These Highlights Do Not Include All The Information Needed To Use Invokana Safely And Effectively. See Full Prescribing Information For Invokana.
b9057d3b-b104-4f09-8a61-c61ef9d4a3f3
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Indications and Usage ( 1 ) 12/2024 Dosage and Administration ( 2.2 , 2.3 ) 12/2024 Dosage and Administration ( 2.5 ) 08/2024 Warnings and Precautions ( 5.1 ) 08/2024 Warnings and Precautions ( 5.2 ) 08/2024
Indications and Usage
INVOKANA (canagliflozin) is indicated: as an adjunct to diet and exercise to improve glycemic control in adults and pediatric patients aged 10 years and older with type 2 diabetes mellitus. to reduce the risk of major adverse cardiovascular events (cardiovascular death, nonfatal myocardial infarction and nonfatal stroke) in adults with type 2 diabetes mellitus and established cardiovascular disease (CVD). to reduce the risk of end-stage kidney disease (ESKD), doubling of serum creatinine, cardiovascular (CV) death, and hospitalization for heart failure in adults with type 2 diabetes mellitus and diabetic nephropathy with albuminuria greater than 300 mg/day.
Dosage and Administration
Assess renal function before initiating and as clinically indicated. Assess volume status and correct volume depletion before initiating ( 2.1 ). The recommended starting dosage in adults and pediatric patients aged 10 years and older with type 2 diabetes mellitus is 100 mg orally once daily, taken before the first meal of the day to improve glycemic control. The dosage can be increased to 300 mg once daily in patients tolerating 100 mg once daily who have an eGFR of 60 mL/min/1.73 m 2 or greater and require additional glycemic control ( 2.2 ). For all other indications in adults, the recommended dosage of INVOKANA is 100 mg orally once daily ( 2.2 ). Dosage adjustments for patients with renal impairment may be required ( 2.3 ). See full prescribing information for INVOKANA dosage modifications due to drug interactions ( 2.4 ). Withhold INVOKANA at least 3 days, if possible, prior to surgery or procedures associated with prolonged fasting ( 2.5 ).
Warnings and Precautions
Diabetic Ketoacidosis in Patients with Type 1 Diabetes Mellitus and Other Ketoacidosis : Consider ketone monitoring in patients at risk for ketoacidosis, as indicated. Assess for ketoacidosis regardless of presenting blood glucose levels and discontinue INVOKANA if ketoacidosis is suspected. Monitor patients for resolution of ketoacidosis before restarting ( 5.1 ). Lower Limb Amputation : Monitor patients for infection or ulcers of lower limb and discontinue if these occur ( 5.2 ). Volume Depletion : May result in acute kidney injury. Before initiating INVOKANA, assess and correct volume status in patients with renal impairment, elderly patients, or patients on loop diuretics. Monitor for signs and symptoms during therapy ( 5.3 ). Urosepsis and Pyelonephritis : Evaluate patients for signs and symptoms of urinary tract infections and treat promptly, if indicated ( 5.4 ). Hypoglycemia with Concomitant Use with Insulin or Insulin Secretagogues : Consider a lower dose of insulin or the insulin secretagogue to reduce the risk of hypoglycemia when used in combination with INVOKANA ( 5.5 ). Necrotizing Fasciitis of the Perineum (Fournier's Gangrene) : Serious, life-threatening cases have occurred in both females and males. Assess patients presenting with pain or tenderness, erythema, or swelling in the genital or perineal area, along with fever or malaise. If suspected, institute prompt treatment ( 5.6 ). Genital Mycotic Infections : Monitor and treat if indicated ( 5.7 ). Hypersensitivity Reactions : Discontinue INVOKANA and monitor until signs and symptoms resolve ( 5.8 ). Bone Fracture : Consider factors that contribute to fracture risk before initiating INVOKANA ( 5.9 ).
Contraindications
INVOKANA is contraindicated in patients with a serious hypersensitivity reaction to INVOKANA, such as anaphylaxis or angioedema [see Warnings and Precautions (5.8) and Adverse Reactions (6.1 , 6.2) ] .
Adverse Reactions
The following important adverse reactions are described below and elsewhere in the labeling: Diabetic Ketoacidosis in Patients with Type 1 Diabetes and Other Ketoacidosis [see Warnings and Precautions (5.1) ] Lower Limb Amputation [see Warnings and Precautions (5.2) ] Volume Depletion [see Warnings and Precautions (5.3) ] Urosepsis and Pyelonephritis [see Warnings and Precautions (5.4) ] Hypoglycemia with Concomitant Use with Insulin or Insulin Secretagogues [see Warnings and Precautions (5.5) ] Necrotizing Fasciitis of the Perineum (Fournier's gangrene) [see Warnings and Precautions (5.6) ] Genital Mycotic Infections [see Warnings and Precautions (5.7) ] Hypersensitivity Reactions [see Warnings and Precautions (5.8) ] Bone Fracture [see Warnings and Precautions (5.9) ]
Drug Interactions
Table 7: Clinically Significant Drug Interactions with INVOKANA UGT Enzyme Inducers Clinical Impact: UGT enzyme inducers decrease canagliflozin exposure which may reduce the effectiveness of INVOKANA. Intervention: For patients with eGFR 60 mL/min/1.73 m 2 or greater, if an inducer of UGTs is administered with INVOKANA, increase the dosage to 200 mg daily in patients currently tolerating INVOKANA 100 mg daily. The total daily dosage may be increased to 300 mg daily in patients currently tolerating INVOKANA 200 mg daily who require additional glycemic control. For patients with eGFR less than 60 mL/min/1.73 m 2 , if an inducer of UGTs is administered with INVOKANA, increase the dosage to 200 mg daily in patients currently tolerating INVOKANA 100 mg daily. Consider adding another antihyperglycemic agent in patients who require additional glycemic control [see Dosage and Administration (2.3) and Clinical Pharmacology (12.3) ] . Examples: Rifampin, phenytoin, phenobarbital, ritonavir Insulin or Insulin Secretagogues Clinical Impact: The risk of hypoglycemia is increased when INVOKANA is used concomitantly with insulin secretagogues (e.g., sulfonylurea) or insulin. Intervention: Concomitant use may require a lower dosage of the insulin secretagogue or insulin to reduce the risk of hypoglycemia. Digoxin Clinical Impact: Canagliflozin increases digoxin exposure [see Clinical Pharmacology (12.3) ] . Intervention: Monitor patients taking INVOKANA with concomitant digoxin for a need to adjust the dosage of digoxin. Lithium Clinical Impact: Concomitant use of an SGLT2 inhibitor with lithium may decrease serum lithium concentrations. Intervention: Monitor serum lithium concentration more frequently during INVOKANA initiation and dosage changes. Drug/Laboratory Test Interference Positive Urine Glucose Test Clinical Impact: SGLT2 inhibitors increase urinary glucose excretion which will lead to positive urine glucose tests. Intervention: Monitoring glycemic control with urine glucose tests is not recommended in patients taking SGLT2 inhibitors. Use alternative methods to monitor glycemic control. Interference with 1,5-anhydroglucitol (1,5-AG) Assay Clinical Impact: Measurements of 1,5-AG are unreliable in assessing glycemic control in patients taking SGLT2 inhibitors. Intervention: Monitoring glycemic control with 1,5-AG assay is not recommended in patients taking SGLT2 inhibitors. Use alternative methods to monitor glycemic control.
Storage and Handling
INVOKANA ® (canagliflozin) tablets are available in the strengths and packages listed below: 100 mg tablets are yellow, capsule-shaped, film-coated tablets with "CFZ" on one side and "100" on the other side. NDC 50458-140-30 Bottle of 30 NDC 50458-140-90 Bottle of 90 NDC 50458-140-50 Bottle of 500 300 mg tablets are white, capsule-shaped, film-coated tablets with "CFZ" on one side and "300" on the other side. NDC 50458-141-30 Bottle of 30 NDC 50458-141-90 Bottle of 90 NDC 50458-141-50 Bottle of 500
How Supplied
INVOKANA ® (canagliflozin) tablets are available in the strengths and packages listed below: 100 mg tablets are yellow, capsule-shaped, film-coated tablets with "CFZ" on one side and "100" on the other side. NDC 50458-140-30 Bottle of 30 NDC 50458-140-90 Bottle of 90 NDC 50458-140-50 Bottle of 500 300 mg tablets are white, capsule-shaped, film-coated tablets with "CFZ" on one side and "300" on the other side. NDC 50458-141-30 Bottle of 30 NDC 50458-141-90 Bottle of 90 NDC 50458-141-50 Bottle of 500
Medication Information
Warnings and Precautions
Diabetic Ketoacidosis in Patients with Type 1 Diabetes Mellitus and Other Ketoacidosis : Consider ketone monitoring in patients at risk for ketoacidosis, as indicated. Assess for ketoacidosis regardless of presenting blood glucose levels and discontinue INVOKANA if ketoacidosis is suspected. Monitor patients for resolution of ketoacidosis before restarting ( 5.1 ). Lower Limb Amputation : Monitor patients for infection or ulcers of lower limb and discontinue if these occur ( 5.2 ). Volume Depletion : May result in acute kidney injury. Before initiating INVOKANA, assess and correct volume status in patients with renal impairment, elderly patients, or patients on loop diuretics. Monitor for signs and symptoms during therapy ( 5.3 ). Urosepsis and Pyelonephritis : Evaluate patients for signs and symptoms of urinary tract infections and treat promptly, if indicated ( 5.4 ). Hypoglycemia with Concomitant Use with Insulin or Insulin Secretagogues : Consider a lower dose of insulin or the insulin secretagogue to reduce the risk of hypoglycemia when used in combination with INVOKANA ( 5.5 ). Necrotizing Fasciitis of the Perineum (Fournier's Gangrene) : Serious, life-threatening cases have occurred in both females and males. Assess patients presenting with pain or tenderness, erythema, or swelling in the genital or perineal area, along with fever or malaise. If suspected, institute prompt treatment ( 5.6 ). Genital Mycotic Infections : Monitor and treat if indicated ( 5.7 ). Hypersensitivity Reactions : Discontinue INVOKANA and monitor until signs and symptoms resolve ( 5.8 ). Bone Fracture : Consider factors that contribute to fracture risk before initiating INVOKANA ( 5.9 ).
Indications and Usage
INVOKANA (canagliflozin) is indicated: as an adjunct to diet and exercise to improve glycemic control in adults and pediatric patients aged 10 years and older with type 2 diabetes mellitus. to reduce the risk of major adverse cardiovascular events (cardiovascular death, nonfatal myocardial infarction and nonfatal stroke) in adults with type 2 diabetes mellitus and established cardiovascular disease (CVD). to reduce the risk of end-stage kidney disease (ESKD), doubling of serum creatinine, cardiovascular (CV) death, and hospitalization for heart failure in adults with type 2 diabetes mellitus and diabetic nephropathy with albuminuria greater than 300 mg/day.
Dosage and Administration
Assess renal function before initiating and as clinically indicated. Assess volume status and correct volume depletion before initiating ( 2.1 ). The recommended starting dosage in adults and pediatric patients aged 10 years and older with type 2 diabetes mellitus is 100 mg orally once daily, taken before the first meal of the day to improve glycemic control. The dosage can be increased to 300 mg once daily in patients tolerating 100 mg once daily who have an eGFR of 60 mL/min/1.73 m 2 or greater and require additional glycemic control ( 2.2 ). For all other indications in adults, the recommended dosage of INVOKANA is 100 mg orally once daily ( 2.2 ). Dosage adjustments for patients with renal impairment may be required ( 2.3 ). See full prescribing information for INVOKANA dosage modifications due to drug interactions ( 2.4 ). Withhold INVOKANA at least 3 days, if possible, prior to surgery or procedures associated with prolonged fasting ( 2.5 ).
Contraindications
INVOKANA is contraindicated in patients with a serious hypersensitivity reaction to INVOKANA, such as anaphylaxis or angioedema [see Warnings and Precautions (5.8) and Adverse Reactions (6.1 , 6.2) ] .
