Hydroquinone 4% Cream
ba2b7504-17bb-4b9e-8db1-937f83d75a03
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
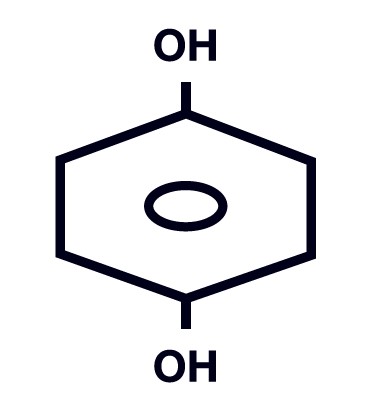
Each gram of Hydroquinone USP, 4% Skin Bleaching Cream contains 40 mg of hydroquinone USP, in a vanishing cream base of aqua (water), BHT, cetyl alcohol, disodium EDTA, glycerin, glycolic acid, methylparaben, propylparaben, saponins, sodium lauryl sulfate, sodium metabisulfite, stearyl alcohol, tetrahexyldecyl ascorbate, and tocopheryl acetate. Chemically, hydroquinone is C 6 H 6 O 2 and has a molecular weight of 110.11. The chemical name is 1,4 dihydroxybenzene, and the structural formula of hydroquinone is:
Contraindications
Prior history of sensitivity or allergic reaction to hydroquinone or to any of the ingredients of the product. The safety of topical hydroquinone use during pregnancy or for children (12 years and under) has not been established.
Adverse Reactions
The following adverse reactions have been reported: dryness and fissuring of paranasal and infraorbital areas, erythema, and stinging. Occasional hypersensitivity (localized contact dermatitis) may develop. If this occurs, the medication should be discontinued and the physician notified immediately.
Drug Interactions
Patients are cautioned on concomitant use of medications that are known to be photosensitizing.
How Supplied
Hydroquinone USP, 4% Skin Bleaching Cream is available as follows: NDC: 72162-2422-2: 1 oz (28.4 g) in a tube. STORAGE Hydroquinone USP, 4% Skin Bleaching Cream should be stored at controlled room temperature (20-25°C) (68-77°F). Darkening of this product is normal. This will not affect performance or safety. Repackaged/Relabeled by: Bryant Ranch Prepack, Inc. Burbank, CA 91504
Medication Information
Contraindications
Prior history of sensitivity or allergic reaction to hydroquinone or to any of the ingredients of the product. The safety of topical hydroquinone use during pregnancy or for children (12 years and under) has not been established.
Adverse Reactions
The following adverse reactions have been reported: dryness and fissuring of paranasal and infraorbital areas, erythema, and stinging.
Occasional hypersensitivity (localized contact dermatitis) may develop. If this occurs, the medication should be discontinued and the physician notified immediately.
Drug Interactions
Patients are cautioned on concomitant use of medications that are known to be photosensitizing.
How Supplied
Hydroquinone USP, 4% Skin Bleaching Cream is available as follows:
NDC: 72162-2422-2: 1 oz (28.4 g) in a tube.
STORAGE
Hydroquinone USP, 4% Skin Bleaching Cream should be stored at controlled room temperature (20-25°C) (68-77°F). Darkening of this product is normal.
This will not affect performance or safety.
Repackaged/Relabeled by:
Bryant Ranch Prepack, Inc.
Burbank, CA 91504
Description
Each gram of Hydroquinone USP, 4% Skin Bleaching Cream contains 40 mg of hydroquinone USP, in a vanishing cream base of aqua (water), BHT, cetyl alcohol, disodium EDTA, glycerin, glycolic acid, methylparaben, propylparaben, saponins, sodium lauryl sulfate, sodium metabisulfite, stearyl alcohol, tetrahexyldecyl ascorbate, and tocopheryl acetate. Chemically, hydroquinone is C6H6O2 and has a molecular weight of 110.11. The chemical name is 1,4 dihydroxybenzene, and the structural formula of hydroquinone is:
Section 51945-4
Hydroquinone 4% Cream #28.4
Warnings
Contains sodium metabisulfite, a sulfite that may cause allergic-type reactions including anaphylactic symptoms and life-threatening or less severe asthmatic episodes in certain susceptible people. The overall prevalence of sulfite sensitivity in the general population is unknown and probably low. Sulfite sensitivity is seen more frequently in asthmatic than in non-asthmatic people. Since this product contains no sunscreen, an effective broad spectrum sun blocking agent should be used and unnecessary solar exposure avoided, or protective clothing should be worn to cover bleached skin in order to prevent repigmentation from occurring. Hydroquinone may produce exogenous ochronosis, a gradual blue-black darkening of the skin. If this condition occurs, discontinue treatment and consult your physician. The majority of patients developing this condition are Black, but it may also occur in Caucasians and Hispanics.
