Janitex
ba3f957d-b477-4c51-8213-b67bb7c00ab3
34390-5
HUMAN OTC DRUG LABEL
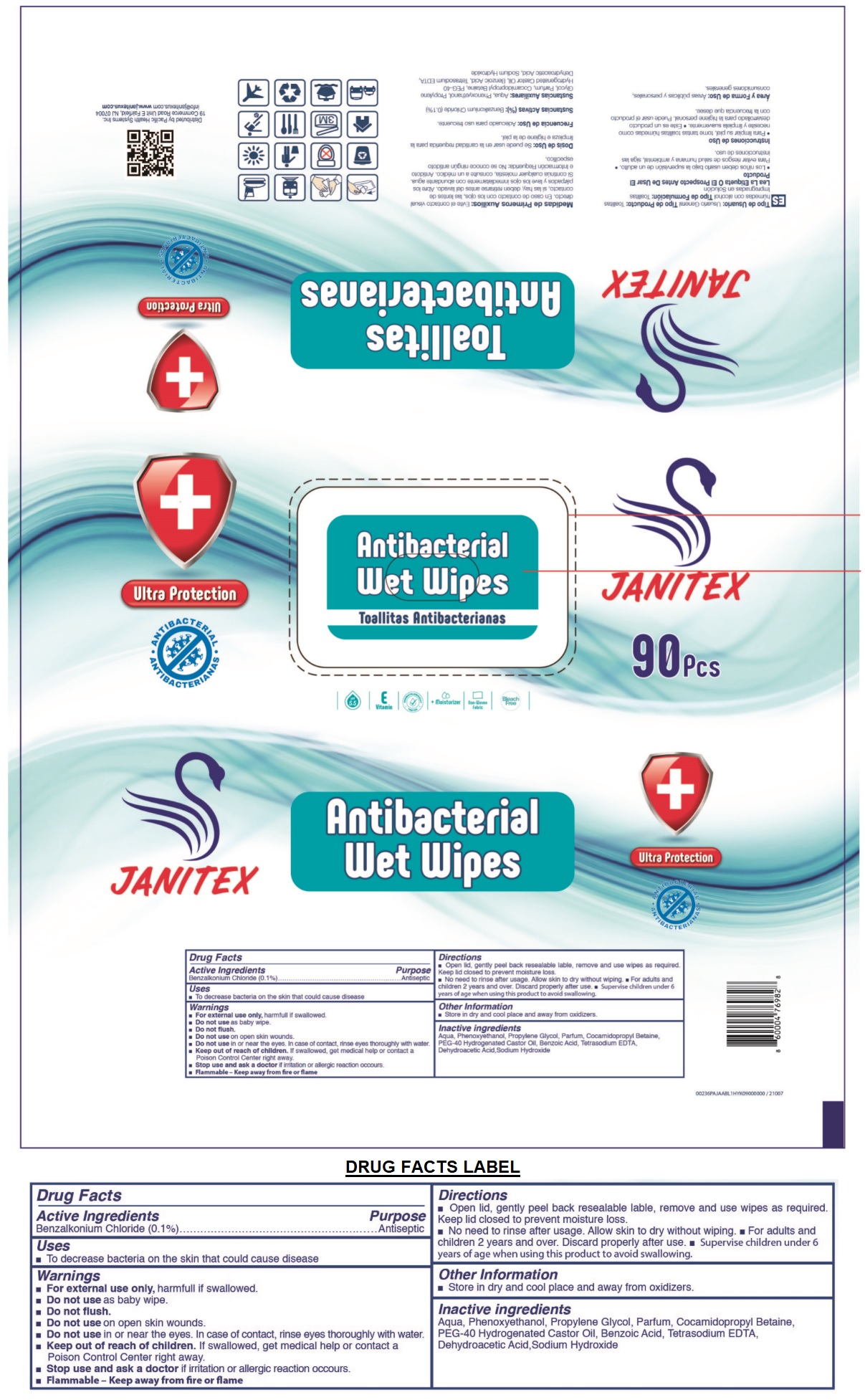
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Benzalkonium Chloride (0.1%)
Purpose
Antiseptic
Medication Information
Purpose
Antiseptic
Description
Benzalkonium Chloride (0.1%)
Uses
- To decrease bacteria on the skin that could cause disease
Section 42229-5
pH 5.5
E Vitamin
DERMATOLOGICALLY TESTED
+ moisturizer
non-woven fabric
Bleach Free
Ultra Protection
ANTIBACTERIAL
Distributed by Pacific Health Systems Inc.
19 Commerce Road Unit E Fairfield, NJ 07004
[email protected] www.janitexus.com
Section 50565-1
- Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center right away.
Warnings
- For external use only, harmful if swallowed.
- Do not use as baby wipe.
- Do not flush.
- Do not use on open skin wounds.
- Do not use in or near the eyes. In case of contact, rinse eyes thoroughly with water.
- Stop use and ask a doctor if irritation or allergic reaction occurs.
- Flammable - Keep away from fire or flame
Packaging
Directions
• Open lid, gently peel back resealable label, remove and use wipes as required. Keep lid closed to prevent moisture loss.
• No need to rinse after usage. Allow skin to dry without wiping. • For adults and children 2 years and over. Discard properly after use. • Supervise children under 6 years of age when using this product to avoid swallowing.
Drug Facts
Other Information
- Store in dry and cool place and away from oxidizers.
Active Ingredients
Benzalkonium Chloride (0.1%)
Inactive Ingredients
Aqua, Phenoxyethanol, Propylene Glycol, Parfum, Cocamidopropyl Betaine, PEG-40 Hydrogenated Castor Oil, Benzoic Acid, Tetrasodium EDTA, Dehydroacetic Acid, Sodium Hydroxide
Structured Label Content
Uses
- To decrease bacteria on the skin that could cause disease
Section 42229-5 (42229-5)
pH 5.5
E Vitamin
DERMATOLOGICALLY TESTED
+ moisturizer
non-woven fabric
Bleach Free
Ultra Protection
ANTIBACTERIAL
Distributed by Pacific Health Systems Inc.
19 Commerce Road Unit E Fairfield, NJ 07004
[email protected] www.janitexus.com
Section 50565-1 (50565-1)
- Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center right away.
Purpose
Antiseptic
Warnings
- For external use only, harmful if swallowed.
- Do not use as baby wipe.
- Do not flush.
- Do not use on open skin wounds.
- Do not use in or near the eyes. In case of contact, rinse eyes thoroughly with water.
- Stop use and ask a doctor if irritation or allergic reaction occurs.
- Flammable - Keep away from fire or flame
Packaging
Directions
• Open lid, gently peel back resealable label, remove and use wipes as required. Keep lid closed to prevent moisture loss.
• No need to rinse after usage. Allow skin to dry without wiping. • For adults and children 2 years and over. Discard properly after use. • Supervise children under 6 years of age when using this product to avoid swallowing.
Drug Facts
Other Information
- Store in dry and cool place and away from oxidizers.
Active Ingredients
Benzalkonium Chloride (0.1%)
Inactive Ingredients (Inactive ingredients)
Aqua, Phenoxyethanol, Propylene Glycol, Parfum, Cocamidopropyl Betaine, PEG-40 Hydrogenated Castor Oil, Benzoic Acid, Tetrasodium EDTA, Dehydroacetic Acid, Sodium Hydroxide
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:58:29.200897 · Updated: 2026-03-14T22:58:01.908948