These Highlights Do Not Include All The Information Needed To Use Tetracaine Hydrochloride Ophthalmic Solution Safely And Effectively. See Full Prescribing Information For Tetracaine Hydrochloride Ophthalmic Solution
bab63a18-3069-48a1-896e-433725f1dbaf
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Warnings and Precautions (5.4) 2/2022
Indications and Usage
Tetracaine hydrochloride ophthalmic solution, 0.5% is indicated for procedures requiring a rapid and short-acting topical ophthalmic anesthetic.
Dosage and Administration
One drop topically in the eye(s) as needed.
Warnings and Precautions
Do not use intracamerally since use may damage corneal endothelial cells. (5.1) Prolonged use or abuse may lead to corneal epithelial toxicity and may manifest as epithelial defects which may progress to permanent corneal damage. (5.2) Patients should not touch the eye for at least 10-20 minutes after using anesthetic as accidental injuries can occur due to insensitivity of the eye. (5.3) For Administration by Healthcare Provider : Tetracaine Hydrochloride Ophthalmic Solution, USP 0.5% is not intended for patient self- administration. (5.4)
Contraindications
Tetracaine hydrochloride ophthalmic solution, 0.5% should not be used in patients with a history of hypersensitivity to any component of this preparation.
Adverse Reactions
The following serious ocular adverse reactions are described elsewhere in the labeling: Corneal Injury with Intracameral Use [ see Warnings and Precautions (5.1)] Corneal Toxicity [ see Warnings and Precautions ( 5.2)] Corneal Injury Due to Insensitivity [ see Warnings and Precautions ( 5.3)] The following adverse reactions have been identified following use of tetracaine hydrochloride ophthalmic solution, 0.5%. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure. Ocular Adverse Reactions Transient stinging, burning, and conjunctival redness, eye irritation, eye pain, ocular discomfort.
Storage and Handling
Tetracaine hydrochloride ophthalmic solution, USP 0.5% is supplied as a sterile, aqueous, topical ophthalmic solution in a white LDPE bottle, plugged with white LDPE nozzle and capped with white HDPE cap in the following sizes: NDC 70069-596-01, 5 mL fill in 5 mL Bottle NDC 70069-597-01, 15 mL fill in 15 mL Bottle After opening, this product can be used until the expiration date stamped on the bottle. Storage: Store at 15° to 25°C (59° to 77°F). Protect from light. Do not use if solution contains crystals, cloudy, or discolored.
How Supplied
Tetracaine hydrochloride ophthalmic solution, USP 0.5% is supplied as a sterile, aqueous, topical ophthalmic solution in a white LDPE bottle, plugged with white LDPE nozzle and capped with white HDPE cap in the following sizes: NDC 70069-596-01, 5 mL fill in 5 mL Bottle NDC 70069-597-01, 15 mL fill in 15 mL Bottle After opening, this product can be used until the expiration date stamped on the bottle. Storage: Store at 15° to 25°C (59° to 77°F). Protect from light. Do not use if solution contains crystals, cloudy, or discolored.
Medication Information
Recent Major Changes
| Warnings and Precautions (5.4) |
2/2022 |
Warnings and Precautions
Do not use intracamerally since use may damage corneal endothelial cells. (5.1) Prolonged use or abuse may lead to corneal epithelial toxicity and may manifest as epithelial defects which may progress to permanent corneal damage. (5.2) Patients should not touch the eye for at least 10-20 minutes after using anesthetic as accidental injuries can occur due to insensitivity of the eye. (5.3) For Administration by Healthcare Provider : Tetracaine Hydrochloride Ophthalmic Solution, USP 0.5% is not intended for patient self- administration. (5.4)
Indications and Usage
Tetracaine hydrochloride ophthalmic solution, 0.5% is indicated for procedures requiring a rapid and short-acting topical ophthalmic anesthetic.
Dosage and Administration
One drop topically in the eye(s) as needed.
Contraindications
Tetracaine hydrochloride ophthalmic solution, 0.5% should not be used in patients with a history of hypersensitivity to any component of this preparation.
Adverse Reactions
The following serious ocular adverse reactions are described elsewhere in the labeling: Corneal Injury with Intracameral Use [ see Warnings and Precautions (5.1)] Corneal Toxicity [ see Warnings and Precautions ( 5.2)] Corneal Injury Due to Insensitivity [ see Warnings and Precautions ( 5.3)] The following adverse reactions have been identified following use of tetracaine hydrochloride ophthalmic solution, 0.5%. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure. Ocular Adverse Reactions Transient stinging, burning, and conjunctival redness, eye irritation, eye pain, ocular discomfort.
