Loperamide Hydrochloride Tablets Usp, 2 Mg
c26c15a1-3a8a-4562-8c5e-ebbc8b81e0c6
34390-5
HUMAN OTC DRUG LABEL
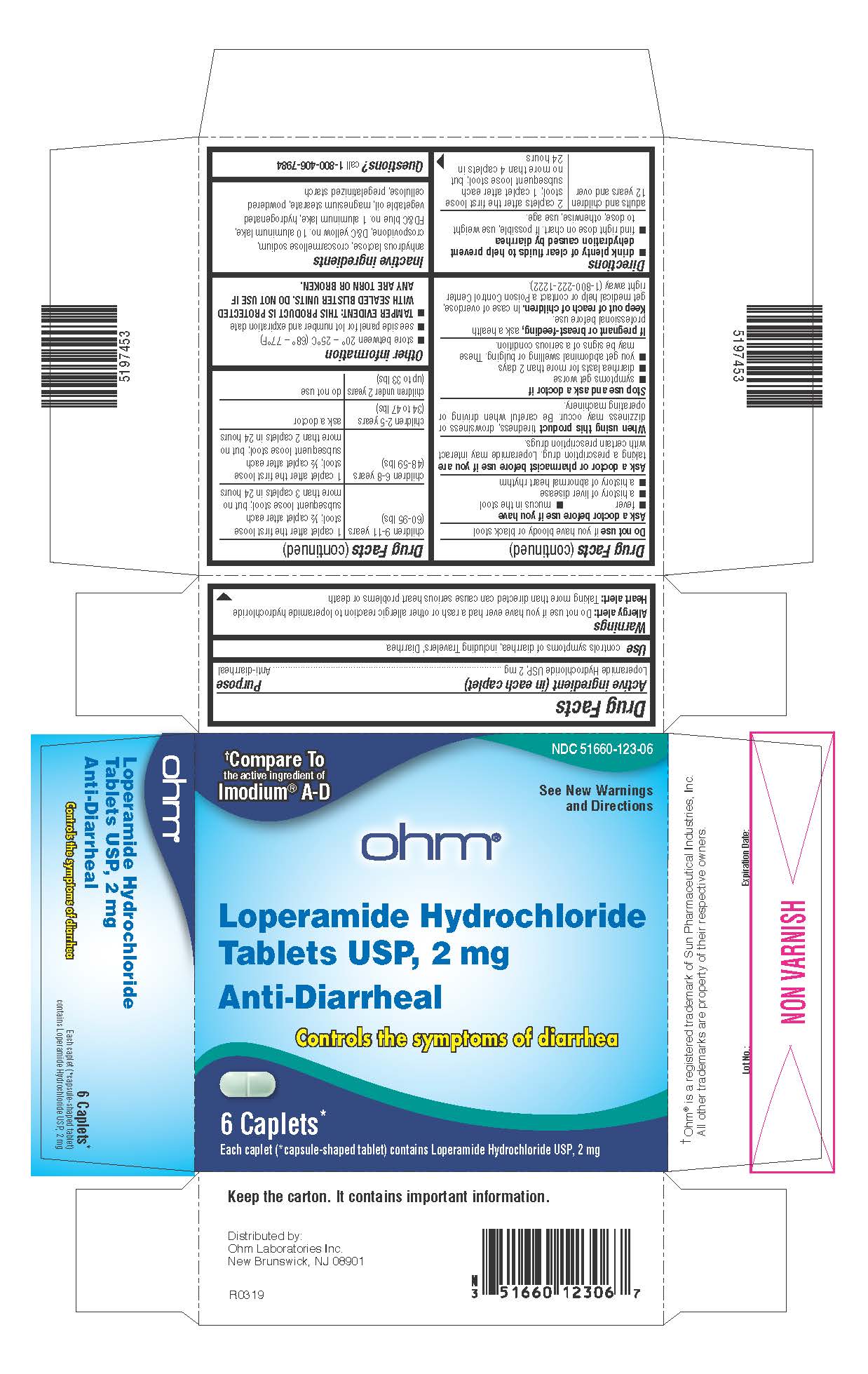
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Drug Facts
Purpose
Anti-diarrheal
Medication Information
Purpose
Anti-diarrheal
Description
Drug Facts
Use
controls symptoms of diarrhea, including Travelers' Diarrhea
Section 42229-5
Drug Facts
Section 50565-1
Keep out of reach of children.In case of overdose, get medical help or contact a Poison Control Center right away (1-800-222-1222).
Section 50567-7
When using this producttiredness, drowsiness or dizziness may occur. Be careful when driving or operating machinery.
Section 50568-5
Ask a doctor or pharmacist before use if you aretaking a prescription drug. Loperamide may interact with certain prescription drugs.
Section 50570-1
Do not useif you have bloody or black stool
Section 53414-9
If pregnant or breast-feeding,ask a health professional before use.
