These Highlights Do Not Include All The Information Needed To Use Gadobutrol Injection Safely And Effectively. See Full Prescribing Information For Gadobutrol Injection.
c0dd1618-0bb3-4974-846b-e5afc2311ae6
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Warnings and Precautions, Acute Respiratory Distress Syndrome ( 5.4 ) 3/2025
Indications and Usage
Gadobutrol injection is a gadolinium-based contrast agent indicated for use with magnetic resonance imaging (MRI): • To detect and visualize areas with disrupted blood brain barrier and/or abnormal vascularity of the central nervous system in adult and pediatric patients (including term neonates) ( 1.1 ) • To assess the presence and extent of malignant breast disease in adult patients ( 1.2 ) • To evaluate known or suspected supra-aortic or renal artery disease in adult and pediatric patients (including term neonates) ( 1.3 ) • To assess myocardial perfusion (stress, rest) and late gadolinium enhancement in adult patients with known or suspected coronary artery disease (CAD). ( 1.4 ).
Dosage and Administration
• Recommended dose for adults and pediatric patients (including term neonates) is 0.1 mL/kg body weight ( 2.1 ) • Administer as an intravenous bolus injection ( 2.2 ) • Follow injection with a normal saline flush ( 2.2 )
Warnings and Precautions
• Hypersensitivity Reactions: Anaphylactic and other hypersensitivity reactions with cardiovascular, respiratory or cutaneous manifestations, ranging from mild to severe, including death, have occurred. Monitor patients closely during and after administration of gadobutrol. ( 5.3 ) • Acute Respiratory Distress Syndrome: For patients demonstrating respiratory distress after administration, assess oxygen requirement and monitor for worsening respiratory function. ( 5.4 ) • Gadolinium Retention: Gadolinium is retained for months or years in brain, bone, and other organs. ( 5.5 )
Contraindications
Gadobutrol injection is contraindicated in patients with history of severe hypersensitivity reactions to gadobutrol.
Adverse Reactions
The following clinically significant adverse reactions are discussed elsewhere in labeling: • Nephrogenic Systemic Fibrosis (NSF) [see Boxed Warning and Warnings and Precautions ( 5.2 )] . • Hypersensitivity reactions [see Contraindications ( 4 ) and Warnings and Precautions ( 5.3 )] . • Acute Respiratory Distress Syndrome [see Warnings and Precautions ( 5.4 )]. • Gadolinium Retention [see Warnings and Precautions ( 5.5 )].
Storage and Handling
Store at 25°C (77°F); excursions permitted to 15° to 30°C (59° to 86°F) [see USP Controlled Room Temperature]. Should freezing occur, gadobutrol injection should be brought to room temperature before use. If allowed to stand at room temperature, gadobutrol injection should return to a clear and colorless to pale yellow solution. Visually inspect gadobutrol injection for particulate matter and discoloration prior to administration. Do not use the solution if it is discolored, if particulate matter is present or if the container appears damaged.
How Supplied
Gadobutrol injection is a sterile, clear and colorless to pale yellow solution containing 604.72 mg gadobutrol per mL (equivalent to 1 mmol gadobutrol per mL). Gadobutrol injection is supplied in the following Multiple-Dose container sizes: Product Code Unit of Sale Each 287230 NDC 65219-287-30 Packaged in cartons of 10. NDC 65219-287-10 30 mL Imaging Bulk Package with rubber stopper. 287265 NDC 65219-289-65 Packaged in cartons of 10. NDC 65219-289-10 65 mL Imaging Bulk Package with rubber stopper.
Medication Information
Warnings and Precautions
• Hypersensitivity Reactions: Anaphylactic and other hypersensitivity reactions with cardiovascular, respiratory or cutaneous manifestations, ranging from mild to severe, including death, have occurred. Monitor patients closely during and after administration of gadobutrol. ( 5.3 ) • Acute Respiratory Distress Syndrome: For patients demonstrating respiratory distress after administration, assess oxygen requirement and monitor for worsening respiratory function. ( 5.4 ) • Gadolinium Retention: Gadolinium is retained for months or years in brain, bone, and other organs. ( 5.5 )
Indications and Usage
Gadobutrol injection is a gadolinium-based contrast agent indicated for use with magnetic resonance imaging (MRI): • To detect and visualize areas with disrupted blood brain barrier and/or abnormal vascularity of the central nervous system in adult and pediatric patients (including term neonates) ( 1.1 ) • To assess the presence and extent of malignant breast disease in adult patients ( 1.2 ) • To evaluate known or suspected supra-aortic or renal artery disease in adult and pediatric patients (including term neonates) ( 1.3 ) • To assess myocardial perfusion (stress, rest) and late gadolinium enhancement in adult patients with known or suspected coronary artery disease (CAD). ( 1.4 ).
Dosage and Administration
• Recommended dose for adults and pediatric patients (including term neonates) is 0.1 mL/kg body weight ( 2.1 ) • Administer as an intravenous bolus injection ( 2.2 ) • Follow injection with a normal saline flush ( 2.2 )
Contraindications
Gadobutrol injection is contraindicated in patients with history of severe hypersensitivity reactions to gadobutrol.
Adverse Reactions
The following clinically significant adverse reactions are discussed elsewhere in labeling: • Nephrogenic Systemic Fibrosis (NSF) [see Boxed Warning and Warnings and Precautions ( 5.2 )] . • Hypersensitivity reactions [see Contraindications ( 4 ) and Warnings and Precautions ( 5.3 )] . • Acute Respiratory Distress Syndrome [see Warnings and Precautions ( 5.4 )]. • Gadolinium Retention [see Warnings and Precautions ( 5.5 )].
Storage and Handling
Store at 25°C (77°F); excursions permitted to 15° to 30°C (59° to 86°F) [see USP Controlled Room Temperature]. Should freezing occur, gadobutrol injection should be brought to room temperature before use. If allowed to stand at room temperature, gadobutrol injection should return to a clear and colorless to pale yellow solution. Visually inspect gadobutrol injection for particulate matter and discoloration prior to administration. Do not use the solution if it is discolored, if particulate matter is present or if the container appears damaged.
How Supplied
Gadobutrol injection is a sterile, clear and colorless to pale yellow solution containing 604.72 mg gadobutrol per mL (equivalent to 1 mmol gadobutrol per mL). Gadobutrol injection is supplied in the following Multiple-Dose container sizes: Product Code Unit of Sale Each 287230 NDC 65219-287-30 Packaged in cartons of 10. NDC 65219-287-10 30 mL Imaging Bulk Package with rubber stopper. 287265 NDC 65219-289-65 Packaged in cartons of 10. NDC 65219-289-10 65 mL Imaging Bulk Package with rubber stopper.
Description
Warnings and Precautions, Acute Respiratory Distress Syndrome ( 5.4 ) 3/2025
Data
Human Data
Contrast enhancement is visualized in the placenta and fetal tissues after maternal GBCA administration.
Cohort studies and case reports on exposure to GBCAs during pregnancy have not reported a clear association between GBCAs and adverse effects in the exposed neonates. However, a retrospective cohort study, comparing pregnant women who had a GBCA MRI to pregnant women who did not have an MRI, reported a higher occurrence of stillbirths and neonatal deaths in the group receiving GBCA MRI. Limitations of this study include a lack of comparison with non-contrast MRI and lack of information about the maternal indication for MRI. Overall, these data preclude a reliable evaluation of the potential risk of adverse fetal outcomes with the use of GBCAs in pregnancy.
Animal Data
Gadolinium Retention
GBCAs administered to pregnant non-human primates (0.1 mmol/kg on gestational days 85 and 135) result in measurable gadolinium concentration in the offspring in bone, brain, skin, liver, kidney, and spleen for at least 7 months. GBCAs administered to pregnant mice (2 mmol/kg daily on gestational days 16 through 19) result in measurable gadolinium concentrations in the pups in bone, brain, kidney, liver, blood, muscle, and spleen at one month postnatal age.
Reproductive Toxicology
Embryolethality was observed when gadobutrol was administered intravenously to monkeys during organogenesis at doses 8 times the recommended single human dose (based on body surface area); gadobutrol was not maternally toxic or teratogenic at this dose. Embryolethality and retardation of embryonal development also occurred in pregnant rats receiving maternally toxic doses of gadobutrol (≥ 7.5 mmol/kg body weight; equivalent to 12 times the human dose based on body surface area) and in pregnant rabbits (≥ 2.5 mmol/kg body weight; equivalent to 8 times the recommended human dose based on body surface area). In rabbits, this finding occurred without evidence of pronounced maternal toxicity and with minimal placental transfer (0.01% of the administered dose detected in the fetuses).
Because pregnant animals received repeated daily doses of gadobutrol, their overall exposure was significantly higher than that achieved with the standard single dose administered to humans.
Section 42229-5
Risk Summary
GBCAs cross the placenta and result in fetal exposure and gadolinium retention. The human data on the association between GBCAs and adverse fetal outcomes are limited and inconclusive (see Data). In animal reproduction studies, although teratogenicity was not observed, embryolethality was observed in monkeys, rabbits and rats receiving intravenous gadobutrol during organogenesis at doses 8 times and above the recommended human dose. Retardation of embryonal development was observed in rabbits and rats receiving intravenous gadobutrol during organogenesis at doses 8 and 12 times, respectively, the recommended human dose (see Data). Because of the potential risks of gadolinium to the fetus, use gadobutrol only if imaging is essential during pregnancy and cannot be delayed.
The estimated background risk of major birth defects and miscarriage for the indicated population is unknown. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2% to 4% and is 15% to 20%, respectively.
Section 43683-2
Warnings and Precautions,
Acute Respiratory Distress Syndrome (5.4) 3/2025
Section 51945-4
PRINCIPAL DISPLAY PANEL – 30 mmol/30 mL (1 mmol/mL) – Shelf Carton
Dose: 0.1 mL/kg
NDC 65219-287-30
Gadobutrol Injection
30 mmol/30 mL
(1 mmol/mL)
Imaging Bulk Package - Not for Direct Infusion
For Intravenous Use Only
Discard 24 hours after initial puncture
Dispense the enclosed Medication Guide to each patient.
14.3 Mra
Patients with known or suspected disease of the supra-aortic arteries (for evaluation up to but excluding the basilar artery) were enrolled in Study C, and patients with known or suspected disease of the renal arteries were enrolled in Study D. In both studies, non-contrast, 2D time-of-flight (ToF) magnetic resonance angiography (MRA) was performed prior to gadobutrol MRA using a single intravenous injection of 0.1 mmol/kg. The injection rate of 1.5 mL/second was selected to extend the injection duration to at least half of the imaging duration. Imaging was performed with parallel-channel, 1.5T MRI devices and an automatic bolus tracking technique to trigger the image acquisition following gadobutrol administration using elliptically encoded, T1-weighted, 3D gradient-echo image acquisition and single breath hold. Three central readers blinded to clinical information interpreted the ToF and gadobutrol MRA images. Three additional central readers interpreted separately acquired computed tomographic angiography (CTA) images, which were used as the standard of reference (SoR) in each study.
The studies included 749 subjects: 457 were evaluated in Study C, with an average age of 68 (range 25 to 93); 64% were male; 80% white, 28% black, and 16% Asian. An additional 292 subjects were evaluated in Study D, with an average age of 55 (range 18 to 88); 54% were male; 68% white, 7% black, and 22% Asian.
Efficacy was evaluated based on anatomical visualization and performance for distinguishing between normal and abnormal anatomy. The visualization metric depended on whether readers selected, “Yes, it can be visualized along its entire length...” when responding to the question, “Is this segment assessable?”. Twenty-one segments in Study C and six segments in Study D were presented per subject to each reader. The performance metrics, sensitivity and specificity, depended on digital caliper-based quantitation of arterial narrowing in visualized, non-occluded, abnormal-appearing segments. Significant stenosis was defined as at least 70% in Study C and 50% in Study D. Performance of gadobutrol MRA compared to ToF MRA was calculated using an imputation method for non-visualized segments by assigning them as a 50% match with SoR and a 50% mismatch. Performance of gadobutrol MRA compared to a pre-specified threshold of 50% was calculated after excluding non-visualized segments. Measurement variability and visualization of accessory renal arteries was also evaluated.
Results were analyzed for each of the three central readers.
| 1Number of segments varied between readers; number for majority-reader shown. | |||||||||
| 2Standard of Reference based on aggregate interpretation of three central CTA readers. | |||||||||
| 395.1/95% (Study C/D) confidence interval for two-sided comparison. | |||||||||
| 490.1/90% (Study C/D) confidence interval for one-sided comparison against non-inferiority margin of -7.5. | |||||||||
| GAD MRA = Post-contrast Gadobutrol Magnetic Resonance Angiography, ToF = Non-contrast 2D-Time of Flight. | |||||||||
|
STUDY C: SUPRA-AORTIC ARTERIES (457 patients)
|
|||||||||
|
VISUALIZATION (%) |
SENSITIVITY (%) |
SPECIFICITY (%) |
|||||||
|
READER |
GAD MRA |
ToF MRA |
GAD − ToF (CI3) |
GAD MRA |
ToF MRA |
GAD − ToF (CI4) |
GAD MRA |
ToF MRA |
GAD − ToF (CI4) |
|
1 |
88 |
24 |
64 |
60 |
54 |
6 |
92 |
62 |
30 |
|
2 |
95 |
75 |
20 |
60 |
54 |
6 |
95 |
85 |
10 |
|
3 |
97 |
82 |
15 |
58 |
55 |
3 |
97 |
89 |
8 |
|
|
|||||||||
|
STUDY D: RENAL ARTERIES (292 patients)
|
|||||||||
|
4 |
98 |
82 |
16 |
52 |
51 |
1 |
94 |
83 |
11 |
|
5 |
96 |
72 |
24 |
54 |
39 |
15 |
95 |
85 |
10 |
|
6 |
96 |
78 |
17 |
53 |
50 |
3 |
94 |
81 |
13 |
For all three supra-aortic artery readers in Study C, the lower bound of confidence for the sensitivity of gadobutrol MRA did not exceed 54%. For all three renal artery readers in Study D, the lower bound of confidence for the sensitivity of gadobutrol MRA did not exceed 46%.