Adverse Reactions
The following important adverse reactions are described below and elsewhere in the labeling: Diabetic Ketoacidosis in Patients with Type 1 Diabetes and Other Ketoacidosis [see Warnings and Precautions (5.1) ] Lower Limb Amputation [see Warnings and Precautions (5.2) ] Volume Depletion [see Warnings and Precautions (5.3) ] Urosepsis and Pyelonephritis [see Warnings and Precautions (5.4) ] Hypoglycemia with Concomitant Use with Insulin or Insulin Secretagogues [see Warnings and Precautions (5.5) ] Necrotizing Fasciitis of the Perineum (Fournier's gangrene) [see Warnings and Precautions (5.6) ] Genital Mycotic Infections [see Warnings and Precautions (5.7) ] Hypersensitivity Reactions [see Warnings and Precautions (5.8) ] Bone Fracture [see Warnings and Precautions (5.9) ]
Drug Interactions
Table 7: Clinically Significant Drug Interactions with INVOKANA UGT Enzyme Inducers Clinical Impact: UGT enzyme inducers decrease canagliflozin exposure which may reduce the effectiveness of INVOKANA. Intervention: For patients with eGFR 60 mL/min/1.73 m 2 or greater, if an inducer of UGTs is administered with INVOKANA, increase the dosage to 200 mg daily in patients currently tolerating INVOKANA 100 mg daily. The total daily dosage may be increased to 300 mg daily in patients currently tolerating INVOKANA 200 mg daily who require additional glycemic control. For patients with eGFR less than 60 mL/min/1.73 m 2 , if an inducer of UGTs is administered with INVOKANA, increase the dosage to 200 mg daily in patients currently tolerating INVOKANA 100 mg daily. Consider adding another antihyperglycemic agent in patients who require additional glycemic control [see Dosage and Administration (2.3) and Clinical Pharmacology (12.3) ] . Examples: Rifampin, phenytoin, phenobarbital, ritonavir Insulin or Insulin Secretagogues Clinical Impact: The risk of hypoglycemia is increased when INVOKANA is used concomitantly with insulin secretagogues (e.g., sulfonylurea) or insulin. Intervention: Concomitant use may require a lower dosage of the insulin secretagogue or insulin to reduce the risk of hypoglycemia. Digoxin Clinical Impact: Canagliflozin increases digoxin exposure [see Clinical Pharmacology (12.3) ] . Intervention: Monitor patients taking INVOKANA with concomitant digoxin for a need to adjust the dosage of digoxin. Lithium Clinical Impact: Concomitant use of an SGLT2 inhibitor with lithium may decrease serum lithium concentrations. Intervention: Monitor serum lithium concentration more frequently during INVOKANA initiation and dosage changes. Drug/Laboratory Test Interference Positive Urine Glucose Test Clinical Impact: SGLT2 inhibitors increase urinary glucose excretion which will lead to positive urine glucose tests. Intervention: Monitoring glycemic control with urine glucose tests is not recommended in patients taking SGLT2 inhibitors. Use alternative methods to monitor glycemic control. Interference with 1,5-anhydroglucitol (1,5-AG) Assay Clinical Impact: Measurements of 1,5-AG are unreliable in assessing glycemic control in patients taking SGLT2 inhibitors. Intervention: Monitoring glycemic control with 1,5-AG assay is not recommended in patients taking SGLT2 inhibitors. Use alternative methods to monitor glycemic control.
Storage and Handling
INVOKANA ® (canagliflozin) tablets are available in the strengths and packages listed below: 100 mg tablets are yellow, capsule-shaped, film-coated tablets with "CFZ" on one side and "100" on the other side. NDC 50458-140-30 Bottle of 30 NDC 50458-140-90 Bottle of 90 NDC 50458-140-50 Bottle of 500 300 mg tablets are white, capsule-shaped, film-coated tablets with "CFZ" on one side and "300" on the other side. NDC 50458-141-30 Bottle of 30 NDC 50458-141-90 Bottle of 90 NDC 50458-141-50 Bottle of 500
How Supplied
INVOKANA ® (canagliflozin) tablets are available in the strengths and packages listed below: 100 mg tablets are yellow, capsule-shaped, film-coated tablets with "CFZ" on one side and "100" on the other side. NDC 50458-140-30 Bottle of 30 NDC 50458-140-90 Bottle of 90 NDC 50458-140-50 Bottle of 500 300 mg tablets are white, capsule-shaped, film-coated tablets with "CFZ" on one side and "300" on the other side. NDC 50458-141-30 Bottle of 30 NDC 50458-141-90 Bottle of 90 NDC 50458-141-50 Bottle of 500
Description
Indications and Usage ( 1 ) 12/2024 Dosage and Administration ( 2.2 , 2.3 ) 12/2024 Dosage and Administration ( 2.5 ) 08/2024 Warnings and Precautions ( 5.1 ) 08/2024 Warnings and Precautions ( 5.2 ) 08/2024
Section 42229-5
Limitations of Use
INVOKANA is not recommended for use to improve glycemic control in patients with type 1 diabetes mellitus [see Warnings and Precautions (5.1)] .
INVOKANA is not recommended for use to improve glycemic control in patients with type 2 diabetes mellitus with an eGFR less than 30 mL/min/1.73 m 2. INVOKANA is likely to be ineffective in this setting based upon its mechanism of action.
Section 42231-1
| This Medication Guide has been approved by the U.S. Food and Drug Administration. | Revised: 12/2024 | ||||||
|
Medication Guide
|
|||||||
|
What is the most important information I should know about INVOKANA? INVOKANA can cause serious side effects, including:
|
|||||||
|
|
||||||
Call your healthcare provider right away if you have new pain or tenderness, any sores, ulcers, or infections in your leg or foot.Your healthcare provider may decide to stop your INVOKANA for a while if you have any of these signs or symptoms. Talk to your healthcare provider about proper foot care. |
|||||||
Talk to your healthcare provider about what you can do to prevent dehydration including how much fluid you should drink on a daily basis. Call your healthcare provider right away if you reduce the amount of food or liquid you drink, for example if you cannot eat or you start to lose liquids from your body, for example from vomiting, diarrhea, or being in the sun too long. |
|||||||
|
|||||||
|
|
||||||
|
Talk to your healthcare provider about what to do if you get symptoms of a yeast infection of the vagina or penis. Your healthcare provider may suggest you use an over-the-counter antifungal medicine. Talk to your healthcare provider right away if you use an over-the-counter antifungal medication and your symptoms do not go away. |
|||||||
|
What is INVOKANA?
|
|||||||
|
Do not take INVOKANA if you:
|
|||||||
|
Before taking INVOKANA, tell your healthcare provider about all of your medical conditions, including if you:
Tell your healthcare provider about all the medicines you take,including prescription and over-the-counter medicines, vitamins, and herbal supplements. INVOKANA may affect the way other medicines work, and other medicines may affect how INVOKANA works. Know the medicines you take. Keep a list of them and show it to your healthcare provider and pharmacist when you get a new medicine. |
|||||||
|
How should I take INVOKANA?
|
|||||||
|
What are the possible side effects of INVOKANA? INVOKANA may cause serious side effects including: See " What is the most important information I should know about INVOKANA? "
|
|||||||
|
|
|
|||||
|
|||||||
|
|
|
|||||
The most common side effects of INVOKANA include:
These are not all the possible side effects of INVOKANA. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. You may also report side effects to Janssen Pharmaceuticals, Inc. at 1-800-526-7736. |
|||||||
|
How should I store INVOKANA?
|
|||||||
|
General information about the safe and effective use of INVOKANA. Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use INVOKANA for a condition for which it was not prescribed. Do not give INVOKANA to other people, even if they have the same symptoms that you have. It may harm them. You can ask your pharmacist or healthcare provider for information about INVOKANA that is written for health professionals. |
|||||||
|
What are the ingredients in INVOKANA? Active ingredient: canagliflozin Inactive ingredients: croscarmellose sodium (E468), hydroxypropyl cellulose (E463), lactose anhydrous, magnesium stearate (E572), and microcrystalline cellulose (E460[i]). In addition, the tablet coating contains iron oxide yellow (E172) (100 mg tablet only), macrogol/PEG3350 (E1521), polyvinyl alcohol (E1203) (partially hydrolyzed), talc (E553b), and titanium dioxide (E171). |
|||||||
|
Active ingredient made in Belgium. Manufactured for: Janssen Pharmaceuticals, Inc., Titusville, NJ 08560, USA.
|
|||||||
Section 43683-2
Section 44425-7
Storage and Handling
Keep out of reach of children.
Store at 20 °C to 25 °C (68 °F to 77 °F); excursions permitted between 15 °C to 30 °C (59 °F to 86 °F) [see USP Controlled Room Temperature].
10 Overdosage
In the event of an overdose, contact the Poison Help line (1-800-222-1222) or a medical toxicologist for additional overdosage management recommendations. It is also reasonable to employ the usual supportive measures, e.g., remove unabsorbed material from the gastrointestinal tract, employ clinical monitoring, and institute supportive treatment as dictated by the patient's clinical status. Canagliflozin was negligibly removed during a 4-hour hemodialysis session. Canagliflozin is not expected to be dialyzable by peritoneal dialysis.
11 Description
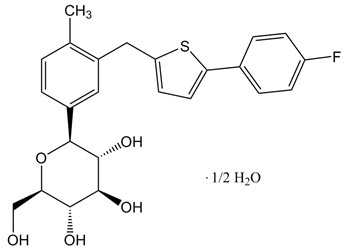
INVOKANA ®(canagliflozin) contains canagliflozin, an inhibitor of SGLT2, the transporter responsible for reabsorbing the majority of glucose filtered by the kidney. Canagliflozin, the active ingredient of INVOKANA, is chemically known as (1 S)-1,5-anhydro-1-[3-[[5-(4-fluorophenyl)-2-thienyl]methyl]-4-methylphenyl]-D-glucitol hemihydrate and its molecular formula and weight are C 24H 25FO 5S∙1/2 H 2O and 453.53, respectively. The structural formula for canagliflozin is:
Canagliflozin is practically insoluble in aqueous media from pH 1.1 to 12.9.
INVOKANA is supplied as film-coated tablets for oral administration, containing 102 and 306 mg of canagliflozin in each tablet strength, corresponding to 100 mg and 300 mg of canagliflozin (anhydrous), respectively.
Inactive ingredients of the core tablet are croscarmellose sodium (E468), hydroxypropyl cellulose (E463), lactose anhydrous, magnesium stearate (E572), and microcrystalline cellulose (E460[i]). The magnesium stearate is vegetable-sourced. The tablets are finished with a commercially available film-coating consisting of the following excipients: iron oxide yellow (E172) (100 mg tablet only), macrogol/PEG3350 (E1521), polyvinyl alcohol (E1203) (partially hydrolyzed), talc (E553b), and titanium dioxide (E171).
5.9 Bone Fracture
An increased risk of bone fracture, occurring as early as 12 weeks after treatment initiation, was observed in adult patients using INVOKANA in the CANVAS trial [see Clinical Studies (14.3)] . Consider factors that contribute to fracture risk prior to initiating INVOKANA [see Adverse Reactions (6.1)] .
8.4 Pediatric Use
The safety and effectiveness of INVOKANA as an adjunct to diet and exercise to improve glycemic control in type 2 diabetes mellitus have been established in pediatric patients aged 10 years and older.
Use of INVOKANA for this indication is supported by evidence from a 52-week double-blind, placebo-controlled trial in 171 pediatric patients aged 10 to 17 years with type 2 diabetes mellitus and in a pediatric pharmacokinetic study [see Clinical Pharmacology (12.3)and Clinical Studies (14.2)] . The safety profile of pediatric patients treated with INVOKANA was similar to that observed in adults with type 2 diabetes mellitus.
The safety and effectiveness of INVOKANA for glycemic control in patients with type 2 diabetes have not been established in pediatric patients under 10 years of age.
The safety and effectiveness of INVOKANA have not been established in pediatric patients to reduce the risk of:
- major adverse cardiovascular events (cardiovascular death, nonfatal myocardial infarction and nonfatal stroke) in patients with type 2 diabetes mellitus and established cardiovascular disease (CVD).
- end-stage kidney disease (ESKD), doubling of serum creatinine, cardiovascular (CV) death, and hospitalization for heart failure in patients with type 2 diabetes mellitus and diabetic nephropathy with albuminuria greater than 300 mg/day.
8.5 Geriatric Use
In 13 clinical trials of INVOKANA, 2,294 patients 65 years and older, and 351 patients 75 years and older were exposed to INVOKANA [see Clinical Studies (14.1)] .
Patients 65 years and older had a higher incidence of adverse reactions related to reduced intravascular volume with INVOKANA (such as hypotension, postural dizziness, orthostatic hypotension, syncope, and dehydration), particularly with the 300 mg daily dose, compared to younger patients; a more prominent increase in the incidence was seen in patients who were 75 years and older [see Dosage and Administration (2.1)and Adverse Reactions (6.1)]. Smaller reductions in HbA 1Cwith INVOKANA relative to placebo were seen in older (65 years and older; -0.61% with INVOKANA 100 mg and -0.74% with INVOKANA 300 mg relative to placebo) compared to younger patients (-0.72% with INVOKANA 100 mg and -0.87% with INVOKANA 300 mg relative to placebo).
4 Contraindications
INVOKANA is contraindicated in patients with a serious hypersensitivity reaction to INVOKANA, such as anaphylaxis or angioedema [see Warnings and Precautions (5.8)and Adverse Reactions (6.1, 6.2)] .
6 Adverse Reactions
The following important adverse reactions are described below and elsewhere in the labeling:
- Diabetic Ketoacidosis in Patients with Type 1 Diabetes and Other Ketoacidosis [see Warnings and Precautions (5.1)]
- Lower Limb Amputation [see Warnings and Precautions (5.2)]
- Volume Depletion [see Warnings and Precautions (5.3)]
- Urosepsis and Pyelonephritis [see Warnings and Precautions (5.4)]
- Hypoglycemia with Concomitant Use with Insulin or Insulin Secretagogues [see Warnings and Precautions (5.5)]
- Necrotizing Fasciitis of the Perineum (Fournier's gangrene) [see Warnings and Precautions (5.6)]
- Genital Mycotic Infections [see Warnings and Precautions (5.7)]
- Hypersensitivity Reactions [see Warnings and Precautions (5.8)]
- Bone Fracture [see Warnings and Precautions (5.9)]
7 Drug Interactions
| UGT Enzyme Inducers | |
| Clinical Impact: | UGT enzyme inducers decrease canagliflozin exposure which may reduce the effectiveness of INVOKANA. |
| Intervention: | For patients with eGFR 60 mL/min/1.73 m
2or greater, if an inducer of UGTs is administered with INVOKANA, increase the dosage to 200 mg daily in patients currently tolerating INVOKANA 100 mg daily. The total daily dosage may be increased to 300 mg daily in patients currently tolerating INVOKANA 200 mg daily who require additional glycemic control.
