These Highlights Do Not Include All The Information Needed To Use Donepezil Hydrochloride Tablets Safely And Effectively. See Full Prescribing Information For Donepezil Hydrochloride Tablets.
c26d9bf3-e148-48bb-8f4f-e8c73804b311
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Donepezil hydrochloride is indicated for the treatment of dementia of the Alzheimer's type. Efficacy has been demonstrated in patients with mild, moderate, and severe Alzheimer's disease.
Indications and Usage
Donepezil hydrochloride is indicated for the treatment of dementia of the Alzheimer's type. Efficacy has been demonstrated in patients with mild, moderate, and severe Alzheimer's disease.
Dosage and Administration
• Mild to Moderate Alzheimer's disease : 5 mg to 10 mg administered once daily ( 2.1 ) • Moderate to Severe Alzheimer’s Disease : 10 mg administered once daily ( 2.2)
Warnings and Precautions
• Cholinesterase inhibitors are likely to exaggerate succinylcholine-type muscle relaxation during anesthesia ( 5.1 ). • Cholinesterase inhibitors may have vagotonic effects on the sinoatrial and atrioventricular nodes manifesting as bradycardia or heart block ( 5.2 ). • Donepezil hydrochloride can cause vomiting. Patients should be observed closely at initiation of treatment and after dose increases ( 5.3 ). • Patients should be monitored closely for symptoms of active or occult gastrointestinal (GI) bleeding, especially those at increased risk for developing ulcers ( 5.4 ). • Cholinomimetics may cause bladder outflow obstructions ( 5.6 ). • Cholinomimetics are believed to have some potential to cause generalized convulsions ( 5.7 ). • Cholinesterase inhibitors should be prescribed with care to patients with a history of asthma or obstructive pulmonary disease ( 5.8 ).
Contraindications
Donepezil hydrochloride is contraindicated in patients with known hypersensitivity to donepezil hydrochloride or to piperidine derivatives.
Adverse Reactions
The following serious adverse reactions are described below and elsewhere in the labeling: • Cardiovascular Conditions [see Warnings and Precautions ( 5.2 )] • Nausea and Vomiting [see Warnings and Precautions ( 5.3 )] • Peptic Ulcer Disease and GI Bleeding [see Warnings and Precautions ( 5.4 )] • Genitourinary Conditions [see Warnings and Precautions ( 5.6 )] • Neurological Conditions: Seizures [see Warnings and Precautions ( 5.7 )] • Pulmonary Conditions [see Warnings and Precautions ( 5.8 )]
Drug Interactions
• Cholinesterase inhibitors have the potential to interfere with the activity of anticholinergic medications ( 7.1 ). • A synergistic effect may be expected with concomitant administration of succinylcholine, similar neuromuscular blocking agents, or cholinergic agonists ( 7.2 ).
Storage and Handling
Donepezil Hydrochloride Tablets The 10 mg tablets are light yellow, round, film-coated tablets, debossed with ‘HH210' on one side. NDC: 71335-0900-1: 30 Tablets in a BOTTLE, PLASTIC NDC: 71335-0900-2: 60 Tablets in a BOTTLE, PLASTIC NDC: 71335-0900-3: 90 Tablets in a BOTTLE, PLASTIC Store at controlled room temperature, 15°C to 30°C (59°F to 86°F). Repackaged/Relabeled by: Bryant Ranch Prepack, Inc. Burbank, CA 9150
How Supplied
Donepezil Hydrochloride Tablets The 10 mg tablets are light yellow, round, film-coated tablets, debossed with ‘HH210' on one side. NDC: 71335-0900-1: 30 Tablets in a BOTTLE, PLASTIC NDC: 71335-0900-2: 60 Tablets in a BOTTLE, PLASTIC NDC: 71335-0900-3: 90 Tablets in a BOTTLE, PLASTIC Store at controlled room temperature, 15°C to 30°C (59°F to 86°F). Repackaged/Relabeled by: Bryant Ranch Prepack, Inc. Burbank, CA 9150
Medication Information
Warnings and Precautions
• Cholinesterase inhibitors are likely to exaggerate succinylcholine-type muscle relaxation during anesthesia ( 5.1 ). • Cholinesterase inhibitors may have vagotonic effects on the sinoatrial and atrioventricular nodes manifesting as bradycardia or heart block ( 5.2 ). • Donepezil hydrochloride can cause vomiting. Patients should be observed closely at initiation of treatment and after dose increases ( 5.3 ). • Patients should be monitored closely for symptoms of active or occult gastrointestinal (GI) bleeding, especially those at increased risk for developing ulcers ( 5.4 ). • Cholinomimetics may cause bladder outflow obstructions ( 5.6 ). • Cholinomimetics are believed to have some potential to cause generalized convulsions ( 5.7 ). • Cholinesterase inhibitors should be prescribed with care to patients with a history of asthma or obstructive pulmonary disease ( 5.8 ).
Indications and Usage
Donepezil hydrochloride is indicated for the treatment of dementia of the Alzheimer's type. Efficacy has been demonstrated in patients with mild, moderate, and severe Alzheimer's disease.
Dosage and Administration
• Mild to Moderate Alzheimer's disease : 5 mg to 10 mg administered once daily ( 2.1 ) • Moderate to Severe Alzheimer’s Disease : 10 mg administered once daily ( 2.2)
Contraindications
Donepezil hydrochloride is contraindicated in patients with known hypersensitivity to donepezil hydrochloride or to piperidine derivatives.
Adverse Reactions
The following serious adverse reactions are described below and elsewhere in the labeling: • Cardiovascular Conditions [see Warnings and Precautions ( 5.2 )] • Nausea and Vomiting [see Warnings and Precautions ( 5.3 )] • Peptic Ulcer Disease and GI Bleeding [see Warnings and Precautions ( 5.4 )] • Genitourinary Conditions [see Warnings and Precautions ( 5.6 )] • Neurological Conditions: Seizures [see Warnings and Precautions ( 5.7 )] • Pulmonary Conditions [see Warnings and Precautions ( 5.8 )]
Drug Interactions
• Cholinesterase inhibitors have the potential to interfere with the activity of anticholinergic medications ( 7.1 ). • A synergistic effect may be expected with concomitant administration of succinylcholine, similar neuromuscular blocking agents, or cholinergic agonists ( 7.2 ).