10 Overdosage
11 Description
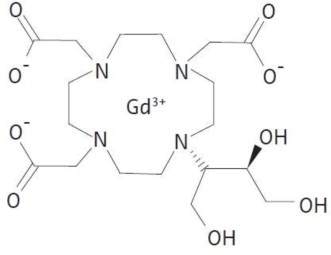
Gadobutrol injection is a paramagnetic macrocyclic contrast agent administered intravenously for magnetic resonance imaging. The chemical name for gadobutrol is 10–[(1SR,2RS)–2,3–dihydroxy–1–hydroxymethylpropyl]–1,4,7,10– tetraazacyclododecane–1,4,7–triacetic acid, gadolinium complex. Gadobutrol has a molecular formula of C18H31GdN4O9 and a molecular weight of 604.72.
Gadobutrol injection is a sterile, clear, colorless to pale yellow solution containing 604.72 mg (1.0 mmol) of gadobutrol per mL as the active ingredient with 0.513 mg of calcobutrol sodium, 1.211 mg of trometamol, hydrochloric acid (for pH adjustment) and water for injection.
Gadobutrol injection contains no preservatives.
The main physicochemical properties of gadobutrol injection (1 mmol/mL solution for injection) are listed below:
|
Density (g/mL at 37°C) |
1.3 |
|
Osmolarity at 37°C (mOsm/L solution) |
1117 |
|
Osmolality at 37°C (mOsm/kg H2O) |
1603 |
|
Viscosity at 37°C (mPa·s) |
4.96 |
|
pH |
6.6 to 8 |
The thermodynamic stability constants for gadobutrol (log Ktherm and log Kcond at pH 7.4) are 21.8 and 15.3, respectively.
1.4 Cardiac Mri
Gadobutrol injection is indicated for use in cardiac MRI (CMRI) to assess myocardial perfusion (stress, rest) and late gadolinium enhancement in adult patients with known or suspected coronary artery disease (CAD).
14.4 Cardiac Mri
Two studies similar in design, Study E and Study F, evaluated the sensitivity and specificity of gadobutrol cardiac MRI (CMRI) for detection of coronary artery disease (CAD) in adult patients with known or suspected CAD. Patients were excluded from study if they had a history of coronary artery bypass grafting, or if it was known in advance that they were unable to hold their breath, or had atrial fibrillation or other arrhythmia likely to prevent electrocardiogram-gated CMRI. The studies were multi-center, open-label, and evaluated 764 subjects for efficacy: 376 in Study E, with an average age of 59 (range 20 to 84); 69% male; 74% white, 1% black, and 25% Asian; and 388 subjects in Study F, with an average age of 59 (range 23 to 82); 61% male; 67% white, 17% black, and 12% Asian.
All subjects underwent dynamic first-pass gadobutrol imaging during vasodilator stress, followed ~10 minutes later by dynamic first-pass gadobutrol imaging at rest, followed ~5 minutes later with imaging during a period of gradual gadobutrol washout from the myocardium (late gadolinium enhancement, LGE). Imaging was performed on 1.5 T or 3.0 T MRI devices equipped with multichannel surface coils to support accelerated acquisitions with parallel imaging, T1-weighted, 2D gradient-echo, dynamic acquisition of perfusion with at least 3 slices per heartbeat. Gadobutrol was administered intravenously at a rate of ~4 mL/second as two separate bolus injections (0.05 mmol/kg each), the first at peak pharmacologic stress (~3 minutes after start of ongoing adenosine infusion, or immediately after completion of regadenoson administration, at approved doses). No additional gadobutrol was administered for LGE imaging.
Images were read by three independent readers blinded to clinical information. Reader detection of CAD depended on visually detecting defective perfusion or scar on gadobutrol CMRI (stress, rest, LGE) imaging. Quantitative coronary angiography (QCA) was used to measure intraluminal narrowing and served as the standard of reference (SoR).
Computed tomographic angiography (CTA) was used as the SoR if disease could be unequivocally excluded, and no coronary angiography (CA) was available. The left ventricular myocardium was divided into six regions. Readers provided per-region (CMRI, CTA) and per-artery (QCA) interpretations for each subject. Subject-level endpoints reflected each subject's most abnormal localized finding.
The sensitivity results for gadobutrol CMRI to detect CAD defined as either maximum stenosis ≥ 50% or ≥ 70% by QCA are presented in Table 12. For each reader, sensitivity of gadobutrol CMRI larger than 60% can be concluded if the lower 95% confidence limit of the sensitivity estimate exceeds the pre-specified threshold of 60%.
| * Stenosis determined by Quantitative Coronary Angiography (QCA) | ||||
| ** CMRI images were assessed by six independent blinded readers, three in each study. | ||||
| *** The bolded value represents the lower limit of the 95% confidence interval, which is compared to a pre-specified threshold of 60% for evaluation of sensitivity. | ||||
|
Study E |
Study F |
|||
|
≥ 50%
|
≥ 70%
|
≥ 50%
|
≥ 70%
|
|
|
Reader 1** |
77 (69, 83)*** |
90 (83, 95) |
65 (57, 72) |
77 (68, 85) |
|
Reader 2** |
65 (57, 73) |
80 (71, 87) |
56 (48, 64) |
71 (62, 80) |
|
Reader 3** |
65 (56, 72) |
79 (70, 86) |
61 (53, 69) |
76 (67, 84) |
The specificity results for gadobutrol CMRI to detect CAD defined as either maximum stenosis ≥ 50% or ≥ 70% by QCA are presented in Table 13. For each reader, specificity of gadobutrol CMRI larger than 55% can be concluded if the lower 95% confidence limit of the specificity estimate exceeds the pre-specified threshold of 55%.
| * Stenosis determined by Quantitative Coronary Angiography (QCA) | ||||
| ** CMRI images were assessed by six independent blinded readers, three in each study. | ||||
| *** The bolded value represents the lower limit of the 95% confidence interval, which is compared to a pre-specified threshold of 55% for evaluation of specificity. | ||||
|
Study E |
Study F |
|||
|
≥ 50%
|
≥ 70%
|
≥ 50%
|
≥ 70%
|
|
|
|
85 (80, 89)*** |
83 (78, 87) |
85 (80, 90) |
82 (77, 86) |
|
|
92 (88, 95) |
91 (87, 94) |
89 (84, 92) |
87 (83, 91) |
|
|
92 (88, 95) |
91 (87, 94) |
90 (85, 93) |
87 (82, 91) |
In Study E, among the 33 patients with maximum stenosis by QCA between 50% and <70%, the proportion of gadobutrol-CMRI positive detections of CAD ranged from 15% to 33%. In Study F, among the 45 patients with maximum stenosis by QCA between 50% and < 70%, the proportion of gadobutrol-CMRI positive detections of CAD ranged from 20% to 35%. The results of gadobutrol-CMRI reads to detect CAD in patients with maximum stenosis between 50% and < 70% are summarized in Table 14.
| * Stenosis determined by Quantitative Coronary Angiography (QCA). | ||
| **CMRI images were assessed by six independent blinded readers, three in each study. | ||
|
Study E (n=33) |
Study F (n=45) |
|
|
Gadobutrol-CMRI positive |
Gadobutrol-CMRI positive |
|
|
Reader 1** |
11 (33%) |
16 (35%) |
|
Reader 2** |
5(15%) |
9 (20%) |
|
Reader 3** |
6(18%) |
12 (26%) |
Medication Guide
|
Gadobutrol Injection
|
|
|
What is the most important information I should know about gadobutrol injection?
|
|
|
What is gadobutrol injection?
|
|
|
Do not receive gadobutrol injection if you have had a severe allergic reaction to gadobutrol injection. |
|
|
Before receiving gadobutrol injection, tell your healthcare provider about all your medical conditions, including if you:
|
|
|
What are the possible side effects of gadobutrol injection?
|
|
|
The most common side effects of gadobutrol injection include: headache, nausea, and dizziness.
|
|
|
General information about the safe and effective use of gadobutrol injection.
|
|
|
What are the ingredients in gadobutrol injection?
Lake Zurich, IL 60047 www.fresenius-kabi.com/us For more information, call Fresenius Kabi USA, LLC at 1-800-551-7176. |
|
|
This Medication Guide has been approved by the U.S. Food and Drug Administration. |
5/2025 |
|
26EUF04 |
451796C |
16.1 How Supplied
Gadobutrol injection is a sterile, clear and colorless to pale yellow solution containing 604.72 mg gadobutrol per mL (equivalent to 1 mmol gadobutrol per mL). Gadobutrol injection is supplied in the following Multiple-Dose container sizes:
|
Product Code |
Unit of Sale |
Each |
|
287230 |
NDC 65219-287-30 |
NDC 65219-287-10 |
|
287265 |
NDC 65219-289-65 |
NDC 65219-289-10 |
2.3 Drug Handling
-
•Visually inspect gadobutrol injection for particulate matter and discoloration prior to administration. Do not use the solution if it is discolored, if particulate matter is present or if the container appears damaged.
-
•Do not mix gadobutrol injection with other medications and do not administer gadobutrol injection in the same intravenous line simultaneously with other medications because of the potential for chemical incompatibility.
-
•Instructions of the device manufacturer must be followed.
8.4 Pediatric Use
The safety and effectiveness of gadobutrol have been established in pediatric patients, including term neonates, for use with MRI to detect and visualize areas with disrupted blood brain barrier and/or abnormal vascularity of the central nervous system and for use in MRA to evaluate known or suspected supra-aortic or renal artery disease. Use of gadobutrol in these indications is supported by adequate and well-controlled studies in adults and supportive imaging data in two studies in 135 patients 2 to less than 18 years of age and 44 patients less than 2 years of age with CNS and non-CNS lesions, and pharmacokinetic data in 130 patients 2 to less than 18 years of age and 43 patients less than 2 years of age, including term neonates [see Clinical Pharmacology (12.3) and Clinical Studies (14.1)]. The frequency, type, and severity of adverse reactions in pediatric patients were similar to adverse reactions in adults [see Adverse Reactions (6.1)]. No dose adjustment according to age is necessary in pediatric patients [see Dosage and Administration (2.1), Clinical Pharmacology (12.3), and Clinical Studies (14.1)]. The safety and effectiveness of gadobutrol have not been established in preterm neonates for any indication or in pediatric patients of any age for use with MRI to assess the presence and extent of malignant breast disease, or for use in CMRI to assess myocardial perfusion (stress, rest) and late gadolinium enhancement in patients with known or suspected coronary artery disease (CAD).
8.5 Geriatric Use
In clinical studies of gadobutrol, 1,377 patients were 65 years of age and over, while 104 patients were 80 years of age and over. No overall differences in safety or effectiveness were observed between these subjects and younger subjects, and other reported clinical experience has not identified differences in responses between the elderly and younger patients. In general, use of gadobutrol in elderly patients should be cautious, reflecting the greater frequency of impaired renal function and concomitant disease or other drug therapy. No dose adjustment according to age is necessary in this population.
14.1 Mri of the Cns
Patients referred for MRI of the central nervous system with contrast were enrolled in two clinical trials that evaluated the visualization characteristics of lesions. In both studies, patients underwent a baseline, pre-contrast MRI prior to administration of gadobutrol at a dose of 0.1 mmol/kg, followed by a post-contrast MRI. In Study A, patients also underwent an MRI before and after the administration of gadoteridol. The studies were designed to demonstrate superiority of gadobutrol MRI to non-contrast MRI for lesion visualization. For both studies, pre-contrast and pre-plus-post contrast images (paired images) were independently evaluated by three readers for contrast enhancement and border delineation using a scale of 1 to 4, and for internal morphology using a scale of 1 to 3 (Table 5). Lesion counting was also performed to demonstrate non-inferiority of paired gadobutrol image sets to pre-contrast MRI. Readers were blinded to clinical information.
|
Score |
Visualization Characteristics |
||
|
Contrast Enhancement |
Border Delineation |
Internal Morphology |
|
|
1 |
None |
None |
Poorly visible |
|
2 |
Weak |
Moderate |
Moderately visible |
|
3 |
Clear |
Clear but incomplete |
Sufficiently visible |
|
4 |
Clear and bright |
Clear and complete |
N/A |
Efficacy was determined in 657 subjects. The average age was 49 years (range 18 to 85 years) and 42% were male. The ethnic representations were 39% Caucasian, 4% Black, 16% Hispanic, 38% Asian, and 3% of other ethnic groups.