For patients with eGFR less than 60 mL/min/1.73 m 2, if an inducer of UGTs is administered with INVOKANA, increase the dosage to 200 mg daily in patients currently tolerating INVOKANA 100 mg daily. Consider adding another antihyperglycemic agent in patients who require additional glycemic control [see Dosage and Administration (2.3)and Clinical Pharmacology (12.3)] . |
| Examples: | Rifampin, phenytoin, phenobarbital, ritonavir |
| Insulin or Insulin Secretagogues | |
| Clinical Impact: | The risk of hypoglycemia is increased when INVOKANA is used concomitantly with insulin secretagogues (e.g., sulfonylurea) or insulin. |
| Intervention: | Concomitant use may require a lower dosage of the insulin secretagogue or insulin to reduce the risk of hypoglycemia. |
| Digoxin | |
| Clinical Impact: | Canagliflozin increases digoxin exposure [see Clinical Pharmacology (12.3)] . |
| Intervention: | Monitor patients taking INVOKANA with concomitant digoxin for a need to adjust the dosage of digoxin. |
| Lithium | |
| Clinical Impact: | Concomitant use of an SGLT2 inhibitor with lithium may decrease serum lithium concentrations. |
| Intervention: | Monitor serum lithium concentration more frequently during INVOKANA initiation and dosage changes. |
| Drug/Laboratory Test Interference | |
| Positive Urine Glucose Test | |
| Clinical Impact: | SGLT2 inhibitors increase urinary glucose excretion which will lead to positive urine glucose tests. |
| Intervention: | Monitoring glycemic control with urine glucose tests is not recommended in patients taking SGLT2 inhibitors. Use alternative methods to monitor glycemic control. |
| Interference with 1,5-anhydroglucitol (1,5-AG) Assay | |
| Clinical Impact: | Measurements of 1,5-AG are unreliable in assessing glycemic control in patients taking SGLT2 inhibitors. |
| Intervention: | Monitoring glycemic control with 1,5-AG assay is not recommended in patients taking SGLT2 inhibitors. Use alternative methods to monitor glycemic control. |
5.3 Volume Depletion
INVOKANA can cause intravascular volume contraction which may sometimes manifest as symptomatic hypotension or acute transient changes in creatinine [see Adverse Reactions (6.1)] . There have been post-marketing reports of acute kidney injury which are likely related to volume depletion, some requiring hospitalizations and dialysis, in patients with type 2 diabetes mellitus receiving SGLT2 inhibitors, including INVOKANA. Patients with impaired renal function (eGFR less than 60 mL/min/1.73 m 2), elderly patients, or patients on loop diuretics may be at increased risk for volume depletion or hypotension. Before initiating INVOKANA in patients with one or more of these characteristics, assess and correct volume status. Monitor for signs and symptoms of volume depletion after initiating therapy.
8.6 Renal Impairment
The efficacy and safety of INVOKANA for glycemic control were evaluated in a trial that included adult patients with moderate renal impairment (eGFR 30 to less than 50 mL/min/1.73 m 2) [see Clinical Studies (14.1)] . These patients had less overall glycemic efficacy, and patients treated with 300 mg per day had increases in serum potassium, which were transient and similar by the end of the trial. Patients with renal impairment using INVOKANA for glycemic control may also be more likely to experience hypotension and may be at higher risk for acute kidney injury [see Warnings and Precautions (5.3)] .
Efficacy and safety trials with INVOKANA did not enroll adult patients with ESKD on dialysis or patients with an eGFR less than 30 mL/min/1.73 m 2 [see Clinical Pharmacology (12.3)] .
12.2 Pharmacodynamics
Following single and multiple oral doses of canagliflozin in adult patients with type 2 diabetes, dose-dependent decreases in RT Gand increases in urinary glucose excretion were observed. From a starting RT Gvalue of approximately 240 mg/dL, canagliflozin at 100 mg and 300 mg once daily suppressed RT Gthroughout the 24-hour period. Data from single oral doses of canagliflozin in healthy volunteers indicate that, on average, the elevation in urinary glucose excretion approaches baseline by about 3 days for doses up to 300 mg once daily. Maximal suppression of mean RT Gover the 24-hour period was seen with the 300 mg daily dose to approximately 70 to 90 mg/dL in patients with type 2 diabetes in Phase 1 trials. The reductions in RT Gled to increases in mean UGE of approximately 100 g/day in patients with type 2 diabetes treated with either 100 mg or 300 mg of canagliflozin. In patients with type 2 diabetes given 100 to 300 mg once daily over a 16-day dosing period, reductions in RT Gand increases in urinary glucose excretion were observed over the dosing period. In this trial, plasma glucose declined in a dose-dependent fashion within the first day of dosing. In single-dose trials in healthy and type 2 diabetic patients, treatment with canagliflozin 300 mg before a mixed-meal delayed intestinal glucose absorption and reduced postprandial glucose.
12.3 Pharmacokinetics
The pharmacokinetics of canagliflozin is similar in healthy subjects and patients with type 2 diabetes. Following single-dose oral administration of 100 mg and 300 mg of INVOKANA, peak plasma concentrations (median T max) of canagliflozin occurs within 1 to 2 hours post-dose. Plasma C maxand AUC of canagliflozin increased in a dose-proportional manner from 50 mg to 300 mg. The apparent terminal half-life (t 1/2) was 10.6 hours and 13.1 hours for the 100 mg and 300 mg doses, respectively. Steady-state was reached after 4 to 5 days of once-daily dosing with canagliflozin 100 mg to 300 mg. Canagliflozin does not exhibit time-dependent pharmacokinetics and accumulated in plasma up to 36% following multiple doses of 100 mg and 300 mg.
8.7 Hepatic Impairment
No dosage adjustment is necessary in patients with mild or moderate hepatic impairment. The use of INVOKANA has not been studied in patients with severe hepatic impairment and is therefore not recommended [see Clinical Pharmacology (12.3)] .
1 Indications and Usage
INVOKANA (canagliflozin) is indicated:
- as an adjunct to diet and exercise to improve glycemic control in adults and pediatric patients aged 10 years and older with type 2 diabetes mellitus.
- to reduce the risk of major adverse cardiovascular events (cardiovascular death, nonfatal myocardial infarction and nonfatal stroke) in adults with type 2 diabetes mellitus and established cardiovascular disease (CVD).
- to reduce the risk of end-stage kidney disease (ESKD), doubling of serum creatinine, cardiovascular (CV) death, and hospitalization for heart failure in adults with type 2 diabetes mellitus and diabetic nephropathy with albuminuria greater than 300 mg/day.
12.1 Mechanism of Action
SGLT2, expressed in the proximal renal tubules, is responsible for the majority of the reabsorption of filtered glucose from the tubular lumen. Canagliflozin is an inhibitor of SGLT2. By inhibiting SGLT2, canagliflozin reduces reabsorption of filtered glucose and lowers the renal threshold for glucose (RT G), and thereby increases urinary glucose excretion (UGE).
Canagliflozin increases the delivery of sodium to the distal tubule by blocking SGLT2-dependent glucose and sodium reabsorption. This is believed to increase tubuloglomerular feedback and reduce intraglomerular pressure.
5.2 Lower Limb Amputation
An increased risk of lower limb amputations associated with INVOKANA use versus placebo was observed in CANVAS (5.9 vs 2.8 events per 1,000 patient-years) and CANVAS-R (7.5 vs 4.2 events per 1,000 patient-years), two randomized, placebo-controlled trials evaluating adult patients with type 2 diabetes mellitus who had either established cardiovascular disease or were at risk for cardiovascular disease. The risk of lower limb amputations was observed at both the 100 mg and 300 mg once daily dosage regimens. The amputation data for CANVAS and CANVAS-R are shown in Tables 3 and 4, respectively [see Adverse Reactions (6.1)].
Amputations of the toe and midfoot (99 out of 140 patients with amputations receiving INVOKANA in the two trials) were the most frequent; however, amputations involving the leg, below and above the knee, were also observed (41 out of 140 patients with amputations receiving INVOKANA in the two trials). Some patients had multiple amputations, some involving both lower limbs.
Lower limb infections, gangrene, and diabetic foot ulcers were the most common precipitating medical events leading to the need for an amputation. The risk of amputation was highest in patients with a baseline history of prior amputation, peripheral vascular disease, and neuropathy.
Counsel patients about the importance of routine preventative foot care. Monitor patients receiving INVOKANA for signs and symptoms of infection (including osteomyelitis), new pain or tenderness, sores or ulcers involving the lower limbs, and discontinue INVOKANA if these complications occur.
5 Warnings and Precautions
- Diabetic Ketoacidosis in Patients with Type 1 Diabetes Mellitus and Other Ketoacidosis: Consider ketone monitoring in patients at risk for ketoacidosis, as indicated. Assess for ketoacidosis regardless of presenting blood glucose levels and discontinue INVOKANA if ketoacidosis is suspected. Monitor patients for resolution of ketoacidosis before restarting ( 5.1).
- Lower Limb Amputation: Monitor patients for infection or ulcers of lower limb and discontinue if these occur ( 5.2).
- Volume Depletion: May result in acute kidney injury. Before initiating INVOKANA, assess and correct volume status in patients with renal impairment, elderly patients, or patients on loop diuretics. Monitor for signs and symptoms during therapy ( 5.3).
- Urosepsis and Pyelonephritis: Evaluate patients for signs and symptoms of urinary tract infections and treat promptly, if indicated ( 5.4).
- Hypoglycemia with Concomitant Use with Insulin or Insulin Secretagogues: Consider a lower dose of insulin or the insulin secretagogue to reduce the risk of hypoglycemia when used in combination with INVOKANA ( 5.5).
- Necrotizing Fasciitis of the Perineum (Fournier's Gangrene): Serious, life-threatening cases have occurred in both females and males. Assess patients presenting with pain or tenderness, erythema, or swelling in the genital or perineal area, along with fever or malaise. If suspected, institute prompt treatment ( 5.6).
- Genital Mycotic Infections: Monitor and treat if indicated ( 5.7).
- Hypersensitivity Reactions: Discontinue INVOKANA and monitor until signs and symptoms resolve ( 5.8).
- Bone Fracture: Consider factors that contribute to fracture risk before initiating INVOKANA ( 5.9).
2 Dosage and Administration
- Assess renal function before initiating and as clinically indicated. Assess volume status and correct volume depletion before initiating ( 2.1).
- The recommended starting dosage in adults and pediatric patients aged 10 years and older with type 2 diabetes mellitus is 100 mg orally once daily, taken before the first meal of the day to improve glycemic control. The dosage can be increased to 300 mg once daily in patients tolerating 100 mg once daily who have an eGFR of 60 mL/min/1.73 m 2or greater and require additional glycemic control ( 2.2).
- For all other indications in adults, the recommended dosage of INVOKANA is 100 mg orally once daily ( 2.2).
- Dosage adjustments for patients with renal impairment may be required ( 2.3).
- See full prescribing information for INVOKANA dosage modifications due to drug interactions ( 2.4).
- Withhold INVOKANA at least 3 days, if possible, prior to surgery or procedures associated with prolonged fasting ( 2.5).
3 Dosage Forms and Strengths
- INVOKANA 100 mg tablets are yellow, capsule-shaped, tablets with "CFZ" on one side and "100" on the other side.
- INVOKANA 300 mg tablets are white, capsule-shaped, tablets with "CFZ" on one side and "300" on the other side.
6.2 Postmarketing Experience
Additional adverse reactions have been identified during post-approval use of INVOKANA. Because these reactions are reported voluntarily from a population of uncertain size, it is generally not possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Metabolism and Nutrition
Ketoacidosis
Renal and Urinary
Acute Kidney Injury
Immune System
Anaphylaxis
Skin and Subcutaneous Tissue
Angioedema
Infections
Urosepsis and Pyelonephritis, Necrotizing Fasciitis of the Perineum (Fournier's gangrene)
8 Use in Specific Populations
- Pregnancy: Advise females of the potential risk to a fetus especially during the second and third trimesters ( 8.1).
- Lactation: Not recommended when breastfeeding ( 8.2).
- Geriatrics: Higher incidence of adverse reactions related to reduced intravascular volume ( 8.5).
- Renal Impairment: Higher incidence of adverse reactions related to hypotension and renal function ( 8.6).
- Hepatic Impairment: Not recommended in patients with severe hepatic impairment ( 8.7).
5.7 Genital Mycotic Infections
INVOKANA increases the risk of genital mycotic infections. Patients with a history of genital mycotic infections and uncircumcised males were more likely to develop genital mycotic infections [see Adverse Reactions (6.1)] . Monitor and treat appropriately.
5.8 Hypersensitivity Reactions
Hypersensitivity reactions, including angioedema and anaphylaxis, have been reported with INVOKANA. These reactions generally occurred within hours to days after initiating INVOKANA. If hypersensitivity reactions occur, discontinue use of INVOKANA; treat and monitor until signs and symptoms resolve [see Contraindications (4)and Adverse Reactions (6.1, 6.2)] .
6.1 Clinical Studies Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to the rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
INVOKANA has been evaluated in clinical trials in adults and pediatric patients aged 10 years and older with type 2 diabetes mellitus. Additionally, INVOKANA has been studied in clinical trials in adult patients with type 2 diabetes mellitus who also have heart failure or chronic kidney disease. The overall safety profile of INVOKANA was consistent across the studied indications.
5.4 Urosepsis and Pyelonephritis
There have been postmarketing reports of serious urinary tract infections including urosepsis and pyelonephritis requiring hospitalization in patients receiving INVOKANA. Treatment with INVOKANA increases the risk for urinary tract infections. Evaluate patients for signs and symptoms of urinary tract infections and treat promptly, if indicated [see Adverse Reactions (6)] .
