These Highlights Do Not Include All The Information Needed To Use Exparel® Safely And Effectively. See Full Prescribing Information For Exparel.
bb5a9e59-0f51-11df-8a39-0800200c9a66
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Indications and Usage ( 1 ) 11/ 2023 Dosage and Administration ( 2.2 ) 11/2023
Indications and Usage
EXPAREL is indicated to produce postsurgical: Local analgesia via infiltration in patients aged 6 years and older Regional analgesia via an interscalene brachial plexus nerve block in adults Regional analgesia via a sciatic nerve block in the popliteal fossa in adults Regional analgesia via an adductor canal block in adults
Dosage and Administration
EXPAREL is for single administration only ( 2.1 ). EXPAREL is not substitutable with other bupivacaine products even if the strength is the same. Therefore, it is not possible to convert a dose from other bupivacaine products to an EXPAREL dose and vice versa ( 2.1 , 2.5 ). Do not dilute EXPAREL with water or other hypotonic solutions ( 2.1 ). The recommended dose of EXPAREL for: Local infiltration in adults is up to a maximum dose of 266 mg. See Full Prescribing Information for guidance on dose selection ( 2.2 ). Local infiltration in pediatric patients aged 6 to less than 17 years is 4 mg/kg, up to a maximum of 266 mg ( 2.2 ). Interscalene brachial plexus nerve block in adults is 133 mg ( 2.3 ). Sciatic nerve block in the popliteal fossa in adults is 133 mg ( 2.3 ). Adductor canal block in adults is 133 mg (10 mL) admixed with 50 mg (10 mL) 0.5% bupivacaine HCl, for a total volume of 20 mL ( 2.3 ). For all these nerve blocks, administer additional analgesics, which may include other immediate-release local anesthetics, as appropriate ( 2.3 ). See Full Prescribing Information for important preparation and administration instructions and compatibility considerations ( 2.4 , 2.5 ).
Warnings and Precautions
As there is a potential risk of severe life-threatening adverse reactions associated with the administration of bupivacaine, EXPAREL should be administered in a setting where trained personnel and equipment are available to promptly treat patients who show evidence of neurological or cardiac toxicity [See Overdosage (10) ] . Caution should be taken to avoid accidental intravascular injection of EXPAREL. Convulsions and cardiac arrest have occurred following accidental intravascular injection of bupivacaine and other amide-containing products. Avoid additional use of local anesthetics within 96 hours following administration of EXPAREL [see Dosage and Administration (2.4) and Clinical Pharmacology (12.3) ] . EXPAREL has not been evaluated for the following uses and, therefore, is not recommended for these routes of administration or types of analgesia: epidural intrathecal intravascular or intra-articular use regional nerve blocks other than interscalene brachial plexus nerve block, sciatic nerve block in the popliteal fossa, and adductor canal block EXPAREL has not been evaluated for use in the following patient populations and, therefore, is not recommended for administration to these groups. patients younger than 6 years old for infiltration patients younger than 18 years old for interscalene brachial plexus nerve block, sciatic nerve block in the popliteal fossa, and adductor canal block pregnant patients The potential sensory and/or motor loss with EXPAREL is temporary and varies in degree and duration depending on the site of injection and dose administered and may last for up to 5 days as seen in clinical trials.
Contraindications
EXPAREL is contraindicated in obstetrical paracervical block anesthesia [see Use in Specific Populations (8.1) ]. While EXPAREL has not been tested with this technique, the use of bupivacaine HCl with this technique has resulted in fetal bradycardia and death.
Adverse Reactions
The following serious adverse reactions have been associated with bupivacaine hydrochloride in clinical trials and are described in greater detail in other sections of the labeling: Central Nervous System Reactions [see Warnings and Precautions (5.1) ] Cardiovascular System Reactions [see Warnings and Precautions (5.1) ] Allergic Reactions [see Warnings and Precautions (5.1) ] Chondrolysis [see Warnings and Precautions (5.1) ] Methemoglobinemia [see Warnings and Precautions (5.1) ] Accidental intravascular injection [see Warnings and Precautions (5.2) ]
Drug Interactions
The toxic effects of local anesthetics are additive and concomitant use should be used with caution including monitoring for neurologic and cardiovascular effects related to local anesthetic systemic toxicity [see Dosage and Administration (2.2) , Warnings and Precautions (5.1) , and Overdosage (10) ] . Avoid additional use of local anesthetics within 96 hours following administration of EXPAREL. Patients who are administered local anesthetics, including EXPAREL, may be at increased risk of developing methemoglobinemia when concurrently exposed to the following drugs, which could include other local anesthetics: Class Examples Nitrates/Nitrites nitric oxide, nitroglycerin, nitroprusside, nitrous oxide Local anesthetics articaine, benzocaine, bupivacaine, lidocaine, mepivacaine, prilocaine, procaine, ropivacaine, tetracaine Antineoplastic agents cyclophosphamide, flutamide, hydroxyurea, ifosfamide, rasburicase Antibiotics dapsone, nitrofurantoin, para-aminosalicylic acid, sulfonamides Antimalarials chloroquine, primaquine Anticonvulsants Phenobarbital, phenytoin, sodium valproate Other drugs acetaminophen, metoclopramide, quinine, sulfasalazine
Storage and Handling
EXPAREL (bupivacaine liposome injectable suspension) is a white to off-white milky aqueous suspension that is available in the following single-dose vials. 1.3% (266 mg/20 mL) (13.3 mg/mL) single-dose vial, (NDC 65250-266-20) packaged in cartons of 10 (NDC 65250-266-09) and cartons of 4 (NDC 65250-266-04) 1.3% (133 mg/10 mL) (13.3 mg/mL) single-dose vial, (NDC 65250-133-10) packaged in cartons of 10 (NDC-65250-133-09) and cartons of 4 (NDC 65250-133-04)
How Supplied
EXPAREL (bupivacaine liposome injectable suspension) is a white to off-white milky aqueous suspension that is available in the following single-dose vials. 1.3% (266 mg/20 mL) (13.3 mg/mL) single-dose vial, (NDC 65250-266-20) packaged in cartons of 10 (NDC 65250-266-09) and cartons of 4 (NDC 65250-266-04) 1.3% (133 mg/10 mL) (13.3 mg/mL) single-dose vial, (NDC 65250-133-10) packaged in cartons of 10 (NDC-65250-133-09) and cartons of 4 (NDC 65250-133-04)
Medication Information
Warnings and Precautions
As there is a potential risk of severe life-threatening adverse reactions associated with the administration of bupivacaine, EXPAREL should be administered in a setting where trained personnel and equipment are available to promptly treat patients who show evidence of neurological or cardiac toxicity [See Overdosage (10) ] . Caution should be taken to avoid accidental intravascular injection of EXPAREL. Convulsions and cardiac arrest have occurred following accidental intravascular injection of bupivacaine and other amide-containing products. Avoid additional use of local anesthetics within 96 hours following administration of EXPAREL [see Dosage and Administration (2.4) and Clinical Pharmacology (12.3) ] . EXPAREL has not been evaluated for the following uses and, therefore, is not recommended for these routes of administration or types of analgesia: epidural intrathecal intravascular or intra-articular use regional nerve blocks other than interscalene brachial plexus nerve block, sciatic nerve block in the popliteal fossa, and adductor canal block EXPAREL has not been evaluated for use in the following patient populations and, therefore, is not recommended for administration to these groups. patients younger than 6 years old for infiltration patients younger than 18 years old for interscalene brachial plexus nerve block, sciatic nerve block in the popliteal fossa, and adductor canal block pregnant patients The potential sensory and/or motor loss with EXPAREL is temporary and varies in degree and duration depending on the site of injection and dose administered and may last for up to 5 days as seen in clinical trials.
