patients with severe hepatic impairment [see Dosage and Administration (2.7), Clinical Pharmacology (12.3)].
1 Indications and Usage
Clobazam tablets are indicated for the adjunctive treatment of seizures associated with Lennox-Gastaut syndrome (LGS) in patients 2 years of age or older.
5.4 Withdrawal Symptoms
Abrupt discontinuation of clobazam should be avoided. Clobazam should be tapered by decreasing the dose every week by 5 mg/day to 10 mg/day until discontinuation [see Dosage and Administration (2.2)].
Withdrawal symptoms occurred following abrupt discontinuation of clobazam; the risk of withdrawal symptoms is greater with higher doses.
As with all antiepileptic drugs, clobazam should be withdrawn gradually to minimize the risk of precipitating seizures, seizure exacerbation, or status epilepticus.
Withdrawal symptoms (e.g., convulsions, psychosis, hallucinations, behavioral disorder, tremor, and anxiety) have been reported following abrupt discontinuance of benzodiazepines. The more severe withdrawal symptoms have usually been limited to patients who received excessive doses over an extended period of time, followed by an abrupt discontinuation. Generally milder withdrawal symptoms (e.g., dysphoria, anxiety, and insomnia) have been reported following abrupt discontinuance of benzodiazepines taken continuously at therapeutic doses for several months.
12.1 Mechanism of Action
The exact mechanism of action for clobazam, a 1,5-benzodiazepine, is not fully understood but is thought to involve potentiation of GABAergic neurotransmission resulting from binding at the benzodiazepine site of the GABAA receptor.
9.1 Controlled Substance
Clobazam tablets contains clobazam which is a Schedule IV controlled substance.
5 Warnings and Precautions
- Somnolence or Sedation: Monitor for central nervous system (CNS) depression. Risk may be increased with concomitant use of other CNS depressants (5.2, 5.3)
- Withdrawal: Symptoms may occur with rapid dose reduction or discontinuation. Discontinue clobazam gradually (5.4)
- Serious Dermatological Reactions (including Stevens-Johnson syndrome and toxic epidermal necrolysis): Discontinue clobazam at first sign of rash unless the rash is clearly not drug-related (5.5)
- Physical and Psychological Dependence: Monitor patients with a history of substance abuse for signs of habituation and dependence (5.6, 9)
- Suicidal Behavior and Ideation: Monitor for suicidal thoughts or behaviors (5.7)
5.3 Somnolence Or Sedation
Clobazam causes somnolence and sedation. In clinical trials, somnolence or sedation was reported at all effective doses and was dose-related.
In general, somnolence and sedation begin within the first month of treatment and may diminish with continued treatment. Prescribers should monitor patients for somnolence and sedation, particularly with concomitant use of other central nervous system depressants. Prescribers should caution patients against engaging in hazardous activities requiring mental alertness, such as operating dangerous machinery or motor vehicles, until the effect of clobazam is known.
2 Dosage and Administration
- For doses above 5 mg/day administer in two divided doses (2.1)
- Patients ≤30 kg body weight: Initiate at 5 mg daily and titrate as tolerated up to 20 mg daily (2.1)
- Patients >30 kg body weight: Initiate at 10 mg daily and titrate as tolerated up to 40 mg daily (2.1)
- Dosage adjustment needed in following groups:
- Reduce dose, or discontinue drug gradually (2.2)
- Administer whole, broken in half along the score, or crush and mix in applesauce (2.3)
- Can be taken with or without food (2.3)
3 Dosage Forms and Strengths
Tablets: 10 mg and 20 mg with a functional score for oral administration.
Each clobazam tablet is a white to off-white, caplet shape tablet functionally-scored on both sides debossed with "T" over the score and "10" under the score or a "T" over the score and "20" under the score on one side and plain on the other side.
6.2 Postmarketing Experience
These reactions are reported voluntarily from a population of uncertain size; therefore, it is not possible to estimate their frequency or establish a causal relationship to drug exposure. Adverse reactions are categorized by system organ class.
Blood Disorders: Anemia, eosinophilia, leukopenia, thrombocytopenia
Eye Disorders: Diplopia, vision blurred
Gastrointestinal Disorders: Abdominal distention
General Disorders and Administration Site Conditions: Hypothermia
Investigations: Hepatic enzyme increased
Musculoskeletal: Muscle spasms
Psychiatric Disorders: Agitation, anxiety, apathy, confusional state, depression, delirium, delusion, hallucination
Renal and Urinary Disorders: Urinary retention
Respiratory Disorders: Aspiration, respiratory depression
Skin and Subcutaneous Tissue Disorders: Rash, urticaria, angioedema, and facial and lip edema
10.2 Management of Overdosage
The management of clobazam overdose may include gastric lavage and/or administration of activated charcoal, intravenous fluid replenishment, early control of airway and general supportive measures, in addition to monitoring level of consciousness and vital signs. Hypotension can be treated by replenishment with plasma substitutes and, if necessary, with sympathomimetic agents.
The efficacy of supplementary administration of physostigmine (a cholinergic agent) or of flumazenil (a benzodiazepine antagonist) in clobazam overdose has not been assessed. The administration of flumazenil in cases of benzodiazepine overdose can lead to withdrawal and adverse reactions. Its use in patients with epilepsy is typically not recommended.
8 Use in Specific Populations
Pregnancy: Based on animal data, may cause fetal harm (8.1)
8.6 Cyp2c19 Poor Metabolizers
Concentrations of clobazam's active metabolite, N-desmethylclobazam, are higher in CYP2C19 poor metabolizers than in extensive metabolizers. For this reason, dosage modification is recommended [see Dosage and Administration (2.5), Clinical Pharmacology (12.3)].