Storage and Handling
Donepezil Hydrochloride Tablets The 10 mg tablets are light yellow, round, film-coated tablets, debossed with ‘HH210' on one side. NDC: 71335-0900-1: 30 Tablets in a BOTTLE, PLASTIC NDC: 71335-0900-2: 60 Tablets in a BOTTLE, PLASTIC NDC: 71335-0900-3: 90 Tablets in a BOTTLE, PLASTIC Store at controlled room temperature, 15°C to 30°C (59°F to 86°F). Repackaged/Relabeled by: Bryant Ranch Prepack, Inc. Burbank, CA 9150
How Supplied
Donepezil Hydrochloride Tablets The 10 mg tablets are light yellow, round, film-coated tablets, debossed with ‘HH210' on one side. NDC: 71335-0900-1: 30 Tablets in a BOTTLE, PLASTIC NDC: 71335-0900-2: 60 Tablets in a BOTTLE, PLASTIC NDC: 71335-0900-3: 90 Tablets in a BOTTLE, PLASTIC Store at controlled room temperature, 15°C to 30°C (59°F to 86°F). Repackaged/Relabeled by: Bryant Ranch Prepack, Inc. Burbank, CA 9150
Description
Donepezil hydrochloride is indicated for the treatment of dementia of the Alzheimer's type. Efficacy has been demonstrated in patients with mild, moderate, and severe Alzheimer's disease.
Section 42229-5
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
Donepezil hydrochloride has been administered to over 1,700 individuals during clinical trials worldwide. Approximately 1200 of these patients have been treated for at least 3 months and more than 1,000 patients have been treated for at least 6 months. Controlled and uncontrolled trials in the United States included approximately 900 patients. In regards to the highest dose of 10 mg/day, this population includes 650 patients treated for 3 months, 475 patients treated for 6 months, and 116 patients treated for over 1 year. The range of patient exposure is from 1 to 1,214 days.
Section 51945-4
Donepezil Hcl 10mg Tablet
10 Overdosage
Because strategies for the management of overdose are continually evolving, it is advisable to contact a Poison Control Center to determine the latest recommendations for the management of an overdose of any drug.
As in any case of overdose, general supportive measures should be utilized. Overdosage with cholinesterase inhibitors can result in cholinergic crisis characterized by severe nausea, vomiting, salivation, sweating, bradycardia, hypotension, respiratory depression, collapse and convulsions. Increasing muscle weakness is a possibility and may result in death if respiratory muscles are involved. Tertiary anticholinergics such as atropine may be used as an antidote for donepezil hydrochloride overdosage. Intravenous atropine sulfate titrated to effect is recommended: an initial dose of 1.0 to 2.0 mg IV with subsequent doses based upon clinical response. Atypical responses in blood pressure and heart rate have been reported with other cholinomimetics when co-administered with quaternary anticholinergics such as glycopyrrolate. It is not known whether donepezil hydrochloride and/or its metabolites can be removed by dialysis (hemodialysis, peritoneal dialysis, or hemofiltration).
Dose-related signs of toxicity in animals included reduced spontaneous movement, prone position, staggering gait, lacrimation, clonic convulsions, depressed respiration, salivation, miosis, tremors, fasciculation and lower body surface temperature.
8.2 Lactation
Risk Summary
There are no data on the presence of donepezil or its metabolites in human milk, the effects on the breastfed infant, or on milk production.
The developmental and health benefits of breastfeeding should be considered along with the mother’s clinical need for donepezil hydrochloride and any potential adverse effects on the breastfed infant from donepezil hydrochloride or from the underlying maternal condition.
11 Description
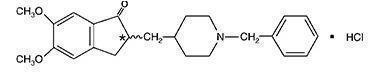
Donepezil hydrochloride is a reversible inhibitor of the enzyme acetylcholinesterase, known chemically as (±)-2, 3-dihydro-5, 6-dimethoxy-2-[[1-(phenylmethyl)-4-piperidinyl]methyl]-1H-inden-1-one hydrochloride. Donepezil hydrochloride is commonly referred to in the pharmacological literature as E2020. It has an empirical formula of C24H29NO3HCl and a molecular weight of 415.96. Donepezil hydrochloride is a white crystalline powder and is freely soluble in chloroform, soluble in water and in glacial acetic acid, slightly soluble in ethanol and in acetonitrile and practically insoluble in ethyl acetate and in n-hexane.
Donepezil Hydrochloride Tablets, USP, are available for oral administration in film-coated tablets containing 5 or 10 mg of donepezil hydrochloride.
Inactive ingredients in 5 mg and 10 mg tablets are lactose monohydrate, hydroxypropyl cellulose, magnesium Stearate, microcrystalline cellulose, corn starch, Croscarmellose sodium. Additionally, the 5 mg tablets coating film contains hydroxypropyl methyl cellulose, titanium dioxide, polyethylene glycol, and FD&C blue. The 10 mg tablets coating film contains hydroxypropyl methyl cellulose, titanium dioxide, polyethylene glycol, iron oxide yellow and iron oxide red.
5.1 Anesthesia
Donepezil hydrochloride, as a cholinesterase inhibitor, is likely to exaggerate succinylcholine-type muscle relaxation during anesthesia.
8.4 Pediatric Use
The safety and effectiveness in pediatric patients have not been established.
8.5 Geriatric Use
Alzheimer's disease is a disorder occurring primarily in individuals over 55 years of age. The mean age of patients enrolled in the clinical studies with donepezil hydrochloride was 73 years; 80% of these patients were between 65 and 84 years old, and 49% of patients were at or above the age of 75. The efficacy and safety data presented in the clinical trials section were obtained from these patients. There were no clinically significant differences in most adverse reactions reported by patient groups ≥ 65 years old and < 65 years old.
4 Contraindications
Donepezil hydrochloride is contraindicated in patients with known hypersensitivity to donepezil hydrochloride or to piperidine derivatives.
6 Adverse Reactions
The following serious adverse reactions are described below and elsewhere in the labeling:
-
•Cardiovascular Conditions [see Warnings and Precautions (5.2)]
-
•Nausea and Vomiting [see Warnings and Precautions (5.3)]
-
•Peptic Ulcer Disease and GI Bleeding [see Warnings and Precautions (5.4)]
-
•Genitourinary Conditions [see Warnings and Precautions (5.6)]
-
•Neurological Conditions: Seizures [see Warnings and Precautions (5.7)]
-
•Pulmonary Conditions [see Warnings and Precautions (5.8)]
7 Drug Interactions
12.3 Pharmacokinetics
Pharmacokinetics of donepezil are linear over a dose range of 1-10 mg given once daily. The rate and extent of absorption of donepezil hydrochloride tablets are not influenced by food.
The elimination half life of donepezil is about 70 hours, and the mean apparent plasma clearance (Cl/F) is 0.13 – 0.19 L/hr/kg. Following multiple dose administration, donepezil accumulates in plasma by 4-7 fold, and steady state is reached within 15 days. The steady state volume of distribution is 12 - 16 L/kg. Donepezil is approximately 96% bound to human plasma proteins, mainly to albumins (about 75%) and alpha1 - acid glycoprotein (about 21%) over the concentration range of 2-1000 ng/mL.
