These Highlights Do Not Include All The Information Needed To Use Risperdal Consta ®
bb34ee82-d2c2-43b8-ba21-2825c0954691
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Elderly patients with dementia-related psychosis treated with antipsychotic drugs are at an increased risk of death. RISPERDAL CONSTA is not approved for the treatment of patients with dementia-related psychosis. [see Warnings and Precautions (5.1) ]
Indications and Usage
RISPERDAL CONSTA is an atypical antipsychotic indicated: for the treatment of schizophrenia. ( 1.1 ) as monotherapy or as adjunctive therapy to lithium or valproate for the maintenance treatment of Bipolar I Disorder. ( 1.2 )
Dosage and Administration
For patients who have never taken oral RISPERDAL, it is recommended to establish tolerability with oral RISPERDAL prior to initiating treatment with RISPERDAL CONSTA. RISPERDAL CONSTA should be administered every 2 weeks by deep intramuscular (IM) deltoid or gluteal injection. Each injection should be administered by a health care professional using the appropriate enclosed safety needle [see Dosage and Administration (2.8) ] . For deltoid administration, use the 1-inch needle alternating injections between the two arms. For gluteal administration, use the 2-inch needle alternating injections between the two buttocks. Do not administer intravenously.
Warnings and Precautions
Cerebrovascular events, including stroke, in elderly patients with dementia-related psychosis. RISPERDAL CONSTA is not approved for use in patients with dementia-related psychosis ( 5.2 ) Neuroleptic Malignant Syndrome: Manage with immediate discontinuation and close monitoring ( 5.3 ) Tardive Dyskinesia: Discontinue treatment if clinically appropriate ( 5.4 ) Metabolic Changes: Atypical antipsychotic drugs have been associated with metabolic changes that may increase cardiovascular/ cerebrovascular risk. These metabolic changes include hyperglycemia, dyslipidemia, and weight gain. ( 5.5 ) Hyperglycemia and Diabetes Mellitus: Monitor patients for symptoms of hyperglycemia including polydipsia, polyuria, polyphagia, and weakness. Monitor glucose regularly in patients with diabetes or at risk for diabetes. ( 5.5 ) Dyslipidemia: Undesirable alterations have been observed in patients treated with atypical antipsychotics. ( 5.5 ) Weight Gain: Significant weight gain has been reported. Monitor weight gain. ( 5.5 ) Hyperprolactinemia: Risperidone treatment may elevate prolactin levels. Long-standing hyperprolactinemia, when associated with hypogonadism, can lead to decreased bone density in men and women. ( 5.6 ) Orthostatic hypotension: associated with dizziness, tachycardia, bradycardia, and syncope can occur, especially during initial dose titration with oral risperidone. Use caution in patients with cardiovascular disease, cerebrovascular disease, and conditions that could affect hemodynamic responses. ( 5.7 ) Leukopenia, Neutropenia, and Agranulocytosis have been reported with antipsychotics, including RISPERDAL CONSTA. Patients with history of a clinically significant low white blood cell count (WBC) or a drug-induced leukopenia/neutropenia should have their complete blood cell count (CBC) monitored frequently during the first few months of therapy and discontinuation of RISPERDAL CONSTA should be considered at the first sign of a clinically significant decline in WBC in the absence of other causative factors. ( 5.9 ) Potential for cognitive and motor impairment: has potential to impair judgment, thinking, and motor skills. Use caution when operating machinery, including automobiles. ( 5.10 ) Seizures: Use cautiously in patients with a history of seizures or with conditions that potentially lower the seizure threshold. ( 5.11 ) Dysphagia: Esophageal dysmotility and aspiration can occur. Use cautiously in patients at risk for aspiration pneumonia. ( 5.12 ) Priapism: has been reported. Severe priapism may require surgical intervention. ( 5.13 ) Avoid inadvertent administration into a blood vessel. ( 5.15 )
Contraindications
RISPERDAL CONSTA is contraindicated in patients with a known hypersensitivity to either risperidone or paliperidone, or to any of the excipients in the RISPERDAL CONSTA formulation. Hypersensitivity reactions, including anaphylactic reactions and angioedema, have been reported in patients treated with risperidone and in patients treated with paliperidone. Paliperidone is a metabolite of risperidone.
Adverse Reactions
The following are discussed in more detail in other sections of the labeling: Increased mortality in elderly patients with dementia-related psychosis [see Boxed Warning and Warnings and Precautions (5.1) ] Cerebrovascular adverse events, including stroke, in elderly patients with dementia-related psychosis [see Warnings and Precautions (5.2) ] Neuroleptic malignant syndrome [see Warnings and Precautions (5.3) ] Tardive dyskinesia [see Warnings and Precautions (5.4) ] Metabolic changes [see Warnings and Precautions (5.5) ] Hyperprolactinemia [see Warnings and Precautions (5.6) ] Orthostatic hypotension [see Warnings and Precautions (5.7) ] Falls [see Warnings and Precautions (5.8) ] Leukopenia/Neutropenia and Agranulocytosis [see Warnings and Precautions (5.9) ] Potential for cognitive and motor impairment [see Warnings and Precautions (5.10) ] Seizures [see Warnings and Precautions (5.11) ] Dysphagia [see Warnings and Precautions (5.12) ] Priapism [see Warnings and Precautions (5.13) ] Disruption of body temperature regulation [see Warnings and Precautions (5.14) ] Avoidance of inadvertent injection into a blood vessel [see Warnings and Precautions (5.15) ] Osteodystrophy and tumors in animals [see Warnings and Precautions (5.16) ] The most common adverse reactions in clinical trials in patients with schizophrenia (≥ 5%) were: headache, parkinsonism, dizziness, akathisia, fatigue, constipation, dyspepsia, sedation, weight increased, pain in extremity, and dry mouth. The most common adverse reactions in the double-blind, placebo-controlled periods of the bipolar disorder trials were weight increased (5% in the monotherapy trial) and tremor and parkinsonism (≥ 10% in the adjunctive treatment trial). The most common adverse reactions that were associated with discontinuation from the 12-week double-blind, placebo-controlled trial in patients with schizophrenia (causing discontinuation in ≥1% of patients) were agitation, depression, anxiety, and akathisia. Adverse reactions that were associated with discontinuation from the double-blind, placebo-controlled periods of the bipolar disorder trials were hyperglycemia (one patient in the monotherapy trial) and hypokinesia and tardive dyskinesia (one patient each in the adjunctive treatment trial). The data described in this section are derived from a clinical trial database consisting of 2392 patients exposed to one or more doses of RISPERDAL CONSTA for the treatment of schizophrenia. Of these 2392 patients, 332 were patients who received RISPERDAL CONSTA while participating in a 12-week double-blind, placebo-controlled trial. Two hundred two (202) of the 332 were schizophrenia patients who received 25 mg or 50 mg RISPERDAL CONSTA. The conditions and duration of treatment with RISPERDAL CONSTA in the other clinical trials varied greatly and included (in overlapping categories) double-blind, fixed- and flexible-dose, placebo- or active-controlled studies and open-label phases of studies, inpatients and outpatients, and short-term (up to 12 weeks) and longer-term (up to 4 years) exposures. Safety was assessed by collecting adverse events and performing physical examinations, vital signs, body weights, laboratory analyses, and ECGs. In addition to the studies in patients with schizophrenia, safety data are presented from a trial assessing the efficacy and safety of RISPERDAL CONSTA when administered as monotherapy for maintenance treatment in patients with bipolar I disorder. The subjects in this multi-center, double-blind, placebo-controlled study were adult patients who met DSM-IV criteria for Bipolar Disorder Type I and who were stable on risperidone (oral or long-acting injection), were stable on other antipsychotics or mood stabilizers, or were experiencing an acute episode. After a 3-week period of treatment with open-label oral risperidone (N=440), subjects who demonstrated an initial response to oral risperidone in this period and those who were stable on risperidone (oral or long-acting injection) at study entry entered into a 26-week stabilization period of open-label RISPERDAL CONSTA (N=501). Subjects who demonstrated a maintained response during this period were then randomized into a 24-month double-blind, placebo-controlled period in which they received RISPERDAL CONSTA (N=154) or placebo (N=149) as monotherapy. Subjects who relapsed or who completed the double-blind period could choose to enter an 8-week open-label RISPERDAL CONSTA extension period (N=160). Safety data are also presented from a trial assessing the efficacy and safety of RISPERDAL CONSTA when administered as adjunctive maintenance treatment in patients with bipolar disorder. The subjects in this multi-center, double-blind, placebo-controlled study were adult patients who met DSM-IV criteria for Bipolar Disorder Type I or Type II and who experienced at least 4 episodes of mood disorder requiring psychiatric/clinical intervention in the previous 12 months, including at least 2 episodes in the 6 months prior to the start of the study. At the start of this study, all patients (N=275) entered into a 16-week open-label treatment phase in which they received RISPERDAL CONSTA in addition to continuing their treatment as usual, which consisted of various mood stabilizers (primarily lithium and valproate), antidepressants, and/or anxiolytics. Patients who reached remission at the end of this 16-week open-label treatment phase (N=139) were then randomized into a 52-week double-blind, placebo-controlled phase in which they received RISPERDAL CONSTA (N=72) or placebo (n=67) as adjunctive treatment in addition to continuing their treatment as usual. Patients who did not reach remission at the end of the 16-week open-label treatment phase could choose to continue to receive RISPERDAL CONSTA as adjunctive therapy in an open-label manner, in addition to continuing their treatment as usual, for up to an additional 36 weeks as clinically indicated for a total period of up to 52 weeks; these patients (N=70) were also included in the evaluation of safety. Adverse events during exposure to study treatment were obtained by general inquiry and recorded by clinical investigators using their own terminology. Consequently, to provide a meaningful estimate of the proportion of individuals experiencing adverse events, events were grouped in standardized categories using MedDRA terminology. Throughout this section, adverse reactions are reported. Adverse reactions are adverse events that were considered to be reasonably associated with the use of RISPERDAL CONSTA (adverse drug reactions) based on the comprehensive assessment of the available adverse event information. A causal association for RISPERDAL CONSTA often cannot be reliably established in individual cases. Further, because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice. The majority of all adverse reactions were mild to moderate in severity.
Drug Interactions
The interactions of RISPERDAL CONSTA with coadministration of other drugs have not been systematically evaluated. The drug interaction data provided in this section is based on studies with oral RISPERDAL.
Storage and Handling
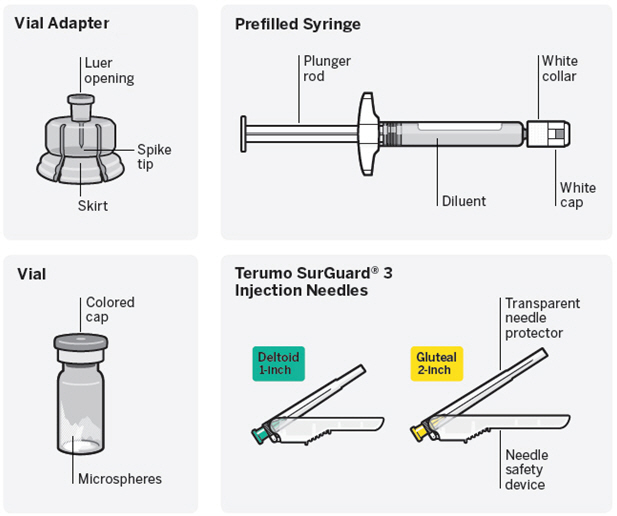
RISPERDAL CONSTA ® (risperidone) is available in dosage strengths of 12.5 mg, 25 mg, 37.5 mg, or 50 mg risperidone. It is provided as a single-use dose pack, consisting of a vial containing the risperidone microspheres, a pre-filled syringe containing 2 mL of diluent for RISPERDAL CONSTA, a vial adapter, and two Terumo SurGuard ® 3 Needles for intramuscular injection (a 21 G UTW 1-inch needle with needle protection device for deltoid administration and a 20 G TW 2-inch needle with needle protection device for gluteal administration). 12.5-mg vial/kit (NDC 50458-309-11): 41 mg (equivalent to 12.5 mg of risperidone) of a white to off-white powder provided in a vial with a violet flip-off cap (NDC 50458-309-01). 25-mg vial/kit (NDC 50458-306-11): 78 mg (equivalent to 25 mg of risperidone) of a white to off-white powder provided in a vial with a pink flip-off cap (NDC 50458-306-01). 37.5-mg vial/kit (NDC 50458-307-11): 116 mg (equivalent to 37.5 mg of risperidone) of a white to off-white powder provided in a vial with a green flip-off cap (NDC 50458-307-01). 50-mg vial/kit (NDC 50458-308-11): 152 mg (equivalent to 50 mg of risperidone) of a white to off-white powder provided in a vial with a blue flip-off cap (NDC 50458-308-01).
How Supplied
RISPERDAL CONSTA ® (risperidone) is available in dosage strengths of 12.5 mg, 25 mg, 37.5 mg, or 50 mg risperidone. It is provided as a single-use dose pack, consisting of a vial containing the risperidone microspheres, a pre-filled syringe containing 2 mL of diluent for RISPERDAL CONSTA, a vial adapter, and two Terumo SurGuard ® 3 Needles for intramuscular injection (a 21 G UTW 1-inch needle with needle protection device for deltoid administration and a 20 G TW 2-inch needle with needle protection device for gluteal administration). 12.5-mg vial/kit (NDC 50458-309-11): 41 mg (equivalent to 12.5 mg of risperidone) of a white to off-white powder provided in a vial with a violet flip-off cap (NDC 50458-309-01). 25-mg vial/kit (NDC 50458-306-11): 78 mg (equivalent to 25 mg of risperidone) of a white to off-white powder provided in a vial with a pink flip-off cap (NDC 50458-306-01). 37.5-mg vial/kit (NDC 50458-307-11): 116 mg (equivalent to 37.5 mg of risperidone) of a white to off-white powder provided in a vial with a green flip-off cap (NDC 50458-307-01). 50-mg vial/kit (NDC 50458-308-11): 152 mg (equivalent to 50 mg of risperidone) of a white to off-white powder provided in a vial with a blue flip-off cap (NDC 50458-308-01).
Medication Information
Warnings and Precautions
Cerebrovascular events, including stroke, in elderly patients with dementia-related psychosis. RISPERDAL CONSTA is not approved for use in patients with dementia-related psychosis ( 5.2 ) Neuroleptic Malignant Syndrome: Manage with immediate discontinuation and close monitoring ( 5.3 ) Tardive Dyskinesia: Discontinue treatment if clinically appropriate ( 5.4 ) Metabolic Changes: Atypical antipsychotic drugs have been associated with metabolic changes that may increase cardiovascular/ cerebrovascular risk. These metabolic changes include hyperglycemia, dyslipidemia, and weight gain. ( 5.5 ) Hyperglycemia and Diabetes Mellitus: Monitor patients for symptoms of hyperglycemia including polydipsia, polyuria, polyphagia, and weakness. Monitor glucose regularly in patients with diabetes or at risk for diabetes. ( 5.5 ) Dyslipidemia: Undesirable alterations have been observed in patients treated with atypical antipsychotics. ( 5.5 ) Weight Gain: Significant weight gain has been reported. Monitor weight gain. ( 5.5 ) Hyperprolactinemia: Risperidone treatment may elevate prolactin levels. Long-standing hyperprolactinemia, when associated with hypogonadism, can lead to decreased bone density in men and women. ( 5.6 ) Orthostatic hypotension: associated with dizziness, tachycardia, bradycardia, and syncope can occur, especially during initial dose titration with oral risperidone. Use caution in patients with cardiovascular disease, cerebrovascular disease, and conditions that could affect hemodynamic responses. ( 5.7 ) Leukopenia, Neutropenia, and Agranulocytosis have been reported with antipsychotics, including RISPERDAL CONSTA. Patients with history of a clinically significant low white blood cell count (WBC) or a drug-induced leukopenia/neutropenia should have their complete blood cell count (CBC) monitored frequently during the first few months of therapy and discontinuation of RISPERDAL CONSTA should be considered at the first sign of a clinically significant decline in WBC in the absence of other causative factors. ( 5.9 ) Potential for cognitive and motor impairment: has potential to impair judgment, thinking, and motor skills. Use caution when operating machinery, including automobiles. ( 5.10 ) Seizures: Use cautiously in patients with a history of seizures or with conditions that potentially lower the seizure threshold. ( 5.11 ) Dysphagia: Esophageal dysmotility and aspiration can occur. Use cautiously in patients at risk for aspiration pneumonia. ( 5.12 ) Priapism: has been reported. Severe priapism may require surgical intervention. ( 5.13 ) Avoid inadvertent administration into a blood vessel. ( 5.15 )
Indications and Usage
RISPERDAL CONSTA is an atypical antipsychotic indicated: for the treatment of schizophrenia. ( 1.1 ) as monotherapy or as adjunctive therapy to lithium or valproate for the maintenance treatment of Bipolar I Disorder. ( 1.2 )
Dosage and Administration
For patients who have never taken oral RISPERDAL, it is recommended to establish tolerability with oral RISPERDAL prior to initiating treatment with RISPERDAL CONSTA. RISPERDAL CONSTA should be administered every 2 weeks by deep intramuscular (IM) deltoid or gluteal injection. Each injection should be administered by a health care professional using the appropriate enclosed safety needle [see Dosage and Administration (2.8) ] . For deltoid administration, use the 1-inch needle alternating injections between the two arms. For gluteal administration, use the 2-inch needle alternating injections between the two buttocks. Do not administer intravenously.
Contraindications
RISPERDAL CONSTA is contraindicated in patients with a known hypersensitivity to either risperidone or paliperidone, or to any of the excipients in the RISPERDAL CONSTA formulation. Hypersensitivity reactions, including anaphylactic reactions and angioedema, have been reported in patients treated with risperidone and in patients treated with paliperidone. Paliperidone is a metabolite of risperidone.
