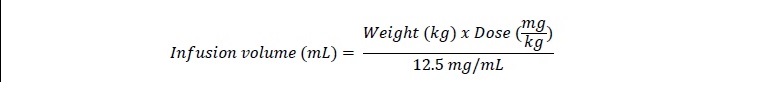These Highlights Do Not Include All The Information Needed To Use Recarbrio Safely And Effectively. See Full Prescribing Information For Recarbrio.
be32f468-738b-40df-a425-591e12f65159
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Indications and Usage ( 1 ) 12/2025 Dosage and Administration ( 2 ) 12/2025
Indications and Usage
RECARBRIO is a combination of imipenem, a penem antibacterial, cilastatin, a renal dehydropeptidase inhibitor, and relebactam, a beta-lactamase inhibitor, indicated in adult and pediatric patients weighing at least 2 kg for the treatment of the following infections caused by susceptible gram-negative microorganisms: Hospital-acquired bacterial pneumonia and ventilator-associated bacterial pneumonia (HABP/VABP). ( 1.1 ) Complicated urinary tract infections, including pyelonephritis (cUTI) in patients who have limited or no alternative treatment options. ( 1.2 ) Complicated intra-abdominal infections (cIAI) in patients who have limited or no alternative treatment options. ( 1.3 ) Approval of the cUTI and cIAI indications is based on limited clinical safety and efficacy data for RECARBRIO. ( 1.2 , 1.3 , 14 ) Limitations of Use RECARBRIO is not recommended in pediatric patients less than 37 weeks post-menstrual age (gestational age at birth plus post-natal age). ( 1.4 , 2.2 ). RECARBRIO is not recommended in pediatric patients weighing less than 30 kg with renal impairment. ( 1.4 , 2.3 ). Usage to Reduce Development of Drug-Resistant Bacteria To reduce the development of drug-resistant bacteria and maintain the effectiveness of RECARBRIO and other antibacterial drugs, RECARBRIO should be used only to treat or prevent infections that are proven or strongly suspected to be caused by bacteria. ( 1.5 )
Dosage and Administration
Adult Patients: The recommended dosage of RECARBRIO is 1.25 grams (imipenem 500 mg, cilastatin 500 mg, relebactam 250 mg) administered by intravenous (IV) infusion over 30 minutes every 6 hours to adult patients with creatinine clearance (CLcr) 90 mL/min or greater. ( 2.1 ) Pediatric Patients weighing at least 2 kg: The recommended dosage of RECARBRIO for pediatric patients weighing at least 2kg varies by patient weight and age, with specific dosing recommendations as shown in the table below, ( 2.2 ): Recommended Dosage of RECARBRIO In Pediatric Patients Weighing at Least 2 kg Age Range Body Weight Dose Dosing Frequency Infusion Duration Birth Pediatric patients from birth (includes pediatric patients at least 37 weeks post-menstrual age) to less than 3 months 2 kg or greater RECARBRIO 37.5 Provides 15 mg/kg imipenem, 15 mg/kg cilastatin, and 7.5 mg/kg relebactam mg/kg Every 8 hours 60 minutes 3 months to less than 18 years less than 30 kg RECARBRIO 37.5 mg/kg Every 6 hours 60 minutes 3 months to less than 18 years 30 kg or greater RECARBRIO 1.25 Provides 500 mg imipenem, 500 mg cilastatin, and 250 mg, relebactam grams Every 6 hours 30 minutes Dose reduction is required in adult and pediatric (weighing at least 30 kg) patients with renal impairment. ( 2.3 , 2.4 ) Do not administer RECARBRIO to adults with CLcr less than 15 mL/min unless hemodialysis is instituted within 48 hours after dose administration. ( 2.3 ) Do not administer RECARBRIO to pediatric patients weighing at least 30 kg with an eGFR less than 15 mL/min/1.73m 2 unless hemodialysis is instituted within 48 hours after dose administration. ( 2.4 ) See Full Prescribing Information for instructions for constituting supplied dry powder and subsequent required dilution. ( 2.5 ) See Full Prescribing Information for drug compatibilities and incompatibilities. ( 2.7 , 2.8 )
Warnings and Precautions
Hypersensitivity Reactions: Serious and occasionally fatal hypersensitivity (anaphylactic) reactions have been reported in patients receiving beta lactam drugs. Discontinue RECARBRIO immediately if a hypersensitivity reaction occurs. ( 5.1 ) Seizures and Central Nervous System Adverse Reactions: CNS adverse reactions such as seizures have been reported with imipenem/cilastatin, a component of RECARBRIO. If focal tremors, myoclonus, or seizures occur, evaluate patients, to determine whether RECARBRIO should be discontinued. ( 5.2 ) Increased Seizure Potential Due to Interaction with Valproic Acid: Concomitant use of RECARBRIO with valproic acid or divalproex sodium may reduce the serum concentration of valproic acid which may increase the risk of breakthrough seizures. Avoid concomitant use or consider alternative antibacterial drugs other than carbapenems. ( 5.3 , 7.2 ) Clostridioides difficile -Associated Diarrhea (CDAD): Has been reported with RECARBRIO. Evaluate if diarrhea occurs. ( 5.4 )
Contraindications
RECARBRIO is contraindicated in patients with a history of known severe hypersensitivity (severe systemic allergic reaction such as anaphylaxis) to any component of RECARBRIO.
Adverse Reactions
CNS adverse reactions, such as seizures, confusional states, and myoclonic activity, have been reported during treatment with imipenem/cilastatin, a component of RECARBRIO, especially when recommended dosages of imipenem were exceeded . These have been reported most commonly in patients with CNS disorders (e.g., brain lesions or history of seizures) and/or compromised renal function . Anticonvulsant therapy should be continued in patients with known seizure disorders. If CNS adverse reactions including seizures occur, patients should undergo a neurological evaluation to determine whether RECARBRIO should be discontinued.
Drug Interactions
Ganciclovir: Avoid concomitant use. ( 7.1 ) Valproic Acid or Divalproex Sodium: Avoid concomitant use. ( 7.2 )
Storage and Handling
RECARBRIO (imipenem, cilastatin, and relebactam) for injection, 1.25 grams is supplied as a white to light yellow sterile powder for constitution in a single-dose glass vial containing imipenem 500 mg (equivalent to 530 mg imipenem monohydrate), cilastatin 500 mg (equivalent to 531 mg cilastatin sodium), and relebactam 250 mg (equivalent to 263 mg relebactam monohydrate). The vials are supplied as a single-dose glass vial (NDC 0006-3856-01) and in cartons containing 25 vials (NDC 0006-3856-02).
How Supplied
RECARBRIO (imipenem, cilastatin, and relebactam) for injection, 1.25 grams is supplied as a white to light yellow sterile powder for constitution in a single-dose glass vial containing imipenem 500 mg (equivalent to 530 mg imipenem monohydrate), cilastatin 500 mg (equivalent to 531 mg cilastatin sodium), and relebactam 250 mg (equivalent to 263 mg relebactam monohydrate). The vials are supplied as a single-dose glass vial (NDC 0006-3856-01) and in cartons containing 25 vials (NDC 0006-3856-02).
Medication Information
Warnings and Precautions
Hypersensitivity Reactions: Serious and occasionally fatal hypersensitivity (anaphylactic) reactions have been reported in patients receiving beta lactam drugs. Discontinue RECARBRIO immediately if a hypersensitivity reaction occurs. ( 5.1 ) Seizures and Central Nervous System Adverse Reactions: CNS adverse reactions such as seizures have been reported with imipenem/cilastatin, a component of RECARBRIO. If focal tremors, myoclonus, or seizures occur, evaluate patients, to determine whether RECARBRIO should be discontinued. ( 5.2 ) Increased Seizure Potential Due to Interaction with Valproic Acid: Concomitant use of RECARBRIO with valproic acid or divalproex sodium may reduce the serum concentration of valproic acid which may increase the risk of breakthrough seizures. Avoid concomitant use or consider alternative antibacterial drugs other than carbapenems. ( 5.3 , 7.2 ) Clostridioides difficile -Associated Diarrhea (CDAD): Has been reported with RECARBRIO. Evaluate if diarrhea occurs. ( 5.4 )
Indications and Usage
RECARBRIO is a combination of imipenem, a penem antibacterial, cilastatin, a renal dehydropeptidase inhibitor, and relebactam, a beta-lactamase inhibitor, indicated in adult and pediatric patients weighing at least 2 kg for the treatment of the following infections caused by susceptible gram-negative microorganisms: Hospital-acquired bacterial pneumonia and ventilator-associated bacterial pneumonia (HABP/VABP). ( 1.1 ) Complicated urinary tract infections, including pyelonephritis (cUTI) in patients who have limited or no alternative treatment options. ( 1.2 ) Complicated intra-abdominal infections (cIAI) in patients who have limited or no alternative treatment options. ( 1.3 ) Approval of the cUTI and cIAI indications is based on limited clinical safety and efficacy data for RECARBRIO. ( 1.2 , 1.3 , 14 ) Limitations of Use RECARBRIO is not recommended in pediatric patients less than 37 weeks post-menstrual age (gestational age at birth plus post-natal age). ( 1.4 , 2.2 ). RECARBRIO is not recommended in pediatric patients weighing less than 30 kg with renal impairment. ( 1.4 , 2.3 ). Usage to Reduce Development of Drug-Resistant Bacteria To reduce the development of drug-resistant bacteria and maintain the effectiveness of RECARBRIO and other antibacterial drugs, RECARBRIO should be used only to treat or prevent infections that are proven or strongly suspected to be caused by bacteria. ( 1.5 )
Dosage and Administration
Adult Patients: The recommended dosage of RECARBRIO is 1.25 grams (imipenem 500 mg, cilastatin 500 mg, relebactam 250 mg) administered by intravenous (IV) infusion over 30 minutes every 6 hours to adult patients with creatinine clearance (CLcr) 90 mL/min or greater. ( 2.1 ) Pediatric Patients weighing at least 2 kg: The recommended dosage of RECARBRIO for pediatric patients weighing at least 2kg varies by patient weight and age, with specific dosing recommendations as shown in the table below, ( 2.2 ): Recommended Dosage of RECARBRIO In Pediatric Patients Weighing at Least 2 kg Age Range Body Weight Dose Dosing Frequency Infusion Duration Birth Pediatric patients from birth (includes pediatric patients at least 37 weeks post-menstrual age) to less than 3 months 2 kg or greater RECARBRIO 37.5 Provides 15 mg/kg imipenem, 15 mg/kg cilastatin, and 7.5 mg/kg relebactam mg/kg Every 8 hours 60 minutes 3 months to less than 18 years less than 30 kg RECARBRIO 37.5 mg/kg Every 6 hours 60 minutes 3 months to less than 18 years 30 kg or greater RECARBRIO 1.25 Provides 500 mg imipenem, 500 mg cilastatin, and 250 mg, relebactam grams Every 6 hours 30 minutes Dose reduction is required in adult and pediatric (weighing at least 30 kg) patients with renal impairment. ( 2.3 , 2.4 ) Do not administer RECARBRIO to adults with CLcr less than 15 mL/min unless hemodialysis is instituted within 48 hours after dose administration. ( 2.3 ) Do not administer RECARBRIO to pediatric patients weighing at least 30 kg with an eGFR less than 15 mL/min/1.73m 2 unless hemodialysis is instituted within 48 hours after dose administration. ( 2.4 ) See Full Prescribing Information for instructions for constituting supplied dry powder and subsequent required dilution. ( 2.5 ) See Full Prescribing Information for drug compatibilities and incompatibilities. ( 2.7 , 2.8 )
Contraindications
RECARBRIO is contraindicated in patients with a history of known severe hypersensitivity (severe systemic allergic reaction such as anaphylaxis) to any component of RECARBRIO.
Adverse Reactions
CNS adverse reactions, such as seizures, confusional states, and myoclonic activity, have been reported during treatment with imipenem/cilastatin, a component of RECARBRIO, especially when recommended dosages of imipenem were exceeded . These have been reported most commonly in patients with CNS disorders (e.g., brain lesions or history of seizures) and/or compromised renal function . Anticonvulsant therapy should be continued in patients with known seizure disorders. If CNS adverse reactions including seizures occur, patients should undergo a neurological evaluation to determine whether RECARBRIO should be discontinued.
