Palonosetron Hydrochloride Injection
b93fae8e-2b76-421e-95bc-ba08c211a7ea
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Indications and Usage
Palonosetron Hydrochloride Injection is indicated in adults for prevention of: acute and delayed nausea and vomiting associated with initial and repeat courses of moderately emetogenic cancer chemotherapy (MEC). acute nausea and vomiting associated with initial and repeat courses highly emetogenic cancer chemotherapy (HEC). Palonosetron Hydrochloride Injection is indicated in pediatric patients 1 month to less than 17 years of age for prevention of: acute nausea and vomiting associated with initial and repeat courses of emetogenic cancer chemotherapy, including highly emetogenic cancer chemotherapy.
Dosage and Administration
Chemotherapy-Induced Nausea and Vomiting ( 2.1 ) *Note different dosing units in pediatrics Age Dose* Infusion Time Adults 0.25 mg as a single dose Infuse over 30 seconds beginning approximately 30 minutes before the start of chemotherapy Pediatrics (1 month to less than 17 years) 20 micrograms per kilogram (maximum 1.5 mg) as a single dose Infuse over 15 minutes beginning approximately 30 minutes before the start of chemotherapy Instructions for Intravenous Administration For a dose of 0.25 mg, use the entire contents (5 mL) of the prefilled syringe. Do not use the prefilled syringe to administer a dose of less than 0.25 mg (5 mL). ( 2.2 )
Contraindications
Palonosetron Hydrochloride Injection is contraindicated in patients known to have hypersensitivity to palonosetron [see Warnings and Precautions ( 5.1 )] .
Warnings and Precautions
Hypersensitivity reactions, including anaphylaxis and anaphylactic shock : reported in patients with or without known hypersensitivity to other selective 5-HT 3 receptor antagonists. If symptoms occur, discontinue Palonosetron Hydrochloride Injection and initiate appropriate medical treatment. ( 5.1 ) Serotonin syndrome : reported with 5-HT 3 receptor antagonists alone, but particularly with concomitant use of serotonergic drugs. ( 5.2 , 7.1 )
Adverse Reactions
Serious or otherwise clinically significant adverse reactions reported in other sections of labeling: Hypersensitivity Reactions [ see Warnings and Precautions ( 5.1 ) ] Serotonin Syndrome [ see Warnings and Precautions ( 5.2 )]
Drug Interactions
Serotonergic Drugs : Monitor for serotonin syndrome; if symptoms occur, discontinue Palonosetron Hydrochloride Injection and initiate supportive treatment. ( 7.1 )
How Supplied
Palonosetron Hydrochloride Injection 0.25 mg/5 mL (free base) single-dose prefilled syringe is available as follows: Product No. NDC No. Strength Package 673189 63323-673-89 0.25 mg per 5 mL (0.05 mg per mL) 5 mL single-dose prefilled syringe, individually packaged in cartons of ten.
Storage and Handling
Palonosetron Hydrochloride Injection 0.25 mg/5 mL (free base) single-dose prefilled syringe is available as follows: Product No. NDC No. Strength Package 673189 63323-673-89 0.25 mg per 5 mL (0.05 mg per mL) 5 mL single-dose prefilled syringe, individually packaged in cartons of ten.
Description
Palonosetron Hydrochloride Injection is indicated in adults for prevention of: acute and delayed nausea and vomiting associated with initial and repeat courses of moderately emetogenic cancer chemotherapy (MEC). acute nausea and vomiting associated with initial and repeat courses highly emetogenic cancer chemotherapy (HEC). Palonosetron Hydrochloride Injection is indicated in pediatric patients 1 month to less than 17 years of age for prevention of: acute nausea and vomiting associated with initial and repeat courses of emetogenic cancer chemotherapy, including highly emetogenic cancer chemotherapy.
Medication Information
Warnings and Precautions
Hypersensitivity reactions, including anaphylaxis and anaphylactic shock : reported in patients with or without known hypersensitivity to other selective 5-HT 3 receptor antagonists. If symptoms occur, discontinue Palonosetron Hydrochloride Injection and initiate appropriate medical treatment. ( 5.1 ) Serotonin syndrome : reported with 5-HT 3 receptor antagonists alone, but particularly with concomitant use of serotonergic drugs. ( 5.2 , 7.1 )
Indications and Usage
Palonosetron Hydrochloride Injection is indicated in adults for prevention of: acute and delayed nausea and vomiting associated with initial and repeat courses of moderately emetogenic cancer chemotherapy (MEC). acute nausea and vomiting associated with initial and repeat courses highly emetogenic cancer chemotherapy (HEC). Palonosetron Hydrochloride Injection is indicated in pediatric patients 1 month to less than 17 years of age for prevention of: acute nausea and vomiting associated with initial and repeat courses of emetogenic cancer chemotherapy, including highly emetogenic cancer chemotherapy.
Dosage and Administration
Chemotherapy-Induced Nausea and Vomiting ( 2.1 ) *Note different dosing units in pediatrics Age Dose* Infusion Time Adults 0.25 mg as a single dose Infuse over 30 seconds beginning approximately 30 minutes before the start of chemotherapy Pediatrics (1 month to less than 17 years) 20 micrograms per kilogram (maximum 1.5 mg) as a single dose Infuse over 15 minutes beginning approximately 30 minutes before the start of chemotherapy Instructions for Intravenous Administration For a dose of 0.25 mg, use the entire contents (5 mL) of the prefilled syringe. Do not use the prefilled syringe to administer a dose of less than 0.25 mg (5 mL). ( 2.2 )
Contraindications
Palonosetron Hydrochloride Injection is contraindicated in patients known to have hypersensitivity to palonosetron [see Warnings and Precautions ( 5.1 )] .
