These Highlights Do Not Include All The Information Needed To Use Jaypirca Safely And Effectively. See Full Prescribing Information For Jaypirca.
bd551845-0878-49a4-860f-839b83f6b801
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Indications and Usage, Chronic Lymphocytic Leukemia/Small Lymphocytic Lymphoma ( 1.2 ) 12/2025
Indications and Usage
JAYPIRCA ® is a kinase inhibitor indicated for the treatment of: Adult patients with relapsed or refractory mantle cell lymphoma (MCL) after at least two lines of systemic therapy, including a BTK inhibitor. ( 1.1 ). This indication is approved under accelerated approval based on response rate. Continued approval for this indication may be contingent upon verification and description of clinical benefit in a confirmatory trial. Adult patients with relapsed or refractory chronic lymphocytic leukemia or small lymphocytic lymphoma (CLL/SLL) who have previously been treated with a covalent BTK inhibitor. ( 1.2 ).
Dosage and Administration
Recommended dosage: 200 mg orally once daily; swallow whole with water, with or without food. Do not cut, crush, or chew tablets. ( 2.1 ) Manage toxicity using treatment interruption, dosage reduction, or discontinuation. ( 2.2 ) Reduce dose in patients with severe renal impairment. ( 2.3 , 8.6 )
Warnings and Precautions
Infections: Monitor for signs and symptoms of infection, evaluate promptly, and treat. ( 5.1 ) Hemorrhage: Monitor for bleeding and manage appropriately. ( 5.2 ) Cytopenias: Monitor complete blood counts during treatment. ( 5.3 ) Cardiac Arrythmias: Monitor for symptoms of arrhythmias and manage appropriately. ( 5.4 ) Second Primary Malignancies: Other malignancies have developed, including skin cancers and other carcinomas. Monitor and advise patients to use sun protection. ( 5.5 ) Hepatotoxicity, Including Drug-Induced Liver Injury: Monitor hepatic function throughout treatment. ( 5.6 ) Embryo-Fetal Toxicity: Can cause fetal harm. Advise females of reproductive potential of potential risk to a fetus and to use effective contraception. ( 5.7 , 8.1 , 8.3 )
Contraindications
None.
Adverse Reactions
Recommended dosage modifications of JAYPIRCA for adverse reactions are presented in Table 1 [see Warnings and Precautions ( 5.1 , 5.2 , 5.3 , and 5.4 )] . Table 1: Recommended Dosage Modification of JAYPIRCA for Adverse Reactions Dose modification is not recommended for asymptomatic lymphocytosis. Asymptomatic lipase increase may not necessarily warrant dose modification. a Evaluate the benefit-risk before resuming treatment at the same dose for a Grade 4 non-hematological toxicity. Adverse Reaction Occurrences Requiring Dosage Modification Modification (Starting Dosage: 200 mg once daily) Grade 3 or greater non-hematologic toxicity a Absolute neutrophil count < 1 to 0.5 x 10 9 /L with fever and/or infection Absolute neutrophil count < 0.5 x 10 9 /L lasting 7 or more days Platelet count < 50 to 25 x 10 9 /L with bleeding Platelet count < 25 x 10 9 /L First occurrence Interrupt JAYPIRCA until recovery to Grade 1 or baseline; restart at original dosage (200 mg once daily) a . Second occurrence Interrupt JAYPIRCA until recovery to Grade 1 or baseline; restart at 100 mg once daily. Third occurrence Interrupt JAYPIRCA until recovery to Grade 1 or baseline; restart at 50 mg once daily. Fourth occurrence Discontinue JAYPIRCA.
Drug Interactions
Strong CYP3A Inhibitors: Avoid concomitant use. If concomitant use is unavoidable, reduce the JAYPIRCA dose. ( 2.4 , 7.1 ) Strong or Moderate CYP3A Inducers: Avoid concomitant use. If concomitant use of moderate CYP3A inducers is unavoidable, increase the JAYPIRCA dose. ( 2.5 , 7.1 ) Sensitive CYP2C8, CYP2C19, CYP3A, P-gp, or BCRP Substrates: For substrates where minimal concentration changes may increase the risk of adverse reactions, follow recommendations for co-administration with CYP2C8, CYP2C19, CYP3A, P-gp, or BCRP inhibitors provided in their approved product labeling. ( 7.2 )
Storage and Handling
How Supplied JAYPIRCA tablets are supplied as follows: Tablet Strength Description Package Configuration NDC Number 50 mg Blue, film coated, arc-triangle shaped tablets debossed with “Lilly 50” on one side and “6902” on the other side. Bottle with child-resistant closure. Each bottle contains 30 tablets. 0002-6902-30 100 mg Blue, film coated, round tablets debossed with “Lilly 100” on one side and “7026” on the other side. Bottle with child-resistant closure. Each bottle contains 60 tablets. 0002-7026-60 Storage and Handling Store JAYPIRCA tablets at room temperature 20°C to 25°C (68°F to 77°F); excursions permitted between 15°C and 30°C (59°F and 86°F) ([see USP Controlled Room Temperature]).
How Supplied
How Supplied JAYPIRCA tablets are supplied as follows: Tablet Strength Description Package Configuration NDC Number 50 mg Blue, film coated, arc-triangle shaped tablets debossed with “Lilly 50” on one side and “6902” on the other side. Bottle with child-resistant closure. Each bottle contains 30 tablets. 0002-6902-30 100 mg Blue, film coated, round tablets debossed with “Lilly 100” on one side and “7026” on the other side. Bottle with child-resistant closure. Each bottle contains 60 tablets. 0002-7026-60 Storage and Handling Store JAYPIRCA tablets at room temperature 20°C to 25°C (68°F to 77°F); excursions permitted between 15°C and 30°C (59°F and 86°F) ([see USP Controlled Room Temperature]).
Medication Information
Warnings and Precautions
Infections: Monitor for signs and symptoms of infection, evaluate promptly, and treat. ( 5.1 ) Hemorrhage: Monitor for bleeding and manage appropriately. ( 5.2 ) Cytopenias: Monitor complete blood counts during treatment. ( 5.3 ) Cardiac Arrythmias: Monitor for symptoms of arrhythmias and manage appropriately. ( 5.4 ) Second Primary Malignancies: Other malignancies have developed, including skin cancers and other carcinomas. Monitor and advise patients to use sun protection. ( 5.5 ) Hepatotoxicity, Including Drug-Induced Liver Injury: Monitor hepatic function throughout treatment. ( 5.6 ) Embryo-Fetal Toxicity: Can cause fetal harm. Advise females of reproductive potential of potential risk to a fetus and to use effective contraception. ( 5.7 , 8.1 , 8.3 )
Indications and Usage
JAYPIRCA ® is a kinase inhibitor indicated for the treatment of: Adult patients with relapsed or refractory mantle cell lymphoma (MCL) after at least two lines of systemic therapy, including a BTK inhibitor. ( 1.1 ). This indication is approved under accelerated approval based on response rate. Continued approval for this indication may be contingent upon verification and description of clinical benefit in a confirmatory trial. Adult patients with relapsed or refractory chronic lymphocytic leukemia or small lymphocytic lymphoma (CLL/SLL) who have previously been treated with a covalent BTK inhibitor. ( 1.2 ).
Dosage and Administration
Recommended dosage: 200 mg orally once daily; swallow whole with water, with or without food. Do not cut, crush, or chew tablets. ( 2.1 ) Manage toxicity using treatment interruption, dosage reduction, or discontinuation. ( 2.2 ) Reduce dose in patients with severe renal impairment. ( 2.3 , 8.6 )
Contraindications
None.
Adverse Reactions
Recommended dosage modifications of JAYPIRCA for adverse reactions are presented in Table 1 [see Warnings and Precautions ( 5.1 , 5.2 , 5.3 , and 5.4 )] . Table 1: Recommended Dosage Modification of JAYPIRCA for Adverse Reactions Dose modification is not recommended for asymptomatic lymphocytosis. Asymptomatic lipase increase may not necessarily warrant dose modification. a Evaluate the benefit-risk before resuming treatment at the same dose for a Grade 4 non-hematological toxicity. Adverse Reaction Occurrences Requiring Dosage Modification Modification (Starting Dosage: 200 mg once daily) Grade 3 or greater non-hematologic toxicity a Absolute neutrophil count < 1 to 0.5 x 10 9 /L with fever and/or infection Absolute neutrophil count < 0.5 x 10 9 /L lasting 7 or more days Platelet count < 50 to 25 x 10 9 /L with bleeding Platelet count < 25 x 10 9 /L First occurrence Interrupt JAYPIRCA until recovery to Grade 1 or baseline; restart at original dosage (200 mg once daily) a . Second occurrence Interrupt JAYPIRCA until recovery to Grade 1 or baseline; restart at 100 mg once daily. Third occurrence Interrupt JAYPIRCA until recovery to Grade 1 or baseline; restart at 50 mg once daily. Fourth occurrence Discontinue JAYPIRCA.