Drug Interactions
Ganciclovir: Avoid concomitant use. ( 7.1 ) Valproic Acid or Divalproex Sodium: Avoid concomitant use. ( 7.2 )
Storage and Handling
RECARBRIO (imipenem, cilastatin, and relebactam) for injection, 1.25 grams is supplied as a white to light yellow sterile powder for constitution in a single-dose glass vial containing imipenem 500 mg (equivalent to 530 mg imipenem monohydrate), cilastatin 500 mg (equivalent to 531 mg cilastatin sodium), and relebactam 250 mg (equivalent to 263 mg relebactam monohydrate). The vials are supplied as a single-dose glass vial (NDC 0006-3856-01) and in cartons containing 25 vials (NDC 0006-3856-02).
How Supplied
RECARBRIO (imipenem, cilastatin, and relebactam) for injection, 1.25 grams is supplied as a white to light yellow sterile powder for constitution in a single-dose glass vial containing imipenem 500 mg (equivalent to 530 mg imipenem monohydrate), cilastatin 500 mg (equivalent to 531 mg cilastatin sodium), and relebactam 250 mg (equivalent to 263 mg relebactam monohydrate). The vials are supplied as a single-dose glass vial (NDC 0006-3856-01) and in cartons containing 25 vials (NDC 0006-3856-02).
Description
Indications and Usage ( 1 ) 12/2025 Dosage and Administration ( 2 ) 12/2025
Section 42229-5
Adult Patients
Overview of the Safety Evaluation of RECARBRIO in Adult Patients
Safety was primarily evaluated in three active-controlled, double-blind trials in HABP/VABP, cUTI, and cIAI (Trials 1, 2, and 3, respectively).
In the HABP/VABP trial (Trial 1), patients were treated with either RECARBRIO or piperacillin and tazobactam (4.5 grams).
In the cUTI trial (Trial 2) and cIAI trial (Trial 3), patients in the treatment arms were treated with either imipenem 500 mg/cilastatin 500 mg and relebactam 250 mg or imipenem 500 mg/cilastatin 500 mg and relebactam 125 mg (not an approved dose), and patients in the control arm were treated with imipenem 500 mg/cilastatin 500 mg plus placebo (IV normal saline). Across Trials 2 and 3, the mean duration of IV therapy in patients treated with imipenem/cilastatin plus relebactam 250 mg was approximately 7 days.
Clinical Trial Experience in Adult Patients with HABP/VABP
Trial 1 included 266 adult patients treated with RECARBRIO and 269 patients treated with piperacillin and tazobactam (4.5 grams) administered intravenously over 30 minutes every 6 hours. The mean age was 60 years, 43% of patients were 65 years of age and older, 31% were female and 22% had polymicrobial infection. The mean Acute Physiology and Chronic Health Evaluation (APACHE) II score was 15 and 48% of patients had an APACHE II score greater than or equal to 15 at baseline. Overall, 260 (49%) patients were ventilated at enrollment, including 194 (36%) patients with VABP and 66 (12%) patients with ventilated HABP.
Section 44425-7
Store RECARBRIO vials at 20°C to 25°C (68°F to 77°F), excursions permitted between 15°C to 30°C (between 59°F to 86°F) [see USP Controlled Room Temperature]. Keep vials in the carton.
10 Overdosage
In the event of overdose, discontinue RECARBRIO, treat symptomatically, and institute general supportive treatment. Imipenem, cilastatin, and relebactam can be removed by hemodialysis [see Clinical Pharmacology (12.3)]. No clinical information is available on the use of hemodialysis to treat overdosage.
11 Description
RECARBRIO (imipenem, cilastatin, and relebactam) for injection is an antibacterial combination product consisting of imipenem, a carbapenem antibacterial drug, cilastatin, a renal dehydropeptidase inhibitor, and relebactam, a diazabicyclooctane beta-lactamase inhibitor, for intravenous administration.
7.1 Ganciclovir
Generalized seizures have been reported in patients who received ganciclovir concomitantly with imipenem/cilastatin, a component of RECARBRIO. Ganciclovir should not be used concomitantly with RECARBRIO unless the potential benefits outweigh the risks.
7.2 Valproic Acid
Based on case reports in the literature concomitant use of carbapenems, including imipenem/cilastatin, components of RECARBRIO, with valproic acid or divalproex sodium may decrease valproic acid concentrations which may increase the risk of breakthrough seizures [see Warnings and Precautions (5.3)]. Although the mechanism of this interaction is unknown, data from in vitro and animal studies suggest that carbapenems may inhibit the hydrolysis of valproic acid's glucuronide metabolite (VPA-g) back to valproic acid, thus decreasing the serum concentrations of valproic acid. Avoid concomitant use of RECARBRIO with valproic acid or divalproex sodium. Consider alternative antibacterials other than carbapenems to treat infections in patients whose seizures are well controlled on valproic acid or divalproex sodium.
8.4 Pediatric Use
The safety and effectiveness of RECARBRIO for the treatment of HABP/VABP, and for the treatment of cUTI and cIAI in patients who have limited or no alternative treatment options have been established in pediatric patients weighing at least 2 kg. Use of RECARBRIO in pediatric patients is supported by evidence from an adequate and well-controlled trial of RECARBRIO in adults with HABP/VABP, controlled trials in adults with cUTI and cIAI, and additional pharmacokinetic, safety, and efficacy data from pediatric trials [see Clinical Pharmacology (12.3) and Clinical Studies (14.1, 14.2 and 14.3)].
The safety profile of RECARBRIO in pediatric patients from the pediatric trials was comparable to that in adults treated with RECARBRIO [see Adverse Reactions (6.1)].
RECARBRIO is not recommended in pediatric patients less than 37 weeks post-menstrual age (gestational age at birth plus post-natal age) or weighing less than 30 kg with renal impairment.
The safety and effectiveness of RECARBRIO for the treatment of HABP/VABP, cUTI or cIAI have not been established in pediatric patients weighing less than 2 kg.
8.5 Geriatric Use
Of the 266 patients treated with RECARBRIO in Trial 1, 113 (42.5%) were 65 years of age or older, including 55 (20.7%) patients 75 years of age and older. Of the 216 patients treated with imipenem/cilastatin plus relebactam 250 mg in Trials 2 and 3, 67 (31.0%) were 65 years of age or older, including 25 (11.6%) patients 75 years of age and older. No overall differences in safety or effectiveness were observed between these patients and younger patients, and other reported clinical experience has not identified differences in responses between the elderly and younger patients, but greater sensitivity of some older individuals cannot be ruled out.
RECARBRIO is known to be substantially excreted by the kidney, and the risk of adverse reactions to this drug may be greater in patients with impaired renal function. Because elderly patients are more likely to have decreased renal function, care should be taken in dose selection, and it may be useful to monitor renal function. No dosage adjustment is required based on age. Dosage adjustment for elderly patients should be based on renal function [see Dosage and Administration (2.2) and Clinical Pharmacology (12.3)].
4 Contraindications
RECARBRIO is contraindicated in patients with a history of known severe hypersensitivity (severe systemic allergic reaction such as anaphylaxis) to any component of RECARBRIO.
6 Adverse Reactions
The following serious adverse reactions are described in greater detail in the Warnings and Precautions section.
- Hypersensitivity Reactions [see Warnings and Precautions (5.1)]
- Seizures and Other Central Nervous System Adverse Reactions [see Warnings and Precautions (5.2)]
- Increased Seizure Potential Due to Interaction with Valproic Acid [see Warnings and Precautions (5.3)]
- Clostridioides difficile-Associated Diarrhea (CDAD) [see Warnings and Precautions (5.4)]
7 Drug Interactions
8.6 Renal Impairment
Reduce RECARBRIO dosage in adult patients with a CLcr less than 90 mL/min and in pediatric patients weighing at least 30 kg with an eGFR less than 90 mL/min/1.73 m2 [see Dosage and Administration (2.3, 2.4) and Clinical Pharmacology (12.3)]. RECARBRIO is not recommended in pediatric patients weighing less than 30 kg with renal impairment.
12.2 Pharmacodynamics
For imipenem, the % time of dosing interval that unbound plasma concentrations of imipenem exceed the imipenem/relebactam minimum inhibitory concentration (MIC) (%fT>MIC) against the infecting organism best correlates with antibacterial activity in animal and in vitro models of infection. For relebactam the ratio of the 24-hour unbound plasma relebactam AUC to imipenem/relebactam MIC (fAUC0–24hr/MIC) best predicts the activity of relebactam in animal and in vitro models of infection.
12.3 Pharmacokinetics
The steady-state pharmacokinetic parameters of imipenem, cilastatin, and relebactam in healthy adults with normal renal function (CLcr 90 mL/min or greater), after multiple 30-minute intravenous infusions of RECARBRIO administered every 6 hours are summarized in Table 7. Pharmacokinetic parameters were similar for single and multiple dose administration due to minimal accumulation. Pharmacokinetic parameters in pediatric patients are described in Table 9.
Adult Patients
The steady-state pharmacokinetic parameters of imipenem and relebactam in patients with active bacterial infection with CLcr 90 mL/min or greater following administration of the recommended dosage are summarized in Table 7.
| PK Parameters | cUTI/cIAI Patients | HABP/VABP Patients | |
|---|---|---|---|
| AUC0-24hr=area under the concentration time curve from 0 to 24 hours Cmax=maximum concentration CL=plasma clearance |
|||
| Imipenem | AUC0-24hr (µM-hr) | 570.6 (253.3) | 771 (342.3) |
| Cmax (µM) | 116.1 (52.4) | 122.7 (56.8) | |
| CL (L/hr) | 14 (6.1) | 10.4 (4.5) | |
| Relebactam | AUC0-24hr (µM-hr) | 415.8 (212.6) | 692.9 (354.3) |
| Cmax (µM) | 62.1 (24.7) | 80 (33.3) | |
| CL (L/hr) | 8.7 (4.5) | 5.2 (2.7) |
1.4 Limitations of Use
- RECARBRIO is not recommended in pediatric patients less than 37 weeks post-menstrual age (gestational age at birth plus post-natal age) [see Dosage and Administration (2.2) and Use in Specific Populations (8.4)].
- RECARBRIO is not recommended in pediatric patients weighing less than 30 kg with renal impairment [see Dosage and Administration (2.4), Use in Specific Populations (8.4, 8.6) and Clinical Pharmacology (12.3)].
1 Indications and Usage
RECARBRIO is a combination of imipenem, a penem antibacterial, cilastatin, a renal dehydropeptidase inhibitor, and relebactam, a beta-lactamase inhibitor, indicated in adult and pediatric patients weighing at least 2 kg for the treatment of the following infections caused by susceptible gram-negative microorganisms:
- Hospital-acquired bacterial pneumonia and ventilator-associated bacterial pneumonia (HABP/VABP). (1.1)
- Complicated urinary tract infections, including pyelonephritis (cUTI) in patients who have limited or no alternative treatment options. (1.2)
- Complicated intra-abdominal infections (cIAI) in patients who have limited or no alternative treatment options. (1.3)
Approval of the cUTI and cIAI indications is based on limited clinical safety and efficacy data for RECARBRIO. (1.2, 1.3, 14)
Limitations of Use
- RECARBRIO is not recommended in pediatric patients less than 37 weeks post-menstrual age (gestational age at birth plus post-natal age). (1.4, 2.2).
- RECARBRIO is not recommended in pediatric patients weighing less than 30 kg with renal impairment. (1.4, 2.3).
Usage to Reduce Development of Drug-Resistant Bacteria
To reduce the development of drug-resistant bacteria and maintain the effectiveness of RECARBRIO and other antibacterial drugs, RECARBRIO should be used only to treat or prevent infections that are proven or strongly suspected to be caused by bacteria. (1.5)
12.1 Mechanism of Action
RECARBRIO is an antibacterial drug [see Microbiology (12.4)].