Adverse Reactions
Serious or otherwise clinically significant adverse reactions reported in other sections of labeling: Hypersensitivity Reactions [ see Warnings and Precautions ( 5.1 ) ] Serotonin Syndrome [ see Warnings and Precautions ( 5.2 )]
Drug Interactions
Serotonergic Drugs : Monitor for serotonin syndrome; if symptoms occur, discontinue Palonosetron Hydrochloride Injection and initiate supportive treatment. ( 7.1 )
Storage and Handling
Palonosetron Hydrochloride Injection 0.25 mg/5 mL (free base) single-dose prefilled syringe is available as follows: Product No. NDC No. Strength Package 673189 63323-673-89 0.25 mg per 5 mL (0.05 mg per mL) 5 mL single-dose prefilled syringe, individually packaged in cartons of ten.
How Supplied
Palonosetron Hydrochloride Injection 0.25 mg/5 mL (free base) single-dose prefilled syringe is available as follows: Product No. NDC No. Strength Package 673189 63323-673-89 0.25 mg per 5 mL (0.05 mg per mL) 5 mL single-dose prefilled syringe, individually packaged in cartons of ten.
Description
Palonosetron Hydrochloride Injection is indicated in adults for prevention of: acute and delayed nausea and vomiting associated with initial and repeat courses of moderately emetogenic cancer chemotherapy (MEC). acute nausea and vomiting associated with initial and repeat courses highly emetogenic cancer chemotherapy (HEC). Palonosetron Hydrochloride Injection is indicated in pediatric patients 1 month to less than 17 years of age for prevention of: acute nausea and vomiting associated with initial and repeat courses of emetogenic cancer chemotherapy, including highly emetogenic cancer chemotherapy.
Section 42229-5
Prevention of Chemotherapy-Induced Nausea and Vomiting
The recommended dosage of Palonosetron Hydrochloride Injection for prevention of nausea and vomiting associated with HEC and MEC in adults and associated with emetogenic chemotherapy, including HEC in pediatric patients 1 month to less than 17 years of age is shown in Table 1.
|
*Note different dosing units in pediatrics |
||
| Age | Dose* | Infusion Time |
| Adults | 0.25 mg as a single dose | Infuse over 30 seconds beginning approximately 30 minutes before the start of chemotherapy |
| Pediatrics (1 month to less than 17 years) | 20 micrograms per kilogram (max 1.5 mg) as a single dose | Infuse over 15 minutes beginning approximately 30 minutes before the start of chemotherapy |
Section 42230-3
|
This Patient Information has been approved by the U.S. Food and Drug Administration. |
|||
|
Patient Information Palonosetron Hydrochloride
(PAL-oh-NOE-se-tron HYE-dro-KLOR-ide) Injection for Intravenous Use |
|||
| Read this Patient Information before you receive Palonosetron Hydrochloride Injection and each time you receive Palonosetron Hydrochloride Injection. There may be new information. This information does not take the place of talking with your doctor about your medical condition or your treatment. | |||
|
What is Palonosetron Hydrochloride Injection?
Palonosetron Hydrochloride Injection is a prescription medicine called an “antiemetic.” Palonosetron Hydrochloride Injection is used in adults to help prevent the nausea and vomiting that happens:
|
|||
Palonosetron Hydrochloride Injection is used in children 1 month old to less than 17 years of age to help prevent the nausea and vomiting that happens right away with certain anti-cancer medicines (chemotherapy).
|
|||
|
Who should not receive Palonosetron Hydrochloride Injection?
Do not receive Palonosetron Hydrochloride Injection if you are allergic to palonosetron hydrochloride or any of the ingredients in Palonosetron Hydrochloride Injection. See the end of this leaflet for a complete list of ingredients in Palonosetron Hydrochloride Injection. |
|||
|
What should I tell my doctor before receiving Palonosetron Hydrochloride Injection?
Before receiving Palonosetron Hydrochloride Injection, tell your doctor about all of your medical conditions, including if you:
|
|||
|
Tell your doctor about all of the medicines you take, including prescription and over-the-counter medicines, vitamins and herbal supplements. Palonosetron Hydrochloride Injection and certain other medicines can affect each other, causing serious side effects. |
|||
How will I receive Palonosetron Hydrochloride injection?
|
|||
What are the possible side effects of Palonosetron Hydrochloride Injection? Palonosetron Hydrochloride Injection may cause serious side effects, including:
|
|||
|
|
|
|
|
|||
|
|
||
|
The most common side effects in adults who receive Palonosetron Hydrochloride Injection to help prevent nausea and vomiting that happens with certain anti-cancer medicine (chemotherapy) include: headache and constipation. These are not all the possible side effects from Palonosetron Hydrochloride Injection. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
|||
| General information about the safe and effective use of Palonosetron Hydrochloride Injection Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. You can ask your doctor or pharmacist for information about Palonosetron Hydrochloride Injection that is written for health professionals. | |||
|
What are the ingredients in Palonosetron Hydrochloride Injection?
Lake Zurich, IL 60047 Made in Austria For more information, go to www.fresenius-kabi.com/us or call 1-800-551-7176. |
Section 44425-7
Storage
- Store at 20° to 25°C (68° to 77°F) [see USP Controlled Room Temperature].
- Protect from freezing.
- Protect from light.
- Discard unused portion
Section 51945-4
PACKAGE LABEL - PRINCIPAL DISPLAY PANEL - Palonosetron Hydrochloride 0.25 mg Single-Dose Prefilled Syringe Label
NDC 63323-673-21
Palonosetron Hydrochloride Injection
0.25 mg* per 5 mL
(0.05 mg* per mL)
For Intravenous Injection Only.
Discard Unused Portion.
5 mL
Single-Dose Syringe
Rx only
10. Overdosage
There is no known antidote to palonosetron. Overdose should be managed with supportive care.
