bfea2a7d-e77b-b415-e053-2995a90af4c8
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
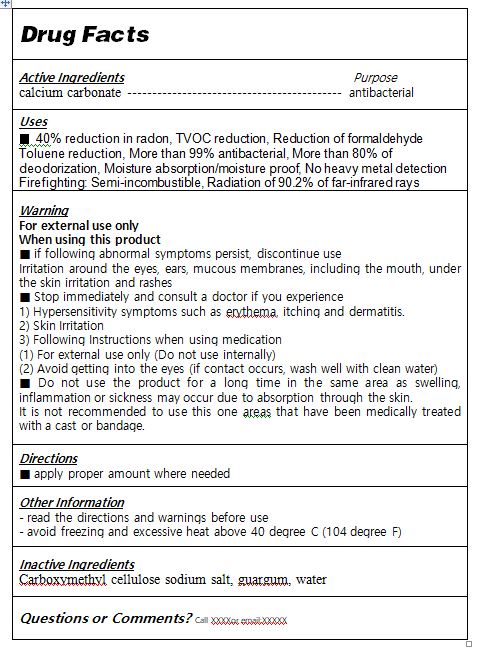
calcium carbonate
Medication Information
Warnings and Precautions
■ if following abnormal symptoms persist, discontinue use
Irritation around the eyes, ears, mucous membranes, including the mouth, under the skin irritation and rashes
■ Stop immediately and consult a doctor if you experience
1) Hypersensitivity symptoms such as erythema, itching and dermatitis.
2) Skin Irritation
3) Following Instructions when using medication
(1) For external use only (Do not use internally)
(2) Avoid getting into the eyes (if contact occurs, wash well with clean water)
■ Be careful not to inhale or use excessively for a long time (ingesting ethanol repeatedly causes irritation to mucous membranes and headaches or other symptoms may appear. When used repeatedly in the same area, skin irritation may occur.
■ Do not use the product for a long time in the same area as swelling, inflammation or sickness may occur due to absorption through the skin.
It is not recommended to use this one areas that have been medically treated with a cast or bandage.
■ Do not use in combination with soap or antibacterial cleansing agents.
Indications and Usage
■ apply proper amount to the skin
Dosage and Administration
for external use only
Description
calcium carbonate
Section 50565-1
• Keep Out of Reach of Children.
Section 51727-6
Carboxymethyl cellulose sodium salt, guargum, water
Section 51945-4
Section 55105-1
40% reduction in radon
TVOC reduction
Reduction of formaldehyde
Toluene reduction
More than 99% antibacterial
More than 80% of deodorization
Moisture absorption/moisture proof
No heavy metal detection
Firefighting: Semi-incombustible
Radiation of 90.2% of far-infrared rays
Section 55106-9
calcium carbonate
Structured Label Content
Indications and Usage (34067-9)
■ apply proper amount to the skin
Dosage and Administration (34068-7)
for external use only
Warnings and Precautions (34071-1)
■ if following abnormal symptoms persist, discontinue use
Irritation around the eyes, ears, mucous membranes, including the mouth, under the skin irritation and rashes
■ Stop immediately and consult a doctor if you experience
1) Hypersensitivity symptoms such as erythema, itching and dermatitis.
2) Skin Irritation
3) Following Instructions when using medication
(1) For external use only (Do not use internally)
(2) Avoid getting into the eyes (if contact occurs, wash well with clean water)
■ Be careful not to inhale or use excessively for a long time (ingesting ethanol repeatedly causes irritation to mucous membranes and headaches or other symptoms may appear. When used repeatedly in the same area, skin irritation may occur.
■ Do not use the product for a long time in the same area as swelling, inflammation or sickness may occur due to absorption through the skin.
It is not recommended to use this one areas that have been medically treated with a cast or bandage.
■ Do not use in combination with soap or antibacterial cleansing agents.
Section 50565-1 (50565-1)
• Keep Out of Reach of Children.
Section 51727-6 (51727-6)
Carboxymethyl cellulose sodium salt, guargum, water
Section 51945-4 (51945-4)
Section 55105-1 (55105-1)
40% reduction in radon
TVOC reduction
Reduction of formaldehyde
Toluene reduction
More than 99% antibacterial
More than 80% of deodorization
Moisture absorption/moisture proof
No heavy metal detection
Firefighting: Semi-incombustible
Radiation of 90.2% of far-infrared rays
Section 55106-9 (55106-9)
calcium carbonate
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:00:27.875766 · Updated: 2026-03-14T23:00:54.830446