These Highlights Do Not Include All The Information Needed To Use Thymoglobulin®
bbd8ab99-552e-4b81-aca4-6b0c7af8b9ae
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
THYMOGLOBULIN is indicated for the prophylaxis and treatment of acute rejection in adult and pediatric patients receiving a kidney transplant in conjunction with concomitant immunosuppression.
Indications and Usage
THYMOGLOBULIN is indicated for the prophylaxis and treatment of acute rejection in adult and pediatric patients receiving a kidney transplant in conjunction with concomitant immunosuppression.
Dosage and Administration
The first dose should be infused over at least 6 hours; doses on subsequent days should be infused over at least 4 hours. ( 2.2 ) Premedication with corticosteroids, acetaminophen, and/or an antihistamine prior to each infusion is recommended. ( 2.2 ) The THYMOGLOBULIN dose should be reduced by one-half if the white blood cell (WBC) count is between 2,000 and 3,000 cells/mm 3 or if the platelet count is between 50,000 and 75,000 cells/mm 3 . Stopping THYMOGLOBULIN treatment should be considered if the WBC count falls below 2,000 cells/mm 3 or if the platelet count falls below 50,000 cells/mm 3 . ( 2.3 ) Indication Dose Prophylaxis of acute rejection 1.5 mg/kg of body weight administered daily for 4 to 7 days Treatment of acute rejection 1.5 mg/kg of body weight administered daily for 7 to 14 days For complete dosing instructions, see full prescribing information. ( 2 )
Warnings and Precautions
THYMOGLOBULIN should only be used by physicians experienced in immunosuppressant therapy in transplantation. ( 5.1 ) Hypersensitivity and infusion-related reactions: THYMOGLOBULIN infusion could result in an anaphylactic reaction. Close compliance with the recommended infusion time may reduce the incidence and severity of infusion-related reactions. ( 5.2 ) Cytopenias including anemia, neutropenia, and thrombocytopenia have occurred with THYMOGLOBULIN administration ( 6 ) and require monitoring of blood counts. Adjust dose accordingly to reverse cytopenias. ( 5.3 ) Infection: Infections and reactivation of infections have been reported. Monitor patients and administer anti-infective prophylaxis. ( 5.4 ) Malignancy: Incidence of malignancies may increase. ( 5.5 ) Immunization with attenuated live vaccines is not recommended for patients who have recently received THYMOGLOBULIN. ( 5.6 ) THYMOGLOBULIN may interfere with rabbit antibody–based immunoassays and with cross-match or panel-reactive antibody cytotoxicity assays. ( 5.7 )
Contraindications
THYMOGLOBULIN is contraindicated in patients with history of allergy or anaphylactic reaction to rabbit proteins or to any product excipients, or who have active acute or chronic infections that contraindicate any additional immunosuppression [see Warnings and Precautions (5.2 , 5.4) and Adverse Reactions (6.2) ] .
Adverse Reactions
The most common adverse reactions and laboratory abnormalities (incidence >5% higher than comparator) are urinary tract infection, abdominal pain, hypertension, nausea, shortness of breath, fever, headache, anxiety, chills, increased potassium levels in the blood, low counts of platelets and white blood cells. ( 6 ) To report SUSPECTED ADVERSE REACTIONS, contact Genzyme Corporation at 1-800-633-1610 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Drug Interactions
No drug interaction studies have been performed. THYMOGLOBULIN can stimulate the production of antibodies that cross-react with rabbit immune globulins [see Clinical Pharmacology (12.3) ] .
Storage and Handling
Store in refrigerator at 2°C to 8°C (36°F to 46°F). Protect from light. Do not freeze. Do not use after the expiration date indicated on the label. Reconstituted THYMOGLOBULIN is physically and chemically stable for up to 24 hours at room temperature; however, room temperature storage is not recommended. As THYMOGLOBULIN contains no preservatives, reconstituted product should be used immediately. Infusion solutions of THYMOGLOBULIN must be used immediately. Any unused drug remaining after infusion must be discarded.
How Supplied
THYMOGLOBULIN is supplied as a single-dose clear glass 10 mL vial containing 25 mg of lyophilized (solid) THYMOGLOBULIN. Each carton contains one THYMOGLOBULIN vial (NDC 58468-0080-1).