Adverse Reactions
The following are discussed in more detail in other sections of the labeling: Increased mortality in elderly patients with dementia-related psychosis [see Boxed Warning and Warnings and Precautions (5.1) ] Cerebrovascular adverse events, including stroke, in elderly patients with dementia-related psychosis [see Warnings and Precautions (5.2) ] Neuroleptic malignant syndrome [see Warnings and Precautions (5.3) ] Tardive dyskinesia [see Warnings and Precautions (5.4) ] Metabolic changes [see Warnings and Precautions (5.5) ] Hyperprolactinemia [see Warnings and Precautions (5.6) ] Orthostatic hypotension [see Warnings and Precautions (5.7) ] Falls [see Warnings and Precautions (5.8) ] Leukopenia/Neutropenia and Agranulocytosis [see Warnings and Precautions (5.9) ] Potential for cognitive and motor impairment [see Warnings and Precautions (5.10) ] Seizures [see Warnings and Precautions (5.11) ] Dysphagia [see Warnings and Precautions (5.12) ] Priapism [see Warnings and Precautions (5.13) ] Disruption of body temperature regulation [see Warnings and Precautions (5.14) ] Avoidance of inadvertent injection into a blood vessel [see Warnings and Precautions (5.15) ] Osteodystrophy and tumors in animals [see Warnings and Precautions (5.16) ] The most common adverse reactions in clinical trials in patients with schizophrenia (≥ 5%) were: headache, parkinsonism, dizziness, akathisia, fatigue, constipation, dyspepsia, sedation, weight increased, pain in extremity, and dry mouth. The most common adverse reactions in the double-blind, placebo-controlled periods of the bipolar disorder trials were weight increased (5% in the monotherapy trial) and tremor and parkinsonism (≥ 10% in the adjunctive treatment trial). The most common adverse reactions that were associated with discontinuation from the 12-week double-blind, placebo-controlled trial in patients with schizophrenia (causing discontinuation in ≥1% of patients) were agitation, depression, anxiety, and akathisia. Adverse reactions that were associated with discontinuation from the double-blind, placebo-controlled periods of the bipolar disorder trials were hyperglycemia (one patient in the monotherapy trial) and hypokinesia and tardive dyskinesia (one patient each in the adjunctive treatment trial). The data described in this section are derived from a clinical trial database consisting of 2392 patients exposed to one or more doses of RISPERDAL CONSTA for the treatment of schizophrenia. Of these 2392 patients, 332 were patients who received RISPERDAL CONSTA while participating in a 12-week double-blind, placebo-controlled trial. Two hundred two (202) of the 332 were schizophrenia patients who received 25 mg or 50 mg RISPERDAL CONSTA. The conditions and duration of treatment with RISPERDAL CONSTA in the other clinical trials varied greatly and included (in overlapping categories) double-blind, fixed- and flexible-dose, placebo- or active-controlled studies and open-label phases of studies, inpatients and outpatients, and short-term (up to 12 weeks) and longer-term (up to 4 years) exposures. Safety was assessed by collecting adverse events and performing physical examinations, vital signs, body weights, laboratory analyses, and ECGs. In addition to the studies in patients with schizophrenia, safety data are presented from a trial assessing the efficacy and safety of RISPERDAL CONSTA when administered as monotherapy for maintenance treatment in patients with bipolar I disorder. The subjects in this multi-center, double-blind, placebo-controlled study were adult patients who met DSM-IV criteria for Bipolar Disorder Type I and who were stable on risperidone (oral or long-acting injection), were stable on other antipsychotics or mood stabilizers, or were experiencing an acute episode. After a 3-week period of treatment with open-label oral risperidone (N=440), subjects who demonstrated an initial response to oral risperidone in this period and those who were stable on risperidone (oral or long-acting injection) at study entry entered into a 26-week stabilization period of open-label RISPERDAL CONSTA (N=501). Subjects who demonstrated a maintained response during this period were then randomized into a 24-month double-blind, placebo-controlled period in which they received RISPERDAL CONSTA (N=154) or placebo (N=149) as monotherapy. Subjects who relapsed or who completed the double-blind period could choose to enter an 8-week open-label RISPERDAL CONSTA extension period (N=160). Safety data are also presented from a trial assessing the efficacy and safety of RISPERDAL CONSTA when administered as adjunctive maintenance treatment in patients with bipolar disorder. The subjects in this multi-center, double-blind, placebo-controlled study were adult patients who met DSM-IV criteria for Bipolar Disorder Type I or Type II and who experienced at least 4 episodes of mood disorder requiring psychiatric/clinical intervention in the previous 12 months, including at least 2 episodes in the 6 months prior to the start of the study. At the start of this study, all patients (N=275) entered into a 16-week open-label treatment phase in which they received RISPERDAL CONSTA in addition to continuing their treatment as usual, which consisted of various mood stabilizers (primarily lithium and valproate), antidepressants, and/or anxiolytics. Patients who reached remission at the end of this 16-week open-label treatment phase (N=139) were then randomized into a 52-week double-blind, placebo-controlled phase in which they received RISPERDAL CONSTA (N=72) or placebo (n=67) as adjunctive treatment in addition to continuing their treatment as usual. Patients who did not reach remission at the end of the 16-week open-label treatment phase could choose to continue to receive RISPERDAL CONSTA as adjunctive therapy in an open-label manner, in addition to continuing their treatment as usual, for up to an additional 36 weeks as clinically indicated for a total period of up to 52 weeks; these patients (N=70) were also included in the evaluation of safety. Adverse events during exposure to study treatment were obtained by general inquiry and recorded by clinical investigators using their own terminology. Consequently, to provide a meaningful estimate of the proportion of individuals experiencing adverse events, events were grouped in standardized categories using MedDRA terminology. Throughout this section, adverse reactions are reported. Adverse reactions are adverse events that were considered to be reasonably associated with the use of RISPERDAL CONSTA (adverse drug reactions) based on the comprehensive assessment of the available adverse event information. A causal association for RISPERDAL CONSTA often cannot be reliably established in individual cases. Further, because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice. The majority of all adverse reactions were mild to moderate in severity.
Drug Interactions
The interactions of RISPERDAL CONSTA with coadministration of other drugs have not been systematically evaluated. The drug interaction data provided in this section is based on studies with oral RISPERDAL.
Storage and Handling
RISPERDAL CONSTA ® (risperidone) is available in dosage strengths of 12.5 mg, 25 mg, 37.5 mg, or 50 mg risperidone. It is provided as a single-use dose pack, consisting of a vial containing the risperidone microspheres, a pre-filled syringe containing 2 mL of diluent for RISPERDAL CONSTA, a vial adapter, and two Terumo SurGuard ® 3 Needles for intramuscular injection (a 21 G UTW 1-inch needle with needle protection device for deltoid administration and a 20 G TW 2-inch needle with needle protection device for gluteal administration). 12.5-mg vial/kit (NDC 50458-309-11): 41 mg (equivalent to 12.5 mg of risperidone) of a white to off-white powder provided in a vial with a violet flip-off cap (NDC 50458-309-01). 25-mg vial/kit (NDC 50458-306-11): 78 mg (equivalent to 25 mg of risperidone) of a white to off-white powder provided in a vial with a pink flip-off cap (NDC 50458-306-01). 37.5-mg vial/kit (NDC 50458-307-11): 116 mg (equivalent to 37.5 mg of risperidone) of a white to off-white powder provided in a vial with a green flip-off cap (NDC 50458-307-01). 50-mg vial/kit (NDC 50458-308-11): 152 mg (equivalent to 50 mg of risperidone) of a white to off-white powder provided in a vial with a blue flip-off cap (NDC 50458-308-01).
How Supplied
RISPERDAL CONSTA ® (risperidone) is available in dosage strengths of 12.5 mg, 25 mg, 37.5 mg, or 50 mg risperidone. It is provided as a single-use dose pack, consisting of a vial containing the risperidone microspheres, a pre-filled syringe containing 2 mL of diluent for RISPERDAL CONSTA, a vial adapter, and two Terumo SurGuard ® 3 Needles for intramuscular injection (a 21 G UTW 1-inch needle with needle protection device for deltoid administration and a 20 G TW 2-inch needle with needle protection device for gluteal administration). 12.5-mg vial/kit (NDC 50458-309-11): 41 mg (equivalent to 12.5 mg of risperidone) of a white to off-white powder provided in a vial with a violet flip-off cap (NDC 50458-309-01). 25-mg vial/kit (NDC 50458-306-11): 78 mg (equivalent to 25 mg of risperidone) of a white to off-white powder provided in a vial with a pink flip-off cap (NDC 50458-306-01). 37.5-mg vial/kit (NDC 50458-307-11): 116 mg (equivalent to 37.5 mg of risperidone) of a white to off-white powder provided in a vial with a green flip-off cap (NDC 50458-307-01). 50-mg vial/kit (NDC 50458-308-11): 152 mg (equivalent to 50 mg of risperidone) of a white to off-white powder provided in a vial with a blue flip-off cap (NDC 50458-308-01).
Description
Elderly patients with dementia-related psychosis treated with antipsychotic drugs are at an increased risk of death. RISPERDAL CONSTA is not approved for the treatment of patients with dementia-related psychosis. [see Warnings and Precautions (5.1) ]
Section 42229-5
Elderly
For elderly patients treated with RISPERDAL CONSTA, the recommended dosage is 25 mg IM every 2 weeks. Oral RISPERDAL (or another antipsychotic medication) should be given with the first injection of RISPERDAL CONSTA and should be continued for 3 weeks to ensure that adequate therapeutic plasma concentrations are maintained prior to the main release phase of risperidone from the injection site [see Clinical Pharmacology (12.3)] .
Section 43683-2
| Warnings and Precautions ( 5.6) | 1/2025 |
Section 44425-7
Storage and Handling
The entire dose pack should be stored in the refrigerator (36 °F to 46 °F; 2 °C to 8 °C) and protected from light.
If refrigeration is unavailable, RISPERDAL CONSTA can be stored at temperatures not exceeding 77 °F (25 °C) for no more than 7 days prior to administration. Do not expose unrefrigerated product to temperatures above 77 °F (25 °C).
Keep out of reach of children.
5.8 Falls
Somnolence, postural hypotension, motor and sensory instability have been reported with the use of antipsychotics, including RISPERDAL CONSTA, which may lead to falls and, consequently, fractures or other fall-related injuries. For patients, particularly the elderly, with diseases, conditions, or medications that could exacerbate these effects, assess the risk of falls when initiating antipsychotic treatment and recurrently for patients on long-term antipsychotic therapy.
9.2 Abuse
RISPERDAL CONSTA has not been systematically studied in animals or humans for its potential for abuse. Because RISPERDAL CONSTA is to be administered by health care professionals, the potential for misuse or abuse by patients is low.
7.8 Lithium
Repeated doses of oral RISPERDAL (3 mg twice daily) did not affect the exposure (AUC) or peak plasma concentrations (C max) of lithium (N=13).
7.10 Digoxin
Oral RISPERDAL (0.25 mg twice daily) did not show a clinically relevant effect on the pharmacokinetics of digoxin.
5.11 Seizures
During premarketing testing, seizures occurred in 0.3% (5/1499 patients) of patients treated with RISPERDAL CONSTA. Therefore, RISPERDAL CONSTA should be used cautiously in patients with a history of seizures.
5.13 Priapism
Priapism has been reported during postmarketing surveillance [see Adverse Reactions (6.2)] . Severe priapism may require surgical intervention.
7.7 Clozapine
Chronic administration of clozapine with risperidone may decrease the clearance of risperidone.
7.9 Valproate
Repeated doses of oral RISPERDAL (4 mg once daily) did not affect the pre-dose or average plasma concentrations and exposure (AUC) of valproate (1000 mg/day in three divided doses) compared to placebo (N=21). However, there was a 20% increase in valproate peak plasma concentration (C max) after concomitant administration of oral RISPERDAL.
11 Description
RISPERDAL CONSTA ®contains risperidone, an atypical antipsychotic belonging to the chemical class of benzisoxazole derivatives. The chemical designation is 3-[2-[4-(6-fluoro-1,2-benzisoxazol-3-yl)-1-piperidinyl]ethyl]-6,7,8,9-tetrahydro-2-methyl-4H-pyrido[1,2-a]pyrimidin-4-one. Its molecular formula is C 23 H 27 FN 4O 2 and its molecular weight is 410.49. The structural formula is:
Risperidone is practically insoluble in water, freely soluble in methylene chloride, and soluble in methanol and 0.1 NHCl.
RISPERDAL CONSTA (risperidone) Long-Acting Injection is a combination of extended-release microspheres for injection and diluent for parenteral use.
The extended-release microspheres formulation is a white to off-white, free-flowing powder that is available in dosage strengths of 12.5 mg, 25 mg, 37.5 mg, or 50 mg risperidone per vial. Risperidone is micro-encapsulated in 7525 polylactide-co-glycolide (PLG) at a concentration of 381 mg risperidone per gram of microspheres.
The diluent for parenteral use is a clear, colorless solution. Composition of the diluent includes 1 mg/mL citric acid anhydrous, 1.27 mg/mL disodium hydrogen phosphate dihydrate, 1 mg/mL polysorbate 20, 22.5 mg/mL sodium carboxymethyl cellulose, 6 mg/mL sodium chloride, 0.54 mg/mL sodium hydroxide, and water for injection. The microspheres are suspended in the diluent prior to injection.
RISPERDAL CONSTA is provided as a single-use dose pack, consisting of a vial containing the microspheres, a pre-filled syringe containing the diluent, a vial adapter, and two Terumo SurGuard 3 Needles (a 21 G UTW 1-inch needle with needle protection device for deltoid administration and a 20 G TW 2-inch needle with needle protection device for gluteal administration).
5.12 Dysphagia
Esophageal dysmotility and aspiration have been associated with antipsychotic drug use. Aspiration pneumonia is a common cause of morbidity and mortality in patients with advanced Alzheimer's dementia. RISPERDAL CONSTA and other antipsychotic drugs should be used cautiously in patients at risk for aspiration pneumonia. [see also Boxed Warning and Warnings and Precautions (5.1)]
9.3 Dependence
RISPERDAL CONSTA has not been systematically studied in animals or humans for its potential for tolerance or physical dependence.
7.11 Topiramate
Oral RISPERDAL administered at doses from 1–6 mg/day concomitantly with topiramate 400 mg/day resulted in a 23% decrease in risperidone C max and a 33% decrease in risperidone AUC 0–12 hour at steady state. Minimal reductions in the exposure to risperidone and 9-hydroxyrisperidone combined, and no change for 9-hydroxyrisperidone were observed. This interaction is unlikely to be of clinical significance. There was no clinically relevant effect of oral RISPERDAL on the pharmacokinetics of topiramate.
1.1 Schizophrenia
RISPERDAL CONSTA (risperidone) is indicated for the treatment of schizophrenia [see Clinical Studies (14.1)].
2.1 Schizophrenia
The recommended dose for the treatment of schizophrenia is 25 mg IM every 2 weeks. Although dose response for effectiveness has not been established for RISPERDAL CONSTA, some patients not responding to 25 mg may benefit from a higher dose of 37.5 mg or 50 mg. The maximum dose should not exceed 50 mg RISPERDAL CONSTA every 2 weeks. No additional benefit was observed with dosages greater than 50 mg RISPERDAL CONSTA; however, a higher incidence of adverse effects was observed.
The efficacy of RISPERDAL CONSTA in the treatment of schizophrenia has not been evaluated in controlled clinical trials for longer than 12 weeks. Although controlled studies have not been conducted to answer the question of how long patients with schizophrenia should be treated with RISPERDAL CONSTA, oral risperidone has been shown to be effective in delaying time to relapse in longer-term use. It is recommended that responding patients be continued on treatment with RISPERDAL CONSTA at the lowest dose needed. The physician who elects to use RISPERDAL CONSTA for extended periods should periodically re-evaluate the long-term risks and benefits of the drug for the individual patient.
7.4 Amitriptyline
Amitriptyline did not affect the pharmacokinetics of risperidone or of risperidone and 9-hydroxyrisperidone combined following concomitant administration with oral RISPERDAL.
8.4 Pediatric Use
Safety and effectiveness of RISPERDAL CONSTA in pediatric patients have not been established. However, juvenile animal toxicology studies have been conducted with oral risperidone.
8.5 Geriatric Use
In an open-label study, 57 clinically stable, elderly patients (≥ 65 years old) with schizophrenia or schizoaffective disorder received RISPERDAL CONSTA every 2 weeks for up to 12 months. In general, no differences in the tolerability of RISPERDAL CONSTA were observed between otherwise healthy elderly and nonelderly patients. Therefore, dosing recommendations for otherwise healthy elderly patients are the same as for nonelderly patients. Because elderly patients exhibit a greater tendency to orthostatic hypotension than nonelderly patients, elderly patients should be instructed in nonpharmacologic interventions that help to reduce the occurrence of orthostatic hypotension (e.g., sitting on the edge of the bed for several minutes before attempting to stand in the morning and slowly rising from a seated position). In addition, monitoring of orthostatic vital signs should be considered in elderly patients for whom orthostatic hypotension is of concern [see Warnings and Precautions (5.7)] .
14.1 Schizophrenia
The effectiveness of RISPERDAL CONSTA in the treatment of schizophrenia was established, in part, on the basis of extrapolation from the established effectiveness of the oral formulation of risperidone. In addition, the effectiveness of RISPERDAL CONSTA in the treatment of schizophrenia was established in a 12-week, placebo-controlled trial in adult psychotic inpatients and outpatients who met the DSM-IV criteria for schizophrenia.
Efficacy data were obtained from 400 patients with schizophrenia who were randomized to receive injections of 25 mg, 50 mg, or 75 mg RISPERDAL CONSTA or placebo every 2 weeks. During a 1-week run-in period, patients were discontinued from other antipsychotics and were titrated to a dose of 4 mg oral RISPERDAL. Patients who received RISPERDAL CONSTA were given doses of oral RISPERDAL (2 mg for patients in the 25-mg group, 4 mg for patients in the 50-mg group, and 6 mg for patients in the 75-mg group) for the 3 weeks after the first injection to provide therapeutic plasma concentrations until the main release phase of risperidone from the injection site had begun. Patients who received placebo injections were given placebo tablets.
Efficacy was evaluated using the Positive and Negative Syndrome Scale (PANSS), a validated, multi-item inventory, composed of five subscales to evaluate positive symptoms, negative symptoms, disorganized thoughts, uncontrolled hostility/excitement, and anxiety/depression.
The primary efficacy variable in this trial was change from baseline to endpoint in the total PANSS score. The mean total PANSS score at baseline for schizophrenic patients in this study was 81.5.
Total PANSS scores showed significant improvement in the change from baseline to endpoint in schizophrenic patients treated with each dose of RISPERDAL CONSTA (25 mg, 50 mg, or 75 mg) compared with patients treated with placebo. While there were no statistically significant differences between the treatment effects for the three dose groups, the effect size for the 75 mg dose group was actually numerically less than that observed for the 50 mg dose group.
Subgroup analyses did not indicate any differences in treatment outcome as a function of age, race, or gender.
4 Contraindications
RISPERDAL CONSTA is contraindicated in patients with a known hypersensitivity to either risperidone or paliperidone, or to any of the excipients in the RISPERDAL CONSTA formulation. Hypersensitivity reactions, including anaphylactic reactions and angioedema, have been reported in patients treated with risperidone and in patients treated with paliperidone. Paliperidone is a metabolite of risperidone.
5.15 Administration
RISPERDAL CONSTA should be injected into the deltoid or gluteal muscle, and care must be taken to avoid inadvertent injection into a blood vessel. [see Dosage and Administration (2) and Adverse Reactions (6)] .
6 Adverse Reactions
The following are discussed in more detail in other sections of the labeling:
- Increased mortality in elderly patients with dementia-related psychosis [see Boxed Warningand Warnings and Precautions (5.1)]
- Cerebrovascular adverse events, including stroke, in elderly patients with dementia-related psychosis [see Warnings and Precautions (5.2)]
- Neuroleptic malignant syndrome [see Warnings and Precautions (5.3)]
- Tardive dyskinesia [see Warnings and Precautions (5.4)]
- Metabolic changes [see Warnings and Precautions (5.5)]
- Hyperprolactinemia [see Warnings and Precautions (5.6)]
- Orthostatic hypotension [see Warnings and Precautions (5.7)]
- Falls [see Warnings and Precautions (5.8)]
- Leukopenia/Neutropenia and Agranulocytosis [see Warnings and Precautions (5.9)]
- Potential for cognitive and motor impairment [see Warnings and Precautions (5.10)]
- Seizures [see Warnings and Precautions (5.11)]
- Dysphagia [see Warnings and Precautions (5.12)]
- Priapism [see Warnings and Precautions (5.13)]
- Disruption of body temperature regulation [see Warnings and Precautions (5.14)]
- Avoidance of inadvertent injection into a blood vessel [see Warnings and Precautions (5.15)]
- Osteodystrophy and tumors in animals [see Warnings and Precautions (5.16)]
The most common adverse reactions in clinical trials in patients with schizophrenia (≥ 5%) were: headache, parkinsonism, dizziness, akathisia, fatigue, constipation, dyspepsia, sedation, weight increased, pain in extremity, and dry mouth. The most common adverse reactions in the double-blind, placebo-controlled periods of the bipolar disorder trials were weight increased (5% in the monotherapy trial) and tremor and parkinsonism (≥ 10% in the adjunctive treatment trial).
The most common adverse reactions that were associated with discontinuation from the 12-week double-blind, placebo-controlled trial in patients with schizophrenia (causing discontinuation in ≥1% of patients) were agitation, depression, anxiety, and akathisia. Adverse reactions that were associated with discontinuation from the double-blind, placebo-controlled periods of the bipolar disorder trials were hyperglycemia (one patient in the monotherapy trial) and hypokinesia and tardive dyskinesia (one patient each in the adjunctive treatment trial).