5 Warnings and Precautions
- Hypersensitivity Reactions: Serious and occasionally fatal hypersensitivity (anaphylactic) reactions have been reported in patients receiving beta lactam drugs. Discontinue RECARBRIO immediately if a hypersensitivity reaction occurs. (5.1)
- Seizures and Central Nervous System Adverse Reactions: CNS adverse reactions such as seizures have been reported with imipenem/cilastatin, a component of RECARBRIO. If focal tremors, myoclonus, or seizures occur, evaluate patients, to determine whether RECARBRIO should be discontinued. (5.2)
- Increased Seizure Potential Due to Interaction with Valproic Acid: Concomitant use of RECARBRIO with valproic acid or divalproex sodium may reduce the serum concentration of valproic acid which may increase the risk of breakthrough seizures. Avoid concomitant use or consider alternative antibacterial drugs other than carbapenems. (5.3, 7.2)
- Clostridioides difficile-Associated Diarrhea (CDAD): Has been reported with RECARBRIO. Evaluate if diarrhea occurs. (5.4)
2 Dosage and Administration
- Adult Patients: The recommended dosage of RECARBRIO is 1.25 grams (imipenem 500 mg, cilastatin 500 mg, relebactam 250 mg) administered by intravenous (IV) infusion over 30 minutes every 6 hours to adult patients with creatinine clearance (CLcr) 90 mL/min or greater. (2.1)
- Pediatric Patients weighing at least 2 kg: The recommended dosage of RECARBRIO for pediatric patients weighing at least 2kg varies by patient weight and age, with specific dosing recommendations as shown in the table below, (2.2):
| Recommended Dosage of RECARBRIO In Pediatric Patients Weighing at Least 2 kg | ||||
|---|---|---|---|---|
| Age Range | Body Weight | Dose | Dosing Frequency | Infusion Duration |
| Birth Pediatric patients from birth (includes pediatric patients at least 37 weeks post-menstrual age) to less than 3 months |
2 kg or greater | RECARBRIO 37.5 Provides 15 mg/kg imipenem, 15 mg/kg cilastatin, and 7.5 mg/kg relebactam mg/kg |
Every 8 hours | 60 minutes |
| 3 months to less than 18 years | less than 30 kg | RECARBRIO 37.5 mg/kg | Every 6 hours | 60 minutes |
| 3 months to less than 18 years | 30 kg or greater | RECARBRIO 1.25 Provides 500 mg imipenem, 500 mg cilastatin, and 250 mg, relebactam grams |
Every 6 hours | 30 minutes |
- Dose reduction is required in adult and pediatric (weighing at least 30 kg) patients with renal impairment. (2.3, 2.4)
- Do not administer RECARBRIO to adults with CLcr less than 15 mL/min unless hemodialysis is instituted within 48 hours after dose administration. (2.3)
- Do not administer RECARBRIO to pediatric patients weighing at least 30 kg with an eGFR less than 15 mL/min/1.73m2 unless hemodialysis is instituted within 48 hours after dose administration. (2.4)
- See Full Prescribing Information for instructions for constituting supplied dry powder and subsequent required dilution. (2.5)
- See Full Prescribing Information for drug compatibilities and incompatibilities. (2.7, 2.8)
3 Dosage Forms and Strengths
RECARBRIO (imipenem, cilastatin, and relebactam) for injection, 1.25 grams is supplied as a white to light yellow sterile powder for constitution in a single-dose glass vial containing imipenem 500 mg (equivalent to 530 mg imipenem monohydrate), cilastatin 500 mg (equivalent to 531 mg cilastatin sodium), and relebactam 250 mg (equivalent to 263 mg relebactam monohydrate).
5.4 Clostridioides Difficile
Clostridioides difficile-associated diarrhea (CDAD) has been reported with use of nearly all antibacterial agents, including RECARBRIO, and may range in severity from mild diarrhea to fatal colitis. Treatment with antibacterial agents alters the normal flora of the colon leading to overgrowth of C. difficile.
C. difficile produces toxins A and B which contribute to the development of CDAD. Hypertoxin producing strains of C. difficile cause increased morbidity and mortality, as these infections can be refractory to antimicrobial therapy and may require colectomy. CDAD must be considered in all patients who present with diarrhea following antibacterial drug use. Careful medical history is necessary since CDAD has been reported to occur over two months after the administration of antibacterial agents.
If CDAD is suspected or confirmed, ongoing antibacterial drug use not directed against C. difficile may need to be discontinued. Appropriate fluid and electrolyte management, protein supplementation, antibacterial drug treatment of C. difficile, and surgical evaluation should be instituted as clinically indicated.
5.1 Hypersensitivity Reactions
Serious and occasionally fatal hypersensitivity (anaphylactic) reactions have been reported in patients receiving therapy with beta lactams. Before initiating therapy with RECARBRIO, careful inquiry should be made concerning previous hypersensitivity reactions to carbapenems, penicillins, cephalosporins, other beta lactams, and other allergens. If a hypersensitivity reaction to RECARBRIO occurs, discontinue the therapy immediately.
RECARBRIO is contraindicated in patients with a history of severe hypersensitivity to any component of RECARBRIO [see Contraindications (4)].
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
2.7 Storage of Constituted Solution
RECARBRIO, as supplied in single-dose glass vials upon constitution with the appropriate diluent and following further dilution in the infusion bag, maintains satisfactory potency for at least 2 hours at room temperature (up to 30°C) or for at least 24 hours under refrigeration at 2°C to 8°C (36°F to 46°F). Do not freeze solutions of RECARBRIO.
16 How Supplied/storage and Handling
RECARBRIO (imipenem, cilastatin, and relebactam) for injection, 1.25 grams is supplied as a white to light yellow sterile powder for constitution in a single-dose glass vial containing imipenem 500 mg (equivalent to 530 mg imipenem monohydrate), cilastatin 500 mg (equivalent to 531 mg cilastatin sodium), and relebactam 250 mg (equivalent to 263 mg relebactam monohydrate).
The vials are supplied as a single-dose glass vial (NDC 0006-3856-01) and in cartons containing 25 vials (NDC 0006-3856-02).
2.8 Compatible Injectable Drug Products
Compatible Drug Products
The physical compatibility of RECARBRIO with selected injectable drug products was evaluated in two commonly available diluents. Compatible drugs with the corresponding compatible diluent (i.e., 5% Dextrose Injection, USP or 0.9% Sodium Chloride Injection, USP) are listed below. RECARBRIO should not be co-administered through the same intravenous line (or cannula), with other drug products not listed below, as no compatibility data are available. Refer to the respective prescribing information of the co-administered drug(s) to confirm compatibility of simultaneous co-administration.
List of Compatible Injectable Drugs for use with 5% Dextrose USP or 0.9% Sodium Chloride USP Injection as Diluents
- dexmedetomidine
- dopamine
- epinephrine
- fentanyl
- heparin
- midazolam
- norepinephrine
- phenylephrine
2.1 Recommended Dosage in Adult Patients
The recommended dosage of RECARBRIO is 1.25 grams (imipenem 500 mg, cilastatin 500 mg, and relebactam 250 mg) administered by intravenous (IV) infusion over 30 minutes every 6 hours in adult patients with creatinine clearance (CLcr) of 90 mL/min or greater). The recommended duration of treatment with RECARBRIO is 4 days to 14 days. The duration of therapy should be guided by the severity and location of infection and clinical response.
2.9 Incompatible Injectable Drug Products
RECARBRIO for injection for intravenous infusion is physically incompatible with propofol in 5% Dextrose USP or 0.9% Sodium Chloride USP.
13.2 Animal Toxicology And/or Pharmacology
Relebactam given as a single entity caused renal tubular degeneration in monkeys at AUC exposure 7-fold the human AUC exposure at the MRHD. Renal tubular degeneration was shown to be reversible after dose discontinuation. There was no evidence of nephrotoxicity at AUC exposures less than or equal to 3-fold the human AUC exposure at the MRHD.
5.5 Development of Drug Resistant Bacteria
Prescribing RECARBRIO in the absence of a proven or strongly suspected bacterial infection or prophylactic indication is unlikely to provide benefit to the patient and increases the risk of the development of drug-resistant bacteria.
1.3 Complicated Intra Abdominal Infections (ciai)
RECARBRIO is indicated in adult and pediatric patients weighing at least 2 kg who have limited or no alternative treatment options for the treatment of complicated intra-abdominal infections (cIAI) caused by the following susceptible gram-negative microorganisms: Bacteroides caccae, Bacteroides fragilis, Bacteroides ovatus, Bacteroides stercoris, Bacteroides thetaiotaomicron, Bacteroides uniformis, Bacteroides vulgatus, Citrobacter freundii, Enterobacter cloacae, Escherichia coli, Fusobacterium nucleatum, Klebsiella aerogenes, Klebsiella oxytoca, Klebsiella pneumoniae, Parabacteroides distasonis, and Pseudomonas aeruginosa.
Approval of this indication is based on limited clinical safety and efficacy data for RECARBRIO [see Clinical Studies (14.2)].
Principal Display Panel 1.25 G Vial Carton Label
NDC 0006-3856-02
Recarbrio™
(imipenem, cilastatin, and relebactam)
for Injection
1.25 g per vial*
Must be constituted and further diluted.
See enclosed package insert for preparation instructions.
For Intravenous Infusion Only
*Each vial contains imipenem 500 mg (equivalent to 530 mg imipenem monohydrate), cilastatin 500 mg (equivalent
to 531 mg cilastatin sodium), and relebactam 250 mg (equivalent to 263 mg relebactam monohydrate).
Inactive ingredient: 20 mg sodium bicarbonate added to each vial as a buffer.
25 single-dose vials
Rx only
1.5 Usage to Reduce Development of Drug Resistant Bacteria
To reduce the development of drug-resistant bacteria and maintain the effectiveness of RECARBRIO and other antibacterial drugs, RECARBRIO should be used only to treat or prevent infections that are proven or strongly suspected to be caused by susceptible bacteria. When culture and susceptibility information are available, they should be considered in selecting or modifying antibacterial therapy. In the absence of such data, local epidemiology and susceptibility patterns may contribute to the empiric selection of therapy.
2.3 Recommended Dosage in Adult Patients With Renal Impairment
Adult patients who have a CLcr less than 90 mL/min require dosage reduction of RECARBRIO. The recommended dosage of RECARBRIO in adult patients with renal impairment is shown in Table 2.
| Creatinine Clearance (CLcr) ([mL/min)] CLcr calculated using the Cockcroft-Gault formula for adult patients.
|
Dose RECARBRIO is provided as a single vial in a fixed-dose combination; the dose for each component will be adjusted equally during preparation [see Dosage and Administration (2.5)].
|
Dosing Frequency | Infusion Duration |
|---|---|---|---|
| 60 to 89 | RECARBRIO 1 gram Provides 400 mg imipenem, 400 mg cilastatin, and 200 mg relebactam
|
Every 6 hours | 30 minutes |
| 30 to 59 | RECARBRIO 0.75 grams Provides 300 mg imipenem, 300 mg cilastatin, and 150 mg relebactam
|
Every 6 hours | 30 minutes |
| 15 to 29 not receiving hemodialysis | RECARBRIO 0.5 grams Provides 200 mg imipenem, 200 mg cilastatin, and 100 mg relebactam
|
Every 6 hours | 30 minutes |
| Receiving hemodialysis Administration should be timed to follow hemodialysis and at intervals timed from the end of that hemodialysis session.
|
RECARBRIO 0.5 grams | Every 6 hours | 30 minutes |
| Less than 15 not receiving hemodialysis | Not recommended |
Do not administer RECARBRIO to adult patients with CLcr less than 15 mL/min unless hemodialysis is instituted within 48 hours after dose administration.
Imipenem, cilastatin, and relebactam are cleared from the circulation during hemodialysis. For adult patients maintained on hemodialysis, administer the recommended dose of RECARBRIO after hemodialysis and at intervals timed from the end of that hemodialysis session.
There is inadequate information to recommend dosage of RECARBRIO for adults undergoing peritoneal dialysis.
2.2 Recommended Dosage in Pediatric Patients Weighing At Least 2 Kg
The recommended dosage of RECARBRIO in pediatric patients weighing at least 2 kg varies by patient weight and age, with specific dosing recommendations shown in Table 1. The recommended duration of treatment with RECARBRIO is 4 days to 14 days. The duration of therapy should be guided by the severity and location of infection and clinical response.
RECARBRIO is not recommended in pediatric patients less than 37 weeks post-menstrual age (gestational age at birth plus post-natal age) [see Indications and Usage (1.4) and Use in Specific Populations (8.4)].