Indications and Usage
EXPAREL is indicated to produce postsurgical: Local analgesia via infiltration in patients aged 6 years and older Regional analgesia via an interscalene brachial plexus nerve block in adults Regional analgesia via a sciatic nerve block in the popliteal fossa in adults Regional analgesia via an adductor canal block in adults
Dosage and Administration
EXPAREL is for single administration only ( 2.1 ). EXPAREL is not substitutable with other bupivacaine products even if the strength is the same. Therefore, it is not possible to convert a dose from other bupivacaine products to an EXPAREL dose and vice versa ( 2.1 , 2.5 ). Do not dilute EXPAREL with water or other hypotonic solutions ( 2.1 ). The recommended dose of EXPAREL for: Local infiltration in adults is up to a maximum dose of 266 mg. See Full Prescribing Information for guidance on dose selection ( 2.2 ). Local infiltration in pediatric patients aged 6 to less than 17 years is 4 mg/kg, up to a maximum of 266 mg ( 2.2 ). Interscalene brachial plexus nerve block in adults is 133 mg ( 2.3 ). Sciatic nerve block in the popliteal fossa in adults is 133 mg ( 2.3 ). Adductor canal block in adults is 133 mg (10 mL) admixed with 50 mg (10 mL) 0.5% bupivacaine HCl, for a total volume of 20 mL ( 2.3 ). For all these nerve blocks, administer additional analgesics, which may include other immediate-release local anesthetics, as appropriate ( 2.3 ). See Full Prescribing Information for important preparation and administration instructions and compatibility considerations ( 2.4 , 2.5 ).
Contraindications
EXPAREL is contraindicated in obstetrical paracervical block anesthesia [see Use in Specific Populations (8.1) ]. While EXPAREL has not been tested with this technique, the use of bupivacaine HCl with this technique has resulted in fetal bradycardia and death.
Adverse Reactions
The following serious adverse reactions have been associated with bupivacaine hydrochloride in clinical trials and are described in greater detail in other sections of the labeling: Central Nervous System Reactions [see Warnings and Precautions (5.1) ] Cardiovascular System Reactions [see Warnings and Precautions (5.1) ] Allergic Reactions [see Warnings and Precautions (5.1) ] Chondrolysis [see Warnings and Precautions (5.1) ] Methemoglobinemia [see Warnings and Precautions (5.1) ] Accidental intravascular injection [see Warnings and Precautions (5.2) ]
Drug Interactions
The toxic effects of local anesthetics are additive and concomitant use should be used with caution including monitoring for neurologic and cardiovascular effects related to local anesthetic systemic toxicity [see Dosage and Administration (2.2) , Warnings and Precautions (5.1) , and Overdosage (10) ] . Avoid additional use of local anesthetics within 96 hours following administration of EXPAREL. Patients who are administered local anesthetics, including EXPAREL, may be at increased risk of developing methemoglobinemia when concurrently exposed to the following drugs, which could include other local anesthetics: Class Examples Nitrates/Nitrites nitric oxide, nitroglycerin, nitroprusside, nitrous oxide Local anesthetics articaine, benzocaine, bupivacaine, lidocaine, mepivacaine, prilocaine, procaine, ropivacaine, tetracaine Antineoplastic agents cyclophosphamide, flutamide, hydroxyurea, ifosfamide, rasburicase Antibiotics dapsone, nitrofurantoin, para-aminosalicylic acid, sulfonamides Antimalarials chloroquine, primaquine Anticonvulsants Phenobarbital, phenytoin, sodium valproate Other drugs acetaminophen, metoclopramide, quinine, sulfasalazine
Storage and Handling
EXPAREL (bupivacaine liposome injectable suspension) is a white to off-white milky aqueous suspension that is available in the following single-dose vials. 1.3% (266 mg/20 mL) (13.3 mg/mL) single-dose vial, (NDC 65250-266-20) packaged in cartons of 10 (NDC 65250-266-09) and cartons of 4 (NDC 65250-266-04) 1.3% (133 mg/10 mL) (13.3 mg/mL) single-dose vial, (NDC 65250-133-10) packaged in cartons of 10 (NDC-65250-133-09) and cartons of 4 (NDC 65250-133-04)
How Supplied
EXPAREL (bupivacaine liposome injectable suspension) is a white to off-white milky aqueous suspension that is available in the following single-dose vials. 1.3% (266 mg/20 mL) (13.3 mg/mL) single-dose vial, (NDC 65250-266-20) packaged in cartons of 10 (NDC 65250-266-09) and cartons of 4 (NDC 65250-266-04) 1.3% (133 mg/10 mL) (13.3 mg/mL) single-dose vial, (NDC 65250-133-10) packaged in cartons of 10 (NDC-65250-133-09) and cartons of 4 (NDC 65250-133-04)
Description
Indications and Usage ( 1 ) 11/ 2023 Dosage and Administration ( 2.2 ) 11/2023
Section 42229-5
Limitations of Use
The safety and effectiveness of EXPAREL have not been established to produce postsurgical regional analgesia via other nerve blocks besides an interscalene brachial plexus nerve block, a sciatic nerve block in the popliteal fossa, or an adductor canal block.
Section 44425-7
Storage
Store EXPAREL vials refrigerated between 2°C to 8°C (36°F to 46°F). EXPAREL may be held at a controlled room temperature of 20°C to 25°C (68°F to 77°F) for up to 30 days in sealed, intact (unopened) vials. Do not re-refrigerate vials.
Do not freeze or expose EXPAREL to high temperatures (greater than 40°C or 104°F) for an extended period. Do not administer EXPAREL if it is suspected of having been frozen or exposed to high temperatures. Do not use the vial if the stopper is bulging.
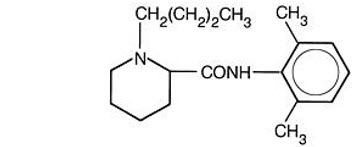
11 Description
EXPAREL (bupivacaine liposome injectable suspension) is a sterile, non-pyrogenic white to off-white preservative-free aqueous suspension consisting of multivesicular liposomes containing bupivacaine. Bupivacaine is present at a concentration of 13.3 mg/mL. After injection of EXPAREL, bupivacaine is released from the multivesicular liposomes. EXPAREL is for infiltration or perineural use.
8.4 Pediatric Use
The safety and effectiveness of EXPAREL to produce postsurgical local analgesia via infiltration have been established in pediatric patients aged 6 years and older. Use of EXPAREL for this indication is supported by evidence from adequate and well-controlled studies in adults, and pharmacokinetic (PK) and safety data in pediatric patients aged 6 years and older from Studies Peds-1 and Peds-2 [see Adverse Reactions (6.1), Clinical Pharmacology (12.3)].
- Study Peds-1 was a multicenter, randomized, open-label, two-part study (NCT03682302) to evaluate the PK and safety of EXPAREL for local infiltration in pediatric patients aged 6 to less than 17 years who were undergoing spine or cardiac surgery (postsurgically, patients were administered opioid rescue medication according to the study site's standard of care).
- Group 1: 61 patients aged 12 to less than 17 years, undergoing spine surgeries, were randomized 1:1 to receive either EXPAREL 4 mg/kg (maximum 266 mg) or bupivacaine HCl 2 mg/kg (maximum 175 mg).
- Group 2: 34 patients aged 6 to less than 12 years, undergoing either spine or cardiac surgeries, received open-label EXPAREL 4 mg/kg (maximum up to 266 mg).
- Study Peds-2 was a phase 1, open-label study that evaluated the PK and safety of 4 mg/kg (maximum 266 mg) of EXPAREL (administered intraoperatively prior to wound closure) in 15 pediatric patients aged 12 to less than 17 who were undergoing spinal surgery.
The safety and effectiveness of EXPAREL have not been established to produce postsurgical:
- Local analgesia via infiltration in pediatric patients aged less than 6 years old.
- Regional analgesia via an interscalene brachial plexus nerve block, sciatic nerve block in the popliteal fossa, or adductor canal block in pediatric patients.