Drug Interactions
Strong CYP3A Inhibitors: Avoid concomitant use. If concomitant use is unavoidable, reduce the JAYPIRCA dose. ( 2.4 , 7.1 ) Strong or Moderate CYP3A Inducers: Avoid concomitant use. If concomitant use of moderate CYP3A inducers is unavoidable, increase the JAYPIRCA dose. ( 2.5 , 7.1 ) Sensitive CYP2C8, CYP2C19, CYP3A, P-gp, or BCRP Substrates: For substrates where minimal concentration changes may increase the risk of adverse reactions, follow recommendations for co-administration with CYP2C8, CYP2C19, CYP3A, P-gp, or BCRP inhibitors provided in their approved product labeling. ( 7.2 )
Storage and Handling
How Supplied JAYPIRCA tablets are supplied as follows: Tablet Strength Description Package Configuration NDC Number 50 mg Blue, film coated, arc-triangle shaped tablets debossed with “Lilly 50” on one side and “6902” on the other side. Bottle with child-resistant closure. Each bottle contains 30 tablets. 0002-6902-30 100 mg Blue, film coated, round tablets debossed with “Lilly 100” on one side and “7026” on the other side. Bottle with child-resistant closure. Each bottle contains 60 tablets. 0002-7026-60 Storage and Handling Store JAYPIRCA tablets at room temperature 20°C to 25°C (68°F to 77°F); excursions permitted between 15°C and 30°C (59°F and 86°F) ([see USP Controlled Room Temperature]).
How Supplied
How Supplied JAYPIRCA tablets are supplied as follows: Tablet Strength Description Package Configuration NDC Number 50 mg Blue, film coated, arc-triangle shaped tablets debossed with “Lilly 50” on one side and “6902” on the other side. Bottle with child-resistant closure. Each bottle contains 30 tablets. 0002-6902-30 100 mg Blue, film coated, round tablets debossed with “Lilly 100” on one side and “7026” on the other side. Bottle with child-resistant closure. Each bottle contains 60 tablets. 0002-7026-60 Storage and Handling Store JAYPIRCA tablets at room temperature 20°C to 25°C (68°F to 77°F); excursions permitted between 15°C and 30°C (59°F and 86°F) ([see USP Controlled Room Temperature]).
Description
Indications and Usage, Chronic Lymphocytic Leukemia/Small Lymphocytic Lymphoma ( 1.2 ) 12/2025
Section 42229-5
Hepatotoxicity, including severe, life-threatening, and potentially fatal cases of drug-induced liver injury (DILI), has occurred in patients treated with Bruton tyrosine kinase inhibitors, including JAYPIRCA.
Evaluate bilirubin and transaminases at baseline and throughout treatment with JAYPIRCA. For patients who develop abnormal liver tests after JAYPIRCA, monitor more frequently for liver test abnormalities and clinical signs and symptoms of hepatic toxicity. If DILI is suspected, withhold JAYPIRCA. Upon confirmation of DILI, discontinue JAYPIRCA.
Section 42230-3
|
This Patient Information has been approved by the U.S. Food and Drug Administration. |
Revised: 12/2025 |
|||
|
PATIENT INFORMATION
JAYPIRCA® (JAY-PIHR-KAA) pirtobrutinib tablets |
||||
|
What is JAYPIRCA?
JAYPIRCA is a prescription medicine used to treat adults with:
|
||||
| It is not known if JAYPIRCA is safe and effective in children. | ||||
Before taking JAYPIRCA, tell your healthcare provider about all of your medical conditions, including if you:
|
||||
| Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. Taking JAYPIRCA with certain other medicines may affect how JAYPIRCA or the other medicines work and can cause side effects. | ||||
How should I take JAYPIRCA?
|
||||
|
What are the possible side effects of JAYPIRCA?
JAYPIRCA can cause serious side effects, including:
|
||||
| Your healthcare provider may decrease your dose, temporarily stop, or permanently stop treatment with JAYPIRCA if you develop severe side effects. The most common side effects of JAYPIRCA include: |
||||
|
|
|||
| These are not all the possible side effects of JAYPIRCA. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
||||
How should I store JAYPIRCA?
|
||||
| Keep JAYPIRCA and all medicines out of the reach of children. | ||||
|
General information about the safe and effective use of JAYPIRCA.
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use JAYPIRCA for a condition for which it was not prescribed. Do not give JAYPIRCA to other people, even if they have the same symptoms that you have. It may harm them. You can ask your pharmacist or healthcare provider for information about JAYPIRCA that is written for health professionals. |
||||
|
What are the ingredients in JAYPIRCA?
Active ingredient: pirtobrutinib Inactive ingredients: croscarmellose sodium, hypromellose acetate succinate, lactose monohydrate, magnesium stearate, microcrystalline cellulose, and silicon dioxide. The tablet film coating contains FD&C Blue #2, hypromellose, titanium dioxide, and triacetin. Marketed by Lilly USA, LLC, Indianapolis, IN 46285, USA JAYPIRCA is a registered trademark of Eli Lilly and Company. Copyright © 2023, 2025, Eli Lilly and Company. All rights reserved. JAY-0005-PPI-20251203 For more information, go to www.jaypirca.com or call 1-800-LillyRx (1-800-545-5979). |
Section 43683-2
| Indications and Usage, Chronic Lymphocytic Leukemia/Small Lymphocytic Lymphoma (1.2) | 12/2025 |
Section 51945-4
PACKAGE LABEL – JAYPIRCA 50 mg Tablets, 30 count bottle
NDC 0002-6902-30
30 tablets Rx only
Jaypirca®
(pirtobrutinib)
tablets
50 mg
Each tablet contains 50 mg pirtobrutinib
www.jaypirca.com
Lilly
11 Description
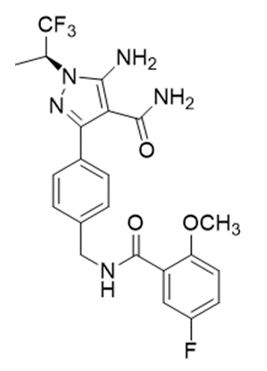
Pirtobrutinib is a kinase inhibitor. It is an orally available, small molecule ATP-competitive inhibitor of BTK. The active pharmaceutical ingredient is pirtobrutinib with the molecular formula C22H21F4N5O3 and a molecular weight of 479.44 g/mol. The chemical name for pirtobrutinib is 5-amino-3-{4-[(5-fluoro-2-methoxybenzamido)methyl]phenyl}-1-[(2S)-1,1,1-trifluoropropan-2-yl]-1H-pyrazole-4-carboxamide.
Pirtobrutinib is a white to practically white to yellow to brown solid. The aqueous solubility of pirtobrutinib is considered practically insoluble, or insoluble, across the pH 1 to pH 7 range.
Pirtobrutinib tablets are supplied as 50 mg or 100 mg film-coated, debossed tablets for oral administration. Each tablet contains inactive ingredients of croscarmellose sodium, hypromellose acetate succinate, lactose monohydrate, magnesium stearate, microcrystalline cellulose and silicon dioxide. The tablet film coating material contains FD&C Blue #2, hypromellose, titanium dioxide and triacetin.
5.1 Infections
Fatal and serious infections (including bacterial, viral, or fungal infections) and opportunistic infections have occurred in patients treated with JAYPIRCA. Across clinical trials, Grade 3 or higher infections occurred in 25% of 704 patients, most commonly pneumonia (20%), with fatal infections occurring in 5% of patients. Sepsis occurred in 6% of patients and febrile neutropenia in 3.8%. In patients with CLL/SLL, Grade 3 or higher infections occurred in 32% of patients, with fatal infections occurring in 8%. Opportunistic infections after treatment with JAYPIRCA have included, but are not limited to, Pneumocystis jirovecii pneumonia and fungal infection [see Adverse Reactions (6.1)].
Consider prophylaxis, including vaccinations and antimicrobial prophylaxis, in patients who are at increased risk for infections, including opportunistic infections. Monitor patients for signs and symptoms of infection, evaluate promptly, and treat appropriately. Based on severity, reduce dose, temporarily withhold, or permanently discontinue JAYPIRCA [see Dosage and Administration (2.2)].
5.2 Hemorrhage
Fatal and serious hemorrhage has occurred with JAYPIRCA. Major hemorrhage (defined as Grade 3 or higher bleeding or any central nervous system bleeding) occurred in 2.6% of 704 patients treated with JAYPIRCA, including gastrointestinal hemorrhage; fatal hemorrhage occurred in 0.3% of patients. Bleeding of any grade, excluding bruising and petechiae, occurred in 16% of patients [see Adverse Reactions (6.1)].
Major hemorrhage occurred in 0.6% of patients taking JAYPIRCA without antithrombotic agents and 2.0% of patients taking JAYPIRCA with antithrombotic agents. Consider the risks and benefits of antithrombotic agents when co-administered with JAYPIRCA. Monitor patients for signs of bleeding. Based on severity of bleeding, reduce dose, temporarily withhold, or permanently discontinue JAYPIRCA [see Dosage and Administration (2.2)].
Consider the benefit-risk of withholding JAYPIRCA for 3 to 7 days pre- and post-surgery depending upon the type of surgery and risk of bleeding.
5.3 Cytopenias
JAYPIRCA can cause cytopenias, including neutropenia, thrombocytopenia, and anemia.