17 Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Medication Guide).
2.1 Prior to Initiation of Invokana
Assess renal function before initiating INVOKANA and as clinically indicated [see Dosage and Administration (2.3)and Warnings and Precautions (5.3)] .
In patients with volume depletion, correct this condition before initiating INVOKANA [see Warnings and Precautions (5.3)and Use in Specific Populations (8.5, 8.6)] .
16 How Supplied/storage and Handling
INVOKANA ®(canagliflozin) tablets are available in the strengths and packages listed below:
100 mg tablets are yellow, capsule-shaped, film-coated tablets with "CFZ" on one side and "100" on the other side.
| NDC 50458-140-30 | Bottle of 30 |
| NDC 50458-140-90 | Bottle of 90 |
| NDC 50458-140-50 | Bottle of 500 |
300 mg tablets are white, capsule-shaped, film-coated tablets with "CFZ" on one side and "300" on the other side.
| NDC 50458-141-30 | Bottle of 30 |
| NDC 50458-141-90 | Bottle of 90 |
| NDC 50458-141-50 | Bottle of 500 |
2.5 Temporary Interruption for Surgery
Withhold INVOKANA at least 3 days, if possible, prior to surgery or procedures associated with prolonged fasting. Resume INVOKANA when the patient is clinically stable and has resumed oral intake [see Warnings and Precautions (5.1)and Clinical Pharmacology (12.2)].
Principal Display Panel 100 Mg Tablet Bottle Label
NDC 50458-140-90
Invokana
®
(canagliflozin) tablets
100 mg
Attention: Dispense the enclosed
Medication Guide to each patient.
Rx only
90 tablets
Principal Display Panel 300 Mg Tablet Bottle Label
NDC 50458-141-90
Invokana
®
(canagliflozin) tablets
300 mg
Attention: Dispense the enclosed
Medication Guide to each patient.
Rx only
90 tablets
5.6 Necrotizing Fasciitis of the Perineum (fournier's Gangrene)
Reports of necrotizing fasciitis of the perineum (Fournier's gangrene), a rare but serious and life-threatening necrotizing infection requiring urgent surgical intervention, have been identified in postmarketing surveillance in patients with diabetes mellitus receiving SGLT2 inhibitors, including INVOKANA. Cases have been reported in both females and males. Serious outcomes have included hospitalization, multiple surgeries, and death.
Patients treated with INVOKANA presenting with pain or tenderness, erythema, or swelling in the genital or perineal area, along with fever or malaise, should be assessed for necrotizing fasciitis. If suspected, start treatment immediately with broad-spectrum antibiotics and, if necessary, surgical debridement. Discontinue INVOKANA, closely monitor blood glucose levels, and provide appropriate alternative therapy for glycemic control.
14.1 Glycemic Control Trials in Adults With Type 2 Diabetes Mellitus
INVOKANA (canagliflozin) has been studied as monotherapy, in combination with metformin HCl, sulfonylurea, metformin HCl and sulfonylurea, metformin HCl and sitagliptin, metformin HCl and a thiazolidinedione (i.e., pioglitazone), and in combination with insulin (with or without other anti-hyperglycemic agents). The efficacy of INVOKANA was compared to a dipeptidyl peptidase-4 (DPP-4) inhibitor (sitagliptin), both as add-on combination therapy with metformin HCl and sulfonylurea, and a sulfonylurea (glimepiride), both as add-on combination therapy with metformin HCl. INVOKANA was also evaluated in adults 55 to 80 years of age and patients with moderate renal impairment.
2.4 Concomitant Use With Udp Glucuronosyl Transferase (ugt) Enzyme Inducers
When co-administering INVOKANA with an inducer of UGT (e.g., rifampin, phenytoin, phenobarbital, ritonavir), increase the dosage of INVOKANA based on renal function [see Drug Interactions (7)]:
- In patients with eGFR 60 mL/min/1.73 m 2or greater, increase the dosage to 200 mg orally once daily in patients currently tolerating INVOKANA 100 mg once daily. The maximum recommended dosage of INVOKANA is 300 mg once daily.
- In patients with eGFR less than 60 mL/min/1.73 m 2, increase to a maximum recommended dosage of 200 mg orally once daily in patients currently tolerating INVOKANA 100 mg once daily.
5.5 Hypoglycemia With Concomitant Use With Insulin Or Insulin Secretagogues
Insulin and insulin secretagogues are known to cause hypoglycemia. INVOKANA may increase the risk of hypoglycemia when combined with insulin or an insulin secretagogue [see Adverse Reactions (6.1)] . The risk of hypoglycemia may be lowered by a reduction in the dose of sulfonylurea (or other concomitantly administered insulin secretagogues) or insulin. Inform patients using these concomitant medications of the risk of hypoglycemia and educate them on the signs and symptoms of hypoglycemia.
14.4 Renal and Cardiovascular Outcomes in Adults With Diabetic Nephropathy and Albuminuria
The Canagliflozin and Renal Events in Diabetes with Established Nephropathy Clinical Evaluation Trial (CREDENCE) was a multinational, randomized, double-blind, placebo-controlled trial comparing canagliflozin with placebo in adult patients with type 2 diabetes mellitus, an eGFR ≥ 30 to < 90 mL/min/1.73 m 2and albuminuria (urine albumin/creatinine > 300 to ≤ 5,000 mg/g) who were receiving standard of care including a maximum-tolerated, labeled daily dose of an angiotensin-converting enzyme inhibitor (ACEi) or angiotensin receptor blocker (ARB).
The primary objective of CREDENCE was to assess the efficacy of canagliflozin relative to placebo in reducing the composite endpoint of end stage kidney disease (ESKD), doubling of serum creatinine, and renal or CV death.
Patients were randomized to receive canagliflozin 100 mg (N=2,202) or placebo (N=2,199) and treatment was continued until the initiation of dialysis or renal transplantation.
The median follow-up duration for the 4,401 randomized subjects was 137 weeks. Vital status was obtained for 99.9% of subjects.
The population was 67% White, 20% Asian, and 5% Black or African American; 32% were of Hispanic or Latino ethnicity. The mean age was 63 years and 66% were male.
At randomization, the mean HbA 1Cwas 8.3%, the median urine albumin/creatinine was 927 mg/g, the mean eGFR was 56.2 mL/min/1.73 m 2, 50% had prior CV disease, and 15% reported a history of heart failure. The most frequent antihyperglycemic agents (AHA) medications used at baseline were insulin (66%), biguanides (58%), and sulfonylureas (29%). Nearly all subjects (99.9%) were on ACEi or ARB at randomization, approximately 60% were taking an anti-thrombotic agent (including aspirin), and 69% were on a statin.
The primary composite endpoint in the CREDENCE trial was the time to first occurrence of ESKD (defined as an eGFR < 15 mL/min/1.73 m 2, initiation of chronic dialysis or renal transplant), doubling of serum creatinine, and renal or CV death. Canagliflozin 100 mg significantly reduced the risk of the primary composite endpoint based on a time-to-event analysis [HR: 0.70; 95% CI: 0.59, 0.82; p<0.0001] (see Figure 4). The treatment effect reflected a reduction in progression to ESKD, doubling of serum creatinine and cardiovascular death as shown in Table 22 and Figure 4. There were few renal deaths during the trial. Canagliflozin 100 mg also significantly reduced the risk of hospitalization for heart failure [HR: 0.61; 95% CI: 0.47 to 0.80; p<0.001].
| Placebo | canagliflozin | ||||
|---|---|---|---|---|---|
| Endpoint | N=2,199 (%) | Event Rate
Event rate per 100 patient-years.
|
N=2,202 (%) | Event Rate | HR
Hazard ratio (canagliflozin compared to placebo), 95% CI and p-value are estimated using a stratified Cox proportional hazards model including treatment as the explanatory variable and stratified by screening eGFR (≥ 30 to < 45, ≥ 45 to < 60, ≥ 60 to < 90 mL/min/1.73 m
2). HR is not presented for renal death due to the small number of events in each group.
(95% CI) |
| Intent-To-Treat Analysis Set (time to first occurrence) | |||||
| The individual components do not represent a breakdown of the composite outcomes, but rather the total number of subjects experiencing an event during the course of the trial. | |||||
| Primary Composite Endpoint (ESKD, doubling of serum creatinine, renal death, or CV death) | 340 (15.5) | 6.1 | 245 (11.1) | 4.3 | 0.70
(0.59, 0.82) P-value <0.0001
|
| ESKD | 165 (7.5) | 2.9 | 116 (5.3) | 2.0 | 0.68
(0.54, 0.86) |
| Doubling of serum creatinine | 188 (8.5) | 3.4 | 118 (5.4) | 2.1 | 0.60
(0.48, 0.76) |
| Renal death | 5 (0.2) | 0.1 | 2 (0.1) | 0.0 | |
| CV death | 140 (6.4) | 2.4 | 110 (5.0) | 1.9 | 0.78
(0.61, 1.00) |
| CV death or hospitalization for heart failure | 253 (11.5) | 4.5 | 179 (8.1) | 3.1 | 0.69
(0.57, 0.83) P-value <0.001
|
| CV death, non-fatal myocardial infarction or non-fatal stroke | 269 (12.2) | 4.9 | 217 (9.9) | 3.9 | 0.80
(0.67, 0.95) P-value <0.02
|
| Non-fatal myocardial infarction | 87 (4.0) | 1.6 | 71 (3.2) | 1.3 | 0.81
(0.59, 1.10) |
| Non-fatal stroke | 66 (3.0) | 1.2 | 53 (2.4) | 0.9 | 0.80
(0.56, 1.15) |
| Hospitalization for heart failure | 141 (6.4) | 2.5 | 89 (4.0) | 1.6 | 0.61
(0.47, 0.80) |
| ESKD, doubling of serum creatinine or renal death | 224 (10.2) | 4.0 | 153 (6.9) | 2.7 | 0.66
(0.53, 0.81) |
The Kaplan-Meier curve (Figure 4) shows time to first occurrence of the primary composite endpoint of ESKD, doubling of serum creatinine, renal death, or CV death. The curves begin to separate by Week 52 and continue to diverge thereafter.
Figure 4: CREDENCE: Time to First Occurrence of the Primary Composite Endpoint
5.1 Diabetic Ketoacidosis in Patients With Type 1 Diabetes Mellitus and Other Ketoacidosis
In patients with type 1 diabetes mellitus, INVOKANA significantly increases the risk of diabetic ketoacidosis, a life-threatening event, beyond the background rate. In placebo-controlled trials of patients with type 1 diabetes mellitus, the risk of ketoacidosis was markedly increased in patients who received sodium glucose transporter 2 (SGLT2) inhibitors compared to patients who received placebo; this risk may be greater with higher doses of INVOKANA. INVOKANA is not indicated for glycemic control in patients with type 1 diabetes mellitus.
Type 2 diabetes mellitus and pancreatic disorders (e.g., history of pancreatitis or pancreatic surgery) are also risk factors for ketoacidosis. There have been postmarketing reports of fatal events of ketoacidosis in patients with type 2 diabetes mellitus using SGLT2 inhibitors, including INVOKANA.
Precipitating conditions for diabetic ketoacidosis or other ketoacidosis include under-insulinization due to insulin dose reduction or missed insulin doses, acute febrile illness, reduced caloric intake, ketogenic diet, surgery, volume depletion, and alcohol abuse.
Signs and symptoms are consistent with dehydration and severe metabolic acidosis and include nausea, vomiting, abdominal pain, generalized malaise, and shortness of breath. Blood glucose levels at presentation may be below those typically expected for diabetic ketoacidosis (e.g., less than 250 mg/dL). Ketoacidosis and glucosuria may persist longer than typically expected.
Urinary glucose excretion persists for 3 days after discontinuing INVOKANA [see Clinical Pharmacology (12.2)]; however, there have been postmarketing reports of ketoacidosis and/or glucosuria lasting greater than 6 days and some up to 2 weeks after discontinuation of SGLT2 inhibitors.
Consider ketone monitoring in patients at risk for ketoacidosis if indicated by the clinical situation. Assess for ketoacidosis regardless of presenting blood glucose levels in patients who present with signs and symptoms consistent with severe metabolic acidosis. If ketoacidosis is suspected, discontinue INVOKANA, promptly evaluate, and treat ketoacidosis, if confirmed. Monitor patients for resolution of ketoacidosis before restarting INVOKANA.
Withhold INVOKANA, if possible, in temporary clinical situations that could predispose patients to ketoacidosis. Resume INVOKANA when the patient is clinically stable and has resumed oral intake [see Dosage and Administration (2.5)] .
Educate all patients on the signs and symptoms of ketoacidosis and instruct patients to discontinue INVOKANA and seek medical attention immediately if signs and symptoms occur.
2.3 Recommended Dosage in Adults and Pediatric Patients Aged 10 Years and Older With Renal Impairment
Table 1 provides dosage recommendations for adults and pediatric patients aged 10 years and older with renal impairment, based on estimated glomerular filtration rate (eGFR).
| Estimated Glomerular Filtration Rate
[eGFR (mL/min/1.73 m 2)] |
Recommended Dosage |
|---|---|
| eGFR 30 to less than 60 | The maximum recommended dosage is 100 mg orally once daily. |
| eGFR less than 30 |
|
14.2 Glycemic Control Trial in Pediatric Patients Aged 10 Years and Older With Type 2 Diabetes Mellitus
In a double-blind, placebo-controlled, parallel-group pediatric trial (NCT03170518), 171 pediatric patients aged 10 to 17 years with inadequately controlled type 2 diabetes mellitus (HbA 1C≥6.5% and ≤11.0%) were randomized to INVOKANA (84 patients) or placebo (87 patients) as add-on to diet and exercise, metformin HCl (≥1,000 mg per day or maximally tolerated dosage), insulin, or a combination of metformin HCl and insulin, for a total of 52-weeks. At Week 13, patients in the INVOKANA arm whose HbA 1Cwas ≥7.0% and eGFR ≥60 mL/min/1.73 m 2were re-randomized to either continue on INVOKANA 100 mg orally once daily (n=16) or to up-titrate to INVOKANA 300 mg orally once daily (n=17).
