bc97f364-9443-47b8-9c2d-b5dcc00cc9b7
34390-5
HUMAN OTC DRUG LABEL
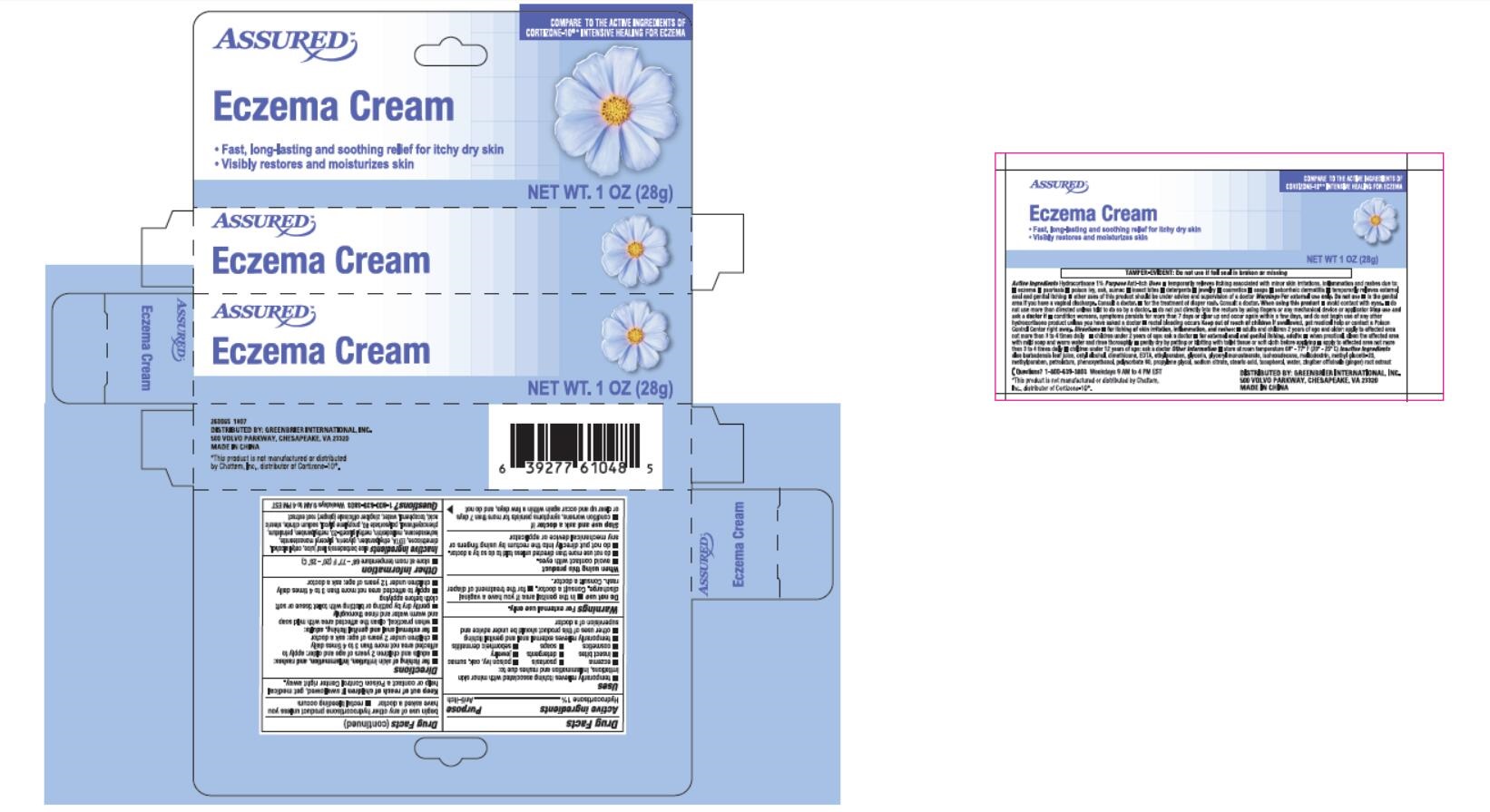
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Active ingredients Hydrocortisone 1%
Medication Information
Warnings and Precautions
. Warnings. For external use only
•in the genital area if you have a vaginal Do not use
discharge. Consult a doctor •for the treatment of diaper
rash. Consult a doctor.
When using this product
•avoid contact with eyes.
•do not use more than directed unless told to do so by a doctor.
•do not put directly into rectum by using fingers or
any mechanical device or applicator.