Donepezil is both excreted in the urine intact and extensively metabolized to four major metabolites, two of which are known to be active, and a number of minor metabolites, not all of which have been identified. Donepezil is metabolized by CYP 450 isoenzymes 2D6 and 3A4 and undergoes glucuronidation. Following administration of 14C-labeled donepezil, plasma radioactivity, expressed as a percent of the administered dose, was present primarily as intact donepezil (53%) and as 6-O-desmethyl donepezil (11%), which has been reported to inhibit AChE to the same extent as donepezil in vitro and was found in plasma at concentrations equal to about 20% of donepezil. Approximately 57% and 15% of the total radioactivity was recovered in urine and feces, respectively, over a period of 10 days, while 28% remained unrecovered, with about 17% of the donepezil dose recovered in the urine as unchanged drug. Examination of the effect of CYP2D6 genotype in Alzheimer's patients showed differences in clearance values among CYP2D6 genotype subgroups. When compared to the extensive metabolizers, poor metabolizers had a 31.5% slower clearance and ultra-rapid metabolizers had a 24% faster clearance.
1 Indications and Usage
Donepezil hydrochloride is indicated for the treatment of dementia of the Alzheimer's type. Efficacy has been demonstrated in patients with mild, moderate, and severe Alzheimer's disease.
5.3 Nausea and Vomiting
Donepezil hydrochloride, as a predictable consequence of its pharmacological properties, has been shown to produce diarrhea, nausea, and vomiting. These effects, when they occur, appear more frequently with the 10 mg/day dose than with the 5 mg/day dose.
Although in most cases, these effects have been transient, sometimes lasting one to three weeks, and have resolved during continued use of donepezil hydrochloride, patients should be observed closely at the initiation of treatment and after dose increases.
12.1 Mechanism of Action
Current theories on the pathogenesis of the cognitive signs and symptoms of Alzheimer's disease attribute some of them to a deficiency of cholinergic neurotransmission.
Donepezil hydrochloride is postulated to exert its therapeutic effect by enhancing cholinergic function. This is accomplished by increasing the concentration of acetylcholine through reversible inhibition of its hydrolysis by acetylcholinesterase. There is no evidence that donepezil alters the course of the underlying dementing process.
5.8 Pulmonary Conditions
Because of their cholinomimetic actions, cholinesterase inhibitors should be prescribed with care to patients with a history of asthma or obstructive pulmonary disease.
5 Warnings and Precautions
-
•Cholinesterase inhibitors are likely to exaggerate succinylcholine-type muscle relaxation during anesthesia (5.1).
-
•Cholinesterase inhibitors may have vagotonic effects on the sinoatrial and atrioventricular nodes manifesting as bradycardia or heart block (5.2).
-
•Donepezil hydrochloride can cause vomiting. Patients should be observed closely at initiation of treatment and after dose increases (5.3).
-
•Patients should be monitored closely for symptoms of active or occult gastrointestinal (GI) bleeding, especially those at increased risk for developing ulcers (5.4).
-
•Cholinomimetics may cause bladder outflow obstructions (5.6).
-
•Cholinomimetics are believed to have some potential to cause generalized convulsions (5.7).
-
•Cholinesterase inhibitors should be prescribed with care to patients with a history of asthma or obstructive pulmonary disease (5.8).
2 Dosage and Administration
3 Dosage Forms and Strengths
Donepezil hydrochloride is supplied as film-coated, round tablets containing 5 mg, 10 mg of donepezil hydrochloride, USP.
The 5 mg tablets are blue, round, film-coated tablets, debossed with ‘HH205' on one side.
The 10 mg tablets are light yellow, round, film-coated tablets, debossed with ‘HH210’ on one side.
5.6 Genitourinary Conditions
Although not observed in clinical trials of donepezil hydrochloride, cholinomimetics may cause bladder outflow obstruction.
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of donepezil hydrochloride. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Abdominal pain, agitation, aggression, cholecystitis, confusion, convulsions, hallucinations, heart block (all types), hemolytic anemia, hepatitis, hyponatremia, neuroleptic malignant syndrome, pancreatitis, rash, rhabdomyolysis, QTc prolongation, and torsade de pointes.
5.2 Cardiovascular Conditions
Because of their pharmacological action, cholinesterase inhibitors may have vagotonic effects on the sinoatrial and atrioventricular nodes. This effect may manifest as bradycardia or heart block in patients both with and without known underlying cardiac conduction abnormalities. Syncopal episodes have been reported in association with the use of donepezil hydrochloride.
7.1 Use With Anticholinergics
- Because of their mechanism of action, cholinesterase inhibitors have the potential to interfere with the activity of anticholinergic medications.
2.3 Administration Information
Donepezil hydrochloride should be taken in the evening, just prior to retiring. Donepezil hydrochloride can be taken with or without food.
8. Use in Specific Populations
Pregnancy: Based on animal data, may cause fetal harm (8.1).
17 Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Patient Information).
Instruct patients and caregivers to take donepezil hydrochloride only once per day, as prescribed.
Instruct patients and caregivers that donepezil hydrochloride can be taken with or without food.
Advise patients and caregivers that donepezil hydrochloride may cause nausea, diarrhea, insomnia, vomiting, muscle cramps, fatigue, and decreased appetite.
Advise patients to notify their healthcare provider if they are pregnant or plan to become pregnant.
Manufactured by:
Zhejiang Huahai Pharmaceutical Co., Ltd.
Xunqiao, Linhai, Zhejiang, 317024, China
Distributed by:
Solco Healthcare US, LLC
Somerset, NJ 08873, USA
Revised: 08/2020
200145-01
DONEPEZIL HYDROCHLORIDE PATIENT PACKAGE INSERT
Donepezil (doe NEP e zil) Hydrochloride Tablets, USP
-
•Tablets: 5 mg and 10 mg
Read this Patient Information that comes with donepezil hydrochloride before you start taking it and each time you get a refill. There may be new information. This leaflet does not take the place of talking with your doctor about Alzheimer’s disease or treatment for it. If you have questions, ask the doctor or pharmacist.
What is donepezil hydrochloride?
Donepezil hydrochloride comes as donepezil hydrochloride film-coated tablets in dosage strengths of 5 mg and 10 mg.
Donepezil hydrochloride is a prescription medicine to treat mild, moderate and severe Alzheimer’s disease. Donepezil hydrochloride can help with mental function and with doing daily tasks. Donepezil hydrochloride does not work the same in all people. Some people may:
-
•Seem much better
-
•Get better in small ways or stay the same
-
•Get worse over time but slower than expected
-
•Not change and then get worse as expected
Donepezil hydrochloride does not cure Alzheimer's disease. All patients with Alzheimer's disease get worse over time, even if they take donepezil hydrochloride.