Table 6 shows a comparison of visualization results between paired images and pre-contrast images. Gadobutrol provided a statistically significant improvement for each of the three lesion visualization parameters when averaged across three independent readers for each study.
| 1 Difference of means = (paired mean) – (pre-contrast mean) | ||||||
| 2 p<0.001 | ||||||
| 3 Met noninferiority margin of -0.35 | ||||||
| 4 Did not meet noninferiority margin of -0.35 | ||||||
|
Endpoint |
Study A
|
Study B N=321 |
||||
|
Pre-contrast |
Paired |
Difference1 |
Pre-contrast |
Paired |
Difference |
|
|
Contrast Enhancement |
0.97 |
2.26 |
1.292 |
0.93 |
2.86 |
1.942 |
|
Border Delineation |
1.98 |
2.58 |
0.602 |
1.92 |
2.94 |
1.022 |
|
Internal Morphology |
1.32 |
1.93 |
0.602 |
1.57 |
2.35 |
0.782 |
|
Average # Lesions Detected |
8.08 |
8.25 |
0.174 |
2.65 |
2.97 |
0.323 |
Performances of gadobutrol and gadoteridol for visualization parameters were similar. Regarding the number of lesions detected, Study B met the prespecified noninferiority margin of -0.35 for paired read versus pre-contrast read while in Study A, gadobutrol and gadoteridol did not.
For the visualization endpoints contrast enhancement, border delineation, and internal morphology, the percentage of patients scoring higher for paired images compared to pre-contrast images ranged from 93% to 99% for Study A, and 95% to 97% for Study B. For both studies, the mean number of lesions detected on paired images exceeded that of the pre-contrast images; 37% for Study A and 24% for Study B. There were 29% and 11% of subjects in which the pre-contrast images detected more lesions for Study A and Study B, respectively.
The percentage of patients whose average reader mean score changed by ≤ 0, up to 1, up to 2, and ≥ 2 scoring categories presented in Table 5 is shown in Table 7. The categorical improvement of (≤ 0) represents higher (< 0) or identical (= 0) scores for the pre-contrast read, the categories with scores > 0 represent the magnitude of improvement seen for the paired read.
|
Study A N=336 |
Study B N=321 |
|||||||
|
Endpoint |
Categorical Improvement
|
Categorical Improvement (Paired – Pre-Contrast) % |
||||||
|
≤ 0 |
> 0 to < 1 |
1 to < 2 |
≥ 2 |
≤ 0 |
> 0 to < 1 |
1 to < 2 |
≥ 2 |
|
|
Contrast Enhancement |
1 |
30 |
55 |
13 |
3 |
6 |
34 |
57 |
|
Border Delineation |
7 |
73 |
18 |
1 |
5 |
38 |
51 |
5 |
|
Internal Morphology |
4 |
79 |
17 |
0 |
5 |
61 |
33 |
1 |
For both studies, the improvement of visualization endpoints in paired gadobutrol images compared to pre-contrast images resulted in improved assessment of normal and abnormal CNS anatomy.
4 Contraindications
Gadobutrol injection is contraindicated in patients with history of severe hypersensitivity reactions to gadobutrol.
6 Adverse Reactions
The following clinically significant adverse reactions are discussed elsewhere in labeling:
-
•Nephrogenic Systemic Fibrosis (NSF) [see Boxed Warning and Warnings and Precautions (5.2)].
-
•Hypersensitivity reactions [see Contraindications (4) and Warnings and Precautions (5.3)].
-
•Acute Respiratory Distress Syndrome [see Warnings and Precautions (5.4)].
-
•Gadolinium Retention [see Warnings and Precautions (5.5)].
2.1 Recommended Dose
The recommended dose of gadobutrol injection for adult and pediatric patients (including term neonates) is 0.1 mL/kg body weight (0.1 mmol/kg). Refer to Table 1 to determine the volume to be administered.
| *For Cardiac MRI, the dose is divided into 2 separate, equal injections | |
|
Body Weight (kg) |
Volume to be Administered (mL) |
|
2.5 |
0.25 |
|
5 |
0.5 |
|
10 |
1 |
|
15 |
1.5 |
|
20 |
2 |
|
25 |
2.5 |
|
30 |
3 |
|
35 |
3.5 |
|
40 |
4 |
|
45 |
4.5 |
|
50 |
5 |
|
60 |
6 |
|
70 |
7 |
|
80 |
8 |
|
90 |
9 |
|
100 |
10 |
|
110 |
11 |
|
120 |
12 |
|
130 |
13 |
|
140 |
14 |
8.6 Renal Impairment
Prior to administration of gadobutrol, screen all patients for renal dysfunction by obtaining a history and/or laboratory tests [see Warnings and Precautions (5.2)]. No dosage adjustment is recommended for patients with renal impairment.
Gadobutrol can be removed from the body by hemodialysis [see Warnings and Precautions (5.2) and Clinical Pharmacology (12.3)].
1.2 Mri of the Breast
Gadobutrol injection is indicated for use with MRI in adult patients to assess the presence and extent of malignant breast disease.
12.2 Pharmacodynamics
Gadobutrol leads to distinct shortening of the relaxation times even in low concentrations. At pH 7, 37°C and 1.5 T, the relaxivity (r1) - determined from the influence on the relaxation times (T1) of protons in plasma - is 5.2 L/(mmol·sec) and the relaxivity (r2) - determined from the influence on the relaxation times (T2) - is 6.1 L/(mmol·sec). These relaxivities display only slight dependence on the strength of the magnetic field. The T1 shortening effect of paramagnetic contrast agents is dependent on concentration and r1 relaxivity (see Table 3). This may improve tissue visualization.
| r1 relaxivity in plasma at 37°C | |
|
Gadolinium-Chelate |
r1 (L·mmol -1 ·s -1) |
|
Gadobenate |
6.3 |
|
Gadobutrol |
5.2 |
|
Gadodiamide |
4.3 |
|
Gadofosveset |
16 |
|
Gadopentetate |
4.1 |
|
Gadoterate |
3.6 |
|
Gadoteridol |
4.1 |
|
Gadoversetamide |
4.7 |
|
Gadoxetate |
6.9 |
Compared to 0.5 molar gadolinium-based contrast agents, the higher concentration of gadobutrol results in half the volume of administration and a more compact contrast bolus injection. At the site of imaging, the relative height and width of the time intensity curve for gadobutrol varies as a function of imaging location and multiple patient, injection, and device-specific factors.
Gadobutrol is a water-soluble, hydrophilic compound with a partition coefficient between n-butanol and buffer at pH 7.6 of about 0.006.
14.2 Mri of the Breast
Patients with recently diagnosed breast cancer were enrolled in two identical clinical trials to evaluate the ability of gadobutrol to assess the presence and extent of malignant breast disease prior to surgery. Patients underwent non-contrast breast MRI (BMR) prior to gadobutrol (0.1 mmol/kg) breast MRI. BMR images and gadobutrol BMR (combined contrast plus non-contrast) images were independently evaluated in each study by three readers blinded to clinical information. In separate reading sessions the BMR images and gadobutrol BMR images were also interpreted together with X-ray mammography images (XRM).
The studies evaluated 787 patients: Study 1 enrolled 390 women with an average age of 56 years, 74% were white, 25% Asian, 0.5% black, and 0.5% other; Study 2 enrolled 396 women and 1 man with an average age of 57 years, 71% were white, 24% Asian, 3% black, and 2% other.
The readers assessed 5 regions per breast for the presence of malignancy using each reading modality. The readings were compared to an independent standard of truth (SoT) consisting of histopathology for all regions where excisions were made and tissue evaluated. XRM plus ultrasound was used for all other regions.
The assessment of malignant disease was performed using a region based within-subject sensitivity. Sensitivity for each reading modality was defined as the mean of the percentage of malignant breast regions correctly interpreted for each subject. The within-subject sensitivity of gadobutrol BMR was superior to that of BMR. The lower bound of the 95% Confidence Interval (CI) for the difference in within-subject sensitivity ranged from 19% to 42% for Study 1 and from 12% to 27% for Study 2. The within-subject sensitivity for gadobutrol BMR and BMR as well as for gadobutrol BMR plus XRM and BMR plus XRM is presented in Table 8.
|
Study 1 |
Study 2 |
||||||||
|
Sensitivity (%)
|
Sensitivity (%)
|
||||||||
|
Reader |
BMR |
BMR + |
Gadobutrol |
Gadobutrol |
Reader |
BMR |
BMR |
Gadobutrol |
Gadobutrol |
|
XRM |
BMR |
BMR |
+ XRM |
BMR |
BMR |
||||
|
+XRM |
+XRM |
||||||||
|
1 |
37 |
71 |
83 |
84 |
4 |
73 |
83 |
87 |
90 |
|
2 |
49 |
76 |
80 |
83 |
5 |
57 |
81 |
89 |
90 |
|
3 |
63 |
75 |
87 |
87 |
6 |
55 |
80 |
86 |
88 |
Specificity was defined as the percentage of non-malignant breasts correctly identified as non-malignant. The lower limit of the 95% confidence interval for specificity of gadobutrol BMR was greater than 80% for 5 of 6 readers. (Table 9)
|
Study 1 |
Study 2 |
||||
|
Specificity (%) |
Specificity (%) |
||||
|
Reader |
Gadobutrol BMR |
Lower Limit |
Reader |
Gadobutrol BMR |
Lower Limit |
|
1 |
86 |
82 |
4 |
92 |
89 |
|
2 |
95 |
93 |
5 |
84 |
80 |
|
3 |
89 |
85 |
6 |
83 |
79 |
Three additional readers in each study read XRM alone. For these readers over both studies, sensitivity ranged from 68% to 73% and specificity in non-malignant breasts ranged from 86% to 94%.
In breasts with malignancy, a false positive detection rate was calculated as the percentage of subjects for which the readers assessed a region as malignant which could not be verified by SoT. The false positive detection rates for gadobutrol BMR ranged from 39% to 53% (95% CI Upper Bounds ranged from 44% to 58%).
1 Indications and Usage
Gadobutrol injection is a gadolinium-based contrast agent indicated for use with magnetic resonance imaging (MRI):
-
•To detect and visualize areas with disrupted blood brain barrier and/or abnormal vascularity of the central nervous system in adult and pediatric patients (including term neonates) (1.1)
-
•To assess the presence and extent of malignant breast disease in adult patients (1.2)
-
•To evaluate known or suspected supra-aortic or renal artery disease in adult and pediatric patients (including term neonates) (1.3)
-
•To assess myocardial perfusion (stress, rest) and late gadolinium enhancement in adult patients with known or suspected coronary artery disease (CAD). (1.4).
5.6 Acute Kidney Injury
In patients with chronic renal impairment, acute kidney injury sometimes requiring dialysis has been observed with the use of some GBCAs. Do not exceed the recommended dose; the risk of acute kidney injury may increase with higher than recommended doses.
12.1 Mechanism of Action
In MRI, visualization of normal and pathological tissue depends in part on variations in the radiofrequency signal intensity that occurs with:
-
•Differences in proton density
-
•Differences of the spin-lattice or longitudinal relaxation times (T1)
-
•Differences in the spin-spin or transverse relaxation time (T2)
When placed in a magnetic field, gadobutrol shortens the T1 and T2 relaxation times. The extent of decrease of T1 and T2 relaxation times, and therefore the amount of signal enhancement obtained from gadobutrol, is based upon several factors including the concentration of gadobutrol in the tissue, the field strength of the MRI system, and the relative ratio of the longitudinal and transverse relaxation times. At the recommended dose, the T1 shortening effect is observed with greatest sensitivity in T1-weighted magnetic resonance sequences. In T2*-weighted sequences the induction of local magnetic field inhomogeneities by the large magnetic moment of gadolinium and at high concentrations (during bolus injection) leads to a signal decrease.
5.5 Gadolinium Retention
Gadolinium is retained for months or years in several organs. The highest concentrations (nanomoles per gram of tissue) have been identified in the bone, followed by other organs (for example, brain, skin, kidney, liver, and spleen). The duration of retention also varies by tissue and is longest in bone. Linear GBCAs cause more retention than macrocyclic GBCAs. At equivalent doses, gadolinium retention varies among the linear agents with Omniscan (gadodiamide) and Optimark (gadoversetamide) causing greater retention than other linear agents [Eovist (gadoxetate disodium), Magnevist (gadopentetate dimeglumine), MultiHance (gadobenate dimeglumine)]. Retention is lowest and similar among the macrocyclic GBCAs [Dotarem (gadoterate meglumine), Gadobutrol injection (gadobutrol), ProHance (gadoteridol)].
Consequences of gadolinium retention in the brain have not been established. Pathologic and clinical consequences of GBCA administration and retention in skin and other organs have been established in patients with impaired renal function [see Warnings and Precautions (5.2)]. There are rare reports of pathologic skin changes in patients with normal renal function. Adverse events involving multiple organ systems have been reported in patients with normal renal function without an established causal link to gadolinium retention [see Adverse Reactions (6.2)].
While clinical consequences of gadolinium retention have not been established in patients with normal renal function, certain patients might be at higher risk. These include patients requiring multiple lifetime doses, pregnant and pediatric patients, and patients with inflammatory conditions. Consider the retention characteristics of the agent when choosing a GBCA for these patients. Minimize repetitive GBCA imaging studies particularly closely spaced studies, when possible.
16.2 Storage and Handling
Store at 25°C (77°F); excursions permitted to 15° to 30°C (59° to 86°F) [see USP Controlled Room Temperature].
Should freezing occur, gadobutrol injection should be brought to room temperature before use. If allowed to stand at room temperature, gadobutrol injection should return to a clear and colorless to pale yellow solution. Visually inspect gadobutrol injection for particulate matter and discoloration prior to administration. Do not use the solution if it is discolored, if particulate matter is present or if the container appears damaged.