Pregnancy
Teratogenic Effects: Pregnancy Category C -
Animal reproduction studies have not been conducted with topical hydroquinone. It is also not known whether topical hydroquinone can cause fetal harm when administered to a pregnant woman or can affect reproduction capacity. Topical hydroquinone should be given to a pregnant woman only if clearly needed.
Overdosage
There have been no systemic reactions reported from the use of topical hydroquinone. However, treatment should be limited to relatively small areas of the body at one time, since some patients experience a transient skin reddening and a mild burning sensation which does not preclude treatment.
Precautions
(see WARNINGS)
Pediatric Use
Safety and effectiveness for pediatric patients below the age of 12 years have not been established.
Nursing Mothers
It is not known whether this drug is excreted in human milk. Because many drugs are excreted in human milk, caution should be exercised when topical hydroquinone is administered to a nursing woman.
General Precautions
Test for skin sensitivity before using by applying a small amount to an unbroken patch of skin; check within 24 hours. Minor redness is not a contraindication, but where there is itching or vesicle formation or excessive inflammatory response further treatment is not advised. Close patient supervision is recommended.
Hydroquinone is a skin bleaching agent which may produce unwanted cosmetic effects if not used as directed. The physician should be familiar with the contents of this insert before prescribing or dispensing this medication.
Indications & Usage
Hydroquinone USP, 4% Skin Bleaching Cream is indicated for the gradual bleaching of hyperpigmented skin conditions such as chloasma, melasma, freckles, senile lentigines, and other unwanted areas of melanin hyperpigmentation.
Clinical Pharmacology
Topical application of hydroquinone produces a reversible depigmentation of the skin by inhibition of the enzymatic oxidation of tyrosine to 3,4-dihydroxyphenylalanine (dopa) (Denton, C. et al., 1952)1 and suppression of other melanocyte metabolic processes (Jimbow, K. et al., 1974)2. Exposure to sunlight or ultraviolet light will cause repigmentation of bleached areas (Parrish, J.A. et al., 1978)3.
Dosage & Administration
Hydroquinone USP, 4% Skin Bleaching Cream should be applied to affected areas and rubbed in well twice daily, in the morning and before bedtime, or as directed by a physician. If no improvement is seen after 2 months of treatment, use of this product should be discontinued. There is no recommended dosage for pediatric patients under 12 years of age except under the advice and supervision of a physician.
Information for Patients
Sunscreen use is an essential aspect of hydroquinone therapy because even minimal sunlight sustains melanocytic activity. To prevent repigmentation, during treatment and maintenance therapy, sun exposure on treated skin should be avoided by application of a broad spectrum sunscreen (SPF 15 or greater) or by use of protective clothing.
Avoid contact with eyes and mucous membranes.
Keep this and all medications out of reach of children. In case of accidental ingestion, call a physician or a poison control center immediately.
Carcinogenesis & Mutagenesis & Impairment of Fertility
Studies of hydroquinone in animals have demonstrated some evidence of carcinogenicity. The carcinogenic potential of hydroquinone in humans is unknown.
Published studies have demonstrated that hydroquinone is a mutagen and a clastogen. Treatment with hydroquinone has resulted in positive findings for genetic toxicity in the Ames assay in bacterial strains sensitive to oxidizing mutagens, in in vitro studies in mammalian cells, and in the in vivo mouse micronucleus assay.
Structured Label Content
Section 51945-4 (51945-4)
Hydroquinone 4% Cream #28.4
Warnings (WARNINGS)
Contains sodium metabisulfite, a sulfite that may cause allergic-type reactions including anaphylactic symptoms and life-threatening or less severe asthmatic episodes in certain susceptible people. The overall prevalence of sulfite sensitivity in the general population is unknown and probably low. Sulfite sensitivity is seen more frequently in asthmatic than in non-asthmatic people. Since this product contains no sunscreen, an effective broad spectrum sun blocking agent should be used and unnecessary solar exposure avoided, or protective clothing should be worn to cover bleached skin in order to prevent repigmentation from occurring. Hydroquinone may produce exogenous ochronosis, a gradual blue-black darkening of the skin. If this condition occurs, discontinue treatment and consult your physician. The majority of patients developing this condition are Black, but it may also occur in Caucasians and Hispanics.
Pregnancy (PREGNANCY)
Teratogenic Effects: Pregnancy Category C -
Animal reproduction studies have not been conducted with topical hydroquinone. It is also not known whether topical hydroquinone can cause fetal harm when administered to a pregnant woman or can affect reproduction capacity. Topical hydroquinone should be given to a pregnant woman only if clearly needed.
Overdosage (OVERDOSAGE)
There have been no systemic reactions reported from the use of topical hydroquinone. However, treatment should be limited to relatively small areas of the body at one time, since some patients experience a transient skin reddening and a mild burning sensation which does not preclude treatment.