Donepezil hydrochloride has not been approved as a treatment for any medical condition in children.
Who should not take donepezil hydrochloride?
Do not take donepezil hydrochloride if you are allergic to any of the ingredients in donepezil hydrochloride tablets or to medicines that contain piperidines. Ask your doctor if you are not sure. See the end of this leaflet for a list of ingredients in donepezil hydrochloride tablets.
What should I tell my doctor before taking donepezil hydrochloride?
Tell the doctor about all of your present or past health problems and conditions. Include:
-
•Any heart problems including problems with irregular, slow, or fast heartbeats
-
•Asthma or lung problems
-
•A seizure
-
•Stomach ulcers
-
•Difficulty passing urine
-
•Liver or kidney problems
-
•Trouble swallowing tablets
-
•Present pregnancy or plans to become pregnant. It is not known if donepezil hydrochloride can harm an unborn baby.
-
•Present breast-feeding. It is not known if donepezil hydrochloride passes into breast milk. Talk to your doctor about the best way to feed your baby if you take donepezil hydrochloride.
Tell the doctor about all the medicines you take, including prescription and non-prescription medicines, vitamins, and herbal products. Donepezil hydrochloride and other medicines may affect each other.
Be particularly sure to tell the doctor if you take aspirin or medicines called nonsteroidal anti-inflammatory drugs (NSAIDs). There are many NSAID medicines, both prescription and non-prescription. Ask the doctor or pharmacist if you are not sure if any of your medicines are NSAIDs. Taking NSAIDs and donepezil hydrochloride together may make you more likely to get stomach ulcers.
Donepezil hydrochloride taken with certain medicines used for anesthesia may cause side effects. Tell the responsible doctor or dentist that you take donepezil hydrochloride before you have:
-
•surgery
-
•medical procedures
-
•dental surgery or procedures
Know the medicines that you take. Keep a list of all your medicines. Show it to your doctor or pharmacist before you start a new medicine..
How should you take donepezil hydrochloride?
-
•Take donepezil hydrochloride exactly as prescribed by the doctor. Do not stop donepezil hydrochloride or change the dose yourself. Talk with your doctor first.
-
•Take donepezil hydrochloride one time each day. Donepezil hydrochloride can be taken with or without food.
-
•If you miss giving the patient a dose of donepezil hydrochloride, just wait. Take only the next dose at the usual time. Do not take 2 doses at the same time.
-
•If donepezil hydrochloride is missed for 7 days or more, talk with your doctor before starting again.
-
•If you take too much donepezil hydrochloride at one time, call your doctor or poison control center, or go to the emergency room right away.
What are the possible side effects of donepezil hydrochloride?
Donepezil hydrochloride may cause the following serious side effects:
-
•slow heartbeat and fainting. This happens more often in people with heart problems. Call your doctor right away if you feel faint or lightheaded while taking donepezil hydrochloride.
-
•more stomach acid. This raises the chance of ulcers and bleeding. The risk is higher for people who have ulcers, or take aspirin or other NSAIDs.
-
•worsening of lung problems in people with asthma or other lung disease.
-
•seizures.
-
•difficulty passing urine.
Call your doctor right away if you have:
-
•fainting.
-
•heartburn or stomach pain that is new or won't go away.
-
•nausea or vomiting, blood in the vomit, dark vomit that looks like coffee grounds.
-
•bowel movements or stools that look like black tar.
-
•new or worse asthma or breathing problems.
-
•seizures.
-
•difficulty passing urine.
The most common side effects of donepezil hydrochloride are:
-
•nausea
-
•diarrhea
-
•not sleeping well
-
•vomiting
-
•muscle cramps
-
•feeling tired
-
•not wanting to eat
These side effects may get better after you take donepezil hydrochloride for a while. This is not a complete list of side effects with donepezil hydrochloride. For more information, ask your doctor or pharmacist.
Call your doctor for medical advice about side effects. You may report side effects to Solco Healthcare U. S., LLC at 1-866-257-2597 or FDA at 1-800-FDA-1088.
How should donepezil hydrochloride be stored?
Store donepezil hydrochloride at room temperature between 59° to 86°F (15° to 30°C).
Keep donepezil hydrochloride and all medicines out of the reach of children.
General information about donepezil hydrochloride
Medicines are sometimes prescribed for conditions that are not mentioned in this Patient Information Leaflet. Do not use donepezil hydrochloride for a condition for which it was not prescribed. Do not give donepezil hydrochloride to other people, even if they have the same symptoms or condition. It may harm them.
This leaflet summarizes the most important information about donepezil hydrochloride. If you would like more information, talk with your doctor. You can ask your pharmacist or doctor for information about donepezil hydrochloride that is written for health professionals.
What are the ingredients in Donepezil Hydrochloride Tablets, USP?
Active ingredient: donepezil hydrochloride
Inactive ingredients:
Donepezil Hydrochloride 5 mg and 10 mg film-coated tablets: lactose monohydrate, hydroxypropyl cellulose, magnesium stearate, microcrystalline cellulose, corn starch, croscarmellose sodium. The film of 5 mg tablets coating contains hydroxypropyl methyl cellulose, titanium dioxide, polyethylene glycol, and FD&C blue. The film of 10 mg tablets coating contains hydroxypropyl methyl cellulose, titanium dioxide, polyethylene glycol, iron oxide yellow and iron oxide red.
Rx Only
Manufactured by:
Zhejiang Huahai Pharmaceutical Co., Ltd.
Xunqiao, Linhai, Zhejiang, 317024, China
Distributed by:
Solco Healthcare US, LLC
Somerset, NJ 08873, USA
Revised: 08/2020
Rx Only
16 How Supplied/storage and Handling
Donepezil Hydrochloride Tablets
The 10 mg tablets are light yellow, round, film-coated tablets, debossed with ‘HH210' on one side.
NDC: 71335-0900-1: 30 Tablets in a BOTTLE, PLASTIC
NDC: 71335-0900-2: 60 Tablets in a BOTTLE, PLASTIC
NDC: 71335-0900-3: 90 Tablets in a BOTTLE, PLASTIC
Store at controlled room temperature, 15°C to 30°C (59°F to 86°F).
Repackaged/Relabeled by:
Bryant Ranch Prepack, Inc.
Burbank, CA 9150
5.7 Neurological Conditions: Seizures
Cholinomimetics are believed to have some potential to cause generalized convulsions. However, seizure activity also may be a manifestation of Alzheimer's disease.