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
During its development for the adjunctive treatment of seizures associated with LGS, clobazam was administered to 333 healthy volunteers and 300 patients with a current or prior diagnosis of LGS, including 197 patients treated for 12 months or more. The conditions and duration of exposure varied greatly and included single- and multiple-dose clinical pharmacology studies in healthy volunteers and two double-blind studies in patients with LGS (Study 1 and 2) [see Clinical Studies (14)]. Only Study 1 included a placebo group, allowing comparison of adverse reaction rates on clobazam at several doses to placebo.
7.2 Cns Depressants and Alcohol
Concomitant use of clobazam with other CNS depressants may increase the risk of sedation and somnolence [see Warnings and Precautions (5.2)].
Alcohol, as a CNS depressant, will interact with clobazam in a similar way and also increases clobazam's maximum plasma exposure by approximately 50%. Therefore, caution patients or their caregivers against simultaneous use with other CNS depressant drugs or alcohol, and caution that the effects of other CNS depressant drugs or alcohol may be potentiated [see Warnings and Precautions (5.2)].
17 Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Medication Guide).
2.6 Patients With Renal Impairment
No dose adjustment is required for patients with mild and moderate renal impairment. There is no experience with clobazam tablets in patients with severe renal impairment or end stage renal disease (ESRD). It is not known if clobazam or its active metabolite, N-desmethylclobazam, is dialyzable [see Use in Specific Populations (8.7), Clinical Pharmacology (12.3)].
5.7 Suicidal Behavior and Ideation
Antiepileptic drugs (AEDs), including clobazam, increase the risk of suicidal thoughts or behavior in patients taking these drugs for any indication. Patients treated with any AED for any indication should be monitored for the emergence or worsening of depression, suicidal thoughts or behavior, and/or any unusual changes in mood or behavior.
Pooled analyses of 199 placebo-controlled clinical trials (mono- and adjunctive therapy) of 11 different AEDs showed that patients randomized to one of the AEDs had approximately twice the risk (adjusted relative risk 1.8, 95% confidence interval [CI]: 1.2, 2.7) of suicidal thinking or behavior compared to patients randomized to placebo. In these trials, which had a median treatment duration of 12 weeks, the estimated incidence rate of suicidal behavior or ideation among 27,863 AED-treated patients was 0.43%, compared to 0.24% among 16,029 placebo-treated patients, representing an increase of approximately one case of suicidal thinking or behavior for every 530 patients treated. There were four suicides in drug-treated patients in the trials and none in placebo-treated patients, but the number is too small to allow any conclusion about drug effect on suicide.
The increased risk of suicidal thoughts or behavior with AEDs was observed as early as one week after starting drug treatment with AEDs and persisted for the duration of treatment assessed. Because most trials included in the analysis did not extend beyond 24 weeks, the risk of suicidal thoughts or behavior beyond 24 weeks could not be assessed.
The risk of suicidal thoughts or behavior was generally consistent among drugs in the data analyzed. The finding of increased risk with AEDs of varying mechanisms of action and across a range of indications suggests that the risk applies to all AEDs used for any indication. The risk did not vary substantially by age (5 years to 100 years) in the clinical trials analyzed. Table 2 shows absolute and relative risk by indication for all evaluated AEDs.
| Indication | Placebo Patients with Events per 1000 Patients | Drug Patients with Events per 1000 Patients | Relative Risk: Incidence of Drug Events in Drug Patients/Incidence in Placebo Patients | Risk Difference: Additional Drug Patients with Events per 1000 Patients |
|---|---|---|---|---|
| Epilepsy | 1.0 | 3.4 | 3.5 | 2.4 |
| Psychiatric | 5.7 | 8.5 | 1.5 | 2.9 |
| Other | 1.0 | 1.8 | 1.9 | 0.9 |
| Total | 2.4 | 4.3 | 1.8 | 1.9 |
The relative risk for suicidal thoughts or behavior was higher in clinical trials for epilepsy than in clinical trials for psychiatric or other conditions, but the absolute risk differences were similar for the epilepsy and psychiatric indications.
Anyone considering prescribing clobazam or any other AED must balance the risk of suicidal thoughts or behavior with the risk of untreated illness. Epilepsy and many other illnesses for which AEDs are prescribed are themselves associated with morbidity and mortality and an increased risk of suicidal thoughts and behavior. Should suicidal thoughts and behavior emerge during treatment, the prescriber needs to consider whether the emergence of these symptoms in any given patient may be related to the illness being treated.
Patients, their caregivers, and families should be informed that AEDs increase the risk of suicidal thoughts and behavior and should be advised of the need to be alert for the emergence or worsening of the signs and symptoms of depression, any unusual changes in mood or behavior, or the emergence of suicidal thoughts, behavior, or thoughts about self-harm. Behaviors of concern should be reported immediately to healthcare providers.
16 How Supplied/storage and Handling
Each clobazam tablet contains 10 mg or 20 mg of clobazam and is a white to off-white, caplet shape tablet functionally-scored on both the sides debossed with "T" over the score and "10" under the score or a "T" over the score and "20" under the score on one side and plain on the other side.