Dialysis studies have not been performed, however, due to the large volume of distribution, dialysis is unlikely to be an effective treatment for palonosetron overdose. A single intravenous dose of palonosetron (HCl) at 30 mg/kg (947 and 474 times the human dose for rats and mice, respectively, based on body surface area) was lethal to rats and mice. The major signs of toxicity were convulsions, gasping, pallor, cyanosis and collapse.
11. Description
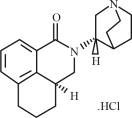
Palonosetron Hydrochloride Injection contains palonosetron as palonosetron HCl, an antiemetic and antinauseant agent. It is a serotonin-3 (5-HT3) receptor antagonist with a strong binding affinity for this receptor. Chemically, palonosetron hydrochloride is: (3aS)-2-[(S)- 1-Azabicyclo [2.2.2]oct-3- yl]-2,3,3a,4,5,6-hexahydro-1-oxo-1Hbenz[de] isoquinoline hydrochloride. Palonosetron hydrochloride exists as a single isomer and has the following structural formula:
| C 19 H 24 N 2 O•HCl | M.W. 332.87 |
Palonosetron Hydrochloride is a white to off-white crystalline powder. It is freely soluble in water, soluble in propylene glycol, and slightly soluble in ethanol and 2-propanol.
Palonosetron Hydrochloride Injection is a sterile, clear, colorless, non-pyrogenic, isotonic, buffered solution for intravenous administration. Palonosetron Hydrochloride Injection is available as a 5 mL single-dose prefilled syringe.
Each 5 mL syringe contains: 0.25 mg palonosetron (equivalent to 0.28 mg palonosetron HCl), 202.4 mg mannitol, 2.5 mg edetate disodium dihydrate, 18 mg trisodium citrate dihydrate, and 7 mg citric acid anhydrous in water for intravenous administration.
The pH of the solution in the 5 mL syringe is 4.5 to 5.5.
8.5 Geriatric Use
Of the 1,374 adult cancer patients in clinical studies of intravenously administered palonosetron HCl, 316 (23%) were 65 years and over, while 71 (5%) were at least 75 years and over. Of the 1,520 adult patients in clinical studies of intravenously administered palonosetron HCl, 73 (5%) were at least 65 years old [see Clinical Studies (14.1)]. No overall differences in safety or effectiveness were observed between these subjects and younger subjects, but greater sensitivity in some older individuals cannot be ruled out. Population pharmacokinetics analysis did not reveal any differences in palonosetron pharmacokinetics between cancer patients 65 years of age and older compared to younger patients [see Clinical Pharmacology (12.3)]. No dose adjustment is required for geriatric patients.
4. Contraindications
Palonosetron Hydrochloride Injection is contraindicated in patients known to have hypersensitivity to palonosetron [see Warnings and Precautions (5.1)].
6. Adverse Reactions
7. Drug Interactions
Serotonergic Drugs: Monitor for serotonin syndrome; if symptoms occur, discontinue Palonosetron Hydrochloride Injection and initiate supportive treatment. (7.1)
12.3 Pharmacokinetics
After intravenous dosing of palonosetron HCl in healthy subjects and cancer patients, an initial decline in palonosetron plasma concentrations is followed by a slow elimination from the body. Mean maximum plasma concentration (Cmax) and area under the concentration-time curve (AUC0-∞) are generally dose-proportional over the dose range of 0.3 to 90 mcg/kg in healthy subjects and in cancer patients. Following a single intravenous dose of palonosetron HCl at 3 mcg/kg (or 0.21 mg/70 kg) to six cancer patients, mean (±SD) maximum plasma concentration was estimated to be 5,630 ± 5,480 ng/L and mean AUC was 35.8 ± 20.9 h•mcg/L.
Following intravenous administration of Palonosetron Hydrochloride Injection 0.25 mg once every other day for 3 doses in 11 cancer patients, the mean increase in plasma palonosetron concentration from Day 1 to Day 5 was 42±34%. Following intravenous administration of Palonosetron Hydrochloride Injection 0.25 mg once daily for 3 days in 12 healthy subjects, the mean (±SD) increase in plasma palonosetron concentration from Day 1 to Day 3 was 110±45%.
5.2 Serotonin Syndrome
The development of serotonin syndrome has been reported with 5-HT3 receptor antagonists. Most reports have been associated with concomitant use of serotonergic drugs (e.g., selective serotonin reuptake inhibitors (SSRIs), serotonin and norepinephrine reuptake inhibitors (SNRIs), monoamine oxidase inhibitors, mirtazapine, fentanyl, lithium, tramadol, and intravenous methylene blue). Some of the reported cases were fatal. Serotonin syndrome occurring with overdose of another 5-HT3 receptor antagonist alone has also been reported. The majority of reports of serotonin syndrome related to 5-HT3 receptor antagonist use occurred in a post-anesthesia care unit or an infusion center.
Symptoms associated with serotonin syndrome may include the following combination of signs and symptoms: mental status changes (e.g. agitation, hallucinations, delirium, and coma), autonomic instability (e.g., tachycardia, labile blood pressure, dizziness, diaphoresis, flushing, hyperthermia), neuromuscular symptoms (e.g., tremor, rigidity, myoclonus, hyperreflexia, incoordination), seizures, with or without gastrointestinal symptoms (e.g., nausea, vomiting, diarrhea). Patients should be monitored for the emergence of serotonin syndrome, especially with concomitant use of Palonosetron Hydrochloride Injection and other serotonergic drugs. If symptoms of serotonin syndrome occur, discontinue Palonosetron Hydrochloride Injection and initiate supportive treatment. Patients should be informed of the increased risk of serotonin syndrome, especially if Palonosetron Hydrochloride Injection is used concomitantly with other serotonergic drugs [see Drug Interactions (7.1)].