Medication Information
Warnings and Precautions
THYMOGLOBULIN should only be used by physicians experienced in immunosuppressant therapy in transplantation. ( 5.1 ) Hypersensitivity and infusion-related reactions: THYMOGLOBULIN infusion could result in an anaphylactic reaction. Close compliance with the recommended infusion time may reduce the incidence and severity of infusion-related reactions. ( 5.2 ) Cytopenias including anemia, neutropenia, and thrombocytopenia have occurred with THYMOGLOBULIN administration ( 6 ) and require monitoring of blood counts. Adjust dose accordingly to reverse cytopenias. ( 5.3 ) Infection: Infections and reactivation of infections have been reported. Monitor patients and administer anti-infective prophylaxis. ( 5.4 ) Malignancy: Incidence of malignancies may increase. ( 5.5 ) Immunization with attenuated live vaccines is not recommended for patients who have recently received THYMOGLOBULIN. ( 5.6 ) THYMOGLOBULIN may interfere with rabbit antibody–based immunoassays and with cross-match or panel-reactive antibody cytotoxicity assays. ( 5.7 )
Indications and Usage
THYMOGLOBULIN is indicated for the prophylaxis and treatment of acute rejection in adult and pediatric patients receiving a kidney transplant in conjunction with concomitant immunosuppression.
Dosage and Administration
The first dose should be infused over at least 6 hours; doses on subsequent days should be infused over at least 4 hours. ( 2.2 ) Premedication with corticosteroids, acetaminophen, and/or an antihistamine prior to each infusion is recommended. ( 2.2 ) The THYMOGLOBULIN dose should be reduced by one-half if the white blood cell (WBC) count is between 2,000 and 3,000 cells/mm 3 or if the platelet count is between 50,000 and 75,000 cells/mm 3 . Stopping THYMOGLOBULIN treatment should be considered if the WBC count falls below 2,000 cells/mm 3 or if the platelet count falls below 50,000 cells/mm 3 . ( 2.3 ) Indication Dose Prophylaxis of acute rejection 1.5 mg/kg of body weight administered daily for 4 to 7 days Treatment of acute rejection 1.5 mg/kg of body weight administered daily for 7 to 14 days For complete dosing instructions, see full prescribing information. ( 2 )
Contraindications
THYMOGLOBULIN is contraindicated in patients with history of allergy or anaphylactic reaction to rabbit proteins or to any product excipients, or who have active acute or chronic infections that contraindicate any additional immunosuppression [see Warnings and Precautions (5.2 , 5.4) and Adverse Reactions (6.2) ] .
Adverse Reactions
The most common adverse reactions and laboratory abnormalities (incidence >5% higher than comparator) are urinary tract infection, abdominal pain, hypertension, nausea, shortness of breath, fever, headache, anxiety, chills, increased potassium levels in the blood, low counts of platelets and white blood cells. ( 6 ) To report SUSPECTED ADVERSE REACTIONS, contact Genzyme Corporation at 1-800-633-1610 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Drug Interactions
No drug interaction studies have been performed. THYMOGLOBULIN can stimulate the production of antibodies that cross-react with rabbit immune globulins [see Clinical Pharmacology (12.3) ] .
Storage and Handling
Store in refrigerator at 2°C to 8°C (36°F to 46°F). Protect from light. Do not freeze. Do not use after the expiration date indicated on the label. Reconstituted THYMOGLOBULIN is physically and chemically stable for up to 24 hours at room temperature; however, room temperature storage is not recommended. As THYMOGLOBULIN contains no preservatives, reconstituted product should be used immediately. Infusion solutions of THYMOGLOBULIN must be used immediately. Any unused drug remaining after infusion must be discarded.
How Supplied
THYMOGLOBULIN is supplied as a single-dose clear glass 10 mL vial containing 25 mg of lyophilized (solid) THYMOGLOBULIN. Each carton contains one THYMOGLOBULIN vial (NDC 58468-0080-1).
Description
THYMOGLOBULIN is indicated for the prophylaxis and treatment of acute rejection in adult and pediatric patients receiving a kidney transplant in conjunction with concomitant immunosuppression.
Section 42229-5
Prophylaxis of Acute Rejection
The recommended dosage of THYMOGLOBULIN for prophylaxis of acute rejection in patients receiving a kidney transplant is 1.5 mg/kg of body weight administered daily with the first dose initiated prior to reperfusion of the donor kidney. The usual duration of administration is 4 to 7 days.
10 Overdosage
THYMOGLOBULIN overdosage may result in leukopenia (including lymphopenia and neutropenia) and/ or thrombocytopenia, which can be managed with dose reduction [see Dosage and Administration (2.1, 2.3)].
5.4 Infection
THYMOGLOBULIN is routinely used in combination with other immunosuppressive agents. Infections (bacterial, fungal, viral and protozoal), reactivation of infection (particularly cytomegalovirus [CMV]) and sepsis have been reported after THYMOGLOBULIN administration in combination with multiple immunosuppressive agents [see Adverse Reactions (6)]. These infections can be fatal.