8.5 Geriatric Use
Of the total number of patients in the EXPAREL local infiltration clinical studies (N=823), 171 patients were greater than or equal to 65 years of age and 47 patients were greater than or equal to 75 years of age. Of the total number of patients in the EXPAREL nerve block clinical studies (N= 1046), 312 patients were greater than or equal to 65 years of age and 70 patients were greater than or equal to 75 years of age. No overall differences in safety or effectiveness of EXPAREL have been observed between patients 65 years of age and older and younger adult patients.
In clinical studies, differences in various pharmacokinetic parameters have been observed between patients 65 years of age and older and younger adult patients. Bupivacaine is known to be substantially excreted by the kidney, and the risk of adverse reactions to bupivacaine may be greater in patients with renal impairment than in patients with normal renal function. Because patients 65 years of age and older are more likely to have renal impairment, increase monitoring for EXPAREL-associated adverse reactions [see Adverse Reactions (6)].
4 Contraindications
EXPAREL is contraindicated in obstetrical paracervical block anesthesia [see Use in Specific Populations (8.1)]. While EXPAREL has not been tested with this technique, the use of bupivacaine HCl with this technique has resulted in fetal bradycardia and death.
6 Adverse Reactions
The following serious adverse reactions have been associated with bupivacaine hydrochloride in clinical trials and are described in greater detail in other sections of the labeling:
- Central Nervous System Reactions [see Warnings and Precautions (5.1)]
- Cardiovascular System Reactions [see Warnings and Precautions (5.1)]
- Allergic Reactions [see Warnings and Precautions (5.1)]
- Chondrolysis [see Warnings and Precautions (5.1)]
- Methemoglobinemia [see Warnings and Precautions (5.1)]
- Accidental intravascular injection [see Warnings and Precautions (5.2)]
7 Drug Interactions
The toxic effects of local anesthetics are additive and concomitant use should be used with caution including monitoring for neurologic and cardiovascular effects related to local anesthetic systemic toxicity [see Dosage and Administration (2.2), Warnings and Precautions (5.1), and Overdosage (10)]. Avoid additional use of local anesthetics within 96 hours following administration of EXPAREL.
Patients who are administered local anesthetics, including EXPAREL, may be at increased risk of developing methemoglobinemia when concurrently exposed to the following drugs, which could include other local anesthetics:
| Class | Examples |
|---|---|
| Nitrates/Nitrites | nitric oxide, nitroglycerin, nitroprusside, nitrous oxide |
| Local anesthetics | articaine, benzocaine, bupivacaine, lidocaine, mepivacaine, prilocaine, procaine, ropivacaine, tetracaine |
| Antineoplastic agents | cyclophosphamide, flutamide, hydroxyurea, ifosfamide, rasburicase |
| Antibiotics | dapsone, nitrofurantoin, para-aminosalicylic acid, sulfonamides |
| Antimalarials | chloroquine, primaquine |
| Anticonvulsants | Phenobarbital, phenytoin, sodium valproate |
| Other drugs | acetaminophen, metoclopramide, quinine, sulfasalazine |
8.7 Renal Impairment
Bupivacaine is known to be substantially excreted by the kidney, and the risk of adverse reactions to EXPAREL may be greater in patients with renal impairment than in patients with normal renal function. Therefore, in patients with renal impairment, increase monitoring for EXPAREL-associated adverse reactions [see Adverse Reactions (6)].
12.2 Pharmacodynamics
Systemic absorption of local anesthetics produces effects on the cardiovascular and central nervous systems. At blood concentrations achieved with normal therapeutic doses, changes in cardiac conduction, excitability, refractoriness, contractility, and peripheral vascular resistance are minimal. However, toxic blood concentrations depress cardiac conductivity and excitability, which may lead to atrioventricular block, ventricular arrhythmias, and cardiac arrest, sometimes resulting in fatalities. In addition, myocardial contractility is depressed and peripheral vasodilation occurs, leading to decreased cardiac output and arterial blood pressure. Clinical reports and animal research suggest that these cardiovascular changes are more likely to occur after accidental intravascular injection of bupivacaine.
Following systemic absorption, local anesthetics can produce central nervous system stimulation, depression, or both. Apparent central stimulation is manifested as restlessness, tremors, and shivering progressing to convulsions, followed by depression and coma progressing ultimately to respiratory arrest. However, the local anesthetics have a primary depressant effect on the medulla and on higher centers. The depressed stage may occur without a prior excited state.
12.3 Pharmacokinetics
After administration of EXPAREL, the systemic plasma levels of bupivacaine were observed for 96 hours after local infiltration, 120 hours after interscalene brachial plexus nerve block, 168 hours after sciatic nerve block in the popliteal fossa, and 168 hours after adductor canal block [see Warnings and Precautions (5.2)]. In general, peripheral nerve blocks have shown systemic plasma levels of bupivacaine for extended duration when compared to local infiltration. Systemic plasma levels of bupivacaine following administration of EXPAREL are not correlated with local efficacy.
8.6 Hepatic Impairment
Amide-type local anesthetics, such as bupivacaine, are metabolized by the liver. Patients with severe hepatic disease, because of their inability to metabolize local anesthetics normally, are at a greater risk of developing toxic plasma concentrations, and potentially local anesthetic systemic toxicity [see Clinical Pharmacology (12.3)]. Therefore, consider increased monitoring for local anesthetic systemic toxicity in patients with moderate to severe hepatic disease.
1 Indications and Usage
EXPAREL is indicated to produce postsurgical:
- Local analgesia via infiltration in patients aged 6 years and older
- Regional analgesia via an interscalene brachial plexus nerve block in adults
- Regional analgesia via a sciatic nerve block in the popliteal fossa in adults
- Regional analgesia via an adductor canal block in adults
12.1 Mechanism of Action
Local anesthetics block the generation and the conduction of nerve impulses presumably by increasing the threshold for electrical excitation in the nerve, by slowing the propagation of the nerve impulse, and by reducing the rate of rise of the action potential. In general, the progression of anesthesia is related to the diameter, myelination, and conduction velocity of affected nerve fibers. Clinically, the order of loss of nerve function is as follows: (1) pain, (2) temperature, (3) touch, (4) proprioception, and (5) skeletal muscle tone.
5 Warnings and Precautions
- Monitor cardiovascular status, neurological status, and vital signs during and after injection of EXPAREL (5.1).
- Because amide-type local anesthetics, such as bupivacaine, are metabolized by the liver, use EXPAREL cautiously in patients with hepatic disease. Patients with severe hepatic disease, because of their inability to metabolize local anesthetics normally, are at a greater risk of developing toxic plasma concentrations (5.1).
- Methemoglobinemia: Cases of methemoglobinemia have been reported in association with local anesthetic use (5.1).
- Avoid additional use of local anesthetics within 96 hours following administration of EXPAREL (5.2).
2 Dosage and Administration
- EXPAREL is for single administration only (2.1).
- EXPAREL is not substitutable with other bupivacaine products even if the strength is the same. Therefore, it is not possible to convert a dose from other bupivacaine products to an EXPAREL dose and vice versa (2.1, 2.5).
- Do not dilute EXPAREL with water or other hypotonic solutions (2.1).
- The recommended dose of EXPAREL for:
- Local infiltration in adults is up to a maximum dose of 266 mg. See Full Prescribing Information for guidance on dose selection (2.2).
- Local infiltration in pediatric patients aged 6 to less than 17 years is 4 mg/kg, up to a maximum of 266 mg (2.2).
- Interscalene brachial plexus nerve block in adults is 133 mg (2.3).
- Sciatic nerve block in the popliteal fossa in adults is 133 mg (2.3).
- Adductor canal block in adults is 133 mg (10 mL) admixed with 50 mg (10 mL) 0.5% bupivacaine HCl, for a total volume of 20 mL (2.3).
- For all these nerve blocks, administer additional analgesics, which may include other immediate-release local anesthetics, as appropriate (2.3).
- See Full Prescribing Information for important preparation and administration instructions and compatibility considerations (2.4, 2.5).