Stop use and ask a doctor if
•condition worsens, symptoms persist for more than 7 days
or clear up and occur again within a few days, and do not
begin use of any other hydrocortisone product unless you
have asked a doctor. •rectal bleeding occurs.
Indications and Usage
Uses
•temporarily relieves itching ssociated with minor skin
irritation, inflammation and rashesdue to:
•eczema •psoriasis •poison ivy, oak, sumac
•insect bites •detergents •jewelry
•cosmetics •soaps •seborrheic dermatitis
•temporarily relieves external anal and genital itching
•other uses of this product should be under advice and
supervision of a doctor
Dosage and Administration
Directions
• : for relieving of skin irritation, inflammation, and rashes
•adults and children 2 years of age and older: apply to
affected area not more than 3 to 4 times daily
•children under 2 years of age: ask a doctor
• s: for external anal and genital itching, adult
•when practical, clean the affected area with mild soap
and warm water and rinse thoroughly
•gently dry by patting or blotting with toilet tissue or soft
cloth before applying
•apply to affected area not more than 3 to 4 times daily
•children under 12 years of age: ask a doctor
Description
Active ingredients Hydrocortisone 1%
Section 44425-7
Other information
•store at room temperature 68° - 77° F (20° - 25° C)
Section 50565-1
. If swallowed, get medical Keep out of reach of children
help or contract a Poison Control Center right away.
Section 51727-6
aloe barbadensis leaf juice, cetyl alcohol, Inactive ingredients
dimethicone, EDTA, ethylparaben, glycerin, glyceryl monostearate,
isohexadecane, maltodextrin, methyl gluceth-20, methyparaben, petrolatum,
phenoxyethanol, polysorbate 80, propylene glycol, sodium citrate, stearic
acid, tocopherol, water, zingiber officinale (ginger) root extract
Section 51945-4
Section 53413-1
1-800-639-3803 Weekdays 9 AM to 4 PM EST Questions?
Section 55105-1
Purpose
Anti-itch
Section 55106-9
Active ingredients
Hydrocortisone 1%
Structured Label Content
Indications and Usage (34067-9)
Uses
•temporarily relieves itching ssociated with minor skin
irritation, inflammation and rashesdue to:
•eczema •psoriasis •poison ivy, oak, sumac
•insect bites •detergents •jewelry
•cosmetics •soaps •seborrheic dermatitis
•temporarily relieves external anal and genital itching
•other uses of this product should be under advice and
supervision of a doctor
Dosage and Administration (34068-7)
Directions
• : for relieving of skin irritation, inflammation, and rashes
•adults and children 2 years of age and older: apply to
affected area not more than 3 to 4 times daily
•children under 2 years of age: ask a doctor
• s: for external anal and genital itching, adult
•when practical, clean the affected area with mild soap
and warm water and rinse thoroughly
•gently dry by patting or blotting with toilet tissue or soft
cloth before applying
•apply to affected area not more than 3 to 4 times daily
•children under 12 years of age: ask a doctor
Warnings and Precautions (34071-1)
. Warnings. For external use only
•in the genital area if you have a vaginal Do not use
discharge. Consult a doctor •for the treatment of diaper
rash. Consult a doctor.
When using this product
•avoid contact with eyes.
•do not use more than directed unless told to do so by a doctor.
•do not put directly into rectum by using fingers or
any mechanical device or applicator.
Stop use and ask a doctor if
•condition worsens, symptoms persist for more than 7 days
or clear up and occur again within a few days, and do not
begin use of any other hydrocortisone product unless you
have asked a doctor. •rectal bleeding occurs.
Section 44425-7 (44425-7)
Other information
•store at room temperature 68° - 77° F (20° - 25° C)
Section 50565-1 (50565-1)
. If swallowed, get medical Keep out of reach of children
help or contract a Poison Control Center right away.
Section 51727-6 (51727-6)
aloe barbadensis leaf juice, cetyl alcohol, Inactive ingredients
dimethicone, EDTA, ethylparaben, glycerin, glyceryl monostearate,
isohexadecane, maltodextrin, methyl gluceth-20, methyparaben, petrolatum,
phenoxyethanol, polysorbate 80, propylene glycol, sodium citrate, stearic
acid, tocopherol, water, zingiber officinale (ginger) root extract
Section 51945-4 (51945-4)
Section 53413-1 (53413-1)
1-800-639-3803 Weekdays 9 AM to 4 PM EST Questions?
Section 55105-1 (55105-1)
Purpose
Anti-itch
Section 55106-9 (55106-9)
Active ingredients
Hydrocortisone 1%
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:01:42.626394 · Updated: 2026-03-14T23:01:30.191020