7.1 Serotonergic Drugs
Serotonin syndrome (including altered mental status, autonomic instability, and neuromuscular symptoms) has been described following the concomitant use of 5-HT3 receptor antagonists and other serotonergic drugs, including selective serotonin reuptake inhibitors (SSRIs) and serotonin and noradrenaline reuptake inhibitors (SNRIs). Monitor for the emergence of serotonin syndrome. If symptoms occur, discontinue Palonosetron Hydrochloride Injection and initiate supportive treatment [see Warnings and Precautions (5.2)].
1. Indications and Usage
Palonosetron Hydrochloride Injection is indicated in adults for prevention of:
- acute and delayed nausea and vomiting associated with initial and repeat courses of moderately emetogenic cancer chemotherapy (MEC).
- acute nausea and vomiting associated with initial and repeat courses highly emetogenic cancer chemotherapy (HEC).
Palonosetron Hydrochloride Injection is indicated in pediatric patients 1 month to less than 17 years of age for prevention of:
- acute nausea and vomiting associated with initial and repeat courses of emetogenic cancer chemotherapy, including highly emetogenic cancer chemotherapy.
12.1 Mechanism of Action
Palonosetron is a 5-HT3 receptor antagonist with a strong binding affinity for this receptor and little or no affinity for other receptors.
Cancer chemotherapy may be associated with a high incidence of nausea and vomiting, particularly when certain agents, such as cisplatin, are used. 5-HT3 receptors are located on the nerve terminals of the vagus in the periphery and centrally in the chemoreceptor trigger zone of the area postrema. It is thought that chemotherapeutic agents produce nausea and vomiting by releasing serotonin from the enterochromaffin cells of the small intestine and that the released serotonin then activates 5-HT3 receptors located on vagal afferents to initiate the vomiting reflex.
5. Warnings and Precautions
- Hypersensitivity reactions, including anaphylaxis and anaphylactic shock: reported in patients with or without known hypersensitivity to other selective 5-HT3 receptor antagonists. If symptoms occur, discontinue Palonosetron Hydrochloride Injection and initiate appropriate medical treatment. (5.1)
- Serotonin syndrome: reported with 5-HT3 receptor antagonists alone, but particularly with concomitant use of serotonergic drugs. (5.2, 7.1)
2. Dosage and Administration
Chemotherapy-Induced Nausea and Vomiting (2.1)
|
*Note different dosing units in pediatrics |
||
| Age | Dose* | Infusion Time |
| Adults | 0.25 mg as a single dose | Infuse over 30 seconds beginning approximately 30 minutes before the start of chemotherapy |
| Pediatrics (1 month to less than 17 years) |
20 micrograms per
kilogram (maximum 1.5 mg) as a single dose |
Infuse over 15 minutes beginning approximately 30 minutes before the start of chemotherapy |
Instructions for Intravenous Administration
- For a dose of 0.25 mg, use the entire contents (5 mL) of the prefilled syringe. Do not use the prefilled syringe to administer a dose of less than 0.25 mg (5 mL). (2.2)
3. Dosage Form and Strengths
Palonosetron Hydrochloride Injection is supplied as a single-dose sterile, clear, colorless solution in a prefilled syringe that provides:
- 0.25 mg (free base) per 5 mL (concentration: 0.05 mg per mL, 50 mcg per mL)
6.2 Postmarketing Experience
The following adverse reactions have been identified during postapproval use of palonosetron HCl. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
- Hypersensitivity reactions: including dyspnea, bronchospasm, swelling/edema, erythema, pruritus, rash, urticaria, anaphylaxis and anaphylactic shock [see Warnings and Precautions (5.1)]
- Injection site reactions: including burning, induration, discomfort and pain
5.1 Hypersensitivity Reactions
Hypersensitivity reactions, including anaphylaxis and anaphylactic shock, have been reported with administration of Palonosetron Hydrochloride Injection [see Adverse Reactions (6.2)]. These reactions occurred in patients with or without known hypersensitivity to other 5-HT3 receptor antagonists. If hypersensitivity reactions occur, discontinue Palonosetron Hydrochloride Injection and initiate appropriate medical treatment. Do not reinitiate Palonosetron Hydrochloride Injection in patients who have previously experienced symptoms of hypersensitivity [see Contraindications (4)].
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
17. Patient Counseling Information
Advise the patient or caregiver to read the FDA-approved patient labeling (Patient Information).
16. How Supplied/storage and Handling
Palonosetron Hydrochloride Injection 0.25 mg/5 mL (free base) single-dose prefilled syringe is available as follows:
|
Product
No. |
NDC
No. |
Strength |
Package |
| 673189 | 63323-673-89 | 0.25 mg per 5 mL (0.05 mg per mL) |
5 mL single-dose prefilled syringe, individually packaged in cartons of ten. |
2.2 Instructions for Intravenous Administration
- Palonosetron Hydrochloride Injection is supplied ready for intravenous administration at a concentration of 0.05 mg/mL (50 mcg/mL).
- Do not mix Palonosetron Hydrochloride Injection with other drugs.
- Flush the infusion line with normal saline before and after administration of Palonosetron Hydrochloride Injection.
- Inspect Palonosetron Hydrochloride Injection visually for particulate matter and discoloration before administration.
- Expel air from syringe prior to administration. For a dose of 0.25 mg, use the entire contents (5mL) of the prefilled syringe.
- Do not use the prefilled syringe to administer a dose less than 0.25mg (5mL)
- Use aseptic technique while handling the syringe
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
In a 104-week carcinogenicity study in CD-1 mice, animals were treated with oral doses of palonosetron HCl at 10, 30 and 60 mg/kg/day. Treatment with palonosetron was not tumorigenic. The highest tested dose produced a systemic exposure to palonosetron (Plasma AUC) of about 150 to 289 times the human exposure (AUC= 29.8 h•mcg/L) at the recommended intravenous dose of 0.25 mg. In a 104-week carcinogenicity study in Sprague-Dawley rats, male and female rats were treated with oral doses of 15, 30 and 60 mg/kg/day and 15, 45 and 90 mg/kg/day, respectively. The highest doses produced a systemic exposure to palonosetron (Plasma AUC) of 137 and 308 times the human exposure at the recommended dose. Treatment with palonosetron produced increased incidences of adrenal benign pheochromocytoma and combined benign and malignant pheochromocytoma, increased incidences of pancreatic Islet cell adenoma and combined adenoma and carcinoma and pituitary adenoma in male rats. In female rats, it produced hepatocellular adenoma and carcinoma and increased the incidences of thyroid C-cell adenoma and combined adenoma and carcinoma.