Description (DESCRIPTION)
Each gram of Hydroquinone USP, 4% Skin Bleaching Cream contains 40 mg of hydroquinone USP, in a vanishing cream base of aqua (water), BHT, cetyl alcohol, disodium EDTA, glycerin, glycolic acid, methylparaben, propylparaben, saponins, sodium lauryl sulfate, sodium metabisulfite, stearyl alcohol, tetrahexyldecyl ascorbate, and tocopheryl acetate. Chemically, hydroquinone is C6H6O2 and has a molecular weight of 110.11. The chemical name is 1,4 dihydroxybenzene, and the structural formula of hydroquinone is:
Precautions (PRECAUTIONS)
(see WARNINGS)
How Supplied (HOW SUPPLIED)
Hydroquinone USP, 4% Skin Bleaching Cream is available as follows:
NDC: 72162-2422-2: 1 oz (28.4 g) in a tube.
STORAGE
Hydroquinone USP, 4% Skin Bleaching Cream should be stored at controlled room temperature (20-25°C) (68-77°F). Darkening of this product is normal.
This will not affect performance or safety.
Repackaged/Relabeled by:
Bryant Ranch Prepack, Inc.
Burbank, CA 91504
Pediatric Use (PEDIATRIC USE)
Safety and effectiveness for pediatric patients below the age of 12 years have not been established.
Nursing Mothers (NURSING MOTHERS)
It is not known whether this drug is excreted in human milk. Because many drugs are excreted in human milk, caution should be exercised when topical hydroquinone is administered to a nursing woman.
Adverse Reactions (ADVERSE REACTIONS)
The following adverse reactions have been reported: dryness and fissuring of paranasal and infraorbital areas, erythema, and stinging.
Occasional hypersensitivity (localized contact dermatitis) may develop. If this occurs, the medication should be discontinued and the physician notified immediately.
Contraindications (CONTRAINDICATIONS)
Prior history of sensitivity or allergic reaction to hydroquinone or to any of the ingredients of the product. The safety of topical hydroquinone use during pregnancy or for children (12 years and under) has not been established.
Drug Interactions (DRUG INTERACTIONS)
Patients are cautioned on concomitant use of medications that are known to be photosensitizing.
General Precautions (GENERAL PRECAUTIONS)
Test for skin sensitivity before using by applying a small amount to an unbroken patch of skin; check within 24 hours. Minor redness is not a contraindication, but where there is itching or vesicle formation or excessive inflammatory response further treatment is not advised. Close patient supervision is recommended.
Hydroquinone is a skin bleaching agent which may produce unwanted cosmetic effects if not used as directed. The physician should be familiar with the contents of this insert before prescribing or dispensing this medication.
Indications & Usage (INDICATIONS & USAGE)
Hydroquinone USP, 4% Skin Bleaching Cream is indicated for the gradual bleaching of hyperpigmented skin conditions such as chloasma, melasma, freckles, senile lentigines, and other unwanted areas of melanin hyperpigmentation.
Clinical Pharmacology (CLINICAL PHARMACOLOGY)
Topical application of hydroquinone produces a reversible depigmentation of the skin by inhibition of the enzymatic oxidation of tyrosine to 3,4-dihydroxyphenylalanine (dopa) (Denton, C. et al., 1952)1 and suppression of other melanocyte metabolic processes (Jimbow, K. et al., 1974)2. Exposure to sunlight or ultraviolet light will cause repigmentation of bleached areas (Parrish, J.A. et al., 1978)3.
Dosage & Administration (DOSAGE & ADMINISTRATION)
Hydroquinone USP, 4% Skin Bleaching Cream should be applied to affected areas and rubbed in well twice daily, in the morning and before bedtime, or as directed by a physician. If no improvement is seen after 2 months of treatment, use of this product should be discontinued. There is no recommended dosage for pediatric patients under 12 years of age except under the advice and supervision of a physician.
Information for Patients (INFORMATION FOR PATIENTS)
Sunscreen use is an essential aspect of hydroquinone therapy because even minimal sunlight sustains melanocytic activity. To prevent repigmentation, during treatment and maintenance therapy, sun exposure on treated skin should be avoided by application of a broad spectrum sunscreen (SPF 15 or greater) or by use of protective clothing.
Avoid contact with eyes and mucous membranes.
Keep this and all medications out of reach of children. In case of accidental ingestion, call a physician or a poison control center immediately.
Carcinogenesis & Mutagenesis & Impairment of Fertility (CARCINOGENESIS & MUTAGENESIS & IMPAIRMENT OF FERTILITY)
Studies of hydroquinone in animals have demonstrated some evidence of carcinogenicity. The carcinogenic potential of hydroquinone in humans is unknown.
Published studies have demonstrated that hydroquinone is a mutagen and a clastogen. Treatment with hydroquinone has resulted in positive findings for genetic toxicity in the Ames assay in bacterial strains sensitive to oxidizing mutagens, in in vitro studies in mammalian cells, and in the in vivo mouse micronucleus assay.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:43:43.782504 · Updated: 2026-03-14T22:17:00.667686