Piperacillin And Tazobactam For Injection
c18109cf-d5db-4cc1-83da-537c8ab0e05c
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Indications and Usage
Piperacillin and tazobactam for injection is a combination of piperacillin, a penicillin-class antibacterial and tazobactam, a beta-lactamase inhibitor, indicated for the treatment of: Intra-abdominal infections in adult and pediatric patients 2 months of age and older ( 1.1 ) Nosocomial pneumonia in adult and pediatric patients 2 months of age and older ( 1.2 ) Skin and skin structure infections in adults ( 1.3 ) Female pelvic infections in adults ( 1.4 ) Community-acquired pneumonia in adults ( 1.5 ) To reduce the development of drug-resistant bacteria and maintain the effectiveness of piperacillin and tazobactam for injection and other antibacterial drugs, piperacillin and tazobactam for injection should be used only to treat or prevent infections that are proven or strongly suspected to be caused by bacteria. ( 1.6 )
Dosage and Administration
Adult Patients With Indications Other Than Nosocomial Pneumonia; The usual daily dosage of piperacillin and tazobactam for injection for adults is 3.375 g every six hours totaling 13.5 g (12.0 g piperacillin and 1.5 g tazobactam). ( 2.1 ) Adult Patients with Nosocomial Pneumonia: Initial presumptive treatment of patients with nosocomial pneumonia should start with piperacillin and tazobactam for injection at a dosage of 4.5 g every six hours plus an aminoglycoside, totaling 18.0 g (16.0 g piperacillin and 2.0 g tazobactam). ( 2.2 ) Adult Patients with Renal Impairment : Dosage in patients with renal impairment (creatinine clearance ≤40 mL/min) and dialysis patients should be reduced, based on the degree of renal impairment. ( 2.3 ) Pediatric Patients by Indication and Age: See Table below ( 2.4 ) Recommended Dosage of Piperacillin and Tazobactam for Injection for Pediatric Patients 2 months of Age and Older, Weighing up to 40 Kg and With Normal Renal Function Age Appendicitis and /or Peritonitis Nosocomial Pneumonia 2 months to 9 months 90 mg/kg (80 mg piperacillin and 10 mg tazobactam) every 8 ( eight ) hours 90 mg/kg (80 mg piperacillin and 10 mg tazobactam) every 6 ( six ) hours Older than 9 months 112.5 mg/kg (100 mg piperacillin and 12.5 mg tazobactam) every 8 ( eight) hours 112.5 mg/kg (100 mg piperacillin and 12.5 mg tazobactam) every 6 ( six) hours Administer piperacillin and tazobactam for injection by intravenous infusion over 30 minutes to both adult and pediatric patients ( 2.1 , 2.2 , 2.3 , 2.4 ). Piperacillin and tazobactam for injection and aminoglycosides should be reconstituted, diluted, and administered separately. Co-administration via Y-site can be done under certain conditions. ( 2.6 ) See the full prescribing information for the preparation and administration instructions for piperacillin and tazobactam for injection pharmacy bulk vials.
Contraindications
Piperacillin and tazobactam for injection is contraindicated in patients with a history of allergic reactions to any of the penicillins, cephalosporins, or beta-lactamase inhibitors.
Warnings and Precautions
Serious hypersensitivity reactions (anaphylactic/anaphylactoid) reactions have been reported in patients receiving piperacillin and tazobactam for injection. Discontinue piperacillin and tazobactam for injection if a reaction occurs. ( 5.1 ) Piperacillin and tazobactam for injection may cause severe cutaneous adverse reactions, such as Stevens-Johnson syndrome, toxic epidermal necrolysis, drug reaction with eosinophilia and systemic symptoms, and acute generalized exanthematous pustulosis. Discontinue piperacillin and tazobactam for injection for progressive rashes. ( 5.2 ) Hemophagocytic lymphohistiocytosis (HLH) has been reported with the use of piperacillin and tazobactam for injection. If HLH is suspected, discontinue piperacillin and tazobactam for injection immediately. ( 5.3 ) Rhabdomyolysis: If signs or symptoms of rhabdomyolysis are observed, discontinue Piperacillin and tazobactam for injection and initiate appropriate therapy. ( 5.4 ) Hematological effects (including bleeding, leukopenia and neutropenia) have occurred. Monitor hematologic tests during prolonged therapy. ( 5.5 ) As with other penicillins, piperacillin and tazobactam for injection may cause neuromuscular excitability or seizures. Patients receiving higher doses, especially in the presence of renal impairment may be at greater risk. Closely monitor patients with renal impairment or seizure disorders for signs and symptoms of neuromuscular excitability or seizures. ( 5.6 ) Nephrotoxicity in critically ill patients has been observed; the use of piperacillin and tazobactam for injection was found to be an independent risk factor for renal failure and was associated with delayed recovery of renal function as compared to other beta-lactam antibacterial drugs in a randomized, multicenter, controlled trial in critically ill patients. Based on this study, alternative treatment options should be considered in the critically ill population. If alternative treatment options are inadequate or unavailable, monitor renal function during treatment with piperacillin and tazobactam for injection. ( 5.7 ) Clostridioides difficile- associated diarrhea: evaluate patients if diarrhea occurs. ( 5.9 )
Adverse Reactions
Bleeding manifestations have occurred in some patients receiving beta-lactam drugs, including piperacillin. These reactions have sometimes been associated with abnormalities of coagulation tests such as clotting time, platelet aggregation and prothrombin time, and are more likely to occur in patients with renal failure. If bleeding manifestations occur, piperacillin and tazobactam for injection should be discontinued and appropriate therapy instituted. The leukopenia/neutropenia associated with piperacillin and tazobactam for injection administration appears to be reversible and most frequently associated with prolonged administration. Periodic assessment of hematopoietic function should be performed, especially with prolonged therapy, i.e., ≥ 21 days [see Adverse Reactions (6.1) ] .
Drug Interactions
Piperacillin and tazobactam for injection administration can significantly reduce tobramycin concentrations in hemodialysis patients. Monitor tobramycin concentrations in these patients. ( 7.1 ) Probenecid prolongs the half-lives of piperacillin and tazobactam and should not be co-administered with piperacillin and tazobactam for injection unless the benefit outweighs the risk. ( 7.2 ) Co-administration of piperacillin and tazobactam for injection with vancomycin may increase the incidence of acute kidney injury. Monitor kidney function in patients receiving piperacillin and tazobactam for injection and vancomycin. ( 7.3 ) Monitor coagulation parameters in patients receiving piperacillin and tazobactam for injection and heparin or oral anticoagulants. ( 7.4 ) Piperacillin and tazobactam for injection may prolong the neuromuscular blockade of vecuronium and other non-depolarizing neuromuscular blockers. Monitor for adverse reactions related to neuromuscular blockade. ( 7.5 )
How Supplied
Piperacillin and tazobactam for injection, USP is supplied as a white to off-white sterile, cryodesiccated powder in the pharmacy bulk vial as follows: Each piperacillin and tazobactam for injection, USP 40.5 g pharmacy bulk vial provides piperacillin sodium equivalent to 36 grams of piperacillin and tazobactam sodium equivalent to 4.5 grams of tazobactam. Each pharmacy bulk vial contains 84.5 mEq (1,944 mg) of sodium. NDC 60505-6262-0 Piperacillin and tazobactam for injection, USP pharmacy bulk vials should be stored at 20° to 25°C (68° to 77°F) [See USP Controlled Room Temperature] prior to reconstitution.
Storage and Handling
Piperacillin and tazobactam for injection, USP is supplied as a white to off-white sterile, cryodesiccated powder in the pharmacy bulk vial as follows: Each piperacillin and tazobactam for injection, USP 40.5 g pharmacy bulk vial provides piperacillin sodium equivalent to 36 grams of piperacillin and tazobactam sodium equivalent to 4.5 grams of tazobactam. Each pharmacy bulk vial contains 84.5 mEq (1,944 mg) of sodium. NDC 60505-6262-0 Piperacillin and tazobactam for injection, USP pharmacy bulk vials should be stored at 20° to 25°C (68° to 77°F) [See USP Controlled Room Temperature] prior to reconstitution.
Description
Warnings and Precautions, Rhabdomyolysis ( 5.4 ) 08/2024
Medication Information
Warnings and Precautions
Serious hypersensitivity reactions (anaphylactic/anaphylactoid) reactions have been reported in patients receiving piperacillin and tazobactam for injection. Discontinue piperacillin and tazobactam for injection if a reaction occurs. ( 5.1 ) Piperacillin and tazobactam for injection may cause severe cutaneous adverse reactions, such as Stevens-Johnson syndrome, toxic epidermal necrolysis, drug reaction with eosinophilia and systemic symptoms, and acute generalized exanthematous pustulosis. Discontinue piperacillin and tazobactam for injection for progressive rashes. ( 5.2 ) Hemophagocytic lymphohistiocytosis (HLH) has been reported with the use of piperacillin and tazobactam for injection. If HLH is suspected, discontinue piperacillin and tazobactam for injection immediately. ( 5.3 ) Rhabdomyolysis: If signs or symptoms of rhabdomyolysis are observed, discontinue Piperacillin and tazobactam for injection and initiate appropriate therapy. ( 5.4 ) Hematological effects (including bleeding, leukopenia and neutropenia) have occurred. Monitor hematologic tests during prolonged therapy. ( 5.5 ) As with other penicillins, piperacillin and tazobactam for injection may cause neuromuscular excitability or seizures. Patients receiving higher doses, especially in the presence of renal impairment may be at greater risk. Closely monitor patients with renal impairment or seizure disorders for signs and symptoms of neuromuscular excitability or seizures. ( 5.6 ) Nephrotoxicity in critically ill patients has been observed; the use of piperacillin and tazobactam for injection was found to be an independent risk factor for renal failure and was associated with delayed recovery of renal function as compared to other beta-lactam antibacterial drugs in a randomized, multicenter, controlled trial in critically ill patients. Based on this study, alternative treatment options should be considered in the critically ill population. If alternative treatment options are inadequate or unavailable, monitor renal function during treatment with piperacillin and tazobactam for injection. ( 5.7 ) Clostridioides difficile- associated diarrhea: evaluate patients if diarrhea occurs. ( 5.9 )
Indications and Usage
Piperacillin and tazobactam for injection is a combination of piperacillin, a penicillin-class antibacterial and tazobactam, a beta-lactamase inhibitor, indicated for the treatment of: Intra-abdominal infections in adult and pediatric patients 2 months of age and older ( 1.1 ) Nosocomial pneumonia in adult and pediatric patients 2 months of age and older ( 1.2 ) Skin and skin structure infections in adults ( 1.3 ) Female pelvic infections in adults ( 1.4 ) Community-acquired pneumonia in adults ( 1.5 ) To reduce the development of drug-resistant bacteria and maintain the effectiveness of piperacillin and tazobactam for injection and other antibacterial drugs, piperacillin and tazobactam for injection should be used only to treat or prevent infections that are proven or strongly suspected to be caused by bacteria. ( 1.6 )
Dosage and Administration
Adult Patients With Indications Other Than Nosocomial Pneumonia; The usual daily dosage of piperacillin and tazobactam for injection for adults is 3.375 g every six hours totaling 13.5 g (12.0 g piperacillin and 1.5 g tazobactam). ( 2.1 ) Adult Patients with Nosocomial Pneumonia: Initial presumptive treatment of patients with nosocomial pneumonia should start with piperacillin and tazobactam for injection at a dosage of 4.5 g every six hours plus an aminoglycoside, totaling 18.0 g (16.0 g piperacillin and 2.0 g tazobactam). ( 2.2 ) Adult Patients with Renal Impairment : Dosage in patients with renal impairment (creatinine clearance ≤40 mL/min) and dialysis patients should be reduced, based on the degree of renal impairment. ( 2.3 ) Pediatric Patients by Indication and Age: See Table below ( 2.4 ) Recommended Dosage of Piperacillin and Tazobactam for Injection for Pediatric Patients 2 months of Age and Older, Weighing up to 40 Kg and With Normal Renal Function Age Appendicitis and /or Peritonitis Nosocomial Pneumonia 2 months to 9 months 90 mg/kg (80 mg piperacillin and 10 mg tazobactam) every 8 ( eight ) hours 90 mg/kg (80 mg piperacillin and 10 mg tazobactam) every 6 ( six ) hours Older than 9 months 112.5 mg/kg (100 mg piperacillin and 12.5 mg tazobactam) every 8 ( eight) hours 112.5 mg/kg (100 mg piperacillin and 12.5 mg tazobactam) every 6 ( six) hours Administer piperacillin and tazobactam for injection by intravenous infusion over 30 minutes to both adult and pediatric patients ( 2.1 , 2.2 , 2.3 , 2.4 ). Piperacillin and tazobactam for injection and aminoglycosides should be reconstituted, diluted, and administered separately. Co-administration via Y-site can be done under certain conditions. ( 2.6 ) See the full prescribing information for the preparation and administration instructions for piperacillin and tazobactam for injection pharmacy bulk vials.
