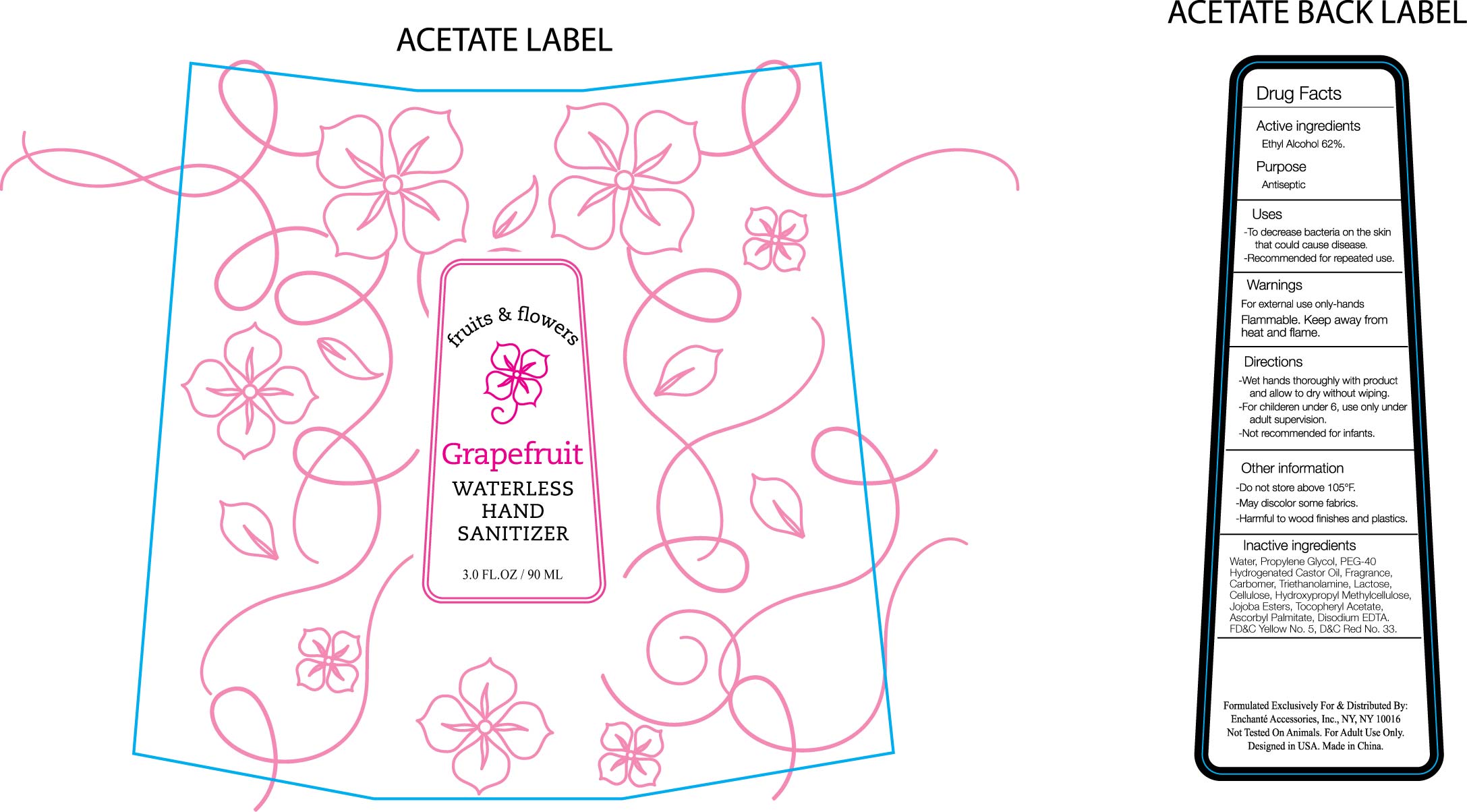
Drug Fact
c209a4c0-fe95-4427-a47a-7d007f3bce41
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Active ingredients Ethyl Alcohol 62%
Medication Information
Warnings and Precautions
For external use only
Flammable: Keep away from heat and flame
Other Information:
Do not store above 105F
May discolor some fabrics
Harmful to wood finishes and plastics
Indications and Usage
To decrease bacteria on hands that can cause disease
Recommended for repeated use.
Dosage and Administration
Wet hands thoroughly with products and allow to dry without wiping.
Description
Active ingredients Ethyl Alcohol 62%
Section 50565-1
For children under 6, use under adult supervision
Not recommended for infants.
Section 51727-6
Water, Propylene Glycol, PEG-40 Hydrogenated Castor Oil, Fragrance, Carbomer, Triethanolamine, Lactose, Cellulose, Hydroxypropyl Methylcellulose, Jojoba Ester, Tocopheryl Acetate, Ascorbyl Palmitate, Disodium EDTA, D and C red No. 33, FD and C Yellow No. 5
Section 51945-4
label
Section 55105-1
Antiseptic
.
Section 55106-9
Active ingredients
Ethyl Alcohol 62%
Structured Label Content
Indications and Usage (34067-9)
To decrease bacteria on hands that can cause disease
Recommended for repeated use.
Dosage and Administration (34068-7)
Wet hands thoroughly with products and allow to dry without wiping.
Warnings and Precautions (34071-1)
For external use only
Flammable: Keep away from heat and flame
Other Information:
Do not store above 105F
May discolor some fabrics
Harmful to wood finishes and plastics
Section 50565-1 (50565-1)
For children under 6, use under adult supervision
Not recommended for infants.
Section 51727-6 (51727-6)
Water, Propylene Glycol, PEG-40 Hydrogenated Castor Oil, Fragrance, Carbomer, Triethanolamine, Lactose, Cellulose, Hydroxypropyl Methylcellulose, Jojoba Ester, Tocopheryl Acetate, Ascorbyl Palmitate, Disodium EDTA, D and C red No. 33, FD and C Yellow No. 5
Section 51945-4 (51945-4)
label
Section 55105-1 (55105-1)
Antiseptic
.
Section 55106-9 (55106-9)
Active ingredients
Ethyl Alcohol 62%
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:55:07.192984 · Updated: 2026-03-14T22:52:27.184723