Across clinical trials, Grade 3 or 4 cytopenias, including decreased neutrophils (27%), decreased platelets (13%), and decreased hemoglobin (11%) developed in patients treated with JAYPIRCA. Grade 4 decreased neutrophils developed in 15% of patients and Grade 4 decreased platelets developed in 6% of patients [see Adverse Reactions (6.1)].
Monitor complete blood counts regularly during treatment. Based on severity, reduce dose, temporarily withhold, or permanently discontinue JAYPIRCA [see Dosage and Administration (2.2)].
8.4 Pediatric Use
Safety and effectiveness of JAYPIRCA have not been established in pediatric patients.
8.5 Geriatric Use
Of the patients with MCL who received the 200 mg dose of JAYPIRCA in BRUIN, 93 (78%) were 65 years of age and older and 39 (33%) were 75 years and older [see Clinical Studies (14.1)]. Clinical studies of JAYPIRCA did not include sufficient numbers of patients with MCL who were less than 65 years of age to determine whether older patients respond differently from younger adult patients. Of the patients with CLL/SLL who received the 200 mg once daily dose of JAYPIRCA in BRUIN and BRUIN-321, 220 (65%) were 65 years of age and older and 78 (33%) were 75 years and older [see Clinical Studies (14.2)]. No overall differences in effectiveness were observed between younger and older patients.
In the pooled safety population in patients with hematologic malignancies in BRUIN and BRUIN CLL-321, 467 (66%) were 65 years of age and older, while 181 (26%) were 75 years of age and older. Patients aged 65 years and older experienced higher rates of Grade 3 and higher adverse reactions and serious adverse reactions compared to patients who were less than 65 years of age.
4 Contraindications
None.
6 Adverse Reactions
The following clinically significant adverse reactions are described elsewhere in the labeling:
- Infections [see Warnings and Precautions (5.1)]
- Hemorrhage [see Warnings and Precautions (5.2)]
- Cytopenias [see Warnings and Precautions (5.3)]
- Atrial Fibrillation and Atrial Flutter [see Warnings and Precautions (5.4)]
- Second Primary Malignancies [see Warnings and Precautions (5.5)]
- Hepatotoxicity, including DILI [see Warnings and Precautions (5.6)]
7 Drug Interactions
- Strong CYP3A Inhibitors: Avoid concomitant use. If concomitant use is unavoidable, reduce the JAYPIRCA dose. (2.4, 7.1)
- Strong or Moderate CYP3A Inducers: Avoid concomitant use. If concomitant use of moderate CYP3A inducers is unavoidable, increase the JAYPIRCA dose. (2.5, 7.1)
- Sensitive CYP2C8, CYP2C19, CYP3A, P-gp, or BCRP Substrates: For substrates where minimal concentration changes may increase the risk of adverse reactions, follow recommendations for co-administration with CYP2C8, CYP2C19, CYP3A, P-gp, or BCRP inhibitors provided in their approved product labeling. (7.2)
8.6 Renal Impairment
Severe renal impairment (eGFR15-29 mL/min) increases pirtobrutinib exposure [see Clinical Pharmacology (12.3)]. Reduce the JAYPIRCA dosage in patients with severe renal impairment [see Dosage and Administration (2.3)]. No dosage adjustment of JAYPIRCA is recommended in patients with mild (60-89 mL/min) or moderate (30-59 mL/min) renal impairment.
12.2 Pharmacodynamics
At the recommended dosage of 200 mg once daily, pirtobrutinib trough concentrations exceeded the BTK IC96. BTK occupancy is maintained throughout the dosing interval, regardless of the intrinsic rate of BTK turnover.
12.3 Pharmacokinetics
The pharmacokinetics of pirtobrutinib were characterized in healthy subjects and in patients with cancer. Pirtobrutinib exposure (AUC) and Cmax increases proportionally following single oral doses ranging from 300 mg to 800 mg (1.5 to 4 times the approved recommended dosage) and once daily doses ranging from 25 – 300 mg (0.125 to 1.5 times the recommended dosage). Steady state was achieved within 5 days of once daily dosing, and the mean (CV%) accumulation ratio was 1.63 (26.7%) based on AUC after administration of 200 mg dosages.
Following administration of the recommended dosage, the geometric mean (CV%) steady-state AUC and Cmax of pirtobrutinib were 92705 h*ng/mL (39%) and 6503 ng/mL (25%), respectively. The geometric mean (CV%) AUC0-24 and Cmax of pirtobrutinib on Cycle 1 Day 8 were 81800 h*ng/mL (66.6%) and 3670 ng/mL (89.5%), respectively.
2.1 Recommended Dosage
The recommended dosage of JAYPIRCA is 200 mg orally once daily until disease progression or unacceptable toxicity.
Advise patients of the following:
- Swallow tablets whole with water. Do not cut, crush, or chew tablets.
- Take JAYPIRCA at the same time each day. JAYPIRCA may be taken with or without food.
- If a dose of JAYPIRCA is missed by more than 12 hours, do not make up the dose and take the next dose as scheduled.
8.7 Hepatic Impairment
No dosage adjustment of JAYPIRCA is recommended in patients with mild hepatic impairment (total bilirubin ≤ upper limit of normal (ULN) and aspartate aminotransferase (AST) > ULN or total bilirubin > 1 to 1.5 × ULN and any AST), moderate hepatic impairment (total bilirubin > 1.5 to 3 × ULN and any AST), or severe hepatic impairment (total bilirubin > 3 × ULN and any AST) [see Clinical Pharmacology (12.3 )].
1 Indications and Usage
JAYPIRCA® is a kinase inhibitor indicated for the treatment of:
- Adult patients with relapsed or refractory mantle cell lymphoma (MCL) after at least two lines of systemic therapy, including a BTK inhibitor. (1.1).
This indication is approved under accelerated approval based on response rate. Continued approval for this indication may be contingent upon verification and description of clinical benefit in a confirmatory trial. - Adult patients with relapsed or refractory chronic lymphocytic leukemia or small lymphocytic lymphoma (CLL/SLL) who have previously been treated with a covalent BTK inhibitor. (1.2).
5.4 Cardiac Arrhythmias
Cardiac arrhythmias, including atrial fibrillation and atrial flutter, were reported in recipients of JAYPIRCA. Atrial fibrillation or flutter were reported in 3.4% of patients, with Grade 3 or 4 atrial fibrillation or flutter reported in 1.6% of 704 patients across clinical trials [see Adverse Reactions (6.1)]. Other serious cardiac arrhythmias such as supraventricular tachycardia and cardiac arrest occurred in 0.4% of patients. Patients with cardiac risk factors, such as hypertension, or previous arrhythmias may be at increased risk.
Monitor for signs and symptoms of arrhythmias (e.g., palpitations, dizziness, syncope, dyspnea) and manage appropriately. Based on severity, reduce dose, temporarily withhold, or permanently discontinue JAYPIRCA [see Dosage and Administration (2.2)].
1.1 Mantle Cell Lymphoma
JAYPIRCA® is indicated for the treatment of adult patients with relapsed or refractory mantle cell lymphoma (MCL) after at least two lines of systemic therapy, including a BTK inhibitor.
This indication is approved under accelerated approval based on response rate [see Clinical Studies (14.1)]. Continued approval for this indication may be contingent upon verification and description of clinical benefit in a confirmatory trial.
12.1 Mechanism of Action
Pirtobrutinib is a small molecule, noncovalent inhibitor of BTK. BTK is a signaling protein of the B-cell antigen receptor (BCR) and cytokine receptor pathways. In B-cells, BTK signaling results in activation of pathways necessary for B-cell proliferation, trafficking, chemotaxis, and adhesion. Pirtobrutinib binds to wild type BTK and BTK harboring C481 mutations, leading to inhibition of BTK kinase activity. In nonclinical studies, pirtobrutinib inhibited BTK-mediated B-cell CD69 expression and inhibited malignant B-cell proliferation. Pirtobrutinib showed dose-dependent anti-tumor activities in BTK wild type and BTK C481S mutant mouse xenograft models.
14.1 Mantle Cell Lymphoma
The efficacy of JAYPIRCA in patients with MCL was evaluated in BRUIN [NCT03740529], an open-label, international, multicohort, single-arm study of JAYPIRCA as monotherapy. Efficacy was based on 120 patients with MCL treated with JAYPIRCA who were previously treated with a BTK inhibitor. JAYPIRCA was given orally at a dose of 200 mg once daily and was continued until disease progression or unacceptable toxicity. Patients with active central nervous system lymphoma or allogeneic hematopoietic stem cell transplantation (HSCT) or CAR-T cell therapy within 60 days were excluded.
The median age was 71 years (range: 46 to 88 years); 79% were male; 78% were White, 14% Asian, 1.7% Black or African American. Seventy-eight percent of patients had the classic/leukemic variant of MCL, 12% had pleomorphic MCL, and 11% had blastoid MCL. The simplified Mantle Cell Lymphoma International Prognostic Index (sMIPI) score was low in 15%, intermediate in 59%, and high in 26% of patients. Patients received a median number of 3 prior lines of therapy (range: 1 to 9) with 93% having received 2 or more prior lines. All received 1 or more prior lines of therapy containing a BTK inhibitor; other prior therapies included chemoimmunotherapy in 88%, HSCT in 20%, lenalidomide in 18%, and CAR-T therapy in 9%. The most common prior BTK inhibitors received were ibrutinib (67%), acalabrutinib (30%), and zanubrutinib (8%). Patients may have received more than one prior BTK inhibitor; 83% of patients discontinued the last BTK inhibitor for refractory or progressive disease, 10% discontinued for toxicity, and 5% discontinued for other reasons.