Monitor patients carefully and administer appropriate anti-infective treatment when indicated [see Dosage and Administration (2.4)].
11 Description
THYMOGLOBULIN® (anti-thymocyte globulin [rabbit]) is a purified, pasteurized, immunoglobulin G, obtained by immunization of rabbits with human thymocytes. This immunosuppressive product contains cytotoxic antibodies directed against antigens expressed on human T-lymphocytes.
THYMOGLOBULIN is a sterile, lyophilized powder for intravenous administration after reconstitution with sterile Water for Injection, USP (SWFI). Each single-dose 10 mL vial contains 25 mg of anti-thymocyte globulin (rabbit), 50 mg glycine, 10 mg sodium chloride, and 50 mg mannitol.
After reconstitution with 5 mL SWFI, each vial of reconstituted product contains approximately 5 mg/mL of THYMOGLOBULIN, of which >90% is rabbit gamma immune globulin (IgG). The reconstituted solution has a pH of 6.5 to 7.2.
Human red blood cells are used in the manufacturing process to deplete cross-reactive antibodies to non–T-cell antigens. The manufacturing process is validated to remove or inactivate potential exogenous viruses. All human red blood cells are from U.S.-registered or FDA-licensed blood banks. A virus removal step (nanofiltration, using a 20 nm filter) and a viral inactivation step (pasteurization, i.e., heat treatment of active ingredient at 60°C/10 hr) are performed for each lot. Each THYMOGLOBULIN lot is released following potency testing (lymphocytotoxicity and anti-CD2 binding inhibition), and cross-reactive antibody testing (hemagglutination, platelet agglutination, and fibroblast toxicity assays on every lot).
5.3 Cytopenias
Cytopenias including anemia, neutropenia, and thrombocytopenia have occurred with THYMOGLOBULIN administration [see Adverse Reactions (6)]. Monitors blood counts after THYMOGLOBULIN administration. Adjust dose accordingly to reverse cytopenias [see Dosage and Administration (2.3)].
5.5 Malignancy
Malignancies with fatal outcomes have been reported in patients treated with THYMOGLOBULIN [see Adverse Reactions (6)]. Use of immunosuppressive agents, including THYMOGLOBULIN, may increase the risk of malignancies, including lymphoma or lymphoproliferative disorders.
16.1 How Supplied
THYMOGLOBULIN is supplied as a single-dose clear glass 10 mL vial containing 25 mg of lyophilized (solid) THYMOGLOBULIN. Each carton contains one THYMOGLOBULIN vial (NDC 58468-0080-1).
5.6 Immunizations
The safety of immunization with attenuated live vaccines following THYMOGLOBULIN therapy has not been studied; therefore, immunization with attenuated live vaccines is not recommended for patients who have recently received THYMOGLOBULIN.
8.4 Pediatric Use
The safety and effectiveness of THYMOGLOBULIN in pediatric patients have been established in pediatric patients for the prophylaxis and treatment of acute rejection. The use of THYMOGLOBULIN in pediatric patients was supported by extrapolation of adult data from Study 1, Study 2 and Study 3 [see Adverse Reactions (6) and Clinical Studies (14)].
4 Contraindications
THYMOGLOBULIN is contraindicated in patients with history of allergy or anaphylactic reaction to rabbit proteins or to any product excipients, or who have active acute or chronic infections that contraindicate any additional immunosuppression [see Warnings and Precautions (5.2, 5.4) and Adverse Reactions (6.2)].
6 Adverse Reactions
The most common adverse reactions and laboratory abnormalities (incidence >5% higher than comparator) are urinary tract infection, abdominal pain, hypertension, nausea, shortness of breath, fever, headache, anxiety, chills, increased potassium levels in the blood, low counts of platelets and white blood cells. (6)
To report SUSPECTED ADVERSE REACTIONS, contact Genzyme Corporation at 1-800-633-1610 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
7 Drug Interactions
No drug interaction studies have been performed.
THYMOGLOBULIN can stimulate the production of antibodies that cross-react with rabbit immune globulins [see Clinical Pharmacology (12.3)].
5.7 Laboratory Tests
THYMOGLOBULIN may interfere with rabbit antibody–based immunoassays and with cross-match or panel-reactive antibody cytotoxicity assays. THYMOGLOBULIN has not been shown to interfere with any routine clinical laboratory tests that do not use immunoglobulins.
12.3 Pharmacokinetics
After an intravenous dose of 1.25 to 1.5 mg/kg/day (over 4 hours for 7–11 days) 4–8 hours post infusion, THYMOGLOBULIN levels were on average 21.5 mcg/mL (10–40 mcg/mL) with a half-life of 2–3 days after the first dose, and 87 mcg/mL (23–170 mcg/mL) after the last dose.