NDC 51672-4202-1: 10 mg scored tablet, Bottles of 100
NDC 51672-4203-1: 20 mg scored tablet, Bottles of 100
5.5 Serious Dermatological Reactions
Serious skin reactions, including Stevens-Johnson syndrome (SJS) and toxic epidermal necrolysis (TEN), have been reported with clobazam in both children and adults during the postmarketing period. Patients should be closely monitored for signs or symptoms of SJS/TEN, especially during the first 8 weeks of treatment initiation or when re-introducing therapy. Clobazam should be discontinued at the first sign of rash, unless the rash is clearly not drug-related. If signs or symptoms suggest SJS/TEN, use of this drug should not be resumed and alternative therapy should be considered [see Contraindications (4)].
10.1 Signs and Symptoms of Overdosage
Overdose and intoxication with benzodiazepines, including clobazam, may lead to CNS depression, associated with drowsiness, confusion and lethargy, possibly progressing to ataxia, respiratory depression, hypotension, and, rarely, coma or death. The risk of a fatal outcome is increased in cases of combined poisoning with other CNS depressants, including opioids and alcohol.
5.6 Physical and Psychological Dependence
Patients with a history of substance abuse should be under careful surveillance when receiving clobazam or other psychotropic agents because of the predisposition of such patients to habituation and dependence [see Drug Abuse and Dependence (9)].
5.1 Risks From Concomitant Use With Opioids
Concomitant use of benzodiazepines, including clobazam, and opioids may result in profound sedation, respiratory depression, coma, and death. Because of these risks, reserve concomitant prescribing of benzodiazepines and opioids for use in patients for whom alternative treatment options are inadequate.
Observational studies have demonstrated that concomitant use of opioid analgesics and benzodiazepines increases the risk of drug-related mortality compared to use of opioids alone. If a decision is made to prescribe clobazam concomitantly with opioids, prescribe the lowest effective dosages and minimum durations of concomitant use, and follow patients closely for signs and symptoms of respiratory depression and sedation. Advise both patients and caregivers about the risks of respiratory depression and sedation when clobazam is used with opioids [see Drug Interactions (7.1)].
2.4 Dosage Adjustments in Geriatric Patients
Plasma concentrations at any given dose are generally higher in the elderly: proceed slowly with dose escalation. The starting dose should be 5 mg/day for all elderly patients. Then titrate elderly patients according to weight, but to half the dose presented in Table 1, as tolerated. If necessary and based upon clinical response, an additional titration to the maximum dose (20 mg/day or 40 mg/day, depending on weight) may be started on day 21 [see Use in Specific Populations (8.5)].
8.3 Females and Males of Reproductive Potential
Administration of clobazam to rats prior to and during mating and early gestation resulted in adverse effects on fertility and early embryonic development at plasma exposures for clobazam and its major active metabolite, N-desmethylclobazam, below those in humans at the MRHD [see Nonclinical Toxicology (13.1)].
Warning: Risks From Concomitant Use With Opioids
Concomitant use of benzodiazepines and opioids may result in profound sedation, respiratory depression, coma, and death [see Warnings and Precautions (5.1), Drug Interactions (7.1)].
- Reserve concomitant prescribing of these drugs for use in patients for whom alternative treatment options are inadequate.
- Limit dosages and durations to the minimum required.
- Follow patients for signs and symptoms of respiratory depression and sedation.
2.5 Dosage Adjustments in Cyp2c19 Poor Metabolizers
In CYP2C19 poor metabolizers, levels of N-desmethylclobazam, clobazam's active metabolite, will be increased. Therefore, in patients known to be CYP2C19 poor metabolizers, the starting dose should be 5 mg/day and dose titration should proceed slowly according to weight, but to half the dose presented in Table 1, as tolerated. If necessary and based upon clinical response, an additional titration to the maximum dose (20 mg/day or 40 mg/day, depending on the weight group) may be started on day 21 [see Use in Specific Populations (8.6), Clinical Pharmacology (12.5)].
Principal Display Panel 10 Mg Tablet Bottle Label
NDC 51672-4202-1
100 Tablets
Clobazam
Tablets
CIV
10 mg
DISPENSE THE ENCLOSED MEDICATION
GUIDE WITH EACH PRESCRIPTION.
Rx only
Principal Display Panel 20 Mg Tablet Bottle Label
NDC 51672-4203-1
100 Tablets
CIV
Clobazam
Tablets
20 mg
DISPENSE THE ENCLOSED MEDICATION GUIDE
WITH EACH PRESCRIPTION.
Rx only
2.7 Dosage Adjustments in Patients With Hepatic Impairment
Clobazam is hepatically metabolized; however, there are limited data to characterize the effect of hepatic impairment on the pharmacokinetics of clobazam. For this reason, proceed slowly with dosing escalations. For patients with mild to moderate hepatic impairment (Child-Pugh score 5 to 9), the starting dose should be 5 mg/day in both weight groups. Then titrate patients according to weight, but to half the dose presented in Table 1, as tolerated. If necessary and based upon clinical response, start an additional titration on day 21 to the maximum dose (20 mg/day or 40 mg/day, depending on the weight group). There is inadequate information about metabolism of clobazam in patients with severe hepatic impairment. Therefore no dosing recommendation in those patients can be given [see Use in Specific Populations (8.8), Clinical Pharmacology (12.3)].
5.2 Potentiation of Sedation From Concomitant Use With Central Nervous System Depressants
Since clobazam has a central nervous system (CNS) depressant effect, patients or their caregivers should be cautioned against simultaneous use with other CNS depressant drugs or alcohol, and cautioned that the effects of other CNS depressant drugs or alcohol may be potentiated [see Drug Interactions (7.2)].
Structured Label Content
Section 42229-5 (42229-5)
Clobazam Tablet Oral Administration
Clobazam tablets can be taken with or without food.
Clobazam tablets can be administered whole, broken in half along the score, or crushed and mixed in applesauce.