5 Warnings and Precautions
-
•Hypersensitivity Reactions: Anaphylactic and other hypersensitivity reactions with cardiovascular, respiratory or cutaneous manifestations, ranging from mild to severe, including death, have occurred. Monitor patients closely during and after administration of gadobutrol. (5.3)
-
•Acute Respiratory Distress Syndrome: For patients demonstrating respiratory distress after administration, assess oxygen requirement and monitor for worsening respiratory function. (5.4)
-
•Gadolinium Retention: Gadolinium is retained for months or years in brain, bone, and other organs. (5.5)
2 Dosage and Administration
3 Dosage Forms and Strengths
Gadobutrol injection is a sterile, clear, and colorless to pale yellow solution for injection containing 604.72 mg gadobutrol per mL (equivalent to 1 mmol gadobutrol/mL).
6.2 Postmarketing Experience
The following additional adverse reactions have been identified during postmarketing use of gadobutrol or other GBCAs. Because these reactions are reported voluntarily from a population of uncertain size, it is not possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
-
•Cardiac arrest
-
•Nephrogenic Systemic Fibrosis (NSF)
-
•Hypersensitivity reactions (anaphylactic shock, circulatory collapse, respiratory arrest, bronchospasm, cyanosis, oropharyngeal swelling, laryngeal edema, blood pressure increased, chest pain, angioedema, conjunctivitis, hyperhidrosis, cough, sneezing, burning sensation, and pallor).
-
•Respiratory, Thoracic, and Mediastinal Disorders: Acute respiratory distress syndrome, pulmonary edema
-
•General Disorders and Administration Site Conditions: Adverse reactions with variable onset and duration have been reported after GBCA administration. These include fatigue, asthenia, pain syndromes, and heterogeneous clusters of symptoms in the neurological, cutaneous, and musculoskeletal systems.
-
•Skin: Gadolinium associated plaques
-
•Gastrointestinal Disorders: Acute pancreatitis with onset within 48 hours after GBCA administration
2.2 Administration Guidelines
Gadobutrol injection is formulated at a higher concentration (1 mmol/mL) compared to certain other gadolinium based contrast agents, resulting in a lower volume of administration. Use Table 1 to determine the volume to be administered.
Use sterile technique when preparing and administering gadobutrol injection.
MRI of the Central Nervous System
-
•Administer gadobutrol injection as an intravenous injection, manually or by power injector, at a flow rate of approximately 2 mL/second.
-
•Follow gadobutrol injection with flush of 0.9% Sodium Chloride Injection, USP to ensure complete administration of the contrast.
-
•Post contrast MRI can commence immediately following contrast administration.
MRI of the Breast
-
•Administer gadobutrol injection as an intravenous bolus by power injector, followed by a flush of 0.9% Sodium Chloride Injection, USP to ensure complete administration of the contrast.
-
•Start image acquisition following contrast administration and then repeat sequentially to determine peak intensity and wash-out.
MR Angiography
Image acquisition should coincide with peak arterial concentration, which varies among patients.
Adults
-
•Administer gadobutrol injection by power injector, at a flow rate of approximately 1.5 mL/second, followed by a 30 mL flush of 0.9% Sodium Chloride Injection, USP at the same rate to ensure complete administration of the contrast.
Pediatric patients
-
•Administer gadobutrol injection by power injector or manually, followed by a flush of 0.9% Sodium Chloride Injection, USP to ensure complete administration of the contrast.
Cardiac MRI
-
•Administer gadobutrol injection through a separate intravenous line in the contralateral arm if concomitantly providing a continuous infusion of a pharmacologic stress agent.
-
•Administer gadobutrol injection as two (2) separate bolus injections: 0.05 mL/kg (0.05 mmol/kg) body weight at peak pharmacologic stress followed by 0.05 mL/kg (0.05 mmol/kg) body weight at rest.
-
•Administer gadobutrol injection via a power injector at a flow rate of approximately 4 mL/second and follow each injection with a flush of 20 mL of 0.9% Sodium Chloride Injection, USP at the same flow rate.
8 Use in Specific Populations
Pregnancy: Use only if imaging is essential during pregnancy and cannot be delayed. (8.1)
5.3 Hypersensitivity Reactions
Anaphylactic and other hypersensitivity reactions with cardiovascular, respiratory or cutaneous manifestations, ranging from mild to severe, including death, have uncommonly occurred following gadobutrol administration [see Adverse Reactions (6)].
-
•Before gadobutrol administration, assess all patients for any history of a reaction to contrast media, bronchial asthma and/or allergic disorders. These patients may have an increased risk for a hypersensitivity reaction to gadobutrol.
-
•Administer gadobutrol only in situations where trained personnel and therapies are promptly available for the treatment of hypersensitivity reactions, including personnel trained in resuscitation.
Most hypersensitivity reactions to gadobutrol have occurred within half an hour after administration. Delayed reactions can occur up to several days after administration. Observe patients for signs and symptoms of hypersensitivity reactions during and following gadobutrol administration.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
The adverse reactions described in this section reflect gadobutrol exposure in 7713 subjects (including 184 pediatric patients, ages 0 to 17 years) with the majority receiving the recommended dose. Approximately 52% of the subjects were male and the ethnic distribution was 62% Caucasian, 28% Asian, 5% Hispanic, 2.5% Black, and 2.5% patients of other ethnic groups. The average age was 56 years (range from 1 week to 93 years).
Overall, approximately 4% of subjects reported one or more adverse reactions during a follow-up period that ranged from 24 hours to 7 days after gadobutrol administration.
Adverse reactions associated with the use of gadobutrol were usually mild to moderate in severity and transient in nature.
Table 2 lists adverse reactions that occurred in ≥ 0.1% subjects who received gadobutrol.
|
Reaction |
Rate (%)
|
|
Headache |
1.7 |
|
Nausea |
1.2 |
|
Dizziness |
0.5 |
|
Dysgeusia |
0.4 |
|
Feeling Hot |
0.4 |
|
Injection site reactions |
0.4 |
|
Vomiting |
0.4 |
|
Rash (includes generalized, macular, papular, pruritic) |
0.3 |
|
Erythema |
0.2 |
|
Paresthesia |
0.2 |
|
Pruritus (includes generalized) |
0.2 |
|
Dyspnea |
0.1 |
|
Urticaria |
0.1 |
Adverse reactions that occurred with a frequency of < 0.1% in subjects who received gadobutrol include: hypersensitivity/anaphylactic reaction, loss of consciousness, convulsion, parosmia, tachycardia, palpitation, dry mouth, malaise and feeling cold.
17 Patient Counseling Information
-
•Advise the patient to read the FDA-approved patient labeling (Medication Guide).
Nephrogenic Systemic Fibrosis
Instruct patients to inform their physician if they:
-
•Have a history of kidney disease and/or liver disease, or
-
•Have recently received a GBCA
GBCAs increase the risk of NSF among patients with impaired elimination of drugs. To counsel patients at risk of NSF:
-
•Describe the clinical manifestation of NSF
-
•Describe procedures to screen for the detection of renal impairment
Instruct the patients to contact their physician if they develop signs or symptoms of NSF following gadobutrol injection administration, such as burning, itching, swelling, scaling, hardening and tightening of the skin; red or dark patches on the skin; stiffness in joints with trouble moving, bending or straightening the arms, hands, legs or feet; pain in the hip bones or ribs; or muscle weakness.
Common Adverse Reactions
Inform patients that they may experience:
-
•Reactions along the venous injection site, such as mild and transient burning or pain or feeling of warmth or coldness at the injection site
-
•Side effects of headache, nausea, abnormal taste and feeling hot
Acute Respiratory Distress Syndrome
-
•Advise patients that acute respiratory distress syndrome (ARDS) has occurred with gadobutrol injection. Inform patients on the symptoms of the observed ARDS cases, and instruct patients to inform their healthcare provider if they experience these symptoms [see Warnings and Precautions (5.4)].
General Precautions
Gadolinium Retention
-
•Advise patients that gadolinium is retained for months or years in brain, bone, skin, and other organs in patients with normal renal function. The clinical consequences of retention are unknown. Retention depends on multiple factors and is greater following administration of linear GBCAs than following administration of macrocyclic GBCAs [see Warnings and Precautions (5.5)].
Instruct patients receiving gadobutrol injection to inform their physician if they:
-
•Are pregnant or breastfeeding
-
•Have a history of allergic reaction to contrast media, bronchial asthma or allergic respiratory disorder
Manufactured for:
Lake Zurich, IL 60047
www.fresenius-kabi.com/us
Product of China
Revised: May 2025
451796C
26EUF04
5.2 Nephrogenic Systemic Fibrosis
GBCAs increase the risk for nephrogenic systemic fibrosis (NSF) among patients with impaired elimination of the drugs. Avoid use of gadobutrol injection among these patients unless the diagnostic information is essential and not available with non-contrast MRI or other modalities. The GBCA-associated NSF risk appears highest for patients with chronic, severe kidney disease (GFR < 30 mL/min/1.73m2) as well as patients with acute kidney injury. The risk appears lower for patients with chronic, moderate kidney disease (GFR 30 to 59 mL/min/1.73m2) and little, if any, for patients with chronic, mild kidney disease (GFR 60 to 89 mL/min/1.73m2). NSF may result in fatal or debilitating fibrosis affecting the skin, muscle and internal organs. Report any diagnosis of NSF following gadobutrol administration to Fresenius Kabi USA, LLC at 1-800-551-7176 or FDA (1-800-FDA-1088 or www.fda.gov/medwatch).
Screen patients for acute kidney injury and other conditions that may reduce renal function. Features of acute kidney injury consist of rapid (over hours to days) and usually reversible decrease in kidney function, commonly in the setting of surgery, severe infection, injury or drug-induced kidney toxicity. Serum creatinine levels and estimated GFR may not reliably assess renal function in the setting of acute kidney injury. For patients at risk for chronically reduced renal function (for example, age > 60 years, diabetes mellitus or chronic hypertension), estimate the GFR through laboratory testing.
Among the factors that may increase the risk for NSF are repeated or higher than recommended doses of a GBCA and degree of renal impairment at the time of exposure. Record the specific GBCA and the dose administered to a patient. For patients at highest risk for NSF, do not exceed the recommended gadobutrol dose and allow a sufficient period of time for elimination of the drug prior to re-administration. For patients receiving hemodialysis, consider the prompt initiation of hemodialysis following the administration of a GBCA in order to enhance the contrast agent's elimination [see Use in Specific Populations (8.6) and Clinical Pharmacology (12.3)]. The usefulness of hemodialysis in the prevention of NSF is unknown [see Clinical Pharmacology (12.3)].
5.4 Acute Respiratory Distress Syndrome
Acute respiratory distress syndrome (ARDS) has been reported in patients administered gadobutrol injection and may be characterized by severe hypoxemia requiring oxygen support and mechanical ventilation. These manifestations may resemble an immediate hypersensitivity reaction with onset of respiratory distress within <30 minutes to 24 hours after gadobutrol injection administration. For patients demonstrating respiratory distress after gadobutrol injection administration, assess oxygen requirement and monitor for worsening respiratory function.
1.3 Magnetic Resonance Angiography (mra)
Gadobutrol injection is indicated for use in magnetic resonance angiography (MRA) in adult and pediatric patients, including term neonates, to evaluate known or suspected supra-aortic or renal artery disease.
5.1 Risk Associated With Intrathecal Use
Intrathecal administration of GBCAs can cause serious adverse reactions including death, coma, encephalopathy, and seizures. The safety and effectiveness of Gadobutrol injection have not been established with intrathecal use. Gadobutrol injection is not approved for intrathecal use [see Dosage and Administration (2.2)].
13.2 Animal Toxicology And/or Pharmacology
Local intolerance reactions, including moderate irritation associated with infiltration of inflammatory cells was observed after paravenous administration to rabbits, suggesting the possibility of occurrence of local irritation if the contrast medium leaks around veins in a clinical setting [see Warnings and Precautions (5.7)].
5.7 Extravasation and Injection Site Reactions
Ensure catheter and venous patency before the injection of gadobutrol. Extravasation into tissues during gadobutrol administration may result in moderate irritation [see Nonclinical Toxicology (13.2)].
2.4 Imaging Bulk Package Preparation Instructions
Gadobutrol injection Imaging Bulk Package (IBP) is a container of a sterile preparation for parenteral use that contains many single doses of gadobutrol for use with a medical imaging device. Gadobutrol injection Imaging Bulk Package is for intravenous use and not for direct infusion. Gadobutrol injection Imaging Bulk Package is for use only with an automated contrast injection system, contrast management system, or contrast media transfer set approved or cleared for use with this contrast agent in this Imaging Bulk Package. Please see drug and device labeling for information on devices indicated for use with this Imaging Bulk Package and techniques to help assure safe use.
-
1.The Gadobutrol injection Imaging Bulk Package is to be used only in a room designated for radiological procedures that involve intravascular administration of a contrast agent.
-
2.Utilize aseptic technique for penetrating the container closure of the Gadobutrol injection Imaging Bulk Package and transferring Gadobutrol injection.
-
3.The container closure must be penetrated only one time with a suitable sterile component of the automated contrast injection system, contrast management system, or contrast media transfer set (e.g., transfer spike) approved or cleared for use with this contrast agent in this Imaging Bulk Package.
-
4.Once the Gadobutrol injection Imaging Bulk Package is punctured, do not remove it from the work area during the entire period of use. Storage temperature of Gadobutrol injection Imaging Bulk Package after the closure has been entered is 20°C to 25°C (68°F to 77°F).
-
5.A maximum use time of 24 hours from initial puncture is permitted to complete fluid transfer. Discard any unused Gadobutrol injection 24 hours after initial puncture of the Imaging Bulk Package.