The data described in this section are derived from a clinical trial database consisting of 2392 patients exposed to one or more doses of RISPERDAL CONSTA for the treatment of schizophrenia. Of these 2392 patients, 332 were patients who received RISPERDAL CONSTA while participating in a 12-week double-blind, placebo-controlled trial. Two hundred two (202) of the 332 were schizophrenia patients who received 25 mg or 50 mg RISPERDAL CONSTA. The conditions and duration of treatment with RISPERDAL CONSTA in the other clinical trials varied greatly and included (in overlapping categories) double-blind, fixed- and flexible-dose, placebo- or active-controlled studies and open-label phases of studies, inpatients and outpatients, and short-term (up to 12 weeks) and longer-term (up to 4 years) exposures. Safety was assessed by collecting adverse events and performing physical examinations, vital signs, body weights, laboratory analyses, and ECGs.
In addition to the studies in patients with schizophrenia, safety data are presented from a trial assessing the efficacy and safety of RISPERDAL CONSTA when administered as monotherapy for maintenance treatment in patients with bipolar I disorder. The subjects in this multi-center, double-blind, placebo-controlled study were adult patients who met DSM-IV criteria for Bipolar Disorder Type I and who were stable on risperidone (oral or long-acting injection), were stable on other antipsychotics or mood stabilizers, or were experiencing an acute episode. After a 3-week period of treatment with open-label oral risperidone (N=440), subjects who demonstrated an initial response to oral risperidone in this period and those who were stable on risperidone (oral or long-acting injection) at study entry entered into a 26-week stabilization period of open-label RISPERDAL CONSTA (N=501). Subjects who demonstrated a maintained response during this period were then randomized into a 24-month double-blind, placebo-controlled period in which they received RISPERDAL CONSTA (N=154) or placebo (N=149) as monotherapy. Subjects who relapsed or who completed the double-blind period could choose to enter an 8-week open-label RISPERDAL CONSTA extension period (N=160).
Safety data are also presented from a trial assessing the efficacy and safety of RISPERDAL CONSTA when administered as adjunctive maintenance treatment in patients with bipolar disorder. The subjects in this multi-center, double-blind, placebo-controlled study were adult patients who met DSM-IV criteria for Bipolar Disorder Type I or Type II and who experienced at least 4 episodes of mood disorder requiring psychiatric/clinical intervention in the previous 12 months, including at least 2 episodes in the 6 months prior to the start of the study. At the start of this study, all patients (N=275) entered into a 16-week open-label treatment phase in which they received RISPERDAL CONSTA in addition to continuing their treatment as usual, which consisted of various mood stabilizers (primarily lithium and valproate), antidepressants, and/or anxiolytics. Patients who reached remission at the end of this 16-week open-label treatment phase (N=139) were then randomized into a 52-week double-blind, placebo-controlled phase in which they received RISPERDAL CONSTA (N=72) or placebo (n=67) as adjunctive treatment in addition to continuing their treatment as usual. Patients who did not reach remission at the end of the 16-week open-label treatment phase could choose to continue to receive RISPERDAL CONSTA as adjunctive therapy in an open-label manner, in addition to continuing their treatment as usual, for up to an additional 36 weeks as clinically indicated for a total period of up to 52 weeks; these patients (N=70) were also included in the evaluation of safety.
Adverse events during exposure to study treatment were obtained by general inquiry and recorded by clinical investigators using their own terminology. Consequently, to provide a meaningful estimate of the proportion of individuals experiencing adverse events, events were grouped in standardized categories using MedDRA terminology.
Throughout this section, adverse reactions are reported. Adverse reactions are adverse events that were considered to be reasonably associated with the use of RISPERDAL CONSTA (adverse drug reactions) based on the comprehensive assessment of the available adverse event information. A causal association for RISPERDAL CONSTA often cannot be reliably established in individual cases. Further, because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
The majority of all adverse reactions were mild to moderate in severity.
7 Drug Interactions
The interactions of RISPERDAL CONSTA with coadministration of other drugs have not been systematically evaluated. The drug interaction data provided in this section is based on studies with oral RISPERDAL.
7.6 Methylphenidate
Concomitant use with methylphenidate, when there is change in dosage of either medication, may increase the risk of extrapyramidal symptoms (EPS). Monitor for symptoms of EPS with concomitant use of RISPERDAL CONSTA and methylphenidate [see Adverse Reactions (6.2)] .
1.2 Bipolar Disorder
RISPERDAL CONSTA is indicated as monotherapy or as adjunctive therapy to lithium or valproate for the maintenance treatment of Bipolar I Disorder [see Clinical Studies (14.2 , 14.3)] .
2.2 Bipolar Disorder
The recommended dose for monotherapy or adjunctive therapy to lithium or valproate for the maintenance treatment of Bipolar I Disorder is 25 mg IM every 2 weeks. Some patients may benefit from a higher dose of 37.5 mg or 50 mg. Dosages above 50 mg have not been studied in this population. The physician who elects to use RISPERDAL CONSTA for extended periods should periodically re-evaluate the long-term risks and benefits of the drug for the individual patient.
Instructions for Use
RISPERDAL CONSTA
®
(risperidone)
Long-Acting Injection
12.5 mg, 25 mg, 37.5 mg, 50 mg
For deltoid or gluteal intramuscular injection only
Each injection should be administered by a healthcare professional.
IMPORTANT RESOURCES
For additional information, visit www.risperdalconsta.com or call Janssen Pharmaceuticals, Inc. at 1-800-526-7736.
Manufactured for:
Janssen Pharmaceuticals, Inc.
Titusville, NJ 08560, USA
For patent information: www.janssenpatents.com
© Johnson & Johnson and its affiliates 2007
Revised: 4/2025
Instructions for use
RISPERDAL CONSTA ® requires close attention to these step-by-step Instructions for Use to help ensure successful administration.
SINGLE-USE DEVICE
Do not reuse.Medical devices require specific material characteristics to perform as intended. These characteristics have been verified for single use only. Any attempt to re-process the device for subsequent re-use may adversely affect the integrity of the device or lead to deterioration in performance.
Important Information
Use components provided
The components in this dose pack are specifically designed for use with RISPERDAL CONSTA ®. RISPERDAL CONSTA ® must be reconstituted only in the diluent supplied in the dose pack.
Do not substitute ANY components of the dose pack.
Do not store suspension after reconstitution
Administer dose as soon as possible after reconstitution to avoid settling.
Proper dosing
The entire contents of the vial must be administered to ensure intended dose of RISPERDAL CONSTA ®is delivered.
Dose pack contents
| Step 1 Assemble components | |||
|---|---|---|---|
| Take out dose pack | Connect vial adapter to vial | ||
|
|
|
|
|
|
Wait 30 minutes
Remove dose pack from the refrigerator and allow to sit at room temperature for at least 30 minutes before reconstituting. Do not warm any other way. |
Remove cap from vial
Flip off colored cap from vial. Wipe top of the grey stopper with an alcohol swab. Allow to air dry. Do not remove grey rubber stopper. |
Prepare vial adapter
Hold sterile blister as shown. Peel back and remove paper backing. Do not remove vial adapter from blister. Do not touch spike tip at any time. This will result in contamination. |
Connect vial adapter to vial
Place vial on a hard surface and hold by the base. Center vial adapter over the grey rubber stopper. Push vial adapter straight down onto vial top until it snaps securely into place. Do not place vial adapter on at an angle or diluent may leak upon transfer to the vial.
|
| Connect prefilled syringe to vial adapter | |||
|
|
|
|
|
|
Remove sterile blister
Keep vial vertical to prevent leakage. Hold base of vial and pull up on the sterile blister to remove. Do not shake. Do not touch exposed luer opening on vial adapter. This will result in contamination. |
Use proper grip
Hold by white collar at the tip of the syringe. Do not hold syringe by the glass barrel during assembly.
|
Remove cap
Holding the white collar, snap off the white cap. Do not twist or cut off the white cap. Do not touch syringe tip. This will result in contamination. The broken-off cap can be discarded. |
Connect syringe to vial adapter
Hold vial adapter by skirt to keep stationary. Hold syringe by white collarthen insert tip into the luer opening of the vial adapter. Do not hold the glass syringe barrel. This may cause the white collar to loosen or detach. Attach the syringe to the vial adapter with a firm clockwise twisting motion until it feels snug. Do not over-tighten. Over-tightening may cause the syringe tip to break. |
| Step 2 Reconstitute microspheres | |||
|---|---|---|---|
|
|
|
|
|
|
Inject diluent
Inject entire amount of diluent from syringe into the vial.
|
Suspend microspheres in diluent
Continuing to hold down the plunger rod, shake vigorously for at least 10 seconds, as shown. Check the suspension. When properly mixed, the suspension appears uniform, thick and milky in color. Microspheres will be visible in the liquid. Immediately proceed to the next step so suspension does not settle. |
Transfer suspension to syringe
Invert vial completely. Slowly pull plunger rod down to withdraw entire contents from the vial into the syringe. |
Remove vial adapter
Hold white collar on the syringe and unscrew from vial adapter. Tear section of the vial label at the perforation. Apply detached label to the syringe for identification purposes. Discard both vial and vial adapter appropriately. |
| Step 3 Attach needle | ||
|---|---|---|
|
|
|
|
|
Select appropriate needle
Choose needle based on injection location (gluteal or deltoid). |
Attach needle
Peel blister pouch open part way and use to grasp the base of the needle, as shown. Holding the white collar on the syringe, attach syringe to needle luer connection with a firm clockwise twisting motion until snug. Do not touch needle luer opening. This will result in contamination. |
Resuspend microspheres
Fully remove the blister pouch. Just before injection, shake syringe vigorously again, as some settling will have occurred. |
| Step 4 Inject dose | ||||
|---|---|---|---|---|
|
|
|
|
|
|
|
Remove transparent needle protector
Move the needle safety device back towards the syringe, as shown. Then hold white collar on syringe and carefully pull the transparent needle protector straight off. Do not twist transparent needle protector, as the luer connection may loosen. |
Remove air bubbles
Hold needle upright and tap gently to make any air bubbles rise to the top. Slowly and carefully press plunger rod upward to remove air. |
Inject
Immediately inject entire contents of syringe intramuscularly (IM) into the gluteal or deltoid muscle of the patient. Gluteal injection should be made into the upper-outer quadrant of the gluteal area. Do not administer intravenously. |
Secure needle in safety device
Using one hand, place needle safety device at a 45-degree angle on a hard, flat surface. Press down with a firm, quick motion until needle is fully engaged in safety device. Avoid needle stick injury: Do not use two hands. Do not intentionally disengage or mishandle the needle safety device. Do not attempt to straighten the needle or engage the safety device if the needle is bent or damaged. |
Properly dispose of needles
Check to confirm needle safety device is fully engaged. Discard in an approved sharps container. Also discard the unused needle provided in the dose pack. |
10.1 Human Experience
No cases of overdose were reported in premarketing studies with RISPERDAL CONSTA. Because RISPERDAL CONSTA is to be administered by health care professionals, the potential for overdosage by patients is low.
In premarketing experience with oral RISPERDAL, there were eight reports of acute RISPERDAL overdosage, with estimated doses ranging from 20 to 300 mg and no fatalities. In general, reported signs and symptoms were those resulting from an exaggeration of the drug's known pharmacological effects, i.e., drowsiness and sedation, tachycardia and hypotension, and extrapyramidal symptoms. One case, involving an estimated overdose of 240 mg, was associated with hyponatremia, hypokalemia, prolonged QT, and widened QRS. Another case, involving an estimated overdose of 36 mg, was associated with a seizure.
Postmarketing experience with oral RISPERDAL includes reports of acute overdose, with estimated doses of up to 360 mg. In general, the most frequently reported signs and symptoms are those resulting from an exaggeration of the drug's known pharmacological effects, i.e., drowsiness, sedation, tachycardia, hypotension, and extrapyramidal symptoms. Other adverse reactions reported since market introduction related to oral RISPERDAL overdose include prolonged QT interval and convulsions. Torsade de pointes has been reported in association with combined overdose of oral RISPERDAL and paroxetine.
12.2 Pharmacodynamics
Risperidone is a monoaminergic antagonist with high affinity (Ki of 0.12 to 7.3 nM) for the serotonin Type 2 (5HT 2), dopamine Type 2 (D 2), α1 and α2 adrenergic, and H 1 histaminergic receptors. Risperidone showed low to moderate affinity (Ki of 47 to 253 nM) for the serotonin 5HT 1C, 5HT 1D, and 5HT 1A receptors, weak affinity (Ki of 620 to 800 nM) for the dopamine D 1 and haloperidol-sensitive sigma site, and no affinity (when tested at concentrations >10 -5 M) for cholinergic muscarinic or β1 and β2 adrenergic receptors.
5.5 Metabolic Changes
Atypical antipsychotic drugs have been associated with metabolic changes that may increase cardiovascular/cerebrovascular risk. These metabolic changes include hyperglycemia, dyslipidemia, and body weight gain. While all of the drugs in the class have been shown to produce some metabolic changes, each drug has its own specific risk profile.
5.4 Tardive Dyskinesia
Tardive dyskinesia, a syndrome consisting of potentially irreversible, involuntary, dyskinetic movements, may develop in patients treated with antipsychotic drugs. Although the prevalence of the syndrome appears to be highest among the elderly, especially elderly women, it is impossible to predict which patients will develop the syndrome. Whether antipsychotic drug products differ in their potential to cause tardive dyskinesia is unknown.
The risk of developing tardive dyskinesia and the likelihood that it will become irreversible increase with the duration of treatment and the cumulative dose. The syndrome can develop after relatively brief treatment periods, even at low doses. It may also occur after discontinuation of treatment.
Tardive dyskinesia may remit, partially or completely, if antipsychotic treatment is discontinued. Antipsychotic treatment, itself, however, may suppress (or partially suppress) the signs and symptoms of the syndrome, possibly masking the underlying process. The effect that symptomatic suppression has upon the long-term course of the syndrome is unknown.
Given these considerations, RISPERDAL CONSTA should be prescribed in a manner that is most likely to minimize the occurrence of tardive dyskinesia. Chronic antipsychotic treatment should generally be reserved for patients: (1) who suffer from a chronic illness that is known to respond to antipsychotic drugs, and (2) for whom alternative, equally effective, but potentially less harmful treatments are not available or appropriate. In patients who do require chronic treatment, the lowest dose and the shortest duration of treatment producing a satisfactory clinical response. Periodically reassess the need for continued treatment.
If signs and symptoms of tardive dyskinesia appear in a patient treated with RISPERDAL CONSTA, drug discontinuation should be considered. However, some patients may require treatment with RISPERDAL CONSTA despite the presence of the syndrome.
5.6 Hyperprolactinemia
As with other drugs that antagonize dopamine D 2receptors, risperidone elevates prolactin levels and the elevation persists during chronic administration. Risperidone is associated with higher levels of prolactin elevation than other antipsychotic agents.
Hyperprolactinemia may suppress hypothalamic GnRH, resulting in reduced pituitary gonadotropin secretion. This, in turn, may inhibit reproductive function by impairing gonadal steroidogenesis in both female and male patients. Galactorrhea, amenorrhea, gynecomastia, and impotence have been reported in patients receiving prolactin-elevating compounds. Long-standing hyperprolactinemia when associated with hypogonadism may lead to decreased bone density in both female and male subjects.
Tissue culture experiments indicate that approximately one-third of human breast cancers are prolactin dependent in vitro, a factor of potential importance if the prescription of these drugs is contemplated in a patient with previously detected breast cancer. An increase in pituitary gland, mammary gland, and pancreatic islet cell neoplasia (mammary adenocarcinomas, pituitary and pancreatic adenomas) was observed in the risperidone carcinogenicity studies conducted in mice and rats [see Nonclinical Toxicology (13.1)] . Published epidemiologic studies have shown inconsistent results when exploring the potential association between hyperprolactinemia and breast cancer.
1 Indications and Usage
12.1 Mechanism of Action
The mechanism of action of risperidone in schizophrenia is unclear. The drug's therapeutic activity in schizophrenia could be mediated through a combination of dopamine Type 2 (D 2) and serotonin Type 2 (5HT 2) receptor antagonism. The clinical effect from risperidone results from the combined concentrations of risperidone and its major active metabolite, 9-hydroxyrisperidone (paliperidone) [see Clinical Pharmacology (12.3)] . Antagonism at receptors other than D 2 and 5HT 2 may explain some of the other effects of risperidone [see Clinical Pharmacology (12.1)] .
2.8 Instructions for Use
For deltoid or gluteal intramuscular injection only
IMPORTANT RESOURCES
For additional information, visit www.risperdalconsta.com or call Janssen Pharmaceuticals, Inc. at 1-800-526-7736.
Important Information
RISPERDAL CONSTA requires close attention to these step-by-step Instructions for Use to help ensure successful administration.
Use components provided
The components in this dose pack are specifically designed for use with RISPERDAL CONSTA. RISPERDAL CONSTA must be reconstituted only in the diluent supplied in the dose pack.
Do notsubstitute ANY components of the dose pack.
Do not store suspension after reconstitution
Administer dose as soon as possible after reconstitution to avoid settling.
Proper dosing
The entire contents of the vial must be administered to ensure intended dose of RISPERDAL CONSTA is delivered.
SINGLE-USE DEVICE
Do not reuse.Medical devices require specific material characteristics to perform as intended. These characteristics have been verified for single use only. Any attempt to re-process the device for subsequent re-use may adversely affect the integrity of the device or lead to deterioration in performance.
Dose pack contents
| Step 1 Assemble components | |||
|---|---|---|---|
| Take out dose pack Connect vial adapter to vial | |||
|
|
|
|
|
|
Wait 30 minutes
Remove dose pack from the refrigerator and allow to sit at room temperature for at least 30 minutesbefore reconstituting. Do notwarm any other way. |
Remove cap from vial
Flip off colored cap from vial. Wipe top of the grey stopper with an alcohol swab. Allow to air dry. Do notremove grey rubber stopper. |
Prepare vial adapter
Hold sterile blister as shown. Peel back and remove paper backing. Do notremove vial adapter from blister. Do nottouch spike tip at any time. This will result in contamination. |
Connect vial adapter to vial
Place vial on a hard surface and hold by the base. Center vial adapter over the grey rubber stopper. Push vial adapter straight down onto vial top until it snaps securely into place. Do notplace vial adapter on at an angle or diluent may leak upon transfer to the vial.
|
| Connect prefilled syringe to vial adapter | |||
|
|
|
|
|
|
Remove sterile blister
Keep vial vertical to prevent leakage. Hold base of vial and pull up on the sterile blister to remove. Do notshake. Do nottouch exposed luer opening on vial adapter. This will result in contamination. |
Use proper grip
Hold by white collar at the tip of the syringe. Do nothold syringe by the glass barrel during assembly.
|
Remove cap
Holding the white collar, snap off the white cap. Do nottwist or cut off the white cap. Do nottouch syringe tip. This will result in contamination. The broken-off cap can be discarded. |
Connect syringe to vial adapter
Hold vial adapter by skirt to keep stationary. Hold syringe by white collarthen insert tip into the luer opening of the vial adapter. Do nothold the glass syringe barrel. This may cause the white collar to loosen or detach. Attach the syringe to the vial adapter with a firm clockwise twisting motion until it feels snug. Do notover-tighten. Over-tightening may cause the syringe tip to break. |
| Step 2 Reconstitute microspheres | |||
|---|---|---|---|
|
|
|
|
|
|
Inject diluent
Inject entire amount of diluent from syringe into the vial.
|
Suspend microspheres in diluent
Continuing to hold down the plunger rod, shake vigorously for at least 10 seconds, as shown. Check the suspension. When properly mixed, the suspension appears uniform, thick and milky in color. Microspheres will be visible in the liquid. Immediately proceed to the next step so suspension does not settle. |
Transfer suspension to syringe
Invert vial completely. Slowly pull plunger rod down to withdraw entire contents from the vial into the syringe. |
Remove vial adapter
Hold white collar on the syringe and unscrew from vial adapter. Tear section of the vial label at the perforation. Apply detached label to the syringe for identification purposes. Discard both vial and vial adapter appropriately. |
| Step 3 Attach needle | ||
|---|---|---|
|
|
|
|
|
Select appropriate needle
Choose needle based on injection location (gluteal or deltoid). |
Attach needle
Peel blister pouch open part way and use to grasp the base of the needle, as shown. Holding the white collar on the syringe, attach syringe to needle luer connection with a firm clockwise twisting motion until snug. Do nottouch needle luer opening. This will result in contamination. |
Resuspend microspheres
Fully remove the blister pouch. Just before injection, shake syringe vigorously again, as some settling will have occurred. |
| Step 4 Inject dose | ||||
|---|---|---|---|---|
|
|
|
|
|
|
|
Remove transparent needle protector
Move the needle safety device back towards the syringe, as shown. Then hold white collar on syringe and carefully pull the transparent needle protector straight off. Do nottwist transparent needle protector, as the luer connection may loosen. |
Remove air bubbles
Hold needle upright and tap gently to make any air bubbles rise to the top. Slowly and carefully press plunger rod upward to remove air. |
Inject
Immediately inject entire contents of syringe intramuscularly (IM) into the gluteal or deltoid muscle of the patient. Gluteal injection should be made into the upper-outer quadrant of the gluteal area. Do not administer intravenously. |
Secure needle in safety device
Using one hand, place needle safety device at a 45-degree angle on a hard, flat surface. Press down with a firm, quick motion until needle is fully engaged in safety device. Avoid needle stick injury: Do notuse two hands. Do notintentionally disengage or mishandle the needle safety device. Do notattempt to straighten the needle or engage the safety device if the needle is bent or damaged. |
Properly dispose of needles
Check to confirm needle safety device is fully engaged. Discard in an approved sharps container. Also discard the unused needle provided in the dose pack. |
9.1 Controlled Substance
RISPERDAL CONSTA (risperidone) is not a controlled substance.