Section 42231-1 (42231-1)
| This Medication Guide has been approved by the U.S. Food and Drug Administration. | ||
| Revised: June 2018 | 20999-0618-1 | |
|
MEDICATION GUIDE CIV Clobazam (KLOE ba zam) Tablets |
||
|
What is the most important information I should know about clobazam tablets?
Call your healthcare provider right away if you have any of these symptoms, especially if they are new, worse, or worry you: |
||
|
|
|
|
How can I watch for early symptoms of suicidal thoughts and actions?
Call your healthcare provider between visits as needed, especially if you are worried about symptoms. Suicidal thoughts or actions can be caused by things other than medicines. If you have suicidal thoughts or actions, your healthcare provider may check for other causes. |
||
|
What are clobazam tablets? Clobazam tablet is a prescription medicine used along with other medicines to treat seizures associated with Lennox-Gastaut syndrome in people 2 years of age or older. It is not known if clobazam tablets are safe and effective in children less than 2 years old. |
||
|
Do not take clobazam tablets if you:
|
||
|
Before you take clobazam tablets, tell your healthcare provider about all your medical conditions, including if you:
Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. Taking clobazam tablets with certain other medicines can cause side effects or affect how well clobazam tablets or the other medicines work. Do not start or stop other medicines without talking to your healthcare provider. |
||
|
How should I take clobazam tablets?
|
||
|
What should I avoid while taking clobazam tablets?
|
||
|
What are the possible side effects of clobazam tablets? Clobazam tablets may cause serious side effects, including: See "What is the most important information I should know about clobazam tablets?" The most common side effects of clobazam tablets include: |
||
|
|
|
|
These are not all the possible side effects of clobazam tablets. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
||
|
How should I store clobazam tablets?
|
||
|
General information about the safe and effective use of clobazam tablets. Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use clobazam tablets for a condition for which it was not prescribed. Do not give clobazam tablets to other people, even if they have the same symptoms that you have. It may harm them. You can ask your pharmacist or healthcare provider for information about clobazam tablets that is written for health professionals. |
||
|
What are the ingredients in clobazam tablets? Active ingredient: clobazam Inactive ingredients: colloidal silicone dioxide, lactose monohydrate, magnesium stearate, pregelatinized maize starch, and talc. Mfd. by: For more information about clobazam tablets, go to www.taro.com or call Taro Pharmaceuticals U.S.A., Inc. at 1-866-923-4914. |
Section 44425-7 (44425-7)
Store tablets at 20°C to 25°C (68°F to 77°F) [See USP Controlled Room Temperature].
9.2 Abuse
Clobazam can be abused in a similar manner as other benzodiazepines, such as diazepam.
The pharmacological profile of clobazam is similar to that of other benzodiazepines listed in Schedule IV of the Controlled Substance Act, particularly in its potentiation of GABAergic transmission through its action on GABAA receptors, which leads to sedation and somnolence.
The World Health Organization epidemiology database contains reports of drug abuse, misuse, and overdoses associated with clobazam.
Drug abuse is the intentional non-therapeutic use of a drug, repeatedly or even sporadically, for its rewarding psychological or physiological effects.
7.1 Opioids
The concomitant use of benzodiazepines and opioids increases the risk of respiratory depression because of actions at different receptor sites in the CNS that control respiration. Benzodiazepines interact at GABAA sites, and opioids interact primarily at mu receptors. When benzodiazepines and opioids are combined, the potential for benzodiazepines to significantly worsen opioid-related respiratory depression exists. Limit dosage and duration of concomitant use of benzodiazepines and opioids, and follow patients closely for respiratory depression and sedation [see Warnings and Precautions (5.1)].
11 Description (11 DESCRIPTION)
| Established Name: | Clobazam Tablets |
| Dosage Form: | Tablet |
| Route of Administration: | Oral |
| Established Pharmacologic Class of Drug: | Benzodiazepine |
| Chemical Name: | 7-Chloro-1-methyl-5-phenyl-1H-1,5 benzodiazepine-2,4(3H,5H)-dione |
| Structural Formula: |
|
Clobazam is a white or almost white, crystalline powder with a slightly bitter taste; is slightly soluble in water, sparingly soluble in ethanol, and freely soluble in methylene chloride. The melting range of clobazam is from 182°C to 185°C. The molecular formula is C16H13O2N2Cl and the molecular weight is 300.7.
Each clobazam tablet contains 10 mg or 20 mg of clobazam. Tablets also contain as inactive ingredients: colloidal silicone dioxide, lactose monohydrate, magnesium stearate, pregelatinized maize starch, and talc.
8.4 Pediatric Use
Safety and effectiveness in patients less than 2 years of age have not been established.
In a study in which clobazam (0 mg/kg/day, 4 mg/kg/day, 36 mg/kg/day, or 120 mg/kg/day) was orally administered to rats during the juvenile period of development (postnatal days 14 to 48), adverse effects on growth (decreased bone density and bone length) and behavior (altered motor activity and auditory startle response; learning deficit) were observed at the high dose. The effect on bone density, but not on behavior, was reversible when drug was discontinued. The no-effect level for juvenile toxicity (36 mg/kg/day) was associated with plasma exposures (AUC) to clobazam and its major active metabolite, N-desmethylclobazam, less than those expected at therapeutic doses in pediatric patients.
8.5 Geriatric Use
Clinical studies of clobazam did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects. However, elderly subjects appear to eliminate clobazam more slowly than younger subjects based on population pharmacokinetic analysis. For these reasons, the initial dose in elderly patients should be 5 mg/day. Patients should be titrated initially to 10 mg/day to 20 mg/day. Patients may be titrated further to a maximum daily dose of 40 mg if tolerated [see Dosage and Administration (2.4), Clinical Pharmacology (12.3)].