-
6.After the container closure is punctured, if the integrity of the Imaging Bulk Package and the delivery system cannot be assured through direct continuous supervision, the Imaging Bulk Package and all associated disposables for the automated contrast injection system, contrast management system, or contrast media transfer set (e.g., transfer spike) should be discarded.
5.9 Low Sensitivity for Significant Arterial Stenosis
The performance of gadobutrol MRA for detecting arterial segments with significant stenosis (>50% renal, >70% supra-aortic) has not been shown to exceed 55%. Therefore, a negative MRA study alone should not be used to rule out significant stenosis [see Clinical Studies (14.3)].
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
No carcinogenicity studies of gadobutrol have been conducted.
Gadobutrol was not mutagenic in in vitro reverse mutation tests in bacteria, in the HGPRT (hypoxanthine-guanine phosphoribosyl transferase) test using cultured Chinese hamster V79 cells, or in chromosome aberration tests in human peripheral blood lymphocytes, and was negative in an in vivo micronucleus test in mice after intravenous injection of 0.5 mmol/kg.
Gadobutrol had no effect on fertility and general reproductive performance of male and female rats when given in doses 12.2 times the human equivalent dose (based on body surface area).
5.8 Overestimation of Extent of Malignant Disease in Mri of the Breast
Gadobutrol MRI of the breast overestimated the histologically confirmed extent of malignancy in the diseased breast in up to 50% of the patients [see Clinical Studies (14.2)].
1.1 Magnetic Resonance Imaging (mri) of the Central Nervous System (cns)
Gadobutrol injection is indicated for use with magnetic resonance imaging (MRI) in adult and pediatric patients, including term neonates, to detect and visualize areas with disrupted blood brain barrier and/or abnormal vascularity of the central nervous system.
Warning: Risk Associated With Intrathecal Use and Nephrogenic Systemic Fibrosis
Risk Associated with Intrathecal Use
Intrathecal administration of gadolinium-based contrast agents (GBCAs) can cause serious adverse reactions including death, coma, encephalopathy, and seizures. Gadobutrol injection is not approved for intrathecal use [see Warnings and Precautions (5.1)].
Nephrogenic Systemic Fibrosis
GBCAs increase the risk for nephrogenic systemic fibrosis (NSF) among patients with impaired elimination of drugs. Avoid use of gadobutrol injection in these patients unless the diagnostic information is essential and not available with non-contrasted MRI or other modalities. NSF may result in fatal or debilitating fibrosis affecting the skin, muscle and internal organs.
-
•The risk for NSF appears highest among patients with:
-
oChronic, severe kidney disease (GFR < 30 mL/min/1.73m2), or
-
oAcute kidney injury.
-
-
•Screen patients for acute kidney injury and other conditions that may reduce renal function. For patients at risk for chronically reduced renal function (for example, age > 60 years, hypertension or diabetes), estimate the glomerular filtration rate (GFR) through laboratory testing.
-
•For patients at highest risk for NSF, do not exceed the recommended gadobutrol dose and allow a sufficient period of time for elimination of the drug from the body prior to any re-administration [see Warnings and Precautions (5.2)].
Structured Label Content
Data
Human Data
Contrast enhancement is visualized in the placenta and fetal tissues after maternal GBCA administration.
Cohort studies and case reports on exposure to GBCAs during pregnancy have not reported a clear association between GBCAs and adverse effects in the exposed neonates. However, a retrospective cohort study, comparing pregnant women who had a GBCA MRI to pregnant women who did not have an MRI, reported a higher occurrence of stillbirths and neonatal deaths in the group receiving GBCA MRI. Limitations of this study include a lack of comparison with non-contrast MRI and lack of information about the maternal indication for MRI. Overall, these data preclude a reliable evaluation of the potential risk of adverse fetal outcomes with the use of GBCAs in pregnancy.
Animal Data
Gadolinium Retention
GBCAs administered to pregnant non-human primates (0.1 mmol/kg on gestational days 85 and 135) result in measurable gadolinium concentration in the offspring in bone, brain, skin, liver, kidney, and spleen for at least 7 months. GBCAs administered to pregnant mice (2 mmol/kg daily on gestational days 16 through 19) result in measurable gadolinium concentrations in the pups in bone, brain, kidney, liver, blood, muscle, and spleen at one month postnatal age.
Reproductive Toxicology
Embryolethality was observed when gadobutrol was administered intravenously to monkeys during organogenesis at doses 8 times the recommended single human dose (based on body surface area); gadobutrol was not maternally toxic or teratogenic at this dose. Embryolethality and retardation of embryonal development also occurred in pregnant rats receiving maternally toxic doses of gadobutrol (≥ 7.5 mmol/kg body weight; equivalent to 12 times the human dose based on body surface area) and in pregnant rabbits (≥ 2.5 mmol/kg body weight; equivalent to 8 times the recommended human dose based on body surface area). In rabbits, this finding occurred without evidence of pronounced maternal toxicity and with minimal placental transfer (0.01% of the administered dose detected in the fetuses).
Because pregnant animals received repeated daily doses of gadobutrol, their overall exposure was significantly higher than that achieved with the standard single dose administered to humans.
Section 42229-5 (42229-5)
Risk Summary
GBCAs cross the placenta and result in fetal exposure and gadolinium retention. The human data on the association between GBCAs and adverse fetal outcomes are limited and inconclusive (see Data). In animal reproduction studies, although teratogenicity was not observed, embryolethality was observed in monkeys, rabbits and rats receiving intravenous gadobutrol during organogenesis at doses 8 times and above the recommended human dose. Retardation of embryonal development was observed in rabbits and rats receiving intravenous gadobutrol during organogenesis at doses 8 and 12 times, respectively, the recommended human dose (see Data). Because of the potential risks of gadolinium to the fetus, use gadobutrol only if imaging is essential during pregnancy and cannot be delayed.
The estimated background risk of major birth defects and miscarriage for the indicated population is unknown. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2% to 4% and is 15% to 20%, respectively.
Section 43683-2 (43683-2)
Warnings and Precautions,
Acute Respiratory Distress Syndrome (5.4) 3/2025
Section 51945-4 (51945-4)
PRINCIPAL DISPLAY PANEL – 30 mmol/30 mL (1 mmol/mL) – Shelf Carton
Dose: 0.1 mL/kg
NDC 65219-287-30
Gadobutrol Injection
30 mmol/30 mL
(1 mmol/mL)
Imaging Bulk Package - Not for Direct Infusion
For Intravenous Use Only
Discard 24 hours after initial puncture
Dispense the enclosed Medication Guide to each patient.
14.3 Mra (14.3 MRA)
Patients with known or suspected disease of the supra-aortic arteries (for evaluation up to but excluding the basilar artery) were enrolled in Study C, and patients with known or suspected disease of the renal arteries were enrolled in Study D. In both studies, non-contrast, 2D time-of-flight (ToF) magnetic resonance angiography (MRA) was performed prior to gadobutrol MRA using a single intravenous injection of 0.1 mmol/kg. The injection rate of 1.5 mL/second was selected to extend the injection duration to at least half of the imaging duration. Imaging was performed with parallel-channel, 1.5T MRI devices and an automatic bolus tracking technique to trigger the image acquisition following gadobutrol administration using elliptically encoded, T1-weighted, 3D gradient-echo image acquisition and single breath hold. Three central readers blinded to clinical information interpreted the ToF and gadobutrol MRA images. Three additional central readers interpreted separately acquired computed tomographic angiography (CTA) images, which were used as the standard of reference (SoR) in each study.
The studies included 749 subjects: 457 were evaluated in Study C, with an average age of 68 (range 25 to 93); 64% were male; 80% white, 28% black, and 16% Asian. An additional 292 subjects were evaluated in Study D, with an average age of 55 (range 18 to 88); 54% were male; 68% white, 7% black, and 22% Asian.
Efficacy was evaluated based on anatomical visualization and performance for distinguishing between normal and abnormal anatomy. The visualization metric depended on whether readers selected, “Yes, it can be visualized along its entire length...” when responding to the question, “Is this segment assessable?”. Twenty-one segments in Study C and six segments in Study D were presented per subject to each reader. The performance metrics, sensitivity and specificity, depended on digital caliper-based quantitation of arterial narrowing in visualized, non-occluded, abnormal-appearing segments. Significant stenosis was defined as at least 70% in Study C and 50% in Study D. Performance of gadobutrol MRA compared to ToF MRA was calculated using an imputation method for non-visualized segments by assigning them as a 50% match with SoR and a 50% mismatch. Performance of gadobutrol MRA compared to a pre-specified threshold of 50% was calculated after excluding non-visualized segments. Measurement variability and visualization of accessory renal arteries was also evaluated.
Results were analyzed for each of the three central readers.
| 1Number of segments varied between readers; number for majority-reader shown. | |||||||||
| 2Standard of Reference based on aggregate interpretation of three central CTA readers. | |||||||||
| 395.1/95% (Study C/D) confidence interval for two-sided comparison. | |||||||||
| 490.1/90% (Study C/D) confidence interval for one-sided comparison against non-inferiority margin of -7.5. | |||||||||
| GAD MRA = Post-contrast Gadobutrol Magnetic Resonance Angiography, ToF = Non-contrast 2D-Time of Flight. | |||||||||
|
STUDY C: SUPRA-AORTIC ARTERIES (457 patients)
|
|||||||||
|
VISUALIZATION (%) |
SENSITIVITY (%) |
SPECIFICITY (%) |
|||||||
|
READER |
GAD MRA |
ToF MRA |
GAD − ToF (CI3) |
GAD MRA |
ToF MRA |
GAD − ToF (CI4) |
GAD MRA |
ToF MRA |
GAD − ToF (CI4) |
|
1 |
88 |
24 |
64 |
60 |
54 |
6 |
92 |
62 |
30 |
|
2 |
95 |
75 |
20 |
60 |
54 |
6 |
95 |
85 |
10 |
|
3 |
97 |
82 |
15 |
58 |
55 |
3 |
97 |
89 |
8 |
|
|
|||||||||
|
STUDY D: RENAL ARTERIES (292 patients)
|
|||||||||
|
4 |
98 |
82 |
16 |
52 |
51 |
1 |
94 |
83 |
11 |
|
5 |
96 |
72 |
24 |
54 |
39 |
15 |
95 |
85 |
10 |
|
6 |
96 |
78 |
17 |
53 |
50 |
3 |
94 |
81 |
13 |
For all three supra-aortic artery readers in Study C, the lower bound of confidence for the sensitivity of gadobutrol MRA did not exceed 54%. For all three renal artery readers in Study D, the lower bound of confidence for the sensitivity of gadobutrol MRA did not exceed 46%.
10 Overdosage (10 OVERDOSAGE)
11 Description (11 DESCRIPTION)
Gadobutrol injection is a paramagnetic macrocyclic contrast agent administered intravenously for magnetic resonance imaging. The chemical name for gadobutrol is 10–[(1SR,2RS)–2,3–dihydroxy–1–hydroxymethylpropyl]–1,4,7,10– tetraazacyclododecane–1,4,7–triacetic acid, gadolinium complex. Gadobutrol has a molecular formula of C18H31GdN4O9 and a molecular weight of 604.72.
Gadobutrol injection is a sterile, clear, colorless to pale yellow solution containing 604.72 mg (1.0 mmol) of gadobutrol per mL as the active ingredient with 0.513 mg of calcobutrol sodium, 1.211 mg of trometamol, hydrochloric acid (for pH adjustment) and water for injection.
Gadobutrol injection contains no preservatives.
The main physicochemical properties of gadobutrol injection (1 mmol/mL solution for injection) are listed below:
|
Density (g/mL at 37°C) |
1.3 |
|
Osmolarity at 37°C (mOsm/L solution) |
1117 |
|
Osmolality at 37°C (mOsm/kg H2O) |
1603 |
|
Viscosity at 37°C (mPa·s) |
4.96 |
|
pH |
6.6 to 8 |
The thermodynamic stability constants for gadobutrol (log Ktherm and log Kcond at pH 7.4) are 21.8 and 15.3, respectively.
1.4 Cardiac Mri (1.4 Cardiac MRI)
Gadobutrol injection is indicated for use in cardiac MRI (CMRI) to assess myocardial perfusion (stress, rest) and late gadolinium enhancement in adult patients with known or suspected coronary artery disease (CAD).
14.4 Cardiac Mri (14.4 Cardiac MRI)
Two studies similar in design, Study E and Study F, evaluated the sensitivity and specificity of gadobutrol cardiac MRI (CMRI) for detection of coronary artery disease (CAD) in adult patients with known or suspected CAD. Patients were excluded from study if they had a history of coronary artery bypass grafting, or if it was known in advance that they were unable to hold their breath, or had atrial fibrillation or other arrhythmia likely to prevent electrocardiogram-gated CMRI. The studies were multi-center, open-label, and evaluated 764 subjects for efficacy: 376 in Study E, with an average age of 59 (range 20 to 84); 69% male; 74% white, 1% black, and 25% Asian; and 388 subjects in Study F, with an average age of 59 (range 23 to 82); 61% male; 67% white, 17% black, and 12% Asian.