3 Dosage Forms and Strengths
EXPAREL (bupivacaine liposome injectable suspension) is a white to off-white, milky aqueous suspension that is available in the following vial sizes:
- 1.3% (266 mg/20 mL) (13.3 mg/mL) single-dose vial
- 1.3% (133 mg/10 mL) (13.3 mg/mL) single-dose vial
6.2 Postmarketing Experience
Because adverse reactions reported during postmarketing are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
These adverse reactions are consistent with those observed in clinical studies and most commonly involve the following system organ classes: Injury, Poisoning, and Procedural Complications (e.g., drug-drug interaction, procedural pain), Nervous System Disorders (e.g., palsy, seizure), General Disorders And Administration Site Conditions (e.g., lack of efficacy, pain), Skin And Subcutaneous Tissue Disorders (e.g., erythema, rash), and Cardiac Disorders (e.g., bradycardia, cardiac arrest).
8 Use in Specific Populations
Pregnancy: May cause fetal harm (8.1).
6.1 Clinical Trials Experience
Because clinical studies are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical studies of another drug and may not reflect the rates observed in practice.
2.5 Compatibility Considerations
Some physicochemical incompatibilities exist between EXPAREL and certain other drugs. Direct contact of EXPAREL with these drugs results in a rapid increase in free (unencapsulated) bupivacaine, altering EXPAREL characteristics and potentially affecting the safety and efficacy of EXPAREL. Therefore, admixing EXPAREL with other drugs prior to administration is not recommended [see Drug Interactions (7)].
- Non-bupivacaine based local anesthetics, including lidocaine, may cause an immediate release of bupivacaine from EXPAREL if administered together locally. The administration of EXPAREL may follow the administration of lidocaine after a delay of 20 minutes or more.
- Bupivacaine HCl administered together with EXPAREL may impact the pharmacokinetic and/or physicochemical properties of EXPAREL, and this effect is concentration dependent. Therefore, bupivacaine HCl and EXPAREL may be administered simultaneously in the same syringe, and bupivacaine HCl may be injected immediately before EXPAREL if the ratio of the milligram dose of bupivacaine HCl solution to EXPAREL does not exceed 1:2.
The toxic effects of these drugs are additive, and their administration should be used with caution including monitoring for neurologic and cardiovascular effects related to local anesthetic systemic toxicity [see Warnings and Precautions (5.1) and Overdosage (10)]. - When a topical antiseptic such as povidone iodine (e.g., Betadine) is applied, the site should be allowed to dry before EXPAREL is administered into the site. EXPAREL should not be allowed to come into contact with antiseptics such as povidone iodine in solution.
Studies conducted with EXPAREL demonstrated that the most common implantable materials (polypropylene, PTFE, silicone, stainless steel, and titanium) are not affected by the presence of EXPAREL any more than they are by saline. None of the materials studied had an adverse effect on EXPAREL.
17 Patient Counseling Information
Inform patients that use of local anesthetics may cause methemoglobinemia, a serious condition that must be treated promptly. Advise patients or caregivers to seek immediate medical attention if they or someone in their care experience the following signs or symptoms: pale, gray, or blue colored skin (cyanosis); headache; rapid heart rate; shortness of breath; lightheadedness; or fatigue.
Inform patients in advance that EXPAREL can cause temporary loss of sensation or motor activity that may last for up to 5 days.
16 How Supplied/storage and Handling
EXPAREL (bupivacaine liposome injectable suspension) is a white to off-white milky aqueous suspension that is available in the following single-dose vials.
-
1.3% (266 mg/20 mL) (13.3 mg/mL) single-dose vial, (NDC 65250-266-20) packaged in cartons of 10 (NDC 65250-266-09) and cartons of 4 (NDC 65250-266-04)
-
1.3% (133 mg/10 mL) (13.3 mg/mL) single-dose vial, (NDC 65250-133-10) packaged in cartons of 10 (NDC-65250-133-09) and cartons of 4 (NDC 65250-133-04)
2.3 Recommended Dose for Regional Analgesia
The maximum recommended dose of EXPAREL via perineural use for interscalene brachial plexus nerve block, sciatic nerve block in the popliteal fossa, and adductor canal block is 133 mg. For all these nerve blocks, administer additional analgesics, which may include other immediate-release local anesthetics, as appropriate (for example, Mayo field block for bunionectomy, infiltration between the popliteal artery and capsule of the knee (IPACK) block for total knee arthroplasty).
2.4 Preparation and Administration Instructions
- Invert vials of EXPAREL multiple times to re-suspend the particles immediately prior to withdrawal from the vial.
- Administer EXPAREL (1) undiluted or (2) diluted to increase volume up to a final concentration of 0.89 mg/mL (i.e., 1:14 dilution by volume) with 0.9% preservative-free Sodium Chloride Injection or lactated Ringer's solution. Use diluted EXPAREL within 4 hours of preparation in a syringe.
- Administer EXPAREL with a 25 gauge or larger bore needle to maintain the structural integrity of the liposomal bupivacaine particles.
- Administer EXPAREL slowly via infiltration or perineural use with frequent aspiration to check for blood and minimize the risk of inadvertent intravascular injection.
- Discard unused portion.
5.2 Warnings and Precautions Specific for Exparel
As there is a potential risk of severe life-threatening adverse reactions associated with the administration of bupivacaine, EXPAREL should be administered in a setting where trained personnel and equipment are available to promptly treat patients who show evidence of neurological or cardiac toxicity [See Overdosage (10)].
Caution should be taken to avoid accidental intravascular injection of EXPAREL. Convulsions and cardiac arrest have occurred following accidental intravascular injection of bupivacaine and other amide-containing products.
Avoid additional use of local anesthetics within 96 hours following administration of EXPAREL [see Dosage and Administration (2.4) and Clinical Pharmacology (12.3)].
EXPAREL has not been evaluated for the following uses and, therefore, is not recommended for these routes of administration or types of analgesia:
- epidural
- intrathecal
- intravascular or intra-articular use
- regional nerve blocks other than interscalene brachial plexus nerve block, sciatic nerve block in the popliteal fossa, and adductor canal block
EXPAREL has not been evaluated for use in the following patient populations and, therefore, is not recommended for administration to these groups.
- patients younger than 6 years old for infiltration
- patients younger than 18 years old for interscalene brachial plexus nerve block, sciatic nerve block in the popliteal fossa, and adductor canal block
- pregnant patients
The potential sensory and/or motor loss with EXPAREL is temporary and varies in degree and duration depending on the site of injection and dose administered and may last for up to 5 days as seen in clinical trials.
Principal Display Panel 133 Mg/10 Ml Vial Label
NDC 65250-133-10
EXPAREL®
(bupivacaine liposome injectable suspension)
STERILE
1.3%
Rx Only
133 mg / 10 mL (13.3 mg/mL)
For infiltration, interscalene brachial plexus
nerve block, sciatic nerve block in the
popliteal fossa, and adductor canal block
10 mL vial
Principal Display Panel 266 Mg/20 Ml Vial Label
NDC 65250-266-20
EXPAREL®
(bupivacaine liposome injectable suspension)
STERILE
1.3%
Rx Only
266 mg/20 mL (13.3 mg/mL)
For infiltration, interscalene brachial plexus
nerve block, sciatic nerve block in the
popliteal fossa, and adductor canal block
20 mL Vial
14.1 Overview of Clinical Studies in Adult Patients
In five multicenter, randomized, double-blinded, placebo-controlled clinical studies in adults, the efficacy of EXPAREL was established to produce postsurgical:
- Local analgesia via infiltration: One study evaluated the use of EXPAREL in patients undergoing bunionectomy (Study 1); the other study evaluated the use of EXPAREL in patients undergoing hemorrhoidectomy (Study 2) [see Clinical Studies (14.2)].
- Regional analgesia via perineural use: one study (Study 3) evaluated the use of EXPAREL as a brachial plexus nerve block via interscalene or supraclavicular approach in patients undergoing total shoulder arthroplasty (TSA) or rotator cuff repair (RCR) (only two patients had nerve blocks via the supraclavicular approach); one study evaluated the use of EXPAREL as a sciatic nerve block in the popliteal fossa in patients undergoing bunionectomy (Study 4); and one study evaluated the use of EXPAREL as an adductor canal block in patients undergoing total knee arthroplasty (TKA) (Study 5) [see Clinical Studies (14.3)].