Efficacy was based on overall response rate (ORR) and duration of response (DOR), as assessed by an independent review committee (IRC) using 2014 Lugano criteria. Efficacy results are shown in Table 8. Additionally, the Kaplan-Meier estimate for the DOR rate at 6 months was 65.3% (95% CI: 49.8, 77.1).
|
CI, confidence interval; CR, complete response; DOR, duration of response; PR, partial response; NE, not estimable. |
|
|
a PET-CT scans were utilized in response assessments (in 41% of patients), with the remainder being assessed by CT scans only. |
|
|
b ORR using CT scan-based assessments in all patients was 48% (95% CI: 38, 57) and CR rate was 13%. |
|
|
c Based on Kaplan-Meier estimation. Estimated median follow-up was 7.3 months. |
|
| Outcome |
JAYPIRCA 200 mg once daily
(N = 120) |
| Overall Response Rate a,b | |
| ORR, n | 60 (50%) |
| (95% CI, %) | 41, 59 |
| CR, n | 15 (13%) |
| PR, n | 45 (38%) |
| Time to Response | |
| Median (range), months | 1.8 (0.8, 4.2) |
| Duration of Response c | |
| Number censored, n | 36 |
| Median DOR, months (95% CI) | 8.3 (5.7, NE) |
5.7 Embryo Fetal Toxicity
Based on findings in animals, JAYPIRCA can cause fetal harm when administered to a pregnant woman. In animal reproduction studies, administration of pirtobrutinib to pregnant rats during the period of organogenesis caused embryo-fetal toxicity including embryo-fetal mortality and malformations at maternal exposures (AUC) approximately 3-times the recommended dose of 200 mg once daily. Advise pregnant women of the potential risk to a fetus. Advise females of reproductive potential to use effective contraception during treatment with JAYPIRCA and for one week after the last dose [see Use in Specific Populations (8.1, 8.3)].
5 Warnings and Precautions
- Infections: Monitor for signs and symptoms of infection, evaluate promptly, and treat. (5.1)
- Hemorrhage: Monitor for bleeding and manage appropriately. (5.2)
- Cytopenias: Monitor complete blood counts during treatment. (5.3)
- Cardiac Arrythmias: Monitor for symptoms of arrhythmias and manage appropriately. (5.4)
- Second Primary Malignancies: Other malignancies have developed, including skin cancers and other carcinomas. Monitor and advise patients to use sun protection. (5.5)
- Hepatotoxicity, Including Drug-Induced Liver Injury: Monitor hepatic function throughout treatment. (5.6)
- Embryo-Fetal Toxicity: Can cause fetal harm. Advise females of reproductive potential of potential risk to a fetus and to use effective contraception. (5.7, 8.1, 8.3)
2 Dosage and Administration
3 Dosage Forms and Strengths
Tablets:
Each 50 mg tablet is blue, arc-triangle shaped, film-coated, and debossed with “Lilly 50” on one side and “6902” on the other side.
Each 100 mg tablet is blue, round, film-coated, and debossed with “Lilly 100” on one side and “7026” on the other side.
8 Use in Specific Populations
Lactation: Advise not to breastfeed (8.2)
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be compared to rates in the clinical trials of another drug and may not reflect the rates observed in the general patient population.
The data in the WARNINGS AND PRECAUTIONS reflect exposure to JAYPIRCA as a single-agent, administered at 200 mg once daily in 704 patients with hematologic malignancies in the BRUIN and the BRUIN-CLL-321 studies. Among these 704 patients, the median duration of exposure was 12 months; 65% were exposed for at least 6 months and 50% were exposed for at least one year.
In this pooled safety population, the most common (≥ 30%) adverse reactions, including laboratory abnormalities, were decreased neutrophil count (54%), decreased hemoglobin (43%), decreased leukocytes (32%), fatigue (31%), decreased platelets (31%), decreased lymphocyte count (31%), and calcium decreased (30%)
5.5 Second Primary Malignancies
Second primary malignancies, including non-skin carcinomas, developed in 9% of 704 patients treated with JAYPIRCA monotherapy across clinical trials. The most frequent malignancy was non-melanoma skin cancer, reported in 4.4% of 704 patients. Other second primary malignancies included solid tumors (including genitourinary and breast cancers) and melanoma. Advise patients to use sun protection and monitor patients for the development of second primary malignancies.
17 Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Patient Information).
Infections
Advise patients that JAYPIRCA can cause serious infections that may be fatal. Advise patients to report any signs or symptoms of infection (e.g., fever, chills, weakness) [see Warnings and Precautions (5.1)].
Hemorrhage
Inform patients to report signs or symptoms of bleeding. Inform patients that JAYPIRCA may need to be interrupted for major surgeries [see Warnings and Precautions (5.2)].
Cytopenias
Advise patients of the need for periodic monitoring of blood counts during treatment with JAYPIRCA [see Warnings and Precautions (5.3)].
Cardiac Arrhythmias
Counsel patients to report any signs of palpitations, dizziness, fainting, chest discomfort, and shortness of breath [see Warnings and Precautions (5.4 )].
Second Primary Malignancies
Inform patients that other malignancies have been reported in patients who have been treated with JAYPIRCA, including skin cancer and other solid tumors. Advise patients to use sun protection and to have monitoring for development of other cancers [see Warnings and Precautions (5.5)].
Hepatotoxicity, Including Drug-Induced Liver Injury
Inform patients that liver problems, including severe, life-threatening, or fatal hepatitis, drug-induced liver injury and abnormalities in liver tests, may develop during JAYPIRCA treatment. Advise patients to contact their healthcare provider immediately if they experience abdominal discomfort, dark urine, or jaundice [see Warnings and Precautions (5.6)].
Embryo-Fetal Toxicity
Advise pregnant women and females of reproductive potential of the potential risk to a fetus. Advise females of reproductive potential to inform their healthcare provider of a known or suspected pregnancy [see Warnings and Precautions (5.7), Use in Specific Populations (8.1, 8.3)].
Advise females of reproductive potential to use effective contraception during treatment with JAYPIRCA and for one week after the last dose [see Use in Specific Populations (8.3)].
Lactation
Advise women not to breastfeed during treatment with JAYPIRCA and for one week after the last dose [see Use in Specific Populations (8.2)].
Administration
Inform patients to take JAYPIRCA orally once daily at approximately the same time each day with or without food and how to make up a missed dose. Advise patients to swallow tablets whole with water. Advise patients not to cut, crush, or chew tablets [see Dosage and Administration (2.1)].
Marketed by: Lilly USA, LLC, Indianapolis, IN 46285, USA
Copyright © 2023, 2025, Eli Lilly and Company. All rights reserved.
JAY-0005-USPI-20251203
16 How Supplied/storage and Handling
How Supplied
JAYPIRCA tablets are supplied as follows:
| Tablet Strength | Description | Package Configuration | NDC Number |
| 50 mg | Blue, film coated, arc-triangle shaped tablets debossed with “Lilly 50” on one side and “6902” on the other side. | Bottle with child-resistant closure. Each bottle contains 30 tablets. |
0002-6902-30 |
| 100 mg | Blue, film coated, round tablets debossed with “Lilly 100” on one side and “7026” on the other side. | Bottle with child-resistant closure. Each bottle contains 60 tablets. |
0002-7026-60 |
Storage and Handling
Store JAYPIRCA tablets at room temperature 20°C to 25°C (68°F to 77°F); excursions permitted between 15°C and 30°C (59°F and 86°F) ([see USP Controlled Room Temperature]).
2.2 Dosage Modifications for Adverse Reactions
Recommended dosage modifications of JAYPIRCA for adverse reactions are presented in Table 1 [see Warnings and Precautions (5.1, 5.2, 5.3, and 5.4)].
|
Dose modification is not recommended for asymptomatic lymphocytosis. Asymptomatic lipase increase may not necessarily warrant dose modification. |
||
|
a Evaluate the benefit-risk before resuming treatment at the same dose for a Grade 4 non-hematological toxicity. |
||
| Adverse Reaction | Occurrences Requiring Dosage Modification |
Modification
(Starting Dosage: 200 mg once daily) |
|
First occurrence | Interrupt JAYPIRCA until recovery to Grade 1 or baseline; restart at original dosage (200 mg once daily)a. |
| Second occurrence | Interrupt JAYPIRCA until recovery to Grade 1 or baseline; restart at 100 mg once daily. | |
| Third occurrence | Interrupt JAYPIRCA until recovery to Grade 1 or baseline; restart at 50 mg once daily. | |
| Fourth occurrence | Discontinue JAYPIRCA. |
8.3 Females and Males of Reproductive Potential
Based on findings from animal studies, JAYPIRCA can cause fetal harm when administered to a pregnant woman [see Use in Specific Populations (8.1)].