All subjects underwent dynamic first-pass gadobutrol imaging during vasodilator stress, followed ~10 minutes later by dynamic first-pass gadobutrol imaging at rest, followed ~5 minutes later with imaging during a period of gradual gadobutrol washout from the myocardium (late gadolinium enhancement, LGE). Imaging was performed on 1.5 T or 3.0 T MRI devices equipped with multichannel surface coils to support accelerated acquisitions with parallel imaging, T1-weighted, 2D gradient-echo, dynamic acquisition of perfusion with at least 3 slices per heartbeat. Gadobutrol was administered intravenously at a rate of ~4 mL/second as two separate bolus injections (0.05 mmol/kg each), the first at peak pharmacologic stress (~3 minutes after start of ongoing adenosine infusion, or immediately after completion of regadenoson administration, at approved doses). No additional gadobutrol was administered for LGE imaging.
Images were read by three independent readers blinded to clinical information. Reader detection of CAD depended on visually detecting defective perfusion or scar on gadobutrol CMRI (stress, rest, LGE) imaging. Quantitative coronary angiography (QCA) was used to measure intraluminal narrowing and served as the standard of reference (SoR).
Computed tomographic angiography (CTA) was used as the SoR if disease could be unequivocally excluded, and no coronary angiography (CA) was available. The left ventricular myocardium was divided into six regions. Readers provided per-region (CMRI, CTA) and per-artery (QCA) interpretations for each subject. Subject-level endpoints reflected each subject's most abnormal localized finding.
The sensitivity results for gadobutrol CMRI to detect CAD defined as either maximum stenosis ≥ 50% or ≥ 70% by QCA are presented in Table 12. For each reader, sensitivity of gadobutrol CMRI larger than 60% can be concluded if the lower 95% confidence limit of the sensitivity estimate exceeds the pre-specified threshold of 60%.
| * Stenosis determined by Quantitative Coronary Angiography (QCA) | ||||
| ** CMRI images were assessed by six independent blinded readers, three in each study. | ||||
| *** The bolded value represents the lower limit of the 95% confidence interval, which is compared to a pre-specified threshold of 60% for evaluation of sensitivity. | ||||
|
Study E |
Study F |
|||
|
≥ 50%
|
≥ 70%
|
≥ 50%
|
≥ 70%
|
|
|
Reader 1** |
77 (69, 83)*** |
90 (83, 95) |
65 (57, 72) |
77 (68, 85) |
|
Reader 2** |
65 (57, 73) |
80 (71, 87) |
56 (48, 64) |
71 (62, 80) |
|
Reader 3** |
65 (56, 72) |
79 (70, 86) |
61 (53, 69) |
76 (67, 84) |
The specificity results for gadobutrol CMRI to detect CAD defined as either maximum stenosis ≥ 50% or ≥ 70% by QCA are presented in Table 13. For each reader, specificity of gadobutrol CMRI larger than 55% can be concluded if the lower 95% confidence limit of the specificity estimate exceeds the pre-specified threshold of 55%.
| * Stenosis determined by Quantitative Coronary Angiography (QCA) | ||||
| ** CMRI images were assessed by six independent blinded readers, three in each study. | ||||
| *** The bolded value represents the lower limit of the 95% confidence interval, which is compared to a pre-specified threshold of 55% for evaluation of specificity. | ||||
|
Study E |
Study F |
|||
|
≥ 50%
|
≥ 70%
|
≥ 50%
|
≥ 70%
|
|
|
|
85 (80, 89)*** |
83 (78, 87) |
85 (80, 90) |
82 (77, 86) |
|
|
92 (88, 95) |
91 (87, 94) |
89 (84, 92) |
87 (83, 91) |
|
|
92 (88, 95) |
91 (87, 94) |
90 (85, 93) |
87 (82, 91) |
In Study E, among the 33 patients with maximum stenosis by QCA between 50% and <70%, the proportion of gadobutrol-CMRI positive detections of CAD ranged from 15% to 33%. In Study F, among the 45 patients with maximum stenosis by QCA between 50% and < 70%, the proportion of gadobutrol-CMRI positive detections of CAD ranged from 20% to 35%. The results of gadobutrol-CMRI reads to detect CAD in patients with maximum stenosis between 50% and < 70% are summarized in Table 14.
| * Stenosis determined by Quantitative Coronary Angiography (QCA). | ||
| **CMRI images were assessed by six independent blinded readers, three in each study. | ||
|
Study E (n=33) |
Study F (n=45) |
|
|
Gadobutrol-CMRI positive |
Gadobutrol-CMRI positive |
|
|
Reader 1** |
11 (33%) |
16 (35%) |
|
Reader 2** |
5(15%) |
9 (20%) |
|
Reader 3** |
6(18%) |
12 (26%) |
Medication Guide (MEDICATION GUIDE)
|
Gadobutrol Injection
|
|
|
What is the most important information I should know about gadobutrol injection?
|
|
|
What is gadobutrol injection?
|
|
|
Do not receive gadobutrol injection if you have had a severe allergic reaction to gadobutrol injection. |
|
|
Before receiving gadobutrol injection, tell your healthcare provider about all your medical conditions, including if you:
|
|
|
What are the possible side effects of gadobutrol injection?
|
|
|
The most common side effects of gadobutrol injection include: headache, nausea, and dizziness.
|
|
|
General information about the safe and effective use of gadobutrol injection.
|
|
|
What are the ingredients in gadobutrol injection?
Lake Zurich, IL 60047 www.fresenius-kabi.com/us For more information, call Fresenius Kabi USA, LLC at 1-800-551-7176. |
|
|
This Medication Guide has been approved by the U.S. Food and Drug Administration. |
5/2025 |
|
26EUF04 |
451796C |
16.1 How Supplied
Gadobutrol injection is a sterile, clear and colorless to pale yellow solution containing 604.72 mg gadobutrol per mL (equivalent to 1 mmol gadobutrol per mL). Gadobutrol injection is supplied in the following Multiple-Dose container sizes:
|
Product Code |
Unit of Sale |
Each |
|
287230 |
NDC 65219-287-30 |
NDC 65219-287-10 |
|
287265 |
NDC 65219-289-65 |
NDC 65219-289-10 |
2.3 Drug Handling
-
•Visually inspect gadobutrol injection for particulate matter and discoloration prior to administration. Do not use the solution if it is discolored, if particulate matter is present or if the container appears damaged.
-
•Do not mix gadobutrol injection with other medications and do not administer gadobutrol injection in the same intravenous line simultaneously with other medications because of the potential for chemical incompatibility.
-
•Instructions of the device manufacturer must be followed.
8.4 Pediatric Use
The safety and effectiveness of gadobutrol have been established in pediatric patients, including term neonates, for use with MRI to detect and visualize areas with disrupted blood brain barrier and/or abnormal vascularity of the central nervous system and for use in MRA to evaluate known or suspected supra-aortic or renal artery disease. Use of gadobutrol in these indications is supported by adequate and well-controlled studies in adults and supportive imaging data in two studies in 135 patients 2 to less than 18 years of age and 44 patients less than 2 years of age with CNS and non-CNS lesions, and pharmacokinetic data in 130 patients 2 to less than 18 years of age and 43 patients less than 2 years of age, including term neonates [see Clinical Pharmacology (12.3) and Clinical Studies (14.1)]. The frequency, type, and severity of adverse reactions in pediatric patients were similar to adverse reactions in adults [see Adverse Reactions (6.1)]. No dose adjustment according to age is necessary in pediatric patients [see Dosage and Administration (2.1), Clinical Pharmacology (12.3), and Clinical Studies (14.1)]. The safety and effectiveness of gadobutrol have not been established in preterm neonates for any indication or in pediatric patients of any age for use with MRI to assess the presence and extent of malignant breast disease, or for use in CMRI to assess myocardial perfusion (stress, rest) and late gadolinium enhancement in patients with known or suspected coronary artery disease (CAD).
8.5 Geriatric Use
In clinical studies of gadobutrol, 1,377 patients were 65 years of age and over, while 104 patients were 80 years of age and over. No overall differences in safety or effectiveness were observed between these subjects and younger subjects, and other reported clinical experience has not identified differences in responses between the elderly and younger patients. In general, use of gadobutrol in elderly patients should be cautious, reflecting the greater frequency of impaired renal function and concomitant disease or other drug therapy. No dose adjustment according to age is necessary in this population.
14.1 Mri of the Cns (14.1 MRI of the CNS)
Patients referred for MRI of the central nervous system with contrast were enrolled in two clinical trials that evaluated the visualization characteristics of lesions. In both studies, patients underwent a baseline, pre-contrast MRI prior to administration of gadobutrol at a dose of 0.1 mmol/kg, followed by a post-contrast MRI. In Study A, patients also underwent an MRI before and after the administration of gadoteridol. The studies were designed to demonstrate superiority of gadobutrol MRI to non-contrast MRI for lesion visualization. For both studies, pre-contrast and pre-plus-post contrast images (paired images) were independently evaluated by three readers for contrast enhancement and border delineation using a scale of 1 to 4, and for internal morphology using a scale of 1 to 3 (Table 5). Lesion counting was also performed to demonstrate non-inferiority of paired gadobutrol image sets to pre-contrast MRI. Readers were blinded to clinical information.
|
Score |
Visualization Characteristics |
||
|
Contrast Enhancement |
Border Delineation |
Internal Morphology |
|
|
1 |
None |
None |
Poorly visible |
|
2 |
Weak |
Moderate |
Moderately visible |
|
3 |
Clear |
Clear but incomplete |
Sufficiently visible |
|
4 |
Clear and bright |
Clear and complete |
N/A |
Efficacy was determined in 657 subjects. The average age was 49 years (range 18 to 85 years) and 42% were male. The ethnic representations were 39% Caucasian, 4% Black, 16% Hispanic, 38% Asian, and 3% of other ethnic groups.
Table 6 shows a comparison of visualization results between paired images and pre-contrast images. Gadobutrol provided a statistically significant improvement for each of the three lesion visualization parameters when averaged across three independent readers for each study.
| 1 Difference of means = (paired mean) – (pre-contrast mean) | ||||||
| 2 p<0.001 | ||||||
| 3 Met noninferiority margin of -0.35 | ||||||
| 4 Did not meet noninferiority margin of -0.35 | ||||||
|
Endpoint |
Study A
|
Study B N=321 |
||||
|
Pre-contrast |
Paired |
Difference1 |
Pre-contrast |
Paired |
Difference |
|
|
Contrast Enhancement |
0.97 |
2.26 |
1.292 |
0.93 |
2.86 |
1.942 |
|
Border Delineation |
1.98 |
2.58 |
0.602 |
1.92 |
2.94 |
1.022 |
|
Internal Morphology |
1.32 |
1.93 |
0.602 |
1.57 |
2.35 |
0.782 |
|
Average # Lesions Detected |
8.08 |
8.25 |
0.174 |
2.65 |
2.97 |
0.323 |
Performances of gadobutrol and gadoteridol for visualization parameters were similar. Regarding the number of lesions detected, Study B met the prespecified noninferiority margin of -0.35 for paired read versus pre-contrast read while in Study A, gadobutrol and gadoteridol did not.
For the visualization endpoints contrast enhancement, border delineation, and internal morphology, the percentage of patients scoring higher for paired images compared to pre-contrast images ranged from 93% to 99% for Study A, and 95% to 97% for Study B. For both studies, the mean number of lesions detected on paired images exceeded that of the pre-contrast images; 37% for Study A and 24% for Study B. There were 29% and 11% of subjects in which the pre-contrast images detected more lesions for Study A and Study B, respectively.
The percentage of patients whose average reader mean score changed by ≤ 0, up to 1, up to 2, and ≥ 2 scoring categories presented in Table 5 is shown in Table 7. The categorical improvement of (≤ 0) represents higher (< 0) or identical (= 0) scores for the pre-contrast read, the categories with scores > 0 represent the magnitude of improvement seen for the paired read.
|
Study A N=336 |
Study B N=321 |
|||||||
|
Endpoint |
Categorical Improvement
|
Categorical Improvement (Paired – Pre-Contrast) % |
||||||
|
≤ 0 |
> 0 to < 1 |
1 to < 2 |
≥ 2 |
≤ 0 |
> 0 to < 1 |
1 to < 2 |
≥ 2 |
|
|
Contrast Enhancement |
1 |
30 |
55 |
13 |
3 |
6 |
34 |
57 |
|
Border Delineation |
7 |
73 |
18 |
1 |
5 |
38 |
51 |
5 |
|
Internal Morphology |
4 |
79 |
17 |
0 |
5 |
61 |
33 |
1 |
For both studies, the improvement of visualization endpoints in paired gadobutrol images compared to pre-contrast images resulted in improved assessment of normal and abnormal CNS anatomy.
4 Contraindications (4 CONTRAINDICATIONS)
Gadobutrol injection is contraindicated in patients with history of severe hypersensitivity reactions to gadobutrol.
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following clinically significant adverse reactions are discussed elsewhere in labeling:
-
•Nephrogenic Systemic Fibrosis (NSF) [see Boxed Warning and Warnings and Precautions (5.2)].
-
•Hypersensitivity reactions [see Contraindications (4) and Warnings and Precautions (5.3)].
-
•Acute Respiratory Distress Syndrome [see Warnings and Precautions (5.4)].
-
•Gadolinium Retention [see Warnings and Precautions (5.5)].