5.4 Peptic Ulcer Disease and Gi Bleeding
Through their primary action, cholinesterase inhibitors may be expected to increase gastric acid secretion due to increased cholinergic activity. Therefore, patients should be monitored closely for symptoms of active or occult gastrointestinal bleeding, especially those at increased risk for developing ulcers, e.g., those with a history of ulcer disease or those receiving concurrent nonsteroidal anti-inflammatory drugs (NSAIDs). Clinical studies of donepezil hydrochloride in a dose of 5 mg/day to 10 mg/day have shown no increase, relative to placebo, in the incidence of either peptic ulcer disease or gastrointestinal bleeding.
14.1 Mild to Moderate Alzheimer's Disease
The effectiveness of donepezil hydrochloride as a treatment for mild to moderate Alzheimer's disease is demonstrated by the results of two randomized, double-blind, placebo-controlled clinical investigations in patients with Alzheimer's disease (diagnosed by NINCDS and DSM III-R criteria, Mini-Mental State Examination ≥ 10 and ≤ 26 and Clinical Dementia Rating of 1 or 2). The mean age of patients participating in donepezil hydrochloride trials was 73 years with a range of 50 to 94. Approximately 62% of patients were women and 38% were men. The racial distribution was white 95%, black 3% and other races 2%.
The higher dose of 10 mg did not provide a statistically significantly greater clinical benefit than 5 mg. There is a suggestion, however, based upon order of group mean scores and dose trend analyses of data from these clinical trials, that a daily dose of 10 mg of donepezil hydrochloride might provide additional benefit for some patients. Accordingly, whether or not to employ a dose of 10 mg is a matter of prescriber and patient preference.
13.2 Animal Toxicology And/or Pharmacology
In an acute dose neurotoxicity study in female rats, oral administration of donepezil and memantine in combination resulted in increased incidence, severity, and distribution of neurodegeneration compared with memantine alone. The no-effect levels of the combination were associated with clinically relevant plasma donepezil and memantine levels.
The relevance of this finding to humans is unknown.
2.1 Dosing in Mild to Moderate Alzheimer's Disease
The recommended starting dosage of donepezil hydrochloride is 5 mg administered once per day in the evening, just prior to retiring. The maximum recommended dosage of donepezil hydrochloride in patients with mild to moderate Alzheimer’s disease is 10 mg per day. A dose of 10 mg should not be administered until patients have been on a daily dose of 5 mg for 4 to 6 weeks.
2.2 Dosing in Moderate to Severe Alzheimer’s Disease
The recommended starting dosage of donepezil hydrochloride is 5 mg administered once per day in the evening, just prior to retiring. A dose of 10 mg should not be administered until patients have been on a daily dose of 5 mg for 4 to 6 weeks.
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
No evidence of carcinogenic potential was obtained in an 88-week carcinogenicity study of donepezil conducted in mice at oral doses up to 180 mg/kg/day (approximately 86 times the maximum recommended human dose [MRHD] of 10 mg/day on a mg/m2 basis), or in a 104-week carcinogenicity study in rats at oral doses up to 30 mg/kg/day (approximately 29 times the MRHD on a mg/m2 basis).
Donepezil was negative in a battery of genotoxicity assays (in vitro bacterial reverse mutation, in vitro mouse lymphoma tk, in vitro chromosomal aberration, and in vivo mouse micronucleus).
Donepezil had no effect on fertility in rats at oral doses up to 10 mg/kg/day (approximately 10 times the MRHD on a mg/m2 basis) when administered to males and females prior to and during mating and continuing in females through implantation.
7.2 Use With Cholinomimetics and Other Cholinesterase Inhibitors
A synergistic effect may be expected when cholinesterase inhibitors are given concurrently with succinylcholine, similar neuromuscular blocking agents or cholinergic agonists such as bethanechol.
Structured Label Content
Section 42229-5 (42229-5)
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
Donepezil hydrochloride has been administered to over 1,700 individuals during clinical trials worldwide. Approximately 1200 of these patients have been treated for at least 3 months and more than 1,000 patients have been treated for at least 6 months. Controlled and uncontrolled trials in the United States included approximately 900 patients. In regards to the highest dose of 10 mg/day, this population includes 650 patients treated for 3 months, 475 patients treated for 6 months, and 116 patients treated for over 1 year. The range of patient exposure is from 1 to 1,214 days.
Section 51945-4 (51945-4)
Donepezil Hcl 10mg Tablet
10 Overdosage (10 OVERDOSAGE)
Because strategies for the management of overdose are continually evolving, it is advisable to contact a Poison Control Center to determine the latest recommendations for the management of an overdose of any drug.
As in any case of overdose, general supportive measures should be utilized. Overdosage with cholinesterase inhibitors can result in cholinergic crisis characterized by severe nausea, vomiting, salivation, sweating, bradycardia, hypotension, respiratory depression, collapse and convulsions. Increasing muscle weakness is a possibility and may result in death if respiratory muscles are involved. Tertiary anticholinergics such as atropine may be used as an antidote for donepezil hydrochloride overdosage. Intravenous atropine sulfate titrated to effect is recommended: an initial dose of 1.0 to 2.0 mg IV with subsequent doses based upon clinical response. Atypical responses in blood pressure and heart rate have been reported with other cholinomimetics when co-administered with quaternary anticholinergics such as glycopyrrolate. It is not known whether donepezil hydrochloride and/or its metabolites can be removed by dialysis (hemodialysis, peritoneal dialysis, or hemofiltration).
Dose-related signs of toxicity in animals included reduced spontaneous movement, prone position, staggering gait, lacrimation, clonic convulsions, depressed respiration, salivation, miosis, tremors, fasciculation and lower body surface temperature.
8.2 Lactation
Risk Summary
There are no data on the presence of donepezil or its metabolites in human milk, the effects on the breastfed infant, or on milk production.
The developmental and health benefits of breastfeeding should be considered along with the mother’s clinical need for donepezil hydrochloride and any potential adverse effects on the breastfed infant from donepezil hydrochloride or from the underlying maternal condition.
11 Description (11 DESCRIPTION)
Donepezil hydrochloride is a reversible inhibitor of the enzyme acetylcholinesterase, known chemically as (±)-2, 3-dihydro-5, 6-dimethoxy-2-[[1-(phenylmethyl)-4-piperidinyl]methyl]-1H-inden-1-one hydrochloride. Donepezil hydrochloride is commonly referred to in the pharmacological literature as E2020. It has an empirical formula of C24H29NO3HCl and a molecular weight of 415.96. Donepezil hydrochloride is a white crystalline powder and is freely soluble in chloroform, soluble in water and in glacial acetic acid, slightly soluble in ethanol and in acetonitrile and practically insoluble in ethyl acetate and in n-hexane.
Donepezil Hydrochloride Tablets, USP, are available for oral administration in film-coated tablets containing 5 or 10 mg of donepezil hydrochloride.