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenicity studies have not been conducted with pirtobrutinib.
Pirtobrutinib was not mutagenic in a bacterial mutagenicity (Ames) assay. Pirtobrutinib was aneugenic in in vitro micronucleus assays using human peripheral blood lymphocytes. Pirtobrutinib was not genotoxic in an in vivo rat bone marrow micronucleus assay at doses up to 2000 mg/kg.
Studies to assess the effects of pirtobrutinib on fertility have not been conducted. In repeat-dose toxicity studies of up to 3-months duration conducted with pirtobrutinib in rats and dogs, no effects on male or female reproductive organs were identified.
1.2 Chronic Lymphocytic Leukemia/small Lymphocytic Lymphoma
JAYPIRCA is indicated for the treatment of adult patients with relapsed or refractory chronic lymphocytic leukemia or small lymphocytic lymphoma (CLL/SLL) who have previously been treated with a covalent BTK inhibitor.
14.2 Chronic Lymphocytic Leukemia/small Lymphocytic Lymphoma
BRUIN
The efficacy of JAYPIRCA in patients with CLL/SLL was evaluated in BRUIN [NCT03740529] an open-label, international, single-arm, multicohort study of JAYPIRCA as monotherapy. Efficacy was based on 108 patients with CLL/SLL treated with JAYPIRCA who were previously treated with at least two prior lines of therapy, including a BTK inhibitor and a BCL-2 inhibitor. JAYPIRCA was given orally at a dose of 200 mg once daily and was continued until disease progression or unacceptable toxicity. The trial required a platelet count ≥ 50 x 109/L, absolute neutrophil count ≥ 0.75 x 109/L, hepatic transaminases ≤ 2.5 times upper limit of normal (ULN), and an ECOG performance status of 0 to 2. The trial excluded patients with significant cardiovascular disease, major bleeding, uncontrolled or symptomatic arrhythmias, prolonged QTc interval, or need for a strong CYP3A inhibitor or inducer or strong P-gp inhibitor. Patients with active central nervous system (CNS) involvement by lymphoma or allogeneic hematopoietic stem cell transplantation (HSCT) within 60 days were excluded.
The median age was 68 years (range: 41 to 88 years); 69% were male; 89% were White, 4.6% Black or African American, 1.9% Asian and 1.9% were Hispanic or Latino. Baseline ECOG performance status was 0 or 1 in 91% of patients and 48% of patients had Rai stage III or IV disease. Among those patients with central testing available, 42% (37 of 88 patients) had a C481 BTK mutation, 54% (43 of 79 patients) had 17p deletion and/or TP53 mutation, 93% (77 of 83 patients) had unmutated IGHV, and 22% (16 of 72 patients) had 11q deletion. Patients received a median number of 5 prior lines of therapy (range: 2 to 11). The most common prior BTK inhibitors received were ibrutinib (97%), acalabrutinib (9%), and zanubrutinib (0.9%). Seventy-seven percent of patients discontinued the last BTK inhibitor for refractory or progressive disease, 13% discontinued for toxicity, and 10% discontinued for other reasons.
Efficacy was established based on overall response rate (ORR) and duration of response (DOR), as assessed by an independent review committee (IRC) using 2018 iwCLL criteria. Efficacy results are shown in Table 9 -. The median time to response was 3.7 months (range: 1.7, 27.9 months).
|
CI, confidence interval; PR, partial response. |
|
|
a Based on Kaplan-Meier estimation. Estimated median follow-up was 15.7 months. |
|
| Outcome |
JAYPIRCA 200 mg once daily
(N = 108) |
| Overall Response Rate | |
| ORR, n | 78 (72%) |
| (95% CI, %) | 63, 80 |
| PR, n | 78 (72%) |
| Duration of Response a | |
| Median DOR, months (95% CI) | 12.2 (9.3, 14.7) |
2.5 Dosage Modifications for Concomitant Use With Cyp3a Inducers
Avoid concomitant use of strong or moderate CYP3A inducers with JAYPIRCA [see Drug Interactions (7.1), Clinical Pharmacology (12.3)]. If concomitant use with moderate CYP3A inducers is unavoidable and the current dosage of JAYPIRCA is 200 mg once daily, increase the dose to 300 mg. If the current dosage is 50 mg or 100 mg once daily, increase the dose by 50 mg.
2.3 Dosage Modifications for Patients With Severe Renal Impairment
For patients with severe renal impairment (eGFR 15-29 mL/min), reduce the JAYPIRCA dose to 100 mg once daily if the current dose is 200 mg once daily otherwise reduce the dose by 50 mg. If the current dosage is 50 mg once daily, discontinue JAYPIRCA [see Use in Specific Populations (8.7), Clinical Pharmacology (12.3)]. No dosage adjustment of JAYPIRCA is recommended in patients with mild to moderate renal impairment (eGFR 30-89 mL/min).
2.4 Dosage Modifications for Concomitant Use With Strong Cyp3a Inhibitors
Avoid concomitant use of strong CYP3A inhibitors with JAYPIRCA [see Drug Interactions (7.1), Clinical Pharmacology (12.3)]. If concomitant use of a strong CYP3A inhibitor is unavoidable, reduce the JAYPIRCA dose by 50 mg. If the current dosage is 50 mg once daily, interrupt JAYPIRCA treatment for the duration of strong CYP3A inhibitor use. After discontinuation of a strong CYP3A inhibitor for 5 half-lives, resume the JAYPIRCA dose that was taken prior to initiating the strong CYP3A inhibitor.
Structured Label Content
Section 42229-5 (42229-5)
Hepatotoxicity, including severe, life-threatening, and potentially fatal cases of drug-induced liver injury (DILI), has occurred in patients treated with Bruton tyrosine kinase inhibitors, including JAYPIRCA.
Evaluate bilirubin and transaminases at baseline and throughout treatment with JAYPIRCA. For patients who develop abnormal liver tests after JAYPIRCA, monitor more frequently for liver test abnormalities and clinical signs and symptoms of hepatic toxicity. If DILI is suspected, withhold JAYPIRCA. Upon confirmation of DILI, discontinue JAYPIRCA.
Section 42230-3 (42230-3)
|
This Patient Information has been approved by the U.S. Food and Drug Administration. |
Revised: 12/2025 |
|||
|
PATIENT INFORMATION
JAYPIRCA® (JAY-PIHR-KAA) pirtobrutinib tablets |
||||
|
What is JAYPIRCA?
JAYPIRCA is a prescription medicine used to treat adults with:
|
||||
| It is not known if JAYPIRCA is safe and effective in children. | ||||
Before taking JAYPIRCA, tell your healthcare provider about all of your medical conditions, including if you:
|
||||
| Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. Taking JAYPIRCA with certain other medicines may affect how JAYPIRCA or the other medicines work and can cause side effects. | ||||
How should I take JAYPIRCA?
|
||||
|
What are the possible side effects of JAYPIRCA?
JAYPIRCA can cause serious side effects, including:
|
||||
| Your healthcare provider may decrease your dose, temporarily stop, or permanently stop treatment with JAYPIRCA if you develop severe side effects. The most common side effects of JAYPIRCA include: |
||||
|
|
|||
| These are not all the possible side effects of JAYPIRCA. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
||||
How should I store JAYPIRCA?
|
||||
| Keep JAYPIRCA and all medicines out of the reach of children. | ||||
|
General information about the safe and effective use of JAYPIRCA.
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use JAYPIRCA for a condition for which it was not prescribed. Do not give JAYPIRCA to other people, even if they have the same symptoms that you have. It may harm them. You can ask your pharmacist or healthcare provider for information about JAYPIRCA that is written for health professionals. |
||||
|
What are the ingredients in JAYPIRCA?
Active ingredient: pirtobrutinib Inactive ingredients: croscarmellose sodium, hypromellose acetate succinate, lactose monohydrate, magnesium stearate, microcrystalline cellulose, and silicon dioxide. The tablet film coating contains FD&C Blue #2, hypromellose, titanium dioxide, and triacetin. Marketed by Lilly USA, LLC, Indianapolis, IN 46285, USA JAYPIRCA is a registered trademark of Eli Lilly and Company. Copyright © 2023, 2025, Eli Lilly and Company. All rights reserved. JAY-0005-PPI-20251203 For more information, go to www.jaypirca.com or call 1-800-LillyRx (1-800-545-5979). |
Section 43683-2 (43683-2)
| Indications and Usage, Chronic Lymphocytic Leukemia/Small Lymphocytic Lymphoma (1.2) | 12/2025 |
Section 51945-4 (51945-4)
PACKAGE LABEL – JAYPIRCA 50 mg Tablets, 30 count bottle
NDC 0002-6902-30
30 tablets Rx only
Jaypirca®
(pirtobrutinib)
tablets
50 mg
Each tablet contains 50 mg pirtobrutinib
www.jaypirca.com
Lilly
11 Description (11 DESCRIPTION)
Pirtobrutinib is a kinase inhibitor. It is an orally available, small molecule ATP-competitive inhibitor of BTK. The active pharmaceutical ingredient is pirtobrutinib with the molecular formula C22H21F4N5O3 and a molecular weight of 479.44 g/mol. The chemical name for pirtobrutinib is 5-amino-3-{4-[(5-fluoro-2-methoxybenzamido)methyl]phenyl}-1-[(2S)-1,1,1-trifluoropropan-2-yl]-1H-pyrazole-4-carboxamide.
