aftaseptic analgesic liquid
beb32fba-503a-f5e5-e053-2a95a90a57e5
34390-5
HUMAN OTC DRUG LABEL
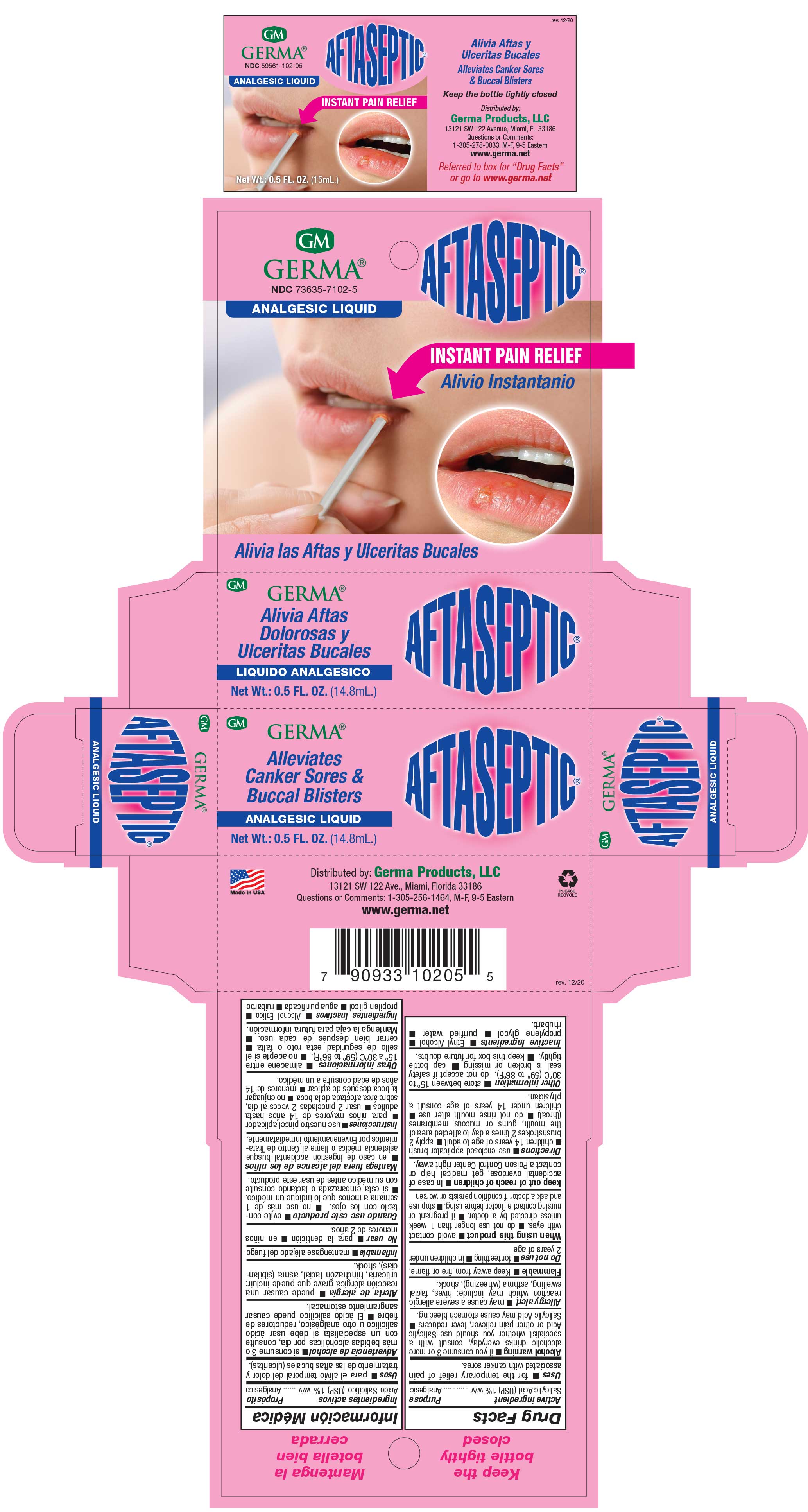
Drug Facts
Composition & Product
Identifiers & Packaging
Purpose
Analgesic
Description
Salicylic Acid 1% w/v
Medication Information
Warnings
Alcohol warning: if you consume 3 or more alcoholic drinks everyday, consult with a specialist whether you should use Salicylic Acid or other pain reliever, fever reducers n Salicylic Acid may cause stomach bleeding.
Allergy alert: may cause a severe allergic reaction which may include: hives, facial swelling, asthma (wheezing), shock.
Uses
For the temporary relief of pain associated with canker sores.
Indications and Usage
for the temporary relief of pain associated with canker sores.
Purpose
Analgesic
Directions
Use enclosed applicator brush
Children 14 years of age to adult. Apply 2 brushstrokes 2 times a day to affected area of the mouth, gums or mucous membranes (throat). Do not rinse mouth after use. Children under 14 years of age consult a physician.
Other Information
Store between 15º to 30ºC (59º to 86ºF). Cap bottle tightly. Do not accept if safety seal is broken or missing. Cap bottle tightly. Keep this box for future doubts.
Inactive Ingredients
Ethyl Alcohol, propylene glycol, purified water, rhubarb.
Description
Salicylic Acid 1% w/v
Section 51945-4
Aftaseptic
Active Ingredient
Salicylic Acid 1% w/v
Structured Label Content
Warnings
Alcohol warning: if you consume 3 or more alcoholic drinks everyday, consult with a specialist whether you should use Salicylic Acid or other pain reliever, fever reducers n Salicylic Acid may cause stomach bleeding.
Allergy alert: may cause a severe allergic reaction which may include: hives, facial swelling, asthma (wheezing), shock.
Uses
For the temporary relief of pain associated with canker sores.
Indications and Usage (34067-9)
for the temporary relief of pain associated with canker sores.
Purpose
Analgesic
Directions
Use enclosed applicator brush
Children 14 years of age to adult. Apply 2 brushstrokes 2 times a day to affected area of the mouth, gums or mucous membranes (throat). Do not rinse mouth after use. Children under 14 years of age consult a physician.
Other Information
Store between 15º to 30ºC (59º to 86ºF). Cap bottle tightly. Do not accept if safety seal is broken or missing. Cap bottle tightly. Keep this box for future doubts.
Inactive Ingredients
Ethyl Alcohol, propylene glycol, purified water, rhubarb.
Section 51945-4 (51945-4)
Aftaseptic
Active Ingredient
Salicylic Acid 1% w/v
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:03:49.727955 · Updated: 2026-03-14T23:13:00.870746