Storage and Handling
Tetracaine hydrochloride ophthalmic solution, USP 0.5% is supplied as a sterile, aqueous, topical ophthalmic solution in a white LDPE bottle, plugged with white LDPE nozzle and capped with white HDPE cap in the following sizes: NDC 70069-596-01, 5 mL fill in 5 mL Bottle NDC 70069-597-01, 15 mL fill in 15 mL Bottle After opening, this product can be used until the expiration date stamped on the bottle. Storage: Store at 15° to 25°C (59° to 77°F). Protect from light. Do not use if solution contains crystals, cloudy, or discolored.
How Supplied
Tetracaine hydrochloride ophthalmic solution, USP 0.5% is supplied as a sterile, aqueous, topical ophthalmic solution in a white LDPE bottle, plugged with white LDPE nozzle and capped with white HDPE cap in the following sizes: NDC 70069-596-01, 5 mL fill in 5 mL Bottle NDC 70069-597-01, 15 mL fill in 15 mL Bottle After opening, this product can be used until the expiration date stamped on the bottle. Storage: Store at 15° to 25°C (59° to 77°F). Protect from light. Do not use if solution contains crystals, cloudy, or discolored.
Description
Warnings and Precautions (5.4) 2/2022
10 Overdosage
Prolonged use of a topical ocular anesthetic including tetracaine hydrochloride ophthalmic solution, 0.5% may produce permanent corneal opacification and ulceration with accompanying visual loss.
8.1 Pregnancy
Risk Summary
There are no adequate and well-controlled studies with tetracaine hydrochloride ophthalmic solution, 0.5% in pregnant women. Animal developmental and reproductive toxicity studies with tetracaine hydrochloride have not been reported in the published literature.
8.2 Lactation
Risk Summary
There are no data to assess whether tetracaine hydrochloride ophthalmic solution, 0.5% is excreted in human milk or to assess its effects on milk production/excretion. The developmental and health benefits of breastfeeding should be considered along with the mother's clinical need for tetracaine hydrochloride ophthalmic solution, 0.5% and any potential adverse effects on the breastfed child from tetracaine hydrochloride ophthalmic solution, 0.5%.
11 Description
Tetracaine hydrochloride ophthalmic solution, USP 0.5% is a sterile, clear, colorless, topical local anesthetic for ophthalmic use only containing tetracaine hydrochloride as the active pharmaceutical ingredient.
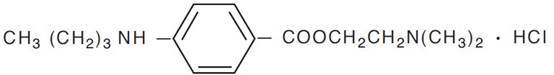
Tetracaine hydrochloride is chemically designated as benzoic acid, 4-(butylamino)-, 2-(dimethylamino) ethyl ester, monohydrochloride. Its chemical formula is C15H24N2O2 ● HCl and it is represented by the chemical structure:
Tetracaine hydrochloride is a white crystalline powder with a molecular weight of 300.82
Active ingredient: tetracaine hydrochloride 0.5% w/v (equivalent to 0.44% w/v tetracaine)
Preservative: chlorobutanol 0.4%
Inactive ingredients: boric acid, potassium chloride, edetate disodium dihydrate, water for injection. Sodium hydroxide and/or hydrochloric acid may be added to adjust pH (3.7 – 6.0).
Spl Unclassified
Manufactured for:
Somerset Therapeutics, LLC
Somerset, NJ 08873
Made in India
Code No.: KR/DRUGS/KTK/28/289/97
ST-TEC/P/00
1200992
8.4 Pediatric Use
Safety of tetracaine hydrochloride ophthalmic solution, 0.5% in the pediatric population has been demonstrated in clinical trials. Efficacy of tetracaine hydrochloride ophthalmic solution, 0.5% for use in pediatric patients has been extrapolated from adequate and well controlled clinical trials in the adult population.
8.5 Geriatric Use
No overall differences in safety or effectiveness of tetracaine hydrochloride ophthalmic solution, 0.5% have been observed between elderly and younger patients.
14 Clinical Studies
Topical administration of tetracaine hydrochloride ophthalmic solution, 0.5% results in localized temporary anesthesia. The maximum effect is achieved within 10–20 seconds after instillation, with efficacy lasting 10–20 minutes. Duration of effect can be extended with repeated dosing [ see Warnings and Precautions ( 5.2) and Overdosage (10)].
4 Contraindications
Tetracaine hydrochloride ophthalmic solution, 0.5% should not be used in patients with a history of hypersensitivity to any component of this preparation.