14 Clinical Studies (14 CLINICAL STUDIES)
The effectiveness of clobazam for the adjunctive treatment of seizures associated with Lennox-Gastaut syndrome was established in two multicenter controlled studies (Study 1 and Study 2). Both studies were similar in terms of disease characteristics and concomitant AED treatments. The most common concomitant AED treatments at baseline included: valproate, lamotrigine, levetiracetam, and topiramate.
4 Contraindications (4 CONTRAINDICATIONS)
Clobazam tablets are contraindicated in patients with a history of hypersensitivity to the drug or its ingredients. Hypersensitivity reactions have included serious dermatological reactions [see Warnings and Precautions (5.5)].
6 Adverse Reactions (6 ADVERSE REACTIONS)
Clinically significant adverse reactions that appear in other sections of the labeling include the following:
- Risks from Concomitant Use with Opioids [see Warnings and Precautions (5.1)]
- Potentiation of Sedation from Concomitant Use with Central Nervous System Depressants [see Warnings and Precautions (5.2)]
- Somnolence or Sedation [see Warnings and Precautions (5.3)]
- Withdrawal Symptoms [see Warnings and Precautions (5.4)]
- Serious Dermatological Reactions [see Contraindications (4), Warnings and Precautions (5.5)]
- Physical and Psychological Dependence [see Warnings and Precautions (5.6)]
- Suicidal Behavior and Ideation [see Warnings and Precautions (5.7)]
7 Drug Interactions (7 DRUG INTERACTIONS)
8.7 Renal Impairment
The pharmacokinetics of clobazam were evaluated in patients with mild and moderate renal impairment. There were no significant differences in systemic exposure (AUC and Cmax) between patients with mild or moderate renal impairment and healthy subjects. No dose adjustment is required for patients with mild and moderate renal impairment. There is essentially no experience with clobazam in patients with severe renal impairment or ESRD. It is not known if clobazam or its active metabolite, N-desmethylclobazam, is dialyzable [see Dosage and Administration (2.6), Clinical Pharmacology (12.3)].
12.3 Pharmacokinetics
The peak plasma levels (Cmax) and the area under the curve (AUC) of clobazam are dose-proportional over the dose range of 10 mg to 80 mg following single- or multiple-dose administration of clobazam. Based on a population pharmacokinetic analysis, the pharmacokinetics of clobazam are linear from 5 mg/day to 160 mg/day. Clobazam is converted to N-desmethylclobazam which has about 1/5 the activity of clobazam. The estimated mean elimination half-lives (t½) of clobazam and N-desmethylclobazam were 36 hours to 42 hours and 71 hours to 82 hours, respectively.
12.5 Pharmacogenomics
The polymorphic CYP2C19 is the main enzyme that metabolizes the pharmacologically active N-desmethylclobazam. Compared to CYP2C19 extensive metabolizers, N-desmethylclobazam AUC and Cmax are approximately 3 times to 5 times higher in poor metabolizers (e.g., subjects with *2/*2 genotype) and 2 times higher in intermediate metabolizers (e.g., subjects with *1/*2 genotype). The prevalence of CYP2C19 poor metabolism differs depending on racial/ethnic background. Dosage in patients who are known CYP2C19 poor metabolizers may need to be adjusted [see Dosage and Administration (2.5)].
The systemic exposure of clobazam is similar for both CYP2C19 poor and extensive metabolizers.
2.1 Dosing Information
A daily dose of clobazam tablets greater than 5 mg should be administered in divided doses twice daily; a 5 mg daily dose can be administered as a single dose. Dose patients according to body weight. Individualize dosing within each body weight group, based on clinical efficacy and tolerability. Each dose in Table 1 (e.g., 5 mg to 20 mg in ≤30 kg weight group) has been shown to be effective, although effectiveness increases with increasing dose [see Clinical Studies (14)]. Do not proceed with dose escalation more rapidly than weekly, because serum concentrations of clobazam and its active metabolite require 5 and 9 days, respectively, to reach steady-state.
| ≤30 kg Body Weight | >30 kg Body Weight | |
|---|---|---|
| Starting Dose | 5 mg | 10 mg |
| Starting Day 7 | 10 mg | 20 mg |
| Starting Day 14 | 20 mg | 40 mg |
2.2 Gradual Withdrawal
As with all antiepileptic drugs and benzodiazepines, withdraw clobazam tablets gradually. Taper by decreasing the total daily dose by 5 mg/day to 10 mg/day on a weekly basis until discontinued [see Warnings and Precautions (5.4)].
8.8 Hepatic Impairment
Clobazam is hepatically metabolized; however, there are limited data to characterize the effect of hepatic impairment on the pharmacokinetics of clobazam. For this reason, dosage adjustment is recommended in patients with mild to moderate hepatic impairment (Child-Pugh score 5 to 9). There is inadequate information about metabolism of clobazam in patients with severe hepatic impairment [see Dosage and Administration (2.7), Clinical Pharmacology (12.3)].
1 Indications and Usage (1 INDICATIONS AND USAGE)
Clobazam tablets are indicated for the adjunctive treatment of seizures associated with Lennox-Gastaut syndrome (LGS) in patients 2 years of age or older.
5.4 Withdrawal Symptoms
Abrupt discontinuation of clobazam should be avoided. Clobazam should be tapered by decreasing the dose every week by 5 mg/day to 10 mg/day until discontinuation [see Dosage and Administration (2.2)].