Four additional studies (Studies 6, 7, 8, and 9) [see Clinical Studies (14.4)] did not provide sufficient efficacy and/or safety data to support an indication for the following nerve blocks: femoral block in patients undergoing total knee arthroplasty, intercostal nerve block in patients undergoing posterolateral thoracotomy, and combined sciatic (in popliteal fossa) and saphenous (in adductor canal) nerve block [see Indications and Usage (1)].
2.1 Important Dose, Preparation, and Administration Instructions
- EXPAREL is for single administration only.
- EXPAREL is not substitutable with other bupivacaine products even if the strength is the same. Therefore, it is not possible to convert a dose from other bupivacaine products to an EXPAREL dose and vice versa.
- Do not dilute EXPAREL with water or other hypotonic agents, as it will result in disruption of the liposomal particles.
- Do not administer EXPAREL if it is suspected that the vial has been frozen or exposed to high temperature (greater than 40°C or 104°F) for an extended period.
- Inspect EXPAREL visually for particulate matter and discoloration prior to administration, whenever solution and container permit. Do not administer EXPAREL if the product is discolored.
- Do not heat or autoclave before use.
- Do not filter during administration.
5.1 Warnings and Precautions for Bupivacaine Containing Products
The safety and effectiveness of EXPAREL, other bupivacaine products, and other amide-containing products depend on proper dosage, correct technique, adequate precautions, and readiness for emergencies. As there is a potential risk of severe life-threatening adverse reactions associated with the administration of bupivacaine, any bupivacaine-containing product should be administered in a setting where trained personnel and equipment are available to promptly treat patients who show evidence of neurological or cardiac toxicity [see Overdosage (10)].
Careful and constant monitoring of cardiovascular and respiratory (adequacy of ventilation) vital signs and the patient's state of consciousness should be performed after injection of bupivacaine and other amide-containing products. Restlessness, anxiety, incoherent speech, lightheadedness, numbness and tingling of the mouth and lips, metallic taste, tinnitus, dizziness, blurred vision, tremors, twitching, depression, or drowsiness may be early warning signs of central nervous system toxicity.
EXPAREL, other bupivacaine products, and other amide-containing products should also be used with caution in patients with impaired cardiovascular function because they may be less able to compensate for functional changes associated with the prolongation of AV conduction produced by these drugs.
Injection of multiple doses of EXPAREL, other bupivacaine products, and other amide-containing products may cause significant increases in plasma concentrations with each repeated dose due to slow accumulation of the drug or its metabolites, or to slow metabolic degradation. Tolerance to elevated blood concentrations varies with the status of the patient.
Because amide-type local anesthetics, such as bupivacaine, are metabolized by the liver, these drugs should be used cautiously in patients with hepatic disease. Patients with severe hepatic disease, because of their inability to metabolize local anesthetics normally, are at a greater risk of developing toxic plasma concentrations.
Principal Display Panel 133 Mg/10 Ml Vial Carton Ndc 65250 133 04
NDC 65250-133-04
Contains 4
10 mL Vials
EXPAREL®
(bupivacaine liposome injectable suspension)
1.3%
133 mg/10 mL (13.3 mg/mL)
Rx Only
For infiltration, interscalene brachial plexus nerve block, sciatic nerve block
in the popliteal fossa, and adductor canal block
Contents: Each 10 mL vial contains 133 mg of bupivacaine free base.
Usual Dosage: see Prescribing Information. Do not substitute for or with other
formulations containing bupivacaine or bupivacaine HCl. Single dose vial;
discard any unused portion.
Do not freeze. Do not use the vial if the stopper is bulging.
Principal Display Panel 133 Mg/10 Ml Vial Carton Ndc 65250 133 09
NDC 65250-133-09
Contains 10
10 mL Vials
EXPAREL®
(bupivacaine liposome injectable suspension)
1.3%
133 mg/10 mL (13.3 mg/mL)
Rx Only
For infiltration, interscalene brachial plexus nerve block, sciatic nerve block
in the popliteal fossa, and adductor canal block
Contents: Each 10 mL vial contains 133 mg of bupivacaine free base.
Usual Dosage: see Prescribing Information. Do not substitute for or with other
formulations containing bupivacaine or bupivacaine HCl. Single dose vial;
discard any unused portion.
Do not freeze. Do not use the vial if the stopper is bulging.
Principal Display Panel 266 Mg/20 Ml Vial Carton Ndc 65250 266 04
NDC 65250-266-04
Contains 4
20 mL Vials
EXPAREL®
(bupivacaine liposome injectable suspension)
1.3%
266 mg/20 mL (13.3 mg/mL)
Rx Only
For infiltration, interscalene brachial plexus nerve block, sciatic nerve block
in the popliteal fossa, and adductor canal block
Contents: Each 20 mL vial contains 266 mg of bupivacaine free base.
Usual Dosage: see Prescribing Information. Do not substitute for or with other
formulations containing bupivacaine or bupivacaine HCl. Single dose vial;
discard any unused portion.
Do not freeze. Do not use the vial if the stopper is bulging.
Principal Display Panel 266 Mg/20 Ml Vial Carton Ndc 65250 266 09
NDC 65250-266-09
Contains 10
20 mL Vials
EXPAREL®
(bupivacaine liposome injectable suspension)
1.3%
266 mg/20 mL (13.3 mg/mL)
Rx Only
For infiltration, interscalene brachial plexus nerve block, sciatic nerve block
in the popliteal fossa, and adductor canal block
Contents: Each 20 mL vial contains 266 mg of bupivacaine free base.
Usual Dosage: see Prescribing Information. Do not substitute for or with other
formulations containing bupivacaine or bupivacaine HCl. Single dose vial;
discard any unused portion.
Do not freeze. Do not use the vial if the stopper is bulging.
Structured Label Content
Section 42229-5 (42229-5)
Limitations of Use
The safety and effectiveness of EXPAREL have not been established to produce postsurgical regional analgesia via other nerve blocks besides an interscalene brachial plexus nerve block, a sciatic nerve block in the popliteal fossa, or an adductor canal block.
Section 43683-2 (43683-2)
Section 44425-7 (44425-7)
Storage
Store EXPAREL vials refrigerated between 2°C to 8°C (36°F to 46°F). EXPAREL may be held at a controlled room temperature of 20°C to 25°C (68°F to 77°F) for up to 30 days in sealed, intact (unopened) vials. Do not re-refrigerate vials.
Do not freeze or expose EXPAREL to high temperatures (greater than 40°C or 104°F) for an extended period. Do not administer EXPAREL if it is suspected of having been frozen or exposed to high temperatures. Do not use the vial if the stopper is bulging.
11 Description (11 DESCRIPTION)
EXPAREL (bupivacaine liposome injectable suspension) is a sterile, non-pyrogenic white to off-white preservative-free aqueous suspension consisting of multivesicular liposomes containing bupivacaine. Bupivacaine is present at a concentration of 13.3 mg/mL. After injection of EXPAREL, bupivacaine is released from the multivesicular liposomes. EXPAREL is for infiltration or perineural use.
8.4 Pediatric Use
The safety and effectiveness of EXPAREL to produce postsurgical local analgesia via infiltration have been established in pediatric patients aged 6 years and older. Use of EXPAREL for this indication is supported by evidence from adequate and well-controlled studies in adults, and pharmacokinetic (PK) and safety data in pediatric patients aged 6 years and older from Studies Peds-1 and Peds-2 [see Adverse Reactions (6.1), Clinical Pharmacology (12.3)].
- Study Peds-1 was a multicenter, randomized, open-label, two-part study (NCT03682302) to evaluate the PK and safety of EXPAREL for local infiltration in pediatric patients aged 6 to less than 17 years who were undergoing spine or cardiac surgery (postsurgically, patients were administered opioid rescue medication according to the study site's standard of care).