Palonosetron was not genotoxic in the Ames test, the Chinese hamster ovarian cell (CHO/HGPRT) forward mutation test, the ex vivo hepatocyte unscheduled DNA synthesis (UDS) test or the mouse micronucleus test. It was, however, positive for clastogenic effects in the Chinese hamster ovarian (CHO) cell chromosomal aberration test.
Palonosetron HCl at oral doses up to 60 mg/kg/day (about 1,894 times the recommended human intravenous dose based on body surface area) was found to have no effect on fertility and reproductive performance of male and female rats.
14.1 Prevention of Nausea and Vomiting Associated With Mec and Hec in Adults
Efficacy of a single intravenous dose of Palonosetron Hydrochloride Injection in preventing acute and delayed nausea and vomiting associated with MEC or HEC were studied in 4 trials. In these double-blind studies, complete response rates (no emetic episodes and no rescue medication) and other efficacy parameters were assessed through at least 120 hours after administration of chemotherapy.
The safety and efficacy of Palonosetron Hydrochloride Injection in repeated courses of chemotherapy was also assessed.
14.2 Prevention of Nausea and Vomiting Associated With Emetogenic Chemotherapy, Including Hec in Pediatric Patients
One double-blind, active-controlled clinical trial was conducted in pediatric cancer patients. The total population (N = 327) had a mean age of 8.3 years (range 2 months to 16.9 years) and were 53% male; and 96% white. Patients were randomized and received a 20 mcg/kg (maximum 1.5 mg) intravenous infusion of Palonosetron Hydrochloride Injection 30 minutes prior to the start of emetogenic chemotherapy (followed by placebo infusions 4 and 8 hours after the dose of Palonosetron Hydrochloride injection) or 0.15 mg/kg of intravenous ondansetron 30 minutes prior to the start of emetogenic chemotherapy (followed by ondansetron 0.15 mg/kg infusions 4 and 8 hours after the first dose of ondansetron, with a maximum total dose of 32 mg). Emetogenic chemotherapies administered included doxorubicin, cyclophosphamide (< 1,500 mg/m2), ifosfamide, cisplatin, dactinomycin, carboplatin, and daunorubicin. Adjuvant corticosteroids, including dexamethasone, were administered with chemotherapy in 55% of patients.
Complete Response in the acute phase of the first cycle of chemotherapy was defined as no vomiting, no retching, and no rescue medication in the first 24 hours after starting chemotherapy. Efficacy was based on demonstrating non-inferiority of intravenous Palonosetron Hydrochloride injection compared to intravenous ondansetron. Non-inferiority criteria were met if the lower bound of the 97.5% confidence interval for the difference in Complete Response rates of intravenous Palonosetron Hydrochloride injection minus intravenous ondansetron was larger than -15%. The non-inferiority margin was 15%.
Structured Label Content
Section 42229-5 (42229-5)
Prevention of Chemotherapy-Induced Nausea and Vomiting
The recommended dosage of Palonosetron Hydrochloride Injection for prevention of nausea and vomiting associated with HEC and MEC in adults and associated with emetogenic chemotherapy, including HEC in pediatric patients 1 month to less than 17 years of age is shown in Table 1.
|
*Note different dosing units in pediatrics |
||
| Age | Dose* | Infusion Time |
| Adults | 0.25 mg as a single dose | Infuse over 30 seconds beginning approximately 30 minutes before the start of chemotherapy |
| Pediatrics (1 month to less than 17 years) | 20 micrograms per kilogram (max 1.5 mg) as a single dose | Infuse over 15 minutes beginning approximately 30 minutes before the start of chemotherapy |
Section 42230-3 (42230-3)
|
This Patient Information has been approved by the U.S. Food and Drug Administration. |
|||
|
Patient Information Palonosetron Hydrochloride
(PAL-oh-NOE-se-tron HYE-dro-KLOR-ide) Injection for Intravenous Use |
|||
| Read this Patient Information before you receive Palonosetron Hydrochloride Injection and each time you receive Palonosetron Hydrochloride Injection. There may be new information. This information does not take the place of talking with your doctor about your medical condition or your treatment. | |||
|
What is Palonosetron Hydrochloride Injection?
Palonosetron Hydrochloride Injection is a prescription medicine called an “antiemetic.” Palonosetron Hydrochloride Injection is used in adults to help prevent the nausea and vomiting that happens:
|
|||
Palonosetron Hydrochloride Injection is used in children 1 month old to less than 17 years of age to help prevent the nausea and vomiting that happens right away with certain anti-cancer medicines (chemotherapy).
|
|||
|
Who should not receive Palonosetron Hydrochloride Injection?
Do not receive Palonosetron Hydrochloride Injection if you are allergic to palonosetron hydrochloride or any of the ingredients in Palonosetron Hydrochloride Injection. See the end of this leaflet for a complete list of ingredients in Palonosetron Hydrochloride Injection. |
|||
|
What should I tell my doctor before receiving Palonosetron Hydrochloride Injection?
