Loratidine Orally Disintegrating Tablets, Usp
c20b916d-7dd0-1a56-e053-2995a90a3c1b
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Drug Facts
Purpose
Antihistamine
Medication Information
Purpose
Antihistamine
Description
Drug Facts
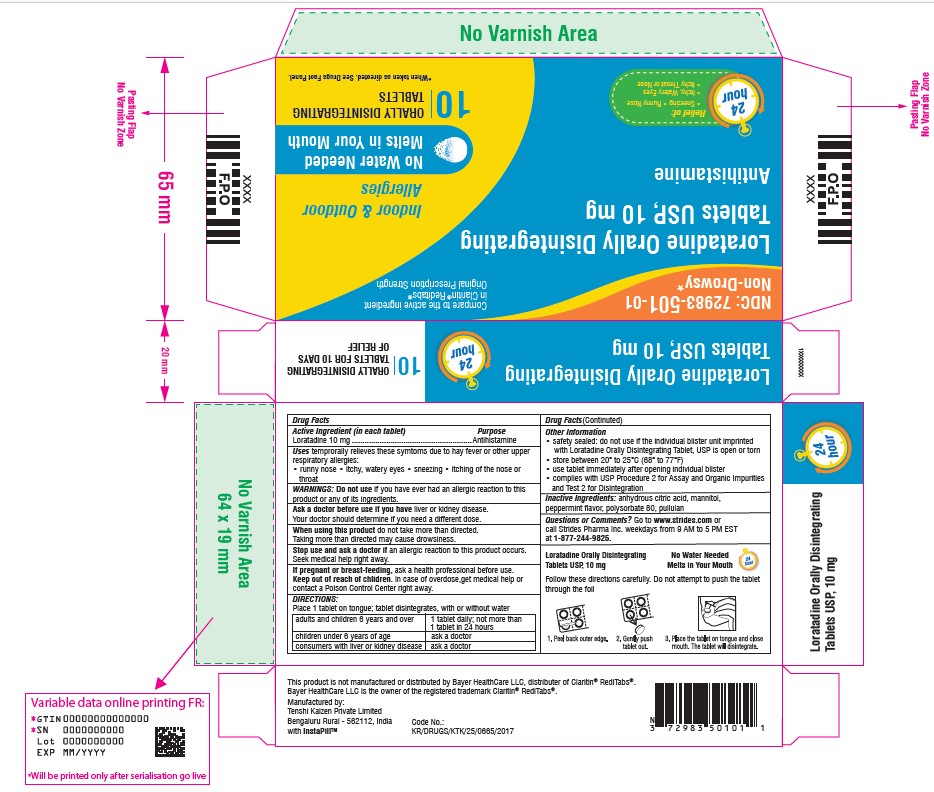
Uses
temporarily relieves these symptoms due to hay fever or other upper respiratory allergies:
- runny nose
- itchy, watery eyes
- sneezing
- itching of the nose or throat
Section 42229-5
Drug Facts
Section 50565-1
Keep out of reach of children. In case of overdose, get medical help or contact a Poison Control Center right away.
Section 50566-9
Stop use and ask a doctor if an allergic reaction to this product occurs. Seek medical help right away.
Section 50567-7
When using this product do not take more than directed. Taking more than directed may cause drowsiness.
Section 50569-3
Ask a doctor before use if you have liver or kidney disease. Your doctor should determine if you need a different dose.
Section 50570-1
Do not use if you have ever had an allergic reaction to this product or any of its ingredients.
Section 53414-9
If pregnant or breast-feeding, ask a health professional before use.
Warnings
Directions
- place 1 tablet on tongue; tablet disintegrates, with or without water
| adults and children 6 years and over | 1 tablet daily; not more than 1 tablet in 24 hours |
| children under 6 years of age | ask a doctor |
| consumers with liver or kidney disease | ask a doctor |
Other Information
- safety sealed: do not use if the individual blister unit imprinted with Loratadine Orally Disintegrating Tablet, USP is open or torn
- store between 20° to 25°C (68° to 77°F)
- use tablet immediately after opening individual blister
- complies with USP Procedure 2 for Assay and Organic Impurities and Test 2 for Disintegration
Inactive Ingredients
anhydrous citric acid, mannitol, peppermint flavor, polysorbate 80, pullulan
Questions Or Comments?
1-877-244-9825
Active Ingredient (in Each Tablet)
Loratadine 10 mg
Package Label.principal Display Panel
NDC 72983-501-01
Non-Drowsy*
Loratadine Orally Disintegrating Tablets USP, 10 mg
antihistamine
Indoor & Outdoor
Allergies
*When taken as directed. See Drug Facts Panel.
24 Hour Relief of:
Sneezing
Runny Nose
Itchy, Watery Eyes
Itchy Throat or Nose
No Water Needed
Melts in Your Mouth
10
ORALLY
DISINTEGRATING TABLETS
Structured Label Content
Uses
temporarily relieves these symptoms due to hay fever or other upper respiratory allergies:
- runny nose
- itchy, watery eyes
- sneezing
- itching of the nose or throat
Section 42229-5 (42229-5)
Drug Facts
Section 50565-1 (50565-1)
Keep out of reach of children. In case of overdose, get medical help or contact a Poison Control Center right away.
Section 50566-9 (50566-9)
Stop use and ask a doctor if an allergic reaction to this product occurs. Seek medical help right away.
Section 50567-7 (50567-7)
When using this product do not take more than directed. Taking more than directed may cause drowsiness.
Section 50569-3 (50569-3)
Ask a doctor before use if you have liver or kidney disease. Your doctor should determine if you need a different dose.
Section 50570-1 (50570-1)
Do not use if you have ever had an allergic reaction to this product or any of its ingredients.
Section 53414-9 (53414-9)
If pregnant or breast-feeding, ask a health professional before use.
Purpose
Antihistamine
Warnings
Directions
- place 1 tablet on tongue; tablet disintegrates, with or without water
| adults and children 6 years and over | 1 tablet daily; not more than 1 tablet in 24 hours |
| children under 6 years of age | ask a doctor |
| consumers with liver or kidney disease | ask a doctor |
Other Information (Other information)
- safety sealed: do not use if the individual blister unit imprinted with Loratadine Orally Disintegrating Tablet, USP is open or torn
- store between 20° to 25°C (68° to 77°F)
- use tablet immediately after opening individual blister
- complies with USP Procedure 2 for Assay and Organic Impurities and Test 2 for Disintegration
Inactive Ingredients (Inactive ingredients)
anhydrous citric acid, mannitol, peppermint flavor, polysorbate 80, pullulan
Questions Or Comments? (Questions or comments?)
1-877-244-9825
Active Ingredient (in Each Tablet) (Active ingredient (in each tablet))
Loratadine 10 mg
Package Label.principal Display Panel (PACKAGE LABEL.PRINCIPAL DISPLAY PANEL)
NDC 72983-501-01
Non-Drowsy*
Loratadine Orally Disintegrating Tablets USP, 10 mg
antihistamine
Indoor & Outdoor
Allergies
*When taken as directed. See Drug Facts Panel.
24 Hour Relief of:
Sneezing
Runny Nose
Itchy, Watery Eyes
Itchy Throat or Nose
No Water Needed
Melts in Your Mouth
10
ORALLY
DISINTEGRATING TABLETS
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:02:38.103699 · Updated: 2026-03-14T23:02:18.218337