- Group 1: 61 patients aged 12 to less than 17 years, undergoing spine surgeries, were randomized 1:1 to receive either EXPAREL 4 mg/kg (maximum 266 mg) or bupivacaine HCl 2 mg/kg (maximum 175 mg).
- Group 2: 34 patients aged 6 to less than 12 years, undergoing either spine or cardiac surgeries, received open-label EXPAREL 4 mg/kg (maximum up to 266 mg).
- Study Peds-2 was a phase 1, open-label study that evaluated the PK and safety of 4 mg/kg (maximum 266 mg) of EXPAREL (administered intraoperatively prior to wound closure) in 15 pediatric patients aged 12 to less than 17 who were undergoing spinal surgery.
The safety and effectiveness of EXPAREL have not been established to produce postsurgical:
- Local analgesia via infiltration in pediatric patients aged less than 6 years old.
- Regional analgesia via an interscalene brachial plexus nerve block, sciatic nerve block in the popliteal fossa, or adductor canal block in pediatric patients.
8.5 Geriatric Use
Of the total number of patients in the EXPAREL local infiltration clinical studies (N=823), 171 patients were greater than or equal to 65 years of age and 47 patients were greater than or equal to 75 years of age. Of the total number of patients in the EXPAREL nerve block clinical studies (N= 1046), 312 patients were greater than or equal to 65 years of age and 70 patients were greater than or equal to 75 years of age. No overall differences in safety or effectiveness of EXPAREL have been observed between patients 65 years of age and older and younger adult patients.
In clinical studies, differences in various pharmacokinetic parameters have been observed between patients 65 years of age and older and younger adult patients. Bupivacaine is known to be substantially excreted by the kidney, and the risk of adverse reactions to bupivacaine may be greater in patients with renal impairment than in patients with normal renal function. Because patients 65 years of age and older are more likely to have renal impairment, increase monitoring for EXPAREL-associated adverse reactions [see Adverse Reactions (6)].
4 Contraindications (4 CONTRAINDICATIONS)
EXPAREL is contraindicated in obstetrical paracervical block anesthesia [see Use in Specific Populations (8.1)]. While EXPAREL has not been tested with this technique, the use of bupivacaine HCl with this technique has resulted in fetal bradycardia and death.
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following serious adverse reactions have been associated with bupivacaine hydrochloride in clinical trials and are described in greater detail in other sections of the labeling:
- Central Nervous System Reactions [see Warnings and Precautions (5.1)]
- Cardiovascular System Reactions [see Warnings and Precautions (5.1)]
- Allergic Reactions [see Warnings and Precautions (5.1)]
- Chondrolysis [see Warnings and Precautions (5.1)]
- Methemoglobinemia [see Warnings and Precautions (5.1)]
- Accidental intravascular injection [see Warnings and Precautions (5.2)]
7 Drug Interactions (7 DRUG INTERACTIONS)
The toxic effects of local anesthetics are additive and concomitant use should be used with caution including monitoring for neurologic and cardiovascular effects related to local anesthetic systemic toxicity [see Dosage and Administration (2.2), Warnings and Precautions (5.1), and Overdosage (10)]. Avoid additional use of local anesthetics within 96 hours following administration of EXPAREL.
Patients who are administered local anesthetics, including EXPAREL, may be at increased risk of developing methemoglobinemia when concurrently exposed to the following drugs, which could include other local anesthetics:
| Class | Examples |
|---|---|
| Nitrates/Nitrites | nitric oxide, nitroglycerin, nitroprusside, nitrous oxide |
| Local anesthetics | articaine, benzocaine, bupivacaine, lidocaine, mepivacaine, prilocaine, procaine, ropivacaine, tetracaine |
| Antineoplastic agents | cyclophosphamide, flutamide, hydroxyurea, ifosfamide, rasburicase |
| Antibiotics | dapsone, nitrofurantoin, para-aminosalicylic acid, sulfonamides |
| Antimalarials | chloroquine, primaquine |
| Anticonvulsants | Phenobarbital, phenytoin, sodium valproate |
| Other drugs | acetaminophen, metoclopramide, quinine, sulfasalazine |
8.7 Renal Impairment
Bupivacaine is known to be substantially excreted by the kidney, and the risk of adverse reactions to EXPAREL may be greater in patients with renal impairment than in patients with normal renal function. Therefore, in patients with renal impairment, increase monitoring for EXPAREL-associated adverse reactions [see Adverse Reactions (6)].
12.2 Pharmacodynamics
Systemic absorption of local anesthetics produces effects on the cardiovascular and central nervous systems. At blood concentrations achieved with normal therapeutic doses, changes in cardiac conduction, excitability, refractoriness, contractility, and peripheral vascular resistance are minimal. However, toxic blood concentrations depress cardiac conductivity and excitability, which may lead to atrioventricular block, ventricular arrhythmias, and cardiac arrest, sometimes resulting in fatalities. In addition, myocardial contractility is depressed and peripheral vasodilation occurs, leading to decreased cardiac output and arterial blood pressure. Clinical reports and animal research suggest that these cardiovascular changes are more likely to occur after accidental intravascular injection of bupivacaine.
Following systemic absorption, local anesthetics can produce central nervous system stimulation, depression, or both. Apparent central stimulation is manifested as restlessness, tremors, and shivering progressing to convulsions, followed by depression and coma progressing ultimately to respiratory arrest. However, the local anesthetics have a primary depressant effect on the medulla and on higher centers. The depressed stage may occur without a prior excited state.
12.3 Pharmacokinetics
After administration of EXPAREL, the systemic plasma levels of bupivacaine were observed for 96 hours after local infiltration, 120 hours after interscalene brachial plexus nerve block, 168 hours after sciatic nerve block in the popliteal fossa, and 168 hours after adductor canal block [see Warnings and Precautions (5.2)]. In general, peripheral nerve blocks have shown systemic plasma levels of bupivacaine for extended duration when compared to local infiltration. Systemic plasma levels of bupivacaine following administration of EXPAREL are not correlated with local efficacy.
8.6 Hepatic Impairment
Amide-type local anesthetics, such as bupivacaine, are metabolized by the liver. Patients with severe hepatic disease, because of their inability to metabolize local anesthetics normally, are at a greater risk of developing toxic plasma concentrations, and potentially local anesthetic systemic toxicity [see Clinical Pharmacology (12.3)]. Therefore, consider increased monitoring for local anesthetic systemic toxicity in patients with moderate to severe hepatic disease.
1 Indications and Usage (1 INDICATIONS AND USAGE)
EXPAREL is indicated to produce postsurgical:
- Local analgesia via infiltration in patients aged 6 years and older
- Regional analgesia via an interscalene brachial plexus nerve block in adults
- Regional analgesia via a sciatic nerve block in the popliteal fossa in adults
- Regional analgesia via an adductor canal block in adults
12.1 Mechanism of Action
Local anesthetics block the generation and the conduction of nerve impulses presumably by increasing the threshold for electrical excitation in the nerve, by slowing the propagation of the nerve impulse, and by reducing the rate of rise of the action potential. In general, the progression of anesthesia is related to the diameter, myelination, and conduction velocity of affected nerve fibers. Clinically, the order of loss of nerve function is as follows: (1) pain, (2) temperature, (3) touch, (4) proprioception, and (5) skeletal muscle tone.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Monitor cardiovascular status, neurological status, and vital signs during and after injection of EXPAREL (5.1).
- Because amide-type local anesthetics, such as bupivacaine, are metabolized by the liver, use EXPAREL cautiously in patients with hepatic disease. Patients with severe hepatic disease, because of their inability to metabolize local anesthetics normally, are at a greater risk of developing toxic plasma concentrations (5.1).
- Methemoglobinemia: Cases of methemoglobinemia have been reported in association with local anesthetic use (5.1).
- Avoid additional use of local anesthetics within 96 hours following administration of EXPAREL (5.2).
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
- EXPAREL is for single administration only (2.1).