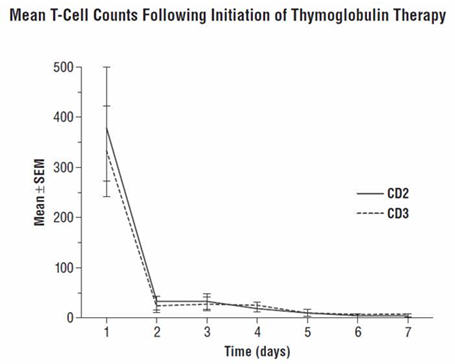
During the THYMOGLOBULIN Phase 3 randomized trial for the treatment of acute rejection, of the 108 of 163 patients evaluated, anti-rabbit antibodies developed in 68% of the THYMOGLOBULIN-treated patients, and anti-horse antibodies developed in 78% of the Active Comparator-treated patients. No controlled studies have been conducted to study the effect of anti-rabbit antibodies on repeat use of THYMOGLOBULIN. However, to ensure that T-cell depletion is achieved upon retreatment with THYMOGLOBULIN, monitoring the lymphocyte count is recommended. T-cell counts based on data collected from a limited number of patients (n=12) in this study, are presented in the chart below. These data were collected using flow cytometry (FACSCAN, Becton-Dickinson).
2.1 Dosing Information
For intravenous use only
2.3 Dose Modifications
Monitor patients for adverse reactions during and after infusion. Monitor total white blood cell and platelet counts during and after THYMOGLOBULIN therapy.
Reduce the THYMOGLOBULIN dose by one-half if the white blood cell (WBC) count is between 2,000 and 3,000 cells/mm3 or if the platelet count is between 50,000 and 75,000 cells/mm3. Consider stopping THYMOGLOBULIN treatment if the WBC count falls below 2,000 cells/mm3 or if the platelet count falls below 50,000 cells/mm3.
1 Indications and Usage
THYMOGLOBULIN is indicated for the prophylaxis and treatment of acute rejection in adult and pediatric patients receiving a kidney transplant in conjunction with concomitant immunosuppression.
12.1 Mechanism of Action
The mechanism of action by which polyclonal antilymphocyte preparations suppress immune responses is not fully understood. Possible mechanisms by which THYMOGLOBULIN may induce immunosuppression in vivo include: T-cell clearance from the circulation and modulation of T-cell activation, homing, and cytotoxic activities. THYMOGLOBULIN includes antibodies against T-cell markers such as CD2, CD3, CD4, CD8, CD11a, CD18, CD25, CD44, CD45, HLA-DR, HLA Class I heavy chains, and ß2 micro-globulin. In vitro, THYMOGLOBULIN (concentrations >0.1 mg/mL) mediates T-cell suppressive effects via inhibition of proliferative responses to several mitogens. In patients, T-cell depletion is usually observed within a day after initiating THYMOGLOBULIN therapy.
THYMOGLOBULIN has not been shown to be effective for treating antibody-mediated (humoral) rejections.
16.2 Storage and Handling
- Store in refrigerator at 2°C to 8°C (36°F to 46°F).
- Protect from light.
- Do not freeze.
- Do not use after the expiration date indicated on the label.
- Reconstituted THYMOGLOBULIN is physically and chemically stable for up to 24 hours at room temperature; however, room temperature storage is not recommended. As THYMOGLOBULIN contains no preservatives, reconstituted product should be used immediately.
- Infusion solutions of THYMOGLOBULIN must be used immediately.
- Any unused drug remaining after infusion must be discarded.
5 Warnings and Precautions
- THYMOGLOBULIN should only be used by physicians experienced in immunosuppressant therapy in transplantation. (5.1)
- Hypersensitivity and infusion-related reactions: THYMOGLOBULIN infusion could result in an anaphylactic reaction. Close compliance with the recommended infusion time may reduce the incidence and severity of infusion-related reactions. (5.2)
- Cytopenias including anemia, neutropenia, and thrombocytopenia have occurred with THYMOGLOBULIN administration (6) and require monitoring of blood counts. Adjust dose accordingly to reverse cytopenias. (5.3)
- Infection: Infections and reactivation of infections have been reported. Monitor patients and administer anti-infective prophylaxis. (5.4)
- Malignancy: Incidence of malignancies may increase. (5.5)
- Immunization with attenuated live vaccines is not recommended for patients who have recently received THYMOGLOBULIN. (5.6)
- THYMOGLOBULIN may interfere with rabbit antibody–based immunoassays and with cross-match or panel-reactive antibody cytotoxicity assays. (5.7)
2 Dosage and Administration
- The first dose should be infused over at least 6 hours; doses on subsequent days should be infused over at least 4 hours. (2.2)
- Premedication with corticosteroids, acetaminophen, and/or an antihistamine prior to each infusion is recommended. (2.2)
- The THYMOGLOBULIN dose should be reduced by one-half if the white blood cell (WBC) count is between 2,000 and 3,000 cells/mm3 or if the platelet count is between 50,000 and 75,000 cells/mm3. Stopping THYMOGLOBULIN treatment should be considered if the WBC count falls below 2,000 cells/mm3 or if the platelet count falls below 50,000 cells/mm3. (2.3)
| Indication | Dose |
|---|---|
| Prophylaxis of acute rejection | 1.5 mg/kg of body weight administered daily for 4 to 7 days |
| Treatment of acute rejection | 1.5 mg/kg of body weight administered daily for 7 to 14 days |
For complete dosing instructions, see full prescribing information. (2)
3 Dosage Forms and Strengths
THYMOGLOBULIN for injection: 25 mg anti-thymocyte globulin (rabbit) as a sterile lyophilized powder, in single-dose 10 mL vials for reconstitution.