Contraindications
Piperacillin and tazobactam for injection is contraindicated in patients with a history of allergic reactions to any of the penicillins, cephalosporins, or beta-lactamase inhibitors.
Adverse Reactions
Bleeding manifestations have occurred in some patients receiving beta-lactam drugs, including piperacillin. These reactions have sometimes been associated with abnormalities of coagulation tests such as clotting time, platelet aggregation and prothrombin time, and are more likely to occur in patients with renal failure. If bleeding manifestations occur, piperacillin and tazobactam for injection should be discontinued and appropriate therapy instituted. The leukopenia/neutropenia associated with piperacillin and tazobactam for injection administration appears to be reversible and most frequently associated with prolonged administration. Periodic assessment of hematopoietic function should be performed, especially with prolonged therapy, i.e., ≥ 21 days [see Adverse Reactions (6.1) ] .
Drug Interactions
Piperacillin and tazobactam for injection administration can significantly reduce tobramycin concentrations in hemodialysis patients. Monitor tobramycin concentrations in these patients. ( 7.1 ) Probenecid prolongs the half-lives of piperacillin and tazobactam and should not be co-administered with piperacillin and tazobactam for injection unless the benefit outweighs the risk. ( 7.2 ) Co-administration of piperacillin and tazobactam for injection with vancomycin may increase the incidence of acute kidney injury. Monitor kidney function in patients receiving piperacillin and tazobactam for injection and vancomycin. ( 7.3 ) Monitor coagulation parameters in patients receiving piperacillin and tazobactam for injection and heparin or oral anticoagulants. ( 7.4 ) Piperacillin and tazobactam for injection may prolong the neuromuscular blockade of vecuronium and other non-depolarizing neuromuscular blockers. Monitor for adverse reactions related to neuromuscular blockade. ( 7.5 )
Storage and Handling
Piperacillin and tazobactam for injection, USP is supplied as a white to off-white sterile, cryodesiccated powder in the pharmacy bulk vial as follows: Each piperacillin and tazobactam for injection, USP 40.5 g pharmacy bulk vial provides piperacillin sodium equivalent to 36 grams of piperacillin and tazobactam sodium equivalent to 4.5 grams of tazobactam. Each pharmacy bulk vial contains 84.5 mEq (1,944 mg) of sodium. NDC 60505-6262-0 Piperacillin and tazobactam for injection, USP pharmacy bulk vials should be stored at 20° to 25°C (68° to 77°F) [See USP Controlled Room Temperature] prior to reconstitution.
How Supplied
Piperacillin and tazobactam for injection, USP is supplied as a white to off-white sterile, cryodesiccated powder in the pharmacy bulk vial as follows: Each piperacillin and tazobactam for injection, USP 40.5 g pharmacy bulk vial provides piperacillin sodium equivalent to 36 grams of piperacillin and tazobactam sodium equivalent to 4.5 grams of tazobactam. Each pharmacy bulk vial contains 84.5 mEq (1,944 mg) of sodium. NDC 60505-6262-0 Piperacillin and tazobactam for injection, USP pharmacy bulk vials should be stored at 20° to 25°C (68° to 77°F) [See USP Controlled Room Temperature] prior to reconstitution.
Description
Warnings and Precautions, Rhabdomyolysis ( 5.4 ) 08/2024
Section 42229-5
Reconstitution of Piperacillin and Tazobactam for Injection for Adult Patients and Pediatric Patients Weighing Over 40 kg
Section 43679-0
Mechanism of Action
Piperacillin sodium exerts bactericidal activity by inhibiting septum formation and cell wall synthesis of susceptible bacteria. In vitro, piperacillin is active against a variety of gram-positive and gram-negative aerobic and anaerobic bacteria. Tazobactam sodium has little clinically relevant in vitro activity against bacteria due to its reduced affinity to penicillin-binding proteins. It is, however, a beta-lactamase inhibitor of the Molecular class A enzymes, including Richmond-Sykes class III (Bush class 2b & 2b') penicillinases and cephalosporinases. It varies in its ability to inhibit class II and IV (2a & 4) penicillinases. Tazobactam does not induce chromosomally-mediated beta-lactamases at tazobactam concentrations achieved with the recommended dosage regimen.
Section 43683-2
| Warnings and Precautions, Rhabdomyolysis (5.4) | 08/2024 |
1.6 Usage
To reduce the development of drug-resistant bacteria and maintain the effectiveness of piperacillin and tazobactam for injection and other antibacterial drugs, piperacillin and tazobactam for injection should be used only to treat or prevent infections that are proven or strongly suspected to be caused by bacteria. When culture and susceptibility information are available, they should be considered in selecting or modifying antibacterial therapy. In the absence of such data, local epidemiology and susceptibility patterns may contribute to the empiric selection of therapy.
10 Overdosage
There have been postmarketing reports of overdose with piperacillin and tazobactam. The majority of those events experienced, including nausea, vomiting, and diarrhea, have also been reported with the usual recommended dosages. Patients may experience neuromuscular excitability or seizures if higher than recommended doses are given intravenously (particularly in the presence of renal failure) [see Warnings and Precautions (5.6)].
Treatment should be supportive and symptomatic according to the patient's clinical presentation. Excessive serum concentrations of either piperacillin or tazobactam may be reduced by hemodialysis. Following a single 3.375 g dose of piperacillin and tazobactam, the percentage of the piperacillin and tazobactam dose removed by hemodialysis was approximately 31% and 39%, respectively [see Clinical Pharmacology (12)].
15 References
- Jensen J-US, Hein L, Lundgren B, et al. BMJ Open 2012; 2:e000635. doi:10.1136.
11 Description
Piperacillin and tazobactam for injection, USP is an injectable antibacterial combination product consisting of the semisynthetic antibacterial piperacillin sodium and the beta-lactamase inhibitor tazobactam sodium for intravenous administration.
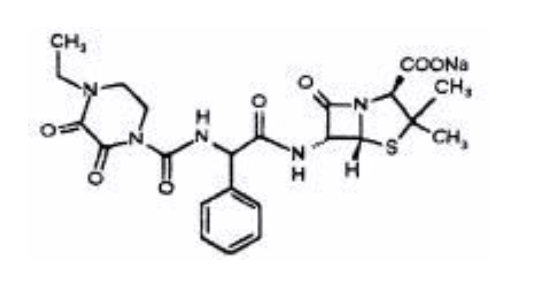
Piperacillin sodium is derived from D(-)-α-aminobenzyl-penicillin. The chemical name of piperacillin sodium is sodium (2S,5R,6R)-6-[(R)-2-(4-ethyl-2,3-dioxo-1-piperazine-carboxamido)-2-phenylacetamido]-3,3-dimethyl-7-oxo-4-thia-1-azabicyclo[3.2.0]heptane-2-carboxylate. The chemical formula is C23H26N5NaO7S and the molecular weight is 539.5. The chemical structure of piperacillin sodium is:
Tazobactam sodium, a derivative of the penicillin nucleus, is a penicillanic acid sulfone. Its chemical name is sodium (2S,3S,5R)-3-methyl-7-oxo-3-(1H-1,2,3-triazol-1-ylmethyl)-4-thia-1-azabicyclo[3.2.0]heptane-2-carboxylate-4,4-dioxide. The chemical formula is C10H11N4NaO5S and the molecular weight is 322.3. The chemical structure of tazobactam sodium is:
Piperacillin and tazobactam for injection contains a total of 2.35 mEq (54 mg) of sodium (Na+) per gram of piperacillin in the combination product.
Piperacillin and tazobactam for injection, USP is a white to off-white sterile, cryodesiccated powder consisting of piperacillin and tazobactam as their sodium salts packaged in glass vials. The product does not contain excipients or preservatives.
Each piperacillin and tazobactam for injection, USP 40.5 g pharmacy bulk vial contains piperacillin sodium equivalent to 36 grams of piperacillin and tazobactam sodium equivalent to 4.5 g of tazobactam sufficient for delivery of multiple doses.
7.2 Probenecid
Probenecid administered concomitantly with piperacillin and tazobactam for injection prolongs the half-life of piperacillin by 21% and that of tazobactam by 71% because probenecid inhibits tubular renal secretion of both piperacillin and tazobactam. Probenecid should not be co-administered with piperacillin and tazobactam for injection unless the benefit outweighs the risk.
7.3 Vancomycin
Studies have detected an increased incidence of acute kidney injury in patients concomitantly administered piperacillin and tazobactam and vancomycin as compared to vancomycin alone [see Warnings and Precautions (5.7)].
Monitor kidney function in patients concomitantly administered with piperacillin and tazobactam and vancomycin.
No pharmacokinetic interactions have been noted between piperacillin and tazobactam and vancomycin.