Inactive ingredients in 5 mg and 10 mg tablets are lactose monohydrate, hydroxypropyl cellulose, magnesium Stearate, microcrystalline cellulose, corn starch, Croscarmellose sodium. Additionally, the 5 mg tablets coating film contains hydroxypropyl methyl cellulose, titanium dioxide, polyethylene glycol, and FD&C blue. The 10 mg tablets coating film contains hydroxypropyl methyl cellulose, titanium dioxide, polyethylene glycol, iron oxide yellow and iron oxide red.
5.1 Anesthesia
Donepezil hydrochloride, as a cholinesterase inhibitor, is likely to exaggerate succinylcholine-type muscle relaxation during anesthesia.
8.4 Pediatric Use
The safety and effectiveness in pediatric patients have not been established.
8.5 Geriatric Use
Alzheimer's disease is a disorder occurring primarily in individuals over 55 years of age. The mean age of patients enrolled in the clinical studies with donepezil hydrochloride was 73 years; 80% of these patients were between 65 and 84 years old, and 49% of patients were at or above the age of 75. The efficacy and safety data presented in the clinical trials section were obtained from these patients. There were no clinically significant differences in most adverse reactions reported by patient groups ≥ 65 years old and < 65 years old.
4 Contraindications (4 CONTRAINDICATIONS)
Donepezil hydrochloride is contraindicated in patients with known hypersensitivity to donepezil hydrochloride or to piperidine derivatives.
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following serious adverse reactions are described below and elsewhere in the labeling:
-
•Cardiovascular Conditions [see Warnings and Precautions (5.2)]
-
•Nausea and Vomiting [see Warnings and Precautions (5.3)]
-
•Peptic Ulcer Disease and GI Bleeding [see Warnings and Precautions (5.4)]
-
•Genitourinary Conditions [see Warnings and Precautions (5.6)]
-
•Neurological Conditions: Seizures [see Warnings and Precautions (5.7)]
-
•Pulmonary Conditions [see Warnings and Precautions (5.8)]
7 Drug Interactions (7 DRUG INTERACTIONS)
12.3 Pharmacokinetics
Pharmacokinetics of donepezil are linear over a dose range of 1-10 mg given once daily. The rate and extent of absorption of donepezil hydrochloride tablets are not influenced by food.
The elimination half life of donepezil is about 70 hours, and the mean apparent plasma clearance (Cl/F) is 0.13 – 0.19 L/hr/kg. Following multiple dose administration, donepezil accumulates in plasma by 4-7 fold, and steady state is reached within 15 days. The steady state volume of distribution is 12 - 16 L/kg. Donepezil is approximately 96% bound to human plasma proteins, mainly to albumins (about 75%) and alpha1 - acid glycoprotein (about 21%) over the concentration range of 2-1000 ng/mL.
Donepezil is both excreted in the urine intact and extensively metabolized to four major metabolites, two of which are known to be active, and a number of minor metabolites, not all of which have been identified. Donepezil is metabolized by CYP 450 isoenzymes 2D6 and 3A4 and undergoes glucuronidation. Following administration of 14C-labeled donepezil, plasma radioactivity, expressed as a percent of the administered dose, was present primarily as intact donepezil (53%) and as 6-O-desmethyl donepezil (11%), which has been reported to inhibit AChE to the same extent as donepezil in vitro and was found in plasma at concentrations equal to about 20% of donepezil. Approximately 57% and 15% of the total radioactivity was recovered in urine and feces, respectively, over a period of 10 days, while 28% remained unrecovered, with about 17% of the donepezil dose recovered in the urine as unchanged drug. Examination of the effect of CYP2D6 genotype in Alzheimer's patients showed differences in clearance values among CYP2D6 genotype subgroups. When compared to the extensive metabolizers, poor metabolizers had a 31.5% slower clearance and ultra-rapid metabolizers had a 24% faster clearance.
1 Indications and Usage (1 INDICATIONS AND USAGE)
Donepezil hydrochloride is indicated for the treatment of dementia of the Alzheimer's type. Efficacy has been demonstrated in patients with mild, moderate, and severe Alzheimer's disease.
5.3 Nausea and Vomiting
Donepezil hydrochloride, as a predictable consequence of its pharmacological properties, has been shown to produce diarrhea, nausea, and vomiting. These effects, when they occur, appear more frequently with the 10 mg/day dose than with the 5 mg/day dose.
Although in most cases, these effects have been transient, sometimes lasting one to three weeks, and have resolved during continued use of donepezil hydrochloride, patients should be observed closely at the initiation of treatment and after dose increases.
12.1 Mechanism of Action
Current theories on the pathogenesis of the cognitive signs and symptoms of Alzheimer's disease attribute some of them to a deficiency of cholinergic neurotransmission.
Donepezil hydrochloride is postulated to exert its therapeutic effect by enhancing cholinergic function. This is accomplished by increasing the concentration of acetylcholine through reversible inhibition of its hydrolysis by acetylcholinesterase. There is no evidence that donepezil alters the course of the underlying dementing process.
5.8 Pulmonary Conditions
Because of their cholinomimetic actions, cholinesterase inhibitors should be prescribed with care to patients with a history of asthma or obstructive pulmonary disease.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
-
•Cholinesterase inhibitors are likely to exaggerate succinylcholine-type muscle relaxation during anesthesia (5.1).
-
•Cholinesterase inhibitors may have vagotonic effects on the sinoatrial and atrioventricular nodes manifesting as bradycardia or heart block (5.2).
-
•Donepezil hydrochloride can cause vomiting. Patients should be observed closely at initiation of treatment and after dose increases (5.3).
-
•Patients should be monitored closely for symptoms of active or occult gastrointestinal (GI) bleeding, especially those at increased risk for developing ulcers (5.4).
-
•Cholinomimetics may cause bladder outflow obstructions (5.6).
-
•Cholinomimetics are believed to have some potential to cause generalized convulsions (5.7).
-
•Cholinesterase inhibitors should be prescribed with care to patients with a history of asthma or obstructive pulmonary disease (5.8).
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Donepezil hydrochloride is supplied as film-coated, round tablets containing 5 mg, 10 mg of donepezil hydrochloride, USP.
The 5 mg tablets are blue, round, film-coated tablets, debossed with ‘HH205' on one side.
The 10 mg tablets are light yellow, round, film-coated tablets, debossed with ‘HH210’ on one side.
5.6 Genitourinary Conditions
Although not observed in clinical trials of donepezil hydrochloride, cholinomimetics may cause bladder outflow obstruction.
