Levonorgestrel Tablet 1.5 Mg
be8b7ace-9af8-4417-85fa-863e19072887
34390-5
HUMAN OTC DRUG LABEL
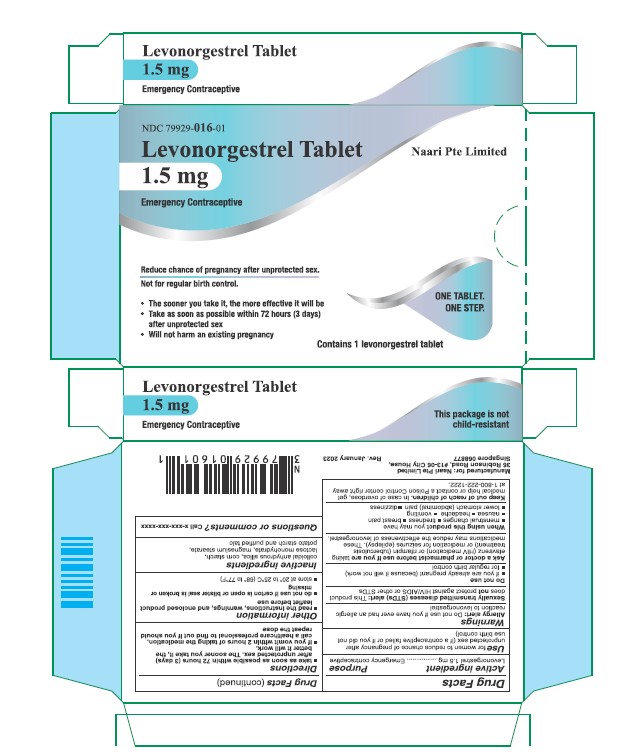
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Drug Facts
Purpose
Emergency contraceptive
Medication Information
Purpose
Emergency contraceptive
Description
Drug Facts
Use
for women to reduce chance of pregnancy after unprotected sex (if a contraceptive failed or if you did not use birth control)
Section 42229-5
Drug Facts
Section 50565-1
Keep out of reach of children. In case of overdose, get medical help or contact a Poison Control center right away.
Section 50567-7
When using this product you may have
- menstrual changes
- tiredness
- breast pain
- nausea
- headache
- vomiting
- lower stomach (abdominal) pain
- dizziness
Section 50568-5
Ask a doctor or pharmacist before use if you are taking efavirenz (HIV medication) or rifampin (tuberculosis treatment) or medication for seizures (epilepsy). These medications may reduce the effectiveness of levonorgestrel.
Directions
- take as soon as possible within 72 hours (3 days) after unprotected sex. The sooner you take it the better it will work.
- if you vomit within 2 hours after taking the medication, call a healthcare professional to find out if you should repeat the dose
Do Not Use
- if you are already pregnant (because it will not work)
- for regular birth control
Questions?
If you have questions or need more information, call our toll-free number, x-xxx-xxx-xxxx.
Allergy Alert
Do not use if you have ever had an allergic reaction to levonorgestrel
Active Ingredient
Levonorgestrel 1.5 mg
Other Information
- read the instructions, warnings and enclosed product leaflet before use
- do not use if carton is open or tear strip is removed or blister seal is broken or missing
- store at 20-25°C (68-77°F)
Inactive Ingredients
colloidal anhydrous silica, corn starch, lactose monohydrate, magnesium stearate, potato starch and purified talc
Principal Display Panel
FOR CLINIC USE ONLY.
NOT FOR RETAIL SALE.
NDC 79929-016-01
levonorgestrel 1.5 mg
emergency contraceptive
Reduces chance of
pregnancy after
unprotected sex.
Not for regular birth control.
One Tablet.
One Step.
1 Tablet Levonorgestrel 1.5 mg
Sexually Transmitted Diseases (stds) Alert
This product does not protect against HIV/AIDS or other STDs
Structured Label Content
Use
for women to reduce chance of pregnancy after unprotected sex (if a contraceptive failed or if you did not use birth control)
Section 42229-5 (42229-5)
Drug Facts
Section 50565-1 (50565-1)
Keep out of reach of children. In case of overdose, get medical help or contact a Poison Control center right away.
Section 50567-7 (50567-7)
When using this product you may have
- menstrual changes
- tiredness
- breast pain
- nausea
- headache
- vomiting
- lower stomach (abdominal) pain
- dizziness
Section 50568-5 (50568-5)
Ask a doctor or pharmacist before use if you are taking efavirenz (HIV medication) or rifampin (tuberculosis treatment) or medication for seizures (epilepsy). These medications may reduce the effectiveness of levonorgestrel.
Purpose
Emergency contraceptive
Directions
- take as soon as possible within 72 hours (3 days) after unprotected sex. The sooner you take it the better it will work.
- if you vomit within 2 hours after taking the medication, call a healthcare professional to find out if you should repeat the dose
Do Not Use (Do not use)
- if you are already pregnant (because it will not work)
- for regular birth control
Questions?
If you have questions or need more information, call our toll-free number, x-xxx-xxx-xxxx.
Allergy Alert (Allergy alert)
Do not use if you have ever had an allergic reaction to levonorgestrel
Active Ingredient (Active ingredient)
Levonorgestrel 1.5 mg
Other Information (Other information)
- read the instructions, warnings and enclosed product leaflet before use
- do not use if carton is open or tear strip is removed or blister seal is broken or missing
- store at 20-25°C (68-77°F)
Inactive Ingredients (Inactive ingredients)
colloidal anhydrous silica, corn starch, lactose monohydrate, magnesium stearate, potato starch and purified talc
Principal Display Panel (PRINCIPAL DISPLAY PANEL )
FOR CLINIC USE ONLY.
NOT FOR RETAIL SALE.
NDC 79929-016-01
levonorgestrel 1.5 mg
emergency contraceptive
Reduces chance of
pregnancy after
unprotected sex.
Not for regular birth control.
One Tablet.
One Step.
1 Tablet Levonorgestrel 1.5 mg
Sexually Transmitted Diseases (stds) Alert (Sexually transmitted diseases (STDs) alert)
This product does not protect against HIV/AIDS or other STDs
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:05:58.722659 · Updated: 2026-03-14T23:08:25.015175