At baseline, background therapies included diet and exercise only (14%), insulin monotherapy (11%), metformin HCl and insulin (29%), and metformin HCl monotherapy (46%). The mean HbA 1Cat baseline was 8.0% and the mean duration of type 2 diabetes mellitus was 2 years. The mean eGFR at baseline was 157.3 mL/min/1.73 m 2, and approximately 16% (24/151) of the trial population with measurements had microalbuminuria or macroalbuminuria. Patients with an eGFR less than 60 mL/min/1.73 m 2were not enrolled in the trial; no patients in the trial reached an eGFR < 60 mL/min/1.73 m 2. The mean age was 14.3 years, 47% were under 15 years of age, and 68% were female. Approximately 42% were Asian, 42% were White, 11% were Black or African American, 5% were American Indian/Alaska Native, and 36% were of Hispanic or Latino ethnicity. The mean BMI was 30.8 kg/m 2(range 18–57 kg/m 2) and the mean BMI Z-score was 1.84.
At Week 26, treatment with canagliflozin provided statistically significant improvement in HbA 1Cfrom baseline, compared with placebo (see Table 20).
| Efficacy Parameter | Placebo
(N=87) |
INVOKANA
(N=84) |
|---|---|---|
| HbA 1C(%) | ||
| Baseline (mean) | 8.3 | 7.8 |
| Change from baseline
Multiple imputation using retrieved dropout approach with 1000 iterations for missing data (canagliflozin N=7 (8.3%), placebo N=7 (8.1%) for HbA
1C and canagliflozin N=9 (10.7%), placebo N=7 (8.1%) for FPG).
Least-Square Mean from Analysis of Covariance (ANCOVA) adjusted for baseline value, baseline age stratum (< 15 years vs 15 to < 18 years) and baseline antihyperglycemic agent (AHA) background (i.e, diet and exercise only, metformin monotherapy, insulin monotherapy, or combination of insulin and metformin).
|
0.34 | -0.38 |
| Difference from placebo 95% CI | -0.73 (-1.26, -0.19)
P-value=0.008 (two-sided)
|
|
| FPG (mg/dL) | ||
| Baseline (mean) | 156.5 | 154.8 |
| Change from baseline | 17.29 | -8.22 |
| Difference from placebo 95% CI | -25.51 (-49.55, -1.47)
Not evaluated for statistical significance, not part of sequential testing procedure.
|
14.3 Cardiovascular Outcomes in Adults With Type 2 Diabetes Mellitus and Atherosclerotic Cardiovascular Disease
The CANVAS and CANVAS-R trials were multicenter, multi-national, randomized, double-blind parallel group, with similar inclusion and exclusion criteria. Patients eligible for enrollment in both CANVAS and CANVAS-R trials were: 30 years of age or older and had established, stable, cardiovascular, cerebrovascular, peripheral artery disease (66% of the enrolled population) or were 50 years of age or older and had two or more other specified risk factors for cardiovascular disease (34% of the enrolled population).
The integrated analysis of the CANVAS and CANVAS-R trials compared the risk of Major Adverse Cardiovascular Event (MACE) between canagliflozin and placebo when these were added to and used concomitantly with standard of care treatments for diabetes and atherosclerotic cardiovascular disease. The primary endpoint, MACE, was the time to first occurrence of a three-part composite outcome which included cardiovascular death, non-fatal myocardial infarction and non-fatal stroke.
In CANVAS, patients were randomly assigned 1:1:1 to canagliflozin 100 mg, canagliflozin 300 mg, or matching placebo. In CANVAS-R, patients were randomly assigned 1:1 to canagliflozin 100 mg or matching placebo, and titration to 300 mg was permitted at the investigator's discretion (based on tolerability and glycemic needs) after Week 13. Concomitant antidiabetic and atherosclerotic therapies could be adjusted, at the discretion of investigators, to ensure participants were treated according to the standard care for these diseases.
A total of 10,134 adult patients were treated (4,327 in CANVAS and 5,807 in CANVAS-R; total of 4,344 randomly assigned to placebo and 5,790 to canagliflozin) for a mean exposure duration of 149 weeks (223 weeks [4.3 years] in CANVAS and 94 weeks [1.8 years] in CANVAS-R) .Approximately 78% of the trial population was White, 13% was Asian, and 3% was Black or African American. The mean age was 63 years and approximately 64% were male.
The mean HbA 1Cat baseline was 8.2% and mean duration of diabetes was 13.5 years with 70% of patients having had diabetes for 10 years or more. Approximately 31%, 21% and 17% reported a past history of neuropathy, retinopathy and nephropathy, respectively, and the mean eGFR 76 mL/min/1.73 m 2. At baseline, patients were treated with one (19%) or more (80%) antidiabetic medications including metformin HCl (77%), insulin (50%), and sulfonylurea (43%).
At baseline, the mean systolic blood pressure was 137 mmHg, the mean diastolic blood pressure was 78 mmHg, the mean LDL was 89 mg/dL, the mean HDL was 46 mg/dL, and the mean urinary albumin to creatinine ratio (UACR) was 115 mg/g. At baseline, approximately 80% of patients were treated with renin angiotensin system inhibitors, 53% with beta-blockers, 13% with loop diuretics, 36% with non-loop diuretics, 75% with statins, and 74% with antiplatelet agents (mostly aspirin). During the trial, investigators could modify anti-diabetic and cardiovascular therapies to achieve local standard of care treatment targets with respect to blood glucose, lipid, and blood pressure. More patients receiving canagliflozin compared to placebo initiated anti-thrombotics (5.2% vs 4.2%) and statins (5.8% vs 4.8%) during the trial.
For the primary analysis, a stratified Cox proportional hazards model was used to test for non-inferiority against a pre-specified risk margin of 1.3 for the hazard ratio of MACE.
In the integrated analysis of CANVAS and CANVAS-R trials, canagliflozin reduced the risk of first occurrence of MACE. The estimated hazard ratio (95% CI) for time to first MACE was 0.86 (0.75, 0.97). Refer to Table 21. Vital status was obtained for 99.6% of patients across the trials. The Kaplan-Meier curve depicting time to first occurrence of MACE is shown in Figure 3.
| Placebo
N=4,347(%) |
Canagliflozin
N=5,795 (%) |
Hazard ratio
(95% C.I.) Stratified Cox-proportional hazards model with treatment as a factor and stratified by the trial and by prior CV disease
|
|
|---|---|---|---|
| Composite of cardiovascular death, non-fatal myocardial infarction, non-fatal stroke
(time to first occurrence) P-value for superiority (2-sided) = 0.0158
,
Number and percentage of first events
,
Due to pooling of unequal randomization ratios, Cochran-Mantel-Haenszel weights were applied to calculate percentages
|
426 (10.4) | 585 (9.2) | 0.86 (0.75, 0.97) |
| Non-fatal myocardial infarction , | 159 (3.9) | 215 (3.4) | 0.85 (0.69, 1.05) |
| Non-fatal Stroke , | 116 (2.8) | 158 (2.5) | 0.90 (0.71, 1.15) |
| Cardiovascular Death , | 185 (4.6) | 268 (4.1) | 0.87 (0.72, 1.06) |
Figure 3: Time to First Occurrence of MACE in Adults with Type 2 Diabetes Mellitus
Structured Label Content
Section 42229-5 (42229-5)
Limitations of Use
INVOKANA is not recommended for use to improve glycemic control in patients with type 1 diabetes mellitus [see Warnings and Precautions (5.1)] .
INVOKANA is not recommended for use to improve glycemic control in patients with type 2 diabetes mellitus with an eGFR less than 30 mL/min/1.73 m 2. INVOKANA is likely to be ineffective in this setting based upon its mechanism of action.
Section 42231-1 (42231-1)
| This Medication Guide has been approved by the U.S. Food and Drug Administration. | Revised: 12/2024 | ||||||
|
Medication Guide
|
|||||||
|
What is the most important information I should know about INVOKANA? INVOKANA can cause serious side effects, including:
|
|||||||
|
|
||||||
Call your healthcare provider right away if you have new pain or tenderness, any sores, ulcers, or infections in your leg or foot.Your healthcare provider may decide to stop your INVOKANA for a while if you have any of these signs or symptoms. Talk to your healthcare provider about proper foot care. |
|||||||
Talk to your healthcare provider about what you can do to prevent dehydration including how much fluid you should drink on a daily basis. Call your healthcare provider right away if you reduce the amount of food or liquid you drink, for example if you cannot eat or you start to lose liquids from your body, for example from vomiting, diarrhea, or being in the sun too long. |
|||||||
|
|||||||
|
|
||||||
|
Talk to your healthcare provider about what to do if you get symptoms of a yeast infection of the vagina or penis. Your healthcare provider may suggest you use an over-the-counter antifungal medicine. Talk to your healthcare provider right away if you use an over-the-counter antifungal medication and your symptoms do not go away. |
|||||||
|
What is INVOKANA?
|
|||||||
|
Do not take INVOKANA if you:
|
|||||||
|
Before taking INVOKANA, tell your healthcare provider about all of your medical conditions, including if you:
Tell your healthcare provider about all the medicines you take,including prescription and over-the-counter medicines, vitamins, and herbal supplements. INVOKANA may affect the way other medicines work, and other medicines may affect how INVOKANA works. Know the medicines you take. Keep a list of them and show it to your healthcare provider and pharmacist when you get a new medicine. |
|||||||
|
How should I take INVOKANA?
|
|||||||
|
What are the possible side effects of INVOKANA? INVOKANA may cause serious side effects including: See " What is the most important information I should know about INVOKANA? "
|
|||||||
|
|
|
|||||
|
|||||||
|
|
|
|||||
The most common side effects of INVOKANA include:
These are not all the possible side effects of INVOKANA. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. You may also report side effects to Janssen Pharmaceuticals, Inc. at 1-800-526-7736. |
|||||||
|
How should I store INVOKANA?
|
|||||||
|
General information about the safe and effective use of INVOKANA. Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use INVOKANA for a condition for which it was not prescribed. Do not give INVOKANA to other people, even if they have the same symptoms that you have. It may harm them. You can ask your pharmacist or healthcare provider for information about INVOKANA that is written for health professionals. |
|||||||
|
What are the ingredients in INVOKANA? Active ingredient: canagliflozin Inactive ingredients: croscarmellose sodium (E468), hydroxypropyl cellulose (E463), lactose anhydrous, magnesium stearate (E572), and microcrystalline cellulose (E460[i]). In addition, the tablet coating contains iron oxide yellow (E172) (100 mg tablet only), macrogol/PEG3350 (E1521), polyvinyl alcohol (E1203) (partially hydrolyzed), talc (E553b), and titanium dioxide (E171). |
|||||||
|
Active ingredient made in Belgium. Manufactured for: Janssen Pharmaceuticals, Inc., Titusville, NJ 08560, USA.
|
|||||||
Section 43683-2 (43683-2)
Section 44425-7 (44425-7)
Storage and Handling
Keep out of reach of children.
Store at 20 °C to 25 °C (68 °F to 77 °F); excursions permitted between 15 °C to 30 °C (59 °F to 86 °F) [see USP Controlled Room Temperature].
10 Overdosage (10 OVERDOSAGE)
In the event of an overdose, contact the Poison Help line (1-800-222-1222) or a medical toxicologist for additional overdosage management recommendations. It is also reasonable to employ the usual supportive measures, e.g., remove unabsorbed material from the gastrointestinal tract, employ clinical monitoring, and institute supportive treatment as dictated by the patient's clinical status. Canagliflozin was negligibly removed during a 4-hour hemodialysis session. Canagliflozin is not expected to be dialyzable by peritoneal dialysis.
11 Description (11 DESCRIPTION)
INVOKANA ®(canagliflozin) contains canagliflozin, an inhibitor of SGLT2, the transporter responsible for reabsorbing the majority of glucose filtered by the kidney. Canagliflozin, the active ingredient of INVOKANA, is chemically known as (1 S)-1,5-anhydro-1-[3-[[5-(4-fluorophenyl)-2-thienyl]methyl]-4-methylphenyl]-D-glucitol hemihydrate and its molecular formula and weight are C 24H 25FO 5S∙1/2 H 2O and 453.53, respectively. The structural formula for canagliflozin is:
Canagliflozin is practically insoluble in aqueous media from pH 1.1 to 12.9.
INVOKANA is supplied as film-coated tablets for oral administration, containing 102 and 306 mg of canagliflozin in each tablet strength, corresponding to 100 mg and 300 mg of canagliflozin (anhydrous), respectively.
Inactive ingredients of the core tablet are croscarmellose sodium (E468), hydroxypropyl cellulose (E463), lactose anhydrous, magnesium stearate (E572), and microcrystalline cellulose (E460[i]). The magnesium stearate is vegetable-sourced. The tablets are finished with a commercially available film-coating consisting of the following excipients: iron oxide yellow (E172) (100 mg tablet only), macrogol/PEG3350 (E1521), polyvinyl alcohol (E1203) (partially hydrolyzed), talc (E553b), and titanium dioxide (E171).