Warnings
Allergy alert:Do not use if you have ever had a rash or other allergic reaction to loperamide hydrochloride
Heart alert:Taking more than directed can cause serious heart problems or death
Directions
- drink plenty of clear fluids to help prevent dehydration caused by diarrhea
- find right dose on chart. If possible, use weight to dose; otherwise, use age.
|
adults and children 12 years and over |
2 caplets after the first loose stool; 1 caplet after each subsequent loose stool; but no more than 4 caplets in 24 hours |
|
children 9-11 years (60-95 lbs) |
1 caplet after the first loose stool; ½ caplet after each subsequent loose stool; but no more than 3 caplets in 24 hours |
|
children 6-8 years (48-59 lbs) |
1 caplet after the first loose stool; ½ caplet after each subsequent loose stool; but no more than 2 caplets in 24 hours |
|
children 2-5 years (34 to 47 lbs) |
ask a doctor |
|
children under 2 years (up to 33 lbs) |
do not use |
Questions?
call 1-800-406-7984
Other Information
- store between 20° – 25°C (68° – 77°F)
- see side panel for lot number and expiration date
- TAMPER EVIDENT: THIS PRODUCT IS PROTECTED WITH SEALED BLISTER UNITS. DO NOT USE IF ANY ARE TORN OR BROKEN.
Inactive Ingredients
anhydrous lactose, croscarmellose sodium, crospovidone, D&C yellow no. 10 aluminum lake, FD&C blue no. 1 aluminum lake, hydrogenated vegetable oil, magnesium stearate, powdered cellulose, pregelatinized starch
Principal Display Panel
NDC 51660-123-06
†Compare To
the active ingredient of
Imodium
®A-D
See New Warnings
and Directions
ohm ®
Loperamide Hydrochloride
Tablets USP, 2 mg
Anti-Diarrheal
Controls the symptoms of diarrhea
6 Caplets*
Each caplet (*capsule-shaped tablet) contains Loperamide Hydrochloride USP, 2 mg
†Ohm ®is a registered trademark of Sun Pharmaceutical Industries, Inc. All other trademarks are property of their respective owners.
Stop Use and Ask A Doctor If
- symptoms get worse
- diarrhea lasts for more than 2 days
- you get abdominal swelling or bulging. These may be signs of a serious condition.
Active Ingredient (in Each Caplet)
Loperamide Hydrochloride USP, 2 mg
Ask A Doctor Before Use If You Have
- fever
- mucus in the stool
- a history of liver disease
- a history of abnormal heart rhythm
Structured Label Content
Use
controls symptoms of diarrhea, including Travelers' Diarrhea
Section 42229-5 (42229-5)
Drug Facts
Section 50565-1 (50565-1)
Keep out of reach of children.In case of overdose, get medical help or contact a Poison Control Center right away (1-800-222-1222).
Section 50567-7 (50567-7)
When using this producttiredness, drowsiness or dizziness may occur. Be careful when driving or operating machinery.
Section 50568-5 (50568-5)
Ask a doctor or pharmacist before use if you aretaking a prescription drug. Loperamide may interact with certain prescription drugs.
Section 50570-1 (50570-1)
Do not useif you have bloody or black stool
Section 53414-9 (53414-9)
If pregnant or breast-feeding,ask a health professional before use.
Purpose
Anti-diarrheal
Warnings
Allergy alert:Do not use if you have ever had a rash or other allergic reaction to loperamide hydrochloride
Heart alert:Taking more than directed can cause serious heart problems or death
Directions
- drink plenty of clear fluids to help prevent dehydration caused by diarrhea
- find right dose on chart. If possible, use weight to dose; otherwise, use age.
|
adults and children 12 years and over |
2 caplets after the first loose stool; 1 caplet after each subsequent loose stool; but no more than 4 caplets in 24 hours |
|
children 9-11 years (60-95 lbs) |
1 caplet after the first loose stool; ½ caplet after each subsequent loose stool; but no more than 3 caplets in 24 hours |
|
children 6-8 years (48-59 lbs) |
1 caplet after the first loose stool; ½ caplet after each subsequent loose stool; but no more than 2 caplets in 24 hours |
|
children 2-5 years (34 to 47 lbs) |
ask a doctor |
|
children under 2 years (up to 33 lbs) |
do not use |
Questions?
call 1-800-406-7984
Other Information (Other information)
- store between 20° – 25°C (68° – 77°F)
- see side panel for lot number and expiration date
- TAMPER EVIDENT: THIS PRODUCT IS PROTECTED WITH SEALED BLISTER UNITS. DO NOT USE IF ANY ARE TORN OR BROKEN.
Inactive Ingredients (Inactive ingredients)
anhydrous lactose, croscarmellose sodium, crospovidone, D&C yellow no. 10 aluminum lake, FD&C blue no. 1 aluminum lake, hydrogenated vegetable oil, magnesium stearate, powdered cellulose, pregelatinized starch
Principal Display Panel (PRINCIPAL DISPLAY PANEL)
NDC 51660-123-06
†Compare To
the active ingredient of
Imodium
®A-D
See New Warnings
and Directions
ohm ®
Loperamide Hydrochloride
Tablets USP, 2 mg
Anti-Diarrheal
Controls the symptoms of diarrhea
6 Caplets*
Each caplet (*capsule-shaped tablet) contains Loperamide Hydrochloride USP, 2 mg
†Ohm ®is a registered trademark of Sun Pharmaceutical Industries, Inc. All other trademarks are property of their respective owners.
Stop Use and Ask A Doctor If (Stop use and ask a doctor if)
- symptoms get worse
- diarrhea lasts for more than 2 days
- you get abdominal swelling or bulging. These may be signs of a serious condition.
Active Ingredient (in Each Caplet) (Active ingredient (in each caplet))
Loperamide Hydrochloride USP, 2 mg
Ask A Doctor Before Use If You Have (Ask a doctor before use if you have)
- fever
- mucus in the stool
- a history of liver disease
- a history of abnormal heart rhythm
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:02:41.380962 · Updated: 2026-03-14T23:02:23.126471