Before receiving Palonosetron Hydrochloride Injection, tell your doctor about all of your medical conditions, including if you:
|
|||
|
Tell your doctor about all of the medicines you take, including prescription and over-the-counter medicines, vitamins and herbal supplements. Palonosetron Hydrochloride Injection and certain other medicines can affect each other, causing serious side effects. |
|||
How will I receive Palonosetron Hydrochloride injection?
|
|||
What are the possible side effects of Palonosetron Hydrochloride Injection? Palonosetron Hydrochloride Injection may cause serious side effects, including:
|
|||
|
|
|
|
|
|||
|
|
||
|
The most common side effects in adults who receive Palonosetron Hydrochloride Injection to help prevent nausea and vomiting that happens with certain anti-cancer medicine (chemotherapy) include: headache and constipation. These are not all the possible side effects from Palonosetron Hydrochloride Injection. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
|||
| General information about the safe and effective use of Palonosetron Hydrochloride Injection Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. You can ask your doctor or pharmacist for information about Palonosetron Hydrochloride Injection that is written for health professionals. | |||
|
What are the ingredients in Palonosetron Hydrochloride Injection?
Lake Zurich, IL 60047 Made in Austria For more information, go to www.fresenius-kabi.com/us or call 1-800-551-7176. |
Section 44425-7 (44425-7)
Storage
- Store at 20° to 25°C (68° to 77°F) [see USP Controlled Room Temperature].
- Protect from freezing.
- Protect from light.
- Discard unused portion
Section 51945-4 (51945-4)
PACKAGE LABEL - PRINCIPAL DISPLAY PANEL - Palonosetron Hydrochloride 0.25 mg Single-Dose Prefilled Syringe Label
NDC 63323-673-21
Palonosetron Hydrochloride Injection
0.25 mg* per 5 mL
(0.05 mg* per mL)
For Intravenous Injection Only.
Discard Unused Portion.
5 mL
Single-Dose Syringe
Rx only
10. Overdosage (10. OVERDOSAGE)
There is no known antidote to palonosetron. Overdose should be managed with supportive care.
Dialysis studies have not been performed, however, due to the large volume of distribution, dialysis is unlikely to be an effective treatment for palonosetron overdose. A single intravenous dose of palonosetron (HCl) at 30 mg/kg (947 and 474 times the human dose for rats and mice, respectively, based on body surface area) was lethal to rats and mice. The major signs of toxicity were convulsions, gasping, pallor, cyanosis and collapse.
11. Description (11. DESCRIPTION)
Palonosetron Hydrochloride Injection contains palonosetron as palonosetron HCl, an antiemetic and antinauseant agent. It is a serotonin-3 (5-HT3) receptor antagonist with a strong binding affinity for this receptor. Chemically, palonosetron hydrochloride is: (3aS)-2-[(S)- 1-Azabicyclo [2.2.2]oct-3- yl]-2,3,3a,4,5,6-hexahydro-1-oxo-1Hbenz[de] isoquinoline hydrochloride. Palonosetron hydrochloride exists as a single isomer and has the following structural formula:
| C 19 H 24 N 2 O•HCl | M.W. 332.87 |
Palonosetron Hydrochloride is a white to off-white crystalline powder. It is freely soluble in water, soluble in propylene glycol, and slightly soluble in ethanol and 2-propanol.
Palonosetron Hydrochloride Injection is a sterile, clear, colorless, non-pyrogenic, isotonic, buffered solution for intravenous administration. Palonosetron Hydrochloride Injection is available as a 5 mL single-dose prefilled syringe.
Each 5 mL syringe contains: 0.25 mg palonosetron (equivalent to 0.28 mg palonosetron HCl), 202.4 mg mannitol, 2.5 mg edetate disodium dihydrate, 18 mg trisodium citrate dihydrate, and 7 mg citric acid anhydrous in water for intravenous administration.
The pH of the solution in the 5 mL syringe is 4.5 to 5.5.
8.5 Geriatric Use
Of the 1,374 adult cancer patients in clinical studies of intravenously administered palonosetron HCl, 316 (23%) were 65 years and over, while 71 (5%) were at least 75 years and over. Of the 1,520 adult patients in clinical studies of intravenously administered palonosetron HCl, 73 (5%) were at least 65 years old [see Clinical Studies (14.1)]. No overall differences in safety or effectiveness were observed between these subjects and younger subjects, but greater sensitivity in some older individuals cannot be ruled out. Population pharmacokinetics analysis did not reveal any differences in palonosetron pharmacokinetics between cancer patients 65 years of age and older compared to younger patients [see Clinical Pharmacology (12.3)]. No dose adjustment is required for geriatric patients.
4. Contraindications (4. CONTRAINDICATIONS)
Palonosetron Hydrochloride Injection is contraindicated in patients known to have hypersensitivity to palonosetron [see Warnings and Precautions (5.1)].
6. Adverse Reactions (6. ADVERSE REACTIONS)
7. Drug Interactions (7. DRUG INTERACTIONS)
Serotonergic Drugs: Monitor for serotonin syndrome; if symptoms occur, discontinue Palonosetron Hydrochloride Injection and initiate supportive treatment. (7.1)
12.3 Pharmacokinetics
After intravenous dosing of palonosetron HCl in healthy subjects and cancer patients, an initial decline in palonosetron plasma concentrations is followed by a slow elimination from the body. Mean maximum plasma concentration (Cmax) and area under the concentration-time curve (AUC0-∞) are generally dose-proportional over the dose range of 0.3 to 90 mcg/kg in healthy subjects and in cancer patients. Following a single intravenous dose of palonosetron HCl at 3 mcg/kg (or 0.21 mg/70 kg) to six cancer patients, mean (±SD) maximum plasma concentration was estimated to be 5,630 ± 5,480 ng/L and mean AUC was 35.8 ± 20.9 h•mcg/L.
Following intravenous administration of Palonosetron Hydrochloride Injection 0.25 mg once every other day for 3 doses in 11 cancer patients, the mean increase in plasma palonosetron concentration from Day 1 to Day 5 was 42±34%. Following intravenous administration of Palonosetron Hydrochloride Injection 0.25 mg once daily for 3 days in 12 healthy subjects, the mean (±SD) increase in plasma palonosetron concentration from Day 1 to Day 3 was 110±45%.