6 Adverse Reactions
The following serious ocular adverse reactions are described elsewhere in the labeling:
- Corneal Injury with Intracameral Use [ see Warnings and Precautions (5.1)]
- Corneal Toxicity [ see Warnings and Precautions ( 5.2)]
- Corneal Injury Due to Insensitivity [see Warnings and Precautions ( 5.3)]
The following adverse reactions have been identified following use of tetracaine hydrochloride ophthalmic solution, 0.5%. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Ocular Adverse Reactions
Transient stinging, burning, and conjunctival redness, eye irritation, eye pain, ocular discomfort.
5.2 Corneal Toxicity
Prolonged use or abuse may lead to corneal epithelial toxicity and may manifest as epithelial defects which may progress to permanent corneal damage.
12.3 Pharmacokinetics
The systemic exposure to tetracaine following topical ocular administration of tetracaine hydrochloride ophthalmic solution, 0.5% has not been studied. Tetracaine hydrochloride is metabolized by plasma pseudocholinesterases and nonspecific esterases in ocular tissues.
1 Indications and Usage
Tetracaine hydrochloride ophthalmic solution, 0.5% is indicated for procedures requiring a rapid and short-acting topical ophthalmic anesthetic.
12.1 Mechanism of Action
Tetracaine blocks sodium ion channels required for the initiation and conduction of neuronal impulses thereby affecting local anesthesia.
5 Warnings and Precautions
Do not use intracamerally since use may damage corneal endothelial cells. (5.1)
- Prolonged use or abuse may lead to corneal epithelial toxicity and may manifest as epithelial defects which may progress to permanent corneal damage. (5.2)
- Patients should not touch the eye for at least 10-20 minutes after using anesthetic as accidental injuries can occur due to insensitivity of the eye. (5.3)
- For Administration by Healthcare Provider : Tetracaine Hydrochloride Ophthalmic Solution, USP 0.5% is not intended for patient self- administration. (5.4)
2 Dosage and Administration
One drop topically in the eye(s) as needed.
3 Dosage Forms and Strengths
Tetracaine hydrochloride ophthalmic solution, USP 0.5% is a clear, colorless, ophthalmic solution containing 0.5% w/v tetracaine hydrochloride equivalent to tetracaine 0.44% w/v.
17 Patient Counseling Information
Eye Care Precaution
Do not touch the dropper tip to any surface as this may contaminate the solution.
Advise patients that, due to the effect of the anesthetic, their eyes will be insensitive for up to 20 minutes and that care should be taken to avoid accidental injuries.
16 How Supplied/storage and Handling
Tetracaine hydrochloride ophthalmic solution, USP 0.5% is supplied as a sterile, aqueous, topical ophthalmic solution in a white LDPE bottle, plugged with white LDPE nozzle and capped with white HDPE cap in the following sizes:
NDC 70069-596-01, 5 mL fill in 5 mL Bottle
NDC 70069-597-01, 15 mL fill in 15 mL Bottle
After opening, this product can be used until the expiration date stamped on the bottle.
Storage: Store at 15° to 25°C (59° to 77°F). Protect from light. Do not use if solution contains crystals, cloudy, or discolored.
Package Label.principal Display Panel
Container Label
NDC 70069-596-01
Tetracaine Hydrochloride Ophthalmic Solution USP, 0.5%
Sterile
Rx only
5 mL
Carton Label
NDC 70069-596-01
Tetracaine Hydrochloride Ophthalmic Solution USP, 0.5%
Sterile
Rx only
5 mL
Container Label
NDC 70069-597-01
Tetracaine Hydrochloride Ophthalmic Solution USP, 0.5%
Sterile
Rx only
15 mL
Carton Label
NDC 70069-597-01
Tetracaine Hydrochloride Ophthalmic Solution USP, 0.5%
Sterile
Rx only
15 mL
5.3 Corneal Injury Due to Insensitivity
Patients should not touch the eye for at least 10-20 minutes after using anesthetic as accidental injuries can occur due to insensitivity of the eye.
5.1 Corneal Injury With Intracameral Use
Not for injection or intraocular use. Do not use intracamerally because use of tetracaine hydrochloride ophthalmic solution, 0.5% may lead to damage of the corneal endothelial cells.
5.4 for Administration By Healthcare Provider
Tetracaine hydrochloride ophthalmic solution, 0.5% is indicated for administration under the direct supervision of a healthcare provider. Tetracaine Hydrochloride Ophthalmic Solution, 0.5% is not intended for patient self-administration [see Warnings and Precautions (5.2)].
