Vagisil Maximum Strength Anti-itch Creme
bb73ebd7-ceb0-4b54-9e8f-77486d9037fb
34390-5
HUMAN OTC DRUG LABEL
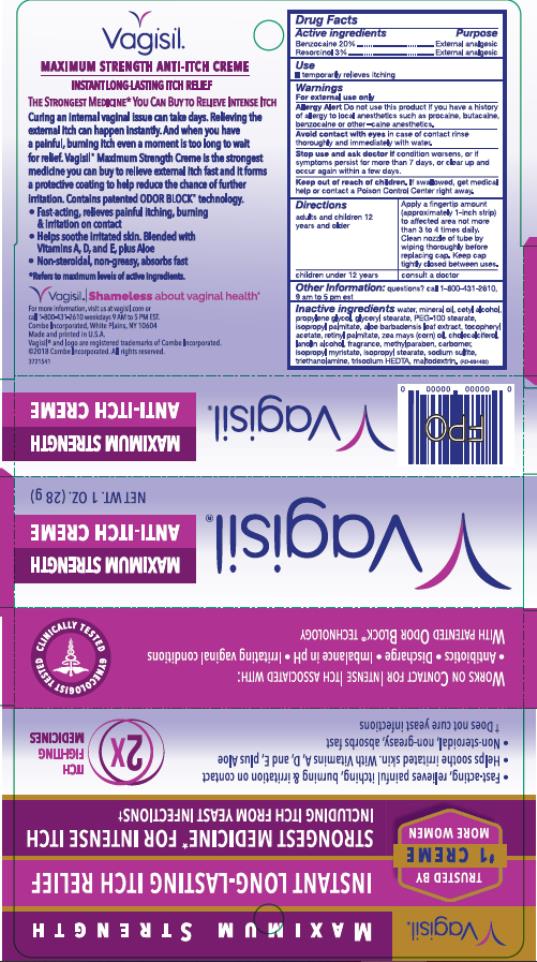
Drug Facts
Composition & Product
Identifiers & Packaging
Description
VAGISIL Maximum Strength Anti-Itch Creme Drug Facts
Purpose
External analgesic
Medication Information
Purpose
External analgesic
Description
VAGISIL Maximum Strength Anti-Itch Creme Drug Facts
Use
temporarily relieves itching
Section 42229-5
VAGISIL Maximum Strength Anti-Itch Creme
Drug Facts
Warnings
For external use only
Allergy Allert
Do not use this product if you have a history of allergy to local anesthetics such as procaine, butacaine, benzocaine or other –caine anesthetics.
Avoid contact with eyes
in case of contact rinse thoroughly and immediately with water.
Directions
| adults and children 12 years and older |
Apply a fingertip amount (approximately 1-inch strip) to affected area not more than 3 to 4 times daily. Clean nozzle of tube by wiping thoroughly before replacing cap. Keep cap tightly closed between uses. |
| children under 12 years | consult a doctor |
Active Ingredient
Benzocaine 20%
Other Information:
questions? call 1-800-431-2610, 9 am to 5 pm est
Inactive Ingredients
Water, Mineral Oil, Cetyl Alcohol, Propylene Glycol, Glyceryl Stearate, PEG-100 Stearate, Isopropyl Palmitate, Aloe Barbadensis Leaf Extract, Tocopheryl Acetate, Retinyl Palmitate, Zea Mays (Corn) Oil, Cholecalciferol, Lanolin Alcohol, Fragrance, Methylparaben, Carbomer, Isopropyl Myristate, Isopropyl Stearate, Sodium Sulfite, Triethanolamine, Trisodium HEDTA, Maltodextrin
Principal Display Panel
Vagisil
®
Maximum Strength
Anti-Itch
Creme
Net Wt. 1 oz. (28 g)
Stop Use and Ask Doctor If
condition worsens, or if symptoms persist for more than 7 days, or clear up and occur again within a few days
Keep Out of Reach of Children.
If swallowed, get medical help or contact a Poison Control Center right away.
Structured Label Content
Use
temporarily relieves itching
Section 42229-5 (42229-5)
VAGISIL Maximum Strength Anti-Itch Creme
Drug Facts
Purpose
External analgesic
Warnings
For external use only
Allergy Allert
Do not use this product if you have a history of allergy to local anesthetics such as procaine, butacaine, benzocaine or other –caine anesthetics.
Avoid contact with eyes
in case of contact rinse thoroughly and immediately with water.
Directions
| adults and children 12 years and older |
Apply a fingertip amount (approximately 1-inch strip) to affected area not more than 3 to 4 times daily. Clean nozzle of tube by wiping thoroughly before replacing cap. Keep cap tightly closed between uses. |
| children under 12 years | consult a doctor |
Active Ingredient (Active ingredient)
Benzocaine 20%
Other Information:
questions? call 1-800-431-2610, 9 am to 5 pm est
Inactive Ingredients (Inactive ingredients)
Water, Mineral Oil, Cetyl Alcohol, Propylene Glycol, Glyceryl Stearate, PEG-100 Stearate, Isopropyl Palmitate, Aloe Barbadensis Leaf Extract, Tocopheryl Acetate, Retinyl Palmitate, Zea Mays (Corn) Oil, Cholecalciferol, Lanolin Alcohol, Fragrance, Methylparaben, Carbomer, Isopropyl Myristate, Isopropyl Stearate, Sodium Sulfite, Triethanolamine, Trisodium HEDTA, Maltodextrin
Principal Display Panel
Vagisil
®
Maximum Strength
Anti-Itch
Creme
Net Wt. 1 oz. (28 g)
Stop Use and Ask Doctor If (Stop use and ask doctor if)
condition worsens, or if symptoms persist for more than 7 days, or clear up and occur again within a few days
Keep Out of Reach of Children. (Keep out of reach of children.)
If swallowed, get medical help or contact a Poison Control Center right away.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:04:41.283657 · Updated: 2026-03-14T23:05:35.877028