5 Warnings and Precautions
- Cerebrovascular events, including stroke, in elderly patients with dementia-related psychosis. RISPERDAL CONSTA is not approved for use in patients with dementia-related psychosis ( 5.2)
- Neuroleptic Malignant Syndrome: Manage with immediate discontinuation and close monitoring ( 5.3)
- Tardive Dyskinesia: Discontinue treatment if clinically appropriate ( 5.4)
- Metabolic Changes: Atypical antipsychotic drugs have been associated with metabolic changes that may increase cardiovascular/ cerebrovascular risk. These metabolic changes include hyperglycemia, dyslipidemia, and weight gain. (
5.5)
- Hyperglycemia and Diabetes Mellitus:Monitor patients for symptoms of hyperglycemia including polydipsia, polyuria, polyphagia, and weakness. Monitor glucose regularly in patients with diabetes or at risk for diabetes. ( 5.5)
- Dyslipidemia:Undesirable alterations have been observed in patients treated with atypical antipsychotics. ( 5.5)
- Weight Gain:Significant weight gain has been reported. Monitor weight gain. ( 5.5)
- Hyperprolactinemia: Risperidone treatment may elevate prolactin levels. Long-standing hyperprolactinemia, when associated with hypogonadism, can lead to decreased bone density in men and women. ( 5.6)
- Orthostatic hypotension: associated with dizziness, tachycardia, bradycardia, and syncope can occur, especially during initial dose titration with oral risperidone. Use caution in patients with cardiovascular disease, cerebrovascular disease, and conditions that could affect hemodynamic responses. ( 5.7)
- Leukopenia, Neutropenia, and Agranulocytosis have been reported with antipsychotics, including RISPERDAL CONSTA. Patients with history of a clinically significant low white blood cell count (WBC) or a drug-induced leukopenia/neutropenia should have their complete blood cell count (CBC) monitored frequently during the first few months of therapy and discontinuation of RISPERDAL CONSTA should be considered at the first sign of a clinically significant decline in WBC in the absence of other causative factors. ( 5.9)
- Potential for cognitive and motor impairment: has potential to impair judgment, thinking, and motor skills. Use caution when operating machinery, including automobiles. ( 5.10)
- Seizures: Use cautiously in patients with a history of seizures or with conditions that potentially lower the seizure threshold. ( 5.11)
- Dysphagia: Esophageal dysmotility and aspiration can occur. Use cautiously in patients at risk for aspiration pneumonia. ( 5.12)
- Priapism: has been reported. Severe priapism may require surgical intervention. ( 5.13)
- Avoid inadvertent administration into a blood vessel. ( 5.15)
2 Dosage and Administration
For patients who have never taken oral RISPERDAL, it is recommended to establish tolerability with oral RISPERDAL prior to initiating treatment with RISPERDAL CONSTA.
RISPERDAL CONSTA should be administered every 2 weeks by deep intramuscular (IM) deltoid or gluteal injection. Each injection should be administered by a health care professional using the appropriate enclosed safety needle [see Dosage and Administration (2.8)] . For deltoid administration, use the 1-inch needle alternating injections between the two arms. For gluteal administration, use the 2-inch needle alternating injections between the two buttocks. Do not administer intravenously.
5.7 Orthostatic Hypotension
RISPERDAL CONSTA may induce orthostatic hypotension associated with dizziness, tachycardia, and in some patients, syncope, especially during the initial dose-titration period with oral risperidone, probably reflecting its alpha-adrenergic antagonistic properties. Syncope was reported in 0.8% (12/1499 patients) of patients treated with RISPERDAL CONSTA in multiple-dose studies. Patients should be instructed in nonpharmacologic interventions that help to reduce the occurrence of orthostatic hypotension (e.g., sitting on the edge of the bed for several minutes before attempting to stand in the morning and slowly rising from a seated position).
RISPERDAL CONSTA should be used with particular caution in (1) patients with known cardiovascular disease (history of myocardial infarction or ischemia, heart failure, or conduction abnormalities), cerebrovascular disease, and conditions which would predispose patients to hypotension, e.g., dehydration and hypovolemia, and (2) in the elderly and patients with renal or hepatic impairment. Monitoring of orthostatic vital signs should be considered in all such patients, and a dose reduction should be considered if hypotension occurs. Clinically significant hypotension has been observed with concomitant use of oral RISPERDAL and antihypertensive medication.
3 Dosage Forms and Strengths
RISPERDAL CONSTA is available in dosage strengths of 12.5 mg, 25 mg, 37.5 mg, and 50 mg risperidone. It is provided as a single-use dose pack, consisting of a vial containing the risperidone microspheres, a pre-filled syringe containing 2 mL of diluent for RISPERDAL CONSTA, a vial adapter, and two Terumo SurGuard 3 Needles for intramuscular injection (a 21 G UTW 1-inch needle with needle protection device for deltoid administration and a 20 G TW 2-inch needle with needle protection device for gluteal administration).
6.2 Postmarketing Experience
The following adverse reactions have been identified during postapproval use of risperidone; because these reactions are reported voluntarily from a population of uncertain size, it is not possible to reliably estimate their frequency: agranulocytosis, alopecia, anaphylactic reaction, angioedema, atrial fibrillation, blood cholesterol increased, blood triglycerides increased, catatonia, diabetes mellitus, diabetic ketoacidosis in patients with impaired glucose metabolism, drug withdrawal syndrome neonatal, dysgeusia, hypoglycemia, hypothermia, ileus, inappropriate antidiuretic hormone secretion, intestinal obstruction, jaundice, mania, pancreatitis, priapism, QT prolongation, sleep apnea syndrome, somnambulism, Stevens-Johnson syndrome and toxic epidermal necrolysis (SJS/TEN), thrombocytopenia, thrombotic thrombocytopenic purpura, urinary retention, and water intoxication. In addition, the following adverse reactions have been observed during postapproval use of RISPERDAL CONSTA: cerebrovascular disorders, including cerebrovascular accidents, and diabetes mellitus aggravated.
Retinal artery occlusion after injection of RISPERDAL CONSTA has been reported during postmarketing surveillance. This has been reported in the presence of abnormal arteriovenous anastomosis.
Serious injection site reactions including abscess, cellulitis, cyst, hematoma, necrosis, nodule, and ulcer have been reported with RISPERDAL CONSTA during postmarketing surveillance. Isolated cases required surgical intervention.
Very rarely, cases of anaphylactic reaction after injection with RISPERDAL CONSTA have been reported during postmarketing experience in patients who have previously tolerated oral risperidone.
Postmarketing cases of extrapyramidal symptoms (dystonia and dyskinesia) have been reported in patients concomitantly taking methylphenidate and risperidone when there was an increase or decrease in dosage, initiation, or discontinuation of either or both medications.
10.2 Management of Overdosage
In case of acute overdosage, establish and maintain an airway and ensure adequate oxygenation and ventilation. Cardiovascular monitoring should commence immediately and should include continuous electrocardiographic monitoring to detect possible arrhythmias. If antiarrhythmic therapy is administered, disopyramide, procainamide, and quinidine carry a theoretical hazard of QT prolonging effects that might be additive to those of risperidone. Similarly, it is reasonable to expect that the alpha-blocking properties of bretylium might be additive to those of risperidone, resulting in problematic hypotension.
There is no specific antidote to risperidone. Therefore, appropriate supportive measures should be instituted. The possibility of multiple drug involvement should be considered. Hypotension and circulatory collapse should be treated with appropriate measures, such as intravenous fluids and/or sympathomimetic agents (epinephrine and dopamine should not be used, since beta stimulation may worsen hypotension in the setting of risperidone-induced alpha blockade). In cases of severe extrapyramidal symptoms, anticholinergic medication should be administered. Close medical supervision and monitoring should continue until the patient recovers.
7.5 Cimetidine and Ranitidine
Cimetidine and ranitidine increased the bioavailability of oral risperidone by 64% and 26%, respectively. However, cimetidine did not affect the AUC of risperidone and 9-hydroxyrisperidone combined, whereas ranitidine increased the AUC of risperidone and 9-hydroxyrisperidone combined by 20%.
8 Use in Specific Populations
- Pregnancy: May cause extrapyramidal and/or withdrawal symptoms in neonates with third trimester exposure. ( 8.1)
- Renal or Hepatic Impairment: dose appropriately with oral RISPERDAL prior to initiating treatment with RISPERDAL CONSTA. A lower starting dose of RISPERDAL CONSTA of 12.5 mg may be appropriate in some patients. ( 2.4)
- Pediatric Use: safety and effectiveness not established in patients less than 18 years of age. ( 8.4)
- Elderly: dosing for otherwise healthy elderly patients is the same as for healthy nonelderly. Elderly may be more predisposed to orthostatic effects than nonelderly. ( 8.5)
2.3 General Dosing Information
A lower initial dose of 12.5 mg may be appropriate when clinical factors warrant dose adjustment, such as in patients with hepatic or renal impairment, for certain drug interactions that increase risperidone plasma concentrations [see Drug Interactions (7.11)] or in patients who have a history of poor tolerability to psychotropic medications. The efficacy of the 12.5 mg dose has not been investigated in clinical trials.
Oral RISPERDAL (or another antipsychotic medication) should be given with the first injection of RISPERDAL CONSTA and continued for 3 weeks (and then discontinued) to ensure that adequate therapeutic plasma concentrations are maintained prior to the main release phase of risperidone from the injection site [see Clinical Pharmacology (12.3)] .
Upward dose adjustment should not be made more frequently than every 4 weeks. The clinical effects of this dose adjustment should not be anticipated earlier than 3 weeks after the first injection with the higher dose.
In patients with clinical factors such as hepatic or renal impairment or certain drug interactions that increase risperidone plasma concentrations [see Drug Interactions (7.11)] , dose reduction as low as 12.5 mg may be appropriate. The efficacy of the 12.5 mg dose has not been investigated in clinical trials.
Do not combine two different dose strengths of RISPERDAL CONSTA in a single administration.
8.6 Renal Or Hepatic Impairment
In patients with renal or hepatic impairment, carefully titrate with oral risperidone prior to initiating treatment with RISPERDAL CONSTA [see Dosage and Administration (2.4)].
Patients with renal impairment may have less ability to eliminate risperidone than patients with normal renal function. Patients with impaired hepatic function may have an increase in the free fraction of risperidone, possibly resulting in an enhanced effect [see Clinical Pharmacology (12.3)] .
5.14 Body Temperature Regulation
Disruption of body temperature regulation has been attributed to antipsychotic agents. Both hyperthermia and hypothermia have been reported in association with oral RISPERDAL or RISPERDAL CONSTA use. Caution is advised when prescribing RISPERDAL CONSTA for patients who will be exposed to temperature extremes.
17 Patient Counseling Information
Physicians are advised to discuss the following issues with patients for whom they prescribe RISPERDAL CONSTA.
7.14 Drugs Metabolized By Cyp 2d6
In vitrostudies indicate that risperidone is a relatively weak inhibitor of CYP 2D6. Therefore, RISPERDAL CONSTA is not expected to substantially inhibit the clearance of drugs that are metabolized by this enzymatic pathway. In drug interaction studies, oral RISPERDAL did not significantly affect the pharmacokinetics of donepezil and galantamine, which are metabolized by CYP 2D6.
5.3 Neuroleptic Malignant Syndrome
Neuroleptic Malignant Syndrome (NMS), a potentially fatal symptom complex, has been reported in association with antipsychotic drugs. Clinical manifestations of NMS are hyperpyrexia, muscle rigidity, altered mental status including delirium, and autonomic instability (irregular pulse or blood pressure, tachycardia, diaphoresis, and cardiac dysrhythmia). Additional signs may include elevated creatine phosphokinase, myoglobinuria (rhabdomyolysis), and acute renal failure.
If NMS is suspected, immediately discontinue RISPERDAL CONSTA and provide symptomatic treatment and monitoring.
7.2 Drugs With Hypotensive Effects
Because of its potential for inducing hypotension, RISPERDAL CONSTA may enhance the hypotensive effects of other therapeutic agents with this potential.
7.3 Levodopa and Dopamine Agonists
RISPERDAL CONSTA may antagonize the effects of levodopa and dopamine agonists.
14.2 Bipolar Disorder Monotherapy
The effectiveness of RISPERDAL CONSTA for the maintenance treatment of Bipolar I Disorder was established in a multicenter, double-blind, placebo-controlled study of adult patients who met DSM-IV criteria for Bipolar Disorder Type I, who were stable on medications or experiencing an acute manic or mixed episode.
A total of 501 patients were treated during a 26-week open-label period with RISPERDAL CONSTA (starting dose of 25 mg, and titrated, if deemed clinically desirable, to 37.5 mg or 50 mg; in patients not tolerating the 25 mg dose, the dose could be reduced to 12.5 mg). In the open-label phase, 303 (60%) patients were judged to be stable and were randomized to double-blind treatment with either the same dose of RISPERDAL CONSTA or placebo and monitored for relapse. The primary endpoint was time to relapse to any mood episode (depression, mania, hypomania, or mixed).
Time to relapse was delayed in patients receiving RISPERDAL CONSTA monotherapy as compared to placebo. The majority of relapses were due to manic rather than depressive symptoms. Based on their bipolar disorder history, subjects entering this study had had, on average, more manic episodes than depressive episodes.
16 How Supplied/storage and Handling
RISPERDAL CONSTA ®(risperidone) is available in dosage strengths of 12.5 mg, 25 mg, 37.5 mg, or 50 mg risperidone. It is provided as a single-use dose pack, consisting of a vial containing the risperidone microspheres, a pre-filled syringe containing 2 mL of diluent for RISPERDAL CONSTA, a vial adapter, and two Terumo SurGuard ®3 Needles for intramuscular injection (a 21 G UTW 1-inch needle with needle protection device for deltoid administration and a 20 G TW 2-inch needle with needle protection device for gluteal administration).
12.5-mg vial/kit (NDC 50458-309-11): 41 mg (equivalent to 12.5 mg of risperidone) of a white to off-white powder provided in a vial with a violet flip-off cap (NDC 50458-309-01).
25-mg vial/kit (NDC 50458-306-11): 78 mg (equivalent to 25 mg of risperidone) of a white to off-white powder provided in a vial with a pink flip-off cap (NDC 50458-306-01).
37.5-mg vial/kit (NDC 50458-307-11): 116 mg (equivalent to 37.5 mg of risperidone) of a white to off-white powder provided in a vial with a green flip-off cap (NDC 50458-307-01).
50-mg vial/kit (NDC 50458-308-11): 152 mg (equivalent to 50 mg of risperidone) of a white to off-white powder provided in a vial with a blue flip-off cap (NDC 50458-308-01).
7.1 Centrally Acting Drugs and Alcohol
Given the primary CNS effects of risperidone, caution should be used when RISPERDAL CONSTA is administered in combination with other centrally-acting drugs or alcohol.
2.6 Switching From Other Antipsychotics
There are no systematically collected data to specifically address switching patients from other antipsychotics to RISPERDAL CONSTA, or concerning concomitant administration with other antipsychotics. Previous antipsychotics should be continued for 3 weeks after the first injection of RISPERDAL CONSTA to ensure that therapeutic concentrations are maintained until the main release phase of risperidone from the injection site has begun [see Clinical Pharmacology (12.3)] . For patients who have never taken oral RISPERDAL, it is recommended to establish tolerability with oral RISPERDAL prior to initiating treatment with RISPERDAL CONSTA. As recommended with other antipsychotic medications, the need for continuing existing EPS medication should be re-evaluated periodically.
5.16 Osteodystrophy and Tumors in Animals
RISPERDAL CONSTA produced osteodystrophy in male and female rats in a 1-year toxicity study and a 2-year carcinogenicity study at a dose of 40 mg/kg administered IM every 2 weeks.
RISPERDAL CONSTA produced renal tubular tumors (adenoma, adenocarcinoma) and adrenomedullary pheochromocytomas in male rats in the 2-year carcinogenicity study at 40 mg/kg administered IM every 2 weeks. In addition, RISPERDAL CONSTA produced an increase in a marker of cellular proliferation in renal tissue in males in the 1-year toxicity study and in renal tumor-bearing males in the 2-year carcinogenicity study at 40 mg/kg administered IM every 2 weeks. (Cellular proliferation was not measured at the low dose or in females in either study.)
The effect dose for osteodystrophy and the tumor findings is 8 times the IM maximum recommended human dose (MRHD) (50 mg) on a mg/m 2basis and is associated with a plasma exposure (AUC) 2 times the expected plasma exposure (AUC) at the IM MRHD. The no-effect dose for these findings was 5 mg/kg (equal to the IM MRHD on a mg/m 2basis). Plasma exposure (AUC) at the no-effect dose was one third the expected plasma exposure (AUC) at the IM MRHD.
Neither the renal or adrenal tumors, nor osteodystrophy, were seen in studies of orally administered risperidone.Osteodystrophy was not observed in dogs at doses up to 14 times (based on AUC) the IM MRHD in a 1-year toxicity study.
The renal tubular and adrenomedullary tumors in male rats and other tumor findings are described in more detail in Section 13.1 (Carcinogenicity, Mutagenesis, Impairment of Fertility).
The relevance of these findings to human risk is unknown.
14.3 Bipolar Disorder Adjunctive Therapy
The effectiveness of RISPERDAL CONSTA as an adjunct to treatment with lithium or valproate for the maintenance treatment of Bipolar Disorder was established in a multi-center, randomized, double-blind, placebo-controlled study of adult patients who met DSM-IV criteria for Bipolar Disorder Type I and who experienced at least 4 episodes of mood disorder requiring psychiatric/clinical intervention in the previous 12 months, including at least 2 episodes in the 6 months prior to the start of the study.
A total of 240 patients were treated during a 16-week open-label period with RISPERDAL CONSTA (starting dose of 25 mg, and titrated, if deemed clinically desirable, to 37.5 mg or 50 mg), as adjunctive therapy in addition to continuing their treatment as usual for their bipolar disorder, which consisted of mood stabilizers (primarily lithium and valproate), antidepressants, and/or anxiolytics. All oral antipsychotics were discontinued after the first three weeks of the initial RISPERDAL CONSTA injection. In the open-label phase, 124 (51.7%) were judged to be stable for at least the last 4 weeks and were randomized to double-blind treatment with either the same dose of RISPERDAL CONSTA or placebo in addition to continuing their treatment as usual and monitored for relapse during a 52-week period. The primary endpoint was time to relapse to any new mood episode (depression, mania, hypomania, or mixed).