Withdrawal symptoms occurred following abrupt discontinuation of clobazam; the risk of withdrawal symptoms is greater with higher doses.
As with all antiepileptic drugs, clobazam should be withdrawn gradually to minimize the risk of precipitating seizures, seizure exacerbation, or status epilepticus.
Withdrawal symptoms (e.g., convulsions, psychosis, hallucinations, behavioral disorder, tremor, and anxiety) have been reported following abrupt discontinuance of benzodiazepines. The more severe withdrawal symptoms have usually been limited to patients who received excessive doses over an extended period of time, followed by an abrupt discontinuation. Generally milder withdrawal symptoms (e.g., dysphoria, anxiety, and insomnia) have been reported following abrupt discontinuance of benzodiazepines taken continuously at therapeutic doses for several months.
12.1 Mechanism of Action
The exact mechanism of action for clobazam, a 1,5-benzodiazepine, is not fully understood but is thought to involve potentiation of GABAergic neurotransmission resulting from binding at the benzodiazepine site of the GABAA receptor.
9.1 Controlled Substance
Clobazam tablets contains clobazam which is a Schedule IV controlled substance.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Somnolence or Sedation: Monitor for central nervous system (CNS) depression. Risk may be increased with concomitant use of other CNS depressants (5.2, 5.3)
- Withdrawal: Symptoms may occur with rapid dose reduction or discontinuation. Discontinue clobazam gradually (5.4)
- Serious Dermatological Reactions (including Stevens-Johnson syndrome and toxic epidermal necrolysis): Discontinue clobazam at first sign of rash unless the rash is clearly not drug-related (5.5)
- Physical and Psychological Dependence: Monitor patients with a history of substance abuse for signs of habituation and dependence (5.6, 9)
- Suicidal Behavior and Ideation: Monitor for suicidal thoughts or behaviors (5.7)
5.3 Somnolence Or Sedation (5.3 Somnolence or Sedation)
Clobazam causes somnolence and sedation. In clinical trials, somnolence or sedation was reported at all effective doses and was dose-related.
In general, somnolence and sedation begin within the first month of treatment and may diminish with continued treatment. Prescribers should monitor patients for somnolence and sedation, particularly with concomitant use of other central nervous system depressants. Prescribers should caution patients against engaging in hazardous activities requiring mental alertness, such as operating dangerous machinery or motor vehicles, until the effect of clobazam is known.
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
- For doses above 5 mg/day administer in two divided doses (2.1)
- Patients ≤30 kg body weight: Initiate at 5 mg daily and titrate as tolerated up to 20 mg daily (2.1)
- Patients >30 kg body weight: Initiate at 10 mg daily and titrate as tolerated up to 40 mg daily (2.1)
- Dosage adjustment needed in following groups:
- Reduce dose, or discontinue drug gradually (2.2)
- Administer whole, broken in half along the score, or crush and mix in applesauce (2.3)
- Can be taken with or without food (2.3)
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Tablets: 10 mg and 20 mg with a functional score for oral administration.
Each clobazam tablet is a white to off-white, caplet shape tablet functionally-scored on both sides debossed with "T" over the score and "10" under the score or a "T" over the score and "20" under the score on one side and plain on the other side.
6.2 Postmarketing Experience
These reactions are reported voluntarily from a population of uncertain size; therefore, it is not possible to estimate their frequency or establish a causal relationship to drug exposure. Adverse reactions are categorized by system organ class.
Blood Disorders: Anemia, eosinophilia, leukopenia, thrombocytopenia
Eye Disorders: Diplopia, vision blurred
Gastrointestinal Disorders: Abdominal distention
General Disorders and Administration Site Conditions: Hypothermia
Investigations: Hepatic enzyme increased
Musculoskeletal: Muscle spasms
Psychiatric Disorders: Agitation, anxiety, apathy, confusional state, depression, delirium, delusion, hallucination
Renal and Urinary Disorders: Urinary retention
Respiratory Disorders: Aspiration, respiratory depression
Skin and Subcutaneous Tissue Disorders: Rash, urticaria, angioedema, and facial and lip edema
10.2 Management of Overdosage
The management of clobazam overdose may include gastric lavage and/or administration of activated charcoal, intravenous fluid replenishment, early control of airway and general supportive measures, in addition to monitoring level of consciousness and vital signs. Hypotension can be treated by replenishment with plasma substitutes and, if necessary, with sympathomimetic agents.
The efficacy of supplementary administration of physostigmine (a cholinergic agent) or of flumazenil (a benzodiazepine antagonist) in clobazam overdose has not been assessed. The administration of flumazenil in cases of benzodiazepine overdose can lead to withdrawal and adverse reactions. Its use in patients with epilepsy is typically not recommended.
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
Pregnancy: Based on animal data, may cause fetal harm (8.1)
8.6 Cyp2c19 Poor Metabolizers (8.6 CYP2C19 Poor Metabolizers)
Concentrations of clobazam's active metabolite, N-desmethylclobazam, are higher in CYP2C19 poor metabolizers than in extensive metabolizers. For this reason, dosage modification is recommended [see Dosage and Administration (2.5), Clinical Pharmacology (12.3)].
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
During its development for the adjunctive treatment of seizures associated with LGS, clobazam was administered to 333 healthy volunteers and 300 patients with a current or prior diagnosis of LGS, including 197 patients treated for 12 months or more. The conditions and duration of exposure varied greatly and included single- and multiple-dose clinical pharmacology studies in healthy volunteers and two double-blind studies in patients with LGS (Study 1 and 2) [see Clinical Studies (14)]. Only Study 1 included a placebo group, allowing comparison of adverse reaction rates on clobazam at several doses to placebo.