6.2 Postmarketing Experience
The following adverse reactions have been identified during postapproval use of THYMOGLOBULIN. Because these adverse reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
- Hepatobiliary disorders: Hepatic dysfunction including transient reversible elevations in aminotransferases without any clinical signs or symptoms, hepatic failure, hyperbilirubinemia.
- Blood and lymphatic system disorders: Febrile neutropenia, coagulopathy without clinical signs or symptoms of bleeding, disseminated intravascular coagulopathy, anemia including hemolytic anemia, thrombotic microangiopathy.
- Immune system disorders: Hypersensitivity reactions including anaphylaxis, CRS.
2.2 Recommended Dosing Regimen
Administer the first dose of THYMOGLOBULIN over a minimum of 6 hours; administer doses on subsequent days over at least 4 hours [see Warnings and Precautions (5.2)].
Premedicate with corticosteroids, acetaminophen, and/or an antihistamine 1 hour prior to each infusion of THYMOGLOBULIN to reduce the incidence and intensity of infusion-related reactions [see Warnings and Precautions (5.2) and Adverse Reactions (6.1)].
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The most common adverse reactions and laboratory abnormalities (incidence >5% higher than comparator) are urinary tract infection, abdominal pain, hypertension, nausea, shortness of breath, fever, headache, anxiety, chills, increased potassium levels in the blood, and low counts of platelets and white blood cells.
17 Patient Counseling Information
Advise patients receiving THYMOGLOBULIN that they will be monitored by a physician experienced in immunosuppressive therapy for the management of kidney transplant patients [see Warnings and Precautions (5.1)].
5.1 Management of Immunosuppression
To prevent over-immunosuppression, physicians may wish to decrease the dose of the maintenance immunosuppression regimen during the period of THYMOGLOBULIN use.
2.4 Recommended Concomitant Medication
THYMOGLOBULIN is used with concomitant immunosuppressants.
Administer prophylactic antifungal and antibacterial therapy if clinically indicated [see Warnings and Precautions (5.4)].
Antiviral prophylactic therapy is recommended for patients who are seropositive for cytomegalovirus (CMV) at the time of transplant and for CMV-seronegative patients scheduled to receive a kidney from a CMV-seropositive donor [see Warnings and Precautions (5.4)].
Principal Display Panel 25 Mg Vial Carton
NDC 58468-0080-1
Rx only
Thymoglobulin®
Anti-thymocyte Globulin (Rabbit)
25 mg per vial
For Injection
For Intravenous Infusion
One single-dose vial
Discard unused portion
SANOFI GENZYME
5.2 Hypersensitivity and Infusion Related Reactions
Severe hypersensitivity and infusion-related reactions, including fatal anaphylaxis and severe cytokine release syndrome (CRS), have been reported with the use of THYMOGLOBULIN [see Adverse Reactions (6)]. Severe acute CRS can cause serious cardiorespiratory events and/or death. Close compliance with the recommended dosage and infusion time may reduce the incidence and severity of infusion-related reactions. Slowing the infusion rate may minimize the risk of infusion-related reactions.
If a hypersensitivity or infusion-related reaction occurs, terminate the infusion immediately and provide supportive treatment according to clinical practice.
Structured Label Content
Section 42229-5 (42229-5)
Prophylaxis of Acute Rejection
The recommended dosage of THYMOGLOBULIN for prophylaxis of acute rejection in patients receiving a kidney transplant is 1.5 mg/kg of body weight administered daily with the first dose initiated prior to reperfusion of the donor kidney. The usual duration of administration is 4 to 7 days.