Time to relapse was delayed in patients receiving adjunctive therapy with RISPERDAL CONSTA as compared to placebo. The relapse types were about half depressive and half manic or mixed episodes.
Principal Display Panel 25 Mg Kit Carton
25 mg
Dose Pack
Store Package
in Refrigerator
(see bottom panel for
storage conditions)
Rx only
Single use only
NDC50458-306-11
Risperdal
CONSTA
®
risperiDONE Long-Acting Injection
DOSE PACK CONTENTS:
- One 25 mg vial of risperidone extended-release microspheres for injection
- One pre-filled syringe containing 2 mL of diluent
- One Vial Adapter
- One 21-gauge UTW 1" Terumo SurGuard ®3 safety needle for deltoid injection
- One 20-gauge TW 2" Terumo SurGuard ®3 safety needle for gluteal injection
Do not substitute any components of the dose pack.
For deltoid or gluteal intramuscular injection only
Each injection should be administered by a healthcare professional.
PLEASE READ COMPLETE INSTRUCTIONS PRIOR TO USE.
Principal Display Panel 50 Mg Kit Carton
50 mg
Dose Pack
Store Package
in Refrigerator
(see bottom panel for
storage conditions)
Rx only
Single use only
NDC50458-308-11
Risperdal
CONSTA
®
risperiDONE Long-Acting Injection
DOSE PACK CONTENTS:
- One 50 mg vial of risperidone extended-release microspheres for injection
- One pre-filled syringe containing 2 mL of diluent
- One Vial Adapter
- One 21-gauge UTW 1" Terumo SurGuard ®3 safety needle for deltoid injection
- One 20-gauge TW 2" Terumo SurGuard ®3 safety needle for gluteal injection
Do not substitute any components of the dose pack.
For deltoid or gluteal intramuscular injection only
Each injection should be administered by a healthcare professional.
PLEASE READ COMPLETE INSTRUCTIONS PRIOR TO USE.
Principal Display Panel 12.5 Mg Kit Carton
12.5 mg
Dose Pack
Store Package
in Refrigerator
(see bottom panel for
storage conditions)
Rx only
Single use only
NDC50458-309-11
Risperdal
CONSTA
®
risperiDONE Long-Acting Injection
DOSE PACK CONTENTS:
- One 12.5 mg vial of risperidone extended-release microspheres for injection
- One pre-filled syringe containing 2 mL of diluent
- One Vial Adapter
- One 21-gauge UTW 1" Terumo SurGuard ®3 safety needle for deltoid injection
- One 20-gauge TW 2" Terumo SurGuard ®3 safety needle for gluteal injection
Do not substitute any components of the dose pack.
For deltoid or gluteal intramuscular injection only
Each injection should be administered by a healthcare professional.
PLEASE READ COMPLETE INSTRUCTIONS PRIOR TO USE.
Principal Display Panel 37.5 Mg Kit Carton
37.5 mg
Dose Pack
Store Package
in Refrigerator
(see bottom panel for
storage conditions)
Rx only
Single use only
NDC50458-307-11
Risperdal
CONSTA
®
risperiDONE Long-Acting Injection
DOSE PACK CONTENTS:
- One 37.5 mg vial of risperidone extended-release microspheres for injection
- One pre-filled syringe containing 2 mL of diluent
- One Vial Adapter
- One 21-gauge UTW 1" Terumo SurGuard ®3 safety needle for deltoid injection
- One 20-gauge TW 2" Terumo SurGuard ®3 safety needle for gluteal injection
Do not substitute any components of the dose pack.
For deltoid or gluteal intramuscular injection only
Each injection should be administered by a healthcare professional.
PLEASE READ COMPLETE INSTRUCTIONS PRIOR TO USE.
5.10 Potential for Cognitive and Motor Impairment
Somnolence was reported by 5% of patients treated with RISPERDAL CONSTA in multiple-dose trials. Since risperidone has the potential to impair judgment, thinking, or motor skills, patients should be cautioned about operating hazardous machinery, including automobiles, until they are reasonably certain that treatment with RISPERDAL CONSTA does not affect them adversely.
7.13 Carbamazepine and Other Cyp 3a4 Enzyme Inducers
Carbamazepine co-administration with oral RISPERDAL decreased the steady-state plasma concentrations of risperidone and 9-hydroxyrisperidone by about 50%. Plasma concentrations of carbamazepine did not appear to be affected. Co-administration of other known CYP 3A4 enzyme inducers (e.g., phenytoin, rifampin, and phenobarbital) with risperidone may cause similar decreases in the combined plasma concentrations of risperidone and 9-hydroxyrisperidone, which could lead to decreased efficacy of RISPERDAL CONSTA treatment. At the initiation of therapy with carbamazepine or other known hepatic enzyme inducers, patients should be closely monitored during the first 4–8 weeks, since the dose of RISPERDAL CONSTA may need to be adjusted. A dose increase, or additional oral RISPERDAL, may need to be considered. On discontinuation of carbamazepine or other CYP 3A4 hepatic enzyme inducers, the dosage of RISPERDAL CONSTA should be re-evaluated and, if necessary, decreased. Patients may be placed on a lower dose of RISPERDAL CONSTA between 2 to 4 weeks before the planned discontinuation of carbamazepine or other CYP 3A4 enzyme inducers to adjust for the expected increase in plasma concentrations of risperidone plus 9-hydroxyrisperidone. For patients treated with the recommended dose of 25 mg RISPERDAL CONSTA and discontinuing from carbamazepine or other CYP 3A4 enzyme inducers, it is recommended to continue treatment with the 25-mg dose unless clinical judgment necessitates lowering the RISPERDAL CONSTA dose to 12.5 mg or necessitates interruption of RISPERDAL CONSTA treatment. The efficacy of the 12.5 mg dose has not been investigated in clinical trials. [see also Dosage and Administration (2.5)]
7.12 Drugs That Inhibit Cyp 2d6 and Other Cyp Isozymes
Risperidone is metabolized to 9-hydroxyrisperidone by CYP 2D6, an enzyme that is polymorphic in the population and that can be inhibited by a variety of psychotropic and other drugs [see Clinical Pharmacology (12.3)] . Drug interactions that reduce the metabolism of risperidone to 9-hydroxyrisperidone would increase the plasma concentrations of risperidone and lower the concentrations of 9-hydroxyrisperidone. Analysis of clinical studies involving a modest number of poor metabolizers (n≅70 patients) does not suggest that poor and extensive metabolizers have different rates of adverse effects. No comparison of effectiveness in the two groups has been made.
In vitrostudies showed that drugs metabolized by other CYP isozymes, including 1A1, 1A2, 2C9, 2C19, and 3A4, are only weak inhibitors of risperidone metabolism.
8.7 Patients With Parkinson's Disease Or Lewy Body Dementia
Patients with Parkinson's Disease or Dementia with Lewy Bodies can experience increased sensitivity to RISPERDAL CONSTA. Manifestations can include confusion, obtundation, postural instability with frequent falls, extrapyramidal symptoms, and clinical features consistent with neuroleptic malignant syndrome.
2.5 Reinitiation of Treatment in Patients Previously Discontinued
There are no data to specifically address reinitiation of treatment. When restarting patients who have had an interval off treatment with RISPERDAL CONSTA, supplementation with oral RISPERDAL (or another antipsychotic medication) should be administered.
2.7 Co Administration of Risperdal Consta With Certain Other Medications
Co-administration of carbamazepine and other CYP 3A4 enzyme inducers (e.g., phenytoin, rifampin, phenobarbital) with risperidone would be expected to cause decreases in the plasma concentrations of the sum of risperidone and 9-hydroxyrisperidone combined, which could lead to decreased efficacy of RISPERDAL CONSTA treatment. The dose of risperidone needs to be titrated accordingly for patients receiving these enzyme inducers, especially during initiation or discontinuation of therapy with these inducers [see Drug Interactions (7.11)] . At the initiation of therapy with carbamazepine or other known CYP 3A4 hepatic enzyme inducers, patients should be closely monitored during the first 4–8 weeks, since the dose of RISPERDAL CONSTA may need to be adjusted. A dose increase, or additional oral RISPERDAL, may need to be considered. On discontinuation of carbamazepine or other CYP 3A4 hepatic enzyme inducers, the dosage of RISPERDAL CONSTA should be re-evaluated and, if necessary, decreased. Patients may be placed on a lower dose of RISPERDAL CONSTA between 2 to 4 weeks before the planned discontinuation of carbamazepine or other CYP 3A4 inducers to adjust for the expected increase in plasma concentrations of risperidone plus 9-hydroxyrisperidone. For patients treated with the recommended dose of 25 mg RISPERDAL CONSTA and discontinuing from carbamazepine or other CYP3A4 enzyme inducers, it is recommended to continue treatment with the 25-mg dose unless clinical judgment necessitates lowering the RISPERDAL CONSTA dose to 12.5 mg or necessitates interruption of RISPERDAL CONSTA treatment. The efficacy of the 12.5 mg dose has not been investigated in clinical trials.
Fluoxetine and paroxetine, CYP 2D6 inhibitors, have been shown to increase the plasma concentration of risperidone 2.5–2.8 fold and 3–9 fold respectively. Fluoxetine did not affect the plasma concentration of 9-hydroxyrisperidone. Paroxetine lowered the concentration of 9-hydroxyrisperidone by about 10%. The dose of risperidone needs to be titrated accordingly when fluoxetine or paroxetine is co-administered. When either concomitant fluoxetine or paroxetine is initiated or discontinued, the physician should re-evaluate the dose of RISPERDAL CONSTA. When initiation of fluoxetine or paroxetine is considered, patients may be placed on a lower dose of RISPERDAL CONSTA between 2 to 4 weeks before the planned start of fluoxetine or paroxetine therapy to adjust for the expected increase in plasma concentrations of risperidone. When fluoxetine or paroxetine is initiated in patients receiving the recommended dose of 25 mg RISPERDAL CONSTA, it is recommended to continue treatment with the 25 mg dose unless clinical judgment necessitates lowering the RISPERDAL CONSTA dose to 12.5 mg or necessitates interruption of RISPERDAL CONSTA treatment. When RISPERDAL CONSTA is initiated in patients already receiving fluoxetine or paroxetine, a starting dose of 12.5 mg can be considered. The efficacy of the 12.5 mg dose has not been investigated in clinical trials. The effects of discontinuation of concomitant fluoxetine or paroxetine therapy on the pharmacokinetics of risperidone and 9-hydroxyrisperidone have not been studied. [see Drug Interactions (7.11)] .
5.1 Increased Mortality in Elderly Patients With Dementia Related Psychosis
Elderly patients with dementia-related psychosis treated with antipsychotic drugs are at an increased risk of death. Analyses of 17 placebo-controlled trials (modal duration of 10 weeks), largely in patients taking atypical antipsychotic drugs, revealed a risk of death in drug-treated patients of between 1.6 to 1.7 times the risk of death in placebo-treated patients. Over the course of a typical 10-week controlled trial, the rate of death in drug-treated patients was about 4.5%, compared to a rate of about 2.6% in the placebo group. Although the causes of death were varied, most of the deaths appeared to be either cardiovascular (e.g., heart failure, sudden death) or infectious (e.g., pneumonia) in nature. Observational studies suggest that, similar to atypical antipsychotic drugs, treatment with conventional antipsychotic drugs may increase mortality. The extent to which the findings of increased mortality in observational studies may be attributed to the antipsychotic drug as opposed to some characteristic(s) of the patients is not clear.
RISPERDAL CONSTA (risperidone) is not approved for the treatment of dementia-related psychosis [see Boxed Warning] .
Warning: Increased Mortality in Elderly Patients With Dementia Related Psychosis
Elderly patients with dementia-related psychosis treated with antipsychotic drugs are at an increased risk of death. RISPERDAL CONSTA is not approved for the treatment of patients with dementia-related psychosis. [see Warnings and Precautions (5.1)]
5.2 Cerebrovascular Adverse Events, Including Stroke, in Elderly Patients With Dementia Related Psychosis
Cerebrovascular adverse events (e.g., stroke, transient ischemic attack), including fatalities, were reported in patients (mean age 85 years; range 73–97) in trials of oral risperidone in elderly patients with dementia-related psychosis. In placebo-controlled trials, there was a significantly higher incidence of cerebrovascular adverse events in patients treated with oral risperidone compared to patients treated with placebo. RISPERDAL CONSTA is not approved for the treatment of patients with dementia-related psychosis. [See also Boxed Warning and Warnings and Precautions (5.1)]
Structured Label Content
Section 42229-5 (42229-5)
Elderly
For elderly patients treated with RISPERDAL CONSTA, the recommended dosage is 25 mg IM every 2 weeks. Oral RISPERDAL (or another antipsychotic medication) should be given with the first injection of RISPERDAL CONSTA and should be continued for 3 weeks to ensure that adequate therapeutic plasma concentrations are maintained prior to the main release phase of risperidone from the injection site [see Clinical Pharmacology (12.3)] .
Section 43683-2 (43683-2)
| Warnings and Precautions ( 5.6) | 1/2025 |
Section 44425-7 (44425-7)
Storage and Handling
The entire dose pack should be stored in the refrigerator (36 °F to 46 °F; 2 °C to 8 °C) and protected from light.
If refrigeration is unavailable, RISPERDAL CONSTA can be stored at temperatures not exceeding 77 °F (25 °C) for no more than 7 days prior to administration. Do not expose unrefrigerated product to temperatures above 77 °F (25 °C).
Keep out of reach of children.
5.8 Falls
Somnolence, postural hypotension, motor and sensory instability have been reported with the use of antipsychotics, including RISPERDAL CONSTA, which may lead to falls and, consequently, fractures or other fall-related injuries. For patients, particularly the elderly, with diseases, conditions, or medications that could exacerbate these effects, assess the risk of falls when initiating antipsychotic treatment and recurrently for patients on long-term antipsychotic therapy.
9.2 Abuse
RISPERDAL CONSTA has not been systematically studied in animals or humans for its potential for abuse. Because RISPERDAL CONSTA is to be administered by health care professionals, the potential for misuse or abuse by patients is low.
7.8 Lithium
Repeated doses of oral RISPERDAL (3 mg twice daily) did not affect the exposure (AUC) or peak plasma concentrations (C max) of lithium (N=13).
7.10 Digoxin
Oral RISPERDAL (0.25 mg twice daily) did not show a clinically relevant effect on the pharmacokinetics of digoxin.
5.11 Seizures
During premarketing testing, seizures occurred in 0.3% (5/1499 patients) of patients treated with RISPERDAL CONSTA. Therefore, RISPERDAL CONSTA should be used cautiously in patients with a history of seizures.
5.13 Priapism
Priapism has been reported during postmarketing surveillance [see Adverse Reactions (6.2)] . Severe priapism may require surgical intervention.
7.7 Clozapine
Chronic administration of clozapine with risperidone may decrease the clearance of risperidone.
7.9 Valproate
Repeated doses of oral RISPERDAL (4 mg once daily) did not affect the pre-dose or average plasma concentrations and exposure (AUC) of valproate (1000 mg/day in three divided doses) compared to placebo (N=21). However, there was a 20% increase in valproate peak plasma concentration (C max) after concomitant administration of oral RISPERDAL.
11 Description (11 DESCRIPTION)
RISPERDAL CONSTA ®contains risperidone, an atypical antipsychotic belonging to the chemical class of benzisoxazole derivatives. The chemical designation is 3-[2-[4-(6-fluoro-1,2-benzisoxazol-3-yl)-1-piperidinyl]ethyl]-6,7,8,9-tetrahydro-2-methyl-4H-pyrido[1,2-a]pyrimidin-4-one. Its molecular formula is C 23 H 27 FN 4O 2 and its molecular weight is 410.49. The structural formula is:
Risperidone is practically insoluble in water, freely soluble in methylene chloride, and soluble in methanol and 0.1 NHCl.
RISPERDAL CONSTA (risperidone) Long-Acting Injection is a combination of extended-release microspheres for injection and diluent for parenteral use.
The extended-release microspheres formulation is a white to off-white, free-flowing powder that is available in dosage strengths of 12.5 mg, 25 mg, 37.5 mg, or 50 mg risperidone per vial. Risperidone is micro-encapsulated in 7525 polylactide-co-glycolide (PLG) at a concentration of 381 mg risperidone per gram of microspheres.
The diluent for parenteral use is a clear, colorless solution. Composition of the diluent includes 1 mg/mL citric acid anhydrous, 1.27 mg/mL disodium hydrogen phosphate dihydrate, 1 mg/mL polysorbate 20, 22.5 mg/mL sodium carboxymethyl cellulose, 6 mg/mL sodium chloride, 0.54 mg/mL sodium hydroxide, and water for injection. The microspheres are suspended in the diluent prior to injection.
RISPERDAL CONSTA is provided as a single-use dose pack, consisting of a vial containing the microspheres, a pre-filled syringe containing the diluent, a vial adapter, and two Terumo SurGuard 3 Needles (a 21 G UTW 1-inch needle with needle protection device for deltoid administration and a 20 G TW 2-inch needle with needle protection device for gluteal administration).
5.12 Dysphagia
Esophageal dysmotility and aspiration have been associated with antipsychotic drug use. Aspiration pneumonia is a common cause of morbidity and mortality in patients with advanced Alzheimer's dementia. RISPERDAL CONSTA and other antipsychotic drugs should be used cautiously in patients at risk for aspiration pneumonia. [see also Boxed Warning and Warnings and Precautions (5.1)]
9.3 Dependence
RISPERDAL CONSTA has not been systematically studied in animals or humans for its potential for tolerance or physical dependence.
7.11 Topiramate
Oral RISPERDAL administered at doses from 1–6 mg/day concomitantly with topiramate 400 mg/day resulted in a 23% decrease in risperidone C max and a 33% decrease in risperidone AUC 0–12 hour at steady state. Minimal reductions in the exposure to risperidone and 9-hydroxyrisperidone combined, and no change for 9-hydroxyrisperidone were observed. This interaction is unlikely to be of clinical significance. There was no clinically relevant effect of oral RISPERDAL on the pharmacokinetics of topiramate.
1.1 Schizophrenia
RISPERDAL CONSTA (risperidone) is indicated for the treatment of schizophrenia [see Clinical Studies (14.1)].
2.1 Schizophrenia
The recommended dose for the treatment of schizophrenia is 25 mg IM every 2 weeks. Although dose response for effectiveness has not been established for RISPERDAL CONSTA, some patients not responding to 25 mg may benefit from a higher dose of 37.5 mg or 50 mg. The maximum dose should not exceed 50 mg RISPERDAL CONSTA every 2 weeks. No additional benefit was observed with dosages greater than 50 mg RISPERDAL CONSTA; however, a higher incidence of adverse effects was observed.
The efficacy of RISPERDAL CONSTA in the treatment of schizophrenia has not been evaluated in controlled clinical trials for longer than 12 weeks. Although controlled studies have not been conducted to answer the question of how long patients with schizophrenia should be treated with RISPERDAL CONSTA, oral risperidone has been shown to be effective in delaying time to relapse in longer-term use. It is recommended that responding patients be continued on treatment with RISPERDAL CONSTA at the lowest dose needed. The physician who elects to use RISPERDAL CONSTA for extended periods should periodically re-evaluate the long-term risks and benefits of the drug for the individual patient.
7.4 Amitriptyline
Amitriptyline did not affect the pharmacokinetics of risperidone or of risperidone and 9-hydroxyrisperidone combined following concomitant administration with oral RISPERDAL.
8.4 Pediatric Use
Safety and effectiveness of RISPERDAL CONSTA in pediatric patients have not been established. However, juvenile animal toxicology studies have been conducted with oral risperidone.