Pirtobrutinib is a white to practically white to yellow to brown solid. The aqueous solubility of pirtobrutinib is considered practically insoluble, or insoluble, across the pH 1 to pH 7 range.
Pirtobrutinib tablets are supplied as 50 mg or 100 mg film-coated, debossed tablets for oral administration. Each tablet contains inactive ingredients of croscarmellose sodium, hypromellose acetate succinate, lactose monohydrate, magnesium stearate, microcrystalline cellulose and silicon dioxide. The tablet film coating material contains FD&C Blue #2, hypromellose, titanium dioxide and triacetin.
5.1 Infections
Fatal and serious infections (including bacterial, viral, or fungal infections) and opportunistic infections have occurred in patients treated with JAYPIRCA. Across clinical trials, Grade 3 or higher infections occurred in 25% of 704 patients, most commonly pneumonia (20%), with fatal infections occurring in 5% of patients. Sepsis occurred in 6% of patients and febrile neutropenia in 3.8%. In patients with CLL/SLL, Grade 3 or higher infections occurred in 32% of patients, with fatal infections occurring in 8%. Opportunistic infections after treatment with JAYPIRCA have included, but are not limited to, Pneumocystis jirovecii pneumonia and fungal infection [see Adverse Reactions (6.1)].
Consider prophylaxis, including vaccinations and antimicrobial prophylaxis, in patients who are at increased risk for infections, including opportunistic infections. Monitor patients for signs and symptoms of infection, evaluate promptly, and treat appropriately. Based on severity, reduce dose, temporarily withhold, or permanently discontinue JAYPIRCA [see Dosage and Administration (2.2)].
5.2 Hemorrhage
Fatal and serious hemorrhage has occurred with JAYPIRCA. Major hemorrhage (defined as Grade 3 or higher bleeding or any central nervous system bleeding) occurred in 2.6% of 704 patients treated with JAYPIRCA, including gastrointestinal hemorrhage; fatal hemorrhage occurred in 0.3% of patients. Bleeding of any grade, excluding bruising and petechiae, occurred in 16% of patients [see Adverse Reactions (6.1)].
Major hemorrhage occurred in 0.6% of patients taking JAYPIRCA without antithrombotic agents and 2.0% of patients taking JAYPIRCA with antithrombotic agents. Consider the risks and benefits of antithrombotic agents when co-administered with JAYPIRCA. Monitor patients for signs of bleeding. Based on severity of bleeding, reduce dose, temporarily withhold, or permanently discontinue JAYPIRCA [see Dosage and Administration (2.2)].
Consider the benefit-risk of withholding JAYPIRCA for 3 to 7 days pre- and post-surgery depending upon the type of surgery and risk of bleeding.
5.3 Cytopenias
JAYPIRCA can cause cytopenias, including neutropenia, thrombocytopenia, and anemia.
Across clinical trials, Grade 3 or 4 cytopenias, including decreased neutrophils (27%), decreased platelets (13%), and decreased hemoglobin (11%) developed in patients treated with JAYPIRCA. Grade 4 decreased neutrophils developed in 15% of patients and Grade 4 decreased platelets developed in 6% of patients [see Adverse Reactions (6.1)].
Monitor complete blood counts regularly during treatment. Based on severity, reduce dose, temporarily withhold, or permanently discontinue JAYPIRCA [see Dosage and Administration (2.2)].
8.4 Pediatric Use
Safety and effectiveness of JAYPIRCA have not been established in pediatric patients.
8.5 Geriatric Use
Of the patients with MCL who received the 200 mg dose of JAYPIRCA in BRUIN, 93 (78%) were 65 years of age and older and 39 (33%) were 75 years and older [see Clinical Studies (14.1)]. Clinical studies of JAYPIRCA did not include sufficient numbers of patients with MCL who were less than 65 years of age to determine whether older patients respond differently from younger adult patients. Of the patients with CLL/SLL who received the 200 mg once daily dose of JAYPIRCA in BRUIN and BRUIN-321, 220 (65%) were 65 years of age and older and 78 (33%) were 75 years and older [see Clinical Studies (14.2)]. No overall differences in effectiveness were observed between younger and older patients.
In the pooled safety population in patients with hematologic malignancies in BRUIN and BRUIN CLL-321, 467 (66%) were 65 years of age and older, while 181 (26%) were 75 years of age and older. Patients aged 65 years and older experienced higher rates of Grade 3 and higher adverse reactions and serious adverse reactions compared to patients who were less than 65 years of age.
4 Contraindications (4 CONTRAINDICATIONS)
None.
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following clinically significant adverse reactions are described elsewhere in the labeling:
- Infections [see Warnings and Precautions (5.1)]
- Hemorrhage [see Warnings and Precautions (5.2)]
- Cytopenias [see Warnings and Precautions (5.3)]
- Atrial Fibrillation and Atrial Flutter [see Warnings and Precautions (5.4)]
- Second Primary Malignancies [see Warnings and Precautions (5.5)]
- Hepatotoxicity, including DILI [see Warnings and Precautions (5.6)]
7 Drug Interactions (7 DRUG INTERACTIONS)
- Strong CYP3A Inhibitors: Avoid concomitant use. If concomitant use is unavoidable, reduce the JAYPIRCA dose. (2.4, 7.1)
- Strong or Moderate CYP3A Inducers: Avoid concomitant use. If concomitant use of moderate CYP3A inducers is unavoidable, increase the JAYPIRCA dose. (2.5, 7.1)
- Sensitive CYP2C8, CYP2C19, CYP3A, P-gp, or BCRP Substrates: For substrates where minimal concentration changes may increase the risk of adverse reactions, follow recommendations for co-administration with CYP2C8, CYP2C19, CYP3A, P-gp, or BCRP inhibitors provided in their approved product labeling. (7.2)
8.6 Renal Impairment
Severe renal impairment (eGFR15-29 mL/min) increases pirtobrutinib exposure [see Clinical Pharmacology (12.3)]. Reduce the JAYPIRCA dosage in patients with severe renal impairment [see Dosage and Administration (2.3)]. No dosage adjustment of JAYPIRCA is recommended in patients with mild (60-89 mL/min) or moderate (30-59 mL/min) renal impairment.
12.2 Pharmacodynamics
At the recommended dosage of 200 mg once daily, pirtobrutinib trough concentrations exceeded the BTK IC96. BTK occupancy is maintained throughout the dosing interval, regardless of the intrinsic rate of BTK turnover.
12.3 Pharmacokinetics
The pharmacokinetics of pirtobrutinib were characterized in healthy subjects and in patients with cancer. Pirtobrutinib exposure (AUC) and Cmax increases proportionally following single oral doses ranging from 300 mg to 800 mg (1.5 to 4 times the approved recommended dosage) and once daily doses ranging from 25 – 300 mg (0.125 to 1.5 times the recommended dosage). Steady state was achieved within 5 days of once daily dosing, and the mean (CV%) accumulation ratio was 1.63 (26.7%) based on AUC after administration of 200 mg dosages.
Following administration of the recommended dosage, the geometric mean (CV%) steady-state AUC and Cmax of pirtobrutinib were 92705 h*ng/mL (39%) and 6503 ng/mL (25%), respectively. The geometric mean (CV%) AUC0-24 and Cmax of pirtobrutinib on Cycle 1 Day 8 were 81800 h*ng/mL (66.6%) and 3670 ng/mL (89.5%), respectively.
2.1 Recommended Dosage
The recommended dosage of JAYPIRCA is 200 mg orally once daily until disease progression or unacceptable toxicity.
Advise patients of the following:
- Swallow tablets whole with water. Do not cut, crush, or chew tablets.
- Take JAYPIRCA at the same time each day. JAYPIRCA may be taken with or without food.
- If a dose of JAYPIRCA is missed by more than 12 hours, do not make up the dose and take the next dose as scheduled.
8.7 Hepatic Impairment
No dosage adjustment of JAYPIRCA is recommended in patients with mild hepatic impairment (total bilirubin ≤ upper limit of normal (ULN) and aspartate aminotransferase (AST) > ULN or total bilirubin > 1 to 1.5 × ULN and any AST), moderate hepatic impairment (total bilirubin > 1.5 to 3 × ULN and any AST), or severe hepatic impairment (total bilirubin > 3 × ULN and any AST) [see Clinical Pharmacology (12.3 )].
1 Indications and Usage (1 INDICATIONS AND USAGE)
JAYPIRCA® is a kinase inhibitor indicated for the treatment of:
- Adult patients with relapsed or refractory mantle cell lymphoma (MCL) after at least two lines of systemic therapy, including a BTK inhibitor. (1.1).
This indication is approved under accelerated approval based on response rate. Continued approval for this indication may be contingent upon verification and description of clinical benefit in a confirmatory trial. - Adult patients with relapsed or refractory chronic lymphocytic leukemia or small lymphocytic lymphoma (CLL/SLL) who have previously been treated with a covalent BTK inhibitor. (1.2).