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of donepezil hydrochloride. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Abdominal pain, agitation, aggression, cholecystitis, confusion, convulsions, hallucinations, heart block (all types), hemolytic anemia, hepatitis, hyponatremia, neuroleptic malignant syndrome, pancreatitis, rash, rhabdomyolysis, QTc prolongation, and torsade de pointes.
5.2 Cardiovascular Conditions
Because of their pharmacological action, cholinesterase inhibitors may have vagotonic effects on the sinoatrial and atrioventricular nodes. This effect may manifest as bradycardia or heart block in patients both with and without known underlying cardiac conduction abnormalities. Syncopal episodes have been reported in association with the use of donepezil hydrochloride.
7.1 Use With Anticholinergics (7.1 Use with Anticholinergics)
- Because of their mechanism of action, cholinesterase inhibitors have the potential to interfere with the activity of anticholinergic medications.
2.3 Administration Information
Donepezil hydrochloride should be taken in the evening, just prior to retiring. Donepezil hydrochloride can be taken with or without food.
8. Use in Specific Populations (8. USE IN SPECIFIC POPULATIONS)
Pregnancy: Based on animal data, may cause fetal harm (8.1).
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient to read the FDA-approved patient labeling (Patient Information).
Instruct patients and caregivers to take donepezil hydrochloride only once per day, as prescribed.
Instruct patients and caregivers that donepezil hydrochloride can be taken with or without food.
Advise patients and caregivers that donepezil hydrochloride may cause nausea, diarrhea, insomnia, vomiting, muscle cramps, fatigue, and decreased appetite.
Advise patients to notify their healthcare provider if they are pregnant or plan to become pregnant.
Manufactured by:
Zhejiang Huahai Pharmaceutical Co., Ltd.
Xunqiao, Linhai, Zhejiang, 317024, China
Distributed by:
Solco Healthcare US, LLC
Somerset, NJ 08873, USA
Revised: 08/2020
200145-01
DONEPEZIL HYDROCHLORIDE PATIENT PACKAGE INSERT
Donepezil (doe NEP e zil) Hydrochloride Tablets, USP
-
•Tablets: 5 mg and 10 mg
Read this Patient Information that comes with donepezil hydrochloride before you start taking it and each time you get a refill. There may be new information. This leaflet does not take the place of talking with your doctor about Alzheimer’s disease or treatment for it. If you have questions, ask the doctor or pharmacist.
What is donepezil hydrochloride?
Donepezil hydrochloride comes as donepezil hydrochloride film-coated tablets in dosage strengths of 5 mg and 10 mg.
Donepezil hydrochloride is a prescription medicine to treat mild, moderate and severe Alzheimer’s disease. Donepezil hydrochloride can help with mental function and with doing daily tasks. Donepezil hydrochloride does not work the same in all people. Some people may:
-
•Seem much better
-
•Get better in small ways or stay the same
-
•Get worse over time but slower than expected
-
•Not change and then get worse as expected
Donepezil hydrochloride does not cure Alzheimer's disease. All patients with Alzheimer's disease get worse over time, even if they take donepezil hydrochloride.
Donepezil hydrochloride has not been approved as a treatment for any medical condition in children.
Who should not take donepezil hydrochloride?
Do not take donepezil hydrochloride if you are allergic to any of the ingredients in donepezil hydrochloride tablets or to medicines that contain piperidines. Ask your doctor if you are not sure. See the end of this leaflet for a list of ingredients in donepezil hydrochloride tablets.
What should I tell my doctor before taking donepezil hydrochloride?
Tell the doctor about all of your present or past health problems and conditions. Include:
-
•Any heart problems including problems with irregular, slow, or fast heartbeats
-
•Asthma or lung problems
-
•A seizure
-
•Stomach ulcers
-
•Difficulty passing urine
-
•Liver or kidney problems
-
•Trouble swallowing tablets
-
•Present pregnancy or plans to become pregnant. It is not known if donepezil hydrochloride can harm an unborn baby.
-
•Present breast-feeding. It is not known if donepezil hydrochloride passes into breast milk. Talk to your doctor about the best way to feed your baby if you take donepezil hydrochloride.
Tell the doctor about all the medicines you take, including prescription and non-prescription medicines, vitamins, and herbal products. Donepezil hydrochloride and other medicines may affect each other.
Be particularly sure to tell the doctor if you take aspirin or medicines called nonsteroidal anti-inflammatory drugs (NSAIDs). There are many NSAID medicines, both prescription and non-prescription. Ask the doctor or pharmacist if you are not sure if any of your medicines are NSAIDs. Taking NSAIDs and donepezil hydrochloride together may make you more likely to get stomach ulcers.
Donepezil hydrochloride taken with certain medicines used for anesthesia may cause side effects. Tell the responsible doctor or dentist that you take donepezil hydrochloride before you have:
-
•surgery
-
•medical procedures
-
•dental surgery or procedures
Know the medicines that you take. Keep a list of all your medicines. Show it to your doctor or pharmacist before you start a new medicine..
How should you take donepezil hydrochloride?
-
•Take donepezil hydrochloride exactly as prescribed by the doctor. Do not stop donepezil hydrochloride or change the dose yourself. Talk with your doctor first.
-
•Take donepezil hydrochloride one time each day. Donepezil hydrochloride can be taken with or without food.
-
•If you miss giving the patient a dose of donepezil hydrochloride, just wait. Take only the next dose at the usual time. Do not take 2 doses at the same time.
-
•If donepezil hydrochloride is missed for 7 days or more, talk with your doctor before starting again.
-
•If you take too much donepezil hydrochloride at one time, call your doctor or poison control center, or go to the emergency room right away.
What are the possible side effects of donepezil hydrochloride?
Donepezil hydrochloride may cause the following serious side effects:
-
•slow heartbeat and fainting. This happens more often in people with heart problems. Call your doctor right away if you feel faint or lightheaded while taking donepezil hydrochloride.
-
•more stomach acid. This raises the chance of ulcers and bleeding. The risk is higher for people who have ulcers, or take aspirin or other NSAIDs.
-
•worsening of lung problems in people with asthma or other lung disease.
-
•seizures.
-
•difficulty passing urine.
Call your doctor right away if you have:
-
•fainting.
-
•heartburn or stomach pain that is new or won't go away.
-
•nausea or vomiting, blood in the vomit, dark vomit that looks like coffee grounds.
-
•bowel movements or stools that look like black tar.
-
•new or worse asthma or breathing problems.
-
•seizures.
-
•difficulty passing urine.
The most common side effects of donepezil hydrochloride are:
-
•nausea
-
•diarrhea
-
•not sleeping well
-
•vomiting
-
•muscle cramps
-
•feeling tired
-
•not wanting to eat
These side effects may get better after you take donepezil hydrochloride for a while. This is not a complete list of side effects with donepezil hydrochloride. For more information, ask your doctor or pharmacist.