8.5 Geriatric Use
In an open-label study, 57 clinically stable, elderly patients (≥ 65 years old) with schizophrenia or schizoaffective disorder received RISPERDAL CONSTA every 2 weeks for up to 12 months. In general, no differences in the tolerability of RISPERDAL CONSTA were observed between otherwise healthy elderly and nonelderly patients. Therefore, dosing recommendations for otherwise healthy elderly patients are the same as for nonelderly patients. Because elderly patients exhibit a greater tendency to orthostatic hypotension than nonelderly patients, elderly patients should be instructed in nonpharmacologic interventions that help to reduce the occurrence of orthostatic hypotension (e.g., sitting on the edge of the bed for several minutes before attempting to stand in the morning and slowly rising from a seated position). In addition, monitoring of orthostatic vital signs should be considered in elderly patients for whom orthostatic hypotension is of concern [see Warnings and Precautions (5.7)] .
14.1 Schizophrenia
The effectiveness of RISPERDAL CONSTA in the treatment of schizophrenia was established, in part, on the basis of extrapolation from the established effectiveness of the oral formulation of risperidone. In addition, the effectiveness of RISPERDAL CONSTA in the treatment of schizophrenia was established in a 12-week, placebo-controlled trial in adult psychotic inpatients and outpatients who met the DSM-IV criteria for schizophrenia.
Efficacy data were obtained from 400 patients with schizophrenia who were randomized to receive injections of 25 mg, 50 mg, or 75 mg RISPERDAL CONSTA or placebo every 2 weeks. During a 1-week run-in period, patients were discontinued from other antipsychotics and were titrated to a dose of 4 mg oral RISPERDAL. Patients who received RISPERDAL CONSTA were given doses of oral RISPERDAL (2 mg for patients in the 25-mg group, 4 mg for patients in the 50-mg group, and 6 mg for patients in the 75-mg group) for the 3 weeks after the first injection to provide therapeutic plasma concentrations until the main release phase of risperidone from the injection site had begun. Patients who received placebo injections were given placebo tablets.
Efficacy was evaluated using the Positive and Negative Syndrome Scale (PANSS), a validated, multi-item inventory, composed of five subscales to evaluate positive symptoms, negative symptoms, disorganized thoughts, uncontrolled hostility/excitement, and anxiety/depression.
The primary efficacy variable in this trial was change from baseline to endpoint in the total PANSS score. The mean total PANSS score at baseline for schizophrenic patients in this study was 81.5.
Total PANSS scores showed significant improvement in the change from baseline to endpoint in schizophrenic patients treated with each dose of RISPERDAL CONSTA (25 mg, 50 mg, or 75 mg) compared with patients treated with placebo. While there were no statistically significant differences between the treatment effects for the three dose groups, the effect size for the 75 mg dose group was actually numerically less than that observed for the 50 mg dose group.
Subgroup analyses did not indicate any differences in treatment outcome as a function of age, race, or gender.
4 Contraindications (4 CONTRAINDICATIONS)
RISPERDAL CONSTA is contraindicated in patients with a known hypersensitivity to either risperidone or paliperidone, or to any of the excipients in the RISPERDAL CONSTA formulation. Hypersensitivity reactions, including anaphylactic reactions and angioedema, have been reported in patients treated with risperidone and in patients treated with paliperidone. Paliperidone is a metabolite of risperidone.
5.15 Administration
RISPERDAL CONSTA should be injected into the deltoid or gluteal muscle, and care must be taken to avoid inadvertent injection into a blood vessel. [see Dosage and Administration (2) and Adverse Reactions (6)] .
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following are discussed in more detail in other sections of the labeling:
- Increased mortality in elderly patients with dementia-related psychosis [see Boxed Warningand Warnings and Precautions (5.1)]
- Cerebrovascular adverse events, including stroke, in elderly patients with dementia-related psychosis [see Warnings and Precautions (5.2)]
- Neuroleptic malignant syndrome [see Warnings and Precautions (5.3)]
- Tardive dyskinesia [see Warnings and Precautions (5.4)]
- Metabolic changes [see Warnings and Precautions (5.5)]
- Hyperprolactinemia [see Warnings and Precautions (5.6)]
- Orthostatic hypotension [see Warnings and Precautions (5.7)]
- Falls [see Warnings and Precautions (5.8)]
- Leukopenia/Neutropenia and Agranulocytosis [see Warnings and Precautions (5.9)]
- Potential for cognitive and motor impairment [see Warnings and Precautions (5.10)]
- Seizures [see Warnings and Precautions (5.11)]
- Dysphagia [see Warnings and Precautions (5.12)]
- Priapism [see Warnings and Precautions (5.13)]
- Disruption of body temperature regulation [see Warnings and Precautions (5.14)]
- Avoidance of inadvertent injection into a blood vessel [see Warnings and Precautions (5.15)]
- Osteodystrophy and tumors in animals [see Warnings and Precautions (5.16)]
The most common adverse reactions in clinical trials in patients with schizophrenia (≥ 5%) were: headache, parkinsonism, dizziness, akathisia, fatigue, constipation, dyspepsia, sedation, weight increased, pain in extremity, and dry mouth. The most common adverse reactions in the double-blind, placebo-controlled periods of the bipolar disorder trials were weight increased (5% in the monotherapy trial) and tremor and parkinsonism (≥ 10% in the adjunctive treatment trial).
The most common adverse reactions that were associated with discontinuation from the 12-week double-blind, placebo-controlled trial in patients with schizophrenia (causing discontinuation in ≥1% of patients) were agitation, depression, anxiety, and akathisia. Adverse reactions that were associated with discontinuation from the double-blind, placebo-controlled periods of the bipolar disorder trials were hyperglycemia (one patient in the monotherapy trial) and hypokinesia and tardive dyskinesia (one patient each in the adjunctive treatment trial).
The data described in this section are derived from a clinical trial database consisting of 2392 patients exposed to one or more doses of RISPERDAL CONSTA for the treatment of schizophrenia. Of these 2392 patients, 332 were patients who received RISPERDAL CONSTA while participating in a 12-week double-blind, placebo-controlled trial. Two hundred two (202) of the 332 were schizophrenia patients who received 25 mg or 50 mg RISPERDAL CONSTA. The conditions and duration of treatment with RISPERDAL CONSTA in the other clinical trials varied greatly and included (in overlapping categories) double-blind, fixed- and flexible-dose, placebo- or active-controlled studies and open-label phases of studies, inpatients and outpatients, and short-term (up to 12 weeks) and longer-term (up to 4 years) exposures. Safety was assessed by collecting adverse events and performing physical examinations, vital signs, body weights, laboratory analyses, and ECGs.
In addition to the studies in patients with schizophrenia, safety data are presented from a trial assessing the efficacy and safety of RISPERDAL CONSTA when administered as monotherapy for maintenance treatment in patients with bipolar I disorder. The subjects in this multi-center, double-blind, placebo-controlled study were adult patients who met DSM-IV criteria for Bipolar Disorder Type I and who were stable on risperidone (oral or long-acting injection), were stable on other antipsychotics or mood stabilizers, or were experiencing an acute episode. After a 3-week period of treatment with open-label oral risperidone (N=440), subjects who demonstrated an initial response to oral risperidone in this period and those who were stable on risperidone (oral or long-acting injection) at study entry entered into a 26-week stabilization period of open-label RISPERDAL CONSTA (N=501). Subjects who demonstrated a maintained response during this period were then randomized into a 24-month double-blind, placebo-controlled period in which they received RISPERDAL CONSTA (N=154) or placebo (N=149) as monotherapy. Subjects who relapsed or who completed the double-blind period could choose to enter an 8-week open-label RISPERDAL CONSTA extension period (N=160).
Safety data are also presented from a trial assessing the efficacy and safety of RISPERDAL CONSTA when administered as adjunctive maintenance treatment in patients with bipolar disorder. The subjects in this multi-center, double-blind, placebo-controlled study were adult patients who met DSM-IV criteria for Bipolar Disorder Type I or Type II and who experienced at least 4 episodes of mood disorder requiring psychiatric/clinical intervention in the previous 12 months, including at least 2 episodes in the 6 months prior to the start of the study. At the start of this study, all patients (N=275) entered into a 16-week open-label treatment phase in which they received RISPERDAL CONSTA in addition to continuing their treatment as usual, which consisted of various mood stabilizers (primarily lithium and valproate), antidepressants, and/or anxiolytics. Patients who reached remission at the end of this 16-week open-label treatment phase (N=139) were then randomized into a 52-week double-blind, placebo-controlled phase in which they received RISPERDAL CONSTA (N=72) or placebo (n=67) as adjunctive treatment in addition to continuing their treatment as usual. Patients who did not reach remission at the end of the 16-week open-label treatment phase could choose to continue to receive RISPERDAL CONSTA as adjunctive therapy in an open-label manner, in addition to continuing their treatment as usual, for up to an additional 36 weeks as clinically indicated for a total period of up to 52 weeks; these patients (N=70) were also included in the evaluation of safety.
Adverse events during exposure to study treatment were obtained by general inquiry and recorded by clinical investigators using their own terminology. Consequently, to provide a meaningful estimate of the proportion of individuals experiencing adverse events, events were grouped in standardized categories using MedDRA terminology.
Throughout this section, adverse reactions are reported. Adverse reactions are adverse events that were considered to be reasonably associated with the use of RISPERDAL CONSTA (adverse drug reactions) based on the comprehensive assessment of the available adverse event information. A causal association for RISPERDAL CONSTA often cannot be reliably established in individual cases. Further, because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
The majority of all adverse reactions were mild to moderate in severity.
7 Drug Interactions (7 DRUG INTERACTIONS)
The interactions of RISPERDAL CONSTA with coadministration of other drugs have not been systematically evaluated. The drug interaction data provided in this section is based on studies with oral RISPERDAL.
7.6 Methylphenidate
Concomitant use with methylphenidate, when there is change in dosage of either medication, may increase the risk of extrapyramidal symptoms (EPS). Monitor for symptoms of EPS with concomitant use of RISPERDAL CONSTA and methylphenidate [see Adverse Reactions (6.2)] .
1.2 Bipolar Disorder
RISPERDAL CONSTA is indicated as monotherapy or as adjunctive therapy to lithium or valproate for the maintenance treatment of Bipolar I Disorder [see Clinical Studies (14.2 , 14.3)] .
2.2 Bipolar Disorder
The recommended dose for monotherapy or adjunctive therapy to lithium or valproate for the maintenance treatment of Bipolar I Disorder is 25 mg IM every 2 weeks. Some patients may benefit from a higher dose of 37.5 mg or 50 mg. Dosages above 50 mg have not been studied in this population. The physician who elects to use RISPERDAL CONSTA for extended periods should periodically re-evaluate the long-term risks and benefits of the drug for the individual patient.
Instructions for Use (INSTRUCTIONS FOR USE)
RISPERDAL CONSTA
®
(risperidone)
Long-Acting Injection
12.5 mg, 25 mg, 37.5 mg, 50 mg
For deltoid or gluteal intramuscular injection only
Each injection should be administered by a healthcare professional.
IMPORTANT RESOURCES
For additional information, visit www.risperdalconsta.com or call Janssen Pharmaceuticals, Inc. at 1-800-526-7736.
Manufactured for:
Janssen Pharmaceuticals, Inc.
Titusville, NJ 08560, USA
For patent information: www.janssenpatents.com
© Johnson & Johnson and its affiliates 2007
Revised: 4/2025
Instructions for use
RISPERDAL CONSTA ® requires close attention to these step-by-step Instructions for Use to help ensure successful administration.
SINGLE-USE DEVICE
Do not reuse.Medical devices require specific material characteristics to perform as intended. These characteristics have been verified for single use only. Any attempt to re-process the device for subsequent re-use may adversely affect the integrity of the device or lead to deterioration in performance.
Important Information
Use components provided
The components in this dose pack are specifically designed for use with RISPERDAL CONSTA ®. RISPERDAL CONSTA ® must be reconstituted only in the diluent supplied in the dose pack.
Do not substitute ANY components of the dose pack.
Do not store suspension after reconstitution
Administer dose as soon as possible after reconstitution to avoid settling.
Proper dosing
The entire contents of the vial must be administered to ensure intended dose of RISPERDAL CONSTA ®is delivered.
Dose pack contents
| Step 1 Assemble components | |||
|---|---|---|---|
| Take out dose pack | Connect vial adapter to vial | ||
|
|
|
|
|
|
Wait 30 minutes
Remove dose pack from the refrigerator and allow to sit at room temperature for at least 30 minutes before reconstituting. Do not warm any other way. |
Remove cap from vial
Flip off colored cap from vial. Wipe top of the grey stopper with an alcohol swab. Allow to air dry. Do not remove grey rubber stopper. |
Prepare vial adapter
Hold sterile blister as shown. Peel back and remove paper backing. Do not remove vial adapter from blister. Do not touch spike tip at any time. This will result in contamination. |
Connect vial adapter to vial
Place vial on a hard surface and hold by the base. Center vial adapter over the grey rubber stopper. Push vial adapter straight down onto vial top until it snaps securely into place. Do not place vial adapter on at an angle or diluent may leak upon transfer to the vial.
|
| Connect prefilled syringe to vial adapter | |||
|
|
|
|
|
|
Remove sterile blister
Keep vial vertical to prevent leakage. Hold base of vial and pull up on the sterile blister to remove. Do not shake. Do not touch exposed luer opening on vial adapter. This will result in contamination. |
Use proper grip
Hold by white collar at the tip of the syringe. Do not hold syringe by the glass barrel during assembly.
|
Remove cap
Holding the white collar, snap off the white cap. Do not twist or cut off the white cap. Do not touch syringe tip. This will result in contamination. The broken-off cap can be discarded. |
Connect syringe to vial adapter
Hold vial adapter by skirt to keep stationary. Hold syringe by white collarthen insert tip into the luer opening of the vial adapter. Do not hold the glass syringe barrel. This may cause the white collar to loosen or detach. Attach the syringe to the vial adapter with a firm clockwise twisting motion until it feels snug. Do not over-tighten. Over-tightening may cause the syringe tip to break. |
| Step 2 Reconstitute microspheres | |||
|---|---|---|---|
|
|
|
|
|
|
Inject diluent
Inject entire amount of diluent from syringe into the vial.
|
Suspend microspheres in diluent
Continuing to hold down the plunger rod, shake vigorously for at least 10 seconds, as shown. Check the suspension. When properly mixed, the suspension appears uniform, thick and milky in color. Microspheres will be visible in the liquid. Immediately proceed to the next step so suspension does not settle. |
Transfer suspension to syringe
Invert vial completely. Slowly pull plunger rod down to withdraw entire contents from the vial into the syringe. |
Remove vial adapter
Hold white collar on the syringe and unscrew from vial adapter. Tear section of the vial label at the perforation. Apply detached label to the syringe for identification purposes. Discard both vial and vial adapter appropriately. |
| Step 3 Attach needle | ||
|---|---|---|
|
|
|
|
|
Select appropriate needle
Choose needle based on injection location (gluteal or deltoid). |
Attach needle
Peel blister pouch open part way and use to grasp the base of the needle, as shown. Holding the white collar on the syringe, attach syringe to needle luer connection with a firm clockwise twisting motion until snug. Do not touch needle luer opening. This will result in contamination. |
Resuspend microspheres
Fully remove the blister pouch. Just before injection, shake syringe vigorously again, as some settling will have occurred. |
| Step 4 Inject dose | ||||
|---|---|---|---|---|
|
|
|
|
|
|
|
Remove transparent needle protector
Move the needle safety device back towards the syringe, as shown. Then hold white collar on syringe and carefully pull the transparent needle protector straight off. Do not twist transparent needle protector, as the luer connection may loosen. |
Remove air bubbles
Hold needle upright and tap gently to make any air bubbles rise to the top. Slowly and carefully press plunger rod upward to remove air. |
Inject
Immediately inject entire contents of syringe intramuscularly (IM) into the gluteal or deltoid muscle of the patient. Gluteal injection should be made into the upper-outer quadrant of the gluteal area. Do not administer intravenously. |
Secure needle in safety device
Using one hand, place needle safety device at a 45-degree angle on a hard, flat surface. Press down with a firm, quick motion until needle is fully engaged in safety device. Avoid needle stick injury: Do not use two hands. Do not intentionally disengage or mishandle the needle safety device. Do not attempt to straighten the needle or engage the safety device if the needle is bent or damaged. |
Properly dispose of needles
Check to confirm needle safety device is fully engaged. Discard in an approved sharps container. Also discard the unused needle provided in the dose pack. |
10.1 Human Experience
No cases of overdose were reported in premarketing studies with RISPERDAL CONSTA. Because RISPERDAL CONSTA is to be administered by health care professionals, the potential for overdosage by patients is low.
In premarketing experience with oral RISPERDAL, there were eight reports of acute RISPERDAL overdosage, with estimated doses ranging from 20 to 300 mg and no fatalities. In general, reported signs and symptoms were those resulting from an exaggeration of the drug's known pharmacological effects, i.e., drowsiness and sedation, tachycardia and hypotension, and extrapyramidal symptoms. One case, involving an estimated overdose of 240 mg, was associated with hyponatremia, hypokalemia, prolonged QT, and widened QRS. Another case, involving an estimated overdose of 36 mg, was associated with a seizure.
Postmarketing experience with oral RISPERDAL includes reports of acute overdose, with estimated doses of up to 360 mg. In general, the most frequently reported signs and symptoms are those resulting from an exaggeration of the drug's known pharmacological effects, i.e., drowsiness, sedation, tachycardia, hypotension, and extrapyramidal symptoms. Other adverse reactions reported since market introduction related to oral RISPERDAL overdose include prolonged QT interval and convulsions. Torsade de pointes has been reported in association with combined overdose of oral RISPERDAL and paroxetine.
12.2 Pharmacodynamics
Risperidone is a monoaminergic antagonist with high affinity (Ki of 0.12 to 7.3 nM) for the serotonin Type 2 (5HT 2), dopamine Type 2 (D 2), α1 and α2 adrenergic, and H 1 histaminergic receptors. Risperidone showed low to moderate affinity (Ki of 47 to 253 nM) for the serotonin 5HT 1C, 5HT 1D, and 5HT 1A receptors, weak affinity (Ki of 620 to 800 nM) for the dopamine D 1 and haloperidol-sensitive sigma site, and no affinity (when tested at concentrations >10 -5 M) for cholinergic muscarinic or β1 and β2 adrenergic receptors.
5.5 Metabolic Changes
Atypical antipsychotic drugs have been associated with metabolic changes that may increase cardiovascular/cerebrovascular risk. These metabolic changes include hyperglycemia, dyslipidemia, and body weight gain. While all of the drugs in the class have been shown to produce some metabolic changes, each drug has its own specific risk profile.
5.4 Tardive Dyskinesia
Tardive dyskinesia, a syndrome consisting of potentially irreversible, involuntary, dyskinetic movements, may develop in patients treated with antipsychotic drugs. Although the prevalence of the syndrome appears to be highest among the elderly, especially elderly women, it is impossible to predict which patients will develop the syndrome. Whether antipsychotic drug products differ in their potential to cause tardive dyskinesia is unknown.
The risk of developing tardive dyskinesia and the likelihood that it will become irreversible increase with the duration of treatment and the cumulative dose. The syndrome can develop after relatively brief treatment periods, even at low doses. It may also occur after discontinuation of treatment.
Tardive dyskinesia may remit, partially or completely, if antipsychotic treatment is discontinued. Antipsychotic treatment, itself, however, may suppress (or partially suppress) the signs and symptoms of the syndrome, possibly masking the underlying process. The effect that symptomatic suppression has upon the long-term course of the syndrome is unknown.
Given these considerations, RISPERDAL CONSTA should be prescribed in a manner that is most likely to minimize the occurrence of tardive dyskinesia. Chronic antipsychotic treatment should generally be reserved for patients: (1) who suffer from a chronic illness that is known to respond to antipsychotic drugs, and (2) for whom alternative, equally effective, but potentially less harmful treatments are not available or appropriate. In patients who do require chronic treatment, the lowest dose and the shortest duration of treatment producing a satisfactory clinical response. Periodically reassess the need for continued treatment.