- EXPAREL is not substitutable with other bupivacaine products even if the strength is the same. Therefore, it is not possible to convert a dose from other bupivacaine products to an EXPAREL dose and vice versa (2.1, 2.5).
- Do not dilute EXPAREL with water or other hypotonic solutions (2.1).
- The recommended dose of EXPAREL for:
- Local infiltration in adults is up to a maximum dose of 266 mg. See Full Prescribing Information for guidance on dose selection (2.2).
- Local infiltration in pediatric patients aged 6 to less than 17 years is 4 mg/kg, up to a maximum of 266 mg (2.2).
- Interscalene brachial plexus nerve block in adults is 133 mg (2.3).
- Sciatic nerve block in the popliteal fossa in adults is 133 mg (2.3).
- Adductor canal block in adults is 133 mg (10 mL) admixed with 50 mg (10 mL) 0.5% bupivacaine HCl, for a total volume of 20 mL (2.3).
- For all these nerve blocks, administer additional analgesics, which may include other immediate-release local anesthetics, as appropriate (2.3).
- See Full Prescribing Information for important preparation and administration instructions and compatibility considerations (2.4, 2.5).
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
EXPAREL (bupivacaine liposome injectable suspension) is a white to off-white, milky aqueous suspension that is available in the following vial sizes:
- 1.3% (266 mg/20 mL) (13.3 mg/mL) single-dose vial
- 1.3% (133 mg/10 mL) (13.3 mg/mL) single-dose vial
6.2 Postmarketing Experience
Because adverse reactions reported during postmarketing are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
These adverse reactions are consistent with those observed in clinical studies and most commonly involve the following system organ classes: Injury, Poisoning, and Procedural Complications (e.g., drug-drug interaction, procedural pain), Nervous System Disorders (e.g., palsy, seizure), General Disorders And Administration Site Conditions (e.g., lack of efficacy, pain), Skin And Subcutaneous Tissue Disorders (e.g., erythema, rash), and Cardiac Disorders (e.g., bradycardia, cardiac arrest).
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
Pregnancy: May cause fetal harm (8.1).
6.1 Clinical Trials Experience
Because clinical studies are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical studies of another drug and may not reflect the rates observed in practice.
2.5 Compatibility Considerations
Some physicochemical incompatibilities exist between EXPAREL and certain other drugs. Direct contact of EXPAREL with these drugs results in a rapid increase in free (unencapsulated) bupivacaine, altering EXPAREL characteristics and potentially affecting the safety and efficacy of EXPAREL. Therefore, admixing EXPAREL with other drugs prior to administration is not recommended [see Drug Interactions (7)].
- Non-bupivacaine based local anesthetics, including lidocaine, may cause an immediate release of bupivacaine from EXPAREL if administered together locally. The administration of EXPAREL may follow the administration of lidocaine after a delay of 20 minutes or more.
- Bupivacaine HCl administered together with EXPAREL may impact the pharmacokinetic and/or physicochemical properties of EXPAREL, and this effect is concentration dependent. Therefore, bupivacaine HCl and EXPAREL may be administered simultaneously in the same syringe, and bupivacaine HCl may be injected immediately before EXPAREL if the ratio of the milligram dose of bupivacaine HCl solution to EXPAREL does not exceed 1:2.
The toxic effects of these drugs are additive, and their administration should be used with caution including monitoring for neurologic and cardiovascular effects related to local anesthetic systemic toxicity [see Warnings and Precautions (5.1) and Overdosage (10)]. - When a topical antiseptic such as povidone iodine (e.g., Betadine) is applied, the site should be allowed to dry before EXPAREL is administered into the site. EXPAREL should not be allowed to come into contact with antiseptics such as povidone iodine in solution.
Studies conducted with EXPAREL demonstrated that the most common implantable materials (polypropylene, PTFE, silicone, stainless steel, and titanium) are not affected by the presence of EXPAREL any more than they are by saline. None of the materials studied had an adverse effect on EXPAREL.
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Inform patients that use of local anesthetics may cause methemoglobinemia, a serious condition that must be treated promptly. Advise patients or caregivers to seek immediate medical attention if they or someone in their care experience the following signs or symptoms: pale, gray, or blue colored skin (cyanosis); headache; rapid heart rate; shortness of breath; lightheadedness; or fatigue.
Inform patients in advance that EXPAREL can cause temporary loss of sensation or motor activity that may last for up to 5 days.
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
EXPAREL (bupivacaine liposome injectable suspension) is a white to off-white milky aqueous suspension that is available in the following single-dose vials.
-
1.3% (266 mg/20 mL) (13.3 mg/mL) single-dose vial, (NDC 65250-266-20) packaged in cartons of 10 (NDC 65250-266-09) and cartons of 4 (NDC 65250-266-04)
-
1.3% (133 mg/10 mL) (13.3 mg/mL) single-dose vial, (NDC 65250-133-10) packaged in cartons of 10 (NDC-65250-133-09) and cartons of 4 (NDC 65250-133-04)
2.3 Recommended Dose for Regional Analgesia
The maximum recommended dose of EXPAREL via perineural use for interscalene brachial plexus nerve block, sciatic nerve block in the popliteal fossa, and adductor canal block is 133 mg. For all these nerve blocks, administer additional analgesics, which may include other immediate-release local anesthetics, as appropriate (for example, Mayo field block for bunionectomy, infiltration between the popliteal artery and capsule of the knee (IPACK) block for total knee arthroplasty).
2.4 Preparation and Administration Instructions
- Invert vials of EXPAREL multiple times to re-suspend the particles immediately prior to withdrawal from the vial.
- Administer EXPAREL (1) undiluted or (2) diluted to increase volume up to a final concentration of 0.89 mg/mL (i.e., 1:14 dilution by volume) with 0.9% preservative-free Sodium Chloride Injection or lactated Ringer's solution. Use diluted EXPAREL within 4 hours of preparation in a syringe.
- Administer EXPAREL with a 25 gauge or larger bore needle to maintain the structural integrity of the liposomal bupivacaine particles.
- Administer EXPAREL slowly via infiltration or perineural use with frequent aspiration to check for blood and minimize the risk of inadvertent intravascular injection.
- Discard unused portion.
5.2 Warnings and Precautions Specific for Exparel (5.2 Warnings and Precautions Specific for EXPAREL)
As there is a potential risk of severe life-threatening adverse reactions associated with the administration of bupivacaine, EXPAREL should be administered in a setting where trained personnel and equipment are available to promptly treat patients who show evidence of neurological or cardiac toxicity [See Overdosage (10)].
Caution should be taken to avoid accidental intravascular injection of EXPAREL. Convulsions and cardiac arrest have occurred following accidental intravascular injection of bupivacaine and other amide-containing products.
Avoid additional use of local anesthetics within 96 hours following administration of EXPAREL [see Dosage and Administration (2.4) and Clinical Pharmacology (12.3)].
EXPAREL has not been evaluated for the following uses and, therefore, is not recommended for these routes of administration or types of analgesia:
- epidural
- intrathecal
- intravascular or intra-articular use
- regional nerve blocks other than interscalene brachial plexus nerve block, sciatic nerve block in the popliteal fossa, and adductor canal block
EXPAREL has not been evaluated for use in the following patient populations and, therefore, is not recommended for administration to these groups.
- patients younger than 6 years old for infiltration
- patients younger than 18 years old for interscalene brachial plexus nerve block, sciatic nerve block in the popliteal fossa, and adductor canal block
- pregnant patients
The potential sensory and/or motor loss with EXPAREL is temporary and varies in degree and duration depending on the site of injection and dose administered and may last for up to 5 days as seen in clinical trials.