5.4 Cardiac Arrhythmias
Cardiac arrhythmias, including atrial fibrillation and atrial flutter, were reported in recipients of JAYPIRCA. Atrial fibrillation or flutter were reported in 3.4% of patients, with Grade 3 or 4 atrial fibrillation or flutter reported in 1.6% of 704 patients across clinical trials [see Adverse Reactions (6.1)]. Other serious cardiac arrhythmias such as supraventricular tachycardia and cardiac arrest occurred in 0.4% of patients. Patients with cardiac risk factors, such as hypertension, or previous arrhythmias may be at increased risk.
Monitor for signs and symptoms of arrhythmias (e.g., palpitations, dizziness, syncope, dyspnea) and manage appropriately. Based on severity, reduce dose, temporarily withhold, or permanently discontinue JAYPIRCA [see Dosage and Administration (2.2)].
1.1 Mantle Cell Lymphoma
JAYPIRCA® is indicated for the treatment of adult patients with relapsed or refractory mantle cell lymphoma (MCL) after at least two lines of systemic therapy, including a BTK inhibitor.
This indication is approved under accelerated approval based on response rate [see Clinical Studies (14.1)]. Continued approval for this indication may be contingent upon verification and description of clinical benefit in a confirmatory trial.
12.1 Mechanism of Action (12.1 Mechanism of action)
Pirtobrutinib is a small molecule, noncovalent inhibitor of BTK. BTK is a signaling protein of the B-cell antigen receptor (BCR) and cytokine receptor pathways. In B-cells, BTK signaling results in activation of pathways necessary for B-cell proliferation, trafficking, chemotaxis, and adhesion. Pirtobrutinib binds to wild type BTK and BTK harboring C481 mutations, leading to inhibition of BTK kinase activity. In nonclinical studies, pirtobrutinib inhibited BTK-mediated B-cell CD69 expression and inhibited malignant B-cell proliferation. Pirtobrutinib showed dose-dependent anti-tumor activities in BTK wild type and BTK C481S mutant mouse xenograft models.
14.1 Mantle Cell Lymphoma
The efficacy of JAYPIRCA in patients with MCL was evaluated in BRUIN [NCT03740529], an open-label, international, multicohort, single-arm study of JAYPIRCA as monotherapy. Efficacy was based on 120 patients with MCL treated with JAYPIRCA who were previously treated with a BTK inhibitor. JAYPIRCA was given orally at a dose of 200 mg once daily and was continued until disease progression or unacceptable toxicity. Patients with active central nervous system lymphoma or allogeneic hematopoietic stem cell transplantation (HSCT) or CAR-T cell therapy within 60 days were excluded.
The median age was 71 years (range: 46 to 88 years); 79% were male; 78% were White, 14% Asian, 1.7% Black or African American. Seventy-eight percent of patients had the classic/leukemic variant of MCL, 12% had pleomorphic MCL, and 11% had blastoid MCL. The simplified Mantle Cell Lymphoma International Prognostic Index (sMIPI) score was low in 15%, intermediate in 59%, and high in 26% of patients. Patients received a median number of 3 prior lines of therapy (range: 1 to 9) with 93% having received 2 or more prior lines. All received 1 or more prior lines of therapy containing a BTK inhibitor; other prior therapies included chemoimmunotherapy in 88%, HSCT in 20%, lenalidomide in 18%, and CAR-T therapy in 9%. The most common prior BTK inhibitors received were ibrutinib (67%), acalabrutinib (30%), and zanubrutinib (8%). Patients may have received more than one prior BTK inhibitor; 83% of patients discontinued the last BTK inhibitor for refractory or progressive disease, 10% discontinued for toxicity, and 5% discontinued for other reasons.
Efficacy was based on overall response rate (ORR) and duration of response (DOR), as assessed by an independent review committee (IRC) using 2014 Lugano criteria. Efficacy results are shown in Table 8. Additionally, the Kaplan-Meier estimate for the DOR rate at 6 months was 65.3% (95% CI: 49.8, 77.1).
|
CI, confidence interval; CR, complete response; DOR, duration of response; PR, partial response; NE, not estimable. |
|
|
a PET-CT scans were utilized in response assessments (in 41% of patients), with the remainder being assessed by CT scans only. |
|
|
b ORR using CT scan-based assessments in all patients was 48% (95% CI: 38, 57) and CR rate was 13%. |
|
|
c Based on Kaplan-Meier estimation. Estimated median follow-up was 7.3 months. |
|
| Outcome |
JAYPIRCA 200 mg once daily
(N = 120) |
| Overall Response Rate a,b | |
| ORR, n | 60 (50%) |
| (95% CI, %) | 41, 59 |
| CR, n | 15 (13%) |
| PR, n | 45 (38%) |
| Time to Response | |
| Median (range), months | 1.8 (0.8, 4.2) |
| Duration of Response c | |
| Number censored, n | 36 |
| Median DOR, months (95% CI) | 8.3 (5.7, NE) |
5.7 Embryo Fetal Toxicity (5.7 Embryo-Fetal Toxicity)
Based on findings in animals, JAYPIRCA can cause fetal harm when administered to a pregnant woman. In animal reproduction studies, administration of pirtobrutinib to pregnant rats during the period of organogenesis caused embryo-fetal toxicity including embryo-fetal mortality and malformations at maternal exposures (AUC) approximately 3-times the recommended dose of 200 mg once daily. Advise pregnant women of the potential risk to a fetus. Advise females of reproductive potential to use effective contraception during treatment with JAYPIRCA and for one week after the last dose [see Use in Specific Populations (8.1, 8.3)].
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Infections: Monitor for signs and symptoms of infection, evaluate promptly, and treat. (5.1)
- Hemorrhage: Monitor for bleeding and manage appropriately. (5.2)
- Cytopenias: Monitor complete blood counts during treatment. (5.3)
- Cardiac Arrythmias: Monitor for symptoms of arrhythmias and manage appropriately. (5.4)
- Second Primary Malignancies: Other malignancies have developed, including skin cancers and other carcinomas. Monitor and advise patients to use sun protection. (5.5)
- Hepatotoxicity, Including Drug-Induced Liver Injury: Monitor hepatic function throughout treatment. (5.6)
- Embryo-Fetal Toxicity: Can cause fetal harm. Advise females of reproductive potential of potential risk to a fetus and to use effective contraception. (5.7, 8.1, 8.3)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Tablets:
Each 50 mg tablet is blue, arc-triangle shaped, film-coated, and debossed with “Lilly 50” on one side and “6902” on the other side.
Each 100 mg tablet is blue, round, film-coated, and debossed with “Lilly 100” on one side and “7026” on the other side.
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
Lactation: Advise not to breastfeed (8.2)
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be compared to rates in the clinical trials of another drug and may not reflect the rates observed in the general patient population.
The data in the WARNINGS AND PRECAUTIONS reflect exposure to JAYPIRCA as a single-agent, administered at 200 mg once daily in 704 patients with hematologic malignancies in the BRUIN and the BRUIN-CLL-321 studies. Among these 704 patients, the median duration of exposure was 12 months; 65% were exposed for at least 6 months and 50% were exposed for at least one year.
In this pooled safety population, the most common (≥ 30%) adverse reactions, including laboratory abnormalities, were decreased neutrophil count (54%), decreased hemoglobin (43%), decreased leukocytes (32%), fatigue (31%), decreased platelets (31%), decreased lymphocyte count (31%), and calcium decreased (30%)
5.5 Second Primary Malignancies
Second primary malignancies, including non-skin carcinomas, developed in 9% of 704 patients treated with JAYPIRCA monotherapy across clinical trials. The most frequent malignancy was non-melanoma skin cancer, reported in 4.4% of 704 patients. Other second primary malignancies included solid tumors (including genitourinary and breast cancers) and melanoma. Advise patients to use sun protection and monitor patients for the development of second primary malignancies.
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient to read the FDA-approved patient labeling (Patient Information).
Infections
Advise patients that JAYPIRCA can cause serious infections that may be fatal. Advise patients to report any signs or symptoms of infection (e.g., fever, chills, weakness) [see Warnings and Precautions (5.1)].
Hemorrhage
Inform patients to report signs or symptoms of bleeding. Inform patients that JAYPIRCA may need to be interrupted for major surgeries [see Warnings and Precautions (5.2)].
Cytopenias
Advise patients of the need for periodic monitoring of blood counts during treatment with JAYPIRCA [see Warnings and Precautions (5.3)].
Cardiac Arrhythmias
Counsel patients to report any signs of palpitations, dizziness, fainting, chest discomfort, and shortness of breath [see Warnings and Precautions (5.4 )].
Second Primary Malignancies
Inform patients that other malignancies have been reported in patients who have been treated with JAYPIRCA, including skin cancer and other solid tumors. Advise patients to use sun protection and to have monitoring for development of other cancers [see Warnings and Precautions (5.5)].
Hepatotoxicity, Including Drug-Induced Liver Injury
Inform patients that liver problems, including severe, life-threatening, or fatal hepatitis, drug-induced liver injury and abnormalities in liver tests, may develop during JAYPIRCA treatment. Advise patients to contact their healthcare provider immediately if they experience abdominal discomfort, dark urine, or jaundice [see Warnings and Precautions (5.6)].