If signs and symptoms of tardive dyskinesia appear in a patient treated with RISPERDAL CONSTA, drug discontinuation should be considered. However, some patients may require treatment with RISPERDAL CONSTA despite the presence of the syndrome.
5.6 Hyperprolactinemia
As with other drugs that antagonize dopamine D 2receptors, risperidone elevates prolactin levels and the elevation persists during chronic administration. Risperidone is associated with higher levels of prolactin elevation than other antipsychotic agents.
Hyperprolactinemia may suppress hypothalamic GnRH, resulting in reduced pituitary gonadotropin secretion. This, in turn, may inhibit reproductive function by impairing gonadal steroidogenesis in both female and male patients. Galactorrhea, amenorrhea, gynecomastia, and impotence have been reported in patients receiving prolactin-elevating compounds. Long-standing hyperprolactinemia when associated with hypogonadism may lead to decreased bone density in both female and male subjects.
Tissue culture experiments indicate that approximately one-third of human breast cancers are prolactin dependent in vitro, a factor of potential importance if the prescription of these drugs is contemplated in a patient with previously detected breast cancer. An increase in pituitary gland, mammary gland, and pancreatic islet cell neoplasia (mammary adenocarcinomas, pituitary and pancreatic adenomas) was observed in the risperidone carcinogenicity studies conducted in mice and rats [see Nonclinical Toxicology (13.1)] . Published epidemiologic studies have shown inconsistent results when exploring the potential association between hyperprolactinemia and breast cancer.
1 Indications and Usage (1 INDICATIONS AND USAGE)
12.1 Mechanism of Action
The mechanism of action of risperidone in schizophrenia is unclear. The drug's therapeutic activity in schizophrenia could be mediated through a combination of dopamine Type 2 (D 2) and serotonin Type 2 (5HT 2) receptor antagonism. The clinical effect from risperidone results from the combined concentrations of risperidone and its major active metabolite, 9-hydroxyrisperidone (paliperidone) [see Clinical Pharmacology (12.3)] . Antagonism at receptors other than D 2 and 5HT 2 may explain some of the other effects of risperidone [see Clinical Pharmacology (12.1)] .
2.8 Instructions for Use
For deltoid or gluteal intramuscular injection only
IMPORTANT RESOURCES
For additional information, visit www.risperdalconsta.com or call Janssen Pharmaceuticals, Inc. at 1-800-526-7736.
Important Information
RISPERDAL CONSTA requires close attention to these step-by-step Instructions for Use to help ensure successful administration.
Use components provided
The components in this dose pack are specifically designed for use with RISPERDAL CONSTA. RISPERDAL CONSTA must be reconstituted only in the diluent supplied in the dose pack.
Do notsubstitute ANY components of the dose pack.
Do not store suspension after reconstitution
Administer dose as soon as possible after reconstitution to avoid settling.
Proper dosing
The entire contents of the vial must be administered to ensure intended dose of RISPERDAL CONSTA is delivered.
SINGLE-USE DEVICE
Do not reuse.Medical devices require specific material characteristics to perform as intended. These characteristics have been verified for single use only. Any attempt to re-process the device for subsequent re-use may adversely affect the integrity of the device or lead to deterioration in performance.
Dose pack contents
| Step 1 Assemble components | |||
|---|---|---|---|
| Take out dose pack Connect vial adapter to vial | |||
|
|
|
|
|
|
Wait 30 minutes
Remove dose pack from the refrigerator and allow to sit at room temperature for at least 30 minutesbefore reconstituting. Do notwarm any other way. |
Remove cap from vial
Flip off colored cap from vial. Wipe top of the grey stopper with an alcohol swab. Allow to air dry. Do notremove grey rubber stopper. |
Prepare vial adapter
Hold sterile blister as shown. Peel back and remove paper backing. Do notremove vial adapter from blister. Do nottouch spike tip at any time. This will result in contamination. |
Connect vial adapter to vial
Place vial on a hard surface and hold by the base. Center vial adapter over the grey rubber stopper. Push vial adapter straight down onto vial top until it snaps securely into place. Do notplace vial adapter on at an angle or diluent may leak upon transfer to the vial.
|
| Connect prefilled syringe to vial adapter | |||
|
|
|
|
|
|
Remove sterile blister
Keep vial vertical to prevent leakage. Hold base of vial and pull up on the sterile blister to remove. Do notshake. Do nottouch exposed luer opening on vial adapter. This will result in contamination. |
Use proper grip
Hold by white collar at the tip of the syringe. Do nothold syringe by the glass barrel during assembly.
|
Remove cap
Holding the white collar, snap off the white cap. Do nottwist or cut off the white cap. Do nottouch syringe tip. This will result in contamination. The broken-off cap can be discarded. |
Connect syringe to vial adapter
Hold vial adapter by skirt to keep stationary. Hold syringe by white collarthen insert tip into the luer opening of the vial adapter. Do nothold the glass syringe barrel. This may cause the white collar to loosen or detach. Attach the syringe to the vial adapter with a firm clockwise twisting motion until it feels snug. Do notover-tighten. Over-tightening may cause the syringe tip to break. |
| Step 2 Reconstitute microspheres | |||
|---|---|---|---|
|
|
|
|
|
|
Inject diluent
Inject entire amount of diluent from syringe into the vial.
|
Suspend microspheres in diluent
Continuing to hold down the plunger rod, shake vigorously for at least 10 seconds, as shown. Check the suspension. When properly mixed, the suspension appears uniform, thick and milky in color. Microspheres will be visible in the liquid. Immediately proceed to the next step so suspension does not settle. |
Transfer suspension to syringe
Invert vial completely. Slowly pull plunger rod down to withdraw entire contents from the vial into the syringe. |
Remove vial adapter
Hold white collar on the syringe and unscrew from vial adapter. Tear section of the vial label at the perforation. Apply detached label to the syringe for identification purposes. Discard both vial and vial adapter appropriately. |
| Step 3 Attach needle | ||
|---|---|---|
|
|
|
|
|
Select appropriate needle
Choose needle based on injection location (gluteal or deltoid). |
Attach needle
Peel blister pouch open part way and use to grasp the base of the needle, as shown. Holding the white collar on the syringe, attach syringe to needle luer connection with a firm clockwise twisting motion until snug. Do nottouch needle luer opening. This will result in contamination. |
Resuspend microspheres
Fully remove the blister pouch. Just before injection, shake syringe vigorously again, as some settling will have occurred. |
| Step 4 Inject dose | ||||
|---|---|---|---|---|
|
|
|
|
|
|
|
Remove transparent needle protector
Move the needle safety device back towards the syringe, as shown. Then hold white collar on syringe and carefully pull the transparent needle protector straight off. Do nottwist transparent needle protector, as the luer connection may loosen. |
Remove air bubbles
Hold needle upright and tap gently to make any air bubbles rise to the top. Slowly and carefully press plunger rod upward to remove air. |
Inject
Immediately inject entire contents of syringe intramuscularly (IM) into the gluteal or deltoid muscle of the patient. Gluteal injection should be made into the upper-outer quadrant of the gluteal area. Do not administer intravenously. |
Secure needle in safety device
Using one hand, place needle safety device at a 45-degree angle on a hard, flat surface. Press down with a firm, quick motion until needle is fully engaged in safety device. Avoid needle stick injury: Do notuse two hands. Do notintentionally disengage or mishandle the needle safety device. Do notattempt to straighten the needle or engage the safety device if the needle is bent or damaged. |
Properly dispose of needles
Check to confirm needle safety device is fully engaged. Discard in an approved sharps container. Also discard the unused needle provided in the dose pack. |
9.1 Controlled Substance
RISPERDAL CONSTA (risperidone) is not a controlled substance.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Cerebrovascular events, including stroke, in elderly patients with dementia-related psychosis. RISPERDAL CONSTA is not approved for use in patients with dementia-related psychosis ( 5.2)
- Neuroleptic Malignant Syndrome: Manage with immediate discontinuation and close monitoring ( 5.3)
- Tardive Dyskinesia: Discontinue treatment if clinically appropriate ( 5.4)
- Metabolic Changes: Atypical antipsychotic drugs have been associated with metabolic changes that may increase cardiovascular/ cerebrovascular risk. These metabolic changes include hyperglycemia, dyslipidemia, and weight gain. (
5.5)
- Hyperglycemia and Diabetes Mellitus:Monitor patients for symptoms of hyperglycemia including polydipsia, polyuria, polyphagia, and weakness. Monitor glucose regularly in patients with diabetes or at risk for diabetes. ( 5.5)
- Dyslipidemia:Undesirable alterations have been observed in patients treated with atypical antipsychotics. ( 5.5)
- Weight Gain:Significant weight gain has been reported. Monitor weight gain. ( 5.5)
- Hyperprolactinemia: Risperidone treatment may elevate prolactin levels. Long-standing hyperprolactinemia, when associated with hypogonadism, can lead to decreased bone density in men and women. ( 5.6)
- Orthostatic hypotension: associated with dizziness, tachycardia, bradycardia, and syncope can occur, especially during initial dose titration with oral risperidone. Use caution in patients with cardiovascular disease, cerebrovascular disease, and conditions that could affect hemodynamic responses. ( 5.7)
- Leukopenia, Neutropenia, and Agranulocytosis have been reported with antipsychotics, including RISPERDAL CONSTA. Patients with history of a clinically significant low white blood cell count (WBC) or a drug-induced leukopenia/neutropenia should have their complete blood cell count (CBC) monitored frequently during the first few months of therapy and discontinuation of RISPERDAL CONSTA should be considered at the first sign of a clinically significant decline in WBC in the absence of other causative factors. ( 5.9)
- Potential for cognitive and motor impairment: has potential to impair judgment, thinking, and motor skills. Use caution when operating machinery, including automobiles. ( 5.10)
- Seizures: Use cautiously in patients with a history of seizures or with conditions that potentially lower the seizure threshold. ( 5.11)
- Dysphagia: Esophageal dysmotility and aspiration can occur. Use cautiously in patients at risk for aspiration pneumonia. ( 5.12)
- Priapism: has been reported. Severe priapism may require surgical intervention. ( 5.13)
- Avoid inadvertent administration into a blood vessel. ( 5.15)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
For patients who have never taken oral RISPERDAL, it is recommended to establish tolerability with oral RISPERDAL prior to initiating treatment with RISPERDAL CONSTA.
RISPERDAL CONSTA should be administered every 2 weeks by deep intramuscular (IM) deltoid or gluteal injection. Each injection should be administered by a health care professional using the appropriate enclosed safety needle [see Dosage and Administration (2.8)] . For deltoid administration, use the 1-inch needle alternating injections between the two arms. For gluteal administration, use the 2-inch needle alternating injections between the two buttocks. Do not administer intravenously.
5.7 Orthostatic Hypotension
RISPERDAL CONSTA may induce orthostatic hypotension associated with dizziness, tachycardia, and in some patients, syncope, especially during the initial dose-titration period with oral risperidone, probably reflecting its alpha-adrenergic antagonistic properties. Syncope was reported in 0.8% (12/1499 patients) of patients treated with RISPERDAL CONSTA in multiple-dose studies. Patients should be instructed in nonpharmacologic interventions that help to reduce the occurrence of orthostatic hypotension (e.g., sitting on the edge of the bed for several minutes before attempting to stand in the morning and slowly rising from a seated position).
RISPERDAL CONSTA should be used with particular caution in (1) patients with known cardiovascular disease (history of myocardial infarction or ischemia, heart failure, or conduction abnormalities), cerebrovascular disease, and conditions which would predispose patients to hypotension, e.g., dehydration and hypovolemia, and (2) in the elderly and patients with renal or hepatic impairment. Monitoring of orthostatic vital signs should be considered in all such patients, and a dose reduction should be considered if hypotension occurs. Clinically significant hypotension has been observed with concomitant use of oral RISPERDAL and antihypertensive medication.
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
RISPERDAL CONSTA is available in dosage strengths of 12.5 mg, 25 mg, 37.5 mg, and 50 mg risperidone. It is provided as a single-use dose pack, consisting of a vial containing the risperidone microspheres, a pre-filled syringe containing 2 mL of diluent for RISPERDAL CONSTA, a vial adapter, and two Terumo SurGuard 3 Needles for intramuscular injection (a 21 G UTW 1-inch needle with needle protection device for deltoid administration and a 20 G TW 2-inch needle with needle protection device for gluteal administration).
6.2 Postmarketing Experience
The following adverse reactions have been identified during postapproval use of risperidone; because these reactions are reported voluntarily from a population of uncertain size, it is not possible to reliably estimate their frequency: agranulocytosis, alopecia, anaphylactic reaction, angioedema, atrial fibrillation, blood cholesterol increased, blood triglycerides increased, catatonia, diabetes mellitus, diabetic ketoacidosis in patients with impaired glucose metabolism, drug withdrawal syndrome neonatal, dysgeusia, hypoglycemia, hypothermia, ileus, inappropriate antidiuretic hormone secretion, intestinal obstruction, jaundice, mania, pancreatitis, priapism, QT prolongation, sleep apnea syndrome, somnambulism, Stevens-Johnson syndrome and toxic epidermal necrolysis (SJS/TEN), thrombocytopenia, thrombotic thrombocytopenic purpura, urinary retention, and water intoxication. In addition, the following adverse reactions have been observed during postapproval use of RISPERDAL CONSTA: cerebrovascular disorders, including cerebrovascular accidents, and diabetes mellitus aggravated.
Retinal artery occlusion after injection of RISPERDAL CONSTA has been reported during postmarketing surveillance. This has been reported in the presence of abnormal arteriovenous anastomosis.
Serious injection site reactions including abscess, cellulitis, cyst, hematoma, necrosis, nodule, and ulcer have been reported with RISPERDAL CONSTA during postmarketing surveillance. Isolated cases required surgical intervention.
Very rarely, cases of anaphylactic reaction after injection with RISPERDAL CONSTA have been reported during postmarketing experience in patients who have previously tolerated oral risperidone.
Postmarketing cases of extrapyramidal symptoms (dystonia and dyskinesia) have been reported in patients concomitantly taking methylphenidate and risperidone when there was an increase or decrease in dosage, initiation, or discontinuation of either or both medications.
10.2 Management of Overdosage
In case of acute overdosage, establish and maintain an airway and ensure adequate oxygenation and ventilation. Cardiovascular monitoring should commence immediately and should include continuous electrocardiographic monitoring to detect possible arrhythmias. If antiarrhythmic therapy is administered, disopyramide, procainamide, and quinidine carry a theoretical hazard of QT prolonging effects that might be additive to those of risperidone. Similarly, it is reasonable to expect that the alpha-blocking properties of bretylium might be additive to those of risperidone, resulting in problematic hypotension.
There is no specific antidote to risperidone. Therefore, appropriate supportive measures should be instituted. The possibility of multiple drug involvement should be considered. Hypotension and circulatory collapse should be treated with appropriate measures, such as intravenous fluids and/or sympathomimetic agents (epinephrine and dopamine should not be used, since beta stimulation may worsen hypotension in the setting of risperidone-induced alpha blockade). In cases of severe extrapyramidal symptoms, anticholinergic medication should be administered. Close medical supervision and monitoring should continue until the patient recovers.
7.5 Cimetidine and Ranitidine
Cimetidine and ranitidine increased the bioavailability of oral risperidone by 64% and 26%, respectively. However, cimetidine did not affect the AUC of risperidone and 9-hydroxyrisperidone combined, whereas ranitidine increased the AUC of risperidone and 9-hydroxyrisperidone combined by 20%.
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
- Pregnancy: May cause extrapyramidal and/or withdrawal symptoms in neonates with third trimester exposure. ( 8.1)
- Renal or Hepatic Impairment: dose appropriately with oral RISPERDAL prior to initiating treatment with RISPERDAL CONSTA. A lower starting dose of RISPERDAL CONSTA of 12.5 mg may be appropriate in some patients. ( 2.4)
- Pediatric Use: safety and effectiveness not established in patients less than 18 years of age. ( 8.4)
- Elderly: dosing for otherwise healthy elderly patients is the same as for healthy nonelderly. Elderly may be more predisposed to orthostatic effects than nonelderly. ( 8.5)
2.3 General Dosing Information
A lower initial dose of 12.5 mg may be appropriate when clinical factors warrant dose adjustment, such as in patients with hepatic or renal impairment, for certain drug interactions that increase risperidone plasma concentrations [see Drug Interactions (7.11)] or in patients who have a history of poor tolerability to psychotropic medications. The efficacy of the 12.5 mg dose has not been investigated in clinical trials.
Oral RISPERDAL (or another antipsychotic medication) should be given with the first injection of RISPERDAL CONSTA and continued for 3 weeks (and then discontinued) to ensure that adequate therapeutic plasma concentrations are maintained prior to the main release phase of risperidone from the injection site [see Clinical Pharmacology (12.3)] .
Upward dose adjustment should not be made more frequently than every 4 weeks. The clinical effects of this dose adjustment should not be anticipated earlier than 3 weeks after the first injection with the higher dose.
In patients with clinical factors such as hepatic or renal impairment or certain drug interactions that increase risperidone plasma concentrations [see Drug Interactions (7.11)] , dose reduction as low as 12.5 mg may be appropriate. The efficacy of the 12.5 mg dose has not been investigated in clinical trials.
Do not combine two different dose strengths of RISPERDAL CONSTA in a single administration.
8.6 Renal Or Hepatic Impairment (8.6 Renal or Hepatic Impairment)
In patients with renal or hepatic impairment, carefully titrate with oral risperidone prior to initiating treatment with RISPERDAL CONSTA [see Dosage and Administration (2.4)].
Patients with renal impairment may have less ability to eliminate risperidone than patients with normal renal function. Patients with impaired hepatic function may have an increase in the free fraction of risperidone, possibly resulting in an enhanced effect [see Clinical Pharmacology (12.3)] .
5.14 Body Temperature Regulation
Disruption of body temperature regulation has been attributed to antipsychotic agents. Both hyperthermia and hypothermia have been reported in association with oral RISPERDAL or RISPERDAL CONSTA use. Caution is advised when prescribing RISPERDAL CONSTA for patients who will be exposed to temperature extremes.
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Physicians are advised to discuss the following issues with patients for whom they prescribe RISPERDAL CONSTA.
7.14 Drugs Metabolized By Cyp 2d6 (7.14 Drugs Metabolized by CYP 2D6)
In vitrostudies indicate that risperidone is a relatively weak inhibitor of CYP 2D6. Therefore, RISPERDAL CONSTA is not expected to substantially inhibit the clearance of drugs that are metabolized by this enzymatic pathway. In drug interaction studies, oral RISPERDAL did not significantly affect the pharmacokinetics of donepezil and galantamine, which are metabolized by CYP 2D6.
5.3 Neuroleptic Malignant Syndrome
Neuroleptic Malignant Syndrome (NMS), a potentially fatal symptom complex, has been reported in association with antipsychotic drugs. Clinical manifestations of NMS are hyperpyrexia, muscle rigidity, altered mental status including delirium, and autonomic instability (irregular pulse or blood pressure, tachycardia, diaphoresis, and cardiac dysrhythmia). Additional signs may include elevated creatine phosphokinase, myoglobinuria (rhabdomyolysis), and acute renal failure.
If NMS is suspected, immediately discontinue RISPERDAL CONSTA and provide symptomatic treatment and monitoring.
7.2 Drugs With Hypotensive Effects (7.2 Drugs with Hypotensive Effects)
Because of its potential for inducing hypotension, RISPERDAL CONSTA may enhance the hypotensive effects of other therapeutic agents with this potential.
7.3 Levodopa and Dopamine Agonists
RISPERDAL CONSTA may antagonize the effects of levodopa and dopamine agonists.