Principal Display Panel 133 Mg/10 Ml Vial Label (PRINCIPAL DISPLAY PANEL - 133 mg/10 mL Vial Label)
NDC 65250-133-10
EXPAREL®
(bupivacaine liposome injectable suspension)
STERILE
1.3%
Rx Only
133 mg / 10 mL (13.3 mg/mL)
For infiltration, interscalene brachial plexus
nerve block, sciatic nerve block in the
popliteal fossa, and adductor canal block
10 mL vial
Principal Display Panel 266 Mg/20 Ml Vial Label (PRINCIPAL DISPLAY PANEL - 266 mg/20 mL Vial Label)
NDC 65250-266-20
EXPAREL®
(bupivacaine liposome injectable suspension)
STERILE
1.3%
Rx Only
266 mg/20 mL (13.3 mg/mL)
For infiltration, interscalene brachial plexus
nerve block, sciatic nerve block in the
popliteal fossa, and adductor canal block
20 mL Vial
14.1 Overview of Clinical Studies in Adult Patients
In five multicenter, randomized, double-blinded, placebo-controlled clinical studies in adults, the efficacy of EXPAREL was established to produce postsurgical:
- Local analgesia via infiltration: One study evaluated the use of EXPAREL in patients undergoing bunionectomy (Study 1); the other study evaluated the use of EXPAREL in patients undergoing hemorrhoidectomy (Study 2) [see Clinical Studies (14.2)].
- Regional analgesia via perineural use: one study (Study 3) evaluated the use of EXPAREL as a brachial plexus nerve block via interscalene or supraclavicular approach in patients undergoing total shoulder arthroplasty (TSA) or rotator cuff repair (RCR) (only two patients had nerve blocks via the supraclavicular approach); one study evaluated the use of EXPAREL as a sciatic nerve block in the popliteal fossa in patients undergoing bunionectomy (Study 4); and one study evaluated the use of EXPAREL as an adductor canal block in patients undergoing total knee arthroplasty (TKA) (Study 5) [see Clinical Studies (14.3)].
Four additional studies (Studies 6, 7, 8, and 9) [see Clinical Studies (14.4)] did not provide sufficient efficacy and/or safety data to support an indication for the following nerve blocks: femoral block in patients undergoing total knee arthroplasty, intercostal nerve block in patients undergoing posterolateral thoracotomy, and combined sciatic (in popliteal fossa) and saphenous (in adductor canal) nerve block [see Indications and Usage (1)].
2.1 Important Dose, Preparation, and Administration Instructions
- EXPAREL is for single administration only.
- EXPAREL is not substitutable with other bupivacaine products even if the strength is the same. Therefore, it is not possible to convert a dose from other bupivacaine products to an EXPAREL dose and vice versa.
- Do not dilute EXPAREL with water or other hypotonic agents, as it will result in disruption of the liposomal particles.
- Do not administer EXPAREL if it is suspected that the vial has been frozen or exposed to high temperature (greater than 40°C or 104°F) for an extended period.
- Inspect EXPAREL visually for particulate matter and discoloration prior to administration, whenever solution and container permit. Do not administer EXPAREL if the product is discolored.
- Do not heat or autoclave before use.
- Do not filter during administration.
5.1 Warnings and Precautions for Bupivacaine Containing Products (5.1 Warnings and Precautions for Bupivacaine-Containing Products)
The safety and effectiveness of EXPAREL, other bupivacaine products, and other amide-containing products depend on proper dosage, correct technique, adequate precautions, and readiness for emergencies. As there is a potential risk of severe life-threatening adverse reactions associated with the administration of bupivacaine, any bupivacaine-containing product should be administered in a setting where trained personnel and equipment are available to promptly treat patients who show evidence of neurological or cardiac toxicity [see Overdosage (10)].
Careful and constant monitoring of cardiovascular and respiratory (adequacy of ventilation) vital signs and the patient's state of consciousness should be performed after injection of bupivacaine and other amide-containing products. Restlessness, anxiety, incoherent speech, lightheadedness, numbness and tingling of the mouth and lips, metallic taste, tinnitus, dizziness, blurred vision, tremors, twitching, depression, or drowsiness may be early warning signs of central nervous system toxicity.
EXPAREL, other bupivacaine products, and other amide-containing products should also be used with caution in patients with impaired cardiovascular function because they may be less able to compensate for functional changes associated with the prolongation of AV conduction produced by these drugs.
Injection of multiple doses of EXPAREL, other bupivacaine products, and other amide-containing products may cause significant increases in plasma concentrations with each repeated dose due to slow accumulation of the drug or its metabolites, or to slow metabolic degradation. Tolerance to elevated blood concentrations varies with the status of the patient.
Because amide-type local anesthetics, such as bupivacaine, are metabolized by the liver, these drugs should be used cautiously in patients with hepatic disease. Patients with severe hepatic disease, because of their inability to metabolize local anesthetics normally, are at a greater risk of developing toxic plasma concentrations.
Principal Display Panel 133 Mg/10 Ml Vial Carton Ndc 65250 133 04 (PRINCIPAL DISPLAY PANEL - 133 mg/10 mL Vial Carton - NDC 65250-133-04)
NDC 65250-133-04
Contains 4
10 mL Vials
EXPAREL®
(bupivacaine liposome injectable suspension)
1.3%
133 mg/10 mL (13.3 mg/mL)
Rx Only
For infiltration, interscalene brachial plexus nerve block, sciatic nerve block
in the popliteal fossa, and adductor canal block
Contents: Each 10 mL vial contains 133 mg of bupivacaine free base.
Usual Dosage: see Prescribing Information. Do not substitute for or with other
formulations containing bupivacaine or bupivacaine HCl. Single dose vial;
discard any unused portion.
Do not freeze. Do not use the vial if the stopper is bulging.
Principal Display Panel 133 Mg/10 Ml Vial Carton Ndc 65250 133 09 (PRINCIPAL DISPLAY PANEL - 133 mg/10 mL Vial Carton - NDC 65250-133-09)
NDC 65250-133-09
Contains 10
10 mL Vials
EXPAREL®
(bupivacaine liposome injectable suspension)
1.3%
133 mg/10 mL (13.3 mg/mL)
Rx Only
For infiltration, interscalene brachial plexus nerve block, sciatic nerve block
in the popliteal fossa, and adductor canal block
Contents: Each 10 mL vial contains 133 mg of bupivacaine free base.
Usual Dosage: see Prescribing Information. Do not substitute for or with other
formulations containing bupivacaine or bupivacaine HCl. Single dose vial;
discard any unused portion.
Do not freeze. Do not use the vial if the stopper is bulging.
Principal Display Panel 266 Mg/20 Ml Vial Carton Ndc 65250 266 04 (PRINCIPAL DISPLAY PANEL - 266 mg/20 mL Vial Carton - NDC 65250-266-04)
NDC 65250-266-04
Contains 4
20 mL Vials
EXPAREL®
(bupivacaine liposome injectable suspension)
1.3%
266 mg/20 mL (13.3 mg/mL)
Rx Only
For infiltration, interscalene brachial plexus nerve block, sciatic nerve block
in the popliteal fossa, and adductor canal block
Contents: Each 20 mL vial contains 266 mg of bupivacaine free base.
Usual Dosage: see Prescribing Information. Do not substitute for or with other
formulations containing bupivacaine or bupivacaine HCl. Single dose vial;
discard any unused portion.
Do not freeze. Do not use the vial if the stopper is bulging.
Principal Display Panel 266 Mg/20 Ml Vial Carton Ndc 65250 266 09 (PRINCIPAL DISPLAY PANEL - 266 mg/20 mL Vial Carton - NDC 65250-266-09)
NDC 65250-266-09
Contains 10
20 mL Vials
EXPAREL®
(bupivacaine liposome injectable suspension)
1.3%
266 mg/20 mL (13.3 mg/mL)
Rx Only
For infiltration, interscalene brachial plexus nerve block, sciatic nerve block
in the popliteal fossa, and adductor canal block
Contents: Each 20 mL vial contains 266 mg of bupivacaine free base.
Usual Dosage: see Prescribing Information. Do not substitute for or with other
formulations containing bupivacaine or bupivacaine HCl. Single dose vial;
discard any unused portion.
Do not freeze. Do not use the vial if the stopper is bulging.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:51:10.266149 · Updated: 2026-03-14T22:39:36.817935