7.5 Vecuronium
Piperacillin when used concomitantly with vecuronium has been implicated in the prolongation of the neuromuscular blockade of vecuronium. piperacillin and tazobactam for injection could produce the same phenomenon if given along with vecuronium. Due to their similar mechanism of action, it is expected that the neuromuscular blockade produced by any of the non-depolarizing neuromuscular blockers could be prolonged in the presence of piperacillin. Monitor for adverse reactions related to neuromuscular blockade (see package insert for vecuronium bromide).
7.6 Methotrexate
Limited data suggests that co-administration of methotrexate and piperacillin may reduce the clearance of methotrexate due to competition for renal secretion. The impact of tazobactam on the elimination of methotrexate has not been evaluated. If concurrent therapy is necessary, serum concentrations of methotrexate as well as the signs and symptoms of methotrexate toxicity should be frequently monitored.
8.4 Pediatric Use
The safety and effectiveness of piperacillin and tazobactam for injection for intra-abdominal infections, and nosocomial pneumonia have been established in pediatric patients 2 months of age and older.
Use of piperacillin and tazobactam for injection in pediatric patients 2 months of age and older with intra-abdominal infections including appendicitis and/or peritonitis is supported by evidence from well-controlled studies and pharmacokinetic studies in adults and in pediatric patients. This includes a prospective, randomized, comparative, open-label clinical trial with 542 pediatric patients 2 to 12 years of age with intra-abdominal infections (including appendicitis and/or peritonitis), in which 273 pediatric patients received piperacillin and tazobactam [see Adverse Reactions (6.1) and Clinical Pharmacology (12.3)].
Use of piperacillin and tazobactam for injection in pediatric patients 2 months of age and older with nosocomial pneumonia is supported by evidence from well-controlled studies in adults with nosocomial pneumonia, a simulation study performed with a population pharmacokinetic model, and a retrospective, cohort study of pediatric patients with nosocomial pneumonia in which 140 pediatric patients were treated with piperacillin and tazobactam for injection and 267 patients treated with comparators (which included ticarcillin-clavulanate, carbapenems, ceftazidime, cefepime, or ciprofloxacin) [see Adverse Reactions (6.1) and Clinical Pharmacology (12.3)].
The safety and effectiveness of piperacillin and tazobactam for injection have not been established in pediatric patients less than 2 months of age [see Clinical Pharmacology (12) and Dosage and Administration (2)].
Dosage of piperacillin and tazobactam for injection in pediatric patients with renal impairment has not been determined.
8.5 Geriatric Use
Patients over 65 years are not at an increased risk of developing adverse effects solely because of age. However, dosage should be adjusted in the presence of renal impairment [see Dosage and Administration (2)].
In general, dose selection for an elderly patient should be cautious, usually starting at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy.
Piperacillin and tazobactam for injection contains 54 mg (2.35 mEq) of sodium per gram of piperacillin in the combination product. At the usual recommended doses, patients would receive between 648 and 864 mg/day (28.2 and 37.6 mEq) of sodium. The geriatric population may respond with a blunted natriuresis to salt loading. This may be clinically important with regard to such diseases as congestive heart failure.
This drug is known to be substantially excreted by the kidney, and the risk of toxic reactions to this drug may be greater in patients with impaired renal function. Because elderly patients are more likely to have decreased renal function, care should be taken in dose selection, and it may be useful to monitor renal function.
5.4 Rhabdomyolysis
Rhabdomyolysis has been reported with the use of Piperacillin and tazobactam for injection [see Adverse Reactions (6.2)] . If signs or symptoms of rhabdomyolysis such as muscle pain, tenderness or weakness, dark urine, or elevated creatine phosphokinase are observed, discontinue Piperacillin and tazobactam for injection and initiate appropriate therapy.
7.4 Anticoagulants
Coagulation parameters should be tested more frequently and monitored regularly during simultaneous administration of high doses of heparin, oral anticoagulants, or other drugs that may affect the blood coagulation system or the thrombocyte function [see Warnings and Precautions (5.5)].
4 Contraindications
Piperacillin and tazobactam for injection is contraindicated in patients with a history of allergic reactions to any of the penicillins, cephalosporins, or beta-lactamase inhibitors.
6 Adverse Reactions
The following clinically significant adverse reactions are described elsewhere in the labeling:
- Hypersensitivity Adverse Reactions [see Warnings and Precautions (5.1)]
- Severe Cutaneous Adverse Reactions [see Warnings and Precautions (5.2)]
- Hemophagocytic Lymphohistiocytosis [see Warnings and Precautions (5.3)]
- Rhabdomyolysis [see Warnings and Precautions (5.4)]
- Hematologic Adverse Reactions [see Warnings and Precautions (5.5)]
- Central Nervous System Adverse Reactions [see Warnings and Precautions (5.6)]
- Nephrotoxicity in Critically Ill Patients [see Warnings and Precautions (5.7)]
- Clostridioides difficile-Associated Diarrhea [see Warnings and Precautions (5.9)]
7 Drug Interactions
- Piperacillin and tazobactam for injection administration can significantly reduce tobramycin concentrations in hemodialysis patients. Monitor tobramycin concentrations in these patients. (7.1)
- Probenecid prolongs the half-lives of piperacillin and tazobactam and should not be co-administered with piperacillin and tazobactam for injection unless the benefit outweighs the risk. (7.2)
- Co-administration of piperacillin and tazobactam for injection with vancomycin may increase the incidence of acute kidney injury. Monitor kidney function in patients receiving piperacillin and tazobactam for injection and vancomycin. (7.3)
- Monitor coagulation parameters in patients receiving piperacillin and tazobactam for injection and heparin or oral anticoagulants. (7.4)
- Piperacillin and tazobactam for injection may prolong the neuromuscular blockade of vecuronium and other non-depolarizing neuromuscular blockers. Monitor for adverse reactions related to neuromuscular blockade. (7.5)
7.1 Aminoglycosides
Piperacillin may inactivate aminoglycosides by converting them to microbiologically inert amides.
8.6 Renal Impairment
In patients with creatinine clearance ≤ 40 mL/min and dialysis patients (hemodialysis and CAPD), the intravenous dose of piperacillin and tazobactam for injection should be reduced to the degree of renal function impairment [see Dosage and Administration (2)].
12.2 Pharmacodynamics
The pharmacodynamic parameter for piperacillin and tazobactam that is most predictive of clinical and microbiological efficacy is time above MIC.
12.3 Pharmacokinetics
The mean and coefficients of variation (CV%) for the pharmacokinetic parameters of piperacillin and tazobactam after multiple intravenous doses are summarized in Table 8.
| Cmax : maximum observed concentration, AUC: Area under the curve, CL=clearance, CLR= Renal clearance V=volume of distribution, T1/2 = elimination half-life |
||||||
| Piperacillin | ||||||
| Piperacillin and Tazobactam Dose Piperacillin and tazobactam were given in combination, infused over 30 minutes.
|
Cmax (mcg/mL) | AUC Numbers in [] parentheses are coefficients of variation [CV%]. (mcg∙h/mL) |
CL (mL/min) | V (L) |
T1/2
(h) |
CLR (mL/min) |
| 2.25 g | 134 | 131 [14] | 257 | 17.4 | 0.79 | -- |
| 3.375 g | 242 | 242 [10] | 207 | 15.1 | 0.84 | 140 |
| 4.5 g | 298 | 322 [16] | 210 | 15.4 | 0.84 | -- |
| Tazobactam | ||||||
| Piperacillin and Tazobactam Dose | Cmax (mcg/mL) | AUC (mcg∙h/mL) | CL (mL/min) | V (L) |
T1/2 (h) | CLR (mL/min) |
| 2.25 g | 15 | 16.0 [21] | 258 | 17.0 | 0.77 | -- |
| 3.375 g | 24 | 25.0 [8] | 251 | 14.8 | 0.68 | 166 |
| 4.5 g | 34 | 39.8 [15] | 206 | 14.7 | 0.82 | -- |
Peak plasma concentrations of piperacillin and tazobactam are attained immediately after completion of an intravenous infusion of piperacillin and tazobactam for injection. Piperacillin plasma concentrations, following a 30-minute infusion of piperacillin and tazobactam for injection, were similar to those attained when equivalent doses of piperacillin were administered alone. Steady-state plasma concentrations of piperacillin and tazobactam were similar to those attained after the first dose due to the short half-lives of piperacillin and tazobactam.
8.7 Hepatic Impairment
Dosage adjustment of piperacillin and tazobactam for injection is not warranted in patients with hepatic cirrhosis [see Clinical Pharmacology (12.3)].
1 Indications and Usage
Piperacillin and tazobactam for injection is a combination of piperacillin, a penicillin-class antibacterial and tazobactam, a beta-lactamase inhibitor, indicated for the treatment of:
- Intra-abdominal infections in adult and pediatric patients 2 months of age and older (1.1)
- Nosocomial pneumonia in adult and pediatric patients 2 months of age and older (1.2)
- Skin and skin structure infections in adults (1.3)
- Female pelvic infections in adults (1.4)
- Community-acquired pneumonia in adults (1.5)
To reduce the development of drug-resistant bacteria and maintain the effectiveness of piperacillin and tazobactam for injection and other antibacterial drugs, piperacillin and tazobactam for injection should be used only to treat or prevent infections that are proven or strongly suspected to be caused by bacteria. (1.6)
5.8 Electrolyte Effects
Piperacillin and tazobactam for injection contains a total of 2.35 mEq (54 mg) of Na+ (sodium) per gram of piperacillin in the combination product. This should be considered when treating patients requiring restricted salt intake. Periodic electrolyte determinations should be performed in patients with low potassium reserves, and the possibility of hypokalemia should be kept in mind with patients who have potentially low potassium reserves and who are receiving cytotoxic therapy or diuretics.
1.2 Nosocomial Pneumonia
Piperacillin and tazobactam for injection is indicated in adults and pediatric patients (2 months of age and older) for the treatment of nosocomial pneumonia (moderate to severe) caused by beta-lactamase producing isolates of Staphylococcus aureus and by piperacillin and tazobactam-susceptible Acinetobacter baumannii, Haemophilus influenzae, Klebsiella pneumoniae, and Pseudomonas aeruginosa (Nosocomial pneumonia caused by P. aeruginosa should be treated in combination with an aminoglycoside) [see Dosage and Administration (2)].
12.1 Mechanism of Action
Piperacillin and tazobactam for injection is an antibacterial drug [see Microbiology (12.4)].