5.9 Bone Fracture
An increased risk of bone fracture, occurring as early as 12 weeks after treatment initiation, was observed in adult patients using INVOKANA in the CANVAS trial [see Clinical Studies (14.3)] . Consider factors that contribute to fracture risk prior to initiating INVOKANA [see Adverse Reactions (6.1)] .
8.4 Pediatric Use
The safety and effectiveness of INVOKANA as an adjunct to diet and exercise to improve glycemic control in type 2 diabetes mellitus have been established in pediatric patients aged 10 years and older.
Use of INVOKANA for this indication is supported by evidence from a 52-week double-blind, placebo-controlled trial in 171 pediatric patients aged 10 to 17 years with type 2 diabetes mellitus and in a pediatric pharmacokinetic study [see Clinical Pharmacology (12.3)and Clinical Studies (14.2)] . The safety profile of pediatric patients treated with INVOKANA was similar to that observed in adults with type 2 diabetes mellitus.
The safety and effectiveness of INVOKANA for glycemic control in patients with type 2 diabetes have not been established in pediatric patients under 10 years of age.
The safety and effectiveness of INVOKANA have not been established in pediatric patients to reduce the risk of:
- major adverse cardiovascular events (cardiovascular death, nonfatal myocardial infarction and nonfatal stroke) in patients with type 2 diabetes mellitus and established cardiovascular disease (CVD).
- end-stage kidney disease (ESKD), doubling of serum creatinine, cardiovascular (CV) death, and hospitalization for heart failure in patients with type 2 diabetes mellitus and diabetic nephropathy with albuminuria greater than 300 mg/day.
8.5 Geriatric Use
In 13 clinical trials of INVOKANA, 2,294 patients 65 years and older, and 351 patients 75 years and older were exposed to INVOKANA [see Clinical Studies (14.1)] .
Patients 65 years and older had a higher incidence of adverse reactions related to reduced intravascular volume with INVOKANA (such as hypotension, postural dizziness, orthostatic hypotension, syncope, and dehydration), particularly with the 300 mg daily dose, compared to younger patients; a more prominent increase in the incidence was seen in patients who were 75 years and older [see Dosage and Administration (2.1)and Adverse Reactions (6.1)]. Smaller reductions in HbA 1Cwith INVOKANA relative to placebo were seen in older (65 years and older; -0.61% with INVOKANA 100 mg and -0.74% with INVOKANA 300 mg relative to placebo) compared to younger patients (-0.72% with INVOKANA 100 mg and -0.87% with INVOKANA 300 mg relative to placebo).
4 Contraindications (4 CONTRAINDICATIONS)
INVOKANA is contraindicated in patients with a serious hypersensitivity reaction to INVOKANA, such as anaphylaxis or angioedema [see Warnings and Precautions (5.8)and Adverse Reactions (6.1, 6.2)] .
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following important adverse reactions are described below and elsewhere in the labeling:
- Diabetic Ketoacidosis in Patients with Type 1 Diabetes and Other Ketoacidosis [see Warnings and Precautions (5.1)]
- Lower Limb Amputation [see Warnings and Precautions (5.2)]
- Volume Depletion [see Warnings and Precautions (5.3)]
- Urosepsis and Pyelonephritis [see Warnings and Precautions (5.4)]
- Hypoglycemia with Concomitant Use with Insulin or Insulin Secretagogues [see Warnings and Precautions (5.5)]
- Necrotizing Fasciitis of the Perineum (Fournier's gangrene) [see Warnings and Precautions (5.6)]
- Genital Mycotic Infections [see Warnings and Precautions (5.7)]
- Hypersensitivity Reactions [see Warnings and Precautions (5.8)]
- Bone Fracture [see Warnings and Precautions (5.9)]
7 Drug Interactions (7 DRUG INTERACTIONS)
| UGT Enzyme Inducers | |
| Clinical Impact: | UGT enzyme inducers decrease canagliflozin exposure which may reduce the effectiveness of INVOKANA. |
| Intervention: | For patients with eGFR 60 mL/min/1.73 m
2or greater, if an inducer of UGTs is administered with INVOKANA, increase the dosage to 200 mg daily in patients currently tolerating INVOKANA 100 mg daily. The total daily dosage may be increased to 300 mg daily in patients currently tolerating INVOKANA 200 mg daily who require additional glycemic control.
For patients with eGFR less than 60 mL/min/1.73 m 2, if an inducer of UGTs is administered with INVOKANA, increase the dosage to 200 mg daily in patients currently tolerating INVOKANA 100 mg daily. Consider adding another antihyperglycemic agent in patients who require additional glycemic control [see Dosage and Administration (2.3)and Clinical Pharmacology (12.3)] . |
| Examples: | Rifampin, phenytoin, phenobarbital, ritonavir |
| Insulin or Insulin Secretagogues | |
| Clinical Impact: | The risk of hypoglycemia is increased when INVOKANA is used concomitantly with insulin secretagogues (e.g., sulfonylurea) or insulin. |
| Intervention: | Concomitant use may require a lower dosage of the insulin secretagogue or insulin to reduce the risk of hypoglycemia. |
| Digoxin | |
| Clinical Impact: | Canagliflozin increases digoxin exposure [see Clinical Pharmacology (12.3)] . |
| Intervention: | Monitor patients taking INVOKANA with concomitant digoxin for a need to adjust the dosage of digoxin. |
| Lithium | |
| Clinical Impact: | Concomitant use of an SGLT2 inhibitor with lithium may decrease serum lithium concentrations. |
| Intervention: | Monitor serum lithium concentration more frequently during INVOKANA initiation and dosage changes. |
| Drug/Laboratory Test Interference | |
| Positive Urine Glucose Test | |
| Clinical Impact: | SGLT2 inhibitors increase urinary glucose excretion which will lead to positive urine glucose tests. |
| Intervention: | Monitoring glycemic control with urine glucose tests is not recommended in patients taking SGLT2 inhibitors. Use alternative methods to monitor glycemic control. |
| Interference with 1,5-anhydroglucitol (1,5-AG) Assay | |
| Clinical Impact: | Measurements of 1,5-AG are unreliable in assessing glycemic control in patients taking SGLT2 inhibitors. |
| Intervention: | Monitoring glycemic control with 1,5-AG assay is not recommended in patients taking SGLT2 inhibitors. Use alternative methods to monitor glycemic control. |
5.3 Volume Depletion
INVOKANA can cause intravascular volume contraction which may sometimes manifest as symptomatic hypotension or acute transient changes in creatinine [see Adverse Reactions (6.1)] . There have been post-marketing reports of acute kidney injury which are likely related to volume depletion, some requiring hospitalizations and dialysis, in patients with type 2 diabetes mellitus receiving SGLT2 inhibitors, including INVOKANA. Patients with impaired renal function (eGFR less than 60 mL/min/1.73 m 2), elderly patients, or patients on loop diuretics may be at increased risk for volume depletion or hypotension. Before initiating INVOKANA in patients with one or more of these characteristics, assess and correct volume status. Monitor for signs and symptoms of volume depletion after initiating therapy.
8.6 Renal Impairment
The efficacy and safety of INVOKANA for glycemic control were evaluated in a trial that included adult patients with moderate renal impairment (eGFR 30 to less than 50 mL/min/1.73 m 2) [see Clinical Studies (14.1)] . These patients had less overall glycemic efficacy, and patients treated with 300 mg per day had increases in serum potassium, which were transient and similar by the end of the trial. Patients with renal impairment using INVOKANA for glycemic control may also be more likely to experience hypotension and may be at higher risk for acute kidney injury [see Warnings and Precautions (5.3)] .
Efficacy and safety trials with INVOKANA did not enroll adult patients with ESKD on dialysis or patients with an eGFR less than 30 mL/min/1.73 m 2 [see Clinical Pharmacology (12.3)] .
12.2 Pharmacodynamics
Following single and multiple oral doses of canagliflozin in adult patients with type 2 diabetes, dose-dependent decreases in RT Gand increases in urinary glucose excretion were observed. From a starting RT Gvalue of approximately 240 mg/dL, canagliflozin at 100 mg and 300 mg once daily suppressed RT Gthroughout the 24-hour period. Data from single oral doses of canagliflozin in healthy volunteers indicate that, on average, the elevation in urinary glucose excretion approaches baseline by about 3 days for doses up to 300 mg once daily. Maximal suppression of mean RT Gover the 24-hour period was seen with the 300 mg daily dose to approximately 70 to 90 mg/dL in patients with type 2 diabetes in Phase 1 trials. The reductions in RT Gled to increases in mean UGE of approximately 100 g/day in patients with type 2 diabetes treated with either 100 mg or 300 mg of canagliflozin. In patients with type 2 diabetes given 100 to 300 mg once daily over a 16-day dosing period, reductions in RT Gand increases in urinary glucose excretion were observed over the dosing period. In this trial, plasma glucose declined in a dose-dependent fashion within the first day of dosing. In single-dose trials in healthy and type 2 diabetic patients, treatment with canagliflozin 300 mg before a mixed-meal delayed intestinal glucose absorption and reduced postprandial glucose.
12.3 Pharmacokinetics
The pharmacokinetics of canagliflozin is similar in healthy subjects and patients with type 2 diabetes. Following single-dose oral administration of 100 mg and 300 mg of INVOKANA, peak plasma concentrations (median T max) of canagliflozin occurs within 1 to 2 hours post-dose. Plasma C maxand AUC of canagliflozin increased in a dose-proportional manner from 50 mg to 300 mg. The apparent terminal half-life (t 1/2) was 10.6 hours and 13.1 hours for the 100 mg and 300 mg doses, respectively. Steady-state was reached after 4 to 5 days of once-daily dosing with canagliflozin 100 mg to 300 mg. Canagliflozin does not exhibit time-dependent pharmacokinetics and accumulated in plasma up to 36% following multiple doses of 100 mg and 300 mg.
8.7 Hepatic Impairment
No dosage adjustment is necessary in patients with mild or moderate hepatic impairment. The use of INVOKANA has not been studied in patients with severe hepatic impairment and is therefore not recommended [see Clinical Pharmacology (12.3)] .
1 Indications and Usage (1 INDICATIONS AND USAGE)
INVOKANA (canagliflozin) is indicated:
- as an adjunct to diet and exercise to improve glycemic control in adults and pediatric patients aged 10 years and older with type 2 diabetes mellitus.
- to reduce the risk of major adverse cardiovascular events (cardiovascular death, nonfatal myocardial infarction and nonfatal stroke) in adults with type 2 diabetes mellitus and established cardiovascular disease (CVD).
- to reduce the risk of end-stage kidney disease (ESKD), doubling of serum creatinine, cardiovascular (CV) death, and hospitalization for heart failure in adults with type 2 diabetes mellitus and diabetic nephropathy with albuminuria greater than 300 mg/day.
12.1 Mechanism of Action
SGLT2, expressed in the proximal renal tubules, is responsible for the majority of the reabsorption of filtered glucose from the tubular lumen. Canagliflozin is an inhibitor of SGLT2. By inhibiting SGLT2, canagliflozin reduces reabsorption of filtered glucose and lowers the renal threshold for glucose (RT G), and thereby increases urinary glucose excretion (UGE).
Canagliflozin increases the delivery of sodium to the distal tubule by blocking SGLT2-dependent glucose and sodium reabsorption. This is believed to increase tubuloglomerular feedback and reduce intraglomerular pressure.
5.2 Lower Limb Amputation
An increased risk of lower limb amputations associated with INVOKANA use versus placebo was observed in CANVAS (5.9 vs 2.8 events per 1,000 patient-years) and CANVAS-R (7.5 vs 4.2 events per 1,000 patient-years), two randomized, placebo-controlled trials evaluating adult patients with type 2 diabetes mellitus who had either established cardiovascular disease or were at risk for cardiovascular disease. The risk of lower limb amputations was observed at both the 100 mg and 300 mg once daily dosage regimens. The amputation data for CANVAS and CANVAS-R are shown in Tables 3 and 4, respectively [see Adverse Reactions (6.1)].
Amputations of the toe and midfoot (99 out of 140 patients with amputations receiving INVOKANA in the two trials) were the most frequent; however, amputations involving the leg, below and above the knee, were also observed (41 out of 140 patients with amputations receiving INVOKANA in the two trials). Some patients had multiple amputations, some involving both lower limbs.
Lower limb infections, gangrene, and diabetic foot ulcers were the most common precipitating medical events leading to the need for an amputation. The risk of amputation was highest in patients with a baseline history of prior amputation, peripheral vascular disease, and neuropathy.
Counsel patients about the importance of routine preventative foot care. Monitor patients receiving INVOKANA for signs and symptoms of infection (including osteomyelitis), new pain or tenderness, sores or ulcers involving the lower limbs, and discontinue INVOKANA if these complications occur.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Diabetic Ketoacidosis in Patients with Type 1 Diabetes Mellitus and Other Ketoacidosis: Consider ketone monitoring in patients at risk for ketoacidosis, as indicated. Assess for ketoacidosis regardless of presenting blood glucose levels and discontinue INVOKANA if ketoacidosis is suspected. Monitor patients for resolution of ketoacidosis before restarting ( 5.1).
- Lower Limb Amputation: Monitor patients for infection or ulcers of lower limb and discontinue if these occur ( 5.2).
- Volume Depletion: May result in acute kidney injury. Before initiating INVOKANA, assess and correct volume status in patients with renal impairment, elderly patients, or patients on loop diuretics. Monitor for signs and symptoms during therapy ( 5.3).