Embryo-Fetal Toxicity
Advise pregnant women and females of reproductive potential of the potential risk to a fetus. Advise females of reproductive potential to inform their healthcare provider of a known or suspected pregnancy [see Warnings and Precautions (5.7), Use in Specific Populations (8.1, 8.3)].
Advise females of reproductive potential to use effective contraception during treatment with JAYPIRCA and for one week after the last dose [see Use in Specific Populations (8.3)].
Lactation
Advise women not to breastfeed during treatment with JAYPIRCA and for one week after the last dose [see Use in Specific Populations (8.2)].
Administration
Inform patients to take JAYPIRCA orally once daily at approximately the same time each day with or without food and how to make up a missed dose. Advise patients to swallow tablets whole with water. Advise patients not to cut, crush, or chew tablets [see Dosage and Administration (2.1)].
Marketed by: Lilly USA, LLC, Indianapolis, IN 46285, USA
Copyright © 2023, 2025, Eli Lilly and Company. All rights reserved.
JAY-0005-USPI-20251203
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
How Supplied
JAYPIRCA tablets are supplied as follows:
| Tablet Strength | Description | Package Configuration | NDC Number |
| 50 mg | Blue, film coated, arc-triangle shaped tablets debossed with “Lilly 50” on one side and “6902” on the other side. | Bottle with child-resistant closure. Each bottle contains 30 tablets. |
0002-6902-30 |
| 100 mg | Blue, film coated, round tablets debossed with “Lilly 100” on one side and “7026” on the other side. | Bottle with child-resistant closure. Each bottle contains 60 tablets. |
0002-7026-60 |
Storage and Handling
Store JAYPIRCA tablets at room temperature 20°C to 25°C (68°F to 77°F); excursions permitted between 15°C and 30°C (59°F and 86°F) ([see USP Controlled Room Temperature]).
2.2 Dosage Modifications for Adverse Reactions
Recommended dosage modifications of JAYPIRCA for adverse reactions are presented in Table 1 [see Warnings and Precautions (5.1, 5.2, 5.3, and 5.4)].
|
Dose modification is not recommended for asymptomatic lymphocytosis. Asymptomatic lipase increase may not necessarily warrant dose modification. |
||
|
a Evaluate the benefit-risk before resuming treatment at the same dose for a Grade 4 non-hematological toxicity. |
||
| Adverse Reaction | Occurrences Requiring Dosage Modification |
Modification
(Starting Dosage: 200 mg once daily) |
|
First occurrence | Interrupt JAYPIRCA until recovery to Grade 1 or baseline; restart at original dosage (200 mg once daily)a. |
| Second occurrence | Interrupt JAYPIRCA until recovery to Grade 1 or baseline; restart at 100 mg once daily. | |
| Third occurrence | Interrupt JAYPIRCA until recovery to Grade 1 or baseline; restart at 50 mg once daily. | |
| Fourth occurrence | Discontinue JAYPIRCA. |
8.3 Females and Males of Reproductive Potential
Based on findings from animal studies, JAYPIRCA can cause fetal harm when administered to a pregnant woman [see Use in Specific Populations (8.1)].
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility (13.1 Carcinogenesis, mutagenesis, impairment of fertility)
Carcinogenicity studies have not been conducted with pirtobrutinib.
Pirtobrutinib was not mutagenic in a bacterial mutagenicity (Ames) assay. Pirtobrutinib was aneugenic in in vitro micronucleus assays using human peripheral blood lymphocytes. Pirtobrutinib was not genotoxic in an in vivo rat bone marrow micronucleus assay at doses up to 2000 mg/kg.
Studies to assess the effects of pirtobrutinib on fertility have not been conducted. In repeat-dose toxicity studies of up to 3-months duration conducted with pirtobrutinib in rats and dogs, no effects on male or female reproductive organs were identified.
1.2 Chronic Lymphocytic Leukemia/small Lymphocytic Lymphoma (1.2 Chronic Lymphocytic Leukemia/Small Lymphocytic Lymphoma)
JAYPIRCA is indicated for the treatment of adult patients with relapsed or refractory chronic lymphocytic leukemia or small lymphocytic lymphoma (CLL/SLL) who have previously been treated with a covalent BTK inhibitor.
14.2 Chronic Lymphocytic Leukemia/small Lymphocytic Lymphoma (14.2 Chronic Lymphocytic Leukemia/Small Lymphocytic Lymphoma)
BRUIN
The efficacy of JAYPIRCA in patients with CLL/SLL was evaluated in BRUIN [NCT03740529] an open-label, international, single-arm, multicohort study of JAYPIRCA as monotherapy. Efficacy was based on 108 patients with CLL/SLL treated with JAYPIRCA who were previously treated with at least two prior lines of therapy, including a BTK inhibitor and a BCL-2 inhibitor. JAYPIRCA was given orally at a dose of 200 mg once daily and was continued until disease progression or unacceptable toxicity. The trial required a platelet count ≥ 50 x 109/L, absolute neutrophil count ≥ 0.75 x 109/L, hepatic transaminases ≤ 2.5 times upper limit of normal (ULN), and an ECOG performance status of 0 to 2. The trial excluded patients with significant cardiovascular disease, major bleeding, uncontrolled or symptomatic arrhythmias, prolonged QTc interval, or need for a strong CYP3A inhibitor or inducer or strong P-gp inhibitor. Patients with active central nervous system (CNS) involvement by lymphoma or allogeneic hematopoietic stem cell transplantation (HSCT) within 60 days were excluded.
The median age was 68 years (range: 41 to 88 years); 69% were male; 89% were White, 4.6% Black or African American, 1.9% Asian and 1.9% were Hispanic or Latino. Baseline ECOG performance status was 0 or 1 in 91% of patients and 48% of patients had Rai stage III or IV disease. Among those patients with central testing available, 42% (37 of 88 patients) had a C481 BTK mutation, 54% (43 of 79 patients) had 17p deletion and/or TP53 mutation, 93% (77 of 83 patients) had unmutated IGHV, and 22% (16 of 72 patients) had 11q deletion. Patients received a median number of 5 prior lines of therapy (range: 2 to 11). The most common prior BTK inhibitors received were ibrutinib (97%), acalabrutinib (9%), and zanubrutinib (0.9%). Seventy-seven percent of patients discontinued the last BTK inhibitor for refractory or progressive disease, 13% discontinued for toxicity, and 10% discontinued for other reasons.
Efficacy was established based on overall response rate (ORR) and duration of response (DOR), as assessed by an independent review committee (IRC) using 2018 iwCLL criteria. Efficacy results are shown in Table 9 -. The median time to response was 3.7 months (range: 1.7, 27.9 months).
|
CI, confidence interval; PR, partial response. |
|
|
a Based on Kaplan-Meier estimation. Estimated median follow-up was 15.7 months. |
|
| Outcome |
JAYPIRCA 200 mg once daily
(N = 108) |
| Overall Response Rate | |
| ORR, n | 78 (72%) |
| (95% CI, %) | 63, 80 |
| PR, n | 78 (72%) |
| Duration of Response a | |
| Median DOR, months (95% CI) | 12.2 (9.3, 14.7) |
2.5 Dosage Modifications for Concomitant Use With Cyp3a Inducers (2.5 Dosage Modifications for Concomitant Use with CYP3A Inducers)
Avoid concomitant use of strong or moderate CYP3A inducers with JAYPIRCA [see Drug Interactions (7.1), Clinical Pharmacology (12.3)]. If concomitant use with moderate CYP3A inducers is unavoidable and the current dosage of JAYPIRCA is 200 mg once daily, increase the dose to 300 mg. If the current dosage is 50 mg or 100 mg once daily, increase the dose by 50 mg.
2.3 Dosage Modifications for Patients With Severe Renal Impairment (2.3 Dosage Modifications for Patients with Severe Renal Impairment)
For patients with severe renal impairment (eGFR 15-29 mL/min), reduce the JAYPIRCA dose to 100 mg once daily if the current dose is 200 mg once daily otherwise reduce the dose by 50 mg. If the current dosage is 50 mg once daily, discontinue JAYPIRCA [see Use in Specific Populations (8.7), Clinical Pharmacology (12.3)]. No dosage adjustment of JAYPIRCA is recommended in patients with mild to moderate renal impairment (eGFR 30-89 mL/min).
2.4 Dosage Modifications for Concomitant Use With Strong Cyp3a Inhibitors (2.4 Dosage Modifications for Concomitant Use with Strong CYP3A Inhibitors)
Avoid concomitant use of strong CYP3A inhibitors with JAYPIRCA [see Drug Interactions (7.1), Clinical Pharmacology (12.3)]. If concomitant use of a strong CYP3A inhibitor is unavoidable, reduce the JAYPIRCA dose by 50 mg. If the current dosage is 50 mg once daily, interrupt JAYPIRCA treatment for the duration of strong CYP3A inhibitor use. After discontinuation of a strong CYP3A inhibitor for 5 half-lives, resume the JAYPIRCA dose that was taken prior to initiating the strong CYP3A inhibitor.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:51:09.672818 · Updated: 2026-03-14T22:39:28.520287