5.2 Serotonin Syndrome
The development of serotonin syndrome has been reported with 5-HT3 receptor antagonists. Most reports have been associated with concomitant use of serotonergic drugs (e.g., selective serotonin reuptake inhibitors (SSRIs), serotonin and norepinephrine reuptake inhibitors (SNRIs), monoamine oxidase inhibitors, mirtazapine, fentanyl, lithium, tramadol, and intravenous methylene blue). Some of the reported cases were fatal. Serotonin syndrome occurring with overdose of another 5-HT3 receptor antagonist alone has also been reported. The majority of reports of serotonin syndrome related to 5-HT3 receptor antagonist use occurred in a post-anesthesia care unit or an infusion center.
Symptoms associated with serotonin syndrome may include the following combination of signs and symptoms: mental status changes (e.g. agitation, hallucinations, delirium, and coma), autonomic instability (e.g., tachycardia, labile blood pressure, dizziness, diaphoresis, flushing, hyperthermia), neuromuscular symptoms (e.g., tremor, rigidity, myoclonus, hyperreflexia, incoordination), seizures, with or without gastrointestinal symptoms (e.g., nausea, vomiting, diarrhea). Patients should be monitored for the emergence of serotonin syndrome, especially with concomitant use of Palonosetron Hydrochloride Injection and other serotonergic drugs. If symptoms of serotonin syndrome occur, discontinue Palonosetron Hydrochloride Injection and initiate supportive treatment. Patients should be informed of the increased risk of serotonin syndrome, especially if Palonosetron Hydrochloride Injection is used concomitantly with other serotonergic drugs [see Drug Interactions (7.1)].
7.1 Serotonergic Drugs
Serotonin syndrome (including altered mental status, autonomic instability, and neuromuscular symptoms) has been described following the concomitant use of 5-HT3 receptor antagonists and other serotonergic drugs, including selective serotonin reuptake inhibitors (SSRIs) and serotonin and noradrenaline reuptake inhibitors (SNRIs). Monitor for the emergence of serotonin syndrome. If symptoms occur, discontinue Palonosetron Hydrochloride Injection and initiate supportive treatment [see Warnings and Precautions (5.2)].
1. Indications and Usage (1. INDICATIONS AND USAGE)
Palonosetron Hydrochloride Injection is indicated in adults for prevention of:
- acute and delayed nausea and vomiting associated with initial and repeat courses of moderately emetogenic cancer chemotherapy (MEC).
- acute nausea and vomiting associated with initial and repeat courses highly emetogenic cancer chemotherapy (HEC).
Palonosetron Hydrochloride Injection is indicated in pediatric patients 1 month to less than 17 years of age for prevention of:
- acute nausea and vomiting associated with initial and repeat courses of emetogenic cancer chemotherapy, including highly emetogenic cancer chemotherapy.
12.1 Mechanism of Action
Palonosetron is a 5-HT3 receptor antagonist with a strong binding affinity for this receptor and little or no affinity for other receptors.
Cancer chemotherapy may be associated with a high incidence of nausea and vomiting, particularly when certain agents, such as cisplatin, are used. 5-HT3 receptors are located on the nerve terminals of the vagus in the periphery and centrally in the chemoreceptor trigger zone of the area postrema. It is thought that chemotherapeutic agents produce nausea and vomiting by releasing serotonin from the enterochromaffin cells of the small intestine and that the released serotonin then activates 5-HT3 receptors located on vagal afferents to initiate the vomiting reflex.
5. Warnings and Precautions (5. WARNINGS AND PRECAUTIONS)
- Hypersensitivity reactions, including anaphylaxis and anaphylactic shock: reported in patients with or without known hypersensitivity to other selective 5-HT3 receptor antagonists. If symptoms occur, discontinue Palonosetron Hydrochloride Injection and initiate appropriate medical treatment. (5.1)
- Serotonin syndrome: reported with 5-HT3 receptor antagonists alone, but particularly with concomitant use of serotonergic drugs. (5.2, 7.1)
2. Dosage and Administration (2. DOSAGE AND ADMINISTRATION)
Chemotherapy-Induced Nausea and Vomiting (2.1)
|
*Note different dosing units in pediatrics |
||
| Age | Dose* | Infusion Time |
| Adults | 0.25 mg as a single dose | Infuse over 30 seconds beginning approximately 30 minutes before the start of chemotherapy |
| Pediatrics (1 month to less than 17 years) |
20 micrograms per
kilogram (maximum 1.5 mg) as a single dose |
Infuse over 15 minutes beginning approximately 30 minutes before the start of chemotherapy |
Instructions for Intravenous Administration
- For a dose of 0.25 mg, use the entire contents (5 mL) of the prefilled syringe. Do not use the prefilled syringe to administer a dose of less than 0.25 mg (5 mL). (2.2)
3. Dosage Form and Strengths (3. DOSAGE FORM AND STRENGTHS)
Palonosetron Hydrochloride Injection is supplied as a single-dose sterile, clear, colorless solution in a prefilled syringe that provides:
- 0.25 mg (free base) per 5 mL (concentration: 0.05 mg per mL, 50 mcg per mL)
6.2 Postmarketing Experience
The following adverse reactions have been identified during postapproval use of palonosetron HCl. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
- Hypersensitivity reactions: including dyspnea, bronchospasm, swelling/edema, erythema, pruritus, rash, urticaria, anaphylaxis and anaphylactic shock [see Warnings and Precautions (5.1)]
- Injection site reactions: including burning, induration, discomfort and pain
5.1 Hypersensitivity Reactions
Hypersensitivity reactions, including anaphylaxis and anaphylactic shock, have been reported with administration of Palonosetron Hydrochloride Injection [see Adverse Reactions (6.2)]. These reactions occurred in patients with or without known hypersensitivity to other 5-HT3 receptor antagonists. If hypersensitivity reactions occur, discontinue Palonosetron Hydrochloride Injection and initiate appropriate medical treatment. Do not reinitiate Palonosetron Hydrochloride Injection in patients who have previously experienced symptoms of hypersensitivity [see Contraindications (4)].