10 Overdosage (10 OVERDOSAGE)
THYMOGLOBULIN overdosage may result in leukopenia (including lymphopenia and neutropenia) and/ or thrombocytopenia, which can be managed with dose reduction [see Dosage and Administration (2.1, 2.3)].
5.4 Infection
THYMOGLOBULIN is routinely used in combination with other immunosuppressive agents. Infections (bacterial, fungal, viral and protozoal), reactivation of infection (particularly cytomegalovirus [CMV]) and sepsis have been reported after THYMOGLOBULIN administration in combination with multiple immunosuppressive agents [see Adverse Reactions (6)]. These infections can be fatal.
Monitor patients carefully and administer appropriate anti-infective treatment when indicated [see Dosage and Administration (2.4)].
11 Description (11 DESCRIPTION)
THYMOGLOBULIN® (anti-thymocyte globulin [rabbit]) is a purified, pasteurized, immunoglobulin G, obtained by immunization of rabbits with human thymocytes. This immunosuppressive product contains cytotoxic antibodies directed against antigens expressed on human T-lymphocytes.
THYMOGLOBULIN is a sterile, lyophilized powder for intravenous administration after reconstitution with sterile Water for Injection, USP (SWFI). Each single-dose 10 mL vial contains 25 mg of anti-thymocyte globulin (rabbit), 50 mg glycine, 10 mg sodium chloride, and 50 mg mannitol.
After reconstitution with 5 mL SWFI, each vial of reconstituted product contains approximately 5 mg/mL of THYMOGLOBULIN, of which >90% is rabbit gamma immune globulin (IgG). The reconstituted solution has a pH of 6.5 to 7.2.
Human red blood cells are used in the manufacturing process to deplete cross-reactive antibodies to non–T-cell antigens. The manufacturing process is validated to remove or inactivate potential exogenous viruses. All human red blood cells are from U.S.-registered or FDA-licensed blood banks. A virus removal step (nanofiltration, using a 20 nm filter) and a viral inactivation step (pasteurization, i.e., heat treatment of active ingredient at 60°C/10 hr) are performed for each lot. Each THYMOGLOBULIN lot is released following potency testing (lymphocytotoxicity and anti-CD2 binding inhibition), and cross-reactive antibody testing (hemagglutination, platelet agglutination, and fibroblast toxicity assays on every lot).
5.3 Cytopenias
Cytopenias including anemia, neutropenia, and thrombocytopenia have occurred with THYMOGLOBULIN administration [see Adverse Reactions (6)]. Monitors blood counts after THYMOGLOBULIN administration. Adjust dose accordingly to reverse cytopenias [see Dosage and Administration (2.3)].
5.5 Malignancy
Malignancies with fatal outcomes have been reported in patients treated with THYMOGLOBULIN [see Adverse Reactions (6)]. Use of immunosuppressive agents, including THYMOGLOBULIN, may increase the risk of malignancies, including lymphoma or lymphoproliferative disorders.
16.1 How Supplied
THYMOGLOBULIN is supplied as a single-dose clear glass 10 mL vial containing 25 mg of lyophilized (solid) THYMOGLOBULIN. Each carton contains one THYMOGLOBULIN vial (NDC 58468-0080-1).
5.6 Immunizations
The safety of immunization with attenuated live vaccines following THYMOGLOBULIN therapy has not been studied; therefore, immunization with attenuated live vaccines is not recommended for patients who have recently received THYMOGLOBULIN.
8.4 Pediatric Use
The safety and effectiveness of THYMOGLOBULIN in pediatric patients have been established in pediatric patients for the prophylaxis and treatment of acute rejection. The use of THYMOGLOBULIN in pediatric patients was supported by extrapolation of adult data from Study 1, Study 2 and Study 3 [see Adverse Reactions (6) and Clinical Studies (14)].
4 Contraindications (4 CONTRAINDICATIONS)
THYMOGLOBULIN is contraindicated in patients with history of allergy or anaphylactic reaction to rabbit proteins or to any product excipients, or who have active acute or chronic infections that contraindicate any additional immunosuppression [see Warnings and Precautions (5.2, 5.4) and Adverse Reactions (6.2)].
6 Adverse Reactions (6 ADVERSE REACTIONS)
The most common adverse reactions and laboratory abnormalities (incidence >5% higher than comparator) are urinary tract infection, abdominal pain, hypertension, nausea, shortness of breath, fever, headache, anxiety, chills, increased potassium levels in the blood, low counts of platelets and white blood cells. (6)
To report SUSPECTED ADVERSE REACTIONS, contact Genzyme Corporation at 1-800-633-1610 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
7 Drug Interactions (7 DRUG INTERACTIONS)
No drug interaction studies have been performed.