RECARBRIO is not recommended for pediatric patients weighing less than 30 kg with renal impairment [see Dosage and administration (2.4) and Use in Specific Populations (8.4)].
| Age Range | Body Weight | Dose | Dosing Frequency | Infusion Duration |
|---|---|---|---|---|
| Birth Pediatric patients from birth (includes pediatric patients at least 37 weeks post-menstrual age) to less than 3 months |
2 kg or greater | RECARBRIO 37.5 Provides 15 mg/kg imipenem, 15 mg/kg cilastatin, and 7.5 mg/kg relebactam mg/kg |
Every 8 hours | 60 minutes |
| 3 months to less than 18 years | less than 30 kg | RECARBRIO 37.5 mg/kg | Every 6 hours | 60 minutes |
| 3 months to less than 18 years | 30 kg or greater | RECARBRIO 1.25 Provides 500 mg imipenem, 500 mg cilastatin, and 250 mg, relebactam grams |
Every 6 hours | 30 minutes |
2.5 Preparation of Recarbrio Solution for Intravenous Administration
RECARBRIO is supplied as a dry powder in a single-dose vial that must be constituted and further diluted using aseptic technique prior to intravenous infusion. To prepare the infusion solution, contents of the vial must be constituted with the appropriate diluent as instructed below. A list of appropriate diluents is as follows:
- 0.9% Sodium Chloride Injection, USP
- 5% Dextrose Injection, USP
- 5% Dextrose Injection, USP + 0.9% Sodium Chloride Injection, USP
- 5% Dextrose Injection, USP + 0.45% Sodium Chloride Injection, USP
- 5% Dextrose Injection, USP + 0.225% Sodium Chloride Injection, USP
RECARBRIO has low aqueous solubility. To ensure complete dissolution of RECARBRIO it is important to adhere to the following instructions:
-
Step 1) For diluents available in 100 mL prefilled infusion bags, proceed to step 2. For diluents not available in 100 mL prefilled infusion bags, aseptically withdraw 100 mL of the desired diluent and transfer it to an empty infusion bag, then proceed to step 2.
-
Step 2) Withdraw 20 mL (as two 10 mL aliquots) of diluent from the appropriate infusion bag and constitute the vial with one 10 mL aliquot of the diluent. The constituted suspension is for intravenous infusion only after dilution in an appropriate infusion solution.
-
Step 3) After constitution, shake vial well and transfer resulting suspension into the remaining 80 mL of the infusion bag.
-
Step 4) Add the second 10 mL aliquot of infusion diluent to the vial and shake well to ensure complete transfer of vial contents; repeat transfer of the resulting suspension to the infusion solution before administering. Agitate the resulting mixture until clear.
Constituted solutions of RECARBRIO range from colorless to yellow. Variations of color within this range do not affect the potency of the product.
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit. Discard if discoloration or visible particles are observed.
The above instructions for preparation of RECARBRIO solution for intravenous administration must be followed for all patients, irrespective of the intended patient’s renal function, age or weight.
The volume of this prepared RECARBRIO solution to be administered in adult and pediatric (weighing at least 30 kg) patients is determined based on estimated renal function [see Dosage and Administration (2.6)].
The volume of the prepared RECARBRIO solution to be administered to pediatric patients weighing 2 kg to less than 30 kg with normal renal function is calculated based on patient weight using the formula below [see Dosage and Administration (2.2)]:
To achieve the appropriate infusion volume, withdraw and discard the excess (100 mL minus the infusion volume) from the prepared 100 mL solution. The remaining volume is the appropriate infusion volume.
5.2 Seizures and Other Central Nervous System (cns) Adverse Reactions
CNS adverse reactions, such as seizures, confusional states, and myoclonic activity, have been reported during treatment with imipenem/cilastatin, a component of RECARBRIO, especially when recommended dosages of imipenem were exceeded. These have been reported most commonly in patients with CNS disorders (e.g., brain lesions or history of seizures) and/or compromised renal function.
Anticonvulsant therapy should be continued in patients with known seizure disorders. If CNS adverse reactions including seizures occur, patients should undergo a neurological evaluation to determine whether RECARBRIO should be discontinued.
5.3 Increased Seizure Potential Due to Interaction With Valproic Acid
Concomitant use of RECARBRIO, with valproic acid or divalproex sodium may increase the risk of breakthrough seizures. Avoid concomitant use of RECARBRIO with valproic acid or divalproex sodium or consider alternative antibacterial drugs other than carbapenems [see Drug Interactions (7.2)].
1.2 Complicated Urinary Tract Infections (cuti), Including Pyelonephritis
RECARBRIO is indicated in adult and pediatric patients weighing at least 2 kg who have limited or no alternative treatment options, for the treatment of complicated urinary tract infections (cUTI), including pyelonephritis, caused by the following susceptible gram-negative microorganisms: Enterobacter cloacae, Escherichia coli, Klebsiella aerogenes, Klebsiella pneumoniae, and Pseudomonas aeruginosa.
Approval of this indication is based on limited clinical safety and efficacy data for RECARBRIO [see Clinical Studies (14.2)].
2.4 Recommended Dosage in Pediatric Patients Weighing At Least 30 Kg With Renal Impairment
Pediatric patients weighing at least 30 kg who have an estimated glomerular filtration rate (eGFR) less than 90 mL/min/1.73m2 require dosage reduction of RECARBRIO. The recommended dosage of RECARBRIO in pediatric patients weighing at least 30 kg with renal impairment is shown in Table 3. There is insufficient information to recommend a dosage of RECARBRIO for pediatric patients weighing less than 30 kg with any degree of renal impairment.
|
eGFR (mL/min/1.73 m2) Calculated using a GFR estimating equation validated in pediatric patients weighing at least 30 kg.
|
Recommended Dose RECARBRIO is provided as a single vial in a fixed-dose combination; the dose for each component will be adjusted equally during preparation [see Dosage and Administration (2.5)].
|
Dosing Frequency | Infusion Duration |
|---|---|---|---|
| 60 to 89 | RECARBRIO 1 gram Provides 400 mg imipenem, 400 mg cilastatin, and 200 mg relebactam
|
Every 6 hours | 30 minutes |
| 30 to 59 | RECARBRIO 0.75 grams Provides 300 mg imipenem, 300 mg cilastatin, and 150 mg relebactam
|
Every 6 hours | 30 minutes |
| 15 to 29 not receiving hemodialysis | RECARBRIO 0.5 grams Provides 200 mg imipenem, 200 mg cilastatin, and 100 mg relebactam
|
Every 6 hours | 30 minutes |
| Receiving hemodialysis Administration should be timed to follow hemodialysis and at intervals timed from the end of that hemodialysis session.
|
RECARBRIO 0.5 grams | Every 6 hours | 30 minutes |
| Less than 15 not receiving hemodialysis | Not recommended |
Do not administer RECARBRIO to pediatric patients weighing at least 30 kg with an eGFR less than 15 mL/min/1.73m2 unless hemodialysis is instituted within 48 hours after dose administration.
Imipenem, cilastatin, and relebactam are cleared from the circulation during hemodialysis. For pediatric patients weighing at least 30 kg maintained on hemodialysis, administer the recommended dose of RECARBRIO after hemodialysis and at intervals timed from the end of that hemodialysis session.
There is inadequate information to recommend a dosage of RECARBRIO for pediatric patients undergoing peritoneal dialysis.
1.1 Hospital Acquired Bacterial Pneumonia and Ventilator Associated Bacterial Pneumonia (habp/vabp)
RECARBRIO® is indicated in adult and pediatric patients weighing at least 2 kg for the treatment of hospital-acquired bacterial pneumonia and ventilator-associated bacterial pneumonia, caused by the following susceptible gram-negative microorganisms: Acinetobacter calcoaceticus-baumannii complex, Enterobacter cloacae, Escherichia coli, Haemophilus influenzae, Klebsiella aerogenes, Klebsiella oxytoca, Klebsiella pneumoniae, Pseudomonas aeruginosa, and Serratia marcescens.
14.1 Hospital Acquired Bacterial Pneumonia and Ventilator Associated Bacterial Pneumonia in Adult Patients
A total of 535 hospitalized adults with HABP/VABP were randomized and received trial medications in a multinational, double-blind trial (Trial 1, NCT02493764) comparing RECARBRIO 1.25 grams (imipenem 500 mg/cilastatin 500 mg/relebactam 250 mg) intravenously every 6 hours to piperacillin and tazobactam (4.5 grams) for 7 to 14 days of therapy.
The modified intent-to-treat (MITT) population, which included all randomized patients who received at least one dose of trial treatment and did not have only gram-positive cocci on Gram stain of the baseline lower respiratory tract (LRT) specimen included 531 patients; the mean age was 60 and 43% were 65 years of age or older. The majority of patients were men (69%), white (78%), and from Europe (61%). The mean APACHE II score was 15 and 47% of the population had an APACHE II score of ≥15. At randomization, 66% of patients were admitted to the ICU, 77% had been in the hospital for ≥5 days, and 48% had a creatinine clearance of <90 mL/min. Concurrent bacteremia was present at baseline in 5.8% of patients.
Table 10 presents the incidence of all-cause mortality through Day 28 and clinical response at the early follow-up (EFU) visit (7 to 14 days after the end of therapy) in the MITT population. Overall results are presented along with subgroup results by pneumonia diagnosis.
| RECARBRIO | Piperacillin/Tazobactam | Treatment Difference | ||||
|---|---|---|---|---|---|---|
| n/m | (%) | n/m | (%) | % Treatment differences and 95% confidence intervals are based on Miettinen & Nurminen method.
|
(95% CI) | |
| EFU = early follow up | ||||||
|
All-Cause Mortality Through Day 28
n/m = number of subjects with survival status of death or unknown / number of modified intent-to-treat subjects.
,
One subject in the RECARBRIO arm had unknown mortality status at Day 28 which was counted as a death.
|
42/264 | (15.9) | 57/267 | (21.3) | -5.3 | (-11.9, 1.2) |
| Non-ventilated HABP | 18/142 | (12.7) | 15/131 | (11.5) | 1.2 | (-6.8, 9.1) |
| Ventilated HABP/VABP | 24/122 | (19.7) | 42/136 | (30.9) | -11.2 | (-21.6, -0.5) |
|
Clinical Response at EFU
n/m = number of subjects with a favorable clinical response / number of modified intent-to-treat subjects.
|
161/264 | (61.0) | 149/267 | (55.8) | 5.0 | (-3.2, 13.2) |
| Non-ventilated HABP | 95/142 | (66.9) | 87/131 | (66.4) | 0.5 | (-10.7, 11.7) |
| Ventilated HABP/VABP | 66/122 | (54.1) | 62/136 | (45.6) | 8.5 | (-3.7, 20.5) |
In the MITT population, in patients with an APACHE II score <15, Day 28 all-cause mortality rates were 17/139 (12.2%) for RECARBRIO-treated patients and 12/140 (8.6%) for piperacillin/tazobactam-treated patients, clinical cure rates were 90/139 (64.7%) and 98/140 (70%), respectively. In patients with an APACHE II score ≥15, Day 28 all-cause mortality rates were 25/125 (20%) for RECARBRIO-treated patients and 45/127 (35.4%) for piperacillin/tazobactam-treated patients, clinical cure rates were 71/125 (56.8%) and 51/127 (40.2%), respectively.