5 Warnings and Precautions
- Serious hypersensitivity reactions (anaphylactic/anaphylactoid) reactions have been reported in patients receiving piperacillin and tazobactam for injection. Discontinue piperacillin and tazobactam for injection if a reaction occurs. (5.1)
- Piperacillin and tazobactam for injection may cause severe cutaneous adverse reactions, such as Stevens-Johnson syndrome, toxic epidermal necrolysis, drug reaction with eosinophilia and systemic symptoms, and acute generalized exanthematous pustulosis. Discontinue piperacillin and tazobactam for injection for progressive rashes. (5.2)
- Hemophagocytic lymphohistiocytosis (HLH) has been reported with the use of piperacillin and tazobactam for injection. If HLH is suspected, discontinue piperacillin and tazobactam for injection immediately. (5.3)
- Rhabdomyolysis: If signs or symptoms of rhabdomyolysis are observed, discontinue Piperacillin and tazobactam for injection and initiate appropriate therapy. (5.4)
- Hematological effects (including bleeding, leukopenia and neutropenia) have occurred. Monitor hematologic tests during prolonged therapy. (5.5)
- As with other penicillins, piperacillin and tazobactam for injection may cause neuromuscular excitability or seizures. Patients receiving higher doses, especially in the presence of renal impairment may be at greater risk. Closely monitor patients with renal impairment or seizure disorders for signs and symptoms of neuromuscular excitability or seizures. (5.6)
- Nephrotoxicity in critically ill patients has been observed; the use of piperacillin and tazobactam for injection was found to be an independent risk factor for renal failure and was associated with delayed recovery of renal function as compared to other beta-lactam antibacterial drugs in a randomized, multicenter, controlled trial in critically ill patients. Based on this study, alternative treatment options should be considered in the critically ill population. If alternative treatment options are inadequate or unavailable, monitor renal function during treatment with piperacillin and tazobactam for injection. (5.7)
- Clostridioides difficile-associated diarrhea: evaluate patients if diarrhea occurs. (5.9)
2 Dosage and Administration
- Adult Patients With Indications Other Than Nosocomial Pneumonia; The usual daily dosage of piperacillin and tazobactam for injection for adults is 3.375 g every six hours totaling 13.5 g (12.0 g piperacillin and 1.5 g tazobactam). (2.1)
- Adult Patients with Nosocomial Pneumonia: Initial presumptive treatment of patients with nosocomial pneumonia should start with piperacillin and tazobactam for injection at a dosage of 4.5 g every six hours plus an aminoglycoside, totaling 18.0 g (16.0 g piperacillin and 2.0 g tazobactam). (2.2)
- Adult Patients with Renal Impairment: Dosage in patients with renal impairment (creatinine clearance ≤40 mL/min) and dialysis patients should be reduced, based on the degree of renal impairment. (2.3)
- Pediatric Patients by Indication and Age: See Table below (2.4)
| Age | Appendicitis and /or Peritonitis | Nosocomial Pneumonia |
|---|---|---|
| 2 months to 9 months | 90 mg/kg (80 mg piperacillin and 10 mg tazobactam) every 8 (eight) hours | 90 mg/kg (80 mg piperacillin and 10 mg tazobactam) every 6 (six) hours |
| Older than 9 months | 112.5 mg/kg (100 mg piperacillin and 12.5 mg tazobactam) every 8 (eight) hours | 112.5 mg/kg (100 mg piperacillin and 12.5 mg tazobactam) every 6 (six) hours |
- Administer piperacillin and tazobactam for injection by intravenous infusion over 30 minutes to both adult and pediatric patients (2.1, 2.2, 2.3, 2.4).
- Piperacillin and tazobactam for injection and aminoglycosides should be reconstituted, diluted, and administered separately. Co-administration via Y-site can be done under certain conditions. (2.6)
- See the full prescribing information for the preparation and administration instructions for piperacillin and tazobactam for injection pharmacy bulk vials.
1.4 Female Pelvic Infections
Piperacillin and tazobactam for injection is indicated in adults for the treatment of postpartum endometritis or pelvic inflammatory disease caused by beta-lactamase producing isolates of Escherichia coli.
3 Dosage Forms and Strengths
Piperacillin and tazobactam for injection, USP is a white to off-white powder in vials :
40.5 g pharmacy bulk vial (piperacillin sodium equivalent to 36 grams of piperacillin and tazobactam sodium equivalent to 4.5 grams tazobactam).
6.2 Postmarketing Experience
In addition to the adverse drug reactions identified in clinical trials in Table 6 and Table 7, the following adverse reactions have been identified during post-approval use of piperacillin and tazobactam for injection. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Hepatobiliary—hepatitis, jaundice
Hematologic—hemolytic anemia, agranulocytosis, pancytopenia
Immune—hypersensitivity reactions, anaphylactic/anaphylactoid reactions (including shock), hemophagocytic lymphohistiocytosis (HLH), acute myocardial ischemia with or without myocardial infarction may occur as part of an allergic reaction
Renal—interstitial nephritis
Nervous system disorders—seizures
Psychiatric disorders—delirium
Respiratory—eosinophilic pneumonia
Skin and Appendages—erythema multiforme, Stevens-Johnson syndrome, toxic epidermal necrolysis, drug reaction with eosinophilia and systemic symptoms, (DRESS), acute generalized exanthematous pustulosis (AGEP), dermatitis exfoliative, and linear IgA bullous dermatosis.
Musculoskeletal—rhabdomyolysis.
Postmarketing experience with piperacillin and tazobactam for injection in pediatric patients suggests a similar safety profile to that seen in adults.
8 Use in Specific Populations
1.1 Intra Abdominal Infections
Piperacillin and tazobactam for injection is indicated in adults and pediatric patients (2 months of age and older) for the treatment of appendicitis (complicated by rupture or abscess) and peritonitis caused by beta-lactamase producing isolates of Escherichia coli or the following members of the Bacteroides fragilis group: B. fragilis, B. ovatus, B. thetaiotaomicron, or B. vulgatus.
5.9 clostridioides Difficile
Clostridioides difficile-associated diarrhea (CDAD) has been reported with use of nearly all antibacterial agents, including piperacillin and tazobactam for injection, and may range in severity from mild diarrhea to fatal colitis. Treatment with antibacterial agents alters the normal flora of the colon leading to overgrowth of C. difficile.
C. difficile produces toxins A and B which contribute to the development of CDAD. Hypertoxin producing strains of C. difficile cause increased morbidity and mortality, as these infections can be refractory to antimicrobial therapy and may require colectomy. CDAD must be considered in all patients who present with diarrhea following antibacterial drug use. Careful medical history is necessary since CDAD has been reported to occur over two months after the administration of antibacterial agents.
If CDAD is suspected or confirmed, ongoing antibacterial drug use not directed against C. difficile may need to be discontinued. Appropriate fluid and electrolyte management, protein supplementation, antibacterial treatment of C. difficile, and surgical evaluation should be instituted as clinically indicated.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
7.7 Effects On Laboratory Tests
There have been reports of positive test results using the Bio-Rad Laboratories Platelia Aspergillus EIA test in patients receiving piperacillin and tazobactam injection who were subsequently found to be free of Aspergillus infection. Cross-reactions with non-Aspergillus polysaccharides and polyfuranoses with the Bio-Rad Laboratories Platelia Aspergillus EIA test have been reported. Therefore, positive test results in patients receiving piperacillin and tazobactam should be interpreted cautiously and confirmed by other diagnostic methods.
As with other penicillins, the administration of piperacillin and tazobactam for injection may result in a false-positive reaction for glucose in the urine using a copper-reduction method (CLINITEST®). It is recommended that glucose tests based on enzymatic glucose oxidase reactions be used.
1.5 Community Acquired Pneumonia
Piperacillin and tazobactam for injection is indicated in adults for the treatment of community-acquired pneumonia (moderate severity only) caused by beta-lactamase producing isolates of Haemophilus influenzae.
5.5 Hematologic Adverse Reactions
Bleeding manifestations have occurred in some patients receiving beta-lactam drugs, including piperacillin. These reactions have sometimes been associated with abnormalities of coagulation tests such as clotting time, platelet aggregation and prothrombin time, and are more likely to occur in patients with renal failure. If bleeding manifestations occur, piperacillin and tazobactam for injection should be discontinued and appropriate therapy instituted.
The leukopenia/neutropenia associated with piperacillin and tazobactam for injection administration appears to be reversible and most frequently associated with prolonged administration.
Periodic assessment of hematopoietic function should be performed, especially with prolonged therapy, i.e., ≥ 21 days [see Adverse Reactions (6.1)].
8.8 Patients With Cystic Fibrosis
As with other semisynthetic penicillins, piperacillin therapy has been associated with an increased incidence of fever and rash in cystic fibrosis patients.
16 How Supplied/storage and Handling
Piperacillin and tazobactam for injection, USP is supplied as a white to off-white sterile, cryodesiccated powder in the pharmacy bulk vial as follows:
Each piperacillin and tazobactam for injection, USP 40.5 g pharmacy bulk vial provides piperacillin sodium equivalent to 36 grams of piperacillin and tazobactam sodium equivalent to 4.5 grams of tazobactam. Each pharmacy bulk vial contains 84.5 mEq (1,944 mg) of sodium. NDC 60505-6262-0
Piperacillin and tazobactam for injection, USP pharmacy bulk vials should be stored at 20° to 25°C (68° to 77°F) [See USP Controlled Room Temperature] prior to reconstitution.
1.3 Skin and Skin Structure Infections
Piperacillin and tazobactam for injection is indicated in adults for the treatment of uncomplicated and complicated skin and skin structure infections, including cellulitis, cutaneous abscesses and ischemic/diabetic foot infections caused by beta-lactamase producing isolates of Staphylococcus aureus.
2.6 Compatibility With Aminoglycosides
Due to the in vitro inactivation of aminoglycosides by piperacillin, piperacillin and tazobactam for injection and aminoglycosides are recommended for separate administration. Piperacillin and tazobactam for injection and aminoglycosides should be reconstituted, diluted, and administered separately when concomitant therapy with aminoglycosides is indicated [see Drug Interactions (7.1)].
In circumstances where co-administration via Y-site is necessary, piperacillin and tazobactam for injection is compatible for simultaneous co-administration via Y-site infusion only with the following aminoglycosides under the following conditions:
| Aminoglycoside | Piperacillin and Tazobactam for Injection Dose (grams) |
Piperacillin and Tazobactam for Injection Diluent Volume Diluent volumes apply only to bulk pharmacy containers
(mL) |
Aminoglycoside Concentration Range The concentration ranges in Table 5 are based on administration of the aminoglycoside in divided doses (10–15 mg/kg/day in two daily doses for amikacin and 3–5 mg/kg/day in three daily doses for gentamicin). Administration of amikacin or gentamicin in a single daily dose or in doses exceeding those stated above via Y-site with piperacillin and tazobactam for injection has not been evaluated. See package insert for each aminoglycoside for complete Dosage and Administration instructions.
(mg/mL) |
Acceptable Diluents |
|---|---|---|---|---|
| Amikacin | 2.25 3.375 4.5 |
50 100 150 |
1.75 – 7.5 | 0.9% sodium chloride or 5% dextrose |
| Gentamicin | 2.25 3.375 4.5 |
50 100 150 |
0.7 – 3.32 | 0.9% sodium chloride or 5% dextrose |
Only the concentration and diluents for amikacin or gentamicin with the dosages of piperacillin and tazobactam for injection listed above have been established as compatible for co-administration via Y-site infusion. Simultaneous co-administration via Y-site infusion in any manner other than listed above may result in inactivation of the aminoglycoside by piperacillin and tazobactam for injection.