14.2 Bipolar Disorder Monotherapy (14.2 Bipolar Disorder - Monotherapy)
The effectiveness of RISPERDAL CONSTA for the maintenance treatment of Bipolar I Disorder was established in a multicenter, double-blind, placebo-controlled study of adult patients who met DSM-IV criteria for Bipolar Disorder Type I, who were stable on medications or experiencing an acute manic or mixed episode.
A total of 501 patients were treated during a 26-week open-label period with RISPERDAL CONSTA (starting dose of 25 mg, and titrated, if deemed clinically desirable, to 37.5 mg or 50 mg; in patients not tolerating the 25 mg dose, the dose could be reduced to 12.5 mg). In the open-label phase, 303 (60%) patients were judged to be stable and were randomized to double-blind treatment with either the same dose of RISPERDAL CONSTA or placebo and monitored for relapse. The primary endpoint was time to relapse to any mood episode (depression, mania, hypomania, or mixed).
Time to relapse was delayed in patients receiving RISPERDAL CONSTA monotherapy as compared to placebo. The majority of relapses were due to manic rather than depressive symptoms. Based on their bipolar disorder history, subjects entering this study had had, on average, more manic episodes than depressive episodes.
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
RISPERDAL CONSTA ®(risperidone) is available in dosage strengths of 12.5 mg, 25 mg, 37.5 mg, or 50 mg risperidone. It is provided as a single-use dose pack, consisting of a vial containing the risperidone microspheres, a pre-filled syringe containing 2 mL of diluent for RISPERDAL CONSTA, a vial adapter, and two Terumo SurGuard ®3 Needles for intramuscular injection (a 21 G UTW 1-inch needle with needle protection device for deltoid administration and a 20 G TW 2-inch needle with needle protection device for gluteal administration).
12.5-mg vial/kit (NDC 50458-309-11): 41 mg (equivalent to 12.5 mg of risperidone) of a white to off-white powder provided in a vial with a violet flip-off cap (NDC 50458-309-01).
25-mg vial/kit (NDC 50458-306-11): 78 mg (equivalent to 25 mg of risperidone) of a white to off-white powder provided in a vial with a pink flip-off cap (NDC 50458-306-01).
37.5-mg vial/kit (NDC 50458-307-11): 116 mg (equivalent to 37.5 mg of risperidone) of a white to off-white powder provided in a vial with a green flip-off cap (NDC 50458-307-01).
50-mg vial/kit (NDC 50458-308-11): 152 mg (equivalent to 50 mg of risperidone) of a white to off-white powder provided in a vial with a blue flip-off cap (NDC 50458-308-01).
7.1 Centrally Acting Drugs and Alcohol (7.1 Centrally-Acting Drugs and Alcohol)
Given the primary CNS effects of risperidone, caution should be used when RISPERDAL CONSTA is administered in combination with other centrally-acting drugs or alcohol.
2.6 Switching From Other Antipsychotics (2.6 Switching from Other Antipsychotics)
There are no systematically collected data to specifically address switching patients from other antipsychotics to RISPERDAL CONSTA, or concerning concomitant administration with other antipsychotics. Previous antipsychotics should be continued for 3 weeks after the first injection of RISPERDAL CONSTA to ensure that therapeutic concentrations are maintained until the main release phase of risperidone from the injection site has begun [see Clinical Pharmacology (12.3)] . For patients who have never taken oral RISPERDAL, it is recommended to establish tolerability with oral RISPERDAL prior to initiating treatment with RISPERDAL CONSTA. As recommended with other antipsychotic medications, the need for continuing existing EPS medication should be re-evaluated periodically.
5.16 Osteodystrophy and Tumors in Animals
RISPERDAL CONSTA produced osteodystrophy in male and female rats in a 1-year toxicity study and a 2-year carcinogenicity study at a dose of 40 mg/kg administered IM every 2 weeks.
RISPERDAL CONSTA produced renal tubular tumors (adenoma, adenocarcinoma) and adrenomedullary pheochromocytomas in male rats in the 2-year carcinogenicity study at 40 mg/kg administered IM every 2 weeks. In addition, RISPERDAL CONSTA produced an increase in a marker of cellular proliferation in renal tissue in males in the 1-year toxicity study and in renal tumor-bearing males in the 2-year carcinogenicity study at 40 mg/kg administered IM every 2 weeks. (Cellular proliferation was not measured at the low dose or in females in either study.)
The effect dose for osteodystrophy and the tumor findings is 8 times the IM maximum recommended human dose (MRHD) (50 mg) on a mg/m 2basis and is associated with a plasma exposure (AUC) 2 times the expected plasma exposure (AUC) at the IM MRHD. The no-effect dose for these findings was 5 mg/kg (equal to the IM MRHD on a mg/m 2basis). Plasma exposure (AUC) at the no-effect dose was one third the expected plasma exposure (AUC) at the IM MRHD.
Neither the renal or adrenal tumors, nor osteodystrophy, were seen in studies of orally administered risperidone.Osteodystrophy was not observed in dogs at doses up to 14 times (based on AUC) the IM MRHD in a 1-year toxicity study.
The renal tubular and adrenomedullary tumors in male rats and other tumor findings are described in more detail in Section 13.1 (Carcinogenicity, Mutagenesis, Impairment of Fertility).
The relevance of these findings to human risk is unknown.
14.3 Bipolar Disorder Adjunctive Therapy (14.3 Bipolar Disorder - Adjunctive Therapy)
The effectiveness of RISPERDAL CONSTA as an adjunct to treatment with lithium or valproate for the maintenance treatment of Bipolar Disorder was established in a multi-center, randomized, double-blind, placebo-controlled study of adult patients who met DSM-IV criteria for Bipolar Disorder Type I and who experienced at least 4 episodes of mood disorder requiring psychiatric/clinical intervention in the previous 12 months, including at least 2 episodes in the 6 months prior to the start of the study.
A total of 240 patients were treated during a 16-week open-label period with RISPERDAL CONSTA (starting dose of 25 mg, and titrated, if deemed clinically desirable, to 37.5 mg or 50 mg), as adjunctive therapy in addition to continuing their treatment as usual for their bipolar disorder, which consisted of mood stabilizers (primarily lithium and valproate), antidepressants, and/or anxiolytics. All oral antipsychotics were discontinued after the first three weeks of the initial RISPERDAL CONSTA injection. In the open-label phase, 124 (51.7%) were judged to be stable for at least the last 4 weeks and were randomized to double-blind treatment with either the same dose of RISPERDAL CONSTA or placebo in addition to continuing their treatment as usual and monitored for relapse during a 52-week period. The primary endpoint was time to relapse to any new mood episode (depression, mania, hypomania, or mixed).
Time to relapse was delayed in patients receiving adjunctive therapy with RISPERDAL CONSTA as compared to placebo. The relapse types were about half depressive and half manic or mixed episodes.
Principal Display Panel 25 Mg Kit Carton (PRINCIPAL DISPLAY PANEL - 25 mg Kit Carton)
25 mg
Dose Pack
Store Package
in Refrigerator
(see bottom panel for
storage conditions)
Rx only
Single use only
NDC50458-306-11
Risperdal
CONSTA
®
risperiDONE Long-Acting Injection
DOSE PACK CONTENTS:
- One 25 mg vial of risperidone extended-release microspheres for injection
- One pre-filled syringe containing 2 mL of diluent
- One Vial Adapter
- One 21-gauge UTW 1" Terumo SurGuard ®3 safety needle for deltoid injection
- One 20-gauge TW 2" Terumo SurGuard ®3 safety needle for gluteal injection
Do not substitute any components of the dose pack.
For deltoid or gluteal intramuscular injection only
Each injection should be administered by a healthcare professional.
PLEASE READ COMPLETE INSTRUCTIONS PRIOR TO USE.
Principal Display Panel 50 Mg Kit Carton (PRINCIPAL DISPLAY PANEL - 50 mg Kit Carton)
50 mg
Dose Pack
Store Package
in Refrigerator
(see bottom panel for
storage conditions)
Rx only
Single use only
NDC50458-308-11
Risperdal
CONSTA
®
risperiDONE Long-Acting Injection
DOSE PACK CONTENTS:
- One 50 mg vial of risperidone extended-release microspheres for injection
- One pre-filled syringe containing 2 mL of diluent
- One Vial Adapter
- One 21-gauge UTW 1" Terumo SurGuard ®3 safety needle for deltoid injection
- One 20-gauge TW 2" Terumo SurGuard ®3 safety needle for gluteal injection
Do not substitute any components of the dose pack.
For deltoid or gluteal intramuscular injection only
Each injection should be administered by a healthcare professional.
PLEASE READ COMPLETE INSTRUCTIONS PRIOR TO USE.
Principal Display Panel 12.5 Mg Kit Carton (PRINCIPAL DISPLAY PANEL - 12.5 mg Kit Carton)
12.5 mg
Dose Pack
Store Package
in Refrigerator
(see bottom panel for
storage conditions)
Rx only
Single use only
NDC50458-309-11
Risperdal
CONSTA
®
risperiDONE Long-Acting Injection
DOSE PACK CONTENTS:
- One 12.5 mg vial of risperidone extended-release microspheres for injection
- One pre-filled syringe containing 2 mL of diluent
- One Vial Adapter
- One 21-gauge UTW 1" Terumo SurGuard ®3 safety needle for deltoid injection
- One 20-gauge TW 2" Terumo SurGuard ®3 safety needle for gluteal injection
Do not substitute any components of the dose pack.
For deltoid or gluteal intramuscular injection only
Each injection should be administered by a healthcare professional.
PLEASE READ COMPLETE INSTRUCTIONS PRIOR TO USE.
Principal Display Panel 37.5 Mg Kit Carton (PRINCIPAL DISPLAY PANEL - 37.5 mg Kit Carton)
37.5 mg
Dose Pack
Store Package
in Refrigerator
(see bottom panel for
storage conditions)
Rx only
Single use only
NDC50458-307-11
Risperdal
CONSTA
®
risperiDONE Long-Acting Injection
DOSE PACK CONTENTS:
- One 37.5 mg vial of risperidone extended-release microspheres for injection
- One pre-filled syringe containing 2 mL of diluent
- One Vial Adapter
- One 21-gauge UTW 1" Terumo SurGuard ®3 safety needle for deltoid injection
- One 20-gauge TW 2" Terumo SurGuard ®3 safety needle for gluteal injection
Do not substitute any components of the dose pack.
For deltoid or gluteal intramuscular injection only
Each injection should be administered by a healthcare professional.
PLEASE READ COMPLETE INSTRUCTIONS PRIOR TO USE.
5.10 Potential for Cognitive and Motor Impairment
Somnolence was reported by 5% of patients treated with RISPERDAL CONSTA in multiple-dose trials. Since risperidone has the potential to impair judgment, thinking, or motor skills, patients should be cautioned about operating hazardous machinery, including automobiles, until they are reasonably certain that treatment with RISPERDAL CONSTA does not affect them adversely.
7.13 Carbamazepine and Other Cyp 3a4 Enzyme Inducers (7.13 Carbamazepine and Other CYP 3A4 Enzyme Inducers)
Carbamazepine co-administration with oral RISPERDAL decreased the steady-state plasma concentrations of risperidone and 9-hydroxyrisperidone by about 50%. Plasma concentrations of carbamazepine did not appear to be affected. Co-administration of other known CYP 3A4 enzyme inducers (e.g., phenytoin, rifampin, and phenobarbital) with risperidone may cause similar decreases in the combined plasma concentrations of risperidone and 9-hydroxyrisperidone, which could lead to decreased efficacy of RISPERDAL CONSTA treatment. At the initiation of therapy with carbamazepine or other known hepatic enzyme inducers, patients should be closely monitored during the first 4–8 weeks, since the dose of RISPERDAL CONSTA may need to be adjusted. A dose increase, or additional oral RISPERDAL, may need to be considered. On discontinuation of carbamazepine or other CYP 3A4 hepatic enzyme inducers, the dosage of RISPERDAL CONSTA should be re-evaluated and, if necessary, decreased. Patients may be placed on a lower dose of RISPERDAL CONSTA between 2 to 4 weeks before the planned discontinuation of carbamazepine or other CYP 3A4 enzyme inducers to adjust for the expected increase in plasma concentrations of risperidone plus 9-hydroxyrisperidone. For patients treated with the recommended dose of 25 mg RISPERDAL CONSTA and discontinuing from carbamazepine or other CYP 3A4 enzyme inducers, it is recommended to continue treatment with the 25-mg dose unless clinical judgment necessitates lowering the RISPERDAL CONSTA dose to 12.5 mg or necessitates interruption of RISPERDAL CONSTA treatment. The efficacy of the 12.5 mg dose has not been investigated in clinical trials. [see also Dosage and Administration (2.5)]
7.12 Drugs That Inhibit Cyp 2d6 and Other Cyp Isozymes (7.12 Drugs That Inhibit CYP 2D6 and Other CYP Isozymes)
Risperidone is metabolized to 9-hydroxyrisperidone by CYP 2D6, an enzyme that is polymorphic in the population and that can be inhibited by a variety of psychotropic and other drugs [see Clinical Pharmacology (12.3)] . Drug interactions that reduce the metabolism of risperidone to 9-hydroxyrisperidone would increase the plasma concentrations of risperidone and lower the concentrations of 9-hydroxyrisperidone. Analysis of clinical studies involving a modest number of poor metabolizers (n≅70 patients) does not suggest that poor and extensive metabolizers have different rates of adverse effects. No comparison of effectiveness in the two groups has been made.
In vitrostudies showed that drugs metabolized by other CYP isozymes, including 1A1, 1A2, 2C9, 2C19, and 3A4, are only weak inhibitors of risperidone metabolism.
8.7 Patients With Parkinson's Disease Or Lewy Body Dementia (8.7 Patients with Parkinson's Disease or Lewy Body Dementia)
Patients with Parkinson's Disease or Dementia with Lewy Bodies can experience increased sensitivity to RISPERDAL CONSTA. Manifestations can include confusion, obtundation, postural instability with frequent falls, extrapyramidal symptoms, and clinical features consistent with neuroleptic malignant syndrome.
2.5 Reinitiation of Treatment in Patients Previously Discontinued
There are no data to specifically address reinitiation of treatment. When restarting patients who have had an interval off treatment with RISPERDAL CONSTA, supplementation with oral RISPERDAL (or another antipsychotic medication) should be administered.
2.7 Co Administration of Risperdal Consta With Certain Other Medications (2.7 Co-Administration of RISPERDAL CONSTA with Certain Other Medications)
Co-administration of carbamazepine and other CYP 3A4 enzyme inducers (e.g., phenytoin, rifampin, phenobarbital) with risperidone would be expected to cause decreases in the plasma concentrations of the sum of risperidone and 9-hydroxyrisperidone combined, which could lead to decreased efficacy of RISPERDAL CONSTA treatment. The dose of risperidone needs to be titrated accordingly for patients receiving these enzyme inducers, especially during initiation or discontinuation of therapy with these inducers [see Drug Interactions (7.11)] . At the initiation of therapy with carbamazepine or other known CYP 3A4 hepatic enzyme inducers, patients should be closely monitored during the first 4–8 weeks, since the dose of RISPERDAL CONSTA may need to be adjusted. A dose increase, or additional oral RISPERDAL, may need to be considered. On discontinuation of carbamazepine or other CYP 3A4 hepatic enzyme inducers, the dosage of RISPERDAL CONSTA should be re-evaluated and, if necessary, decreased. Patients may be placed on a lower dose of RISPERDAL CONSTA between 2 to 4 weeks before the planned discontinuation of carbamazepine or other CYP 3A4 inducers to adjust for the expected increase in plasma concentrations of risperidone plus 9-hydroxyrisperidone. For patients treated with the recommended dose of 25 mg RISPERDAL CONSTA and discontinuing from carbamazepine or other CYP3A4 enzyme inducers, it is recommended to continue treatment with the 25-mg dose unless clinical judgment necessitates lowering the RISPERDAL CONSTA dose to 12.5 mg or necessitates interruption of RISPERDAL CONSTA treatment. The efficacy of the 12.5 mg dose has not been investigated in clinical trials.
Fluoxetine and paroxetine, CYP 2D6 inhibitors, have been shown to increase the plasma concentration of risperidone 2.5–2.8 fold and 3–9 fold respectively. Fluoxetine did not affect the plasma concentration of 9-hydroxyrisperidone. Paroxetine lowered the concentration of 9-hydroxyrisperidone by about 10%. The dose of risperidone needs to be titrated accordingly when fluoxetine or paroxetine is co-administered. When either concomitant fluoxetine or paroxetine is initiated or discontinued, the physician should re-evaluate the dose of RISPERDAL CONSTA. When initiation of fluoxetine or paroxetine is considered, patients may be placed on a lower dose of RISPERDAL CONSTA between 2 to 4 weeks before the planned start of fluoxetine or paroxetine therapy to adjust for the expected increase in plasma concentrations of risperidone. When fluoxetine or paroxetine is initiated in patients receiving the recommended dose of 25 mg RISPERDAL CONSTA, it is recommended to continue treatment with the 25 mg dose unless clinical judgment necessitates lowering the RISPERDAL CONSTA dose to 12.5 mg or necessitates interruption of RISPERDAL CONSTA treatment. When RISPERDAL CONSTA is initiated in patients already receiving fluoxetine or paroxetine, a starting dose of 12.5 mg can be considered. The efficacy of the 12.5 mg dose has not been investigated in clinical trials. The effects of discontinuation of concomitant fluoxetine or paroxetine therapy on the pharmacokinetics of risperidone and 9-hydroxyrisperidone have not been studied. [see Drug Interactions (7.11)] .
5.1 Increased Mortality in Elderly Patients With Dementia Related Psychosis (5.1 Increased Mortality in Elderly Patients with Dementia-Related Psychosis)
Elderly patients with dementia-related psychosis treated with antipsychotic drugs are at an increased risk of death. Analyses of 17 placebo-controlled trials (modal duration of 10 weeks), largely in patients taking atypical antipsychotic drugs, revealed a risk of death in drug-treated patients of between 1.6 to 1.7 times the risk of death in placebo-treated patients. Over the course of a typical 10-week controlled trial, the rate of death in drug-treated patients was about 4.5%, compared to a rate of about 2.6% in the placebo group. Although the causes of death were varied, most of the deaths appeared to be either cardiovascular (e.g., heart failure, sudden death) or infectious (e.g., pneumonia) in nature. Observational studies suggest that, similar to atypical antipsychotic drugs, treatment with conventional antipsychotic drugs may increase mortality. The extent to which the findings of increased mortality in observational studies may be attributed to the antipsychotic drug as opposed to some characteristic(s) of the patients is not clear.
RISPERDAL CONSTA (risperidone) is not approved for the treatment of dementia-related psychosis [see Boxed Warning] .
Warning: Increased Mortality in Elderly Patients With Dementia Related Psychosis (WARNING: INCREASED MORTALITY IN ELDERLY PATIENTS WITH DEMENTIA-RELATED PSYCHOSIS)
Elderly patients with dementia-related psychosis treated with antipsychotic drugs are at an increased risk of death. RISPERDAL CONSTA is not approved for the treatment of patients with dementia-related psychosis. [see Warnings and Precautions (5.1)]
5.2 Cerebrovascular Adverse Events, Including Stroke, in Elderly Patients With Dementia Related Psychosis (5.2 Cerebrovascular Adverse Events, Including Stroke, in Elderly Patients with Dementia-Related Psychosis)
Cerebrovascular adverse events (e.g., stroke, transient ischemic attack), including fatalities, were reported in patients (mean age 85 years; range 73–97) in trials of oral risperidone in elderly patients with dementia-related psychosis. In placebo-controlled trials, there was a significantly higher incidence of cerebrovascular adverse events in patients treated with oral risperidone compared to patients treated with placebo. RISPERDAL CONSTA is not approved for the treatment of patients with dementia-related psychosis. [See also Boxed Warning and Warnings and Precautions (5.1)]
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:46:41.033606 · Updated: 2026-03-14T22:49:12.025925