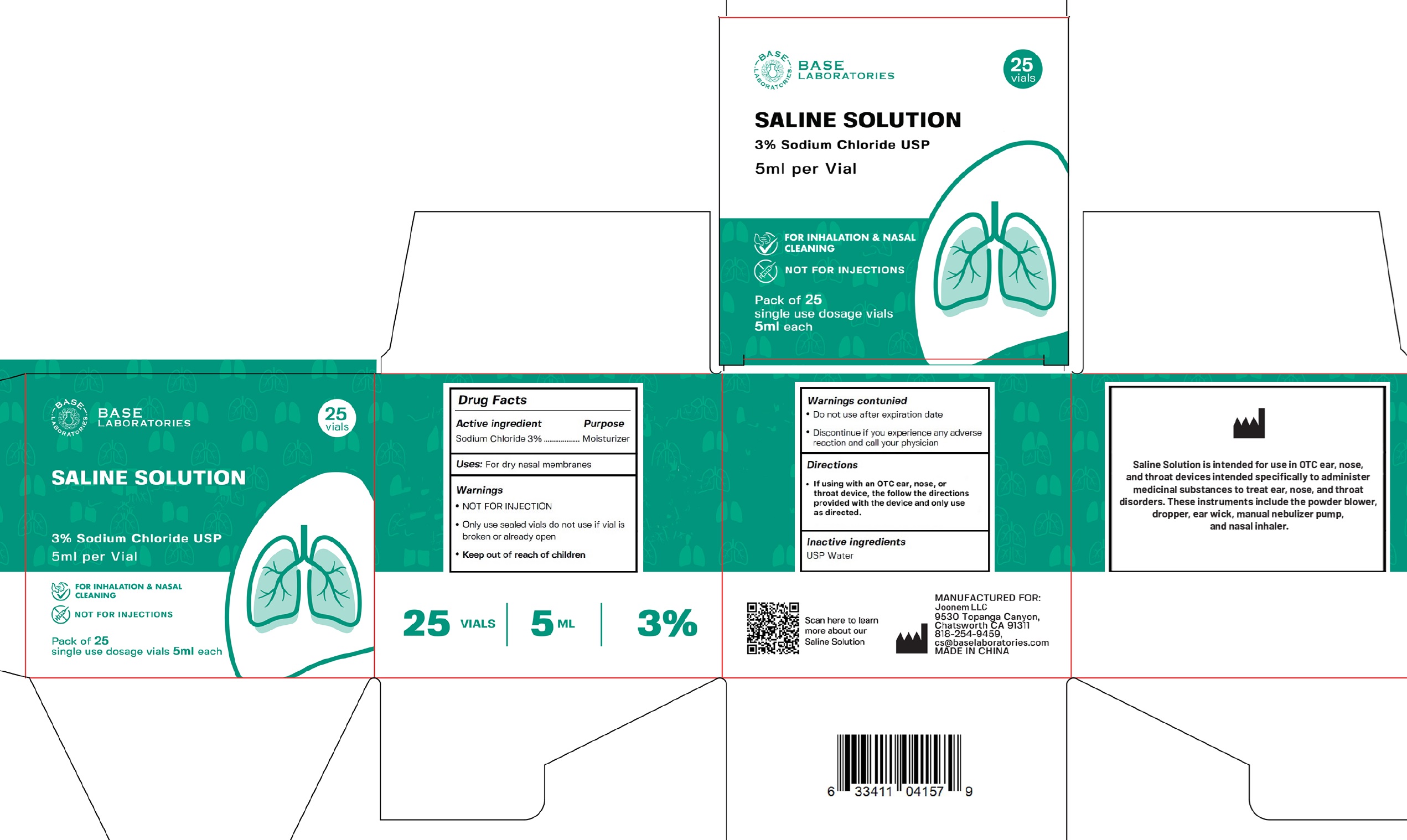
base laboratories saline solution 3% sodium chloride
ba0868f6-d3de-40e2-bcb3-144fa53062c7
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Purpose
Moisturizer
Description
Sodium Chloride 3%
Medication Information
Warnings
- NOT FOR INJECTION
- Only use sealed vials do not use if vial is broken or already open
Purpose
Moisturizer
Directions
- If using with an OTC ear, nose, or throat device, the follow the directions provided with the device and only use as directed.
Inactive Ingredients
USP Water
Description
Sodium Chloride 3%
Drug Facts
Uses:
For dry nasal membranes
Active Ingredient
Sodium Chloride 3%
Package Labelling:
• Do Not Use
after expiration date
- Discontinue if you experience any adverse reaction and call your physician
Structured Label Content
Warnings
- NOT FOR INJECTION
- Only use sealed vials do not use if vial is broken or already open
Purpose
Moisturizer
Directions
- If using with an OTC ear, nose, or throat device, the follow the directions provided with the device and only use as directed.
Inactive Ingredients (Inactive ingredients)
USP Water
Drug Facts
Uses:
For dry nasal membranes
Active Ingredient (Active ingredient)
Sodium Chloride 3%
Package Labelling:
• Do Not Use (• Do not use )
after expiration date
- Discontinue if you experience any adverse reaction and call your physician
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:05:02.348117 · Updated: 2026-03-14T23:06:18.731306