Onelax
be4eeb4d-df29-45dc-94ff-933e52c8b6f6
34390-5
HUMAN OTC DRUG LABEL
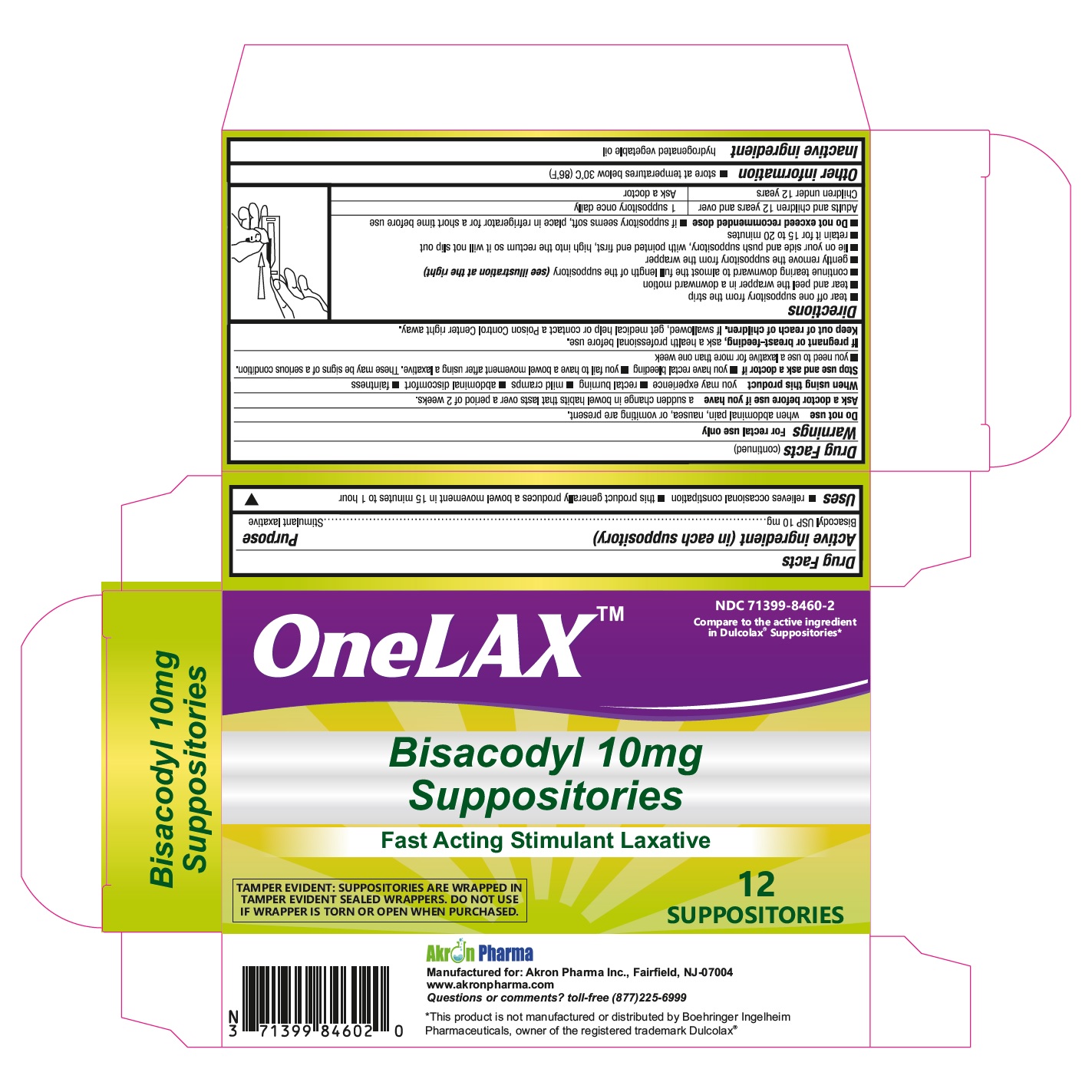
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Bisacodyl USP, 10 mg
Purpose
Laxative
Medication Information
Purpose
Laxative
Description
Bisacodyl USP, 10 mg
Uses
- for relief of occasional constipation
- this product usually produces bowel movement in 1/4 to 1 hour
Section 50565-1
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center right away.
Section 51945-4
Warnings
For rectal use only.
Directions
- adults and children 12 years of age and older
- detach one suppository from the strip
- remove from wrapper before inserting into the rectum
- the rectal suppository dose is one suppository per day or as directed by a doctor
- children under 12 years of age consult a doctor
Other Information
store below 30° C (86° F)
Inactive Ingredient
hydrogenated vegetable oil
Manufactured for:
Akron Pharma Inc.,
Fairfield, NJ-07004
www.akronpharma.com
When Using This Product
it may cause abdominal discomfort, faintness, rectal burning and mild cramps
Do Not Use Laxative Products
- when abdominal pain, nausea or vomiting are present
- for a period longer than one week
Stop Use and Ask A Doctor If
rectal bleeding occurs, or you fail to have a bowel movement after using a laxative. This may indicate a serious condition.
If Pregnant Or Breast Feeding
ask a health professional before use.
Ask A Doctor Before Use If You Have
noticed a sudden change in bowel habits that persist over a period of 2 weeks
Active Ingredients (per Suppository)
Bisacodyl USP, 10 mg
Structured Label Content
Uses
- for relief of occasional constipation
- this product usually produces bowel movement in 1/4 to 1 hour
Section 50565-1 (50565-1)
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center right away.
Section 51945-4 (51945-4)
Purpose
Laxative
Warnings
For rectal use only.
Directions
- adults and children 12 years of age and older
- detach one suppository from the strip
- remove from wrapper before inserting into the rectum
- the rectal suppository dose is one suppository per day or as directed by a doctor
- children under 12 years of age consult a doctor
Other Information (Other information)
store below 30° C (86° F)
Inactive Ingredient (Inactive ingredient)
hydrogenated vegetable oil
Manufactured for:
Akron Pharma Inc.,
Fairfield, NJ-07004
www.akronpharma.com
When Using This Product (When using this product)
it may cause abdominal discomfort, faintness, rectal burning and mild cramps
Do Not Use Laxative Products (Do not use laxative products)
- when abdominal pain, nausea or vomiting are present
- for a period longer than one week
Stop Use and Ask A Doctor If (Stop use and ask a doctor if)
rectal bleeding occurs, or you fail to have a bowel movement after using a laxative. This may indicate a serious condition.
If Pregnant Or Breast Feeding (If pregnant or breast-feeding)
ask a health professional before use.
Ask A Doctor Before Use If You Have (Ask a doctor before use if you have)
noticed a sudden change in bowel habits that persist over a period of 2 weeks
Active Ingredients (per Suppository) (Active Ingredients (per suppository))
Bisacodyl USP, 10 mg
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:03:36.588678 · Updated: 2026-03-14T23:13:25.872506