Piperacillin and tazobactam for injection is not compatible with tobramycin for simultaneous co-administration via Y-site infusion. Compatibility of piperacillin and tazobactam for injection with other aminoglycosides has not been established.
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit.
5.1 Hypersensitivity Adverse Reactions
Serious and occasionally fatal hypersensitivity (anaphylactic/anaphylactoid) reactions (including shock) have been reported in patients receiving therapy with piperacillin and tazobactam for injection. These reactions are more likely to occur in individuals with a history of penicillin, cephalosporin, or carbapenem hypersensitivity or a history of sensitivity to multiple allergens. Before initiating therapy with piperacillin and tazobactam for injection, careful inquiry should be made concerning previous hypersensitivity reactions. If an allergic reaction occurs, piperacillin and tazobactam for injection should be discontinued and appropriate therapy instituted.
5.2 Severe Cutaneous Adverse Reactions
Piperacillin and tazobactam for injection may cause severe cutaneous adverse reactions, such as Stevens-Johnson syndrome, toxic epidermal necrolysis, drug reaction with eosinophilia and systemic symptoms, and acute generalized exanthematous pustulosis. If patients develop a skin rash they should be monitored closely and piperacillin and tazobactam for injection discontinued if lesions progress.
5.3 Hemophagocytic Lymphohistiocytosis
Cases of hemophagocytic lymphohistiocytosis (HLH) have been reported in pediatric and adult patients treated with piperacillin and tazobactam for injection. Signs and symptoms of HLH may include fever, rash, lymphadenopathy, hepatosplenomegaly and cytopenia. If HLH is suspected, discontinue piperacillin and tazobactam for injection immediately and institute appropriate management.
5.10 Development of Drug Resistant Bacteria
Prescribing piperacillin and tazobactam for injection in the absence of a proven or strongly suspected bacterial infection or a prophylactic indication is unlikely to provide benefit to the patient and increases the risk of development of drug-resistant bacteria.
6.3 Additional Experience With Piperacillin
The following adverse reaction has also been reported for piperacillin for injection:
Skeletal—prolonged neuromuscular blockade [see Drug Interactions (7.5)].
Principal Display Panel – 40.5 Grams Vial
NDC 60505-6262-0 Rx only
Piperacillin and Tazobactam for Injection, USP
40.5 gram vial
PHARMACY BULK PACKAGE
Not for Direct Infusion
RECONSTITUTED STOCK SOLUTION MUST
BE TRANSFERRED AND FURTHER
DILUTED FOR INTRAVENOUS INFUSION
5.6 Central Nervous System Adverse Reactions
As with other penicillins, piperacillin and tazobactam for injection may cause neuromuscular excitability or seizures. Patients receiving higher doses, especially patients with renal impairment may be at greater risk for central nervous system adverse reactions. Closely monitor patients with renal impairment or seizure disorders for signs and symptoms of neuromuscular excitability or seizures [see Adverse Reactions (6.2)].
5.7 Nephrotoxicity in Critically Ill Patients
The use of piperacillin and tazobactam for injection was found to be an independent risk factor for renal failure and was associated with delayed recovery of renal function as compared to other beta-lactam antibacterial drugs in a randomized, multicenter, controlled trial in critically ill patients [see Adverse Reactions (6.1)]. Based on this study, alternative treatment options should be considered in the critically ill population. If alternative treatment options are inadequate or unavailable, monitor renal function during treatment with piperacillin and tazobactam for injection [see Dosage and Administration (2.3)].
Combined use of piperacillin and tazobactam and vancomycin may be associated with an increased incidence of acute kidney injury [see Drug Interactions (7.3)].
Principal Display Panel – 40.5 Grams Carton
NDC 60505-6262-0
Rx only
Piperacillin and Tazobactam for Injection, USP
40.5 gram vial
PHARMACY BULK PACKAGE
Not for Direct Infusion
RECONSTITUTED STOCK SOLUTION MUST
BE TRANSFERRED AND FURTHER
DILUTED FOR INTRAVENOUS INFUSION
2.3 Dosage in Adult Patients With Renal Impairment
In adult patients with renal impairment (creatinine clearance ≤ 40 mL/min) and dialysis patients (hemodialysis and CAPD), the intravenous dose of piperacillin and tazobactam for injection should be reduced based on the degree of renal impairment. The recommended daily dosage of piperacillin and tazobactam for injection for patients with renal impairment administered by intravenous infusion over 30 minutes is described in Table 1.
| Creatinine clearance, mL/min | All Indications (except nosocomial pneumonia) | Nosocomial Pneumonia |
|---|---|---|
| Greater than 40 mL/min | 3.375 every 6 hours | 4.5 every 6 hours |
| 20 to 40 mL/min Creatinine clearance for patients not receiving hemodialysis
|
2.25 every 6 hours | 3.375 every 6 hours |
| Less than 20 mL/min | 2.25 every 8 hours | 2.25 every 6 hours |
| Hemodialysis 0.75 g (0.67 g piperacillin and 0.08 g tazobactam) should be administered following each hemodialysis session on hemodialysis days
|
2.25 every 12 hours | 2.25 every 8 hours |
| CAPD | 2.25 every 12 hours | 2.25 every 8 hours |
For patients on hemodialysis, the maximum dose is 2.25 g every twelve hours for all indications other than nosocomial pneumonia and 2.25 g every eight hours for nosocomial pneumonia. Since hemodialysis removes 30% to 40% of the administered dose, an additional dose of 0.75 g piperacillin and tazobactam for injection (0.67 g piperacillin and 0.08 g tazobactam) should be administered following each dialysis period on hemodialysis days. No additional dosage of piperacillin and tazobactam for injection is necessary for CAPD patients.
2.2 Dosage in Adult Patients With Nosocomial Pneumonia
Initial presumptive treatment of adult patients with nosocomial pneumonia should start with piperacillin and tazobactam for injection at a dosage of 4.5 g every six hours plus an aminoglycoside, [totaling 18.0 g (16.0 g piperacillin and 2.0 g tazobactam)], administered by intravenous infusion over 30 minutes. The recommended duration of piperacillin and tazobactam for injection treatment for nosocomial pneumonia is 7 to 14 days. Treatment with the aminoglycoside should be continued in patients from whom P. aeruginosa is isolated.
2.1 Dosage in Adult Patients With Indications Other Than Nosocomial Pneumonia
The usual total daily dosage of piperacillin and tazobactam for injection for adult patients with indications other than nosocomial pneumonia is 3.375 g every six hours [totaling 13.5 g (12.0 g piperacillin and 1.5 g tazobactam)], to be administered by intravenous infusion over 30 minutes. The usual duration of piperacillin and tazobactam for injection treatment is from 7 to 10 days.
2.4 Dosage in Pediatric Patients With Appendicitis/peritonitis Or Nosocomial Pneumonia
The recommended dosage for pediatric patients with appendicitis and/or peritonitis or nosocomial pneumonia aged 2 months of age and older, weighing up to 40 kg, and with normal renal function, is described in Table 2 [see Use in Specific Populations (8.4) and Clinical Pharmacology (12.3)].
| Age | Appendicitis and/or Peritonitis | Nosocomial Pneumonia |
|---|---|---|
| 2 months to 9 months | 90 mg/kg (80 mg piperacillin and 10 mg tazobactam) every 8 (eight) hours |
90 mg/kg (80 mg piperacillin and 10 mg tazobactam) every 6 (six) hours |
| Older than 9 months of age | 112.5 mg/kg (100 mg piperacillin and 12.5 mg tazobactam) every 8 (eight) hours |
112.5 mg/kg (100 mg piperacillin and 12.5 mg tazobactam) every 6 (six) hours |
Pediatric patients weighing over 40 kg and with normal renal function should receive the adult dose [see Dosage and Administration (2.1, 2.2)].
Dosage of piperacillin and tazobactam for injection in pediatric patients with renal impairment has not been determined.
Structured Label Content
Section 42229-5 (42229-5)
Reconstitution of Piperacillin and Tazobactam for Injection for Adult Patients and Pediatric Patients Weighing Over 40 kg
Section 43679-0 (43679-0)
Mechanism of Action
Piperacillin sodium exerts bactericidal activity by inhibiting septum formation and cell wall synthesis of susceptible bacteria. In vitro, piperacillin is active against a variety of gram-positive and gram-negative aerobic and anaerobic bacteria. Tazobactam sodium has little clinically relevant in vitro activity against bacteria due to its reduced affinity to penicillin-binding proteins. It is, however, a beta-lactamase inhibitor of the Molecular class A enzymes, including Richmond-Sykes class III (Bush class 2b & 2b') penicillinases and cephalosporinases. It varies in its ability to inhibit class II and IV (2a & 4) penicillinases. Tazobactam does not induce chromosomally-mediated beta-lactamases at tazobactam concentrations achieved with the recommended dosage regimen.
Section 43683-2 (43683-2)
| Warnings and Precautions, Rhabdomyolysis (5.4) | 08/2024 |
1.6 Usage
To reduce the development of drug-resistant bacteria and maintain the effectiveness of piperacillin and tazobactam for injection and other antibacterial drugs, piperacillin and tazobactam for injection should be used only to treat or prevent infections that are proven or strongly suspected to be caused by bacteria. When culture and susceptibility information are available, they should be considered in selecting or modifying antibacterial therapy. In the absence of such data, local epidemiology and susceptibility patterns may contribute to the empiric selection of therapy.
10 Overdosage (10 OVERDOSAGE)
There have been postmarketing reports of overdose with piperacillin and tazobactam. The majority of those events experienced, including nausea, vomiting, and diarrhea, have also been reported with the usual recommended dosages. Patients may experience neuromuscular excitability or seizures if higher than recommended doses are given intravenously (particularly in the presence of renal failure) [see Warnings and Precautions (5.6)].
Treatment should be supportive and symptomatic according to the patient's clinical presentation. Excessive serum concentrations of either piperacillin or tazobactam may be reduced by hemodialysis. Following a single 3.375 g dose of piperacillin and tazobactam, the percentage of the piperacillin and tazobactam dose removed by hemodialysis was approximately 31% and 39%, respectively [see Clinical Pharmacology (12)].
15 References (15 REFERENCES)
- Jensen J-US, Hein L, Lundgren B, et al. BMJ Open 2012; 2:e000635. doi:10.1136.
11 Description (11 DESCRIPTION)
Piperacillin and tazobactam for injection, USP is an injectable antibacterial combination product consisting of the semisynthetic antibacterial piperacillin sodium and the beta-lactamase inhibitor tazobactam sodium for intravenous administration.