2.1 Recommended Dose
The recommended dose of gadobutrol injection for adult and pediatric patients (including term neonates) is 0.1 mL/kg body weight (0.1 mmol/kg). Refer to Table 1 to determine the volume to be administered.
| *For Cardiac MRI, the dose is divided into 2 separate, equal injections | |
|
Body Weight (kg) |
Volume to be Administered (mL) |
|
2.5 |
0.25 |
|
5 |
0.5 |
|
10 |
1 |
|
15 |
1.5 |
|
20 |
2 |
|
25 |
2.5 |
|
30 |
3 |
|
35 |
3.5 |
|
40 |
4 |
|
45 |
4.5 |
|
50 |
5 |
|
60 |
6 |
|
70 |
7 |
|
80 |
8 |
|
90 |
9 |
|
100 |
10 |
|
110 |
11 |
|
120 |
12 |
|
130 |
13 |
|
140 |
14 |
8.6 Renal Impairment
Prior to administration of gadobutrol, screen all patients for renal dysfunction by obtaining a history and/or laboratory tests [see Warnings and Precautions (5.2)]. No dosage adjustment is recommended for patients with renal impairment.
Gadobutrol can be removed from the body by hemodialysis [see Warnings and Precautions (5.2) and Clinical Pharmacology (12.3)].
1.2 Mri of the Breast (1.2 MRI of the Breast)
Gadobutrol injection is indicated for use with MRI in adult patients to assess the presence and extent of malignant breast disease.
12.2 Pharmacodynamics
Gadobutrol leads to distinct shortening of the relaxation times even in low concentrations. At pH 7, 37°C and 1.5 T, the relaxivity (r1) - determined from the influence on the relaxation times (T1) of protons in plasma - is 5.2 L/(mmol·sec) and the relaxivity (r2) - determined from the influence on the relaxation times (T2) - is 6.1 L/(mmol·sec). These relaxivities display only slight dependence on the strength of the magnetic field. The T1 shortening effect of paramagnetic contrast agents is dependent on concentration and r1 relaxivity (see Table 3). This may improve tissue visualization.
| r1 relaxivity in plasma at 37°C | |
|
Gadolinium-Chelate |
r1 (L·mmol -1 ·s -1) |
|
Gadobenate |
6.3 |
|
Gadobutrol |
5.2 |
|
Gadodiamide |
4.3 |
|
Gadofosveset |
16 |
|
Gadopentetate |
4.1 |
|
Gadoterate |
3.6 |
|
Gadoteridol |
4.1 |
|
Gadoversetamide |
4.7 |
|
Gadoxetate |
6.9 |
Compared to 0.5 molar gadolinium-based contrast agents, the higher concentration of gadobutrol results in half the volume of administration and a more compact contrast bolus injection. At the site of imaging, the relative height and width of the time intensity curve for gadobutrol varies as a function of imaging location and multiple patient, injection, and device-specific factors.
Gadobutrol is a water-soluble, hydrophilic compound with a partition coefficient between n-butanol and buffer at pH 7.6 of about 0.006.
14.2 Mri of the Breast (14.2 MRI of the Breast)
Patients with recently diagnosed breast cancer were enrolled in two identical clinical trials to evaluate the ability of gadobutrol to assess the presence and extent of malignant breast disease prior to surgery. Patients underwent non-contrast breast MRI (BMR) prior to gadobutrol (0.1 mmol/kg) breast MRI. BMR images and gadobutrol BMR (combined contrast plus non-contrast) images were independently evaluated in each study by three readers blinded to clinical information. In separate reading sessions the BMR images and gadobutrol BMR images were also interpreted together with X-ray mammography images (XRM).
The studies evaluated 787 patients: Study 1 enrolled 390 women with an average age of 56 years, 74% were white, 25% Asian, 0.5% black, and 0.5% other; Study 2 enrolled 396 women and 1 man with an average age of 57 years, 71% were white, 24% Asian, 3% black, and 2% other.
The readers assessed 5 regions per breast for the presence of malignancy using each reading modality. The readings were compared to an independent standard of truth (SoT) consisting of histopathology for all regions where excisions were made and tissue evaluated. XRM plus ultrasound was used for all other regions.
The assessment of malignant disease was performed using a region based within-subject sensitivity. Sensitivity for each reading modality was defined as the mean of the percentage of malignant breast regions correctly interpreted for each subject. The within-subject sensitivity of gadobutrol BMR was superior to that of BMR. The lower bound of the 95% Confidence Interval (CI) for the difference in within-subject sensitivity ranged from 19% to 42% for Study 1 and from 12% to 27% for Study 2. The within-subject sensitivity for gadobutrol BMR and BMR as well as for gadobutrol BMR plus XRM and BMR plus XRM is presented in Table 8.
|
Study 1 |
Study 2 |
||||||||
|
Sensitivity (%)
|
Sensitivity (%)
|
||||||||
|
Reader |
BMR |
BMR + |
Gadobutrol |
Gadobutrol |
Reader |
BMR |
BMR |
Gadobutrol |
Gadobutrol |
|
XRM |
BMR |
BMR |
+ XRM |
BMR |
BMR |
||||
|
+XRM |
+XRM |
||||||||
|
1 |
37 |
71 |
83 |
84 |
4 |
73 |
83 |
87 |
90 |
|
2 |
49 |
76 |
80 |
83 |
5 |
57 |
81 |
89 |
90 |
|
3 |
63 |
75 |
87 |
87 |
6 |
55 |
80 |
86 |
88 |
Specificity was defined as the percentage of non-malignant breasts correctly identified as non-malignant. The lower limit of the 95% confidence interval for specificity of gadobutrol BMR was greater than 80% for 5 of 6 readers. (Table 9)
|
Study 1 |
Study 2 |
||||
|
Specificity (%) |
Specificity (%) |
||||
|
Reader |
Gadobutrol BMR |
Lower Limit |
Reader |
Gadobutrol BMR |
Lower Limit |
|
1 |
86 |
82 |
4 |
92 |
89 |
|
2 |
95 |
93 |
5 |
84 |
80 |
|
3 |
89 |
85 |
6 |
83 |
79 |
Three additional readers in each study read XRM alone. For these readers over both studies, sensitivity ranged from 68% to 73% and specificity in non-malignant breasts ranged from 86% to 94%.
In breasts with malignancy, a false positive detection rate was calculated as the percentage of subjects for which the readers assessed a region as malignant which could not be verified by SoT. The false positive detection rates for gadobutrol BMR ranged from 39% to 53% (95% CI Upper Bounds ranged from 44% to 58%).
1 Indications and Usage (1 INDICATIONS AND USAGE)
Gadobutrol injection is a gadolinium-based contrast agent indicated for use with magnetic resonance imaging (MRI):
-
•To detect and visualize areas with disrupted blood brain barrier and/or abnormal vascularity of the central nervous system in adult and pediatric patients (including term neonates) (1.1)
-
•To assess the presence and extent of malignant breast disease in adult patients (1.2)
-
•To evaluate known or suspected supra-aortic or renal artery disease in adult and pediatric patients (including term neonates) (1.3)
-
•To assess myocardial perfusion (stress, rest) and late gadolinium enhancement in adult patients with known or suspected coronary artery disease (CAD). (1.4).
5.6 Acute Kidney Injury
In patients with chronic renal impairment, acute kidney injury sometimes requiring dialysis has been observed with the use of some GBCAs. Do not exceed the recommended dose; the risk of acute kidney injury may increase with higher than recommended doses.
12.1 Mechanism of Action
In MRI, visualization of normal and pathological tissue depends in part on variations in the radiofrequency signal intensity that occurs with:
-
•Differences in proton density
-
•Differences of the spin-lattice or longitudinal relaxation times (T1)
-
•Differences in the spin-spin or transverse relaxation time (T2)
When placed in a magnetic field, gadobutrol shortens the T1 and T2 relaxation times. The extent of decrease of T1 and T2 relaxation times, and therefore the amount of signal enhancement obtained from gadobutrol, is based upon several factors including the concentration of gadobutrol in the tissue, the field strength of the MRI system, and the relative ratio of the longitudinal and transverse relaxation times. At the recommended dose, the T1 shortening effect is observed with greatest sensitivity in T1-weighted magnetic resonance sequences. In T2*-weighted sequences the induction of local magnetic field inhomogeneities by the large magnetic moment of gadolinium and at high concentrations (during bolus injection) leads to a signal decrease.
5.5 Gadolinium Retention
Gadolinium is retained for months or years in several organs. The highest concentrations (nanomoles per gram of tissue) have been identified in the bone, followed by other organs (for example, brain, skin, kidney, liver, and spleen). The duration of retention also varies by tissue and is longest in bone. Linear GBCAs cause more retention than macrocyclic GBCAs. At equivalent doses, gadolinium retention varies among the linear agents with Omniscan (gadodiamide) and Optimark (gadoversetamide) causing greater retention than other linear agents [Eovist (gadoxetate disodium), Magnevist (gadopentetate dimeglumine), MultiHance (gadobenate dimeglumine)]. Retention is lowest and similar among the macrocyclic GBCAs [Dotarem (gadoterate meglumine), Gadobutrol injection (gadobutrol), ProHance (gadoteridol)].
Consequences of gadolinium retention in the brain have not been established. Pathologic and clinical consequences of GBCA administration and retention in skin and other organs have been established in patients with impaired renal function [see Warnings and Precautions (5.2)]. There are rare reports of pathologic skin changes in patients with normal renal function. Adverse events involving multiple organ systems have been reported in patients with normal renal function without an established causal link to gadolinium retention [see Adverse Reactions (6.2)].
While clinical consequences of gadolinium retention have not been established in patients with normal renal function, certain patients might be at higher risk. These include patients requiring multiple lifetime doses, pregnant and pediatric patients, and patients with inflammatory conditions. Consider the retention characteristics of the agent when choosing a GBCA for these patients. Minimize repetitive GBCA imaging studies particularly closely spaced studies, when possible.
16.2 Storage and Handling
Store at 25°C (77°F); excursions permitted to 15° to 30°C (59° to 86°F) [see USP Controlled Room Temperature].
Should freezing occur, gadobutrol injection should be brought to room temperature before use. If allowed to stand at room temperature, gadobutrol injection should return to a clear and colorless to pale yellow solution. Visually inspect gadobutrol injection for particulate matter and discoloration prior to administration. Do not use the solution if it is discolored, if particulate matter is present or if the container appears damaged.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
-
•Hypersensitivity Reactions: Anaphylactic and other hypersensitivity reactions with cardiovascular, respiratory or cutaneous manifestations, ranging from mild to severe, including death, have occurred. Monitor patients closely during and after administration of gadobutrol. (5.3)
-
•Acute Respiratory Distress Syndrome: For patients demonstrating respiratory distress after administration, assess oxygen requirement and monitor for worsening respiratory function. (5.4)
-
•Gadolinium Retention: Gadolinium is retained for months or years in brain, bone, and other organs. (5.5)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Gadobutrol injection is a sterile, clear, and colorless to pale yellow solution for injection containing 604.72 mg gadobutrol per mL (equivalent to 1 mmol gadobutrol/mL).
6.2 Postmarketing Experience
The following additional adverse reactions have been identified during postmarketing use of gadobutrol or other GBCAs. Because these reactions are reported voluntarily from a population of uncertain size, it is not possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
-
•Cardiac arrest
-
•Nephrogenic Systemic Fibrosis (NSF)
-
•Hypersensitivity reactions (anaphylactic shock, circulatory collapse, respiratory arrest, bronchospasm, cyanosis, oropharyngeal swelling, laryngeal edema, blood pressure increased, chest pain, angioedema, conjunctivitis, hyperhidrosis, cough, sneezing, burning sensation, and pallor).
-
•Respiratory, Thoracic, and Mediastinal Disorders: Acute respiratory distress syndrome, pulmonary edema
-
•General Disorders and Administration Site Conditions: Adverse reactions with variable onset and duration have been reported after GBCA administration. These include fatigue, asthenia, pain syndromes, and heterogeneous clusters of symptoms in the neurological, cutaneous, and musculoskeletal systems.
-
•Skin: Gadolinium associated plaques
-
•Gastrointestinal Disorders: Acute pancreatitis with onset within 48 hours after GBCA administration
2.2 Administration Guidelines
Gadobutrol injection is formulated at a higher concentration (1 mmol/mL) compared to certain other gadolinium based contrast agents, resulting in a lower volume of administration. Use Table 1 to determine the volume to be administered.
Use sterile technique when preparing and administering gadobutrol injection.
MRI of the Central Nervous System
-
•Administer gadobutrol injection as an intravenous injection, manually or by power injector, at a flow rate of approximately 2 mL/second.
-
•Follow gadobutrol injection with flush of 0.9% Sodium Chloride Injection, USP to ensure complete administration of the contrast.
-
•Post contrast MRI can commence immediately following contrast administration.
MRI of the Breast
-
•Administer gadobutrol injection as an intravenous bolus by power injector, followed by a flush of 0.9% Sodium Chloride Injection, USP to ensure complete administration of the contrast.
-
•Start image acquisition following contrast administration and then repeat sequentially to determine peak intensity and wash-out.
MR Angiography
Image acquisition should coincide with peak arterial concentration, which varies among patients.
Adults
-
•Administer gadobutrol injection by power injector, at a flow rate of approximately 1.5 mL/second, followed by a 30 mL flush of 0.9% Sodium Chloride Injection, USP at the same rate to ensure complete administration of the contrast.
Pediatric patients
-
•Administer gadobutrol injection by power injector or manually, followed by a flush of 0.9% Sodium Chloride Injection, USP to ensure complete administration of the contrast.
Cardiac MRI
-
•Administer gadobutrol injection through a separate intravenous line in the contralateral arm if concomitantly providing a continuous infusion of a pharmacologic stress agent.
-
•Administer gadobutrol injection as two (2) separate bolus injections: 0.05 mL/kg (0.05 mmol/kg) body weight at peak pharmacologic stress followed by 0.05 mL/kg (0.05 mmol/kg) body weight at rest.
