After Care
c19c5ffe-3b62-43e1-83c2-d89c7b9aa2ae
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Drug Facts
Purpose
Skin protectant
Medication Information
Purpose
Skin protectant
Description
Drug Facts
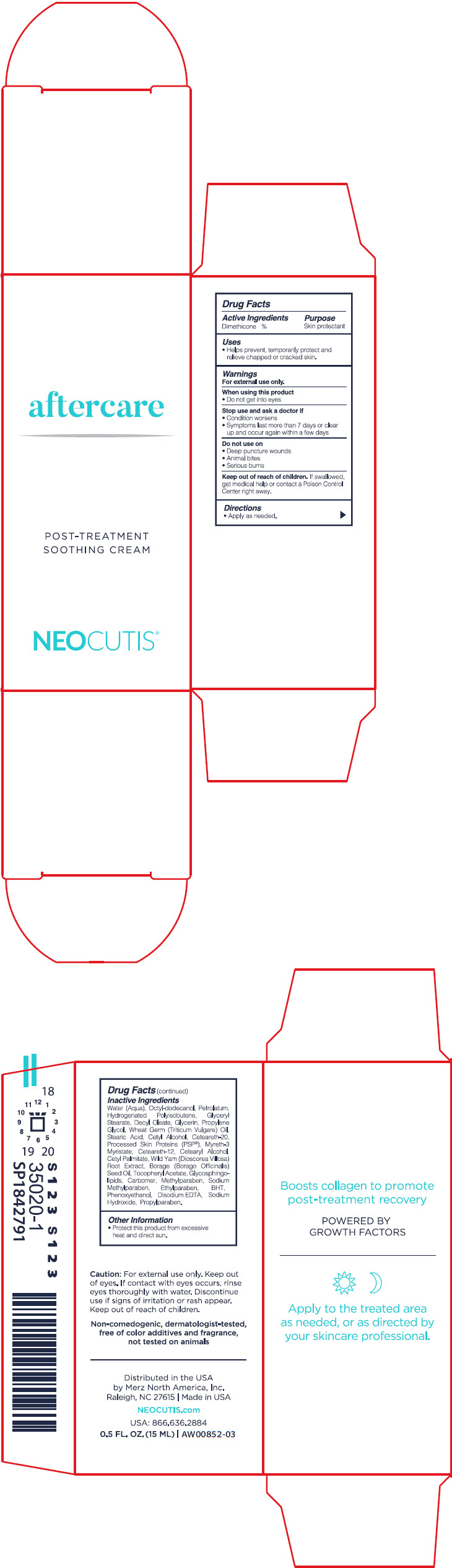
Uses
- Helps prevent, temporarily protect and relieve chapped or cracked skin.
Section 42229-5
Drug Facts
Section 50565-1
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center right away.
Warnings
For external use only.
Directions
- Apply as needed.
Do Not Use On
- Deep or puncture wounds
- Animal bites
- Serious burns
Other Information
- Protect this product from excessive heat and direct sun.
Active Ingredients
Dimethicone 4%
Inactive Ingredients
Water (Aqua), Octyl-Dodecanol, Petrolatum, Hydrogenated Polyisobutene, Glyceryl Stearate, Decyl Oleate, Glycerin, Propylene Glycol, Wheat Germ (Triticum Vulgare) Oil, Stearic Acid, Cetyl Alcohol, Ceteareth-20, Processed Skin Proteins (PSP(R)), Myreth-3 Myristate, Ceteareth-12, Cetearyl Alcohol, Cetyl Palmitate, Wild Yam (Dioscorea Villosa) Root Extract, Borage (Borago Officinalis) Seed Oil, Tocopheryl Acetate, Glycosphingolipids, Carbomer, Methylparaben, Sodium Methylparaben, Ethylparaben, BHT, Phenoxyethanol, Disodium EDTA, Sodium Hydroxide, Propylparaben.
When Using This Product
- Do not get into eyes
Stop Use and Ask A Doctor If
- Condition worsens
- Symptoms last more than 7 days or clear up and occur again within a few days
Principal Display Panel 15 Ml Bottle Carton
aftercare
POST-TREATMENT
SOOTHING CREAM
NEOCUTIS®
Structured Label Content
Uses
- Helps prevent, temporarily protect and relieve chapped or cracked skin.
Section 42229-5 (42229-5)
Drug Facts
Section 50565-1 (50565-1)
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center right away.
Purpose
Skin protectant
Warnings
For external use only.
Directions
- Apply as needed.
Do Not Use On (Do not use on)
- Deep or puncture wounds
- Animal bites
- Serious burns
Other Information (Other information)
- Protect this product from excessive heat and direct sun.
Active Ingredients (Active ingredients)
Dimethicone 4%
Inactive Ingredients (Inactive ingredients)
Water (Aqua), Octyl-Dodecanol, Petrolatum, Hydrogenated Polyisobutene, Glyceryl Stearate, Decyl Oleate, Glycerin, Propylene Glycol, Wheat Germ (Triticum Vulgare) Oil, Stearic Acid, Cetyl Alcohol, Ceteareth-20, Processed Skin Proteins (PSP(R)), Myreth-3 Myristate, Ceteareth-12, Cetearyl Alcohol, Cetyl Palmitate, Wild Yam (Dioscorea Villosa) Root Extract, Borage (Borago Officinalis) Seed Oil, Tocopheryl Acetate, Glycosphingolipids, Carbomer, Methylparaben, Sodium Methylparaben, Ethylparaben, BHT, Phenoxyethanol, Disodium EDTA, Sodium Hydroxide, Propylparaben.
When Using This Product (When using this product)
- Do not get into eyes
Stop Use and Ask A Doctor If (Stop use and ask a doctor if)
- Condition worsens
- Symptoms last more than 7 days or clear up and occur again within a few days
Principal Display Panel 15 Ml Bottle Carton (PRINCIPAL DISPLAY PANEL - 15 ML Bottle Carton)
aftercare
POST-TREATMENT
SOOTHING CREAM
NEOCUTIS®
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:04:20.453479 · Updated: 2026-03-14T23:04:59.717126