7.2 Cns Depressants and Alcohol (7.2 CNS Depressants and Alcohol)
Concomitant use of clobazam with other CNS depressants may increase the risk of sedation and somnolence [see Warnings and Precautions (5.2)].
Alcohol, as a CNS depressant, will interact with clobazam in a similar way and also increases clobazam's maximum plasma exposure by approximately 50%. Therefore, caution patients or their caregivers against simultaneous use with other CNS depressant drugs or alcohol, and caution that the effects of other CNS depressant drugs or alcohol may be potentiated [see Warnings and Precautions (5.2)].
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient to read the FDA-approved patient labeling (Medication Guide).
2.6 Patients With Renal Impairment (2.6 Patients with Renal Impairment)
No dose adjustment is required for patients with mild and moderate renal impairment. There is no experience with clobazam tablets in patients with severe renal impairment or end stage renal disease (ESRD). It is not known if clobazam or its active metabolite, N-desmethylclobazam, is dialyzable [see Use in Specific Populations (8.7), Clinical Pharmacology (12.3)].
5.7 Suicidal Behavior and Ideation
Antiepileptic drugs (AEDs), including clobazam, increase the risk of suicidal thoughts or behavior in patients taking these drugs for any indication. Patients treated with any AED for any indication should be monitored for the emergence or worsening of depression, suicidal thoughts or behavior, and/or any unusual changes in mood or behavior.
Pooled analyses of 199 placebo-controlled clinical trials (mono- and adjunctive therapy) of 11 different AEDs showed that patients randomized to one of the AEDs had approximately twice the risk (adjusted relative risk 1.8, 95% confidence interval [CI]: 1.2, 2.7) of suicidal thinking or behavior compared to patients randomized to placebo. In these trials, which had a median treatment duration of 12 weeks, the estimated incidence rate of suicidal behavior or ideation among 27,863 AED-treated patients was 0.43%, compared to 0.24% among 16,029 placebo-treated patients, representing an increase of approximately one case of suicidal thinking or behavior for every 530 patients treated. There were four suicides in drug-treated patients in the trials and none in placebo-treated patients, but the number is too small to allow any conclusion about drug effect on suicide.
The increased risk of suicidal thoughts or behavior with AEDs was observed as early as one week after starting drug treatment with AEDs and persisted for the duration of treatment assessed. Because most trials included in the analysis did not extend beyond 24 weeks, the risk of suicidal thoughts or behavior beyond 24 weeks could not be assessed.
The risk of suicidal thoughts or behavior was generally consistent among drugs in the data analyzed. The finding of increased risk with AEDs of varying mechanisms of action and across a range of indications suggests that the risk applies to all AEDs used for any indication. The risk did not vary substantially by age (5 years to 100 years) in the clinical trials analyzed. Table 2 shows absolute and relative risk by indication for all evaluated AEDs.
| Indication | Placebo Patients with Events per 1000 Patients | Drug Patients with Events per 1000 Patients | Relative Risk: Incidence of Drug Events in Drug Patients/Incidence in Placebo Patients | Risk Difference: Additional Drug Patients with Events per 1000 Patients |
|---|---|---|---|---|
| Epilepsy | 1.0 | 3.4 | 3.5 | 2.4 |
| Psychiatric | 5.7 | 8.5 | 1.5 | 2.9 |
| Other | 1.0 | 1.8 | 1.9 | 0.9 |
| Total | 2.4 | 4.3 | 1.8 | 1.9 |
The relative risk for suicidal thoughts or behavior was higher in clinical trials for epilepsy than in clinical trials for psychiatric or other conditions, but the absolute risk differences were similar for the epilepsy and psychiatric indications.
Anyone considering prescribing clobazam or any other AED must balance the risk of suicidal thoughts or behavior with the risk of untreated illness. Epilepsy and many other illnesses for which AEDs are prescribed are themselves associated with morbidity and mortality and an increased risk of suicidal thoughts and behavior. Should suicidal thoughts and behavior emerge during treatment, the prescriber needs to consider whether the emergence of these symptoms in any given patient may be related to the illness being treated.
Patients, their caregivers, and families should be informed that AEDs increase the risk of suicidal thoughts and behavior and should be advised of the need to be alert for the emergence or worsening of the signs and symptoms of depression, any unusual changes in mood or behavior, or the emergence of suicidal thoughts, behavior, or thoughts about self-harm. Behaviors of concern should be reported immediately to healthcare providers.
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
Each clobazam tablet contains 10 mg or 20 mg of clobazam and is a white to off-white, caplet shape tablet functionally-scored on both the sides debossed with "T" over the score and "10" under the score or a "T" over the score and "20" under the score on one side and plain on the other side.
NDC 51672-4202-1: 10 mg scored tablet, Bottles of 100
NDC 51672-4203-1: 20 mg scored tablet, Bottles of 100
5.5 Serious Dermatological Reactions
Serious skin reactions, including Stevens-Johnson syndrome (SJS) and toxic epidermal necrolysis (TEN), have been reported with clobazam in both children and adults during the postmarketing period. Patients should be closely monitored for signs or symptoms of SJS/TEN, especially during the first 8 weeks of treatment initiation or when re-introducing therapy. Clobazam should be discontinued at the first sign of rash, unless the rash is clearly not drug-related. If signs or symptoms suggest SJS/TEN, use of this drug should not be resumed and alternative therapy should be considered [see Contraindications (4)].