- Urosepsis and Pyelonephritis: Evaluate patients for signs and symptoms of urinary tract infections and treat promptly, if indicated ( 5.4).
- Hypoglycemia with Concomitant Use with Insulin or Insulin Secretagogues: Consider a lower dose of insulin or the insulin secretagogue to reduce the risk of hypoglycemia when used in combination with INVOKANA ( 5.5).
- Necrotizing Fasciitis of the Perineum (Fournier's Gangrene): Serious, life-threatening cases have occurred in both females and males. Assess patients presenting with pain or tenderness, erythema, or swelling in the genital or perineal area, along with fever or malaise. If suspected, institute prompt treatment ( 5.6).
- Genital Mycotic Infections: Monitor and treat if indicated ( 5.7).
- Hypersensitivity Reactions: Discontinue INVOKANA and monitor until signs and symptoms resolve ( 5.8).
- Bone Fracture: Consider factors that contribute to fracture risk before initiating INVOKANA ( 5.9).
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
- Assess renal function before initiating and as clinically indicated. Assess volume status and correct volume depletion before initiating ( 2.1).
- The recommended starting dosage in adults and pediatric patients aged 10 years and older with type 2 diabetes mellitus is 100 mg orally once daily, taken before the first meal of the day to improve glycemic control. The dosage can be increased to 300 mg once daily in patients tolerating 100 mg once daily who have an eGFR of 60 mL/min/1.73 m 2or greater and require additional glycemic control ( 2.2).
- For all other indications in adults, the recommended dosage of INVOKANA is 100 mg orally once daily ( 2.2).
- Dosage adjustments for patients with renal impairment may be required ( 2.3).
- See full prescribing information for INVOKANA dosage modifications due to drug interactions ( 2.4).
- Withhold INVOKANA at least 3 days, if possible, prior to surgery or procedures associated with prolonged fasting ( 2.5).
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
- INVOKANA 100 mg tablets are yellow, capsule-shaped, tablets with "CFZ" on one side and "100" on the other side.
- INVOKANA 300 mg tablets are white, capsule-shaped, tablets with "CFZ" on one side and "300" on the other side.
6.2 Postmarketing Experience
Additional adverse reactions have been identified during post-approval use of INVOKANA. Because these reactions are reported voluntarily from a population of uncertain size, it is generally not possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Metabolism and Nutrition
Ketoacidosis
Renal and Urinary
Acute Kidney Injury
Immune System
Anaphylaxis
Skin and Subcutaneous Tissue
Angioedema
Infections
Urosepsis and Pyelonephritis, Necrotizing Fasciitis of the Perineum (Fournier's gangrene)
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
- Pregnancy: Advise females of the potential risk to a fetus especially during the second and third trimesters ( 8.1).
- Lactation: Not recommended when breastfeeding ( 8.2).
- Geriatrics: Higher incidence of adverse reactions related to reduced intravascular volume ( 8.5).
- Renal Impairment: Higher incidence of adverse reactions related to hypotension and renal function ( 8.6).
- Hepatic Impairment: Not recommended in patients with severe hepatic impairment ( 8.7).
5.7 Genital Mycotic Infections
INVOKANA increases the risk of genital mycotic infections. Patients with a history of genital mycotic infections and uncircumcised males were more likely to develop genital mycotic infections [see Adverse Reactions (6.1)] . Monitor and treat appropriately.
5.8 Hypersensitivity Reactions
Hypersensitivity reactions, including angioedema and anaphylaxis, have been reported with INVOKANA. These reactions generally occurred within hours to days after initiating INVOKANA. If hypersensitivity reactions occur, discontinue use of INVOKANA; treat and monitor until signs and symptoms resolve [see Contraindications (4)and Adverse Reactions (6.1, 6.2)] .
6.1 Clinical Studies Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to the rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
INVOKANA has been evaluated in clinical trials in adults and pediatric patients aged 10 years and older with type 2 diabetes mellitus. Additionally, INVOKANA has been studied in clinical trials in adult patients with type 2 diabetes mellitus who also have heart failure or chronic kidney disease. The overall safety profile of INVOKANA was consistent across the studied indications.
5.4 Urosepsis and Pyelonephritis
There have been postmarketing reports of serious urinary tract infections including urosepsis and pyelonephritis requiring hospitalization in patients receiving INVOKANA. Treatment with INVOKANA increases the risk for urinary tract infections. Evaluate patients for signs and symptoms of urinary tract infections and treat promptly, if indicated [see Adverse Reactions (6)] .
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient to read the FDA-approved patient labeling (Medication Guide).
2.1 Prior to Initiation of Invokana (2.1 Prior to Initiation of INVOKANA)
Assess renal function before initiating INVOKANA and as clinically indicated [see Dosage and Administration (2.3)and Warnings and Precautions (5.3)] .
In patients with volume depletion, correct this condition before initiating INVOKANA [see Warnings and Precautions (5.3)and Use in Specific Populations (8.5, 8.6)] .
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
INVOKANA ®(canagliflozin) tablets are available in the strengths and packages listed below:
100 mg tablets are yellow, capsule-shaped, film-coated tablets with "CFZ" on one side and "100" on the other side.
| NDC 50458-140-30 | Bottle of 30 |
| NDC 50458-140-90 | Bottle of 90 |
| NDC 50458-140-50 | Bottle of 500 |
300 mg tablets are white, capsule-shaped, film-coated tablets with "CFZ" on one side and "300" on the other side.
| NDC 50458-141-30 | Bottle of 30 |
| NDC 50458-141-90 | Bottle of 90 |
| NDC 50458-141-50 | Bottle of 500 |
2.5 Temporary Interruption for Surgery
Withhold INVOKANA at least 3 days, if possible, prior to surgery or procedures associated with prolonged fasting. Resume INVOKANA when the patient is clinically stable and has resumed oral intake [see Warnings and Precautions (5.1)and Clinical Pharmacology (12.2)].
Principal Display Panel 100 Mg Tablet Bottle Label (PRINCIPAL DISPLAY PANEL - 100 mg Tablet Bottle Label)
NDC 50458-140-90
Invokana
®
(canagliflozin) tablets
100 mg
Attention: Dispense the enclosed
Medication Guide to each patient.
Rx only
90 tablets
Principal Display Panel 300 Mg Tablet Bottle Label (PRINCIPAL DISPLAY PANEL - 300 mg Tablet Bottle Label)
NDC 50458-141-90
Invokana
®
(canagliflozin) tablets
300 mg
Attention: Dispense the enclosed
Medication Guide to each patient.
Rx only
90 tablets
5.6 Necrotizing Fasciitis of the Perineum (fournier's Gangrene) (5.6 Necrotizing Fasciitis of the Perineum (Fournier's Gangrene))
Reports of necrotizing fasciitis of the perineum (Fournier's gangrene), a rare but serious and life-threatening necrotizing infection requiring urgent surgical intervention, have been identified in postmarketing surveillance in patients with diabetes mellitus receiving SGLT2 inhibitors, including INVOKANA. Cases have been reported in both females and males. Serious outcomes have included hospitalization, multiple surgeries, and death.
Patients treated with INVOKANA presenting with pain or tenderness, erythema, or swelling in the genital or perineal area, along with fever or malaise, should be assessed for necrotizing fasciitis. If suspected, start treatment immediately with broad-spectrum antibiotics and, if necessary, surgical debridement. Discontinue INVOKANA, closely monitor blood glucose levels, and provide appropriate alternative therapy for glycemic control.
14.1 Glycemic Control Trials in Adults With Type 2 Diabetes Mellitus (14.1 Glycemic Control Trials in Adults with Type 2 Diabetes Mellitus)
INVOKANA (canagliflozin) has been studied as monotherapy, in combination with metformin HCl, sulfonylurea, metformin HCl and sulfonylurea, metformin HCl and sitagliptin, metformin HCl and a thiazolidinedione (i.e., pioglitazone), and in combination with insulin (with or without other anti-hyperglycemic agents). The efficacy of INVOKANA was compared to a dipeptidyl peptidase-4 (DPP-4) inhibitor (sitagliptin), both as add-on combination therapy with metformin HCl and sulfonylurea, and a sulfonylurea (glimepiride), both as add-on combination therapy with metformin HCl. INVOKANA was also evaluated in adults 55 to 80 years of age and patients with moderate renal impairment.
2.4 Concomitant Use With Udp Glucuronosyl Transferase (ugt) Enzyme Inducers (2.4 Concomitant Use with UDP-Glucuronosyl transferase (UGT) Enzyme Inducers)
When co-administering INVOKANA with an inducer of UGT (e.g., rifampin, phenytoin, phenobarbital, ritonavir), increase the dosage of INVOKANA based on renal function [see Drug Interactions (7)]:
- In patients with eGFR 60 mL/min/1.73 m 2or greater, increase the dosage to 200 mg orally once daily in patients currently tolerating INVOKANA 100 mg once daily. The maximum recommended dosage of INVOKANA is 300 mg once daily.
- In patients with eGFR less than 60 mL/min/1.73 m 2, increase to a maximum recommended dosage of 200 mg orally once daily in patients currently tolerating INVOKANA 100 mg once daily.
5.5 Hypoglycemia With Concomitant Use With Insulin Or Insulin Secretagogues (5.5 Hypoglycemia with Concomitant Use with Insulin or Insulin Secretagogues)
Insulin and insulin secretagogues are known to cause hypoglycemia. INVOKANA may increase the risk of hypoglycemia when combined with insulin or an insulin secretagogue [see Adverse Reactions (6.1)] . The risk of hypoglycemia may be lowered by a reduction in the dose of sulfonylurea (or other concomitantly administered insulin secretagogues) or insulin. Inform patients using these concomitant medications of the risk of hypoglycemia and educate them on the signs and symptoms of hypoglycemia.
14.4 Renal and Cardiovascular Outcomes in Adults With Diabetic Nephropathy and Albuminuria (14.4 Renal and Cardiovascular Outcomes in Adults with Diabetic Nephropathy and Albuminuria)
The Canagliflozin and Renal Events in Diabetes with Established Nephropathy Clinical Evaluation Trial (CREDENCE) was a multinational, randomized, double-blind, placebo-controlled trial comparing canagliflozin with placebo in adult patients with type 2 diabetes mellitus, an eGFR ≥ 30 to < 90 mL/min/1.73 m 2and albuminuria (urine albumin/creatinine > 300 to ≤ 5,000 mg/g) who were receiving standard of care including a maximum-tolerated, labeled daily dose of an angiotensin-converting enzyme inhibitor (ACEi) or angiotensin receptor blocker (ARB).
The primary objective of CREDENCE was to assess the efficacy of canagliflozin relative to placebo in reducing the composite endpoint of end stage kidney disease (ESKD), doubling of serum creatinine, and renal or CV death.
Patients were randomized to receive canagliflozin 100 mg (N=2,202) or placebo (N=2,199) and treatment was continued until the initiation of dialysis or renal transplantation.
The median follow-up duration for the 4,401 randomized subjects was 137 weeks. Vital status was obtained for 99.9% of subjects.
The population was 67% White, 20% Asian, and 5% Black or African American; 32% were of Hispanic or Latino ethnicity. The mean age was 63 years and 66% were male.
At randomization, the mean HbA 1Cwas 8.3%, the median urine albumin/creatinine was 927 mg/g, the mean eGFR was 56.2 mL/min/1.73 m 2, 50% had prior CV disease, and 15% reported a history of heart failure. The most frequent antihyperglycemic agents (AHA) medications used at baseline were insulin (66%), biguanides (58%), and sulfonylureas (29%). Nearly all subjects (99.9%) were on ACEi or ARB at randomization, approximately 60% were taking an anti-thrombotic agent (including aspirin), and 69% were on a statin.
The primary composite endpoint in the CREDENCE trial was the time to first occurrence of ESKD (defined as an eGFR < 15 mL/min/1.73 m 2, initiation of chronic dialysis or renal transplant), doubling of serum creatinine, and renal or CV death. Canagliflozin 100 mg significantly reduced the risk of the primary composite endpoint based on a time-to-event analysis [HR: 0.70; 95% CI: 0.59, 0.82; p<0.0001] (see Figure 4). The treatment effect reflected a reduction in progression to ESKD, doubling of serum creatinine and cardiovascular death as shown in Table 22 and Figure 4. There were few renal deaths during the trial. Canagliflozin 100 mg also significantly reduced the risk of hospitalization for heart failure [HR: 0.61; 95% CI: 0.47 to 0.80; p<0.001].
| Placebo | canagliflozin | ||||
|---|---|---|---|---|---|
| Endpoint | N=2,199 (%) | Event Rate
Event rate per 100 patient-years.
|
N=2,202 (%) | Event Rate | HR
Hazard ratio (canagliflozin compared to placebo), 95% CI and p-value are estimated using a stratified Cox proportional hazards model including treatment as the explanatory variable and stratified by screening eGFR (≥ 30 to < 45, ≥ 45 to < 60, ≥ 60 to < 90 mL/min/1.73 m
2). HR is not presented for renal death due to the small number of events in each group.