-
•Administer gadobutrol injection via a power injector at a flow rate of approximately 4 mL/second and follow each injection with a flush of 20 mL of 0.9% Sodium Chloride Injection, USP at the same flow rate.
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
Pregnancy: Use only if imaging is essential during pregnancy and cannot be delayed. (8.1)
5.3 Hypersensitivity Reactions
Anaphylactic and other hypersensitivity reactions with cardiovascular, respiratory or cutaneous manifestations, ranging from mild to severe, including death, have uncommonly occurred following gadobutrol administration [see Adverse Reactions (6)].
-
•Before gadobutrol administration, assess all patients for any history of a reaction to contrast media, bronchial asthma and/or allergic disorders. These patients may have an increased risk for a hypersensitivity reaction to gadobutrol.
-
•Administer gadobutrol only in situations where trained personnel and therapies are promptly available for the treatment of hypersensitivity reactions, including personnel trained in resuscitation.
Most hypersensitivity reactions to gadobutrol have occurred within half an hour after administration. Delayed reactions can occur up to several days after administration. Observe patients for signs and symptoms of hypersensitivity reactions during and following gadobutrol administration.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
The adverse reactions described in this section reflect gadobutrol exposure in 7713 subjects (including 184 pediatric patients, ages 0 to 17 years) with the majority receiving the recommended dose. Approximately 52% of the subjects were male and the ethnic distribution was 62% Caucasian, 28% Asian, 5% Hispanic, 2.5% Black, and 2.5% patients of other ethnic groups. The average age was 56 years (range from 1 week to 93 years).
Overall, approximately 4% of subjects reported one or more adverse reactions during a follow-up period that ranged from 24 hours to 7 days after gadobutrol administration.
Adverse reactions associated with the use of gadobutrol were usually mild to moderate in severity and transient in nature.
Table 2 lists adverse reactions that occurred in ≥ 0.1% subjects who received gadobutrol.
|
Reaction |
Rate (%)
|
|
Headache |
1.7 |
|
Nausea |
1.2 |
|
Dizziness |
0.5 |
|
Dysgeusia |
0.4 |
|
Feeling Hot |
0.4 |
|
Injection site reactions |
0.4 |
|
Vomiting |
0.4 |
|
Rash (includes generalized, macular, papular, pruritic) |
0.3 |
|
Erythema |
0.2 |
|
Paresthesia |
0.2 |
|
Pruritus (includes generalized) |
0.2 |
|
Dyspnea |
0.1 |
|
Urticaria |
0.1 |
Adverse reactions that occurred with a frequency of < 0.1% in subjects who received gadobutrol include: hypersensitivity/anaphylactic reaction, loss of consciousness, convulsion, parosmia, tachycardia, palpitation, dry mouth, malaise and feeling cold.
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
-
•Advise the patient to read the FDA-approved patient labeling (Medication Guide).
Nephrogenic Systemic Fibrosis
Instruct patients to inform their physician if they:
-
•Have a history of kidney disease and/or liver disease, or
-
•Have recently received a GBCA
GBCAs increase the risk of NSF among patients with impaired elimination of drugs. To counsel patients at risk of NSF:
-
•Describe the clinical manifestation of NSF
-
•Describe procedures to screen for the detection of renal impairment
Instruct the patients to contact their physician if they develop signs or symptoms of NSF following gadobutrol injection administration, such as burning, itching, swelling, scaling, hardening and tightening of the skin; red or dark patches on the skin; stiffness in joints with trouble moving, bending or straightening the arms, hands, legs or feet; pain in the hip bones or ribs; or muscle weakness.
Common Adverse Reactions
Inform patients that they may experience:
-
•Reactions along the venous injection site, such as mild and transient burning or pain or feeling of warmth or coldness at the injection site
-
•Side effects of headache, nausea, abnormal taste and feeling hot
Acute Respiratory Distress Syndrome
-
•Advise patients that acute respiratory distress syndrome (ARDS) has occurred with gadobutrol injection. Inform patients on the symptoms of the observed ARDS cases, and instruct patients to inform their healthcare provider if they experience these symptoms [see Warnings and Precautions (5.4)].
General Precautions
Gadolinium Retention
-
•Advise patients that gadolinium is retained for months or years in brain, bone, skin, and other organs in patients with normal renal function. The clinical consequences of retention are unknown. Retention depends on multiple factors and is greater following administration of linear GBCAs than following administration of macrocyclic GBCAs [see Warnings and Precautions (5.5)].
Instruct patients receiving gadobutrol injection to inform their physician if they:
-
•Are pregnant or breastfeeding
-
•Have a history of allergic reaction to contrast media, bronchial asthma or allergic respiratory disorder
Manufactured for:
Lake Zurich, IL 60047
www.fresenius-kabi.com/us
Product of China
Revised: May 2025
451796C
26EUF04
5.2 Nephrogenic Systemic Fibrosis
GBCAs increase the risk for nephrogenic systemic fibrosis (NSF) among patients with impaired elimination of the drugs. Avoid use of gadobutrol injection among these patients unless the diagnostic information is essential and not available with non-contrast MRI or other modalities. The GBCA-associated NSF risk appears highest for patients with chronic, severe kidney disease (GFR < 30 mL/min/1.73m2) as well as patients with acute kidney injury. The risk appears lower for patients with chronic, moderate kidney disease (GFR 30 to 59 mL/min/1.73m2) and little, if any, for patients with chronic, mild kidney disease (GFR 60 to 89 mL/min/1.73m2). NSF may result in fatal or debilitating fibrosis affecting the skin, muscle and internal organs. Report any diagnosis of NSF following gadobutrol administration to Fresenius Kabi USA, LLC at 1-800-551-7176 or FDA (1-800-FDA-1088 or www.fda.gov/medwatch).
Screen patients for acute kidney injury and other conditions that may reduce renal function. Features of acute kidney injury consist of rapid (over hours to days) and usually reversible decrease in kidney function, commonly in the setting of surgery, severe infection, injury or drug-induced kidney toxicity. Serum creatinine levels and estimated GFR may not reliably assess renal function in the setting of acute kidney injury. For patients at risk for chronically reduced renal function (for example, age > 60 years, diabetes mellitus or chronic hypertension), estimate the GFR through laboratory testing.
Among the factors that may increase the risk for NSF are repeated or higher than recommended doses of a GBCA and degree of renal impairment at the time of exposure. Record the specific GBCA and the dose administered to a patient. For patients at highest risk for NSF, do not exceed the recommended gadobutrol dose and allow a sufficient period of time for elimination of the drug prior to re-administration. For patients receiving hemodialysis, consider the prompt initiation of hemodialysis following the administration of a GBCA in order to enhance the contrast agent's elimination [see Use in Specific Populations (8.6) and Clinical Pharmacology (12.3)]. The usefulness of hemodialysis in the prevention of NSF is unknown [see Clinical Pharmacology (12.3)].
5.4 Acute Respiratory Distress Syndrome
Acute respiratory distress syndrome (ARDS) has been reported in patients administered gadobutrol injection and may be characterized by severe hypoxemia requiring oxygen support and mechanical ventilation. These manifestations may resemble an immediate hypersensitivity reaction with onset of respiratory distress within <30 minutes to 24 hours after gadobutrol injection administration. For patients demonstrating respiratory distress after gadobutrol injection administration, assess oxygen requirement and monitor for worsening respiratory function.
1.3 Magnetic Resonance Angiography (mra) (1.3 Magnetic Resonance Angiography (MRA))
Gadobutrol injection is indicated for use in magnetic resonance angiography (MRA) in adult and pediatric patients, including term neonates, to evaluate known or suspected supra-aortic or renal artery disease.
5.1 Risk Associated With Intrathecal Use (5.1 Risk Associated with Intrathecal Use)
Intrathecal administration of GBCAs can cause serious adverse reactions including death, coma, encephalopathy, and seizures. The safety and effectiveness of Gadobutrol injection have not been established with intrathecal use. Gadobutrol injection is not approved for intrathecal use [see Dosage and Administration (2.2)].
13.2 Animal Toxicology And/or Pharmacology (13.2 Animal Toxicology and/or Pharmacology)
Local intolerance reactions, including moderate irritation associated with infiltration of inflammatory cells was observed after paravenous administration to rabbits, suggesting the possibility of occurrence of local irritation if the contrast medium leaks around veins in a clinical setting [see Warnings and Precautions (5.7)].
5.7 Extravasation and Injection Site Reactions
Ensure catheter and venous patency before the injection of gadobutrol. Extravasation into tissues during gadobutrol administration may result in moderate irritation [see Nonclinical Toxicology (13.2)].
2.4 Imaging Bulk Package Preparation Instructions
Gadobutrol injection Imaging Bulk Package (IBP) is a container of a sterile preparation for parenteral use that contains many single doses of gadobutrol for use with a medical imaging device. Gadobutrol injection Imaging Bulk Package is for intravenous use and not for direct infusion. Gadobutrol injection Imaging Bulk Package is for use only with an automated contrast injection system, contrast management system, or contrast media transfer set approved or cleared for use with this contrast agent in this Imaging Bulk Package. Please see drug and device labeling for information on devices indicated for use with this Imaging Bulk Package and techniques to help assure safe use.
-
1.The Gadobutrol injection Imaging Bulk Package is to be used only in a room designated for radiological procedures that involve intravascular administration of a contrast agent.
-
2.Utilize aseptic technique for penetrating the container closure of the Gadobutrol injection Imaging Bulk Package and transferring Gadobutrol injection.
-
3.The container closure must be penetrated only one time with a suitable sterile component of the automated contrast injection system, contrast management system, or contrast media transfer set (e.g., transfer spike) approved or cleared for use with this contrast agent in this Imaging Bulk Package.
-
4.Once the Gadobutrol injection Imaging Bulk Package is punctured, do not remove it from the work area during the entire period of use. Storage temperature of Gadobutrol injection Imaging Bulk Package after the closure has been entered is 20°C to 25°C (68°F to 77°F).
-
5.A maximum use time of 24 hours from initial puncture is permitted to complete fluid transfer. Discard any unused Gadobutrol injection 24 hours after initial puncture of the Imaging Bulk Package.
-
6.After the container closure is punctured, if the integrity of the Imaging Bulk Package and the delivery system cannot be assured through direct continuous supervision, the Imaging Bulk Package and all associated disposables for the automated contrast injection system, contrast management system, or contrast media transfer set (e.g., transfer spike) should be discarded.
5.9 Low Sensitivity for Significant Arterial Stenosis
The performance of gadobutrol MRA for detecting arterial segments with significant stenosis (>50% renal, >70% supra-aortic) has not been shown to exceed 55%. Therefore, a negative MRA study alone should not be used to rule out significant stenosis [see Clinical Studies (14.3)].
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
No carcinogenicity studies of gadobutrol have been conducted.
Gadobutrol was not mutagenic in in vitro reverse mutation tests in bacteria, in the HGPRT (hypoxanthine-guanine phosphoribosyl transferase) test using cultured Chinese hamster V79 cells, or in chromosome aberration tests in human peripheral blood lymphocytes, and was negative in an in vivo micronucleus test in mice after intravenous injection of 0.5 mmol/kg.
Gadobutrol had no effect on fertility and general reproductive performance of male and female rats when given in doses 12.2 times the human equivalent dose (based on body surface area).
5.8 Overestimation of Extent of Malignant Disease in Mri of the Breast (5.8 Overestimation of Extent of Malignant Disease in MRI of the Breast)
Gadobutrol MRI of the breast overestimated the histologically confirmed extent of malignancy in the diseased breast in up to 50% of the patients [see Clinical Studies (14.2)].
1.1 Magnetic Resonance Imaging (mri) of the Central Nervous System (cns) (1.1 Magnetic Resonance Imaging (MRI) of the Central Nervous System (CNS))
Gadobutrol injection is indicated for use with magnetic resonance imaging (MRI) in adult and pediatric patients, including term neonates, to detect and visualize areas with disrupted blood brain barrier and/or abnormal vascularity of the central nervous system.
Warning: Risk Associated With Intrathecal Use and Nephrogenic Systemic Fibrosis (WARNING: RISK ASSOCIATED WITH INTRATHECAL USE and NEPHROGENIC SYSTEMIC FIBROSIS)
Risk Associated with Intrathecal Use
Intrathecal administration of gadolinium-based contrast agents (GBCAs) can cause serious adverse reactions including death, coma, encephalopathy, and seizures. Gadobutrol injection is not approved for intrathecal use [see Warnings and Precautions (5.1)].
Nephrogenic Systemic Fibrosis
GBCAs increase the risk for nephrogenic systemic fibrosis (NSF) among patients with impaired elimination of drugs. Avoid use of gadobutrol injection in these patients unless the diagnostic information is essential and not available with non-contrasted MRI or other modalities. NSF may result in fatal or debilitating fibrosis affecting the skin, muscle and internal organs.
-
•The risk for NSF appears highest among patients with:
-
oChronic, severe kidney disease (GFR < 30 mL/min/1.73m2), or
-
oAcute kidney injury.
-
-
•Screen patients for acute kidney injury and other conditions that may reduce renal function. For patients at risk for chronically reduced renal function (for example, age > 60 years, hypertension or diabetes), estimate the glomerular filtration rate (GFR) through laboratory testing.
-
•For patients at highest risk for NSF, do not exceed the recommended gadobutrol dose and allow a sufficient period of time for elimination of the drug from the body prior to any re-administration [see Warnings and Precautions (5.2)].
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:50:21.429660 · Updated: 2026-03-14T22:37:11.582303