THYMOGLOBULIN can stimulate the production of antibodies that cross-react with rabbit immune globulins [see Clinical Pharmacology (12.3)].
5.7 Laboratory Tests
THYMOGLOBULIN may interfere with rabbit antibody–based immunoassays and with cross-match or panel-reactive antibody cytotoxicity assays. THYMOGLOBULIN has not been shown to interfere with any routine clinical laboratory tests that do not use immunoglobulins.
12.3 Pharmacokinetics
After an intravenous dose of 1.25 to 1.5 mg/kg/day (over 4 hours for 7–11 days) 4–8 hours post infusion, THYMOGLOBULIN levels were on average 21.5 mcg/mL (10–40 mcg/mL) with a half-life of 2–3 days after the first dose, and 87 mcg/mL (23–170 mcg/mL) after the last dose.
During the THYMOGLOBULIN Phase 3 randomized trial for the treatment of acute rejection, of the 108 of 163 patients evaluated, anti-rabbit antibodies developed in 68% of the THYMOGLOBULIN-treated patients, and anti-horse antibodies developed in 78% of the Active Comparator-treated patients. No controlled studies have been conducted to study the effect of anti-rabbit antibodies on repeat use of THYMOGLOBULIN. However, to ensure that T-cell depletion is achieved upon retreatment with THYMOGLOBULIN, monitoring the lymphocyte count is recommended. T-cell counts based on data collected from a limited number of patients (n=12) in this study, are presented in the chart below. These data were collected using flow cytometry (FACSCAN, Becton-Dickinson).
2.1 Dosing Information
For intravenous use only
2.3 Dose Modifications
Monitor patients for adverse reactions during and after infusion. Monitor total white blood cell and platelet counts during and after THYMOGLOBULIN therapy.
Reduce the THYMOGLOBULIN dose by one-half if the white blood cell (WBC) count is between 2,000 and 3,000 cells/mm3 or if the platelet count is between 50,000 and 75,000 cells/mm3. Consider stopping THYMOGLOBULIN treatment if the WBC count falls below 2,000 cells/mm3 or if the platelet count falls below 50,000 cells/mm3.
1 Indications and Usage (1 INDICATIONS AND USAGE)
THYMOGLOBULIN is indicated for the prophylaxis and treatment of acute rejection in adult and pediatric patients receiving a kidney transplant in conjunction with concomitant immunosuppression.
12.1 Mechanism of Action
The mechanism of action by which polyclonal antilymphocyte preparations suppress immune responses is not fully understood. Possible mechanisms by which THYMOGLOBULIN may induce immunosuppression in vivo include: T-cell clearance from the circulation and modulation of T-cell activation, homing, and cytotoxic activities. THYMOGLOBULIN includes antibodies against T-cell markers such as CD2, CD3, CD4, CD8, CD11a, CD18, CD25, CD44, CD45, HLA-DR, HLA Class I heavy chains, and ß2 micro-globulin. In vitro, THYMOGLOBULIN (concentrations >0.1 mg/mL) mediates T-cell suppressive effects via inhibition of proliferative responses to several mitogens. In patients, T-cell depletion is usually observed within a day after initiating THYMOGLOBULIN therapy.
THYMOGLOBULIN has not been shown to be effective for treating antibody-mediated (humoral) rejections.
16.2 Storage and Handling
- Store in refrigerator at 2°C to 8°C (36°F to 46°F).
- Protect from light.
- Do not freeze.
- Do not use after the expiration date indicated on the label.
- Reconstituted THYMOGLOBULIN is physically and chemically stable for up to 24 hours at room temperature; however, room temperature storage is not recommended. As THYMOGLOBULIN contains no preservatives, reconstituted product should be used immediately.
- Infusion solutions of THYMOGLOBULIN must be used immediately.