Piperacillin sodium is derived from D(-)-α-aminobenzyl-penicillin. The chemical name of piperacillin sodium is sodium (2S,5R,6R)-6-[(R)-2-(4-ethyl-2,3-dioxo-1-piperazine-carboxamido)-2-phenylacetamido]-3,3-dimethyl-7-oxo-4-thia-1-azabicyclo[3.2.0]heptane-2-carboxylate. The chemical formula is C23H26N5NaO7S and the molecular weight is 539.5. The chemical structure of piperacillin sodium is:
Tazobactam sodium, a derivative of the penicillin nucleus, is a penicillanic acid sulfone. Its chemical name is sodium (2S,3S,5R)-3-methyl-7-oxo-3-(1H-1,2,3-triazol-1-ylmethyl)-4-thia-1-azabicyclo[3.2.0]heptane-2-carboxylate-4,4-dioxide. The chemical formula is C10H11N4NaO5S and the molecular weight is 322.3. The chemical structure of tazobactam sodium is:
Piperacillin and tazobactam for injection contains a total of 2.35 mEq (54 mg) of sodium (Na+) per gram of piperacillin in the combination product.
Piperacillin and tazobactam for injection, USP is a white to off-white sterile, cryodesiccated powder consisting of piperacillin and tazobactam as their sodium salts packaged in glass vials. The product does not contain excipients or preservatives.
Each piperacillin and tazobactam for injection, USP 40.5 g pharmacy bulk vial contains piperacillin sodium equivalent to 36 grams of piperacillin and tazobactam sodium equivalent to 4.5 g of tazobactam sufficient for delivery of multiple doses.
7.2 Probenecid
Probenecid administered concomitantly with piperacillin and tazobactam for injection prolongs the half-life of piperacillin by 21% and that of tazobactam by 71% because probenecid inhibits tubular renal secretion of both piperacillin and tazobactam. Probenecid should not be co-administered with piperacillin and tazobactam for injection unless the benefit outweighs the risk.
7.3 Vancomycin
Studies have detected an increased incidence of acute kidney injury in patients concomitantly administered piperacillin and tazobactam and vancomycin as compared to vancomycin alone [see Warnings and Precautions (5.7)].
Monitor kidney function in patients concomitantly administered with piperacillin and tazobactam and vancomycin.
No pharmacokinetic interactions have been noted between piperacillin and tazobactam and vancomycin.
7.5 Vecuronium
Piperacillin when used concomitantly with vecuronium has been implicated in the prolongation of the neuromuscular blockade of vecuronium. piperacillin and tazobactam for injection could produce the same phenomenon if given along with vecuronium. Due to their similar mechanism of action, it is expected that the neuromuscular blockade produced by any of the non-depolarizing neuromuscular blockers could be prolonged in the presence of piperacillin. Monitor for adverse reactions related to neuromuscular blockade (see package insert for vecuronium bromide).
7.6 Methotrexate
Limited data suggests that co-administration of methotrexate and piperacillin may reduce the clearance of methotrexate due to competition for renal secretion. The impact of tazobactam on the elimination of methotrexate has not been evaluated. If concurrent therapy is necessary, serum concentrations of methotrexate as well as the signs and symptoms of methotrexate toxicity should be frequently monitored.
8.4 Pediatric Use
The safety and effectiveness of piperacillin and tazobactam for injection for intra-abdominal infections, and nosocomial pneumonia have been established in pediatric patients 2 months of age and older.
Use of piperacillin and tazobactam for injection in pediatric patients 2 months of age and older with intra-abdominal infections including appendicitis and/or peritonitis is supported by evidence from well-controlled studies and pharmacokinetic studies in adults and in pediatric patients. This includes a prospective, randomized, comparative, open-label clinical trial with 542 pediatric patients 2 to 12 years of age with intra-abdominal infections (including appendicitis and/or peritonitis), in which 273 pediatric patients received piperacillin and tazobactam [see Adverse Reactions (6.1) and Clinical Pharmacology (12.3)].
Use of piperacillin and tazobactam for injection in pediatric patients 2 months of age and older with nosocomial pneumonia is supported by evidence from well-controlled studies in adults with nosocomial pneumonia, a simulation study performed with a population pharmacokinetic model, and a retrospective, cohort study of pediatric patients with nosocomial pneumonia in which 140 pediatric patients were treated with piperacillin and tazobactam for injection and 267 patients treated with comparators (which included ticarcillin-clavulanate, carbapenems, ceftazidime, cefepime, or ciprofloxacin) [see Adverse Reactions (6.1) and Clinical Pharmacology (12.3)].
The safety and effectiveness of piperacillin and tazobactam for injection have not been established in pediatric patients less than 2 months of age [see Clinical Pharmacology (12) and Dosage and Administration (2)].
Dosage of piperacillin and tazobactam for injection in pediatric patients with renal impairment has not been determined.
8.5 Geriatric Use
Patients over 65 years are not at an increased risk of developing adverse effects solely because of age. However, dosage should be adjusted in the presence of renal impairment [see Dosage and Administration (2)].
In general, dose selection for an elderly patient should be cautious, usually starting at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy.
Piperacillin and tazobactam for injection contains 54 mg (2.35 mEq) of sodium per gram of piperacillin in the combination product. At the usual recommended doses, patients would receive between 648 and 864 mg/day (28.2 and 37.6 mEq) of sodium. The geriatric population may respond with a blunted natriuresis to salt loading. This may be clinically important with regard to such diseases as congestive heart failure.
This drug is known to be substantially excreted by the kidney, and the risk of toxic reactions to this drug may be greater in patients with impaired renal function. Because elderly patients are more likely to have decreased renal function, care should be taken in dose selection, and it may be useful to monitor renal function.
5.4 Rhabdomyolysis
Rhabdomyolysis has been reported with the use of Piperacillin and tazobactam for injection [see Adverse Reactions (6.2)] . If signs or symptoms of rhabdomyolysis such as muscle pain, tenderness or weakness, dark urine, or elevated creatine phosphokinase are observed, discontinue Piperacillin and tazobactam for injection and initiate appropriate therapy.
7.4 Anticoagulants
Coagulation parameters should be tested more frequently and monitored regularly during simultaneous administration of high doses of heparin, oral anticoagulants, or other drugs that may affect the blood coagulation system or the thrombocyte function [see Warnings and Precautions (5.5)].
4 Contraindications (4 CONTRAINDICATIONS)
Piperacillin and tazobactam for injection is contraindicated in patients with a history of allergic reactions to any of the penicillins, cephalosporins, or beta-lactamase inhibitors.
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following clinically significant adverse reactions are described elsewhere in the labeling:
- Hypersensitivity Adverse Reactions [see Warnings and Precautions (5.1)]
- Severe Cutaneous Adverse Reactions [see Warnings and Precautions (5.2)]
- Hemophagocytic Lymphohistiocytosis [see Warnings and Precautions (5.3)]
- Rhabdomyolysis [see Warnings and Precautions (5.4)]
- Hematologic Adverse Reactions [see Warnings and Precautions (5.5)]
- Central Nervous System Adverse Reactions [see Warnings and Precautions (5.6)]
- Nephrotoxicity in Critically Ill Patients [see Warnings and Precautions (5.7)]
- Clostridioides difficile-Associated Diarrhea [see Warnings and Precautions (5.9)]
7 Drug Interactions (7 DRUG INTERACTIONS)
- Piperacillin and tazobactam for injection administration can significantly reduce tobramycin concentrations in hemodialysis patients. Monitor tobramycin concentrations in these patients. (7.1)
- Probenecid prolongs the half-lives of piperacillin and tazobactam and should not be co-administered with piperacillin and tazobactam for injection unless the benefit outweighs the risk. (7.2)
- Co-administration of piperacillin and tazobactam for injection with vancomycin may increase the incidence of acute kidney injury. Monitor kidney function in patients receiving piperacillin and tazobactam for injection and vancomycin. (7.3)
- Monitor coagulation parameters in patients receiving piperacillin and tazobactam for injection and heparin or oral anticoagulants. (7.4)
- Piperacillin and tazobactam for injection may prolong the neuromuscular blockade of vecuronium and other non-depolarizing neuromuscular blockers. Monitor for adverse reactions related to neuromuscular blockade. (7.5)
7.1 Aminoglycosides
Piperacillin may inactivate aminoglycosides by converting them to microbiologically inert amides.
8.6 Renal Impairment
In patients with creatinine clearance ≤ 40 mL/min and dialysis patients (hemodialysis and CAPD), the intravenous dose of piperacillin and tazobactam for injection should be reduced to the degree of renal function impairment [see Dosage and Administration (2)].
12.2 Pharmacodynamics
The pharmacodynamic parameter for piperacillin and tazobactam that is most predictive of clinical and microbiological efficacy is time above MIC.
12.3 Pharmacokinetics
The mean and coefficients of variation (CV%) for the pharmacokinetic parameters of piperacillin and tazobactam after multiple intravenous doses are summarized in Table 8.
| Cmax : maximum observed concentration, AUC: Area under the curve, CL=clearance, CLR= Renal clearance V=volume of distribution, T1/2 = elimination half-life |
||||||
| Piperacillin | ||||||
| Piperacillin and Tazobactam Dose Piperacillin and tazobactam were given in combination, infused over 30 minutes.
|
Cmax (mcg/mL) | AUC Numbers in [] parentheses are coefficients of variation [CV%]. (mcg∙h/mL) |
CL (mL/min) | V (L) |
T1/2
(h) |
CLR (mL/min) |
| 2.25 g | 134 | 131 [14] | 257 | 17.4 | 0.79 | -- |
| 3.375 g | 242 | 242 [10] | 207 | 15.1 | 0.84 | 140 |
| 4.5 g | 298 | 322 [16] | 210 | 15.4 | 0.84 | -- |
| Tazobactam | ||||||
| Piperacillin and Tazobactam Dose | Cmax (mcg/mL) | AUC (mcg∙h/mL) | CL (mL/min) | V (L) |
T1/2 (h) | CLR (mL/min) |
| 2.25 g | 15 | 16.0 [21] | 258 | 17.0 | 0.77 | -- |
| 3.375 g | 24 | 25.0 [8] | 251 | 14.8 | 0.68 | 166 |
| 4.5 g | 34 | 39.8 [15] | 206 | 14.7 | 0.82 | -- |
Peak plasma concentrations of piperacillin and tazobactam are attained immediately after completion of an intravenous infusion of piperacillin and tazobactam for injection. Piperacillin plasma concentrations, following a 30-minute infusion of piperacillin and tazobactam for injection, were similar to those attained when equivalent doses of piperacillin were administered alone. Steady-state plasma concentrations of piperacillin and tazobactam were similar to those attained after the first dose due to the short half-lives of piperacillin and tazobactam.
8.7 Hepatic Impairment
Dosage adjustment of piperacillin and tazobactam for injection is not warranted in patients with hepatic cirrhosis [see Clinical Pharmacology (12.3)].