8.3 Females and Males of Reproductive Potential
No human data on the effect of tetracaine hydrochloride ophthalmic solution, 0.5% on fertility are available.
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Studies to assess the genotoxicity of tetracaine hydrochloride have not been reported in the published literature. Long-term animal studies have not been conducted to evaluate the carcinogenic potential of tetracaine hydrochloride. Animal studies to assess the effects of tetracaine hydrochloride on fertility have not been reported in the published literature.
Structured Label Content
10 Overdosage (10 OVERDOSAGE)
Prolonged use of a topical ocular anesthetic including tetracaine hydrochloride ophthalmic solution, 0.5% may produce permanent corneal opacification and ulceration with accompanying visual loss.
8.1 Pregnancy
Risk Summary
There are no adequate and well-controlled studies with tetracaine hydrochloride ophthalmic solution, 0.5% in pregnant women. Animal developmental and reproductive toxicity studies with tetracaine hydrochloride have not been reported in the published literature.
8.2 Lactation
Risk Summary
There are no data to assess whether tetracaine hydrochloride ophthalmic solution, 0.5% is excreted in human milk or to assess its effects on milk production/excretion. The developmental and health benefits of breastfeeding should be considered along with the mother's clinical need for tetracaine hydrochloride ophthalmic solution, 0.5% and any potential adverse effects on the breastfed child from tetracaine hydrochloride ophthalmic solution, 0.5%.
11 Description (11 DESCRIPTION)
Tetracaine hydrochloride ophthalmic solution, USP 0.5% is a sterile, clear, colorless, topical local anesthetic for ophthalmic use only containing tetracaine hydrochloride as the active pharmaceutical ingredient.
Tetracaine hydrochloride is chemically designated as benzoic acid, 4-(butylamino)-, 2-(dimethylamino) ethyl ester, monohydrochloride. Its chemical formula is C15H24N2O2 ● HCl and it is represented by the chemical structure:
Tetracaine hydrochloride is a white crystalline powder with a molecular weight of 300.82
Active ingredient: tetracaine hydrochloride 0.5% w/v (equivalent to 0.44% w/v tetracaine)
Preservative: chlorobutanol 0.4%
Inactive ingredients: boric acid, potassium chloride, edetate disodium dihydrate, water for injection. Sodium hydroxide and/or hydrochloric acid may be added to adjust pH (3.7 – 6.0).
Spl Unclassified (SPL UNCLASSIFIED)
Manufactured for:
Somerset Therapeutics, LLC
Somerset, NJ 08873
Made in India
Code No.: KR/DRUGS/KTK/28/289/97
ST-TEC/P/00
1200992
8.4 Pediatric Use
Safety of tetracaine hydrochloride ophthalmic solution, 0.5% in the pediatric population has been demonstrated in clinical trials. Efficacy of tetracaine hydrochloride ophthalmic solution, 0.5% for use in pediatric patients has been extrapolated from adequate and well controlled clinical trials in the adult population.
8.5 Geriatric Use
No overall differences in safety or effectiveness of tetracaine hydrochloride ophthalmic solution, 0.5% have been observed between elderly and younger patients.
14 Clinical Studies (14 CLINICAL STUDIES)
Topical administration of tetracaine hydrochloride ophthalmic solution, 0.5% results in localized temporary anesthesia. The maximum effect is achieved within 10–20 seconds after instillation, with efficacy lasting 10–20 minutes. Duration of effect can be extended with repeated dosing [ see Warnings and Precautions ( 5.2) and Overdosage (10)].
4 Contraindications (4 CONTRAINDICATIONS)
Tetracaine hydrochloride ophthalmic solution, 0.5% should not be used in patients with a history of hypersensitivity to any component of this preparation.
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following serious ocular adverse reactions are described elsewhere in the labeling:
- Corneal Injury with Intracameral Use [ see Warnings and Precautions (5.1)]
- Corneal Toxicity [ see Warnings and Precautions ( 5.2)]
- Corneal Injury Due to Insensitivity [see Warnings and Precautions ( 5.3)]
The following adverse reactions have been identified following use of tetracaine hydrochloride ophthalmic solution, 0.5%. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Ocular Adverse Reactions
Transient stinging, burning, and conjunctival redness, eye irritation, eye pain, ocular discomfort.
5.2 Corneal Toxicity
Prolonged use or abuse may lead to corneal epithelial toxicity and may manifest as epithelial defects which may progress to permanent corneal damage.