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
17. Patient Counseling Information (17. PATIENT COUNSELING INFORMATION)
Advise the patient or caregiver to read the FDA-approved patient labeling (Patient Information).
16. How Supplied/storage and Handling (16. HOW SUPPLIED/STORAGE AND HANDLING)
Palonosetron Hydrochloride Injection 0.25 mg/5 mL (free base) single-dose prefilled syringe is available as follows:
|
Product
No. |
NDC
No. |
Strength |
Package |
| 673189 | 63323-673-89 | 0.25 mg per 5 mL (0.05 mg per mL) |
5 mL single-dose prefilled syringe, individually packaged in cartons of ten. |
2.2 Instructions for Intravenous Administration
- Palonosetron Hydrochloride Injection is supplied ready for intravenous administration at a concentration of 0.05 mg/mL (50 mcg/mL).
- Do not mix Palonosetron Hydrochloride Injection with other drugs.
- Flush the infusion line with normal saline before and after administration of Palonosetron Hydrochloride Injection.
- Inspect Palonosetron Hydrochloride Injection visually for particulate matter and discoloration before administration.
- Expel air from syringe prior to administration. For a dose of 0.25 mg, use the entire contents (5mL) of the prefilled syringe.
- Do not use the prefilled syringe to administer a dose less than 0.25mg (5mL)
- Use aseptic technique while handling the syringe
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
In a 104-week carcinogenicity study in CD-1 mice, animals were treated with oral doses of palonosetron HCl at 10, 30 and 60 mg/kg/day. Treatment with palonosetron was not tumorigenic. The highest tested dose produced a systemic exposure to palonosetron (Plasma AUC) of about 150 to 289 times the human exposure (AUC= 29.8 h•mcg/L) at the recommended intravenous dose of 0.25 mg. In a 104-week carcinogenicity study in Sprague-Dawley rats, male and female rats were treated with oral doses of 15, 30 and 60 mg/kg/day and 15, 45 and 90 mg/kg/day, respectively. The highest doses produced a systemic exposure to palonosetron (Plasma AUC) of 137 and 308 times the human exposure at the recommended dose. Treatment with palonosetron produced increased incidences of adrenal benign pheochromocytoma and combined benign and malignant pheochromocytoma, increased incidences of pancreatic Islet cell adenoma and combined adenoma and carcinoma and pituitary adenoma in male rats. In female rats, it produced hepatocellular adenoma and carcinoma and increased the incidences of thyroid C-cell adenoma and combined adenoma and carcinoma.
Palonosetron was not genotoxic in the Ames test, the Chinese hamster ovarian cell (CHO/HGPRT) forward mutation test, the ex vivo hepatocyte unscheduled DNA synthesis (UDS) test or the mouse micronucleus test. It was, however, positive for clastogenic effects in the Chinese hamster ovarian (CHO) cell chromosomal aberration test.
Palonosetron HCl at oral doses up to 60 mg/kg/day (about 1,894 times the recommended human intravenous dose based on body surface area) was found to have no effect on fertility and reproductive performance of male and female rats.
14.1 Prevention of Nausea and Vomiting Associated With Mec and Hec in Adults (14.1 Prevention of Nausea and Vomiting Associated with MEC and HEC in Adults)
Efficacy of a single intravenous dose of Palonosetron Hydrochloride Injection in preventing acute and delayed nausea and vomiting associated with MEC or HEC were studied in 4 trials. In these double-blind studies, complete response rates (no emetic episodes and no rescue medication) and other efficacy parameters were assessed through at least 120 hours after administration of chemotherapy.
The safety and efficacy of Palonosetron Hydrochloride Injection in repeated courses of chemotherapy was also assessed.
14.2 Prevention of Nausea and Vomiting Associated With Emetogenic Chemotherapy, Including Hec in Pediatric Patients (14.2 Prevention of Nausea and Vomiting Associated with Emetogenic Chemotherapy, Including HEC in Pediatric Patients)
One double-blind, active-controlled clinical trial was conducted in pediatric cancer patients. The total population (N = 327) had a mean age of 8.3 years (range 2 months to 16.9 years) and were 53% male; and 96% white. Patients were randomized and received a 20 mcg/kg (maximum 1.5 mg) intravenous infusion of Palonosetron Hydrochloride Injection 30 minutes prior to the start of emetogenic chemotherapy (followed by placebo infusions 4 and 8 hours after the dose of Palonosetron Hydrochloride injection) or 0.15 mg/kg of intravenous ondansetron 30 minutes prior to the start of emetogenic chemotherapy (followed by ondansetron 0.15 mg/kg infusions 4 and 8 hours after the first dose of ondansetron, with a maximum total dose of 32 mg). Emetogenic chemotherapies administered included doxorubicin, cyclophosphamide (< 1,500 mg/m2), ifosfamide, cisplatin, dactinomycin, carboplatin, and daunorubicin. Adjuvant corticosteroids, including dexamethasone, were administered with chemotherapy in 55% of patients.
Complete Response in the acute phase of the first cycle of chemotherapy was defined as no vomiting, no retching, and no rescue medication in the first 24 hours after starting chemotherapy. Efficacy was based on demonstrating non-inferiority of intravenous Palonosetron Hydrochloride injection compared to intravenous ondansetron. Non-inferiority criteria were met if the lower bound of the 97.5% confidence interval for the difference in Complete Response rates of intravenous Palonosetron Hydrochloride injection minus intravenous ondansetron was larger than -15%. The non-inferiority margin was 15%.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:35:52.434095 · Updated: 2026-03-14T21:44:38.795279