(95% CI) |
| Intent-To-Treat Analysis Set (time to first occurrence) | |||||
| The individual components do not represent a breakdown of the composite outcomes, but rather the total number of subjects experiencing an event during the course of the trial. | |||||
| Primary Composite Endpoint (ESKD, doubling of serum creatinine, renal death, or CV death) | 340 (15.5) | 6.1 | 245 (11.1) | 4.3 | 0.70
(0.59, 0.82) P-value <0.0001
|
| ESKD | 165 (7.5) | 2.9 | 116 (5.3) | 2.0 | 0.68
(0.54, 0.86) |
| Doubling of serum creatinine | 188 (8.5) | 3.4 | 118 (5.4) | 2.1 | 0.60
(0.48, 0.76) |
| Renal death | 5 (0.2) | 0.1 | 2 (0.1) | 0.0 | |
| CV death | 140 (6.4) | 2.4 | 110 (5.0) | 1.9 | 0.78
(0.61, 1.00) |
| CV death or hospitalization for heart failure | 253 (11.5) | 4.5 | 179 (8.1) | 3.1 | 0.69
(0.57, 0.83) P-value <0.001
|
| CV death, non-fatal myocardial infarction or non-fatal stroke | 269 (12.2) | 4.9 | 217 (9.9) | 3.9 | 0.80
(0.67, 0.95) P-value <0.02
|
| Non-fatal myocardial infarction | 87 (4.0) | 1.6 | 71 (3.2) | 1.3 | 0.81
(0.59, 1.10) |
| Non-fatal stroke | 66 (3.0) | 1.2 | 53 (2.4) | 0.9 | 0.80
(0.56, 1.15) |
| Hospitalization for heart failure | 141 (6.4) | 2.5 | 89 (4.0) | 1.6 | 0.61
(0.47, 0.80) |
| ESKD, doubling of serum creatinine or renal death | 224 (10.2) | 4.0 | 153 (6.9) | 2.7 | 0.66
(0.53, 0.81) |
The Kaplan-Meier curve (Figure 4) shows time to first occurrence of the primary composite endpoint of ESKD, doubling of serum creatinine, renal death, or CV death. The curves begin to separate by Week 52 and continue to diverge thereafter.
Figure 4: CREDENCE: Time to First Occurrence of the Primary Composite Endpoint
5.1 Diabetic Ketoacidosis in Patients With Type 1 Diabetes Mellitus and Other Ketoacidosis (5.1 Diabetic Ketoacidosis in Patients with Type 1 Diabetes Mellitus and Other Ketoacidosis)
In patients with type 1 diabetes mellitus, INVOKANA significantly increases the risk of diabetic ketoacidosis, a life-threatening event, beyond the background rate. In placebo-controlled trials of patients with type 1 diabetes mellitus, the risk of ketoacidosis was markedly increased in patients who received sodium glucose transporter 2 (SGLT2) inhibitors compared to patients who received placebo; this risk may be greater with higher doses of INVOKANA. INVOKANA is not indicated for glycemic control in patients with type 1 diabetes mellitus.
Type 2 diabetes mellitus and pancreatic disorders (e.g., history of pancreatitis or pancreatic surgery) are also risk factors for ketoacidosis. There have been postmarketing reports of fatal events of ketoacidosis in patients with type 2 diabetes mellitus using SGLT2 inhibitors, including INVOKANA.
Precipitating conditions for diabetic ketoacidosis or other ketoacidosis include under-insulinization due to insulin dose reduction or missed insulin doses, acute febrile illness, reduced caloric intake, ketogenic diet, surgery, volume depletion, and alcohol abuse.
Signs and symptoms are consistent with dehydration and severe metabolic acidosis and include nausea, vomiting, abdominal pain, generalized malaise, and shortness of breath. Blood glucose levels at presentation may be below those typically expected for diabetic ketoacidosis (e.g., less than 250 mg/dL). Ketoacidosis and glucosuria may persist longer than typically expected.
Urinary glucose excretion persists for 3 days after discontinuing INVOKANA [see Clinical Pharmacology (12.2)]; however, there have been postmarketing reports of ketoacidosis and/or glucosuria lasting greater than 6 days and some up to 2 weeks after discontinuation of SGLT2 inhibitors.
Consider ketone monitoring in patients at risk for ketoacidosis if indicated by the clinical situation. Assess for ketoacidosis regardless of presenting blood glucose levels in patients who present with signs and symptoms consistent with severe metabolic acidosis. If ketoacidosis is suspected, discontinue INVOKANA, promptly evaluate, and treat ketoacidosis, if confirmed. Monitor patients for resolution of ketoacidosis before restarting INVOKANA.
Withhold INVOKANA, if possible, in temporary clinical situations that could predispose patients to ketoacidosis. Resume INVOKANA when the patient is clinically stable and has resumed oral intake [see Dosage and Administration (2.5)] .
Educate all patients on the signs and symptoms of ketoacidosis and instruct patients to discontinue INVOKANA and seek medical attention immediately if signs and symptoms occur.
2.3 Recommended Dosage in Adults and Pediatric Patients Aged 10 Years and Older With Renal Impairment (2.3 Recommended Dosage in Adults and Pediatric Patients Aged 10 Years and Older with Renal Impairment)
Table 1 provides dosage recommendations for adults and pediatric patients aged 10 years and older with renal impairment, based on estimated glomerular filtration rate (eGFR).
| Estimated Glomerular Filtration Rate
[eGFR (mL/min/1.73 m 2)] |
Recommended Dosage |
|---|---|
| eGFR 30 to less than 60 | The maximum recommended dosage is 100 mg orally once daily. |
| eGFR less than 30 |
|
14.2 Glycemic Control Trial in Pediatric Patients Aged 10 Years and Older With Type 2 Diabetes Mellitus (14.2 Glycemic Control Trial in Pediatric Patients Aged 10 Years and Older with Type 2 Diabetes Mellitus)
In a double-blind, placebo-controlled, parallel-group pediatric trial (NCT03170518), 171 pediatric patients aged 10 to 17 years with inadequately controlled type 2 diabetes mellitus (HbA 1C≥6.5% and ≤11.0%) were randomized to INVOKANA (84 patients) or placebo (87 patients) as add-on to diet and exercise, metformin HCl (≥1,000 mg per day or maximally tolerated dosage), insulin, or a combination of metformin HCl and insulin, for a total of 52-weeks. At Week 13, patients in the INVOKANA arm whose HbA 1Cwas ≥7.0% and eGFR ≥60 mL/min/1.73 m 2were re-randomized to either continue on INVOKANA 100 mg orally once daily (n=16) or to up-titrate to INVOKANA 300 mg orally once daily (n=17).
At baseline, background therapies included diet and exercise only (14%), insulin monotherapy (11%), metformin HCl and insulin (29%), and metformin HCl monotherapy (46%). The mean HbA 1Cat baseline was 8.0% and the mean duration of type 2 diabetes mellitus was 2 years. The mean eGFR at baseline was 157.3 mL/min/1.73 m 2, and approximately 16% (24/151) of the trial population with measurements had microalbuminuria or macroalbuminuria. Patients with an eGFR less than 60 mL/min/1.73 m 2were not enrolled in the trial; no patients in the trial reached an eGFR < 60 mL/min/1.73 m 2. The mean age was 14.3 years, 47% were under 15 years of age, and 68% were female. Approximately 42% were Asian, 42% were White, 11% were Black or African American, 5% were American Indian/Alaska Native, and 36% were of Hispanic or Latino ethnicity. The mean BMI was 30.8 kg/m 2(range 18–57 kg/m 2) and the mean BMI Z-score was 1.84.
At Week 26, treatment with canagliflozin provided statistically significant improvement in HbA 1Cfrom baseline, compared with placebo (see Table 20).
| Efficacy Parameter | Placebo
(N=87) |
INVOKANA
(N=84) |
|---|---|---|
| HbA 1C(%) | ||
| Baseline (mean) | 8.3 | 7.8 |
| Change from baseline
Multiple imputation using retrieved dropout approach with 1000 iterations for missing data (canagliflozin N=7 (8.3%), placebo N=7 (8.1%) for HbA
1C and canagliflozin N=9 (10.7%), placebo N=7 (8.1%) for FPG).
Least-Square Mean from Analysis of Covariance (ANCOVA) adjusted for baseline value, baseline age stratum (< 15 years vs 15 to < 18 years) and baseline antihyperglycemic agent (AHA) background (i.e, diet and exercise only, metformin monotherapy, insulin monotherapy, or combination of insulin and metformin).
|
0.34 | -0.38 |
| Difference from placebo 95% CI | -0.73 (-1.26, -0.19)
P-value=0.008 (two-sided)
|
|
| FPG (mg/dL) | ||
| Baseline (mean) | 156.5 | 154.8 |
| Change from baseline | 17.29 | -8.22 |
| Difference from placebo 95% CI | -25.51 (-49.55, -1.47)
Not evaluated for statistical significance, not part of sequential testing procedure.
|
14.3 Cardiovascular Outcomes in Adults With Type 2 Diabetes Mellitus and Atherosclerotic Cardiovascular Disease (14.3 Cardiovascular Outcomes in Adults with Type 2 Diabetes Mellitus and Atherosclerotic Cardiovascular Disease)
The CANVAS and CANVAS-R trials were multicenter, multi-national, randomized, double-blind parallel group, with similar inclusion and exclusion criteria. Patients eligible for enrollment in both CANVAS and CANVAS-R trials were: 30 years of age or older and had established, stable, cardiovascular, cerebrovascular, peripheral artery disease (66% of the enrolled population) or were 50 years of age or older and had two or more other specified risk factors for cardiovascular disease (34% of the enrolled population).
The integrated analysis of the CANVAS and CANVAS-R trials compared the risk of Major Adverse Cardiovascular Event (MACE) between canagliflozin and placebo when these were added to and used concomitantly with standard of care treatments for diabetes and atherosclerotic cardiovascular disease. The primary endpoint, MACE, was the time to first occurrence of a three-part composite outcome which included cardiovascular death, non-fatal myocardial infarction and non-fatal stroke.
In CANVAS, patients were randomly assigned 1:1:1 to canagliflozin 100 mg, canagliflozin 300 mg, or matching placebo. In CANVAS-R, patients were randomly assigned 1:1 to canagliflozin 100 mg or matching placebo, and titration to 300 mg was permitted at the investigator's discretion (based on tolerability and glycemic needs) after Week 13. Concomitant antidiabetic and atherosclerotic therapies could be adjusted, at the discretion of investigators, to ensure participants were treated according to the standard care for these diseases.
A total of 10,134 adult patients were treated (4,327 in CANVAS and 5,807 in CANVAS-R; total of 4,344 randomly assigned to placebo and 5,790 to canagliflozin) for a mean exposure duration of 149 weeks (223 weeks [4.3 years] in CANVAS and 94 weeks [1.8 years] in CANVAS-R) .Approximately 78% of the trial population was White, 13% was Asian, and 3% was Black or African American. The mean age was 63 years and approximately 64% were male.
The mean HbA 1Cat baseline was 8.2% and mean duration of diabetes was 13.5 years with 70% of patients having had diabetes for 10 years or more. Approximately 31%, 21% and 17% reported a past history of neuropathy, retinopathy and nephropathy, respectively, and the mean eGFR 76 mL/min/1.73 m 2. At baseline, patients were treated with one (19%) or more (80%) antidiabetic medications including metformin HCl (77%), insulin (50%), and sulfonylurea (43%).
At baseline, the mean systolic blood pressure was 137 mmHg, the mean diastolic blood pressure was 78 mmHg, the mean LDL was 89 mg/dL, the mean HDL was 46 mg/dL, and the mean urinary albumin to creatinine ratio (UACR) was 115 mg/g. At baseline, approximately 80% of patients were treated with renin angiotensin system inhibitors, 53% with beta-blockers, 13% with loop diuretics, 36% with non-loop diuretics, 75% with statins, and 74% with antiplatelet agents (mostly aspirin). During the trial, investigators could modify anti-diabetic and cardiovascular therapies to achieve local standard of care treatment targets with respect to blood glucose, lipid, and blood pressure. More patients receiving canagliflozin compared to placebo initiated anti-thrombotics (5.2% vs 4.2%) and statins (5.8% vs 4.8%) during the trial.
For the primary analysis, a stratified Cox proportional hazards model was used to test for non-inferiority against a pre-specified risk margin of 1.3 for the hazard ratio of MACE.
In the integrated analysis of CANVAS and CANVAS-R trials, canagliflozin reduced the risk of first occurrence of MACE. The estimated hazard ratio (95% CI) for time to first MACE was 0.86 (0.75, 0.97). Refer to Table 21. Vital status was obtained for 99.6% of patients across the trials. The Kaplan-Meier curve depicting time to first occurrence of MACE is shown in Figure 3.
| Placebo
N=4,347(%) |
Canagliflozin
N=5,795 (%) |
Hazard ratio
(95% C.I.) Stratified Cox-proportional hazards model with treatment as a factor and stratified by the trial and by prior CV disease
|
|
|---|---|---|---|
| Composite of cardiovascular death, non-fatal myocardial infarction, non-fatal stroke
(time to first occurrence) P-value for superiority (2-sided) = 0.0158
,
Number and percentage of first events
,
Due to pooling of unequal randomization ratios, Cochran-Mantel-Haenszel weights were applied to calculate percentages
|
426 (10.4) | 585 (9.2) | 0.86 (0.75, 0.97) |
| Non-fatal myocardial infarction , | 159 (3.9) | 215 (3.4) | 0.85 (0.69, 1.05) |
| Non-fatal Stroke , | 116 (2.8) | 158 (2.5) | 0.90 (0.71, 1.15) |
| Cardiovascular Death , | 185 (4.6) | 268 (4.1) | 0.87 (0.72, 1.06) |
Figure 3: Time to First Occurrence of MACE in Adults with Type 2 Diabetes Mellitus
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:46:14.520620 · Updated: 2026-03-14T22:24:19.550175