- Any unused drug remaining after infusion must be discarded.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- THYMOGLOBULIN should only be used by physicians experienced in immunosuppressant therapy in transplantation. (5.1)
- Hypersensitivity and infusion-related reactions: THYMOGLOBULIN infusion could result in an anaphylactic reaction. Close compliance with the recommended infusion time may reduce the incidence and severity of infusion-related reactions. (5.2)
- Cytopenias including anemia, neutropenia, and thrombocytopenia have occurred with THYMOGLOBULIN administration (6) and require monitoring of blood counts. Adjust dose accordingly to reverse cytopenias. (5.3)
- Infection: Infections and reactivation of infections have been reported. Monitor patients and administer anti-infective prophylaxis. (5.4)
- Malignancy: Incidence of malignancies may increase. (5.5)
- Immunization with attenuated live vaccines is not recommended for patients who have recently received THYMOGLOBULIN. (5.6)
- THYMOGLOBULIN may interfere with rabbit antibody–based immunoassays and with cross-match or panel-reactive antibody cytotoxicity assays. (5.7)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
- The first dose should be infused over at least 6 hours; doses on subsequent days should be infused over at least 4 hours. (2.2)
- Premedication with corticosteroids, acetaminophen, and/or an antihistamine prior to each infusion is recommended. (2.2)
- The THYMOGLOBULIN dose should be reduced by one-half if the white blood cell (WBC) count is between 2,000 and 3,000 cells/mm3 or if the platelet count is between 50,000 and 75,000 cells/mm3. Stopping THYMOGLOBULIN treatment should be considered if the WBC count falls below 2,000 cells/mm3 or if the platelet count falls below 50,000 cells/mm3. (2.3)
| Indication | Dose |
|---|---|
| Prophylaxis of acute rejection | 1.5 mg/kg of body weight administered daily for 4 to 7 days |
| Treatment of acute rejection | 1.5 mg/kg of body weight administered daily for 7 to 14 days |
For complete dosing instructions, see full prescribing information. (2)
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
THYMOGLOBULIN for injection: 25 mg anti-thymocyte globulin (rabbit) as a sterile lyophilized powder, in single-dose 10 mL vials for reconstitution.
6.2 Postmarketing Experience
The following adverse reactions have been identified during postapproval use of THYMOGLOBULIN. Because these adverse reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
- Hepatobiliary disorders: Hepatic dysfunction including transient reversible elevations in aminotransferases without any clinical signs or symptoms, hepatic failure, hyperbilirubinemia.
- Blood and lymphatic system disorders: Febrile neutropenia, coagulopathy without clinical signs or symptoms of bleeding, disseminated intravascular coagulopathy, anemia including hemolytic anemia, thrombotic microangiopathy.
- Immune system disorders: Hypersensitivity reactions including anaphylaxis, CRS.
2.2 Recommended Dosing Regimen
Administer the first dose of THYMOGLOBULIN over a minimum of 6 hours; administer doses on subsequent days over at least 4 hours [see Warnings and Precautions (5.2)].
Premedicate with corticosteroids, acetaminophen, and/or an antihistamine 1 hour prior to each infusion of THYMOGLOBULIN to reduce the incidence and intensity of infusion-related reactions [see Warnings and Precautions (5.2) and Adverse Reactions (6.1)].
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The most common adverse reactions and laboratory abnormalities (incidence >5% higher than comparator) are urinary tract infection, abdominal pain, hypertension, nausea, shortness of breath, fever, headache, anxiety, chills, increased potassium levels in the blood, and low counts of platelets and white blood cells.
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise patients receiving THYMOGLOBULIN that they will be monitored by a physician experienced in immunosuppressive therapy for the management of kidney transplant patients [see Warnings and Precautions (5.1)].
5.1 Management of Immunosuppression
To prevent over-immunosuppression, physicians may wish to decrease the dose of the maintenance immunosuppression regimen during the period of THYMOGLOBULIN use.
2.4 Recommended Concomitant Medication
THYMOGLOBULIN is used with concomitant immunosuppressants.
Administer prophylactic antifungal and antibacterial therapy if clinically indicated [see Warnings and Precautions (5.4)].
Antiviral prophylactic therapy is recommended for patients who are seropositive for cytomegalovirus (CMV) at the time of transplant and for CMV-seronegative patients scheduled to receive a kidney from a CMV-seropositive donor [see Warnings and Precautions (5.4)].
Principal Display Panel 25 Mg Vial Carton (PRINCIPAL DISPLAY PANEL - 25 mg Vial Carton)
NDC 58468-0080-1
Rx only
Thymoglobulin®
Anti-thymocyte Globulin (Rabbit)
25 mg per vial
For Injection
For Intravenous Infusion
One single-dose vial
Discard unused portion
SANOFI GENZYME
5.2 Hypersensitivity and Infusion Related Reactions (5.2 Hypersensitivity and Infusion-Related Reactions)
Severe hypersensitivity and infusion-related reactions, including fatal anaphylaxis and severe cytokine release syndrome (CRS), have been reported with the use of THYMOGLOBULIN [see Adverse Reactions (6)]. Severe acute CRS can cause serious cardiorespiratory events and/or death. Close compliance with the recommended dosage and infusion time may reduce the incidence and severity of infusion-related reactions. Slowing the infusion rate may minimize the risk of infusion-related reactions.
If a hypersensitivity or infusion-related reaction occurs, terminate the infusion immediately and provide supportive treatment according to clinical practice.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T02:18:20.084474 · Updated: 2026-03-14T22:49:56.242164