1 Indications and Usage (1 INDICATIONS AND USAGE)
Piperacillin and tazobactam for injection is a combination of piperacillin, a penicillin-class antibacterial and tazobactam, a beta-lactamase inhibitor, indicated for the treatment of:
- Intra-abdominal infections in adult and pediatric patients 2 months of age and older (1.1)
- Nosocomial pneumonia in adult and pediatric patients 2 months of age and older (1.2)
- Skin and skin structure infections in adults (1.3)
- Female pelvic infections in adults (1.4)
- Community-acquired pneumonia in adults (1.5)
To reduce the development of drug-resistant bacteria and maintain the effectiveness of piperacillin and tazobactam for injection and other antibacterial drugs, piperacillin and tazobactam for injection should be used only to treat or prevent infections that are proven or strongly suspected to be caused by bacteria. (1.6)
5.8 Electrolyte Effects
Piperacillin and tazobactam for injection contains a total of 2.35 mEq (54 mg) of Na+ (sodium) per gram of piperacillin in the combination product. This should be considered when treating patients requiring restricted salt intake. Periodic electrolyte determinations should be performed in patients with low potassium reserves, and the possibility of hypokalemia should be kept in mind with patients who have potentially low potassium reserves and who are receiving cytotoxic therapy or diuretics.
1.2 Nosocomial Pneumonia
Piperacillin and tazobactam for injection is indicated in adults and pediatric patients (2 months of age and older) for the treatment of nosocomial pneumonia (moderate to severe) caused by beta-lactamase producing isolates of Staphylococcus aureus and by piperacillin and tazobactam-susceptible Acinetobacter baumannii, Haemophilus influenzae, Klebsiella pneumoniae, and Pseudomonas aeruginosa (Nosocomial pneumonia caused by P. aeruginosa should be treated in combination with an aminoglycoside) [see Dosage and Administration (2)].
12.1 Mechanism of Action
Piperacillin and tazobactam for injection is an antibacterial drug [see Microbiology (12.4)].
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Serious hypersensitivity reactions (anaphylactic/anaphylactoid) reactions have been reported in patients receiving piperacillin and tazobactam for injection. Discontinue piperacillin and tazobactam for injection if a reaction occurs. (5.1)
- Piperacillin and tazobactam for injection may cause severe cutaneous adverse reactions, such as Stevens-Johnson syndrome, toxic epidermal necrolysis, drug reaction with eosinophilia and systemic symptoms, and acute generalized exanthematous pustulosis. Discontinue piperacillin and tazobactam for injection for progressive rashes. (5.2)
- Hemophagocytic lymphohistiocytosis (HLH) has been reported with the use of piperacillin and tazobactam for injection. If HLH is suspected, discontinue piperacillin and tazobactam for injection immediately. (5.3)
- Rhabdomyolysis: If signs or symptoms of rhabdomyolysis are observed, discontinue Piperacillin and tazobactam for injection and initiate appropriate therapy. (5.4)
- Hematological effects (including bleeding, leukopenia and neutropenia) have occurred. Monitor hematologic tests during prolonged therapy. (5.5)
- As with other penicillins, piperacillin and tazobactam for injection may cause neuromuscular excitability or seizures. Patients receiving higher doses, especially in the presence of renal impairment may be at greater risk. Closely monitor patients with renal impairment or seizure disorders for signs and symptoms of neuromuscular excitability or seizures. (5.6)
- Nephrotoxicity in critically ill patients has been observed; the use of piperacillin and tazobactam for injection was found to be an independent risk factor for renal failure and was associated with delayed recovery of renal function as compared to other beta-lactam antibacterial drugs in a randomized, multicenter, controlled trial in critically ill patients. Based on this study, alternative treatment options should be considered in the critically ill population. If alternative treatment options are inadequate or unavailable, monitor renal function during treatment with piperacillin and tazobactam for injection. (5.7)
- Clostridioides difficile-associated diarrhea: evaluate patients if diarrhea occurs. (5.9)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
- Adult Patients With Indications Other Than Nosocomial Pneumonia; The usual daily dosage of piperacillin and tazobactam for injection for adults is 3.375 g every six hours totaling 13.5 g (12.0 g piperacillin and 1.5 g tazobactam). (2.1)
- Adult Patients with Nosocomial Pneumonia: Initial presumptive treatment of patients with nosocomial pneumonia should start with piperacillin and tazobactam for injection at a dosage of 4.5 g every six hours plus an aminoglycoside, totaling 18.0 g (16.0 g piperacillin and 2.0 g tazobactam). (2.2)
- Adult Patients with Renal Impairment: Dosage in patients with renal impairment (creatinine clearance ≤40 mL/min) and dialysis patients should be reduced, based on the degree of renal impairment. (2.3)
- Pediatric Patients by Indication and Age: See Table below (2.4)
| Age | Appendicitis and /or Peritonitis | Nosocomial Pneumonia |
|---|---|---|
| 2 months to 9 months | 90 mg/kg (80 mg piperacillin and 10 mg tazobactam) every 8 (eight) hours | 90 mg/kg (80 mg piperacillin and 10 mg tazobactam) every 6 (six) hours |
| Older than 9 months | 112.5 mg/kg (100 mg piperacillin and 12.5 mg tazobactam) every 8 (eight) hours | 112.5 mg/kg (100 mg piperacillin and 12.5 mg tazobactam) every 6 (six) hours |
- Administer piperacillin and tazobactam for injection by intravenous infusion over 30 minutes to both adult and pediatric patients (2.1, 2.2, 2.3, 2.4).
- Piperacillin and tazobactam for injection and aminoglycosides should be reconstituted, diluted, and administered separately. Co-administration via Y-site can be done under certain conditions. (2.6)
- See the full prescribing information for the preparation and administration instructions for piperacillin and tazobactam for injection pharmacy bulk vials.
1.4 Female Pelvic Infections
Piperacillin and tazobactam for injection is indicated in adults for the treatment of postpartum endometritis or pelvic inflammatory disease caused by beta-lactamase producing isolates of Escherichia coli.
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Piperacillin and tazobactam for injection, USP is a white to off-white powder in vials :
40.5 g pharmacy bulk vial (piperacillin sodium equivalent to 36 grams of piperacillin and tazobactam sodium equivalent to 4.5 grams tazobactam).
6.2 Postmarketing Experience
In addition to the adverse drug reactions identified in clinical trials in Table 6 and Table 7, the following adverse reactions have been identified during post-approval use of piperacillin and tazobactam for injection. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Hepatobiliary—hepatitis, jaundice
Hematologic—hemolytic anemia, agranulocytosis, pancytopenia
Immune—hypersensitivity reactions, anaphylactic/anaphylactoid reactions (including shock), hemophagocytic lymphohistiocytosis (HLH), acute myocardial ischemia with or without myocardial infarction may occur as part of an allergic reaction
Renal—interstitial nephritis
Nervous system disorders—seizures
Psychiatric disorders—delirium
Respiratory—eosinophilic pneumonia
Skin and Appendages—erythema multiforme, Stevens-Johnson syndrome, toxic epidermal necrolysis, drug reaction with eosinophilia and systemic symptoms, (DRESS), acute generalized exanthematous pustulosis (AGEP), dermatitis exfoliative, and linear IgA bullous dermatosis.
Musculoskeletal—rhabdomyolysis.
Postmarketing experience with piperacillin and tazobactam for injection in pediatric patients suggests a similar safety profile to that seen in adults.
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
1.1 Intra Abdominal Infections (1.1 Intra-abdominal Infections)
Piperacillin and tazobactam for injection is indicated in adults and pediatric patients (2 months of age and older) for the treatment of appendicitis (complicated by rupture or abscess) and peritonitis caused by beta-lactamase producing isolates of Escherichia coli or the following members of the Bacteroides fragilis group: B. fragilis, B. ovatus, B. thetaiotaomicron, or B. vulgatus.
5.9 clostridioides Difficile (5.9 Clostridioides difficile-)
Clostridioides difficile-associated diarrhea (CDAD) has been reported with use of nearly all antibacterial agents, including piperacillin and tazobactam for injection, and may range in severity from mild diarrhea to fatal colitis. Treatment with antibacterial agents alters the normal flora of the colon leading to overgrowth of C. difficile.
C. difficile produces toxins A and B which contribute to the development of CDAD. Hypertoxin producing strains of C. difficile cause increased morbidity and mortality, as these infections can be refractory to antimicrobial therapy and may require colectomy. CDAD must be considered in all patients who present with diarrhea following antibacterial drug use. Careful medical history is necessary since CDAD has been reported to occur over two months after the administration of antibacterial agents.
If CDAD is suspected or confirmed, ongoing antibacterial drug use not directed against C. difficile may need to be discontinued. Appropriate fluid and electrolyte management, protein supplementation, antibacterial treatment of C. difficile, and surgical evaluation should be instituted as clinically indicated.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
7.7 Effects On Laboratory Tests (7.7 Effects on Laboratory Tests)
There have been reports of positive test results using the Bio-Rad Laboratories Platelia Aspergillus EIA test in patients receiving piperacillin and tazobactam injection who were subsequently found to be free of Aspergillus infection. Cross-reactions with non-Aspergillus polysaccharides and polyfuranoses with the Bio-Rad Laboratories Platelia Aspergillus EIA test have been reported. Therefore, positive test results in patients receiving piperacillin and tazobactam should be interpreted cautiously and confirmed by other diagnostic methods.
As with other penicillins, the administration of piperacillin and tazobactam for injection may result in a false-positive reaction for glucose in the urine using a copper-reduction method (CLINITEST®). It is recommended that glucose tests based on enzymatic glucose oxidase reactions be used.
1.5 Community Acquired Pneumonia (1.5 Community-acquired Pneumonia)
Piperacillin and tazobactam for injection is indicated in adults for the treatment of community-acquired pneumonia (moderate severity only) caused by beta-lactamase producing isolates of Haemophilus influenzae.
5.5 Hematologic Adverse Reactions
Bleeding manifestations have occurred in some patients receiving beta-lactam drugs, including piperacillin. These reactions have sometimes been associated with abnormalities of coagulation tests such as clotting time, platelet aggregation and prothrombin time, and are more likely to occur in patients with renal failure. If bleeding manifestations occur, piperacillin and tazobactam for injection should be discontinued and appropriate therapy instituted.
The leukopenia/neutropenia associated with piperacillin and tazobactam for injection administration appears to be reversible and most frequently associated with prolonged administration.
Periodic assessment of hematopoietic function should be performed, especially with prolonged therapy, i.e., ≥ 21 days [see Adverse Reactions (6.1)].
8.8 Patients With Cystic Fibrosis (8.8 Patients with Cystic Fibrosis)
As with other semisynthetic penicillins, piperacillin therapy has been associated with an increased incidence of fever and rash in cystic fibrosis patients.