Per pathogen favorable clinical response at EFU and Day 28 all-cause mortality were assessed in a microbiological modified intention to treat (mMITT) population, which consisted of all randomized MITT subjects who had at least one baseline LRT pathogen that was susceptible to both study treatments (Table 11).
| Baseline LRT Pathogen |
Day 28 All-Cause Mortality | Clinical Response at EFU | ||
|---|---|---|---|---|
| RECARBRIO n/m n/m = the number of subjects with survival status of death or unknown within each category / the number of microbiological modified intent-to-treat subjects who have the corresponding baseline pathogen from LRT culture. (%) |
Piperacillin/ Tazobactam n/m (%) |
RECARBRIO n/m n/m = the number of subjects with a favorable clinical response within each category / the number of microbiological modified intent-to-treat subjects who have the corresponding baseline pathogen from LRT culture. (%) |
Piperacillin/ Tazobactam n/m (%) |
|
| LRT = lower respiratory tract EFU = early follow-up |
||||
|
Acinetobacter calcoaceticus- baumannii complex |
0/5 Supportive evidence was derived from the imipenem and cilastatin prescribing information. (0.0) |
1/10 (10.0) | 4/5 (80.0) | 6/10 (60.0) |
|
Enterobacter cloacae |
1/7 (14.3) | 3/16 (18.8) | 6/7 (85.7) | 12/16 (75.0) |
| Escherichia coli | 5/27(18.5) | 8/33 (24.2) | 16/27 (59.3) | 19/33 (57.6) |
|
Haemophilus influenzae All H. influenzae isolates were susceptible to imipenem. The susceptible MIC breakpoint for PIP/TAZ is ≤1/4 mcg/mL. At the lowest concentration of PIP/TAZ tested (2/4 mcg/mL) there was no visible growth.
|
2/13 (15.4) | 3/12 (25.0) | 9/13 (69.2) | 8/12 (66.7) |
|
Klebsiella spp Includes Klebsiella aerogenes, Klebsiella oxytoca, Klebsiella pneumoniae.
|
6/42 (14.3) | 8/41 (19.5) | 25/42 (59.5) | 28/41 (68.3) |
|
Pseudomonas aeruginosa |
7/26 (26.9) | 5/35 (14.3) | 12/26 (46.2) | 20/35 (57.1) |
|
Serratia marcescens |
2/10 (20.0) | 1/4 (25.0) | 7/10 (70.0) | 3/4 (75.0) |
14.2 Complicated Urinary Tract Infections, Including Pyelonephritis and Complicated Intra Abdominal Infections in Adult Patients
The determination of efficacy and safety of RECARBRIO was supported in part by the previous findings of the efficacy and safety of imipenem/cilastatin for the treatment of cUTI and cIAI. The contribution of relebactam to RECARBRIO was primarily established in vitro and in animal models of infection [see Microbiology (12.4)]. Imipenem/cilastatin plus relebactam was studied in cUTI including pyelonephritis (Trial 2, NCT01505634) and cIAI (Trial 3, NCT01506271) in randomized, blinded, active-controlled, multicenter trials. These trials provided only limited efficacy and safety information.
2.6 Preparation of Recarbrio Solution for Intravenous Administration in Adult and Pediatric Patients Weighing At Least 30 Kg With Renal Impairment
Prepare a reduced dose of RECARBRIO (1 gram, 0.75 grams, or 0.5 grams) [see Dosage and Administration (2.3, 2.4)] by preparing a 100 mL solution containing 1.25 grams as described above [see Dosage and Administration (2.5)] then withdrawing and discarding the excess according to Table 4.
|
Estimated Renal Function Creatinine Clearance (mL/min) Calculated using the Cockcroft-Gault formula for adult patients
Or Estimated Glomerular Filtration Rate eGFR (mL/min/1.73 m2) Calculated using a GFR estimating equation validated in pediatric patients weighing at least 30 kg
|
Dose | After preparation as instructed above, remove from the 100 mL prepared bag the volume indicated below and discard | Resulting volume that provides the indicated reduced dose |
|---|---|---|---|
| 60 to 89 | RECARBRIO 1 gram Provides 400 mg imipenem, 400 mg cilastatin, and 200 mg relebactam
|
20 mL | 80 mL |
| 30 to 59 | RECARBRIO 0.75 grams Provides 300 mg imipenem, 300 mg cilastatin, and 150 mg relebactam
|
40 mL | 60 mL |
| 15 to 29 not receiving hemodialysis | RECARBRIO 0.5 grams Provides 200 mg imipenem, 200 mg cilastatin, and 100 mg relebactam
|
60 mL | 40 mL |
| Receiving hemodialysis | RECARBRIO 0.5 grams | 60 mL | 40 mL |
14.3 Hospital Acquired Bacterial Pneumonia and Ventilator Associated Bacterial Pneumonia (habp/vabp), Complicated Urinary Tract Infections (cuti), and Complicated Intra Abdominal Infections (ciai) in Pediatric Patients
The safety and efficacy of RECARBRIO in pediatric patients were investigated in a randomized, active-controlled, open-label trial that enrolled hospitalized patients from birth to less than 18 years of age with HABP/VABP, cUTI, or cIAI (Trial 4, NCT03969901). Eligible patients were randomized in a 3:1 ratio with stratification by age group and infection type to receive IV RECARBRIO or active control (defined as investigator’s choice of specified comparators). Patients with HABP/VABP received IV therapy for 7 to 14 days. Patients with cUTI or cIAI received IV therapy for a minimum of 3 days before an optional switch to oral step-down therapy at the discretion of the investigator to complete a total of the 5 to 14 days of antibacterial therapy.
The modified intention to treat (MITT) population consisted of 113 patients who were randomized and received at least one dose of trial treatment (RECARBRIO, n=85; active control, n=28). In patients treated with RECARBRIO, 49% were male and the median age was 5 years (range 21 days to 17 years). The age groups who received RECARBRIO were as follows: 12 to <18 years (n=10), 6 to <12 years (n=31), 2 to <6 years (n=21), 3 months to <2 years (n=15), and birth to <3 months (n=8). The MITT population included 54 patients (48%) with cUTI, 53 (47%) with cIAI, and 6 (5%) with HABP/VABP. Patients were predominantly white (81%) and from Europe (61%).
The primary objective of the study was to evaluate the safety and tolerability of RECARBRIO. Efficacy assessments were not powered for formal hypothesis testing between treatment groups. For HABP/VABP and cIAI, Table 12 presents the clinical cure rates at early follow-up (EFU, 7 to 14 days after the end of therapy) in the MITT population. For cUTI, Table 13 presents the clinical cure and microbiologic response rates at EFU in the mMITT population, which included all patients with cUTI in the MITT population that had a baseline pathogen isolated. No patient died in either intervention group in the study through Day 28.
| RECARBRIO n/N (%) |
Active Control n/N (%) |
|
|---|---|---|
| HABP/VABP | 5/5 (100.0) | 1/1 (100.0) |
| cIAI | 34/39 (87.2) | 13/14 (92.9) |
|
RECARBRIO n/N (%) |
ACTIVE Control n/N (%) |
|
|---|---|---|
| Clinical Cure | 20/30 (66.7) | 7/10 (70.0) |
| Microbiologic Response | 20/30 (66.7) | 8/10 (80.0) |
Structured Label Content
Section 42229-5 (42229-5)
Adult Patients
Overview of the Safety Evaluation of RECARBRIO in Adult Patients
Safety was primarily evaluated in three active-controlled, double-blind trials in HABP/VABP, cUTI, and cIAI (Trials 1, 2, and 3, respectively).
In the HABP/VABP trial (Trial 1), patients were treated with either RECARBRIO or piperacillin and tazobactam (4.5 grams).
In the cUTI trial (Trial 2) and cIAI trial (Trial 3), patients in the treatment arms were treated with either imipenem 500 mg/cilastatin 500 mg and relebactam 250 mg or imipenem 500 mg/cilastatin 500 mg and relebactam 125 mg (not an approved dose), and patients in the control arm were treated with imipenem 500 mg/cilastatin 500 mg plus placebo (IV normal saline). Across Trials 2 and 3, the mean duration of IV therapy in patients treated with imipenem/cilastatin plus relebactam 250 mg was approximately 7 days.
Clinical Trial Experience in Adult Patients with HABP/VABP
Trial 1 included 266 adult patients treated with RECARBRIO and 269 patients treated with piperacillin and tazobactam (4.5 grams) administered intravenously over 30 minutes every 6 hours. The mean age was 60 years, 43% of patients were 65 years of age and older, 31% were female and 22% had polymicrobial infection. The mean Acute Physiology and Chronic Health Evaluation (APACHE) II score was 15 and 48% of patients had an APACHE II score greater than or equal to 15 at baseline. Overall, 260 (49%) patients were ventilated at enrollment, including 194 (36%) patients with VABP and 66 (12%) patients with ventilated HABP.
Section 44425-7 (44425-7)
Store RECARBRIO vials at 20°C to 25°C (68°F to 77°F), excursions permitted between 15°C to 30°C (between 59°F to 86°F) [see USP Controlled Room Temperature]. Keep vials in the carton.
10 Overdosage (10 OVERDOSAGE)
In the event of overdose, discontinue RECARBRIO, treat symptomatically, and institute general supportive treatment. Imipenem, cilastatin, and relebactam can be removed by hemodialysis [see Clinical Pharmacology (12.3)]. No clinical information is available on the use of hemodialysis to treat overdosage.
11 Description (11 DESCRIPTION)
RECARBRIO (imipenem, cilastatin, and relebactam) for injection is an antibacterial combination product consisting of imipenem, a carbapenem antibacterial drug, cilastatin, a renal dehydropeptidase inhibitor, and relebactam, a diazabicyclooctane beta-lactamase inhibitor, for intravenous administration.
7.1 Ganciclovir
Generalized seizures have been reported in patients who received ganciclovir concomitantly with imipenem/cilastatin, a component of RECARBRIO. Ganciclovir should not be used concomitantly with RECARBRIO unless the potential benefits outweigh the risks.
7.2 Valproic Acid
Based on case reports in the literature concomitant use of carbapenems, including imipenem/cilastatin, components of RECARBRIO, with valproic acid or divalproex sodium may decrease valproic acid concentrations which may increase the risk of breakthrough seizures [see Warnings and Precautions (5.3)]. Although the mechanism of this interaction is unknown, data from in vitro and animal studies suggest that carbapenems may inhibit the hydrolysis of valproic acid's glucuronide metabolite (VPA-g) back to valproic acid, thus decreasing the serum concentrations of valproic acid. Avoid concomitant use of RECARBRIO with valproic acid or divalproex sodium. Consider alternative antibacterials other than carbapenems to treat infections in patients whose seizures are well controlled on valproic acid or divalproex sodium.
8.4 Pediatric Use
The safety and effectiveness of RECARBRIO for the treatment of HABP/VABP, and for the treatment of cUTI and cIAI in patients who have limited or no alternative treatment options have been established in pediatric patients weighing at least 2 kg. Use of RECARBRIO in pediatric patients is supported by evidence from an adequate and well-controlled trial of RECARBRIO in adults with HABP/VABP, controlled trials in adults with cUTI and cIAI, and additional pharmacokinetic, safety, and efficacy data from pediatric trials [see Clinical Pharmacology (12.3) and Clinical Studies (14.1, 14.2 and 14.3)].
The safety profile of RECARBRIO in pediatric patients from the pediatric trials was comparable to that in adults treated with RECARBRIO [see Adverse Reactions (6.1)].
RECARBRIO is not recommended in pediatric patients less than 37 weeks post-menstrual age (gestational age at birth plus post-natal age) or weighing less than 30 kg with renal impairment.
The safety and effectiveness of RECARBRIO for the treatment of HABP/VABP, cUTI or cIAI have not been established in pediatric patients weighing less than 2 kg.
8.5 Geriatric Use
Of the 266 patients treated with RECARBRIO in Trial 1, 113 (42.5%) were 65 years of age or older, including 55 (20.7%) patients 75 years of age and older. Of the 216 patients treated with imipenem/cilastatin plus relebactam 250 mg in Trials 2 and 3, 67 (31.0%) were 65 years of age or older, including 25 (11.6%) patients 75 years of age and older. No overall differences in safety or effectiveness were observed between these patients and younger patients, and other reported clinical experience has not identified differences in responses between the elderly and younger patients, but greater sensitivity of some older individuals cannot be ruled out.
RECARBRIO is known to be substantially excreted by the kidney, and the risk of adverse reactions to this drug may be greater in patients with impaired renal function. Because elderly patients are more likely to have decreased renal function, care should be taken in dose selection, and it may be useful to monitor renal function. No dosage adjustment is required based on age. Dosage adjustment for elderly patients should be based on renal function [see Dosage and Administration (2.2) and Clinical Pharmacology (12.3)].
4 Contraindications (4 CONTRAINDICATIONS)
RECARBRIO is contraindicated in patients with a history of known severe hypersensitivity (severe systemic allergic reaction such as anaphylaxis) to any component of RECARBRIO.
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following serious adverse reactions are described in greater detail in the Warnings and Precautions section.
- Hypersensitivity Reactions [see Warnings and Precautions (5.1)]
- Seizures and Other Central Nervous System Adverse Reactions [see Warnings and Precautions (5.2)]
- Increased Seizure Potential Due to Interaction with Valproic Acid [see Warnings and Precautions (5.3)]
- Clostridioides difficile-Associated Diarrhea (CDAD) [see Warnings and Precautions (5.4)]
7 Drug Interactions (7 DRUG INTERACTIONS)
8.6 Renal Impairment
Reduce RECARBRIO dosage in adult patients with a CLcr less than 90 mL/min and in pediatric patients weighing at least 30 kg with an eGFR less than 90 mL/min/1.73 m2 [see Dosage and Administration (2.3, 2.4) and Clinical Pharmacology (12.3)]. RECARBRIO is not recommended in pediatric patients weighing less than 30 kg with renal impairment.