10.1 Signs and Symptoms of Overdosage
Overdose and intoxication with benzodiazepines, including clobazam, may lead to CNS depression, associated with drowsiness, confusion and lethargy, possibly progressing to ataxia, respiratory depression, hypotension, and, rarely, coma or death. The risk of a fatal outcome is increased in cases of combined poisoning with other CNS depressants, including opioids and alcohol.
5.6 Physical and Psychological Dependence
Patients with a history of substance abuse should be under careful surveillance when receiving clobazam or other psychotropic agents because of the predisposition of such patients to habituation and dependence [see Drug Abuse and Dependence (9)].
5.1 Risks From Concomitant Use With Opioids (5.1 Risks from Concomitant Use with Opioids)
Concomitant use of benzodiazepines, including clobazam, and opioids may result in profound sedation, respiratory depression, coma, and death. Because of these risks, reserve concomitant prescribing of benzodiazepines and opioids for use in patients for whom alternative treatment options are inadequate.
Observational studies have demonstrated that concomitant use of opioid analgesics and benzodiazepines increases the risk of drug-related mortality compared to use of opioids alone. If a decision is made to prescribe clobazam concomitantly with opioids, prescribe the lowest effective dosages and minimum durations of concomitant use, and follow patients closely for signs and symptoms of respiratory depression and sedation. Advise both patients and caregivers about the risks of respiratory depression and sedation when clobazam is used with opioids [see Drug Interactions (7.1)].
2.4 Dosage Adjustments in Geriatric Patients
Plasma concentrations at any given dose are generally higher in the elderly: proceed slowly with dose escalation. The starting dose should be 5 mg/day for all elderly patients. Then titrate elderly patients according to weight, but to half the dose presented in Table 1, as tolerated. If necessary and based upon clinical response, an additional titration to the maximum dose (20 mg/day or 40 mg/day, depending on weight) may be started on day 21 [see Use in Specific Populations (8.5)].
8.3 Females and Males of Reproductive Potential
Administration of clobazam to rats prior to and during mating and early gestation resulted in adverse effects on fertility and early embryonic development at plasma exposures for clobazam and its major active metabolite, N-desmethylclobazam, below those in humans at the MRHD [see Nonclinical Toxicology (13.1)].
Warning: Risks From Concomitant Use With Opioids (WARNING: RISKS FROM CONCOMITANT USE WITH OPIOIDS)
Concomitant use of benzodiazepines and opioids may result in profound sedation, respiratory depression, coma, and death [see Warnings and Precautions (5.1), Drug Interactions (7.1)].
- Reserve concomitant prescribing of these drugs for use in patients for whom alternative treatment options are inadequate.
- Limit dosages and durations to the minimum required.
- Follow patients for signs and symptoms of respiratory depression and sedation.
2.5 Dosage Adjustments in Cyp2c19 Poor Metabolizers (2.5 Dosage Adjustments in CYP2C19 Poor Metabolizers)
In CYP2C19 poor metabolizers, levels of N-desmethylclobazam, clobazam's active metabolite, will be increased. Therefore, in patients known to be CYP2C19 poor metabolizers, the starting dose should be 5 mg/day and dose titration should proceed slowly according to weight, but to half the dose presented in Table 1, as tolerated. If necessary and based upon clinical response, an additional titration to the maximum dose (20 mg/day or 40 mg/day, depending on the weight group) may be started on day 21 [see Use in Specific Populations (8.6), Clinical Pharmacology (12.5)].
Principal Display Panel 10 Mg Tablet Bottle Label (PRINCIPAL DISPLAY PANEL - 10 mg Tablet Bottle Label)
NDC 51672-4202-1
100 Tablets
Clobazam
Tablets
CIV
10 mg
DISPENSE THE ENCLOSED MEDICATION
GUIDE WITH EACH PRESCRIPTION.
Rx only
Principal Display Panel 20 Mg Tablet Bottle Label (PRINCIPAL DISPLAY PANEL - 20 mg Tablet Bottle Label)
NDC 51672-4203-1
100 Tablets
CIV
Clobazam
Tablets
20 mg
DISPENSE THE ENCLOSED MEDICATION GUIDE
WITH EACH PRESCRIPTION.
Rx only
2.7 Dosage Adjustments in Patients With Hepatic Impairment (2.7 Dosage Adjustments in Patients with Hepatic Impairment)
Clobazam is hepatically metabolized; however, there are limited data to characterize the effect of hepatic impairment on the pharmacokinetics of clobazam. For this reason, proceed slowly with dosing escalations. For patients with mild to moderate hepatic impairment (Child-Pugh score 5 to 9), the starting dose should be 5 mg/day in both weight groups. Then titrate patients according to weight, but to half the dose presented in Table 1, as tolerated. If necessary and based upon clinical response, start an additional titration on day 21 to the maximum dose (20 mg/day or 40 mg/day, depending on the weight group). There is inadequate information about metabolism of clobazam in patients with severe hepatic impairment. Therefore no dosing recommendation in those patients can be given [see Use in Specific Populations (8.8), Clinical Pharmacology (12.3)].
5.2 Potentiation of Sedation From Concomitant Use With Central Nervous System Depressants (5.2 Potentiation of Sedation from Concomitant Use with Central Nervous System Depressants)
Since clobazam has a central nervous system (CNS) depressant effect, patients or their caregivers should be cautioned against simultaneous use with other CNS depressant drugs or alcohol, and cautioned that the effects of other CNS depressant drugs or alcohol may be potentiated [see Drug Interactions (7.2)].
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:34:23.813656 · Updated: 2026-03-14T21:40:30.329146