Recent Major Changes (RECENT MAJOR CHANGES)
| Warnings and Precautions (5.4) |
2/2022 |
12.3 Pharmacokinetics
The systemic exposure to tetracaine following topical ocular administration of tetracaine hydrochloride ophthalmic solution, 0.5% has not been studied. Tetracaine hydrochloride is metabolized by plasma pseudocholinesterases and nonspecific esterases in ocular tissues.
1 Indications and Usage (1 INDICATIONS AND USAGE)
Tetracaine hydrochloride ophthalmic solution, 0.5% is indicated for procedures requiring a rapid and short-acting topical ophthalmic anesthetic.
12.1 Mechanism of Action
Tetracaine blocks sodium ion channels required for the initiation and conduction of neuronal impulses thereby affecting local anesthesia.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
Do not use intracamerally since use may damage corneal endothelial cells. (5.1)
- Prolonged use or abuse may lead to corneal epithelial toxicity and may manifest as epithelial defects which may progress to permanent corneal damage. (5.2)
- Patients should not touch the eye for at least 10-20 minutes after using anesthetic as accidental injuries can occur due to insensitivity of the eye. (5.3)
- For Administration by Healthcare Provider : Tetracaine Hydrochloride Ophthalmic Solution, USP 0.5% is not intended for patient self- administration. (5.4)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
One drop topically in the eye(s) as needed.
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Tetracaine hydrochloride ophthalmic solution, USP 0.5% is a clear, colorless, ophthalmic solution containing 0.5% w/v tetracaine hydrochloride equivalent to tetracaine 0.44% w/v.
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Eye Care Precaution
Do not touch the dropper tip to any surface as this may contaminate the solution.
Advise patients that, due to the effect of the anesthetic, their eyes will be insensitive for up to 20 minutes and that care should be taken to avoid accidental injuries.
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
Tetracaine hydrochloride ophthalmic solution, USP 0.5% is supplied as a sterile, aqueous, topical ophthalmic solution in a white LDPE bottle, plugged with white LDPE nozzle and capped with white HDPE cap in the following sizes:
NDC 70069-596-01, 5 mL fill in 5 mL Bottle
NDC 70069-597-01, 15 mL fill in 15 mL Bottle
After opening, this product can be used until the expiration date stamped on the bottle.
Storage: Store at 15° to 25°C (59° to 77°F). Protect from light. Do not use if solution contains crystals, cloudy, or discolored.
Package Label.principal Display Panel (PACKAGE LABEL.PRINCIPAL DISPLAY PANEL)
Container Label
NDC 70069-596-01
Tetracaine Hydrochloride Ophthalmic Solution USP, 0.5%
Sterile
Rx only
5 mL
Carton Label
NDC 70069-596-01
Tetracaine Hydrochloride Ophthalmic Solution USP, 0.5%
Sterile
Rx only
5 mL
Container Label
NDC 70069-597-01
Tetracaine Hydrochloride Ophthalmic Solution USP, 0.5%
Sterile
Rx only
15 mL
Carton Label
NDC 70069-597-01
Tetracaine Hydrochloride Ophthalmic Solution USP, 0.5%
Sterile
Rx only
15 mL
5.3 Corneal Injury Due to Insensitivity
Patients should not touch the eye for at least 10-20 minutes after using anesthetic as accidental injuries can occur due to insensitivity of the eye.
5.1 Corneal Injury With Intracameral Use (5.1 Corneal Injury with Intracameral Use)
Not for injection or intraocular use. Do not use intracamerally because use of tetracaine hydrochloride ophthalmic solution, 0.5% may lead to damage of the corneal endothelial cells.
5.4 for Administration By Healthcare Provider (5.4 For Administration by Healthcare Provider)
Tetracaine hydrochloride ophthalmic solution, 0.5% is indicated for administration under the direct supervision of a healthcare provider. Tetracaine Hydrochloride Ophthalmic Solution, 0.5% is not intended for patient self-administration [see Warnings and Precautions (5.2)].
8.3 Females and Males of Reproductive Potential
No human data on the effect of tetracaine hydrochloride ophthalmic solution, 0.5% on fertility are available.
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Studies to assess the genotoxicity of tetracaine hydrochloride have not been reported in the published literature. Long-term animal studies have not been conducted to evaluate the carcinogenic potential of tetracaine hydrochloride. Animal studies to assess the effects of tetracaine hydrochloride on fertility have not been reported in the published literature.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:44:22.682417 · Updated: 2026-03-14T22:19:49.692010