12.2 Pharmacodynamics
For imipenem, the % time of dosing interval that unbound plasma concentrations of imipenem exceed the imipenem/relebactam minimum inhibitory concentration (MIC) (%fT>MIC) against the infecting organism best correlates with antibacterial activity in animal and in vitro models of infection. For relebactam the ratio of the 24-hour unbound plasma relebactam AUC to imipenem/relebactam MIC (fAUC0–24hr/MIC) best predicts the activity of relebactam in animal and in vitro models of infection.
12.3 Pharmacokinetics
The steady-state pharmacokinetic parameters of imipenem, cilastatin, and relebactam in healthy adults with normal renal function (CLcr 90 mL/min or greater), after multiple 30-minute intravenous infusions of RECARBRIO administered every 6 hours are summarized in Table 7. Pharmacokinetic parameters were similar for single and multiple dose administration due to minimal accumulation. Pharmacokinetic parameters in pediatric patients are described in Table 9.
Adult Patients
The steady-state pharmacokinetic parameters of imipenem and relebactam in patients with active bacterial infection with CLcr 90 mL/min or greater following administration of the recommended dosage are summarized in Table 7.
| PK Parameters | cUTI/cIAI Patients | HABP/VABP Patients | |
|---|---|---|---|
| AUC0-24hr=area under the concentration time curve from 0 to 24 hours Cmax=maximum concentration CL=plasma clearance |
|||
| Imipenem | AUC0-24hr (µM-hr) | 570.6 (253.3) | 771 (342.3) |
| Cmax (µM) | 116.1 (52.4) | 122.7 (56.8) | |
| CL (L/hr) | 14 (6.1) | 10.4 (4.5) | |
| Relebactam | AUC0-24hr (µM-hr) | 415.8 (212.6) | 692.9 (354.3) |
| Cmax (µM) | 62.1 (24.7) | 80 (33.3) | |
| CL (L/hr) | 8.7 (4.5) | 5.2 (2.7) |
1.4 Limitations of Use
- RECARBRIO is not recommended in pediatric patients less than 37 weeks post-menstrual age (gestational age at birth plus post-natal age) [see Dosage and Administration (2.2) and Use in Specific Populations (8.4)].
- RECARBRIO is not recommended in pediatric patients weighing less than 30 kg with renal impairment [see Dosage and Administration (2.4), Use in Specific Populations (8.4, 8.6) and Clinical Pharmacology (12.3)].
1 Indications and Usage (1 INDICATIONS AND USAGE)
RECARBRIO is a combination of imipenem, a penem antibacterial, cilastatin, a renal dehydropeptidase inhibitor, and relebactam, a beta-lactamase inhibitor, indicated in adult and pediatric patients weighing at least 2 kg for the treatment of the following infections caused by susceptible gram-negative microorganisms:
- Hospital-acquired bacterial pneumonia and ventilator-associated bacterial pneumonia (HABP/VABP). (1.1)
- Complicated urinary tract infections, including pyelonephritis (cUTI) in patients who have limited or no alternative treatment options. (1.2)
- Complicated intra-abdominal infections (cIAI) in patients who have limited or no alternative treatment options. (1.3)
Approval of the cUTI and cIAI indications is based on limited clinical safety and efficacy data for RECARBRIO. (1.2, 1.3, 14)
Limitations of Use
- RECARBRIO is not recommended in pediatric patients less than 37 weeks post-menstrual age (gestational age at birth plus post-natal age). (1.4, 2.2).
- RECARBRIO is not recommended in pediatric patients weighing less than 30 kg with renal impairment. (1.4, 2.3).
Usage to Reduce Development of Drug-Resistant Bacteria
To reduce the development of drug-resistant bacteria and maintain the effectiveness of RECARBRIO and other antibacterial drugs, RECARBRIO should be used only to treat or prevent infections that are proven or strongly suspected to be caused by bacteria. (1.5)
12.1 Mechanism of Action
RECARBRIO is an antibacterial drug [see Microbiology (12.4)].
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Hypersensitivity Reactions: Serious and occasionally fatal hypersensitivity (anaphylactic) reactions have been reported in patients receiving beta lactam drugs. Discontinue RECARBRIO immediately if a hypersensitivity reaction occurs. (5.1)
- Seizures and Central Nervous System Adverse Reactions: CNS adverse reactions such as seizures have been reported with imipenem/cilastatin, a component of RECARBRIO. If focal tremors, myoclonus, or seizures occur, evaluate patients, to determine whether RECARBRIO should be discontinued. (5.2)
- Increased Seizure Potential Due to Interaction with Valproic Acid: Concomitant use of RECARBRIO with valproic acid or divalproex sodium may reduce the serum concentration of valproic acid which may increase the risk of breakthrough seizures. Avoid concomitant use or consider alternative antibacterial drugs other than carbapenems. (5.3, 7.2)
- Clostridioides difficile-Associated Diarrhea (CDAD): Has been reported with RECARBRIO. Evaluate if diarrhea occurs. (5.4)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
- Adult Patients: The recommended dosage of RECARBRIO is 1.25 grams (imipenem 500 mg, cilastatin 500 mg, relebactam 250 mg) administered by intravenous (IV) infusion over 30 minutes every 6 hours to adult patients with creatinine clearance (CLcr) 90 mL/min or greater. (2.1)
- Pediatric Patients weighing at least 2 kg: The recommended dosage of RECARBRIO for pediatric patients weighing at least 2kg varies by patient weight and age, with specific dosing recommendations as shown in the table below, (2.2):
| Recommended Dosage of RECARBRIO In Pediatric Patients Weighing at Least 2 kg | ||||
|---|---|---|---|---|
| Age Range | Body Weight | Dose | Dosing Frequency | Infusion Duration |
| Birth Pediatric patients from birth (includes pediatric patients at least 37 weeks post-menstrual age) to less than 3 months |
2 kg or greater | RECARBRIO 37.5 Provides 15 mg/kg imipenem, 15 mg/kg cilastatin, and 7.5 mg/kg relebactam mg/kg |
Every 8 hours | 60 minutes |
| 3 months to less than 18 years | less than 30 kg | RECARBRIO 37.5 mg/kg | Every 6 hours | 60 minutes |
| 3 months to less than 18 years | 30 kg or greater | RECARBRIO 1.25 Provides 500 mg imipenem, 500 mg cilastatin, and 250 mg, relebactam grams |
Every 6 hours | 30 minutes |
- Dose reduction is required in adult and pediatric (weighing at least 30 kg) patients with renal impairment. (2.3, 2.4)
- Do not administer RECARBRIO to adults with CLcr less than 15 mL/min unless hemodialysis is instituted within 48 hours after dose administration. (2.3)
- Do not administer RECARBRIO to pediatric patients weighing at least 30 kg with an eGFR less than 15 mL/min/1.73m2 unless hemodialysis is instituted within 48 hours after dose administration. (2.4)
- See Full Prescribing Information for instructions for constituting supplied dry powder and subsequent required dilution. (2.5)
- See Full Prescribing Information for drug compatibilities and incompatibilities. (2.7, 2.8)
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
RECARBRIO (imipenem, cilastatin, and relebactam) for injection, 1.25 grams is supplied as a white to light yellow sterile powder for constitution in a single-dose glass vial containing imipenem 500 mg (equivalent to 530 mg imipenem monohydrate), cilastatin 500 mg (equivalent to 531 mg cilastatin sodium), and relebactam 250 mg (equivalent to 263 mg relebactam monohydrate).
5.4 Clostridioides Difficile (5.4 Clostridioides difficile)
Clostridioides difficile-associated diarrhea (CDAD) has been reported with use of nearly all antibacterial agents, including RECARBRIO, and may range in severity from mild diarrhea to fatal colitis. Treatment with antibacterial agents alters the normal flora of the colon leading to overgrowth of C. difficile.
C. difficile produces toxins A and B which contribute to the development of CDAD. Hypertoxin producing strains of C. difficile cause increased morbidity and mortality, as these infections can be refractory to antimicrobial therapy and may require colectomy. CDAD must be considered in all patients who present with diarrhea following antibacterial drug use. Careful medical history is necessary since CDAD has been reported to occur over two months after the administration of antibacterial agents.
If CDAD is suspected or confirmed, ongoing antibacterial drug use not directed against C. difficile may need to be discontinued. Appropriate fluid and electrolyte management, protein supplementation, antibacterial drug treatment of C. difficile, and surgical evaluation should be instituted as clinically indicated.
5.1 Hypersensitivity Reactions
Serious and occasionally fatal hypersensitivity (anaphylactic) reactions have been reported in patients receiving therapy with beta lactams. Before initiating therapy with RECARBRIO, careful inquiry should be made concerning previous hypersensitivity reactions to carbapenems, penicillins, cephalosporins, other beta lactams, and other allergens. If a hypersensitivity reaction to RECARBRIO occurs, discontinue the therapy immediately.
RECARBRIO is contraindicated in patients with a history of severe hypersensitivity to any component of RECARBRIO [see Contraindications (4)].
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
2.7 Storage of Constituted Solution
RECARBRIO, as supplied in single-dose glass vials upon constitution with the appropriate diluent and following further dilution in the infusion bag, maintains satisfactory potency for at least 2 hours at room temperature (up to 30°C) or for at least 24 hours under refrigeration at 2°C to 8°C (36°F to 46°F). Do not freeze solutions of RECARBRIO.
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
RECARBRIO (imipenem, cilastatin, and relebactam) for injection, 1.25 grams is supplied as a white to light yellow sterile powder for constitution in a single-dose glass vial containing imipenem 500 mg (equivalent to 530 mg imipenem monohydrate), cilastatin 500 mg (equivalent to 531 mg cilastatin sodium), and relebactam 250 mg (equivalent to 263 mg relebactam monohydrate).
The vials are supplied as a single-dose glass vial (NDC 0006-3856-01) and in cartons containing 25 vials (NDC 0006-3856-02).
2.8 Compatible Injectable Drug Products
Compatible Drug Products
The physical compatibility of RECARBRIO with selected injectable drug products was evaluated in two commonly available diluents. Compatible drugs with the corresponding compatible diluent (i.e., 5% Dextrose Injection, USP or 0.9% Sodium Chloride Injection, USP) are listed below. RECARBRIO should not be co-administered through the same intravenous line (or cannula), with other drug products not listed below, as no compatibility data are available. Refer to the respective prescribing information of the co-administered drug(s) to confirm compatibility of simultaneous co-administration.
List of Compatible Injectable Drugs for use with 5% Dextrose USP or 0.9% Sodium Chloride USP Injection as Diluents
- dexmedetomidine
- dopamine
- epinephrine
- fentanyl
- heparin
- midazolam
- norepinephrine
- phenylephrine
2.1 Recommended Dosage in Adult Patients
The recommended dosage of RECARBRIO is 1.25 grams (imipenem 500 mg, cilastatin 500 mg, and relebactam 250 mg) administered by intravenous (IV) infusion over 30 minutes every 6 hours in adult patients with creatinine clearance (CLcr) of 90 mL/min or greater). The recommended duration of treatment with RECARBRIO is 4 days to 14 days. The duration of therapy should be guided by the severity and location of infection and clinical response.
2.9 Incompatible Injectable Drug Products
RECARBRIO for injection for intravenous infusion is physically incompatible with propofol in 5% Dextrose USP or 0.9% Sodium Chloride USP.
13.2 Animal Toxicology And/or Pharmacology (13.2 Animal Toxicology and/or Pharmacology)
Relebactam given as a single entity caused renal tubular degeneration in monkeys at AUC exposure 7-fold the human AUC exposure at the MRHD. Renal tubular degeneration was shown to be reversible after dose discontinuation. There was no evidence of nephrotoxicity at AUC exposures less than or equal to 3-fold the human AUC exposure at the MRHD.
5.5 Development of Drug Resistant Bacteria (5.5 Development of Drug-resistant Bacteria)
Prescribing RECARBRIO in the absence of a proven or strongly suspected bacterial infection or prophylactic indication is unlikely to provide benefit to the patient and increases the risk of the development of drug-resistant bacteria.
