replenix®
c12f9005-a549-4447-9beb-df7126f77df1
34390-5
HUMAN OTC DRUG LABEL
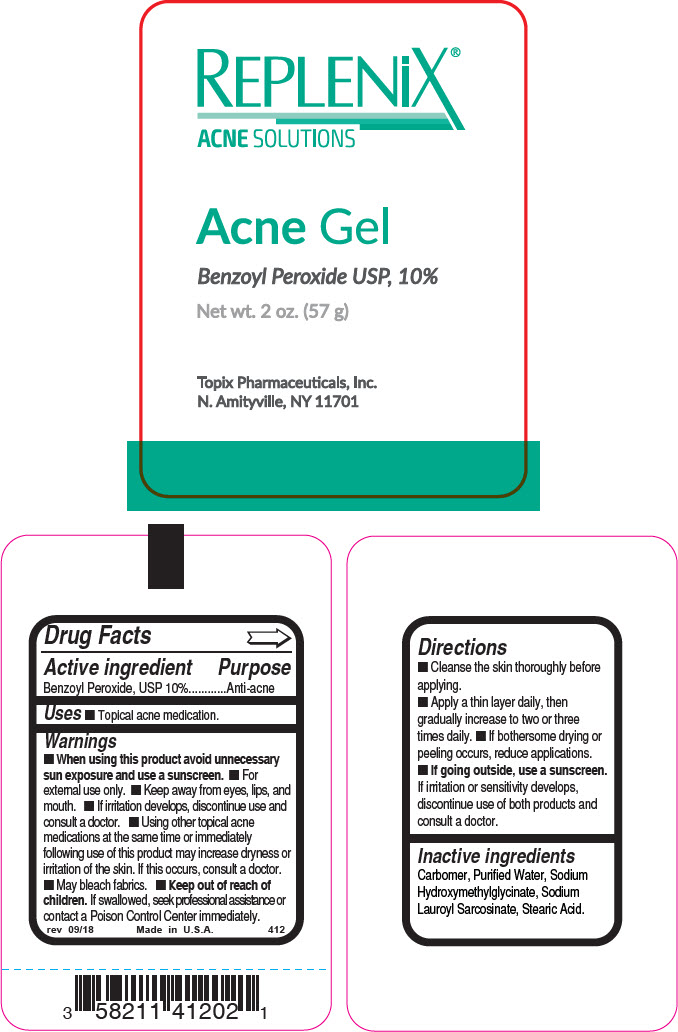
Drug Facts
Composition & Product
Identifiers & Packaging
Purpose
Anti-acne
Description
Drug Facts
Medication Information
Uses
- Topical acne medication.
Purpose
Anti-acne
Directions
- Cleanse the skin thoroughly before applying.
- Apply a thin layer daily, then gradually increase to two or three times daily.
- If bothersome drying or peeling occurs, reduce applications.
- If going outside, use a sunscreen. If irritation or sensitivity develops, discontinue use of both products and consult a doctor.
Inactive Ingredients
Carbomer, Purified Water, Sodium Hydroxymethylglycinate, Sodium Lauroyl Sarcosinate, Stearic Acid.
Description
Drug Facts
Section 42229-5
Drug Facts
Section 50565-1
- Keep out of reach of children. If swallowed, seek professional assistance or contact a Poison Control Center immediately.
Section 50566-9
- If irritation develops, discontinue use and consult a doctor.
Section 50567-7
- When using this product avoid unnecessary sun exposure and use a sunscreen.
- For external use only.
- Keep away from eyes, lips, and mouth.
Section 50569-3
- Using other topical acne medications at the same time or immediately following use of this product may increase dryness or irritation of the skin. If this occurs, consult a doctor.
Active Ingredient
Benzoyl Peroxide, USP 10%
Principal Display Panel 57 G Tube Label
REPLENiX®
ACNE SOLUTIONS
Acne Gel
Benzoyl Peroxide USP, 10%
Net wt. 2 oz. (57 g)
Topix Pharmaceuticals, Inc.
N. Amityville, NY 11701
Structured Label Content
Uses
- Topical acne medication.
Purpose
Anti-acne
Directions
- Cleanse the skin thoroughly before applying.
- Apply a thin layer daily, then gradually increase to two or three times daily.
- If bothersome drying or peeling occurs, reduce applications.
- If going outside, use a sunscreen. If irritation or sensitivity develops, discontinue use of both products and consult a doctor.
Inactive Ingredients (Inactive ingredients)
Carbomer, Purified Water, Sodium Hydroxymethylglycinate, Sodium Lauroyl Sarcosinate, Stearic Acid.
Section 42229-5 (42229-5)
Drug Facts
Section 50565-1 (50565-1)
- Keep out of reach of children. If swallowed, seek professional assistance or contact a Poison Control Center immediately.
Section 50566-9 (50566-9)
- If irritation develops, discontinue use and consult a doctor.
Section 50567-7 (50567-7)
- When using this product avoid unnecessary sun exposure and use a sunscreen.
- For external use only.
- Keep away from eyes, lips, and mouth.
Section 50569-3 (50569-3)
- Using other topical acne medications at the same time or immediately following use of this product may increase dryness or irritation of the skin. If this occurs, consult a doctor.
Active Ingredient (Active ingredient)
Benzoyl Peroxide, USP 10%
Principal Display Panel 57 G Tube Label (PRINCIPAL DISPLAY PANEL - 57 g Tube Label)
REPLENiX®
ACNE SOLUTIONS
Acne Gel
Benzoyl Peroxide USP, 10%
Net wt. 2 oz. (57 g)
Topix Pharmaceuticals, Inc.
N. Amityville, NY 11701
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:02:20.793879 · Updated: 2026-03-14T23:01:50.647319