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
Piperacillin and tazobactam for injection, USP is supplied as a white to off-white sterile, cryodesiccated powder in the pharmacy bulk vial as follows:
Each piperacillin and tazobactam for injection, USP 40.5 g pharmacy bulk vial provides piperacillin sodium equivalent to 36 grams of piperacillin and tazobactam sodium equivalent to 4.5 grams of tazobactam. Each pharmacy bulk vial contains 84.5 mEq (1,944 mg) of sodium. NDC 60505-6262-0
Piperacillin and tazobactam for injection, USP pharmacy bulk vials should be stored at 20° to 25°C (68° to 77°F) [See USP Controlled Room Temperature] prior to reconstitution.
1.3 Skin and Skin Structure Infections
Piperacillin and tazobactam for injection is indicated in adults for the treatment of uncomplicated and complicated skin and skin structure infections, including cellulitis, cutaneous abscesses and ischemic/diabetic foot infections caused by beta-lactamase producing isolates of Staphylococcus aureus.
2.6 Compatibility With Aminoglycosides
Due to the in vitro inactivation of aminoglycosides by piperacillin, piperacillin and tazobactam for injection and aminoglycosides are recommended for separate administration. Piperacillin and tazobactam for injection and aminoglycosides should be reconstituted, diluted, and administered separately when concomitant therapy with aminoglycosides is indicated [see Drug Interactions (7.1)].
In circumstances where co-administration via Y-site is necessary, piperacillin and tazobactam for injection is compatible for simultaneous co-administration via Y-site infusion only with the following aminoglycosides under the following conditions:
| Aminoglycoside | Piperacillin and Tazobactam for Injection Dose (grams) |
Piperacillin and Tazobactam for Injection Diluent Volume Diluent volumes apply only to bulk pharmacy containers
(mL) |
Aminoglycoside Concentration Range The concentration ranges in Table 5 are based on administration of the aminoglycoside in divided doses (10–15 mg/kg/day in two daily doses for amikacin and 3–5 mg/kg/day in three daily doses for gentamicin). Administration of amikacin or gentamicin in a single daily dose or in doses exceeding those stated above via Y-site with piperacillin and tazobactam for injection has not been evaluated. See package insert for each aminoglycoside for complete Dosage and Administration instructions.
(mg/mL) |
Acceptable Diluents |
|---|---|---|---|---|
| Amikacin | 2.25 3.375 4.5 |
50 100 150 |
1.75 – 7.5 | 0.9% sodium chloride or 5% dextrose |
| Gentamicin | 2.25 3.375 4.5 |
50 100 150 |
0.7 – 3.32 | 0.9% sodium chloride or 5% dextrose |
Only the concentration and diluents for amikacin or gentamicin with the dosages of piperacillin and tazobactam for injection listed above have been established as compatible for co-administration via Y-site infusion. Simultaneous co-administration via Y-site infusion in any manner other than listed above may result in inactivation of the aminoglycoside by piperacillin and tazobactam for injection.
Piperacillin and tazobactam for injection is not compatible with tobramycin for simultaneous co-administration via Y-site infusion. Compatibility of piperacillin and tazobactam for injection with other aminoglycosides has not been established.
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit.
5.1 Hypersensitivity Adverse Reactions
Serious and occasionally fatal hypersensitivity (anaphylactic/anaphylactoid) reactions (including shock) have been reported in patients receiving therapy with piperacillin and tazobactam for injection. These reactions are more likely to occur in individuals with a history of penicillin, cephalosporin, or carbapenem hypersensitivity or a history of sensitivity to multiple allergens. Before initiating therapy with piperacillin and tazobactam for injection, careful inquiry should be made concerning previous hypersensitivity reactions. If an allergic reaction occurs, piperacillin and tazobactam for injection should be discontinued and appropriate therapy instituted.
5.2 Severe Cutaneous Adverse Reactions
Piperacillin and tazobactam for injection may cause severe cutaneous adverse reactions, such as Stevens-Johnson syndrome, toxic epidermal necrolysis, drug reaction with eosinophilia and systemic symptoms, and acute generalized exanthematous pustulosis. If patients develop a skin rash they should be monitored closely and piperacillin and tazobactam for injection discontinued if lesions progress.
5.3 Hemophagocytic Lymphohistiocytosis
Cases of hemophagocytic lymphohistiocytosis (HLH) have been reported in pediatric and adult patients treated with piperacillin and tazobactam for injection. Signs and symptoms of HLH may include fever, rash, lymphadenopathy, hepatosplenomegaly and cytopenia. If HLH is suspected, discontinue piperacillin and tazobactam for injection immediately and institute appropriate management.
5.10 Development of Drug Resistant Bacteria (5.10 Development of Drug-Resistant Bacteria)
Prescribing piperacillin and tazobactam for injection in the absence of a proven or strongly suspected bacterial infection or a prophylactic indication is unlikely to provide benefit to the patient and increases the risk of development of drug-resistant bacteria.
6.3 Additional Experience With Piperacillin (6.3 Additional Experience with Piperacillin)
The following adverse reaction has also been reported for piperacillin for injection:
Skeletal—prolonged neuromuscular blockade [see Drug Interactions (7.5)].
Principal Display Panel – 40.5 Grams Vial (PRINCIPAL DISPLAY PANEL – 40.5 grams Vial)
NDC 60505-6262-0 Rx only
Piperacillin and Tazobactam for Injection, USP
40.5 gram vial
PHARMACY BULK PACKAGE
Not for Direct Infusion
RECONSTITUTED STOCK SOLUTION MUST
BE TRANSFERRED AND FURTHER
DILUTED FOR INTRAVENOUS INFUSION
5.6 Central Nervous System Adverse Reactions
As with other penicillins, piperacillin and tazobactam for injection may cause neuromuscular excitability or seizures. Patients receiving higher doses, especially patients with renal impairment may be at greater risk for central nervous system adverse reactions. Closely monitor patients with renal impairment or seizure disorders for signs and symptoms of neuromuscular excitability or seizures [see Adverse Reactions (6.2)].
5.7 Nephrotoxicity in Critically Ill Patients
The use of piperacillin and tazobactam for injection was found to be an independent risk factor for renal failure and was associated with delayed recovery of renal function as compared to other beta-lactam antibacterial drugs in a randomized, multicenter, controlled trial in critically ill patients [see Adverse Reactions (6.1)]. Based on this study, alternative treatment options should be considered in the critically ill population. If alternative treatment options are inadequate or unavailable, monitor renal function during treatment with piperacillin and tazobactam for injection [see Dosage and Administration (2.3)].
Combined use of piperacillin and tazobactam and vancomycin may be associated with an increased incidence of acute kidney injury [see Drug Interactions (7.3)].
Principal Display Panel – 40.5 Grams Carton (PRINCIPAL DISPLAY PANEL – 40.5 grams Carton)
NDC 60505-6262-0
Rx only
Piperacillin and Tazobactam for Injection, USP
40.5 gram vial
PHARMACY BULK PACKAGE
Not for Direct Infusion
RECONSTITUTED STOCK SOLUTION MUST
BE TRANSFERRED AND FURTHER
DILUTED FOR INTRAVENOUS INFUSION
2.3 Dosage in Adult Patients With Renal Impairment
In adult patients with renal impairment (creatinine clearance ≤ 40 mL/min) and dialysis patients (hemodialysis and CAPD), the intravenous dose of piperacillin and tazobactam for injection should be reduced based on the degree of renal impairment. The recommended daily dosage of piperacillin and tazobactam for injection for patients with renal impairment administered by intravenous infusion over 30 minutes is described in Table 1.
| Creatinine clearance, mL/min | All Indications (except nosocomial pneumonia) | Nosocomial Pneumonia |
|---|---|---|
| Greater than 40 mL/min | 3.375 every 6 hours | 4.5 every 6 hours |
| 20 to 40 mL/min Creatinine clearance for patients not receiving hemodialysis
|
2.25 every 6 hours | 3.375 every 6 hours |
| Less than 20 mL/min | 2.25 every 8 hours | 2.25 every 6 hours |
| Hemodialysis 0.75 g (0.67 g piperacillin and 0.08 g tazobactam) should be administered following each hemodialysis session on hemodialysis days
|
2.25 every 12 hours | 2.25 every 8 hours |
| CAPD | 2.25 every 12 hours | 2.25 every 8 hours |
For patients on hemodialysis, the maximum dose is 2.25 g every twelve hours for all indications other than nosocomial pneumonia and 2.25 g every eight hours for nosocomial pneumonia. Since hemodialysis removes 30% to 40% of the administered dose, an additional dose of 0.75 g piperacillin and tazobactam for injection (0.67 g piperacillin and 0.08 g tazobactam) should be administered following each dialysis period on hemodialysis days. No additional dosage of piperacillin and tazobactam for injection is necessary for CAPD patients.
2.2 Dosage in Adult Patients With Nosocomial Pneumonia
Initial presumptive treatment of adult patients with nosocomial pneumonia should start with piperacillin and tazobactam for injection at a dosage of 4.5 g every six hours plus an aminoglycoside, [totaling 18.0 g (16.0 g piperacillin and 2.0 g tazobactam)], administered by intravenous infusion over 30 minutes. The recommended duration of piperacillin and tazobactam for injection treatment for nosocomial pneumonia is 7 to 14 days. Treatment with the aminoglycoside should be continued in patients from whom P. aeruginosa is isolated.
2.1 Dosage in Adult Patients With Indications Other Than Nosocomial Pneumonia
The usual total daily dosage of piperacillin and tazobactam for injection for adult patients with indications other than nosocomial pneumonia is 3.375 g every six hours [totaling 13.5 g (12.0 g piperacillin and 1.5 g tazobactam)], to be administered by intravenous infusion over 30 minutes. The usual duration of piperacillin and tazobactam for injection treatment is from 7 to 10 days.
2.4 Dosage in Pediatric Patients With Appendicitis/peritonitis Or Nosocomial Pneumonia (2.4 Dosage in Pediatric Patients With Appendicitis/Peritonitis or Nosocomial Pneumonia)
The recommended dosage for pediatric patients with appendicitis and/or peritonitis or nosocomial pneumonia aged 2 months of age and older, weighing up to 40 kg, and with normal renal function, is described in Table 2 [see Use in Specific Populations (8.4) and Clinical Pharmacology (12.3)].
| Age | Appendicitis and/or Peritonitis | Nosocomial Pneumonia |
|---|---|---|
| 2 months to 9 months | 90 mg/kg (80 mg piperacillin and 10 mg tazobactam) every 8 (eight) hours |
90 mg/kg (80 mg piperacillin and 10 mg tazobactam) every 6 (six) hours |
| Older than 9 months of age | 112.5 mg/kg (100 mg piperacillin and 12.5 mg tazobactam) every 8 (eight) hours |
112.5 mg/kg (100 mg piperacillin and 12.5 mg tazobactam) every 6 (six) hours |
Pediatric patients weighing over 40 kg and with normal renal function should receive the adult dose [see Dosage and Administration (2.1, 2.2)].
Dosage of piperacillin and tazobactam for injection in pediatric patients with renal impairment has not been determined.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:43:16.044875 · Updated: 2026-03-14T22:14:14.773616