1.3 Complicated Intra Abdominal Infections (ciai) (1.3 Complicated Intra-abdominal Infections (cIAI))
RECARBRIO is indicated in adult and pediatric patients weighing at least 2 kg who have limited or no alternative treatment options for the treatment of complicated intra-abdominal infections (cIAI) caused by the following susceptible gram-negative microorganisms: Bacteroides caccae, Bacteroides fragilis, Bacteroides ovatus, Bacteroides stercoris, Bacteroides thetaiotaomicron, Bacteroides uniformis, Bacteroides vulgatus, Citrobacter freundii, Enterobacter cloacae, Escherichia coli, Fusobacterium nucleatum, Klebsiella aerogenes, Klebsiella oxytoca, Klebsiella pneumoniae, Parabacteroides distasonis, and Pseudomonas aeruginosa.
Approval of this indication is based on limited clinical safety and efficacy data for RECARBRIO [see Clinical Studies (14.2)].
Principal Display Panel 1.25 G Vial Carton Label (PRINCIPAL DISPLAY PANEL - 1.25 g Vial Carton Label)
NDC 0006-3856-02
Recarbrio™
(imipenem, cilastatin, and relebactam)
for Injection
1.25 g per vial*
Must be constituted and further diluted.
See enclosed package insert for preparation instructions.
For Intravenous Infusion Only
*Each vial contains imipenem 500 mg (equivalent to 530 mg imipenem monohydrate), cilastatin 500 mg (equivalent
to 531 mg cilastatin sodium), and relebactam 250 mg (equivalent to 263 mg relebactam monohydrate).
Inactive ingredient: 20 mg sodium bicarbonate added to each vial as a buffer.
25 single-dose vials
Rx only
1.5 Usage to Reduce Development of Drug Resistant Bacteria (1.5 Usage to Reduce Development of Drug-Resistant Bacteria)
To reduce the development of drug-resistant bacteria and maintain the effectiveness of RECARBRIO and other antibacterial drugs, RECARBRIO should be used only to treat or prevent infections that are proven or strongly suspected to be caused by susceptible bacteria. When culture and susceptibility information are available, they should be considered in selecting or modifying antibacterial therapy. In the absence of such data, local epidemiology and susceptibility patterns may contribute to the empiric selection of therapy.
2.3 Recommended Dosage in Adult Patients With Renal Impairment (2.3 Recommended Dosage in Adult Patients with Renal Impairment)
Adult patients who have a CLcr less than 90 mL/min require dosage reduction of RECARBRIO. The recommended dosage of RECARBRIO in adult patients with renal impairment is shown in Table 2.
| Creatinine Clearance (CLcr) ([mL/min)] CLcr calculated using the Cockcroft-Gault formula for adult patients.
|
Dose RECARBRIO is provided as a single vial in a fixed-dose combination; the dose for each component will be adjusted equally during preparation [see Dosage and Administration (2.5)].
|
Dosing Frequency | Infusion Duration |
|---|---|---|---|
| 60 to 89 | RECARBRIO 1 gram Provides 400 mg imipenem, 400 mg cilastatin, and 200 mg relebactam
|
Every 6 hours | 30 minutes |
| 30 to 59 | RECARBRIO 0.75 grams Provides 300 mg imipenem, 300 mg cilastatin, and 150 mg relebactam
|
Every 6 hours | 30 minutes |
| 15 to 29 not receiving hemodialysis | RECARBRIO 0.5 grams Provides 200 mg imipenem, 200 mg cilastatin, and 100 mg relebactam
|
Every 6 hours | 30 minutes |
| Receiving hemodialysis Administration should be timed to follow hemodialysis and at intervals timed from the end of that hemodialysis session.
|
RECARBRIO 0.5 grams | Every 6 hours | 30 minutes |
| Less than 15 not receiving hemodialysis | Not recommended |
Do not administer RECARBRIO to adult patients with CLcr less than 15 mL/min unless hemodialysis is instituted within 48 hours after dose administration.
Imipenem, cilastatin, and relebactam are cleared from the circulation during hemodialysis. For adult patients maintained on hemodialysis, administer the recommended dose of RECARBRIO after hemodialysis and at intervals timed from the end of that hemodialysis session.
There is inadequate information to recommend dosage of RECARBRIO for adults undergoing peritoneal dialysis.
2.2 Recommended Dosage in Pediatric Patients Weighing At Least 2 Kg (2.2 Recommended Dosage in Pediatric Patients Weighing at Least 2 kg)
The recommended dosage of RECARBRIO in pediatric patients weighing at least 2 kg varies by patient weight and age, with specific dosing recommendations shown in Table 1. The recommended duration of treatment with RECARBRIO is 4 days to 14 days. The duration of therapy should be guided by the severity and location of infection and clinical response.
RECARBRIO is not recommended in pediatric patients less than 37 weeks post-menstrual age (gestational age at birth plus post-natal age) [see Indications and Usage (1.4) and Use in Specific Populations (8.4)].
RECARBRIO is not recommended for pediatric patients weighing less than 30 kg with renal impairment [see Dosage and administration (2.4) and Use in Specific Populations (8.4)].
| Age Range | Body Weight | Dose | Dosing Frequency | Infusion Duration |
|---|---|---|---|---|
| Birth Pediatric patients from birth (includes pediatric patients at least 37 weeks post-menstrual age) to less than 3 months |
2 kg or greater | RECARBRIO 37.5 Provides 15 mg/kg imipenem, 15 mg/kg cilastatin, and 7.5 mg/kg relebactam mg/kg |
Every 8 hours | 60 minutes |
| 3 months to less than 18 years | less than 30 kg | RECARBRIO 37.5 mg/kg | Every 6 hours | 60 minutes |
| 3 months to less than 18 years | 30 kg or greater | RECARBRIO 1.25 Provides 500 mg imipenem, 500 mg cilastatin, and 250 mg, relebactam grams |
Every 6 hours | 30 minutes |
2.5 Preparation of Recarbrio Solution for Intravenous Administration (2.5 Preparation of RECARBRIO Solution for Intravenous Administration)
RECARBRIO is supplied as a dry powder in a single-dose vial that must be constituted and further diluted using aseptic technique prior to intravenous infusion. To prepare the infusion solution, contents of the vial must be constituted with the appropriate diluent as instructed below. A list of appropriate diluents is as follows:
- 0.9% Sodium Chloride Injection, USP
- 5% Dextrose Injection, USP
- 5% Dextrose Injection, USP + 0.9% Sodium Chloride Injection, USP
- 5% Dextrose Injection, USP + 0.45% Sodium Chloride Injection, USP
- 5% Dextrose Injection, USP + 0.225% Sodium Chloride Injection, USP
RECARBRIO has low aqueous solubility. To ensure complete dissolution of RECARBRIO it is important to adhere to the following instructions:
-
Step 1) For diluents available in 100 mL prefilled infusion bags, proceed to step 2. For diluents not available in 100 mL prefilled infusion bags, aseptically withdraw 100 mL of the desired diluent and transfer it to an empty infusion bag, then proceed to step 2.
-
Step 2) Withdraw 20 mL (as two 10 mL aliquots) of diluent from the appropriate infusion bag and constitute the vial with one 10 mL aliquot of the diluent. The constituted suspension is for intravenous infusion only after dilution in an appropriate infusion solution.
-
Step 3) After constitution, shake vial well and transfer resulting suspension into the remaining 80 mL of the infusion bag.
-
Step 4) Add the second 10 mL aliquot of infusion diluent to the vial and shake well to ensure complete transfer of vial contents; repeat transfer of the resulting suspension to the infusion solution before administering. Agitate the resulting mixture until clear.
Constituted solutions of RECARBRIO range from colorless to yellow. Variations of color within this range do not affect the potency of the product.
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit. Discard if discoloration or visible particles are observed.
The above instructions for preparation of RECARBRIO solution for intravenous administration must be followed for all patients, irrespective of the intended patient’s renal function, age or weight.
The volume of this prepared RECARBRIO solution to be administered in adult and pediatric (weighing at least 30 kg) patients is determined based on estimated renal function [see Dosage and Administration (2.6)].
The volume of the prepared RECARBRIO solution to be administered to pediatric patients weighing 2 kg to less than 30 kg with normal renal function is calculated based on patient weight using the formula below [see Dosage and Administration (2.2)]:
To achieve the appropriate infusion volume, withdraw and discard the excess (100 mL minus the infusion volume) from the prepared 100 mL solution. The remaining volume is the appropriate infusion volume.
5.2 Seizures and Other Central Nervous System (cns) Adverse Reactions (5.2 Seizures and Other Central Nervous System (CNS) Adverse Reactions)
CNS adverse reactions, such as seizures, confusional states, and myoclonic activity, have been reported during treatment with imipenem/cilastatin, a component of RECARBRIO, especially when recommended dosages of imipenem were exceeded. These have been reported most commonly in patients with CNS disorders (e.g., brain lesions or history of seizures) and/or compromised renal function.
Anticonvulsant therapy should be continued in patients with known seizure disorders. If CNS adverse reactions including seizures occur, patients should undergo a neurological evaluation to determine whether RECARBRIO should be discontinued.
5.3 Increased Seizure Potential Due to Interaction With Valproic Acid (5.3 Increased Seizure Potential Due to Interaction with Valproic Acid)
Concomitant use of RECARBRIO, with valproic acid or divalproex sodium may increase the risk of breakthrough seizures. Avoid concomitant use of RECARBRIO with valproic acid or divalproex sodium or consider alternative antibacterial drugs other than carbapenems [see Drug Interactions (7.2)].
1.2 Complicated Urinary Tract Infections (cuti), Including Pyelonephritis (1.2 Complicated Urinary Tract Infections (cUTI), including Pyelonephritis)
RECARBRIO is indicated in adult and pediatric patients weighing at least 2 kg who have limited or no alternative treatment options, for the treatment of complicated urinary tract infections (cUTI), including pyelonephritis, caused by the following susceptible gram-negative microorganisms: Enterobacter cloacae, Escherichia coli, Klebsiella aerogenes, Klebsiella pneumoniae, and Pseudomonas aeruginosa.
Approval of this indication is based on limited clinical safety and efficacy data for RECARBRIO [see Clinical Studies (14.2)].
2.4 Recommended Dosage in Pediatric Patients Weighing At Least 30 Kg With Renal Impairment (2.4 Recommended Dosage in Pediatric Patients Weighing at Least 30 kg with Renal Impairment)
Pediatric patients weighing at least 30 kg who have an estimated glomerular filtration rate (eGFR) less than 90 mL/min/1.73m2 require dosage reduction of RECARBRIO. The recommended dosage of RECARBRIO in pediatric patients weighing at least 30 kg with renal impairment is shown in Table 3. There is insufficient information to recommend a dosage of RECARBRIO for pediatric patients weighing less than 30 kg with any degree of renal impairment.
|
eGFR (mL/min/1.73 m2) Calculated using a GFR estimating equation validated in pediatric patients weighing at least 30 kg.
|
Recommended Dose RECARBRIO is provided as a single vial in a fixed-dose combination; the dose for each component will be adjusted equally during preparation [see Dosage and Administration (2.5)].
|
Dosing Frequency | Infusion Duration |
|---|---|---|---|
| 60 to 89 | RECARBRIO 1 gram Provides 400 mg imipenem, 400 mg cilastatin, and 200 mg relebactam
|
Every 6 hours | 30 minutes |
| 30 to 59 | RECARBRIO 0.75 grams Provides 300 mg imipenem, 300 mg cilastatin, and 150 mg relebactam
|
Every 6 hours | 30 minutes |
| 15 to 29 not receiving hemodialysis | RECARBRIO 0.5 grams Provides 200 mg imipenem, 200 mg cilastatin, and 100 mg relebactam
|
Every 6 hours | 30 minutes |
| Receiving hemodialysis Administration should be timed to follow hemodialysis and at intervals timed from the end of that hemodialysis session.
|
RECARBRIO 0.5 grams | Every 6 hours | 30 minutes |
| Less than 15 not receiving hemodialysis | Not recommended |
Do not administer RECARBRIO to pediatric patients weighing at least 30 kg with an eGFR less than 15 mL/min/1.73m2 unless hemodialysis is instituted within 48 hours after dose administration.
Imipenem, cilastatin, and relebactam are cleared from the circulation during hemodialysis. For pediatric patients weighing at least 30 kg maintained on hemodialysis, administer the recommended dose of RECARBRIO after hemodialysis and at intervals timed from the end of that hemodialysis session.