Call your doctor for medical advice about side effects. You may report side effects to Solco Healthcare U. S., LLC at 1-866-257-2597 or FDA at 1-800-FDA-1088.
How should donepezil hydrochloride be stored?
Store donepezil hydrochloride at room temperature between 59° to 86°F (15° to 30°C).
Keep donepezil hydrochloride and all medicines out of the reach of children.
General information about donepezil hydrochloride
Medicines are sometimes prescribed for conditions that are not mentioned in this Patient Information Leaflet. Do not use donepezil hydrochloride for a condition for which it was not prescribed. Do not give donepezil hydrochloride to other people, even if they have the same symptoms or condition. It may harm them.
This leaflet summarizes the most important information about donepezil hydrochloride. If you would like more information, talk with your doctor. You can ask your pharmacist or doctor for information about donepezil hydrochloride that is written for health professionals.
What are the ingredients in Donepezil Hydrochloride Tablets, USP?
Active ingredient: donepezil hydrochloride
Inactive ingredients:
Donepezil Hydrochloride 5 mg and 10 mg film-coated tablets: lactose monohydrate, hydroxypropyl cellulose, magnesium stearate, microcrystalline cellulose, corn starch, croscarmellose sodium. The film of 5 mg tablets coating contains hydroxypropyl methyl cellulose, titanium dioxide, polyethylene glycol, and FD&C blue. The film of 10 mg tablets coating contains hydroxypropyl methyl cellulose, titanium dioxide, polyethylene glycol, iron oxide yellow and iron oxide red.
Rx Only
Manufactured by:
Zhejiang Huahai Pharmaceutical Co., Ltd.
Xunqiao, Linhai, Zhejiang, 317024, China
Distributed by:
Solco Healthcare US, LLC
Somerset, NJ 08873, USA
Revised: 08/2020
Rx Only
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
Donepezil Hydrochloride Tablets
The 10 mg tablets are light yellow, round, film-coated tablets, debossed with ‘HH210' on one side.
NDC: 71335-0900-1: 30 Tablets in a BOTTLE, PLASTIC
NDC: 71335-0900-2: 60 Tablets in a BOTTLE, PLASTIC
NDC: 71335-0900-3: 90 Tablets in a BOTTLE, PLASTIC
Store at controlled room temperature, 15°C to 30°C (59°F to 86°F).
Repackaged/Relabeled by:
Bryant Ranch Prepack, Inc.
Burbank, CA 9150
5.7 Neurological Conditions: Seizures
Cholinomimetics are believed to have some potential to cause generalized convulsions. However, seizure activity also may be a manifestation of Alzheimer's disease.
5.4 Peptic Ulcer Disease and Gi Bleeding (5.4 Peptic Ulcer Disease and GI Bleeding)
Through their primary action, cholinesterase inhibitors may be expected to increase gastric acid secretion due to increased cholinergic activity. Therefore, patients should be monitored closely for symptoms of active or occult gastrointestinal bleeding, especially those at increased risk for developing ulcers, e.g., those with a history of ulcer disease or those receiving concurrent nonsteroidal anti-inflammatory drugs (NSAIDs). Clinical studies of donepezil hydrochloride in a dose of 5 mg/day to 10 mg/day have shown no increase, relative to placebo, in the incidence of either peptic ulcer disease or gastrointestinal bleeding.
14.1 Mild to Moderate Alzheimer's Disease
The effectiveness of donepezil hydrochloride as a treatment for mild to moderate Alzheimer's disease is demonstrated by the results of two randomized, double-blind, placebo-controlled clinical investigations in patients with Alzheimer's disease (diagnosed by NINCDS and DSM III-R criteria, Mini-Mental State Examination ≥ 10 and ≤ 26 and Clinical Dementia Rating of 1 or 2). The mean age of patients participating in donepezil hydrochloride trials was 73 years with a range of 50 to 94. Approximately 62% of patients were women and 38% were men. The racial distribution was white 95%, black 3% and other races 2%.
The higher dose of 10 mg did not provide a statistically significantly greater clinical benefit than 5 mg. There is a suggestion, however, based upon order of group mean scores and dose trend analyses of data from these clinical trials, that a daily dose of 10 mg of donepezil hydrochloride might provide additional benefit for some patients. Accordingly, whether or not to employ a dose of 10 mg is a matter of prescriber and patient preference.
13.2 Animal Toxicology And/or Pharmacology (13.2 Animal Toxicology and/or Pharmacology)
In an acute dose neurotoxicity study in female rats, oral administration of donepezil and memantine in combination resulted in increased incidence, severity, and distribution of neurodegeneration compared with memantine alone. The no-effect levels of the combination were associated with clinically relevant plasma donepezil and memantine levels.
The relevance of this finding to humans is unknown.
2.1 Dosing in Mild to Moderate Alzheimer's Disease
The recommended starting dosage of donepezil hydrochloride is 5 mg administered once per day in the evening, just prior to retiring. The maximum recommended dosage of donepezil hydrochloride in patients with mild to moderate Alzheimer’s disease is 10 mg per day. A dose of 10 mg should not be administered until patients have been on a daily dose of 5 mg for 4 to 6 weeks.
2.2 Dosing in Moderate to Severe Alzheimer’s Disease
The recommended starting dosage of donepezil hydrochloride is 5 mg administered once per day in the evening, just prior to retiring. A dose of 10 mg should not be administered until patients have been on a daily dose of 5 mg for 4 to 6 weeks.
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
No evidence of carcinogenic potential was obtained in an 88-week carcinogenicity study of donepezil conducted in mice at oral doses up to 180 mg/kg/day (approximately 86 times the maximum recommended human dose [MRHD] of 10 mg/day on a mg/m2 basis), or in a 104-week carcinogenicity study in rats at oral doses up to 30 mg/kg/day (approximately 29 times the MRHD on a mg/m2 basis).
Donepezil was negative in a battery of genotoxicity assays (in vitro bacterial reverse mutation, in vitro mouse lymphoma tk, in vitro chromosomal aberration, and in vivo mouse micronucleus).
Donepezil had no effect on fertility in rats at oral doses up to 10 mg/kg/day (approximately 10 times the MRHD on a mg/m2 basis) when administered to males and females prior to and during mating and continuing in females through implantation.
7.2 Use With Cholinomimetics and Other Cholinesterase Inhibitors (7.2 Use with Cholinomimetics and Other Cholinesterase Inhibitors)
A synergistic effect may be expected when cholinesterase inhibitors are given concurrently with succinylcholine, similar neuromuscular blocking agents or cholinergic agonists such as bethanechol.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:39:21.001311 · Updated: 2026-03-14T21:57:28.446364