There is inadequate information to recommend a dosage of RECARBRIO for pediatric patients undergoing peritoneal dialysis.
1.1 Hospital Acquired Bacterial Pneumonia and Ventilator Associated Bacterial Pneumonia (habp/vabp) (1.1 Hospital-acquired Bacterial Pneumonia and Ventilator-associated Bacterial Pneumonia (HABP/VABP))
RECARBRIO® is indicated in adult and pediatric patients weighing at least 2 kg for the treatment of hospital-acquired bacterial pneumonia and ventilator-associated bacterial pneumonia, caused by the following susceptible gram-negative microorganisms: Acinetobacter calcoaceticus-baumannii complex, Enterobacter cloacae, Escherichia coli, Haemophilus influenzae, Klebsiella aerogenes, Klebsiella oxytoca, Klebsiella pneumoniae, Pseudomonas aeruginosa, and Serratia marcescens.
14.1 Hospital Acquired Bacterial Pneumonia and Ventilator Associated Bacterial Pneumonia in Adult Patients (14.1 Hospital-acquired Bacterial Pneumonia and Ventilator-associated Bacterial Pneumonia in Adult Patients)
A total of 535 hospitalized adults with HABP/VABP were randomized and received trial medications in a multinational, double-blind trial (Trial 1, NCT02493764) comparing RECARBRIO 1.25 grams (imipenem 500 mg/cilastatin 500 mg/relebactam 250 mg) intravenously every 6 hours to piperacillin and tazobactam (4.5 grams) for 7 to 14 days of therapy.
The modified intent-to-treat (MITT) population, which included all randomized patients who received at least one dose of trial treatment and did not have only gram-positive cocci on Gram stain of the baseline lower respiratory tract (LRT) specimen included 531 patients; the mean age was 60 and 43% were 65 years of age or older. The majority of patients were men (69%), white (78%), and from Europe (61%). The mean APACHE II score was 15 and 47% of the population had an APACHE II score of ≥15. At randomization, 66% of patients were admitted to the ICU, 77% had been in the hospital for ≥5 days, and 48% had a creatinine clearance of <90 mL/min. Concurrent bacteremia was present at baseline in 5.8% of patients.
Table 10 presents the incidence of all-cause mortality through Day 28 and clinical response at the early follow-up (EFU) visit (7 to 14 days after the end of therapy) in the MITT population. Overall results are presented along with subgroup results by pneumonia diagnosis.
| RECARBRIO | Piperacillin/Tazobactam | Treatment Difference | ||||
|---|---|---|---|---|---|---|
| n/m | (%) | n/m | (%) | % Treatment differences and 95% confidence intervals are based on Miettinen & Nurminen method.
|
(95% CI) | |
| EFU = early follow up | ||||||
|
All-Cause Mortality Through Day 28
n/m = number of subjects with survival status of death or unknown / number of modified intent-to-treat subjects.
,
One subject in the RECARBRIO arm had unknown mortality status at Day 28 which was counted as a death.
|
42/264 | (15.9) | 57/267 | (21.3) | -5.3 | (-11.9, 1.2) |
| Non-ventilated HABP | 18/142 | (12.7) | 15/131 | (11.5) | 1.2 | (-6.8, 9.1) |
| Ventilated HABP/VABP | 24/122 | (19.7) | 42/136 | (30.9) | -11.2 | (-21.6, -0.5) |
|
Clinical Response at EFU
n/m = number of subjects with a favorable clinical response / number of modified intent-to-treat subjects.
|
161/264 | (61.0) | 149/267 | (55.8) | 5.0 | (-3.2, 13.2) |
| Non-ventilated HABP | 95/142 | (66.9) | 87/131 | (66.4) | 0.5 | (-10.7, 11.7) |
| Ventilated HABP/VABP | 66/122 | (54.1) | 62/136 | (45.6) | 8.5 | (-3.7, 20.5) |
In the MITT population, in patients with an APACHE II score <15, Day 28 all-cause mortality rates were 17/139 (12.2%) for RECARBRIO-treated patients and 12/140 (8.6%) for piperacillin/tazobactam-treated patients, clinical cure rates were 90/139 (64.7%) and 98/140 (70%), respectively. In patients with an APACHE II score ≥15, Day 28 all-cause mortality rates were 25/125 (20%) for RECARBRIO-treated patients and 45/127 (35.4%) for piperacillin/tazobactam-treated patients, clinical cure rates were 71/125 (56.8%) and 51/127 (40.2%), respectively.
Per pathogen favorable clinical response at EFU and Day 28 all-cause mortality were assessed in a microbiological modified intention to treat (mMITT) population, which consisted of all randomized MITT subjects who had at least one baseline LRT pathogen that was susceptible to both study treatments (Table 11).
| Baseline LRT Pathogen |
Day 28 All-Cause Mortality | Clinical Response at EFU | ||
|---|---|---|---|---|
| RECARBRIO n/m n/m = the number of subjects with survival status of death or unknown within each category / the number of microbiological modified intent-to-treat subjects who have the corresponding baseline pathogen from LRT culture. (%) |
Piperacillin/ Tazobactam n/m (%) |
RECARBRIO n/m n/m = the number of subjects with a favorable clinical response within each category / the number of microbiological modified intent-to-treat subjects who have the corresponding baseline pathogen from LRT culture. (%) |
Piperacillin/ Tazobactam n/m (%) |
|
| LRT = lower respiratory tract EFU = early follow-up |
||||
|
Acinetobacter calcoaceticus- baumannii complex |
0/5 Supportive evidence was derived from the imipenem and cilastatin prescribing information. (0.0) |
1/10 (10.0) | 4/5 (80.0) | 6/10 (60.0) |
|
Enterobacter cloacae |
1/7 (14.3) | 3/16 (18.8) | 6/7 (85.7) | 12/16 (75.0) |
| Escherichia coli | 5/27(18.5) | 8/33 (24.2) | 16/27 (59.3) | 19/33 (57.6) |
|
Haemophilus influenzae All H. influenzae isolates were susceptible to imipenem. The susceptible MIC breakpoint for PIP/TAZ is ≤1/4 mcg/mL. At the lowest concentration of PIP/TAZ tested (2/4 mcg/mL) there was no visible growth.
|
2/13 (15.4) | 3/12 (25.0) | 9/13 (69.2) | 8/12 (66.7) |
|
Klebsiella spp Includes Klebsiella aerogenes, Klebsiella oxytoca, Klebsiella pneumoniae.
|
6/42 (14.3) | 8/41 (19.5) | 25/42 (59.5) | 28/41 (68.3) |
|
Pseudomonas aeruginosa |
7/26 (26.9) | 5/35 (14.3) | 12/26 (46.2) | 20/35 (57.1) |
|
Serratia marcescens |
2/10 (20.0) | 1/4 (25.0) | 7/10 (70.0) | 3/4 (75.0) |
14.2 Complicated Urinary Tract Infections, Including Pyelonephritis and Complicated Intra Abdominal Infections in Adult Patients (14.2 Complicated Urinary Tract Infections, including Pyelonephritis and Complicated Intra-abdominal Infections in Adult Patients)
The determination of efficacy and safety of RECARBRIO was supported in part by the previous findings of the efficacy and safety of imipenem/cilastatin for the treatment of cUTI and cIAI. The contribution of relebactam to RECARBRIO was primarily established in vitro and in animal models of infection [see Microbiology (12.4)]. Imipenem/cilastatin plus relebactam was studied in cUTI including pyelonephritis (Trial 2, NCT01505634) and cIAI (Trial 3, NCT01506271) in randomized, blinded, active-controlled, multicenter trials. These trials provided only limited efficacy and safety information.
2.6 Preparation of Recarbrio Solution for Intravenous Administration in Adult and Pediatric Patients Weighing At Least 30 Kg With Renal Impairment (2.6 Preparation of RECARBRIO Solution for Intravenous Administration in Adult and Pediatric Patients Weighing at Least 30 kg with Renal Impairment)
Prepare a reduced dose of RECARBRIO (1 gram, 0.75 grams, or 0.5 grams) [see Dosage and Administration (2.3, 2.4)] by preparing a 100 mL solution containing 1.25 grams as described above [see Dosage and Administration (2.5)] then withdrawing and discarding the excess according to Table 4.
|
Estimated Renal Function Creatinine Clearance (mL/min) Calculated using the Cockcroft-Gault formula for adult patients
Or Estimated Glomerular Filtration Rate eGFR (mL/min/1.73 m2) Calculated using a GFR estimating equation validated in pediatric patients weighing at least 30 kg
|
Dose | After preparation as instructed above, remove from the 100 mL prepared bag the volume indicated below and discard | Resulting volume that provides the indicated reduced dose |
|---|---|---|---|
| 60 to 89 | RECARBRIO 1 gram Provides 400 mg imipenem, 400 mg cilastatin, and 200 mg relebactam
|
20 mL | 80 mL |
| 30 to 59 | RECARBRIO 0.75 grams Provides 300 mg imipenem, 300 mg cilastatin, and 150 mg relebactam
|
40 mL | 60 mL |
| 15 to 29 not receiving hemodialysis | RECARBRIO 0.5 grams Provides 200 mg imipenem, 200 mg cilastatin, and 100 mg relebactam
|
60 mL | 40 mL |
| Receiving hemodialysis | RECARBRIO 0.5 grams | 60 mL | 40 mL |
14.3 Hospital Acquired Bacterial Pneumonia and Ventilator Associated Bacterial Pneumonia (habp/vabp), Complicated Urinary Tract Infections (cuti), and Complicated Intra Abdominal Infections (ciai) in Pediatric Patients (14.3 Hospital-acquired Bacterial Pneumonia and Ventilator-associated Bacterial Pneumonia (HABP/VABP), Complicated Urinary Tract Infections (cUTI), and Complicated Intra-abdominal Infections (cIAI) in Pediatric Patients)
The safety and efficacy of RECARBRIO in pediatric patients were investigated in a randomized, active-controlled, open-label trial that enrolled hospitalized patients from birth to less than 18 years of age with HABP/VABP, cUTI, or cIAI (Trial 4, NCT03969901). Eligible patients were randomized in a 3:1 ratio with stratification by age group and infection type to receive IV RECARBRIO or active control (defined as investigator’s choice of specified comparators). Patients with HABP/VABP received IV therapy for 7 to 14 days. Patients with cUTI or cIAI received IV therapy for a minimum of 3 days before an optional switch to oral step-down therapy at the discretion of the investigator to complete a total of the 5 to 14 days of antibacterial therapy.
The modified intention to treat (MITT) population consisted of 113 patients who were randomized and received at least one dose of trial treatment (RECARBRIO, n=85; active control, n=28). In patients treated with RECARBRIO, 49% were male and the median age was 5 years (range 21 days to 17 years). The age groups who received RECARBRIO were as follows: 12 to <18 years (n=10), 6 to <12 years (n=31), 2 to <6 years (n=21), 3 months to <2 years (n=15), and birth to <3 months (n=8). The MITT population included 54 patients (48%) with cUTI, 53 (47%) with cIAI, and 6 (5%) with HABP/VABP. Patients were predominantly white (81%) and from Europe (61%).
The primary objective of the study was to evaluate the safety and tolerability of RECARBRIO. Efficacy assessments were not powered for formal hypothesis testing between treatment groups. For HABP/VABP and cIAI, Table 12 presents the clinical cure rates at early follow-up (EFU, 7 to 14 days after the end of therapy) in the MITT population. For cUTI, Table 13 presents the clinical cure and microbiologic response rates at EFU in the mMITT population, which included all patients with cUTI in the MITT population that had a baseline pathogen isolated. No patient died in either intervention group in the study through Day 28.
| RECARBRIO n/N (%) |
Active Control n/N (%) |
|
|---|---|---|
| HABP/VABP | 5/5 (100.0) | 1/1 (100.0) |
| cIAI | 34/39 (87.2) | 13/14 (92.9) |
|
RECARBRIO n/N (%) |
ACTIVE Control n/N (%) |
|
|---|---|---|
| Clinical Cure | 20/30 (66.7) | 7/10 (70.0) |
| Microbiologic Response | 20/30 (66.7) | 8/10 (80.0) |
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:51:23.266323 · Updated: 2026-03-14T22:40:12.922880